Abstract
Phosphorus (P) enters roots as inorganic phosphate (Pi) derived from organic and inorganic P compounds in the soil. Nucleic acids can support plant growth as the sole source of P in axenic culture but are thought to be converted into Pi by plant-derived nucleases and phosphatases prior to uptake. Here, we show that a nuclease-resistant analog of DNA is taken up by plant cells. Fluorescently labeled S-DNA of 25 bp, which is protected against enzymatic breakdown by its phosphorothioate backbone, was taken up and detected in root cells including root hairs and pollen tubes. These results indicate that current views of plant P acquisition may have to be revised to include uptake of DNA into cells. We further show that addition of DNA to Pi-containing growth medium enhanced the growth of lateral roots and root hairs even though plants were P replete and had similar biomass as plants supplied with Pi only. Exogenously supplied DNA increased length growth of pollen tubes, which were studied because they have similar elongated and polarized growth as root hairs. Our results indicate that DNA is not only taken up and used as a P source by plants, but ironically and independent of Pi supply, DNA also induces morphological changes in roots similar to those observed with P limitation. This study provides, to our knowledge, first evidence that exogenous DNA could act nonspecifically as signaling molecules for root development.
Phosphorus (P) is an essential macronutrient that limits plant growth in many situations due to a low availability in soils (for review, see Schachtman et al., 1998; Raghothama, 1999; Vance et al., 2003; Lambers et al., 2008). P enters plant roots as orthophosphates (Pi) via active transport across the plasma membrane (Smith et al., 2003; Park et al., 2007; Xu et al., 2007). Concentrations of Pi in soil solution are generally very low (<10 μm; Bieleski, 1973) and plants have evolved root specializations to access P from inorganic and organic sources (Raghothama, 1999; Hinsinger, 2001; López-Bucio et al., 2003; Vance et al., 2003; Lambers et al., 2008). Roots exude enzymes and chemicals to mobilize P directly from soil compounds or indirectly via enhanced activity of soil microbes, and form symbioses with P-mobilizing mycorrhizal fungi (Schachtman et al., 1998; Raghothama, 1999; Bucher, 2007).
However, similar to other nutrients, notably nitrogen, research on P nutrition of plants has focused on inorganic sources although organic P (Porg) in soil can account for 40% to 80% of the total P pool of mineral and organic soils, respectively (Bower, 1945; Raghothama, 1999; Vance et al., 2003). Porg compounds in soils are derived from plant residues, soil biota, and from synthesis by soil microbes (Jencks et al., 1964). Soil Porg is composed primarily of phospholipids, nucleic acids, and phytin (Dyer and Wrenshall, 1941). Phytic acid (inositol hexaphosphate) and its salts phytate, account for a large proportion of the Porg pool of soils (Anderson, 1980). Nucleic acids (RNA, DNA) represent approximately 1% to 2% of the soil Porg pool (Dalal, 1977). It can be released from prokaryotic and eukaryotic cells after death and protected against nuclease degradation by its adsorption on soil colloids and sand particles (Pietramellara et al., 2009).
Although Porg can be a substantial constituent of the soil P pool, its contribution to the P nutrition of plants is poorly understood. Porg can be converted to Pi via root-exuded enzymes (Tarafdar and Claassen, 1988; Marschner, 1995; Vance et al., 2003). Secretion of nucleolytic enzymes and breakdown of nucleic acid were considered the reason for the observed growth of axenic Arabidopsis (Arabidopsis thaliana) and wheat (Triticum aestivum) on nucleic acid substrates as the sole P source (Chen et al., 2000; Richardson et al., 2000).
Whether plants take up intact DNA has not been reported. We recently showed that roots take up protein, possibly via endocytosis (Paungfoo-Lonhienne et al., 2008). We hypothesized that roots may take up DNA by a similar process and grew Arabidopsis in the presence of phosphorothioate oligonucleotides (S-DNA) labeled with Cy3-fluorescent dye. S-DNA has a sulfur backbone and cannot be digested by plant nucleases, allowing tracking DNA of known size into cells (Spitzer and Eckstein, 1988). We examined if S-DNA of 25 nucleotides in length enters root hairs and pollen tubes as both types of cells are strongly elongated and have similar polarized growth (Schiefelbein et al., 1993; Hepler et al., 2001). We also assessed if addition of DNA to the growth medium affects the morphology of roots and pollen tubes. Here, we present evidence that plants take up DNA and demonstrate that the presence of DNA in the growth medium enhances lateral branching of roots, and the length of root hairs and pollen tubes, irrespective of Pi supply.
RESULTS
Arabidopsis Roots Take Up Externally Supplied DNA
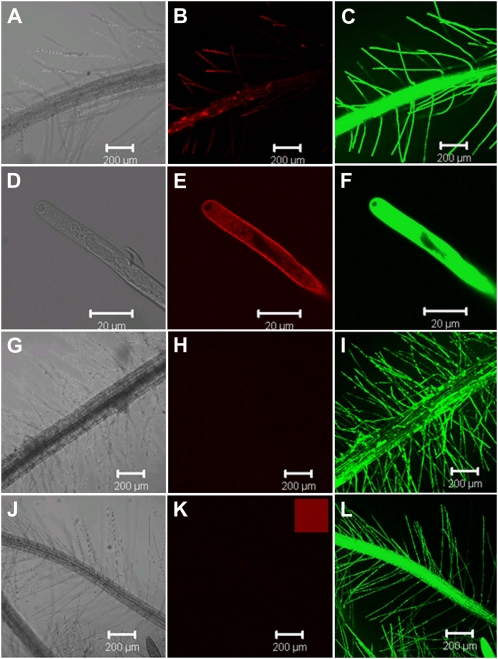
To examine whether DNA, rather than nucleotides or Pi derived from DNA, was taken up by plant roots, nuclease-resistant phosphorothioate DNA (S-DNA) was added to intact roots of axenic Arabidopsis. When 1 μm Cy3-S-DNA (25-bp long) was added to roots of Arabidopsis grown axenically in solid medium, fluorescence was observed in root hairs and root epidermis (Fig. 1, B and E). The same roots showed strong fluorescence when stained with fluorescein diacetate (FDA), indicating that root cells are alive (Fig. 1C; Widholm, 1972). This observation was supported by cytoplasmic streaming in root hair cells in which Cy3-S-DNA was detected (Fig. 1E; Supplemental Video S1).
Figure 1.
Uptake of externally supplied DNA by Arabidopsis roots. Plants were grown on 0.5× MS medium for 11 d before adding 1 μm Cy3-labeled S-DNA (B and E) or 1 μm free Cy3 (H) or 1 μm rhodamine-labeled dextran (K) to the plates and incubated for 3 h. D is an enlarged image of a living root hair cell as indicated by its cytoplasmic streaming (Supplemental Video S1). No fluorescence was observed in plants incubated with free Cy3 (H) or rhodamine-labeled dextran (K). Living root cells were visualized after incubation with 5 μg mL−1 FDA for 1 min (C, F, I, and L). A, D, G, and J are bright-field images of fluorescence images B and C, E and F, H and I, and K and L, respectively. Small square in K shows a fluorescence image of rhodamine-labeled dextran. Images were taken with a confocal microscope.
No fluorescence was observed in plants incubated with free 1 μm Cy3 (Fig. 1H), confirming that fluorescence observed in Cy3-S-DNA incubated roots is not caused by free Cy3 dye entering root cells. We detected no fluorescence in roots of plants incubated with 1 μm Rhodamine Green 3,000 molecular weight dextran (rhodamine dextran; Fig. 1K), a molecule of a molecular mass similar to Cy3-S-DNA, which further lends confidence to the notion that Cy3-S-DNA was actively incorporated into root cells. We also observed internalization of Cy3-S-DNA in roots of Arabidopsis grown axenically in liquid culture (Supplemental Fig. S1), thus excluding the possibility that entry of Cy3-S-DNA was the result of damaging manipulation of roots grown in solid medium. Taken together, the results presented here are strong evidence that Arabidopsis is able to incorporate DNA into root cells by a process specific to DNA.
DNA in Growth Medium Increases Lateral Branching, Lateral Root Length, and Root Hair Length
Plants grown with no or low Pi supply (0.114 μg KH2PO4 per mL growth medium) produced more biomass in the presence of DNA (0.8 mg herring sperm DNA of 120–3,000 nucleotides length per mL growth medium; Supplemental Fig. S2), confirming that DNA was used as a P source as had been previously reported (Chen et al., 2000).
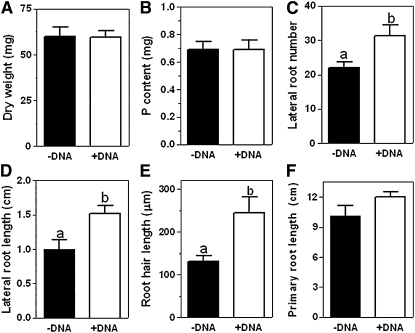
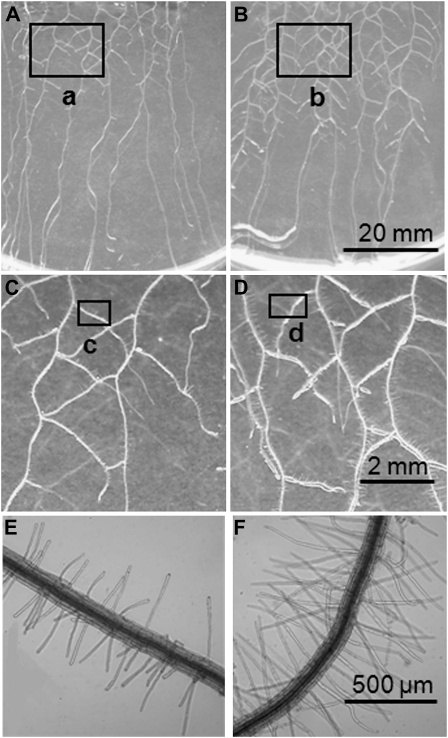
Arabidopsis plants grown with adequate Pi supply (5.7 μg KH2PO4 per mL medium; a standard concentration in Arabidopsis axenic culture) had similar dry weight and P content regardless of the addition of DNA (0.8 mg DNA per mL; Fig. 2, A and B), indicating that plants in both treatments were P replete. However, the presence of DNA in the growth medium induced changes in root morphology (Figs. 2, C–F and 3). P-replete plants grown in the presence of DNA had significantly more lateral roots (31.4 ± 2.9 per plant and 22 ± 1.7 per plant; Figs. 2C and 3, A–D) and greater average lateral root length (1.52 ± 0.1 cm and 0.9 ± 0.1 cm; Fig. 3D) than plants grown with adequate Pi supply alone (P < 0.005). Average root hair length was significantly (P < 0.0001) greater in P-replete plants grown with DNA than with Pi alone (245 ± 11.3 μm and 131 ± 4.2 μm, respectively; Figs. 2E and 3, C–F; Supplemental Fig. S3), while length of the primary root was similar in both treatments (Fig. 2F). Furthermore, these effects on root morphology were also observed when purified plasmid was used as a DNA source (Supplemental Fig. S4), ruling out the possibility that they are triggered by a compound present in the herring sperm DNA.
Figure 2.
Dry weight, P content, and root growth of Arabidopsis plants grown on medium with and without DNA. Dry weight (A), P content (B), and primary root length (F) were similar in plants grown with and without DNA while number of lateral root (C), lateral root length (D), and root hair length (E) increased in response to addition of DNA. Seeds were germinated and cultivated for 14 d (A and B) or 11 d (C–F) on medium containing adequate supply of Pi (5.7 μg KH2PO4 per mL growth medium) without DNA (−DNA) or with DNA (+DNA, 0.8 mg mL−1) added. Bars of A and B represent average and sem of four plates with 100 Arabidopsis plants per plate. Bars represent averages and sem of five plants (C, D, and F) or 10 roots hairs on each of three plants (E). Different letters indicate significant differences at P < 0.005 (C and D) or P < 0.0001 (E). Error bars indicate sem.
Figure 3.
Root growth of axenic Arabidopsis plants on medium with or without externally supplied DNA. Plants were grown for 11 d on medium containing an adequate concentration of Pi (5.7 μg KH2PO4 per mL growth medium without DNA [A, C, and E] or with DNA [0.8 mg DNA per mL growth medium] added [B, D, and F]). Root hair length (D and F) and number of lateral root (B) increased in response to the addition of DNA. C and D are enlarged images of a and b, respectively. E and F are enlarged images of c and d, respectively. Bright-field images of E and F were taken with a confocal microscope.
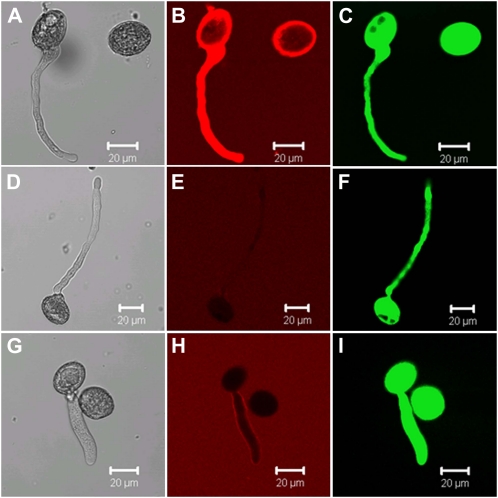
Pollen Tubes of Arabidopsis and Nicotiana benthamiana Take Up S-DNA and Have Enhanced Growth in the Presence of DNA
Root hairs and pollen tubes have similar growth morphology and we therefore tested if pollen tubes take up DNA. Arabidopsis and Nicotiana benthamiana pollen was germinated for 5 or 3 h respectively, in the presence of S-DNA or Cy3-S-DNA to determine the effect on pollen tube growth and possible incorporation of Cy3-S-DNA. As indicated by strong fluorescence inside pollen tubes, Cy3-S-DNA was taken up into Arabidopsis (Fig. 4B) and N. benthamiana pollen (data not shown). Similar to root hairs, pollen tubes did not take up free Cy3 dye (Fig. 4E), confirming that fluorescence observed in pollen incubated with Cy3-S-DNA was due to uptake of the whole molecule (Fig. 4B). The viability of cells taking up S-DNA was assessed using FDA staining and Figure 4C shows that S-DNA detected in pollen tubes was taken up by living cell. Similar to roots, pollen did not take up 1 μm rhodamine dextran (Fig. 4H), supporting the notion that uptake of S-DNA into roots and into pollen tubes occurs via a similar, active process.
Figure 4.
Uptake of Cy3-labeled S-DNA and rhodamine-labeled dextran by Arabidopsis pollen tubes. Bright-field images are shown (A, D, and G) for pollen grown on germination medium for 5 h and incubated with 1 μm Cy3-S-DNA (B), 1 μm Cy3 dye (E), and 1 μm rhodamine dextran (H), respectively, for 1 h. Cy3 (E) and rhodamine dextran (H) were not taken up by pollen. Living root cell were visualized by staining with 5 μg mL−1 FDA for 1 min (C, F, and I). Images were viewed with a confocal microscope.
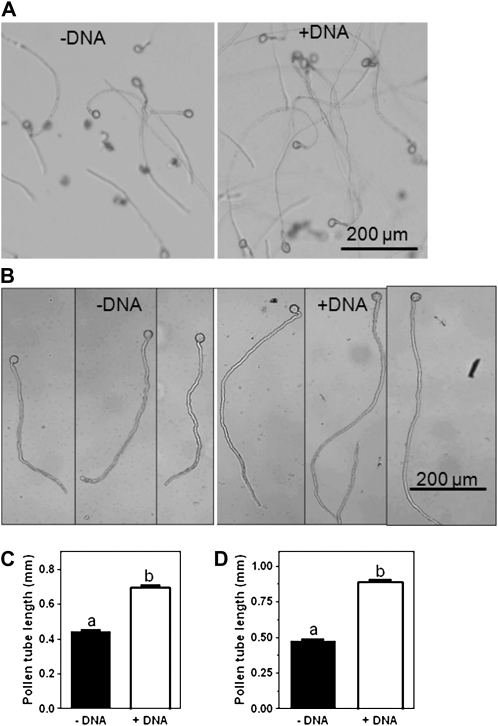
Adding DNA to the germinating medium as plasmid, circular double-stranded DNA of 5,006 nucleotides in length, significantly (P < 0.0001) increased the length of pollen tubes (Fig. 5). Pollen tube growth experiments were carried out with plasmid DNA because, in contrast to fish sperm DNA used in the whole-plant experiments described above, plasmid DNA does not precipitate in the pollen-germinating medium that contains polyethylene glycol. The length of Arabidopsis pollen tubes with and without DNA in the germinating medium differed significantly (P < 0.0001), averaging 0.69 ± 0.012 mm (mean ± sem) and 0.44 ± 0.008 mm, respectively (Fig. 5C). Similarly, average lengths of N. benthamiana pollen tubes were 0.89 ± 0.0169 mm (with DNA added) and 0.47 ± 0.013 mm (without DNA added; Fig. 5D).
Figure 5.
Pollen tube growth in response to external DNA supply. Pollen tubes of Arabidopsis (A) and N. benthamiana (B) grow longer with DNA (0.4 mg plasmid per mL germination buffer) added to the growth medium. The length of Arabidopsis (C) and N. benthamiana (D) pollen tubes was significantly (P < 0.0001) greater in DNA-containing medium as indicated by different letters. Pollen was viewed after 3 h of incubation in germination medium. Different letters indicate significant differences at P < 0.0001 (C–D). Error bars indicate sem.
DISCUSSION
This study provides, to our knowledge, the first evidence that plants take up Porg in the form of DNA. Following the observation that axenically grown Arabidopsis and wheat can grow with DNA as the sole source of P, it was proposed that prior to uptake, DNA is converted into Pi by root-exuded nucleases (Abel et al., 2000; Richardson et al., 2000). We used nuclease-resistant S-DNA, 25-bp long and labeled with Cy3-fluorescent dye, to demonstrate that root hairs and pollen tubes take up S-DNA. S-DNA has a mass of approximately 16.5 kD, somewhat less than GFP (26.9 kD) that is also incorporated into roots (Paungfoo-Lonhienne et al., 2008). Cy3 dye is much smaller than S-DNA and GFP with a mass of 767 D (http://flowcyt.salk.edu/fluo.html) and was not incorporated into roots when administered alone. Furthermore, we show that roots of Arabidopsis cultivated in liquid medium took up S-DNA, confirming that entry of S-DNA is not due to damage and passive leakage into roots or root hairs that could occur during manipulation of roots grown on solid media. The pattern of fluorescence observed following uptake of Cy3-labeled S-DNA is strikingly similar to those seen in roots that have taken up GFP (Paungfoo-Lonhienne et al., 2008) as well as in transgenic plants expressing GFP under the control of trichoblast-specific phosphate transporter promoters (Mudge et al., 2002, 2003). The observation that DNA enters root trichoblasts (i.e. root hairs) and pollen tubes provides further evidence that large exogenous biomolecules can enter plant cells.
Root hairs are tip-growing, tubular-shaped outgrowths that emerge from the basal end of specialized trichoblast cells (Foreman and Dolan, 2001). Root hairs substantially increase the absorbing surface of roots and have a main role for the uptake of water and nutrients into plants (Greulach and Adams, 1967; Parker et al., 2000). The uptake of S-DNA and previously reported uptake of protein (Paungfoo-Lonhienne et al., 2008) appears to be restricted to root-hair-producing trichoblast cells and root hairs. Root hairs are the major site of Pi uptake (Gahoonia and Nielsen, 1998) as trichoblasts express the high-affinity phosphate transporters responsible for Pi uptake (Mudge et al., 2002, 2003; Schunmann et al., 2004). The observation that uptake of larger molecules such as DNA and protein occurs in trichoblasts indicates that these specialized epidermal cells may have a more wide-ranging ability for the acquisition of compounds from the soil solution than previously considered. This ability is not restricted to Arabidopsis, since uptake of Cy3-S-DNA also occurred into root hairs and root-hair-producing trichoblasts of tomato (Solanum lycopersicum; Supplemental Fig. S5), which in contrast to nonmycorrhizal Arabidopsis, forms endomycorrhizal symbioses.
Root hairs and pollen tubes have similar polarized growth (Schiefelbein et al., 1993), and pollen is used to study uptake facilitated by membrane transporters (Komarova et al., 2008). We investigated whether pollen tubes resemble root hairs in their ability to take up DNA. The results showed that pollen tubes of Arabidopsis and N. benthamiana take up Cy3-labeled S-DNA, and, similar to root hairs, pollen tubes grew longer in the presence of DNA. This observation suggests that similar mechanisms of DNA uptake and growth stimulation exist in root hairs and pollen tubes.
The second key finding of the research presented here is that exogenously supplied DNA affects root morphology and pollen tube growth. Plants increase root branching, root length, root hair density, and length in response to P deficiency (Bates and Lynch, 1996; Ma et al., 2001; Gojon et al., 2009), yet results presented here show that root branching, length of lateral roots, and length of root hairs increase in the presence of DNA, irrespective of the plant P nutrition status. Biomass and P contents were similar in plants grown with adequate supply of Pi in the presence or absence of DNA, and the observed increases in root branching and root hair length are therefore not a response to low P supply. Similarly, DNA-derived nitrogen or reduced carbon compounds are an unlikely cause for the observed changes in root morphology because plants were adequately supplied with these compounds in the growth medium.
The ecological significance of the research presented here may be explained by the need of plants to forage for localized supplies of nutrients (Hutchings and Dekroon, 1994). Root development responds to heterogeneous distribution of nutrients and lateral roots proliferate preferentially within nutrient-rich zones (Jackson et al., 1990; Robinson, 1994). Similarly to other P forms in soil, DNA is adsorbed by soil colloids and particles (Pietramellara et al., 2009), and DNA-induced root proliferation could enhance access to not only this P source but to other colocated nutrients. Indeed, it is possible that presence of DNA is an environmental cue for enrichment of multiple nutrients in the soil, especially associated with decaying organic matter. Our results indicate that exogenous DNA could act nonspecifically as signaling molecules for root development, but further research is required to elaborate the mechanisms and the ecological relevance.
CONCLUSION
We have shown that a nuclease-resistant analog of DNA enters root hairs and pollen tubes to provide further evidence that exogenous biomolecules of high molecular mass can enter plant cells. Our study also presents evidence that Porg may be more important as a source of P for plants than previously considered and that exogenous DNA increases lateral root branching and root hair length by a mechanism independent of plant P status.
MATERIALS AND METHODS
DNA Preparations
For Arabidopsis (Arabidopsis thaliana) growth experiments, we used herring sperm DNA fragments, 120 to 3,000 nucleotides in length (10 mg DNA mL−1, Roche; Chen et al., 2000). To eliminate any minor contamination with small DNA fragments or free nucleic acids, herring sperm DNA was solubilized in sterile distilled water, filter sterilized (0.22-μm Millipore Filter), and twice subjected to dialysis at 4°C against distilled water (1:100 v/v) for 12 h. The dialysis tubing (Spectra/Por, Spectrum Laboratories Inc.) has a nominal Mr cutoff of 25 kD to remove traces of any contaminants of low molecular mass. The resulting DNA (8 mg mL−1) and the remaining dialysis water were stored in aliquots at −20°C. The Arabidopsis growth medium (see below) was supplemented either with dialyzed DNA (+DNA treatment), or with an equivalent volume of the remaining dialysis water (−DNA treatment). Because herring sperm DNA precipitates in germination medium of pollen, plasmid DNA was used instead. Plasmid DNA was isolated using a QIAGEN plasmid giga kit, and diluted in distilled water to a final concentration of 4 mg DNA mL−1 (pGem-T Easy vector, Promega) and used for pollen tube growth experiments. To exclude any chance that root growth was altered by biologically active compounds present in the herring sperm DNA, the root growth experiment was repeated using purified plasmid DNA.
To study the uptake of fluorescent DNA by Arabidopsis plants and pollen of Arabidopsis and Nicotiana benthamiana, we used Cy3-DNA (Sigma-Proligo) and Cy3-S-DNA (Sigma-Genosys). Cy3-DNA was 25 nucleotides in length (5′-ATGGTGAGCAAGGGCGAGGAGCTGT-3′) labeled with Cy3 at its 5′ end, and hybridized with its unlabeled antisense oligonucleotide (5′-ACAGCTCCTCGCCCTTGCTCACCAT-3′). Cy3-S-DNA (Sigma-Genosys) was the same as Cy3-DNA, except that the labeled strand had phosphorothioate rather than phosphodiester linkages along its backbone. These sequences showed no significant homology (i.e. ≤14 nucleotide perfect match) with sequences in the Arabidopsis genome. Cy3 was used as the negative control (Molecular Probes).
Plant Material and Growth Conditions
Arabidopsis (ecotype Columbia-0) was used in this study. To meet the specific requirements of individual experiments, one of the following growth conditions was used.
Petri Dish Culture
Surface-sterilized seeds were grown on petri dishes containing 25 mL of P-free Murashige and Skoog (MS; Murashige and Skoog, 1962) basal salt solution (M0529, Sigma-Aldrich) supplemented with 5 g L−1 Suc, 5 mm KNO3, 2 mm MgSO4, 2 mm Ca(NO3)2, and 2.5 mm MES (pH 5.5). The media were adjusted to pH 5.5 and 0.3% phytagel (Phytotechnologies) was used as solidifying substance. Phytagel contains a minor amount of P (1 μm ± 0.5; Bates and Lynch, 1996) and was used for all experiments. P was added as Pi (1.25 mm KH2PO4) or as a combination of Pi (1.25 mm KH2PO4) and DNA (0.8 mg mL−1 of herring sperm DNA, equaling about 2.58 mm P). Plated seeds were incubated in a cold room for 3 d and then transferred to a growth cabinet (21°C, 16 h/8 h day/night, 150 μmol m−2 s−1) for growth in a vertical position. After 14 d, plants were rinsed and cleaned three times in 0.5 mm CaCl2 to remove P from plant surfaces. Plants were dried at 60°C for 2 d, weighed, homogenized, and analyzed for P with a combustion elemental analyzer (Thermo Finnigan EA 1112 Series, CE Instruments). The results represent averages and sem of four plates with 100 Arabidopsis plants per plate.
For the root growth experiment, seedlings were grown vertically for 11 d before root morphology was assessed. The length of 10 randomly selected root hairs was measured in three 2-mm segments at approximately 2-mm distance from the main root axis. Primary root length, lateral root number, and lateral root lengths of five plants were measured using the WinRhizo 2007 program (Reagent Instruments Inc.) and the experiment was repeated twice.
For uptake experiments with fluorescent DNA, axenic Arabidopsis and tomato (Solanum lycopersicum) were grown vertically on MS medium (Murashige and Skoog, 1962) for 11 d. One micrometer of Cy3 (negative control) or Cy3-S-DNA was carefully poured on the surface of the media. Incubation was carried out for 3 h in the dark and at room temperature. Roots were carefully removed with forceps taking the entire plant from the medium and immediately washed twice with water before being viewed under confocal microscope as described below. Concurrently, 1 μm of Rhodamine Green 3,000 molecular weight dextran (rhodamine dextran; Molecular Probes) was used as another control for the uptake of high molecular mass compounds.
Liquid Culture
In addition to solid culture, we grew plants in axenic liquid culture to ensure that injury of roots after removal from the culture medium is not the cause of the observed incorporation of DNA (Supplemental Fig. S1). Briefly, Arabidopsis plants were grown axenically for 7 d in 1 mL 0.5× strength MS medium in 24-well cell culture plates. The plates were aerated by placing on a shaker table rotating at 80 rpm. The roots were rinsed twice with 0.5× strength MS medium, and then Cy3-S-DNA was added to a final concentration of 0.2 μm. Roots were washed after 3 h of incubation and viewed with a confocal microscope (see below).
To ensure that the root growth was not an effect from any biologically active compound contained in herring sperm DNA, Arabidopsis plants were axenically grown for 10 d in a 12-well cell culture plate containing 2 mL 0.5× strength MS medium with or without purified plasmid DNA (0.4 mg mL−1) added. The length of 10 randomly selected root hairs was measured in three 2-mm segments at approximately 2-mm distance from the main root axis. Lateral root number, and lateral root lengths of 18 plants were measured as described above.
In Vitro Pollen Germination
For pollen growth experiments, pollen from three to five flowers of Arabidopsis or N. benthamiana was suspended in 45 μL of germination buffer (Tang et al., 2002) supplemented with 5 μL of distilled water or 5 μL of plasmid solution (4 μg plasmid μL−1). Pollen was germinated on hanging drops (20 μL) at 22°C for 5 h (Arabidopsis) or 6 h (N. benthamiana). Germinated pollen was examined via microscopy (Nikon Eclipse E600) or confocal microscopy (Carl Zeiss). Determination of Arabidopsis and N. benthamiana pollen tube length was based on 100 pollen in three independent experiments.
For studying the uptake of fluorescent DNA, Arabidopsis pollen was germinated as described above with 1 μm of Cy3-S-DNA, 1 μm of Cy3 (negative control), or 1 μm rhodamine dextran incorporated in the germination buffer. After 5 h incubation, pollen was examined with confocal microscopy as described below.
Determination of Cell Viability
Cell viability were assessed with FDA (Sigma-Aldrich) according to the method of Widholm (1972). FDA (0.2%; w/v) stock solution in acetone was diluted to a final concentration of 5 μg mL−1 with culture medium and used for staining plant roots and pollen on a microscopic slide for 1 min. Roots were washed twice before being observed under the confocal microscope.
Confocal Microscopy and Image Analysis
A Zeiss LSM510 META (Carl Zeiss) confocal laser-scanning microscope was used with 10× dry, 20× water immersion objectives, and 40× or 60× oil immersion objectives. DNA or S-DNA labeled with Cy3 dye and dextran labeled with rhodamine were visualized by excitation with a HeNe laser at 543 nm and detection with a 560 to 615 nm band-path filter. Fluorescein, actively converted from nonfluorescent FDA by living cells, was detected by excitation with an Argon laser at 488 nm and detection with a 505 to 530 band-path filter.
Root hairs and pollen tubes were observed and measured with either a fluorescence microscope (Nikon Eclipse E600) or the confocal microscope. Images were captured with a SPOT camera and imported into SPOT RT imaging software v3.5 (SPOT Diagnostic Instrument, Inc.) for the fluorescence microscope or with a Zeiss LSM image browser (Carl Zeiss) for the confocal microscope.
Statistical Analysis
Statistical analyses were performed using Student's t tests (GraphPad Prism4, GraphPad Software, Inc.).
Supplemental Data
The following materials are available in the online version of this article.
Supplemental Figure S1. Uptake of DNA by roots of Arabidopsis plants grown in liquid medium.
Supplemental Figure S2. The presence of externally supplied DNA increased growth of Arabidopsis grown without or with suboptimal amount of Pi.
Supplemental Figure S3. Main root profile of Arabidopsis on medium containing adequate P content with (+DNA) or without (−DNA) the addition of DNA.
Supplemental Figure S4. Dry weight and root growth of Arabidopsis plants grown on medium with and without plasmid DNA.
Supplemental Figure S5. Uptake of DNA by roots of tomato.
Supplemental Video S1. Cytoplasmic streaming of Cy3-labeled S-DNA taken up into a root hair of Arabidopsis.
Supplementary Material
Acknowledgments
We thank Kerry Vinall for assisting with root measurements using Winrhizo. Thanks to Dr. Henrik Svennerstam for technical advice on axenic hydroponic culture and Professor Doris Rentsch on pollen tube growth. We are grateful to the Australian Research Council Centre of Excellence for Integrative Legume Research for access to research facilities.
References
- Abel S, Nurnberger T, Ahnert V, Krauss GJ, Glund K. (2000) Induction of an extracellular cyclic nucleotide phosphodiesterase as an accessory ribonucleolytic activity during phosphate starvation of cultured tomato cells. Plant Physiol 122: 543–552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson G. (1980) Assessing organic phosphorus in soils. Khasawneh F, Sample E, Kamprath E, , The Role of Phosphorus in Agriculture. American Society of Agronomy, Madison, WI, pp 411–432 [Google Scholar]
- Bates TR, Lynch JP. (1996) Stimulation of root hair elongation in Arabidopsis thaliana by low phosphorus availability. Plant Cell Environ 19: 529–538 [Google Scholar]
- Bieleski R. (1973) Phosphate pools, phosphate transport, and phosphate availability. Annu Rev Plant Physiol 24: 225–252 [Google Scholar]
- Bower C. (1945) Separation and identification of phytin and its derivatives from soils. Soil Sci 59: 277–285 [Google Scholar]
- Bucher M. (2007) Functional biology of plant phosphate uptake at root and mycorrhiza interfaces. New Phytol 173: 11–26 [DOI] [PubMed] [Google Scholar]
- Chen DL, Delatorre CA, Bakker A, Abel S. (2000) Conditional identification of phosphate-starvation-response mutants in Arabidopsis thaliana. Planta 211: 13–22 [DOI] [PubMed] [Google Scholar]
- Dalal RC. (1977) Soil organic phosphorus. Adv Agron 29: 83–117 [Google Scholar]
- Dyer B, Wrenshall CL. (1941) Organic P in soils. I. The extraction and separation of organic phosphorus compounds from soils. Soil Sci 51: 159–170 [Google Scholar]
- Foreman J, Dolan L. (2001) Root hairs as a model system for studying plant cell growth. Ann Bot (Lond) 88: 1–7 [Google Scholar]
- Gahoonia TS, Nielsen NE. (1998) Direct evidence on participation of root hairs in phosphorus (32P) uptake from soil. Plant Soil 198: 147–152 [Google Scholar]
- Gojon A, Nacry P, Davidian JC. (2009) Root uptake regulation: a central process for NPS homeostasis in plants. Curr Opin Plant Biol 12: 328–338 [DOI] [PubMed] [Google Scholar]
- Greulach VA, Adams JE. (1967) Tissues and organs. Greulach VA, Adams JE, , Plants: An Introduction to Modern Botany, Ed 2 John Wiley and Sons, Inc, New York, pp 145–203 [Google Scholar]
- Hepler PK, Vidali L, Cheung AY. (2001) Polarized cell growth in higher plants. Annu Rev Cell Dev Biol 17: 159–187 [DOI] [PubMed] [Google Scholar]
- Hinsinger P. (2001) Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: a review. Plant Soil 237: 173–195 [Google Scholar]
- Hutchings MJ, Dekroon H. (1994) Foraging in plants—the role of morphological plasticity in resource acquisition. Adv Ecol Res 25: 159–238 [Google Scholar]
- Jackson R, Manwaring J, Caldwell M. (1990) Rapid physiological adjustment of roots to localized soil enrichment. Nature 344: 58–60 [DOI] [PubMed] [Google Scholar]
- Jencks EM, Raese JT, Reese CD. (1964) Organic Phosphorus Content of Some West Virginia Soils, Bulletin 489. Agricultural Experiment Station, West Virginia University, Morgantown, WV, pp 1–16 [Google Scholar]
- Komarova NY, Thor K, Gubler A, Meier S, Dietrich D, Weichert A, Suter Grotemeyer M, Tegeder M, Rentsch D. (2008) AtPTR1 and AtPTR5 transport dipeptides in planta. Plant Physiol 148: 856–869 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambers H, Raven JA, Shaver GR, Smith SE. (2008) Plant nutrient-acquisition strategies change with soil age. Trends Ecol Evol 23: 95–103 [DOI] [PubMed] [Google Scholar]
- López-Bucio J, Cruz-Ramírez A, Herrera-Estrella L. (2003) The role of nutrient availability in regulating root architecture. Curr Opin Plant Biol 6: 280–287 [DOI] [PubMed] [Google Scholar]
- Ma Z, Bielenberg DG, Brown KM, Lynch JP. (2001) Regulation of root hair density by phosphorus availability in Arabidopsis thaliana. Plant Cell Environ 24: 459–467 [Google Scholar]
- Marschner H. (1995) Mineral Nutrition of Higher Plants, Ed 2 Academic Press Ltd, London, pp 559–561 [Google Scholar]
- Mudge SR, Rae AL, Diatloff E, Smith F. (2002) Expression analysis suggests novel roles for members of the Pht1 family of phosphate transporters in Arabidopsis. Plant J 31: 341–353 [DOI] [PubMed] [Google Scholar]
- Mudge SR, Smith FW, Richardson AE. (2003) Root-specific and phosphate-regulated expression of phytase under the control of a phosphate transporter promoter enables Arabidopsis to grow on phytate as a sole P source. Plant Sci 165: 871–878 [Google Scholar]
- Murashige T, Skoog F. (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15: 473–497 [Google Scholar]
- Park MR, Baek SH, de los Reyes BG, Yun SJ. (2007) Overexpression of a high-affinity phosphate transporter gene from tobacco (NtPT1) enhances phosphate uptake and accumulation in transgenic rice plants. Plant Soil 292: 259–269 [Google Scholar]
- Parker JS, Cavell AC, Dolan L, Roberts K, Grierson CS. (2000) Genetic interactions during root hair morphogenesis in Arabidopsis. Plant Cell 12: 1961–1974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paungfoo-Lonhienne C, Lonhienne TGA, Rentsch D, Robinson N, Christie M, Webb RI, Gamage HK, Carroll BJ, Schenk PM, Schmidt S. (2008) Plants can use protein as a nitrogen source without assistance from other organisms. Proc Natl Acad Sci USA 11: 4524–4529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietramellara G, Ascher J, Borgogni F, Ceccherini MT, Guerri G, Nannipieri P. (2009) Extracellular DNA in soil and sediment: fate and ecological relevance. Biol Fertil Soils 45: 219–235 [Google Scholar]
- Raghothama KG. (1999) Phosphate acquisition. Annu Rev Plant Physiol Plant Mol Biol 50: 665–693 [DOI] [PubMed] [Google Scholar]
- Richardson AE, Hadobas PA, Hayes JE. (2000) Acid phosphomonoesterase and phytase activities of wheat (Triticum aestivum L.) roots and utilization of organic phosphorus substrates by seedlings grown in sterile culture. Plant Cell Environ 23: 397–405 [Google Scholar]
- Robinson D. (1994) The responses of plants to non-uniform supplies of nutrients. New Phytol 127: 635–674 [DOI] [PubMed] [Google Scholar]
- Schachtman DP, Reid RJ, Ayling SM. (1998) Phosphorus uptake by plants: from soil to cell. Plant Physiol 116: 447–453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiefelbein J, Galway M, Masucci J, Ford S. (1993) Pollen tube and root-hair tip growth is disrupted in a mutant of Arabidopsis thaliana. Plant Physiol 103: 979–985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schunmann P, Richardson A, Vickers C, Delhaize E. (2004) Promoter analysis of the barley Pht1;1 phosphate transporter gene identifies regions controlling root expression and responsiveness to phosphate deprivation. Plant Physiol 136: 4205–4214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith FW, Mudge SR, Rae AL, Glassop D. (2003) Phosphate transport in plants. Plant Soil 248: 71–83 [Google Scholar]
- Spitzer S, Eckstein F. (1988) Inhibition of deoxyribonucleases by phosphorothioate groups in oligodeoxyribonucleotides. Nucleic Acids Res 16: 11691–11704 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang W, Ezcurra I, Muschietti J, McCormick S. (2002) A cysteine-rich extracellular protein, LAT52, interacts with the extracellular domain of the pollen receptor kinase LePRK2. Plant Cell 14: 2277–2287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarafdar JC, Claassen N. (1988) Organic phosphorus compounds as a phosphorus source for higher plants through the activity of phosphatases produced by plant roots and microorganisms. Biol Fertil Soils 5: 308–312 [Google Scholar]
- Vance C, Uhde-Stone C, Allan D. (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157: 423–447 [DOI] [PubMed] [Google Scholar]
- Widholm JM. (1972) Use of fluorescein diacetate and phenosafranine for determining viability of cultured plant-cells. Stain Technol 47: 189–194 [DOI] [PubMed] [Google Scholar]
- Xu GH, Chague V, Melamed-Bessudo C, Kapulnik Y, Jain A, Raghothama KG, Levy AA, Silber A. (2007) Functional characterization of LePT4: a phosphate transporter in tomato with mycorrhiza-enhanced expression. J Exp Bot 58: 2491–2501 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.