Abstract
Nitro-oleic acid (OA-NO2), an electrophilic fatty acid by-product of nitric oxide and nitrite reactions, is present in normal and inflamed mammalian tissues at up to micromolar concentrations and exhibits anti-inflammatory signaling actions. The effects of OA-NO2 on cultured dorsal root ganglion (DRG) neurons were examined using fura-2 Ca2+ imaging and patch clamping. OA-NO2 (3.5–35 μM) elicited Ca2+ transients in 20 to 40% of DRG neurons, the majority (60–80%) of which also responded to allyl isothiocyanate (AITC; 1–50 μM), a TRPA1 agonist, and to capsaicin (CAPS; 0.5 μM), a TRPV1 agonist. The OA-NO2-evoked Ca2+ transients were reduced by the TRPA1 antagonist 2-(1,3-dimethyl-2,6-dioxo-1,2,3,6-tetrahydro-7H-purin-7-yl)-N-(4-isopropylphenyl) acetamide (HC-030031; 5–50 μM) and the TRPV1 antagonist capsazepine (10 μM). Patch-clamp recording revealed that OA-NO2 depolarized and induced inward currents in 62% of neurons. The effects of OA-NO2 were elicited by concentrations ≥5 nM and were blocked by 10 mM dithiothreitol. Concentrations of OA-NO2 ≥5 nM reduced action potential (AP) overshoot, increased AP duration, inhibited firing induced by depolarizing current pulses, and inhibited Na+ currents. The effects of OA-NO2 were not prevented or reversed by the NO-scavenger carboxy-2-phenyl-4,4,5,5-tetramethylimidazolineoxyl-1-oxyl-3-oxide. A large percentage (46–57%) of OA-NO2-responsive neurons also responded to CAPS (0.5 μM) or AITC (0.5 μM). OA-NO2 currents were reduced by TRPV1 (diarylpiperazine; 5 μM) or TRPA1 (HC-030031; 5 μM) antagonists. These data reveal that endogenous OA-NO2 generated at sites of inflammation may initially activate transient receptor potential channels on nociceptive afferent nerves, contributing to the initiation of afferent nerve activity, and later suppresses afferent firing.
Nitrated fatty acids, including nitro-oleic acid (OA-NO2), nitro-linoleic acid and nitro-arachidonic acid, are present in tissues at low levels under normal conditions, and their levels are increased postprandially and during inflammation (Ferreira et al., 2009; Rudolph et al., 2010). These agents elicit a variety of cellular effects, including activation of peroxisome proliferator-activated receptor γ, signal transducer and activator of transcription 1, nuclear factor erythroid 2-related factor 2, and mitogen-activated protein phosphatase 1 signaling pathways, as well as induction of heme oxygenase 1 expression and inhibition of nuclear factor-κB-dependent gene expression (Ichikawa et al., 2008; Li et al., 2008; Kansanen et al., 2009; Wright et al., 2009). Exogenous administration of nitrated fatty acids mediates anti-inflammatory effects in cell and animal models. Nitrated fatty acids, bearing a nitroalkene configuration, are electrophilic and modulate intracellular signaling pathways by undergoing post-translational protein adduction reactions that target nucleophilic residues, such as cysteine and histidine (Baker et al., 2007; Li et al., 2008; Nadtochiy et al., 2009; Rudolph et al., 2010).
The TRP family of ion channels, including TRPV1 and TRPA1, have ankyrin-like repeats in the N terminus that are rich in cysteine residues (Bandell et al., 2007; Macpherson et al., 2007) that react with electrophiles and other thiol-modifying species (Bandell et al., 2007; Macpherson et al., 2007; Salazar et al., 2008). TRP channels, which are expressed in nociceptive afferent fibers, can mediate neurogenic inflammation and pain induced by noxious chemicals or thermal stimuli. TRPV1, the temperature-acid-sensing receptor activated by capsaicin (CAPS) (Montell et al., 2002), and TRPA1, the cold-temperature-noxious chemical sensory channel activated by allyl-isothiocyanate (Bandell et al., 2007; Macpherson et al., 2007), both stimulate nociceptive afferent terminals and stimulate the release of neurotransmitters, e.g., neurokinins that induce plasma extravasation and mast cell activation. In addition, neurokinins released at afferent terminals could act in an autofeedback manner to enhance firing in dorsal root ganglion (DRG) neurons (Sculptoreanu and de Groat, 2007) and enhance TRPV1 activity (Sculptoreanu et al., 2008). Induction of hyperexcitability of DRG neurons is a feature of various inflammatory conditions such as cyclophosphamide-induced cystitis (Yoshimura and de Groat, 1999), gastric ulcer (Dang et al., 2004), pancreatitis (Xu et al., 2006), and feline interstitial cystitis (Sculptoreanu et al., 2005).
Here, the effects of nitro-oleic acid on the properties of nociceptive afferent neurons expressing TRPV1 and/or TRPA1 channels were evaluated to determine whether neural actions might contribute to the anti-inflammatory effects of OA-NO2. The results indicate that OA-NO2 elicits multiple effects on cultured DRG neurons from adult rats, including activation of TRPV1 and TRPA1 channels and suppression of excitability and action potential (AP) generation and inhibition of voltage-dependent Na+ currents.
Materials and Methods
Cell Isolation.
Neurons were isolated from L4-S3 DRG of adult male rats using methods described previously (Sculptoreanu et al., 2008). In brief, freshly removed ganglia were minced and enzymatically digested at 37°C for 10 min in Dulbecco's modified Eagle's medium (DMEM) containing 2.5 mg/ml trypsin, followed by 50 min in DMEM containing 2.5 mg/ml collagenase D (Boehringer Mannheim Corp., Louisville, KY) and 4 mg/ml trypsin inhibitor type 2S (Sigma-Aldrich, St. Louis, MO). The ganglia were then dissociated mechanically by trituration using siliconized Pasteur pipettes. The cell suspension was layered on DMEM containing 50% bovine serum and centrifuged for 10 min at 800 rpm (150g) at room temperature to remove most of the debris and broken cells. After withdrawing the supernatant, the pellet containing neurons was resuspended in DMEM containing 10% heat-inactivated horse serum and 5% fetal bovine serum and then plated on collagen-coated 35-mm petri dishes (BioCoat; BD Biosciences, Franklin Lakes, NJ) and kept in a 95% air and 5% CO2 incubator at 37°C until recording. For Ca2+ imaging, neurons were plated on collagen-coated glass coverslips. Medium was replaced after 2 h of incubation. Cells were used within 4 to 48 h after dissociation for Ca2+ imaging and within 2 to 5 days for electrophysiology.
Calcium Imaging.
DRG neurons were loaded with fura-2 acetoxymethyl ester (2 μM; Invitrogen, Carlsbad, CA) for 30 min at 37°C in an atmosphere of 5% CO2. Fura-2 acetoxymethyl ester was dissolved in the bath solution (Hanks' balanced salt solution; HBSS) containing 138 mM NaCl, 5 mM KCl, 0.3 mM KH2PO4, 4 mM NaHCO3, 2 mM CaCl2, 1 mM MgCl2, 10 mM HEPES, and 5.6 mM glucose, pH 7.4; 310 mOsm/l) to which bovine serum albumin (5 mg/ml; Sigma-Aldrich) was added to promote dye loading. Fura-2 Ca2+ imaging was performed as described previously (Kullmann et al., 2008). In short, coverslips were placed on an inverted epifluorescence microscope (IX70; Olympus, Tokyo, Japan) and continuously superfused with HBSS. Fura-2 was excited alternately with UV light at 340 and 380 nm, and the fluorescence emission was detected at 510 nm using a computer-controlled monochromator. Image pairs were acquired every 5 to 30 s using illumination periods between 100 and 250 ms in duration. Wavelength selection, timing of excitation and acquisition of images, were controlled using the program C-Imaging (Compix Inc., Cranberry Township, PA). Digital images were stored for off-line analysis.
Drugs were dissolved in external solution from concentrated stock solutions and delivered via bath application using a gravity-driven application system. A stock solution of capsaicin (33 mM) was made in 10% ethanol, 10% Tween 80, and 80% physiological saline. A stock solution of allyl isothiocyanate (AITC; 600 mM) was made in dimethyl sulfoxide (DMSO) and further diluted in HBSS to final concentration. A stock solution of OA-NO2 (70 mM) was prepared in ethanol and diluted in HBSS to final concentration. The TRPA1 antagonist HC-030031 (Hydra Biosciences, Inc., Cambridge, MA) was dissolved in DMSO (100 mM), and the TRPV1 antagonist capsazepine (Sigma-Aldrich) was dissolved in DMSO (10 mM) before serial dilution in HBSS. Dithiothreitol (DTT; Sigma-Aldrich) was dissolved in aqueous buffer. The OA-NO2 was synthesized as described previously (Baker et al., 2005). At final concentrations, vehicle solutions did not alter intracellular Ca2+ concentration ([Ca2+]i). The water-soluble NO scavenger carboxy-2-phenyl-4,4,5,5-tetramethylimidazolineoxyl-1-oxyl-3-oxide (cPTIO; Maeda et al., 1994) was prepared in the recording buffer at 100× the final concentration just before doing the experiments.
Image Analysis.
Image analysis was performed using the program C-Imaging (Compix Inc.). Background was subtracted to minimize camera dark noise and tissue autofluorescence. An area of interest was drawn around each cell, and the average value of all pixels included in this area was taken as one measurement. The ratio (R) of fluorescence signal measured at 340 nm, divided by the fluorescence signal measured at 380 nm, is proportional to [Ca2+]i. Baseline [Ca2+]i was determined from the average of five to eight measurements obtained before drug application. The agonists were applied alone, washed out, and then applied again in the continuous presence of an antagonist. Amplitudes of peak [Ca2+]i responses were computed as the difference between the peak value and the baseline value. To be considered a drug-induced response, changes in [Ca2+]i had to occur within 2 to 3 min after drug application, and the amplitudes had to exceed baseline by 2 S.D.s. The results are given as changes in R before and after drug application and as percentage increase of R (ΔR/R) above resting levels of [Ca2+]i. Data analysis was performed using Excel (Microsoft, Redmond, WA), Origin (OriginLab Corp., Northampton, MA), and Prism 4 (GraphPad Software Inc., San Diego, CA).
Electrophysiology.
Gigaohm-seal recordings of CAPS currents were obtained in DRG neurons using whole-cell patch-clamp techniques. Neurons from cultures older than 5 days that exhibited extensive processes and large slowly decaying capacitive currents were excluded in our analysis. Patch pipettes were pulled from capillary glass tubes (Accufil 90; Clay-Adams, Batavia, IL) and fire polished. Immediately before recording, the serum-containing medium was replaced with phosphate-buffered saline. Whole-cell currents were voltage-clamped using an Axopatch 200A amplifier (MDS, Inc. Toronto, ON, Canada). Pulse generation, current recording and data analysis used pClamp software (MDS, Inc. Toronto, ON, Canada). Currents were sampled at 500 μs and filtered at 2 kHz. Capacitive currents and up to 80% of the series resistance were compensated.
The extracellular solution for patch clamping was Dulbecco's phosphate buffer (Sigma-Aldrich), which contained 138 mM NaCl, 2.6 mM KCl, 0.9 mM CaCl2, 0.5 mM MgCl2, 1.5 mM KH2PO4, and 8.1 mM Na2HPO4. The pipette (intracellular) solution contained 120 mM KCl, 10 mM K2HPO4, 10 mM NaCl, 2 mM MgCl2, 1 mM EGTA, and 10 mM HEPES, with pH adjusted to 7.4 with HCl. To this solution Mg-ATP (3 mM), cAMP (0.3 mM), and Tris-GTP (0.5 mM) were added just before the experiments. Capsaicin (Calbiochem-Novabiochem, San Diego, CA), the TRPA1 agonist allyl isothiocyanate (Sigma-Aldrich), and the TRPA1 antagonist HC-030031 (Hydra Biosciences, Inc.) were dissolved in DMSO (100 mM). Drugs were used at less than 0.01% of their stock concentration. At these dilutions, DMSO alone did not elicit membrane currents or change firing. Stock solutions (10–100 mM) were stored at −20°C and diluted in the external recording solution just before experiments. The TRPV1 antagonist diarylpiperazine, a gift from Neurogen (Branford, CT), was used in these experiments instead of capsazepine because it is a potent and selective TRPV1 inhibitor (Ki = 6 nM for inhibition of acid pH-evoked responses and 35 nM for inhibition of CAPS-evoked responses; Sculptoreanu et al., 2008). However, it could not be used in the imaging experiments because it interfered with the Ca2+ measurements. Extracellularly applied drugs were pipetted from stock solutions at 50 times the final concentration and rapidly mixed in the recording chamber as described previously (Sculptoreanu et al., 2008). Effects of each drug concentration were measured for periods up to 5 to 8 min before changing the drug concentration or adding a new drug.
Firing and Na+ Currents.
Action potentials were generated by rectangular current pulse injections 5 ms long and 50 to 500 pA in intensity, followed by a 100-ms interpulse at the holding potential and a second pulse, 600 ms long. In general, the sequence consisted of at least two control recordings of evoked APs followed by pharmacological studies in which OA-NO2 evoked currents or Na+-currents before and after application of OA-NO2 were measured in voltage-clamp followed by a second recording of APs. In some experiments, time course of the effect of OA-NO2 was monitored by repeating the above-mentioned sequence at stimulus intensity just above that required to evoke an action potential. The activation curves of Na+ current were determined by a rectangular pulse 200 ms in duration and changed from −120 to 40 mV in steps of 6 mV. Peak currents were plotted against the test pulse potential.
Agonist-Activated Inward Currents.
Whole-cell currents evoked by CAPS, AITC, or OA-NO2 alone were normalized to membrane capacitance in each cell, averaged, and fitted with an equation consisting of one exponential for the rising phase and two exponentials for the decaying phase of the form y = f1 + f2, where f1 is the rising phase [f1 = a × (1 − exp(−(x − l1)/τRISE)) + c1], and f2 is the decaying phase of the response [f2 = b × exp(−(x − l2)/τDecay-fast) + d × exp(−(x − l3)/τDecay-slow)], where x is time; l1 − l3 are lag times; a, b, and d are best-fit amplitudes of currents normalized to the peak; and τRISE, τDecay-fast, and τDecay-slow are rising phase time constant and fast- and slow-decaying phase time constants, respectively. The lag time (time between application of OA-NO2 and onset of the response) was also determined empirically.
The effects of either the TRPV1 (diarylpiperazine) or the TRPA1 (HC-030031) antagonist on OA-NO2 current were evaluated by applying the antagonists within 100 to 200 s after application of OA-NO2 during the declining phase of the OA-NO2 current. Cells were considered responsive to the antagonists if the decay of the OA-NO2-evoked currents was significantly enhanced within 2 to 5 s after antagonist application. The magnitude of the antagonist effect was obtained by comparing the time course of the current after an antagonist to a predicted time course based on the average decaying phase of control OA-NO2 currents (n = 5 cells). The average control curve was constructed by normalizing control responses in each cell to the peak amplitude of the current and then fitting with the sum of three exponentials as described above. The fractional block induced by the antagonist in each cell was then obtained by measuring the maximal difference between the current after the antagonist and the averaged control current. To graph the average responses from multiple cells, an average time of antagonist application (i.e., time after application of OA-NO2, which varied between 100 and 200 s) was calculated and then the curves from individual experiments were scaled to match the average fraction of current remaining (referenced to the peak current) at the time of antagonist application. The individual curves were then averaged and the resulting data for the decay of currents in the presence of antagonists were fitted with a single exponential.
Statistics.
Results are reported as mean ± S.E.M. Statistical testing was carried out using a stepwise procedure depending upon the number of groups being compared. When only two means were involved in a comparison, a two-tailed t test with unequal variances was used. A comparison was considered statistically significant if p < 0.05. When more than two means were involved, a one-way analysis of variance was first carried out to obtain a global test of the null hypothesis. If the global p value for the test of the null hypothesis was <0.05, we performed post hoc comparisons between the different groups using the Holm-Sidak test (Kuzma and Bohnenblust, 2005).
Results
Comparison of Intracellular Ca2+ Transients Induced by OA-NO2, CAPS, and AITC.
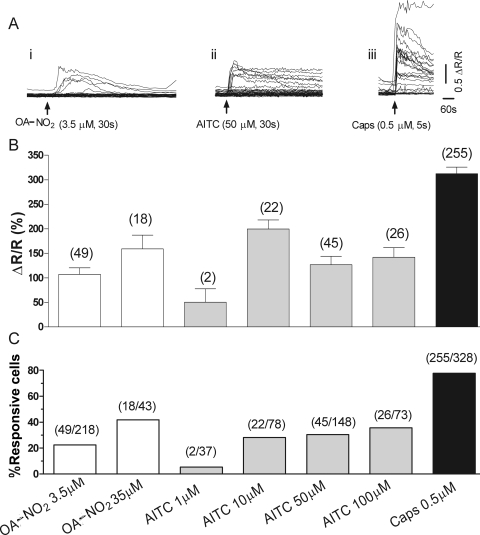
Application of OA-NO2 (3.5 or 35 μM; 30 s) produced transient increases in intracellular Ca2+ concentration in 20 to 40% of neurons tested (Fig. 1, Ai, B, and C). Responses were concentration dependent both in terms of amplitude (Fig. 1B, white bars) and percentage of responding cells (Fig. 1C, white bars). A second application of OA-NO2 of the same concentration as the first (3.5 μM) produced a smaller amplitude response, indicating that partial desensitization occurred. However, the decrease in amplitude was not statistically significant (first application, 112.3 ± 29.5 ΔR/R; second application, 79.5 ± 13.7 ΔR/R; n = 19 cells; p > 0.05). Although Ca2+ transients were detected at lower concentrations of OA-NO2, higher concentrations (3.5–35 μM) were tested in Ca2+ imaging experiments to increase the number of cells giving a detectable signal.
Fig. 1.
Ca2+ transients induced by OA-NO2, AITC, and CAPS in DRG neurons. A, example of [Ca2+]i changes in response to OA-NO2 (3.5 μM; 30-s duration (i), AITC (50 μM; 30-s duration) (ii), and CAPS (0.5 μM; 5-s duration) (iii). Each line represents a response from one DRG neuron. B, summary of the amplitude of responses to different concentrations of OA-NO2 (white bars), AITC (gray bars), and to CAPS (black bar). Numbers in parentheses represent the number of responsive cells. C, summary of the percentage of cells responding to different concentrations of OA-NO2 (white bars), AITC (gray bars), and to capsaicin (black bar). Concentrations are indicated at the bottom of the graph in C also apply to B. Numbers in parentheses represents the number of cells responding and total number of cells tested.
AITC (1–100 μM; 30-s application), a TRPA1 agonist, also produced transient increases in intracellular Ca2+ concentration in 5 to 35% of neurons tested (Fig. 1, Aii, B, and C). At concentrations of AITC ≥10 μM, the amplitude of the responses (Fig. 1B, gray bars) and the percentage of responding cells (Fig. 1C, gray bars) were similar to those induced by OA-NO2. A second application (10 μM) produced a response that was smaller but not significantly different in amplitude (first application, 129.2 ± 21.6 ΔR/R; second application, 100.9 ± 17.5 ΔR/R; n = 26 cells; p > 0.05). Short applications of CAPS (5–10 s; 0.5 μM) increased intracellular Ca2+ concentration in 77.7% DRG neurons (Fig. 1, Aiii, B, and C, black bars). As shown previously (Sculptoreanu et al., 2008), responses to CAPS desensitized with repetitive applications (decreased to 82 ± 14% of control after fourth application; p < 0.05). Among the three agents, CAPS produced the largest Ca2+ transients and in the highest percentage of cells. All responses were detected in small- to medium-diameter cells (diameters of 10.3–26.7 μm; average, 18.9 ± 0.7 μm; n = 417 cells). The average diameters of cells responding to OA-NO2, AITC, or CAPS were not significantly different.
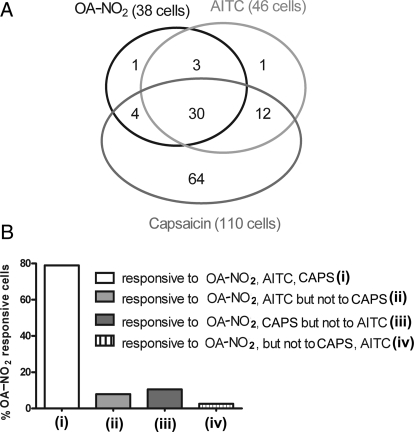
To determine whether neurons could respond to multiple agonists, the three agents were tested in the same neurons in the following order: OA-NO2 (3.5 μM), AITC (10 μM), and CAPS (0.5 μM). The majority of the OA-NO2-responsive neurons (79%; 30/38 cells) were both CAPS- and AITC-responsive (Fig. 2A). However, small percentages of OA-NO2 cells responded to CAPS and AITC as follows: to AITC but not to CAPS (7.9%); to CAPS but not to AITC (10.5%); or did not respond to either AITC or CAPS (2.6%; Fig. 2B).
Fig. 2.
DRG neurons responsive to OA-NO2, AITC, and CAPS. A, Venn diagrams illustrating the overlap of cells exhibiting Ca2+ transients in response to OA-NO2, AITC, and CAPS. B, percentage of OA-NO2-responsive cells that respond to both AITC and CAPS (i), that respond to AITC but not to CAPS (ii), that respond to CAPS but not to AITC (iii), and that do not respond to either CAPS or AITC (iv).
To determine whether the OA-NO2 responses were due to activation of TRP channels, the effects of TRPA1 (HC-030031) and TRPV1 (capsazepine) antagonists were examined. HC-030031 (5, 20, and 50 μM) completely blocked OA-NO2 responses (n = 61 cells), as well as the AITC responses (n = 34 cells) but did not block CAPS responses (n = 15 cells) (Fig. 3A). The responses to AITC recovered approximately 15 min after washing out the antagonist (n = 20 cells). Capsazepine (10 μM) greatly reduced the responses to OA-NO2 (64.6 ± 11.9% reduction from 154.7 ± 24.8 to 45.9 ± 13.9; n = 19 cells; paired t test, p < 0.05) (Fig. 3B) and completely blocked CAPS responses (n = 19 cells) but had no effect on AITC-evoked Ca2+ transients. In contrast, DTT (10 mM) abolished OA-NO2 responses (n = 10 cells) but did not block AITC- (n = 7 cells) or CAPS-evoked (n = 9 cells) responses (data not shown).
Fig. 3.
Inhibition of agonist induced Ca2+ transients in DRG neurons by a TRPA1 (HC-030031) and a TRPV1 (capsazepine) antagonist. A, examples from several cells showing responses to OA-NO2 (3.5 μM; 30-s duration) and AITC (10 μM; 30-s duration) in the absence and in the presence of the TRPA1 antagonist HC-030031 (50 μM). HC-030031 completely blocked OA-NO2 and AITC responses but did not affect CAPS response. B, examples from several cells showing responses to OA-NO2 (3.5 μM; 30-s duration) in the absence and in the presence of the TRPV1 antagonist capsazepine (10 μM). Capsazepine reduced OA-NO2 responses and completely blocked CAPS responses (0.5 μM; 5-s duration). Each line represents the response from one DRG neuron.
Patch-Clamp Recording: Currents Evoked by OA-NO2.
Patch-clamp experiments were conducted on 55 lumbosacral (L4-S3) small- to medium-sized DRG neurons ranging from 20 to 30 μm in diameter, with an average membrane capacitance of 45 ± 2 pF, a resting membrane potential of −53 ± 2 mV, and membrane resistance of 224 ± 15 MΩ. Cells were tested for their responsiveness to CAPS (Fig. 4Ai), AITC (Fig. 4Aii), and -OA-NO2 (Fig. 4Aiii). Thirty-three (62%) of these cells responded to OA-NO2 (Fig. 4Aiii) in a concentration-dependent manner (0.005–0.5 μM; Fig. 4B), with inward currents ranging in magnitude from 0.2 to 17 pA/pF. The number of cells responding to OA-NO2 increased with increasing OA-NO2 concentrations from 0.005 to 0.5 μM (43–62%). Concentrations of oleic acid as high as 5 mM failed to induce inward currents in DRG neurons (n = 8), which responded to OA-NO2.
Fig. 4.
Patch-clamp recordings of agonist-induced inward currents in DRG neurons. A, comparison of currents induced by CAPS (0.5 μM) (i), AITC (0.5 μM) (ii), and OA-NO2 (0.5 μM) (iii) in different cells. In another set of experiments agonists were applied in sequence in the same cell: OA-NO2 (0.05 μM) followed by AITC (0.5 μM (iv) and OA-NO2 (0.05 μM) followed by CAPS (0.5 μM) and AITC (0.5 μM) (v). B, average peak current densities (pA/pF) evoked by three concentrations of OA-NO2 alone or in a combination of OA-NO2 (0.05 μM). Absolute responses after sequential administration of either CAPS (0.5 μM; CAPS post-OA-NO2) or AITC (0.5 μM; AITC post-OA-NO2) were obtained by subtracting the steady state OA-NO2 (0.05 μM) current from the total peak current. For comparison, currents in response to either CAPS (0.5 μM; n = 46) or AITC (0.5 μM; n = 5) were obtained in separate experiments. In both sets of experiments, prior application of OA-NO2 significantly reduced the response to either CAPS (p < 0.001) or AITC (p < 0.05). The responses of combined administration to CAPS and AITC (0.5 μM each) are also shown. Peak currents after addition of each drug were measured separately and presented as the sum total of all currents; C, number of neurons that responded to a combination of OA-NO2 and either CAPS (i) or AITC (ii). Number of neurons that responded to CAPS and AITC applied sequentially after OA-NO2 (iii). Unhatched areas indicate responses to a single agent and hatched areas indicate responses to both agents. Number of neurons that did not respond to any of the agonists tested in each group is also shown below the diagrams.
Cell Responses to OA-NO2 and TRP Channel Agonists.
Among the 55 cells studied, OA-NO2 (0.005–0.50 μM) was tested alone in 15 cells (Fig. 4Aiii), and the remainder was also tested for responses to the subsequent application of either AITC (0.5 μM; n = 8; Fig. 4Aiv), CAPS (0.5 μM; n = 18), or to CAPS (0.5 μM) followed by AITC (0.5 μM; n = 14; Fig. 4Av). The CAPS- and AITC-responsive cells had resting membrane potentials similar to those of the total population of cells studied and were of similar size and average membrane capacitance (58 ± 4 pF). Among this population of cells, 63% (14 of 22 cells) responded to 0.5 μM AITC (Fig. 4A, iv and v), and 65% (21 of 32 cells) responded to CAPS (Fig. 4Av and B), with inward currents 50 pA to 10 nA in amplitude. Compared with responses to CAPS alone or AITC alone, CAPS and AITC responses were smaller in amplitude after application of 0.05 μM OA-NO2 (Fig. 4B). Of 18 cells tested with a combination of OA-NO2 and CAPS (Fig. 4Ci), 50% responded to both agents, 27.8% did not respond to either agent and the remainder responded only to OA-NO2 (11.1%) or only to CAPS (11.1%). Likewise, when cells were tested with OA-NO2 and AITC in sequence (Fig. 4, Aiv and Cii), 50% (four of eight cells) responded to both agents, 37.5% (three of eight) did not respond to either agent, and 12.5% (one of eight) responded to AITC but not to OA-NO2. In the population of cells tested for all three agents (Fig. 4Av), seven cells responded to all three agents and four did not respond to any agent with an inward current. To establish the overlap of TRPV1 and TRPA1 expression, 14 neurons tested with CAPS and AITC in sequence (Fig. 4Ciii), seven (50%) responded to both CAPS and AITC, three (21%) responded to CAPS but not AITC, and four (29%) did not respond to either agonist. When normalized to cell membrane capacitance, the amplitude of the currents elicited by 0.5 μM OA-NO2 (−13 ± 3 pA/pF; n = 32) was similar to currents elicited by AITC (0.5 μM; −18.7 ± 3 pA/pF; n = 5; p > 0.1) and CAPS (0.5 μM; −15 ± 2 pA/pF; n = 46; p > 0.1), but smaller than currents elicited by a combination of AITC and CAPS (0.5 μM each; −30 ± 11 pA/pF; n = 4; p > 0.1).
Comparison of Currents Evoked by OA-NO2 and TRP Channel Agonists.
Data on the kinetics of the agonist evoked currents were obtained from cells where multiple drugs were applied continuously and cumulatively in the following order: OA-NO2, CAPS, and AITC and where CAPS or AITC were administered after partial desensitization of the current induced by the previously administered agents as shown in Fig. 4A, iv and v. The OA-NO2-evoked currents had activation rates that were concentration-dependent (faster at higher concentrations; Fig. 5, A, C, and E) and desensitized slowly (Fig. 5, A, F, and G) in the presence of the drug. Compared at the same agonist concentration (0.5 μM; Fig. 5), the parameters relating to the onset time (time to peak, lag time, and rising phase time constant; Fig. 5, C–E) were significantly slower for OA-NO2-evoked currents than for CAPS- (0.5 μM) or AITC (0.5 μM)-evoked currents (p < 0.001). The decay of the currents evoked by the three agonists (0.5 μM; Fig. 5) appeared to consist of two decaying phases, one fast and the other slow, which could be fitted by a sum of two exponential equations (see Materials and Methods). The decay time constants of the two phases were similar for the three agonists. However, the magnitude of the slow phase of the OA-NO2-evoked currents was much larger (>50% of the best-fit peak amplitude) than the slow phase (<50% of the best-fit peak amplitude) of the CAPS or AITC currents. Thus, the AITC- and CAPS-evoked currents that had a larger contribution from the more rapidly decaying component (Fig. 5, A and B) exhibited a more rapid desensitization.
Fig. 5.
Time course of CAPS-, AITC-, and OA-NO2-evoked currents. A, averaged currents (symbols) and fitted curves (continuous lines) for currents evoked by OA-NO2 at 0.05 μM (n = 21) or 0.5 μM (n = 13). B, averaged currents (symbols) and fitted curves (continuous lines) for currents evoked by of either CAPS (n = 12; 0.5 μM) or AITC (n = 8; 0.5 μM). C, time to peak for average curves in A and B. D, lag time (time from addition of drug to beginning of response). E to G, time constants for the rising phase and fast and slow time constants for the decaying phase of fitted average currents shown in A and B.
Effect of Selective TRPV1 and TRPA1 Antagonists on OA-NO2-Evoked Currents.
The effects of TRPV1 (diarylpiperazine; 5 μM) and TRPA1 (HC-030031; 5 μM) antagonists on OA-NO2-induced currents were examined by applying the antagonists in the presence of OA-NO2 after the peak of the OA-NO2 response (Figs. 6 and 7). Cells were considered responsive to antagonists if the decay of the OA-NO2-evoked currents was significantly enhanced within 2 to 5 s after antagonist application (Fig. 6D, inset). For simplicity, the decay time constants immediately before the application of the antagonists and during the effect of antagonists were each fitted with a single exponential curve (Fig. 7). Application of either antagonist after the OA-NO2 currents had reached a peak and were declining (Fig. 6) increased the decay time constants by more than 4-fold. The rate of decay in the presence of antagonist eventually reached a steady state that paralleled the rate of decay of OA-NO2 currents in the absence of the antagonists (Figs. 6D, inset, and 7). At this point, we assumed that the effect of the antagonist had reached a plateau and therefore the magnitude of the block had reached a maximum. The magnitude of the antagonist effect was obtained by comparing the time course of the current after an antagonist to a predicted time course based on the average decay phase of control OA-NO2 currents (n = 5 cells). The average control curve was constructed by normalizing control responses in each cell to the peak amplitude of the current and then fitting with the sum of three exponentials as described under Materials and Methods. The fractional block induced by the antagonist in each cell was then obtained by measuring the maximal difference between the current after the antagonist and the averaged control current. The inset in Fig. 6D shows examples from two cells in which diarylpiperazine was applied at different times in the presence of OA-NO2. The rate of decay in the presence of antagonist was independent of the time it was applied after OA-NO2 (Fig. 6D, inset). We assume that when in the presence of antagonist the desensitization rates approach those seen before addition of antagonist, the blocking effect of the antagonist has reached a steady state and therefore the block would be less than 100%. Because HC-030031 (McNamara et al., 2007) and diarylpiperazine (Sculptoreanu et al., 2005; Srinivasan et al., 2008) are potent and selective antagonists of TRPA1 and TRPV1 channels, we would expect that at the relatively high concentrations used in our experiments (5 μM), one of the antagonists would rapidly and fully block the OA-NO2-evoked currents if OA-NO2 only activated one type of TRP channel. The selectivity of diarylpiperazine for TRPV1 was shown in two neurons responding to AITC in which the TRPV1 antagonist did not affect the decay of the AITC inward current but HC-3030031 fully blocked the current (data not shown). In three additional neurons that did not respond to CAPS but did respond to AITC and OA-NO2, the TRPV1 antagonist did not affect the decay of the inward currents, but HC-3030031 fully blocked the currents (data not shown). The effects on OA-NO2 currents of antagonists applied at a concentration (5 μM) that completely blocked the currents evoked by either CAPS or AITC varied in different cells, ranging from 28 to 100% for diarylpiperazine and from 21 to 100% for HC-030031. In some cells diarylpiperazine (n = 5 of 9 cells) or HC-030031 (n = 4 of 8 cells; Fig. 6A) alone fully blocked the currents evoked by OA-NO2 (Fig. 6A). However, in other cells diarylpiperazine produced only a partial block (n = 2) or no block (n = 2). A similar result was obtained with HC-030031, which produced a partial block in two cells and no effect in two additional cells.
Fig. 6.
Block of OA-NO2-evoked currents by TRP antagonists. A, inward currents induced by increasing concentrations of OA-NO2. Block of the OA-NO2-evoked current by a TRPA1 antagonist (HC-030031; 5 μM). B, block of an OA-NO2-evoked current by the reducing agent dithiothreitol (10 mM). C, partial block of an OA-NO2-evoked current by the TRPV1 antagonist diarylpiperazine (5 μM), and the remaining current was blocked by subsequent addition of HC-030031 (5 μM). D, average block of an OA-NO2-evoked current by each antagonist alone or the combination of the two antagonists or by dithiothreitol. Inset shows average time course for prolonged application of OA-NO2 (0.05 μM) in five neurons (empty circles) that was fitted with an equation consisting of one exponential for the rising phase and two exponentials for the decaying phase as explained under Materials and Methods. Inset shows the effect of diarylpiperazine (5 μM) in two neurons. Currents were normalized to the peak inward current in all cells. Statistical comparisons in D (**, p < 0.01) are for average percentage of steady-state block after drugs versus decay of current at the same time point in absence of blockers.
Fig. 7.
Average inhibition of OA-NO2 currents by two TRP antagonists. A, effect of diarylpiperazine in six neurons. Average curve of antagonist effect (circles) and its best fit (dotted line) is compared with the best fit (dashed line) of normalized currents averaged in five cells. Time constants immediately before and after antagonist application are also shown. Inset shows a current trace in one neuron in which diarylpiperazine (5 μM) was applied approximately 170 s after OA-NO2; the exponential fits of the current decay before and after antagonist are linearly extrapolated to show the change in the rate of current decline. B, effect of HC-030031 in five neurons. Average curve of antagonist effect (circles) and its best fit (dotted line) is compared with the best fit (dashed continuous line) of normalized currents averaged in five cells. Time constants immediately before and after antagonist application are also shown. Inset shows a current trace in one neuron in which HC-030031 (5 μM) was applied approximately 120 s after OA-NO2, the exponential fits of the current decay before and after antagonist are linearly extrapolated to show the change in the rate of current decline. Statistical difference between control curve and the average curve for the effect of the antagonist was determined by nonlinear regression to be p < 0.001 for the effect of diarylpiperazine in A and p < 0.0001 for the effect of HC-030031 in B.
To graph the average responses from multiple cells, an average time of antagonist application (i.e., time after application of OA-NO2, which varied between 100 and 200 s) was calculated, and then the curves from individual experiments were scaled to match the average fraction of current remaining (referenced to the peak current) at the time of antagonist application. The individual curves were then averaged and the resulting data for the decay of currents in the presence of antagonists were fitted with a single exponential. Curves were averaged (Fig. 7, filled circles), and the resulting data were fitted with a single exponential (Fig. 7, dashed lines). On average, diarylpiperazine produced a slightly larger block (56 ± 8%; p < 0.05) than HC-030031 (48 ± 7%; Figs. 6D and 7). In 10 cells, combined administration of the two antagonists applied sequentially (Fig. 6, C and D) produced a greater block (87 ± 9%; p < 0.01) than the block induced by either antagonist alone.
In cells treated with both antagonists (n = 4) or in cells in which application of a TRPV1 (n = 2) or a TRPA1 (n = 3) antagonist only partially blocked the currents, application of the reducing agent DTT (10 mM; n = 13) inhibited fully the remaining currents evoked by OA-NO2 after partial block with the antagonists (Fig. 6, B and D). The average block of OA-NO2-evoked currents by DTT applied alone was 95 ± 2% (n = 9). The p values in Fig. 6D were determined using one sample test of proportions (Kuzma and Bohnenblust, 2005).
Effect of OA-NO2 and Antagonists on Membrane Potential and Firing.
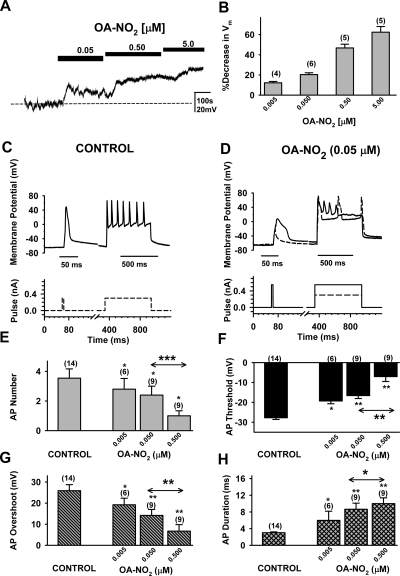
OA-NO2 reduced the resting potential by 5 to 50% in a concentration-dependent manner (Fig. 8, A and B). The depolarization induced by OA-NO2 (Fig. 8A) when applied cumulatively at increasing concentrations did not evoke firing (Fig. 8A). Depolarization in response to 0.5 μM OA-NO2 was partially reversed (20–70%) by application of HC-030031 (5 μM; n = 3) or diarylpiperazine (n = 2).
Fig. 8.
Effect of OA-NO2 on membrane potential (A and B) and firing (C–H) in DRG neurons. A, membrane potential change in response to three concentrations of OA-NO2. B, concentration-dependent reduction in membrane potential in response to increasing concentrations of OA-NO2. C and D, firing triggered by short-duration (10 ms) or long-duration (600 ms) current pulses in control (C) and after addition of OA-NO2 (0.05 μM; D). Current intensity of 0.3 nA (dashed lines in C) was sufficient to trigger an AP in response to short-duration pulses and seven APs during a long-duration pulse. After exposure to OA-NO2, the short-duration 0.4 nA stimulus failed to generate an AP, and the long-duration stimulus produced fewer APs (dashed lines in D). Higher stimulus intensity (continuous line in D) triggers an AP with a short-duration pulse, and a long-duration pulse elicited fewer APs than in control recordings (D). E, effect of OA-NO2 on the maximal number of APs generated by long-duration stimuli of submaximal intensities. F, threshold of AP generated by single short pulses (10 ms). G, overshoot of APs generated by single short pulses (10 ms). H, duration of AP at 50% repolarization generated by single short pulses (10 ms). Data from cells shown in E to H are from 14 OA-NO2-responsive cells that also responded to CAPS and/or AITC with an inward current. Statistical comparisons in E to H (*, p < 0.05; **, p < 0.01; ***, p < 0.001) (n) are between control values before application of OA-NO2 and after OA-NO2; values above the arrow line are for comparisons of effects at 0.05 and 0.50 μM OA-NO2.
When the membrane potential was held at −57 mV, application of rectangular depolarizing current pulses 50 to 500 pA in amplitude and 600 ms in duration elicited one to eight action potentials. The number of APs in cells responsive to CAPS (2.6 ± 0.9 APs/600 ms; n = 11), AITC (3.1 ± 1 APs/600 ms; n = 8), or OA-NO2 (3.2 ± 1 APs/600 ms; n = 21) (Table 1) was similar to the AP number (4.1 ± 1 APs/600 ms; n = 9) (Table 1) in cells that did not respond to these agents (Table 1). The voltage threshold of APs and AP overshoot were also indistinguishable in OA-NO2-responsive and OA-NO2-unresponsive cells (Table 1).
TABLE 1.
Electrophysiological characteristics of DRG neurons that responded or did not respond to OA-NO2
Number of action potentials evoked by a 600-ms depolarizing current pulse (APNmax), voltage threshold (TSH) for initiation of action potential, and action potential overshoot (OS) in cells that responded or did not respond (UNRESP) to OA-NO2. All measurements were made in untreated cells. Data are averages ± S.E.M.; t test relative to values in cells that responded to OA-NO2, two-tailed, unequal variance. p value was below statistical significance in all groups tested.
| Response | n | APNmax | p | TSH | p | OS | p |
|---|---|---|---|---|---|---|---|
| mV | mV | ||||||
| OA-NO2 | 21 | 3.24 ± 0.64 | −27.86 ± 0.97 | 33.57 ± 3.14 | |||
| UNRESP | 9 | 4.11 ± 1.10 | 0.50 | −28.00 ± 1.49 | 0.94 | 32.67 ± 4.01 | 0.86 |
In 14 cells that responded to various OA-NO2 concentrations (0.005–0.5 μM) with an inward current or membrane depolarization, and after adjusting the holding voltage to −57 mV to compensate for the OA-NO2-evoked depolarizations, there were substantial changes in AP generation, notably an increase in the voltage threshold for induction of firing. There was also an increase in the intensity of depolarizing current injection required to generate APs, a decrease in overshoot of the AP, and an increase in the duration of APs (Fig. 8). There was also a decrease in the number of APs triggered by long-duration current injections (600 ms; Fig. 8E). These changes in firing parameters were not reversed by application of HC-030031 (5 μM; n = 5) or diarylpiperazine (n = 7), nor were they prevented by pretreatment with the antagonists (HC-030031: 5 μM, n = 3 and diarylpiperazine: 5 μM, n = 4). Similar inhibitory effects on firing (n = 6; p < 0.05), AP threshold (p < 0.01), AP overshoot (p < 0.001), and an increase in duration (p < 0.01) occurred at higher OA-NO2 concentrations (≥0.05 μM) in CAPS/AITC-unresponsive cells, suggesting that the inhibition was not restricted to putative nociceptive neurons. In cells that did not respond to OA-NO2 with an inward current or membrane depolarization, application of OA-NO2 resulted in measurable changes in firing and AP parameters in one of six cells at 0.005 μM; 8 of 12 cells at 0.05 μM, and 10 of 12 cells at 0.5 μM. In contrast, in cells that did respond to OA-NO2 with a membrane depolarization or an inward current, application of OA-NO2 resulted in measurable changes in firing and AP parameters in four of six cells at 0.005 μM, six of nine cells at 0.05 μM, and nine of nine cells at 0.5 μM (Fig. 8).
OA-NO2 Signaling Actions Are Not Nitric Oxide-Dependent.
As reported previously (Taylor-Clark et al., 2009), the effect of NO donors in DRG neurons is small compared with that seen in response to OA-NO2. In addition, in their experiments pretreatment with the NO scavenger cPTIO did not prevent the OA-NO2-evoked Ca2+ transients. To determine whether the effects of OA-NO2 were due to release of NO, experiments were preformed in the presence of the NO scavenger cPTIO (Fig. 9A). In calcium-imaging experiments, pretreatment of cells with cPTIO (100 μM; 5 min) in six neurons that responded to OA-NO2 alone did not prevent OA-NO2-induced increase in [Ca2+]i (Fig. 9A). Likewise, cPTIO did not prevent a further increase in the inward currents evoked by OA-NO2 when the concentration was changed from 0.05 to 0.5 μM (Fig. 9B) or when 0.05 μM OA-NO2 was applied in the presence of the NO scavenger cPTIO (Fig. 9C).
Fig. 9.
NO scavenger cPTIO did not prevent OA-NO2 effects. A, Ca2+ transients evoked by 30 μM OA-NO2 averaged in six neurons in the absence (left traces) and presence (right traces) of 100 μM cPTIO. OA-NO2 (30 μM) was applied alone for 30 s, and the [Ca2+]i was allowed to recover to baseline. cPTIO was applied for 5 min before and during a subsequent 30-s application of OANO2. B, in an AITC/HC-030031-unresponsive cell, OA-NO2 evoked an inward current that was unaffected by subsequent application of PTIO. Application of 10× more OA-NO2 in the presence of OA-NO2 further increased the OA-NO2-evoked current and was nearly completely blocked by diarylpiperazine. C, in the presence of cPTIO, OA-NO2 evoked an inward current that was further increase by submaximal concentrations of AITC and CAPS. In this neuron, the total slowly desensitizing current in the presence of antagonists was blocked by a combined application of diarylpiperazine and HC-030031. Bars above currents traces in B and C represent the time of drug application. Current-voltage curves of Na+ currents (D and F) and representative current traces (at −10 mV, holding potential, −80 mV; E and G) of OA-NO2 effects applied at various concentrations as shown in the absence (D and E) and presence (F and G) of cPTIO. cPTIO (0.5 mM) was applied 3 to 5 min before addition of OA-NO2 (n = 6).
The inhibition of firing was probably due to inhibition of Na+ channels because OA-NO2 inhibited parameters of firing normally associated with Na+ channels: the threshold of firing, overshoot of AP, and maximal rate of rise (dV/dtmax), which occurred independently of TRP channel activation. OA-NO2 inhibited Na+ currents in a concentration-dependent manner (Fig. 9, D and E), and this effect was also not prevented by cPTIO (Fig. 9, F and G).
Discussion
This study addressed the changes in ion channel function induced by an unsaturated fatty acid nitration product that is formed endogenously by NO and nitrite-dependent oxidative inflammatory reactions. Application of low concentrations of OA-NO2 (0.005–0.50 μM) to DRG neurons activates TRPV1 and TRPA1 channels to elicit membrane depolarizations and inward currents that are similar in kinetics to CAPS- and AITC-evoked currents. Higher concentrations of OA-NO2 (>0.05 μM) also inhibit firing, prolong the AP, reduce AP overshoot, and raise the firing threshold in a majority of neurons regardless of their responsiveness to CAPS and or AITC. The latter effects are consistent with inhibition of both voltage-dependent Na+ and K+ channels. In most cells, OA-NO2-activated currents were blocked by a combination of TRPV1 and TRPA1 antagonists, but the effects of OA-NO2 on firing were not reversed by these antagonists. OA-NO2 also induced Ca2+ transients in DRG neurons that responded to CAPS and AITC. It is concluded that OA-NO2 activates TRPV1 and TRPA1 channels in DRG neurons but may also target unidentified channels.
Although both patch-clamp and Ca2+ imaging experiments revealed that OA-NO2 activated TRP channels, some apparent discrepancies in concentration-response relationships were noted. These differences are not substantive, however, because we chose higher concentrations in Ca2+ imaging experiments to increase the likelihood of a detectable response in a large population of neurons. Although confirming Ca2+ imaging-based findings, patch-clamp measurements were more sensitive for detecting responses to OA-NO2 and AITC.
The major component of the OA-NO2-induced current or Ca2+ transient is attributed to activation of TRPV1 and TRPA1 channels, because in most experiments the inward currents generated by OA-NO2 were markedly reduced by TRPV1- (53 ± 6%) or TRPA1 (56 ± 10%)-selective antagonists (diarylpiperazine and HC-030031, respectively). The minor component (<10–30%) of OA-NO2-activated current that was insensitive to a combination of TRPA1 and TRPV1 antagonists was blocked by DTT, indicating that this response was also dependent on the electrophilic properties of OA-NO2.
The differences between our study and another study (Taylor-Clark et al., 2009) of the effects of OA-NO2 may be attributed to species differences (rats in this study versus mouse) or differences in neuronal populations (DRG versus trigeminal and nodose ganglion neurons). We view that in rat DRG neurons, OA-NO2 activates multiple ion channels (TRPA1, TRPV1, and unidentified channels) and not just TRPA1 channels. This conclusion is based on several observations. First, OA-NO2 activated inward currents in neurons that were unresponsive to the TRPA1 agonist AITC, indicating that channels other than TRPA1 must be responsive to OA-NO2. Second, in these AITC-unresponsive neurons, the OA-NO2-induced inward current was markedly reduced by a TRPV1 antagonist (Fig. 9A). Third, in neurons that were responsive to OA-NO2, CAPS, and AITC, the sequential administration of a TRPV1 and TRPA1 antagonist significantly reduced in a stepwise manner the OA-NO2 current. Fourth, OA-NO2 induced currents in neurons unresponsive to both AITC and capsaicin, indicating that channels other than TRPA1 and TRPV1 are responsive to OA-NO2. Fifth, the TRP channel antagonists were selective because the TRPA1 antagonist did not suppress the response to capsaicin and the TRPV1 antagonists did not suppress the responses to AITC. The selectivity of HC-030031 for TRPA1 versus other TRP channels was also reported in a recent study by other investigators (McNamara et al., 2007). Sixth, the suppression of electrically evoked action potentials was not prevented or reversed by TRPA1 or TRPV1 antagonists, indicating that mechanisms other than TRPA1/TRPV1 activation contribute to the neural inhibitory effects of OA-NO2.
Activation of other channels could also explain the differences in the kinetics of OA-NO2- and AITC/CAPS-activated currents. OA-NO2 currents exhibited a decaying phase dominated by a slow component, unlike the decay phase of CAPS- and AITC-evoked currents that exhibited a dominant rapid and a smaller slow component. In addition, there was a significant delay in the OA-NO2-evoked currents that was reflected in the onset of the response including lag time, the rise time, and time to peak (Fig. 5E). One possible explanation for this delay is that OA-NO2 binds at different sites in the channels than CAPS and AITC that are less accessible to extracellularly applied agonists, thereby requiring a longer time for activation.
Recently, it was reported that omega-3 polyunsaturated fatty acids open TRPV1 channels in a phosphorylation-dependent manner. Thus, pretreatment with a PKC activator (phorbol 12-myristate 13-acetate), which phosphorylates the channel, was necessary to reveal the agonist action of omega-3 fatty acids (Matta et al., 2007). In DRG neurons, expression of a dominant-negative PKCε speeds up desensitization and reduces the amplitude of CAPS-responses, indicating that there is basal PKC-mediated phosphorylation of TRPV1 channels (Sculptoreanu et al., 2008; Srinivasan et al., 2008). Thus, the effect of OA-NO2 might be influenced by the phosphorylation state of the TRPV1 channel, with this possibly contributing to the variability in the response to OA-NO2 in different cells. Studies with oleic acid revealed that the nitroalkene functional group of OA-NO2 is required to activate TRP channels, because concentrations of oleic acid (5 mM) well in excess of OA-NO2 failed to induce inward currents in DRG neurons.
In strictly aqueous milieu, OA-NO2 can undergo a slow Nef-like decay reaction that releases low yields of NO and a carbonyl fatty acid adduct as products, species that might also contribute to the effects of the parent compound on DRG cells (Schopfer et al., 2005). However, the Nef reaction is inhibited when nitro-fatty acids partition into membranes of lipoproteins and cells (Baker et al., 2007; Rudolph et al., 2009), indicating that NO is not a mediator of OA-NO2 actions. Although NO activates TRPC channels by S-nitrosylation of cysteine, an action reversed by DTT, this requires very high S-nitrosothiol and NO concentrations (Yoshida et al., 2006). A modest action of NO donors on TRP channels has been reported by Taylor-Clark et al. (2009); however, these effects were minimal in comparison with those of OA-NO2. In addition, the use of an NO scavenger (carboxy-PTIO) in their experiments and in rat DRG neurons (this study) did not prevent OA-NO2 evoked currents or inhibition of firing and Na+ currents by OA-NO2, supporting the idea that breakdown of OA-NO2 and release of NO is not responsible for the effects of OA-NO2.
The effect of combined application of TRPV1 and TRPA1 antagonists on OA-NO2-evoked currents was comparable with that of the reducing agent DTT that competitively reacts with the electrophilic fatty acid (Salazar et al., 2008; Chen et al., 2009). Thus, the action of DTT on the OA-NO2-evoked currents is highly suggestive of an electrophilic interaction of OA-NO2 with cysteine residues of TRP channels, such as that reported for various pungent compounds and AITC (Hinman et al., 2006; Macpherson et al., 2007).
In CAPS- and AITC-responsive cells, low concentrations of OA-NO2 (0.005–0.50 μM) increased the firing thresholds, dramatically increased the duration of action potentials, and inhibited firing. The effects of OA-NO2 on firing parameters suggest that OA-NO2 may also inhibit voltage-dependent Na+ and K+ channels. Redox modulation of A-type K+ channel activity in DRG neurons has been observed previously (Hsieh, 2008). In addition, high concentrations of polyunsaturated fatty acids, probably containing thiol-reactive oxidized species, also suppress Na+ channels (Xiao et al., 1995; Vreugdenhil et al., 1996). However, to our knowledge, this is the first report of an effect of nanomolar concentrations of a nitro-fatty acid on firing in DRG neurons. These effects would thus be expected to induce antinociceptive and possible anti-inflammatory effects, due to an action on CAPS-sensitive afferent nerve terminals. Similar inhibitory effects also occurred at higher OA-NO2 concentrations (>0.5 μM) in CAPS/AITC-unresponsive cells, suggesting that the inhibition was not restricted to putative nociceptive neurons.
In summary, in DRG neurons OA-NO2 activates TRPV1 and TRPA1 channels and induces an inward current and membrane depolarization. If the OA-NO2 that is generated by cells during oxidative stress reacts with afferent terminals in sufficient concentrations, the initial activation of TRP channels would depolarize the membrane and stimulate release of peptides and other neurotransmitters that could propagate inflammatory responses. Supporting this view are recent findings suggesting that in the spinal cord, oxidative products of linoleic acid mediate inflammatory hyperalgesia by activating TRPV1 channels (Patwardhan et al., 2009). However, although the initial effect of OA-NO2 may be excitatory, prolonged exposure to OA-NO2 may desensitize the TRP channels and in turn suppress nociceptive and inflammatory responses, an event reflective of the delayed antinociceptive effects of CAPS (Dray, 1992; Wrigglesworth et al., 1996). The suppression of afferent nerve excitability by high concentrations of OA-NO2 could thus act in concert with the desensitization of TRP channels to suppress afferent firing and limit the duration and magnitude of inflammatory responses. Thus, OA-NO2 acts by multiple mechanisms to modulate inflammatory responses (Baker et al., 2007; Li et al., 2008; Wright et al., 2009).
Acknowledgments
We thank Dr. Guillermo Romero for helpful suggestions with data analysis and Dr. Richard Day for help with statistical analysis. BAF acknowledges financial interest in Complexa, Inc.
This work was supported by the National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases [Grant R37-DK049430] (to W.C.d.G); the National Institutes of Health National Heart, Lung, and Blood Institute [Grants R01-HL58115, R01-HL64937] (to B.A.F.); and the American Heart Association [Grant ADA-7-08-JF-52] (to F.J.S.).
Results in this article have been presented previously in abstract form: Sculptoreanu A, Schopfer F, Woodcock S, de Groat WC, Freeman B, Moran MM, and Del Camino D (2008) Nitro-oleic acid depolarizes the membrane and induces inward currents in TRPV1 and TRPA1 positive DRG neurons, in Proceedings of the 38th Annual Meeting of the Society for Neuroscience; 2008 Nov 15–19; Washington, DC. Society for Neuroscience, Washington, DC.
Article, publication date, and citation information can be found at http://jpet.aspetjournals.org.
doi:10.1124/jpet.109.163154.
- OA-NO2
- nitro-oleic acid
- TRP
- transient receptor potential
- DRG
- dorsal root ganglion
- DMEM
- Dulbecco's modified Eagle's medium
- HBSS
- Hanks' balanced salt solution
- AITC
- allyl isothiocyanate
- DMSO
- dimethyl sulfoxide
- HC-030031
- 2-(1,3-dimethyl-2,6-dioxo-1,2,3,6-tetrahydro-7H-purin-7-yl)-N-(4-isopropylphenyl) acetamide
- DTT
- dithiothreitol
- [Ca2+]i
- intracellular Ca2+ concentration
- cPTIO
- carboxy-2-phenyl-4,4,5,5-tetramethylimidazolineoxyl-1-oxyl-3-oxide
- R
- ratio
- CAPS
- capsaicin
- AP
- action potential
- PKC
- protein kinase C.
References
- Baker et al., 2007.Baker LM, Baker PR, Golin-Bisello F, Schopfer FJ, Fink M, Woodcock SR, Branchaud BP, Radi R, Freeman BA. (2007) Nitro-fatty acid reaction with glutathione and cysteine. Kinetic analysis of thiol alkylation by a Michael addition reaction. J Biol Chem 282:31085–31093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker et al., 2005.Baker PR, Lin Y, Schopfer FJ, Woodcock SR, Groeger AL, Batthyany C, Sweeney S, Long MH, Iles KE, Baker LM, et al. (2005) Fatty acid transduction of nitric oxide signaling: multiple nitrated unsaturated fatty acid derivatives exist in human blood and urine and serve as endogenous peroxisome proliferator-activated receptor ligands. J Biol Chem 280:42464–42475 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bandell et al., 2007.Bandell M, Macpherson LJ, Patapoutian A. (2007) From chills to chilis: mechanisms for thermosensation and chemesthesis via thermoTRPs. Curr Opin Neurobiol 17:490–497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen et al., 2009.Chen J, Crossland RF, Noorani MM, Marrelli SP. (2009) Inhibition of TRPC1/TRPC3 by PKG contributes to NO-mediated vasorelaxation. Am J Physiol Heart Circ Physiol 297:H417–H424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang et al., 2004.Dang K, Bielefeldt K, Gebhart GF. (2004) Gastric ulcers reduce A-type potassium currents in rat gastric sensory ganglion neurons. Am J Physiol Gastrointest Liver Physiol 286:G573–G579 [DOI] [PubMed] [Google Scholar]
- Dray, 1992.Dray A. (1992) Mechanism of action of capsaicin-like molecules on sensory neurons. Life Sci 51:1759–1765 [DOI] [PubMed] [Google Scholar]
- Ferreira et al., 2009.Ferreira AM, Ferrari MI, Trostchansky A, Batthyany C, Souza JM, Alvarez MN, López GV, Baker PR, Schopfer FJ, O'Donnell V, et al. (2009) Macrophage activation induces formation of the anti-inflammatory lipid cholesteryl-nitrolinoleate. Biochem J 417:223–234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinman et al., 2006.Hinman A, Chuang HH, Bautista DM, Julius D. (2006) TRP channel activation by reversible covalent modification. Proc Natl Acad Sci USA 103:19564–19568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh, 2008.Hsieh CP. (2008) Redox modulation of A-type K+ currents in pain-sensing dorsal root ganglion neurons. Biochem Biophys Res Commun 370:445–449 [DOI] [PubMed] [Google Scholar]
- Ichikawa et al., 2008.Ichikawa T, Zhang J, Chen K, Liu Y, Schopfer FJ, Baker PR, Freeman BA, Chen YE, Cui T. (2008) Nitroalkenes suppress lipopolysaccharide-induced signal transducer and activator of transcription signaling in macrophages: a critical role of mitogen-activated protein kinase phosphatase 1. Endocrinology 149:4086–4094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kansanen et al., 2009.Kansanen E, Jyrkkänen HK, Volger OL, Leinonen H, Kivelä AM, Häkkinen SK, Woodcock SR, Schopfer FJ, Horrevoets AJ, Ylä-Herttuala S, et al. (2009) Nrf2-dependent and -independent responses to nitro-fatty acids in human endothelial cells: identification of heat shock response as the major pathway activated by nitro-oleic acid. J Biol Chem 284:33233–33241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kullmann et al., 2008.Kullmann FA, Artim D, Beckel J, Barrick S, de Groat WC, Birder LA. (2008) Heterogeneity of muscarinic receptor-mediated Ca2+ responses in cultured urothelial cells from rat. Am J Physiol Renal Physiol 294:F971–F981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuzma and Bohnenblust, 2005.Kuzma JW, Bohnenblust SE. (2005) Basic Statistics for the Health Sciences, McGraw-Hill, Boston, MA [Google Scholar]
- Li et al., 2008.Li Y, Zhang J, Schopfer FJ, Martynowski D, Garcia-Barrio MT, Kovach A, Suino-Powell K, Baker PR, Freeman BA, Chen YE, et al. (2008) Molecular recognition of nitrated fatty acids by PPAR gamma. Nat Struct Mol Biol 15:865–867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macpherson et al., 2007.Macpherson LJ, Dubin AE, Evans MJ, Marr F, Schultz PG, Cravatt BF, Patapoutian A. (2007) Noxious compounds activate TRPA1 ion channels through covalent modification of cysteines. Nature 445:541–545 [DOI] [PubMed] [Google Scholar]
- Maeda et al., 1994.Maeda H, Akaike T, Yoshida M, Suga M. (1994) Multiple functions of nitric oxide in pathophysiology and microbiology: analysis by a new nitric oxide scavenger. J Leukoc Biol 56:588–592 [DOI] [PubMed] [Google Scholar]
- Matta et al., 2007.Matta JA, Miyares RL, Ahern GP. (2007) TRPV1 is a novel target for omega-3 polyunsaturated fatty acids. J Physiol 578:397–411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McNamara et al., 2007.McNamara CR, Mandel-Brehm J, Bautista DM, Siemens J, Deranian KL, Zhao M, Hayward NJ, Chong JA, Julius D, Moran MM, et al. (2007) TRPA1 mediates formalin-induced pain. Proc Natl Acad Sci USA 104:13525–13530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montell et al., 2002.Montell C, Birnbaumer L, Flockerzi V, Bindels RJ, Bruford EA, Caterina MJ, Clapham DE, Harteneck C, Heller S, Julius D, et al. (2002) A unified nomenclature for the superfamily of TRP cation channels. Mol Cell 9:229–231 [DOI] [PubMed] [Google Scholar]
- Nadtochiy et al., 2009.Nadtochiy SM, Baker PR, Freeman BA, Brookes PS. (2009) Mitochondrial nitroalkene formation and mild uncoupling in ischaemic preconditioning: implications for cardioprotection. Cardiovasc Res 82:333–340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patwardhan et al., 2009.Patwardhan AM, Scotland PE, Akopian AN, Hargreaves KM. (2009) Activation of TRPV1 in the spinal cord by oxidized linoleic acid metabolites contributes to inflammatory hyperalgesia. Proc Natl Acad Sci USA 106:18820–18824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudolph et al., 2010.Rudolph V, Rudolph TK, Schopfer FJ, Bonacci G, Woodcock SR, Cole MP, Baker PR, Ramani R, Freeman BA. (2010) Endogenous generation and protective effects of nitro-fatty acids in a murine model of focal cardiac ischaemia and reperfusion. Cardiovasc Res 85:155–166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudolph et al., 2009.Rudolph V, Schopfer FJ, Khoo NK, Rudolph TK, Cole MP, Woodcock SR, Bonacci G, Groeger AL, Golin-Bisello F, Chen CS, et al. (2009) Nitro-fatty acid metabolome: saturation, desaturation, beta-oxidation, and protein adduction. J Biol Chem 284:1461–1473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salazar et al., 2008.Salazar H, Llorente I, Jara-Oseguera A, García-Villegas R, Munari M, Gordon SE, Islas LD, Rosenbaum T. (2008) A single N-terminal cysteine in TRPV1 determines activation by pungent compounds from onion and garlic. Nat Neurosci 11:255–261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schopfer et al., 2005.Schopfer FJ, Baker PR, Giles G, Chumley P, Batthyany C, Crawford J, Patel RP, Hogg N, Branchaud BP, Lancaster JR, Jr, et al. (2005) Fatty acid transduction of nitric oxide signaling. Nitrolinoleic acid is a hydrophobically stabilized nitric oxide donor. J Biol Chem 280:19289–19297 [DOI] [PubMed] [Google Scholar]
- Sculptoreanu et al., 2008.Sculptoreanu A, Kullmann AF, de Groat WC. (2008) Neurokinin 2 receptor-mediated activation of protein kinase C modulates capsaicin responses in DRG neurons from adult rats. Eur J Neurosci 27:3171–3181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sculptoreanu and de Groat, 2007.Sculptoreanu A, de Groat WC. (2007) Neurokinins enhance excitability in capsaicin-responsive DRG neurons. Exp Neurol 205:92–100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sculptoreanu et al., 2005.Sculptoreanu A, de Groat WC, Buffington CA, Birder LA. (2005) Protein kinase C contributes to abnormal capsaicin responses in DRG neurons from cats with feline interstitial cystitis. Neurosci Lett 381:42–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Srinivasan et al., 2008.Srinivasan R, Wolfe D, Goss J, Watkins S, de Groat WC, Sculptoreanu A, Glorioso JC. (2008) Protein kinase C epsilon contributes to basal and sensitizing responses of TRPV1 to capsaicin in rat dorsal root ganglion neurons. Eur J Neurosci 28:1241–1254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor-Clark et al., 2009.Taylor-Clark TE, Ghatta S, Bettner W, Undem BJ. (2009) Nitrooleic acid, an endogenous product of nitrative stress, activates nociceptive sensory nerves via the direct activation of TRPA1. Mol Pharmacol 75:820–829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vreugdenhil et al., 1996.Vreugdenhil M, Bruehl C, Voskuyl RA, Kang JX, Leaf A, Wadman WJ. (1996) Polyunsaturated fatty acids modulate sodium and calcium currents in CA1 neurons. Proc Natl Acad Sci USA 93:12559–12563 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wrigglesworth et al., 1996.Wrigglesworth R, Walpole CS, Bevan S, Campbell EA, Dray A, Hughes GA, James I, Masdin KJ, Winter J. (1996) Analogues of capsaicin with agonist activity as novel analgesic agents: structure-activity studies. 4. Potent, orally active analgesics. J Med Chem 39:4942–4951 [DOI] [PubMed] [Google Scholar]
- Wright et al., 2009.Wright MM, Kim J, Hock TD, Leitinger N, Freeman BA, Agarwal A. (2009) Human haem oxygenase-1 induction by nitro-linoleic acid is mediated by cAMP, AP-1 and E-box response element interactions. Biochem J 422:353–361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao et al., 1995.Xiao YF, Kang JX, Morgan JP, Leaf A. (1995) Blocking effects of polyunsaturated fatty acids on Na+ channels of neonatal rat ventricular myocytes. Proc Natl Acad Sci USA 92:11000–11004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu et al., 2006.Xu GY, Winston JH, Shenoy M, Yin H, Pasricha PJ. (2006) Enhanced excitability and suppression of A-type K+ current of pancreas-specific afferent neurons in a rat model of chronic pancreatitis. Am J Physiol Gastrointest Liver Physiol 291:G424–G431 [DOI] [PubMed] [Google Scholar]
- Yoshida et al., 2006.Yoshida T, Inoue R, Morii T, Takahashi N, Yamamoto S, Hara Y, Tominaga M, Shimizu S, Sato Y, Mori Y. (2006) Nitric oxide activates TRP channels by cysteine S-nitrosylation. Nat Chem Biol 2:596–607 [DOI] [PubMed] [Google Scholar]
- Yoshimura and de Groat, 1999.Yoshimura N, de Groat WC. (1999) Increased excitability of afferent neurons innervating rat urinary bladder after chronic bladder inflammation. J Neurosci 19:4644–4653 [DOI] [PMC free article] [PubMed] [Google Scholar]