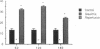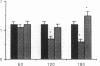Abstract
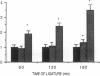
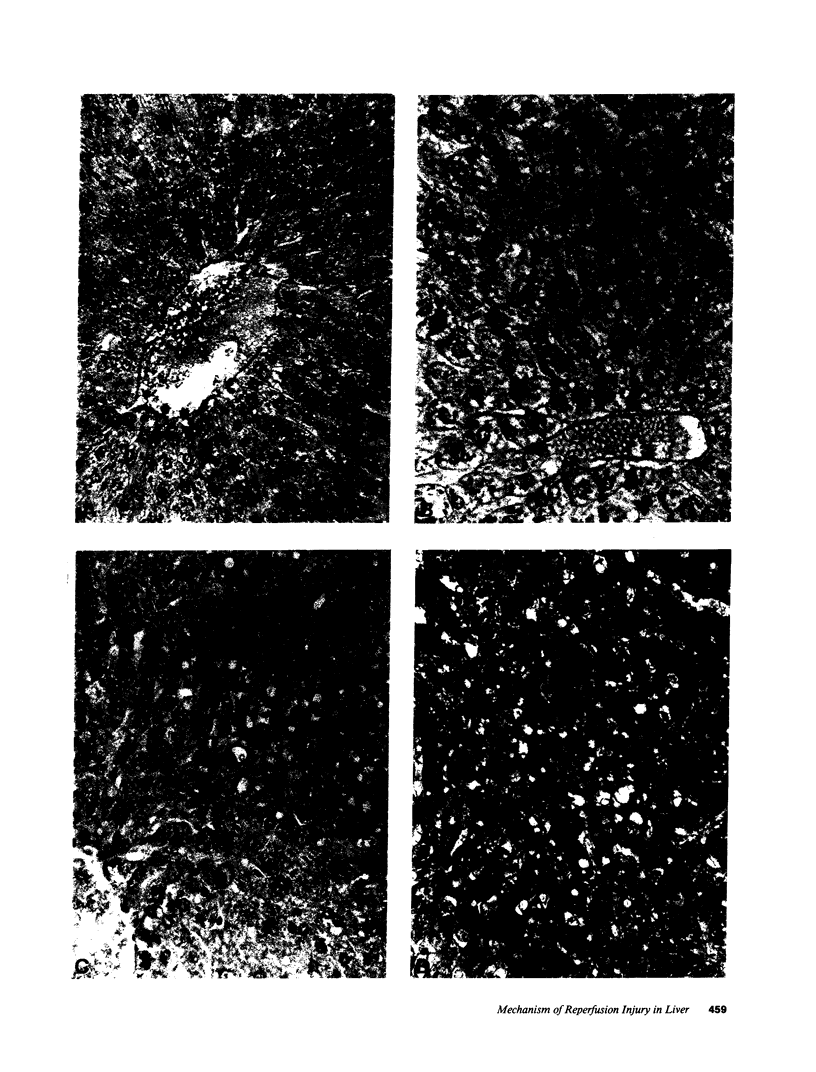
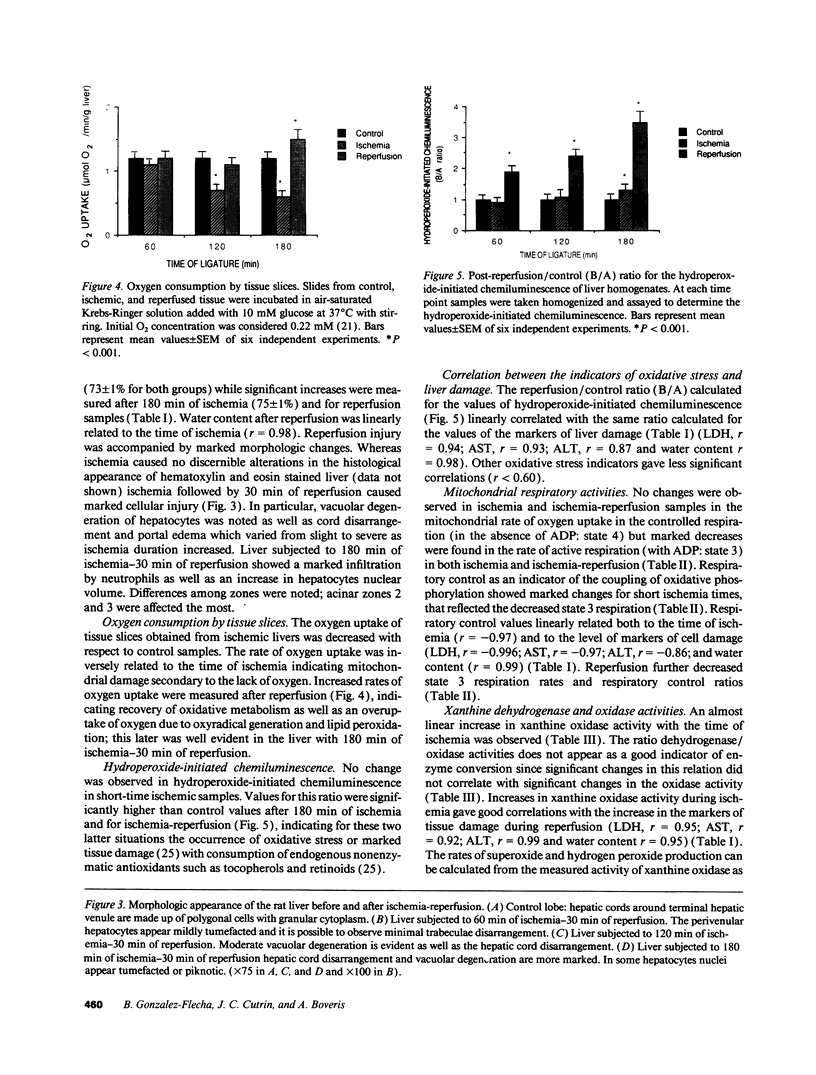
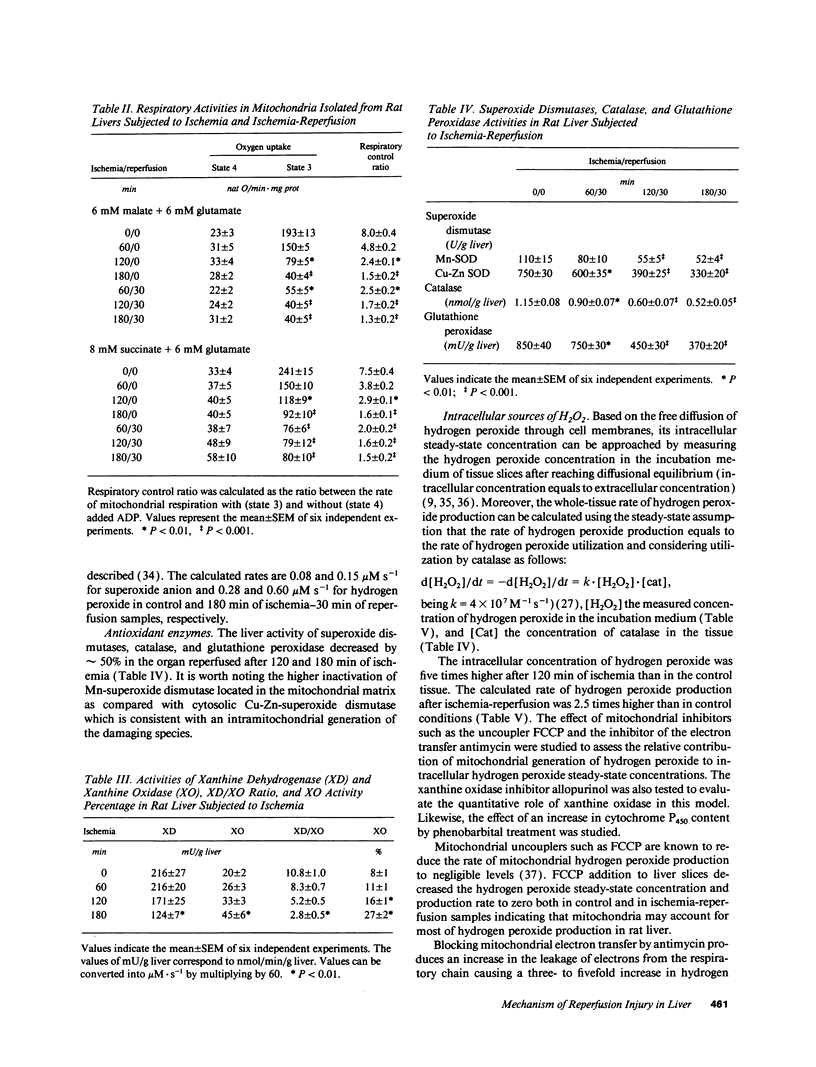
The time course of oxidative stress and tissue damage in zonal liver ischemia-reperfusion in rat liver in vivo was evaluated. After 180 min of ischemia, surface chemiluminescence decreased to zero, state 3 mitochondrial respiration decreased by 70-80%, and xanthine oxidase activity increased by 26% without change in the water content and in the activities of superoxide dismutase, catalase, and glutathione peroxidase. After reperfusion, marked increases in oxyradical production and tissue damage were detected. Mitochondrial oxygen uptake in state 3 and respiratory control as well as the activities of superoxide dismutase, catalase, and glutathione peroxidase and the level of nonenzymatic antioxidants (evaluated by the hydroperoxide-initiated chemiluminescence) were decreased. The severity of the post-reperfusion changes correlated with the time of ischemia. Morphologically, hepatocytes appeared swollen with zonal cord disarrangement which ranged from mild to severe for the tissue reperfused after 60-180 min of ischemia. Neutrophil infiltration was observed after 180 min of ischemia and 30 min of reperfusion. Mitochondria appear as the major source of hydrogen peroxide in control and in reperfused liver, as indicated by the almost complete inhibition of hydrogen peroxide production exerted by the uncoupler carbonylcyanide p-(trifluoromethoxy) phenylhydrazone. Additionally, inhibition of mitochondrial electron transfer by antimycin in liver slices reproduced the inhibition of state 3 mitochondrial respiration and the increase in hydrogen peroxide steady-state concentration found in reperfused liver. Increased rates of oxyradical production by inhibited mitochondria appear as the initial cause of oxidative stress and liver damage during early reperfusion in rat liver.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beauchamp C. O., Fridovich I. Isozymes of superoxide dismutase from wheat germ. Biochim Biophys Acta. 1973 Jul 12;317(1):50–64. doi: 10.1016/0005-2795(73)90198-0. [DOI] [PubMed] [Google Scholar]
- Boveris A., Cadenas E. Mitochondrial production of superoxide anions and its relationship to the antimycin insensitive respiration. FEBS Lett. 1975 Jul 1;54(3):311–314. doi: 10.1016/0014-5793(75)80928-8. [DOI] [PubMed] [Google Scholar]
- Boveris A., Cadenas E., Reiter R., Filipkowski M., Nakase Y., Chance B. Organ chemiluminescence: noninvasive assay for oxidative radical reactions. Proc Natl Acad Sci U S A. 1980 Jan;77(1):347–351. doi: 10.1073/pnas.77.1.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boveris A., Chance B. The mitochondrial generation of hydrogen peroxide. General properties and effect of hyperbaric oxygen. Biochem J. 1973 Jul;134(3):707–716. doi: 10.1042/bj1340707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boveris A., Oshino N., Chance B. The cellular production of hydrogen peroxide. Biochem J. 1972 Jul;128(3):617–630. doi: 10.1042/bj1280617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brass C. A., Narciso J., Gollan J. L. Enhanced activity of the free radical producing enzyme xanthine oxidase in hypoxic rat liver. Regulation and pathophysiologic significance. J Clin Invest. 1991 Feb;87(2):424–431. doi: 10.1172/JCI115013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bustamante J., Guerra L., Bredeston L., Mordoh J., Boveris A. Melanin content and hydroperoxide metabolism in human melanoma cells. Exp Cell Res. 1991 Oct;196(2):172–176. doi: 10.1016/0014-4827(91)90247-r. [DOI] [PubMed] [Google Scholar]
- Cadenas E. Biochemistry of oxygen toxicity. Annu Rev Biochem. 1989;58:79–110. doi: 10.1146/annurev.bi.58.070189.000455. [DOI] [PubMed] [Google Scholar]
- Castillo-Olivares J. L., Gosalvez M., Azpeitia D., Romero E. G., Blanco M., Figuera D. Mitochondrial respiration and oxidative phosphorylation during hepatic preservation. J Surg Res. 1972 Aug;13(2):85–89. doi: 10.1016/0022-4804(72)90048-0. [DOI] [PubMed] [Google Scholar]
- Chance B., Sies H., Boveris A. Hydroperoxide metabolism in mammalian organs. Physiol Rev. 1979 Jul;59(3):527–605. doi: 10.1152/physrev.1979.59.3.527. [DOI] [PubMed] [Google Scholar]
- Chien K. R., Abrams J., Pfau R. G., Farber J. L. Prevention by chlorpromazine of ischemic liver cell death. Am J Pathol. 1977 Sep;88(3):539–557. [PMC free article] [PubMed] [Google Scholar]
- Engerson T. D., McKelvey T. G., Rhyne D. B., Boggio E. B., Snyder S. J., Jones H. P. Conversion of xanthine dehydrogenase to oxidase in ischemic rat tissues. J Clin Invest. 1987 Jun;79(6):1564–1570. doi: 10.1172/JCI112990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferreira R., Burgos M., Llesuy S., Molteni L., Milei J., Flecha B. G., Boveris A. Reduction of reperfusion injury with mannitol cardioplegia. Ann Thorac Surg. 1989 Jul;48(1):77–84. doi: 10.1016/0003-4975(89)90182-3. [DOI] [PubMed] [Google Scholar]
- Fisher A. B., Dodia C., Tan Z. T., Ayene I., Eckenhoff R. G. Oxygen-dependent lipid peroxidation during lung ischemia. J Clin Invest. 1991 Aug;88(2):674–679. doi: 10.1172/JCI115352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flohé L., Günzler W. A. Assays of glutathione peroxidase. Methods Enzymol. 1984;105:114–121. doi: 10.1016/s0076-6879(84)05015-1. [DOI] [PubMed] [Google Scholar]
- Fraga C. G., Arias R. F., Llesuy S. F., Koch O. R., Boveris A. Effect of vitamin E- and selenium-deficiency on rat liver chemiluminescence. Biochem J. 1987 Mar 1;242(2):383–386. doi: 10.1042/bj2420383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fridovich I. Quantitative aspects of the production of superoxide anion radical by milk xanthine oxidase. J Biol Chem. 1970 Aug 25;245(16):4053–4057. [PubMed] [Google Scholar]
- Giulivi C., Turrens J. F., Boveris A. Chemiluminescence enhancement by trypanocidal drugs and by inhibitors of antioxidant enzymes in Trypanosoma cruzi. Mol Biochem Parasitol. 1988 Sep;30(3):243–251. doi: 10.1016/0166-6851(88)90093-x. [DOI] [PubMed] [Google Scholar]
- Gonzalez Flecha B., Llesuy S., Boveris A. Hydroperoxide-initiated chemiluminescence: an assay for oxidative stress in biopsies of heart, liver, and muscle. Free Radic Biol Med. 1991;10(2):93–100. doi: 10.1016/0891-5849(91)90002-k. [DOI] [PubMed] [Google Scholar]
- Guilbault G. G., Brignac P. J., Jr, Juneau M. New substrates for the fluorometric determination of oxidative enzymes. Anal Chem. 1968 Jul;40(8):1256–1263. doi: 10.1021/ac60264a027. [DOI] [PubMed] [Google Scholar]
- Jaeschke H., Farhood A., Smith C. W. Neutrophils contribute to ischemia/reperfusion injury in rat liver in vivo. FASEB J. 1990 Dec;4(15):3355–3359. [PubMed] [Google Scholar]
- Jaeschke H., Mitchell J. R. Mitochondria and xanthine oxidase both generate reactive oxygen species in isolated perfused rat liver after hypoxic injury. Biochem Biophys Res Commun. 1989 Apr 14;160(1):140–147. doi: 10.1016/0006-291x(89)91632-x. [DOI] [PubMed] [Google Scholar]
- Joannidis M., Bonn G., Pfaller W. Lipid peroxidation--an initial event in experimental acute renal failure. Ren Physiol Biochem. 1989 Jan-Feb;12(1):47–55. doi: 10.1159/000173179. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Marotto M. E., Thurman R. G., Lemasters J. J. Early midzonal cell death during low-flow hypoxia in the isolated, perfused rat liver: protection by allopurinol. Hepatology. 1988 May-Jun;8(3):585–590. doi: 10.1002/hep.1840080325. [DOI] [PubMed] [Google Scholar]
- McKelvey T. G., Höllwarth M. E., Granger D. N., Engerson T. D., Landler U., Jones H. P. Mechanisms of conversion of xanthine dehydrogenase to xanthine oxidase in ischemic rat liver and kidney. Am J Physiol. 1988 May;254(5 Pt 1):G753–G760. doi: 10.1152/ajpgi.1988.254.5.G753. [DOI] [PubMed] [Google Scholar]
- Metzger J., Dore S. P., Lauterburg B. H. Oxidant stress during reperfusion of ischemic liver: no evidence for a role of xanthine oxidase. Hepatology. 1988 May-Jun;8(3):580–584. doi: 10.1002/hep.1840080324. [DOI] [PubMed] [Google Scholar]
- Parks D. A., Bulkley G. B., Granger D. N., Hamilton S. R., McCord J. M. Ischemic injury in the cat small intestine: role of superoxide radicals. Gastroenterology. 1982 Jan;82(1):9–15. [PubMed] [Google Scholar]
- Parks D. A., Bulkley G. B., Granger D. N. Role of oxygen-derived free radicals in digestive tract diseases. Surgery. 1983 Sep;94(3):415–422. [PubMed] [Google Scholar]
- Parks D. A., Williams T. K., Beckman J. S. Conversion of xanthine dehydrogenase to oxidase in ischemic rat intestine: a reevaluation. Am J Physiol. 1988 May;254(5 Pt 1):G768–G774. doi: 10.1152/ajpgi.1988.254.5.G768. [DOI] [PubMed] [Google Scholar]
- Puntarulo S., Sánchez R. A., Boveris A. Hydrogen peroxide metabolism in soybean embryonic axes at the onset of germination. Plant Physiol. 1988 Feb;86(2):626–630. doi: 10.1104/pp.86.2.626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roldán E. J., Pinus C. R., Turrens J. F., Boveris A. Chemiluminescence of ischaemic and reperfused intestine in vivo. Gut. 1989 Feb;30(2):184–187. doi: 10.1136/gut.30.2.184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takenaka M., Tatsukawa Y., Dohi K., Ezaki H., Matsukawa K., Kawasaki T. Protective effects of alpha-tocopherol and coenzyme Q10 on warm ischemic damages of the rat kidney. Transplantation. 1981 Aug;32(2):137–141. doi: 10.1097/00007890-198108000-00010. [DOI] [PubMed] [Google Scholar]
- Turrens J. F., Beconi M., Barilla J., Chavez U. B., McCord J. M. Mitochondrial generation of oxygen radicals during reoxygenation of ischemic tissues. Free Radic Res Commun. 1991;12-13 Pt 2:681–689. doi: 10.3109/10715769109145847. [DOI] [PubMed] [Google Scholar]
- Turrens J. F., Boveris A. Generation of superoxide anion by the NADH dehydrogenase of bovine heart mitochondria. Biochem J. 1980 Nov 1;191(2):421–427. doi: 10.1042/bj1910421. [DOI] [PMC free article] [PubMed] [Google Scholar]