Abstract
Pouchitis after total rectocolectomy is the most common complication of ulcerative colitis (UC). The immunological mechanisms involved in the genesis of pouchitis are unclear. Therefore, we evaluated the inflammatory activity in normal ileal pouch mucosa by determining signal transducers and activators of transcription (STAT-1) activation and cytokine expression in patients operated for UC and familial adenomatous polyposis (FAP). Eighteen asymptomatic patients, who underwent total rectocolectomy and J pouch, were evaluated: nine with UC and nine with FAP. The activation of STAT-1 and cytokine expression were determined by immunoblot of total protein extracts from pouch mucosal biopsies. The absence of pouchitis was assessed by clinical, histological and endoscopic parameters, according to the Pouchitis Disease Activity Index. The patients were not receiving any medication. Analysis of variance (anova) and Tukey–Kramer's test were applied. The local ethical committee approved the study and informed consent was signed by all participants. STAT-1 activation was increased in UC when compared to FAP and controls (P < 0·05). Higher levels of interferon (IFN)-γ expression were observed in UC patients when compared to the control group (P < 0·05), but were similar to FAP. In contrast, cytokine signalling (SOCS-3) and interleukin (IL)-10 expression were similar in all groups (P > 0·05). These findings could explain the higher susceptibility to this inflammatory complication in UC when compared to FAP. A tendency towards increased levels of IFN-γ and STAT-1 in patients with UC, even without clinical and endoscopic evidence of pouchitis, was observed; studying inflammatory activity in asymptomatic ileal pouches may help understanding of the pathogenesis of pouchitis.
Keywords: cytokines, familial adenomatous polyposis, ileal pouch, pouchitis, ulcerative colitis
Introduction
Restorative rectocolectomy with ileal pouch is the surgical procedure of choice for ulcerative colitis (UC) and familial adenomatous polyposis (FAP) [1,2]. UC is a complex inflammatory bowel disease (IBD), with environmental and genetic factors involved, although its aetiology is not known completely. High expression of T helper type 2 (Th2) pathway cytokines, such as interleukin (IL)-4, IL-5 and IL-13, is reported in UC patients but is a point of discussion, because higher tumour necrosis factor α (TNF-α) expression (Th1 cytokine) is also seen in UC patients [3]. Mutation of the IL-10 receptor gene was identified to have a role in the pathogenesis of UC [4]. Recently, the Th17 pathway was identified mediating intestinal inflammation in IBD, especially in Crohn's disease (CD), and IL-23 was found to be responsible for inflammatory cytokine cascade involving increased levels of tumour necrosis factor (TNF)-α, IL-6, interferon-γ (IFN-γ) and IL-17 in the intestine [5].
Conversely, FAP is a rare genetic disease, with dominant autosomal heritage, determined in 95% of the cases by adenomatous polyposis coli (APC) gene mutation. The most common pouch design is the ‘J’ pouch, which is 15 cm long, with double bowel [6]. The ileal reservoir improves patients' quality of life because it allows fewer bowel movements and better function than the technique of direct anastomosis of the terminal ileum to anal canal. However, pouchitis is one of the major complications after this procedure and affects up to 50% of patients with UC, and only 5% of patients with FAP [7,8]. The cumulative risk of this complication is approximately 48% in 10 years [9]. Although pouchitis is a commonly reported complication, its aetiology is still unclear [10–14]. Currently, the diagnosis of pouchitis is based on clinical symptoms that must be confirmed by histology and endoscopic findings [15–17].
The difference in the incidence of pouchitis between patients with UC and FAP, which is higher in UC, has led to the suggestion that pathophysiological similarities exist between pouchitis and UC. This hypothesis is boosted further by the fact that some patients with pouchitis have a resurgence of extra-intestinal manifestations of UC in the same way as patients who have active UC [18,19].
Proinflammatory cytokines and signalling pathways involving transcription factors of the signal transducers and activators of transcription (STAT) family play a pivotal role in the pathogenesis of UC. Specifically, increased levels of IFN-γ and STAT-1 have been reported in samples from patients with IBD, particularly in UC [20–23]. Moreover, down-regulation of suppressor of cytokine signalling (SOCS-3) has been studied in UC disease, where this protein is a powerful inhibitor of the STAT family [22]. However, few studies have evaluated inflammatory activity in asymptomatic ileal pouches and pouchitis [7,24–30] and determined whether the pathophysiology is similar to that of UC [31–35]. Therefore, in order to compare inflammatory activity in asymptomatic pouches in highly pouchitis-prone UC patients and pouchitis-protected patients with FAP, we employed immunoblotting assays to determine the expression of pro- and anti-inflammatory cytokines and, in order to understand ileal pouch inflammation more clearly, the activation of a common intracellular proinflammatory signal transducer in biopsies from the pouches of FAP and UC patients.
Materials and methods
Mucosal biopsies were obtained from nine patients with non-inflamed ileo-anal pouches after rectocolectomy for UC [median age, 48·7 (range, 31–63) years; male 44·4%; female 55·6%] and nine patients with non-inflamed ileoanal pouch after rectocolectomy for FAP [median age, 33·8 (range, 21–59) years; male 44·4%; female 55·6%]. Six fragments of biopsy were obtained from each patient during colonoscopic examination. The follow-up after the operation was 73·1 (24–168) months. The reservoir design was of the ‘J’ type in all patients, and the right colonic vascular arcade was preserved as a supplementary blood supply for the terminal ileum [36]. Mucosectomy was also performed, with hand-sewn ileo-anal anastomosis. At the time of the study, the patients had had their ileostomy closed for more than 1 year. The absence of pouchitis was assessed clinically, histology and endoscopically, according to the Pouchitis Disease Activity Index (PDAI) [15]. The control group comprised nine individuals with normal colonoscopy examination, with an average of 40·9 (range, 26–58) years; 55·6% were female. Six fragments of biopsies were obtained from the terminal ileum of each patient by colonoscopy.
The study was performed in accordance with the Declaration of Helsinki and was approved by the ethical committee of the State University of Campinas. All biopsies were taken after informed consent from the patients. The study was carried out at the State University of Campinas, Coloproctology Unit and at the Cell Signaling Laboratory in the Department of Internal Medicine.
Mucosal biopsies from the pouches and from normal ileum were snap-frozen in liquid nitrogen, not buffered and stored at −80°C until use. For total protein extract preparation, the fragments were homogenized in solubilization buffer at 4°C [1% Triton X-100, 100 mM Tris-HCl (pH 7·4), 100 mM sodium pyrophosphate, 100 mM sodium fluoride, 10 mM ethylenediamine tetraacetic acid (EDTA), 10 mM sodium orthovanadate, 2·0 mM phenylmethylsulphonyl fluoride (PMSF) and 0·1 mg aprotinin/ml] with a Polytron PTA 20S generator (model PT 10/35; Brinkmann Instruments, Westbury, NY, USA) operated at maximum speed for 30 s. Insoluble material was removed by centrifugation for 20 min at 9000 g at 4°C. The protein concentrations of the supernatants were determined by the Bradford dye binding method [37]. Aliquots of the resulting supernatants containing 100 µg total proteins were separated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE), transferred to nitrocellulose membranes and blotted with anti-phospho-STAT-1, anti-IFN-γ, anti-IL-10 or anti-SOCS3 antibodies [38,39]. Bovine serum albumin was used for blocking other non-specific proteins. β-actin protein was used to normalize all proteins analysed.
Reagents for SDS-PAGE and immunoblotting were from Bio-Rad Laboratories (Richmond, CA, USA). Phenylmethylsulphonyl fluoride, aprotinin, Triton X-100, Tween 20 and glycerol were obtained from Sigma (St Louis, MO, USA). Nitrocellulose paper (BA85, 0·2 µm) was from Amersham (Aylesbury, UK). The anti-phospho-STAT-1 (sc-7988, goat polyclonal), anti-IFN-γ (sc-9344, goat polyclonal), anti-IL-10 (sc-1783, goat polyclonal) and anti-SOCS3 (sc-7009, goat polyclonal) antibodies were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). The proteins in the membrane were detected by chemiluminescent reaction (SuperSignal® West Pico chemiluminescent substrate from Pierce Biothechnology, Inc., Rockford, IL, USA) and the resulting bands were detected by exposure X-ray films.
All numerical results are arbitrary units expressed as the mean ± standard error of the mean (s.e.m.) of the indicated number of experiments. Results of the blots are presented as direct comparisons of bands in autoradiographs and quantified by densitometry using Gel-Pro Analyzer 3·1 software (Exon-Intron Inc., Farrell, MD, USA). Data were analysed by analysis of variance (anova) followed by analysis of significance (Tukey–Kramer's multiple comparisons test) comparing UC, FAP and control groups. The level of significance was set at P < 0·05.
Results
The level of activated tyrosine phosphorylated STAT-1 was significantly higher in UC when compared with FAP and controls (P < 0·05), demonstrating that it can be an inflammatory pathway predisposing to pouchitis. In addition, UC patients had significantly higher levels of IFN-γ when compared to controls (P < 0·05), correlating with STAT-1 activation. Although there was a tendency towards higher IFN-γ expression in UC than FAP, these were considered similar using statistical tests (P > 0·05) (Fig. 1).
Fig. 1.
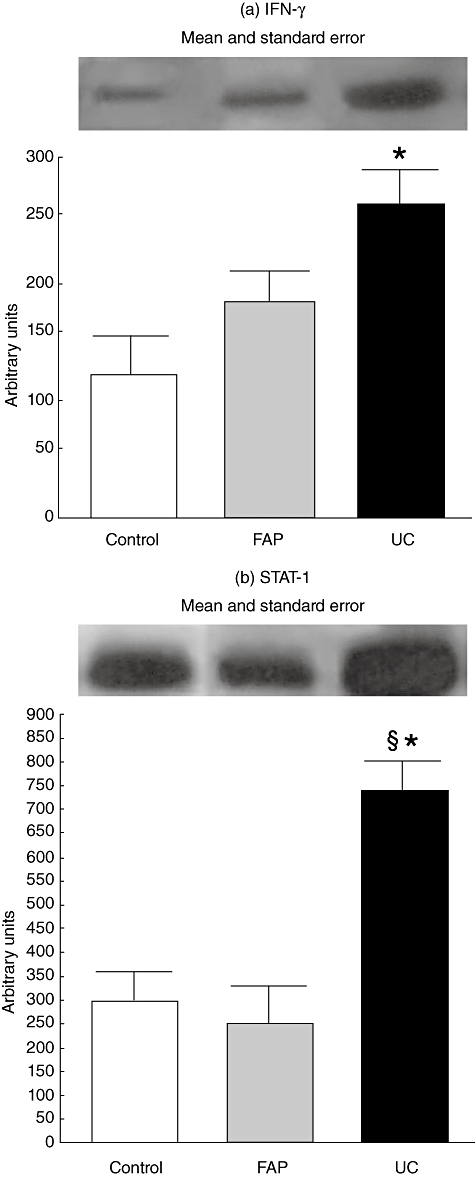
Activation of signal transducer and activator of transcription-1 (STAT-1) and interferon (IFN)-γ protein expression in control, familial adenomatous polyposis (FAP) and ulcerative colitis (UC) groups. For illustration purposes each line band represents one patient. For all conditions, n = 09, FAP*versus control, P < 0·05; UC§versus FAP, P < 0·05.
The levels of IL-10 and SOCS-3 expressions were similar in UC, FAP and control patients (P > 0·05), although the graphs reveal a tendency for a higher expression of these proteins in ileal pouch of UC patients (Fig. 2). β-actin expression was similar among the groups in all experiments.
Fig. 2.
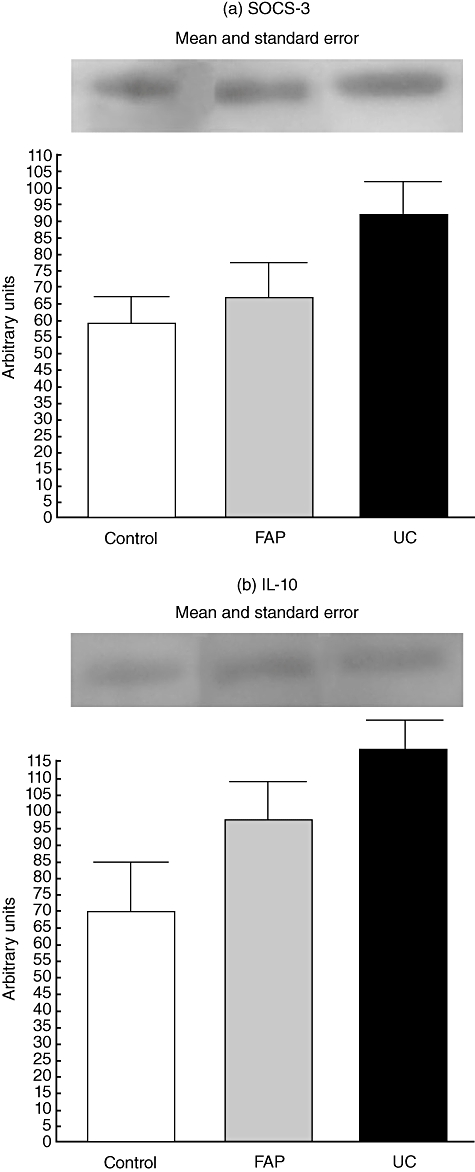
Expression of interleukin (IL)-10 and suppressor of cytokine signalling (SOCS-3) in control, familial adenomatous polyposis (FAP) and ulcerative colitis (UC) groups. For illustration purposes each line band represents one patient. For all conditions, n = 09, FAP*versus control, P < 0·05; UC§versus FAP, P < 0·05.
Discussion
The aetiology of primary pouchitis remains unclear, and this has precluded the development of appropriate prophylaxis and treatment [40,41]. A previous diagnosis of UC seems to play an important role when compared to FAP, due to the higher frequency of this postoperative complication in UC patients [30].
Development of colon inflammation in UC involves increased levels of proinflammatory cytokines, such as IFN-γ and activators of transcription of TNF-α, such as STAT-1, which is associated with an imbalance of T cell immunoregulatory cytokines such as IL-10 [4,21,22,42]. The expression of IFN-γ and IL-10 and activation of STAT-1 have been reported in IBD, especially in UC. However, few studies have evaluated the immunological activity in ileal pouches and whether there is a tendency for inflammation in asymptomatic patients with ileal pouches [43]. It has been shown that the development of pouchitis correlates with high levels of TNF-α, IL-1β, IL-6, IL-8 and decreased levels of IL-10 in UC [24–26,28,29,35]. Patients with FAP, however, demonstrate low proinflammatory cytokine expression, which correlates with a low incidence of pouchitis [7].
Although TNF-α has an important role in inflammatory pathways seen in IBD, IFN-γ seems to play a pivotal role in UC patients. Indeed, STAT-1, and not nuclear factor kappa B (NF-κB), has been suggested to be the major nuclear transcription factor of TNF-α activated by IFN-γ in cases of UC, detected by Western blotting [22,44]. Wu et al. [45] studied gene expression in IBD patients and observed differences between Crohn's disease and UC. There was preferential overexpression of STAT-1 and STAT-3 genes in Crohn's disease when compared to UC and controls. However, Mudter et al. [46] reported phosphorylated STAT-1 augmented in IBD, predominantly in UC, without significant differences among UC, Crohn's disease and controls, although the same study revealed higher levels of STAT-1 in T cells of Crohn's disease without statistical significance among the groups. This could be due to a different method of measuring STAT-1 activation [immunofluorescence staining and quantitative real-time reverse transcription–polymerase chain reaction (RT–PCR)]. Low levels of suppressor of cytokine signalling (SOCS-3) expression, an inhibitor of STAT activation, were observed in UC disease [22]. In light of this, some authors have studied IFN-γ and STAT-1 in ileal pouches and determined an increased expression of these proteins in UC patients [30–33]. However, only one study has reported the expression of IFN-γ in the ileal pouches of FAP patients [34]; in contrast, STAT-1 has not yet been studied in FAP. Furthermore, SOCS-3 expression has not been studied in the ileal pouch of UC and FAP patients.
We observed that IFN-γ expression was increased in the pouch mucosa of UC patients compared with controls. In agreement with this, STAT-1, a transcription factor responsive to IFN-γ, was also increased in UC patients. It is extremely interesting that, even in such optimal clinical conditions, the local levels of IFN-γ and STAT-1 were higher in UC. IFN-γ, together with TNF-α, is known to play an important role in the early steps of installation of inflammation, recruiting and activating monocytes and playing important roles in the induction of expression of other inflammatory cytokines. IFN-γ expression was similar in UC and FAP patients in this study: this finding may be explained due to the fact that patients were asymptomatic and that differences in cytokine levels may not be detectable in the absence of endoscopic inflammation and other anti-inflammatory cytokines, such as transforming growth factor (TGF)-β[47], could be blocking the main proinflammatory cytokines, such as IFN-γ, in the normal pouch considering endoscopic and histological perspectives. Therefore, UC patients had a tendency to present inflammatory activity. Such activity may have a close connection with primary disease in these patients, and may predispose them to frequent development of local inflammation.
IFN-γ expression and STAT-1 activation were found to be similar in FAP patients when compared to control individuals, demonstrating that FAP patients do not have tendency for pouch inflammation. IL-10 and SOCS-3 expression was also similar in these groups, due to the fact that these are asymptomatic; as such, a balance between pro- and anti-inflammatory pathways may exist. It should be emphasized that, on a clinical basis, FAP and UC patients behave in very distinct manners. UC patients are prone to inflammatory activity, which is the basis of their primary disease. By retaining some subclinical inflammatory activity, as illustrated in this study by the increased local levels of IFN-γ and STAT-1, UC patients may become predisposed to pouchitis. Here, inflammation of the pouch mucosa may represent a recurrence of UC, and this aspect must be relevant because of the higher incidence of pouchitis in UC patients compared with FAP patients. It must be remembered that ileal pouch surgery has been performed by surgeons for the treatment of the two distinct conditions, inflammatory (UC) and autosomal dominant proliferative disease (FAP). Studies comparing different expression of inflammatory cytokines in the ileal pouch of asymptomatic UC and FAP patients are rare, and the majority of these studies concerned patients with pouchitis. Because it may be an extremely difficult condition to treat, it should be considered that the beginning of this unpredictable condition is not completely known; studies that focus on this aspect should be relevant.
In summary, the present study shows that, even in asymptomatic conditions, patients with UC have an inflammatory activity condition in pouches that may be similar to that observed in UC disease. The results help to explain the higher incidence of pouchitis in UC patients compared to FAP.
Acknowledgments
We thank A. L. N. Domingues for technical assistance (Inflammatory Bowel Disease Ambulatory – Coloproctology Unit). These studies were supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo and Fundo de Apoio ao Ensino, Pesquisa e Extensão from the University of Campinas.
Disclosure
None.
References
- 1.M'Koma AE. Serum biochemical evaluation of patients with functional pouches tem to 20 years after restorative proctocolectomy. Int J Colorectal Dis. 2006;26:1–10. doi: 10.1007/s00384-005-0076-3. [DOI] [PubMed] [Google Scholar]
- 2.Mc Guire BB, Brannigan AE, O'Connell PR. Ileal pouch-anal anastomosis. Br J Surg. 2007;94:812–23. doi: 10.1002/bjs.5866. [DOI] [PubMed] [Google Scholar]
- 3.Hanauer SB. Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflamm Bowel Dis. 2006;12:S3–9. doi: 10.1097/01.mib.0000195385.19268.68. [DOI] [PubMed] [Google Scholar]
- 4.Franke A, Balschun T, Karlsen TH, et al. Sequence variants in IL-10, ARPC2 and multiple other loci contribute to ulcerative colitis susceptibility. Nat Genet. 2008;40:1319–23. doi: 10.1038/ng.221. [DOI] [PubMed] [Google Scholar]
- 5.McGovern D, Powrie F. The IL23 axis plays a key role in the pathogenesis of IBD. Gut. 2007;56:1333–6. doi: 10.1136/gut.2006.115402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Utsunomiya J, Iwama T, Imajo M, et al. Total colectomy, mucosal proctectomy, and ileoanal anastomosis. Dis Colon Rectum. 1980;23:459–66. doi: 10.1007/BF02987076. [DOI] [PubMed] [Google Scholar]
- 7.Kiehne K, Brunke G, Wegner F, et al. Defensin expression in chronic pouchitis in patients with ulcerative colitis or familial adenomatous polyposis coli. World J Gastroenterol. 2006;12:1056–62. doi: 10.3748/wjg.v12.i7.1056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shen B, Fazio VW, Renzi FH, et al. Risk factors for disease of ileal pouch-anal anastomosis after restorative proctocolectomy for ulcerative colitis. Clin Gastroenterol Hepatol. 2006;4:81–9. doi: 10.1016/j.cgh.2005.10.004. [DOI] [PubMed] [Google Scholar]
- 9.Meagher AP, Farouk R, Dozois RR, et al. J ileal pouch-anal anastomosis for chronic ulcerative colitis: complications and long-term outcome in 1310 patients. Br J Surg. 1998;85:800–3. doi: 10.1046/j.1365-2168.1998.00689.x. [DOI] [PubMed] [Google Scholar]
- 10.Laake KO, Bjorneklett A, Aamodt G, et al. Outcome of four weeks' intervention with probiotics on symptoms and endoscopic appearance after surgical reconstruction with a J-configurated ileal-pouch-anal-anastomosis in ulcerative colitis. Scand J Gastroenterol. 2005;40:43–51. doi: 10.1080/00365520410009339. [DOI] [PubMed] [Google Scholar]
- 11.Ohge H, Furne JK, Springfield J, et al. Association between fecal hydrogen sulfide production and pouchitis. Dis Colon Rectum. 2005;48:469–75. doi: 10.1007/s10350-004-0820-8. [DOI] [PubMed] [Google Scholar]
- 12.Kuehbacher T, Ott SJ, Helwig U, et al. Bacterial and fungal microbiota in relation to probiotic therapy in pouchitis. Gut. 2006;55:833–41. doi: 10.1136/gut.2005.078303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kienle P, Weitz J, Reinshagen S, et al. Association of decreased perfusion of the ileoanal pouch mucosa with early postoperative pouchitis and local septic complications. Arch Surg. 2001;136:1124–30. doi: 10.1001/archsurg.136.10.1124. [DOI] [PubMed] [Google Scholar]
- 14.Heuschen G, Leowardi C, Hinz U, et al. Differential expression of toll-like receptor 3 and 5 in ileal pouch mucosa of ulcerative colitis patients. Int J Colorectal Dis. 2007;22:293–301. doi: 10.1007/s00384-006-0133-6. [DOI] [PubMed] [Google Scholar]
- 15.Sandborn WJ, Tremaine WJ, Batts KP, et al. Pouchitis after ileal pouch-anal anastomosis: a Pouchitis Disease Activity Index. Mayo Clin Proc. 1994;69:409–15. doi: 10.1016/s0025-6196(12)61634-6. [DOI] [PubMed] [Google Scholar]
- 16.Shen B, Achkar JP, Connor JT, et al. Modified Pouchitis Disease Activity Index. A simplified approach to the diagnosis of pouchitis. Dis Colon Rectum. 2003;46:748–53. doi: 10.1007/s10350-004-6652-8. [DOI] [PubMed] [Google Scholar]
- 17.Akerlung JE, Lofberg R. Pouchitis. Curr Opin Gastroenterol. 2004;20:341–4. doi: 10.1097/00001574-200407000-00007. [DOI] [PubMed] [Google Scholar]
- 18.Lohmuller JL, Pemberton JH, Dozois RR, et al. Pouchitis and extraintestinal manifestations of inflammatory bowel disease after ileal pouch-anal anastomosis. Ann Surg. 1990;211:622–9. [PMC free article] [PubMed] [Google Scholar]
- 19.Hata K, Watanabe T, Shinozaki M, et al. Patients with extraintestinal manifestations have a higher risk of developing pouchitis in ulcerative colitis: multivariate analysis. Scand J Gastroenterol. 2003;38:1055–8. doi: 10.1080/00365520310005938. [DOI] [PubMed] [Google Scholar]
- 20.O'Shea J, Ma A, Lipsky P. Cytokines and autoimmunity. Nat Rev. 2002;2:37–45. doi: 10.1038/nri702. [DOI] [PubMed] [Google Scholar]
- 21.Mudter J, Weigmann B, Bartsch B, et al. Activation pattern of signal transducers and activators of transcription (STAT) factors in inflammatory bowel disease. Am J Gastroenterol. 2005;100:64–72. doi: 10.1111/j.1572-0241.2005.40615.x. [DOI] [PubMed] [Google Scholar]
- 22.Schreiber S, Rosenstiel P, Hampe J, et al. Activation of signal tranducer and activator of transcription (STAT) 1 in human chronic inflammatory bowel disease. Gut. 2002;51:379–85. doi: 10.1136/gut.51.3.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Klampfer L, Huang J, Sasazuki T, et al. Inhibition of interferon γ signaling by the short chain fatty acid butyrate. Mol Cancer Res. 2003;1:855–62. [PubMed] [Google Scholar]
- 24.Gionchetti P, Campieri M, Belluzzi A, et al. Mucosal concentrations of interleukin-1 beta, interleukin-6, interleukin-8, and tumor necrosis factor-alpha in pelvic ileal pouches. Dig Dis Sci. 1994;39:1525–31. doi: 10.1007/BF02088059. [DOI] [PubMed] [Google Scholar]
- 25.Evgenikos N, Bartolo DCC, Hamer-Hodges DW, et al. Assessment of ileoanal pouch inflammation by interleukin 1β and interleukin 8 concentrations in the gut lumen. Dis Colon Rectum. 2002;45:249–55. doi: 10.1007/s10350-004-6156-6. [DOI] [PubMed] [Google Scholar]
- 26.Bulois P, Tremaine WJ, Maunoury V, et al. Pouchitis is associated with mucosal imbalance between interleukin-8 and interleukin-10. Inflamm Bowel Dis. 2000;6:157–67. doi: 10.1097/00054725-200008000-00001. [DOI] [PubMed] [Google Scholar]
- 27.Lammers KM, Vergopoulos A, Babel N, et al. Probiotic therapy in the prevention of pouchitis onset: decreased interleukin-1β, interleukin-8 and interferon-γ gene expression. Inflamm Bowel Dis. 2005;11:447–54. doi: 10.1097/01.mpa.0000160302.40931.7b. [DOI] [PubMed] [Google Scholar]
- 28.Schmidt C, Giese T, Ludwig B, et al. Increased cytokine transcripts in pouchitis reflect the degree of inflammation but not the underlying entity. Int J Colorectal Dis. 2006;21:419–26. doi: 10.1007/s00384-005-0024-2. [DOI] [PubMed] [Google Scholar]
- 29.Helwig U, Gionchetti P, Rizello F, et al. CXC and CC chemokine expression in inflamed and noninflamed pelvic ileal pouch tissue. Int J Colorectal Dis. 2004;19:165–70. doi: 10.1007/s00384-003-0500-5. [DOI] [PubMed] [Google Scholar]
- 30.Ulisse S, Gionchetti P, D'Aló S, et al. Expression of cytokines, inducible nitric oxide synthase, and matrix metalloproteinases in pouchitis: effects of probiotic treatment. Am J Gastroenterol. 2001;96:2691–9. doi: 10.1111/j.1572-0241.2001.04139.x. [DOI] [PubMed] [Google Scholar]
- 31.Kuhbacher T, Gionchetti P, Hampe J, et al. Activation of signal-transducer and activator of transcription 1 (STAT1) in pouchitis. Clin Exp Immunol. 2001;123:395–401. doi: 10.1046/j.1365-2249.2001.01455.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Boerr L, Sambuelli A, Sugay E, et al. Serum TGF-β1 and interferon-γ levels in ileal pouch-anal anastomosis in ulcerative colitis. Gastroenterology. 1998;114:A936. abstract. [Google Scholar]
- 33.Schreiber S, Gionchetti P, Hampe J, et al. Pouchitis shares similarities with ulcerative colitis in the pattern of cytokine transcription factor activation. Gastroenterology. 1997;112:A1086. [Google Scholar]
- 34.Stallmach A, Schafer F, Hoffmann S, et al. Increased state of activation of CD4 positive T cells and elevated interferon γ production in pouchitis. Gut. 1998;43:499–505. doi: 10.1136/gut.43.4.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Patel RT, Bain I, Youngs D, et al. Cytokine production in pouchitis is similar to that in ulcerative colitis. Dis Colon Rectum. 1995;38:831–7. doi: 10.1007/BF02049839. [DOI] [PubMed] [Google Scholar]
- 36.Góes JRN, Coy CSR, Amaral CA, et al. Superior mesenteric artery syndrome as a complication of ileal pouch-anal anastomosis. Dis Colon Rectum. 1995;38:543–4. doi: 10.1007/BF02148857. [DOI] [PubMed] [Google Scholar]
- 37.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–54. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- 38.Velloso LA, Folli F, Sun XJ, et al. Cross-talk between the insulin and angiotensin signaling systems. Proc Natl Acad Sci USA. 1996;93:12490–5. doi: 10.1073/pnas.93.22.12490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Araújo EP, De Souza CT, Gasparetti AL, et al. Short-term in vivo inhibition of insulin receptor substrate-1 expression leads to insulin resistence, hyperinsulinemia, and increased adiposity. Endocrinology. 2005;146:1428–37. doi: 10.1210/en.2004-0778. [DOI] [PubMed] [Google Scholar]
- 40.Kuhbacher T, Schreiber S, Runkel N. Pouchitis: pathophysiology and treatment. Int J Colorectal Dis. 1998;13:196–207. doi: 10.1007/s003840050162. [DOI] [PubMed] [Google Scholar]
- 41.Tulchinsky H, Hawley PR, Nicholls J. Long-term failure after restorative proctocolectomy for ulcerative colitis. Ann Surg. 2003;238:229–34. doi: 10.1097/01.sla.0000082121.84763.4c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kuhn K, Lohler J, Rennick D, et al. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263–74. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- 43.Leal RF, Coy CSR, Ayrizono MLS, et al. Differential expression of pro-inflammatory cytokines and a pro-apoptotic protein in ileal pouches for ulcerative colitis and familial adenomatous polyposis. Tech Coloproctol. 2008;12:33–8. doi: 10.1007/s10151-008-0395-9. [DOI] [PubMed] [Google Scholar]
- 44.Schreiber S, Nikolaus S, Hampe J. Activation of nuclear factor KB in inflammatory bowel disease. Gut. 1998;42:477–84. doi: 10.1136/gut.42.4.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wu F, Dassopoulos T, Cope L, et al. Genome-wide gene expression differences in Crohn's disease and ulcerative colitis from endoscopic pinch biopsies: insights into distinctive pathogenesis. Inflamm Bowel Dis. 2007;13:807–21. doi: 10.1002/ibd.20110. [DOI] [PubMed] [Google Scholar]
- 46.Mudter J, Weigmann B, Bartsch B, et al. Activation pattern of signal transducers and activators of transcription (STAT) factors in inflammatory bowel diseases. Am J Gastroenterol. 2005;100:64–72. doi: 10.1111/j.1572-0241.2005.40615.x. [DOI] [PubMed] [Google Scholar]
- 47.Sartor RB. Mechanisms of disease: pathogenesis of Crohn's disease and ulcerative colitis. Nat Clin Pract Gastroenterol Hepatol. 2006;3:390–407. doi: 10.1038/ncpgasthep0528. [DOI] [PubMed] [Google Scholar]


