Abstract
Noninvasive methods for the bacteriological diagnosis of early-stage Mycobacterium ulcerans infection are not available. It was recently shown that fine-needle aspiration (FNA) could be used for diagnosing M. ulcerans infection in ulcerative lesions. We report that FNA is an appropriate sampling method for diagnosing M. ulcerans infection in nonulcerative lesions.
Mycobacterium ulcerans infection (Buruli ulcer) is one of the 13 most neglected tropical diseases (9) and the third most common mycobacterial infection after tuberculosis and leprosy in immunocompetent humans (2, 6, 13-14). In general, this skin disease initially manifests as a painless nodule or papule, plaque, or edema (2). Without early intervention, these symptoms evolve into painless ulcers with undermined edges. The epidemiological, scientific, and management aspects of this disease have been well described (12). Over recent years, the management of Buruli ulcer patients has considerably changed with advances in antibiotherapy (3, 5).
Laboratory diagnosis of this mycobacterial infection is based on detection of acid-fast bacilli (AFB) through the direct examination of samples, isolation of mycobacteria by culture, histological analysis, and detection of M. ulcerans DNA by PCR (12). Ulcerative lesion specimens are collected using swabs (12). Swabbing from the undermined edges of ulcers may sometimes be difficult and painful. Collecting specimens from patients with nonulcerative lesions necessitates invasive procedures, such as incisional, excisional, or punch biopsies, which require hospital infrastructure not available in remote rural areas in Africa where M. ulcerans infection is endemic. Two studies recently reported that fine-needle aspirates could be used to diagnose M. ulcerans infection in ulcerative lesions. In both studies, the number of patients enrolled was not large enough to draw conclusions on the effectiveness of this technique in diagnosing nonulcerative forms.
First, we compared the diagnostic sensitivities of fine-needle aspiration (FNA) and swabbing in 64 patients with ulcerative lesions. These patients had skin lesions consistent with active M. ulcerans infection, based on the clinical definition of the World Health Organization (12). For each patient with ulcerative lesions, two swab samples were taken from beneath the undermined edges of the ulcers and one FNA sample was taken from the edge of the lesion. The FNA procedure was similar to that described previously (4, 11); however, we used 20-gauge, 25-mm needles (attached to 5-ml syringes) instead of the 21-gauge and 23-gauge needles used in other studies. All samples were placed in sterile Venosafe tubes (Terumo) and sent, at room temperature, to the bacteriology unit of Angers University Hospital, France, within 7 days of collection for processing.
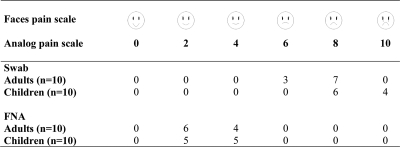
Significant differences were observed in the efficacies of the two sampling methods. PCR using FNA samples detected M. ulcerans DNA in 56 of the 71 patients (diagnostic sensitivity of 79%), and PCR using swab samples detected M. ulcerans DNA in 68 of 71 patients (sensitivity of 95%) (Table 1). Chi-square tests showed that the number of positive smears (direct smear examination) was significantly different (P < 0.0001) between swab (50.7%) and FNA (9.9%) samples. The number of positive FNA PCR results was not significantly lower than those for swab samples (P < 0.46). However, there was a significant difference in the number of negative PCR results between fine-needle aspiration (21.1%) and swab (4%) samples (P < 0.007). Overall, these comparisons showed that PCR analysis of swab samples was more accurate than that of FNA samples for diagnosing ulcerative forms. For each swab collection and FNA, the patient's response to pain was assessed according to standard pain assessment methods (15). Twenty adults (aged 15 to 35 years) and 20 children (aged 5 to 12 years) presenting ulcerative lesions (5 to 15 cm in diameter) localized on right or left limbs were enrolled. The analysis of results clearly demonstrated that FNA was less painful and thus more comfortable for the patient than swabbing (Fig. 1).
TABLE 1.
Results of direct smear examination and M. ulcerans DNA detection from swabs and fine-needle aspirations from ulcerative and nonulcerative lesions
| Lesion specimen and sampling method | No. (%) of specimens with indicated result: |
|||
|---|---|---|---|---|
| DSEa |
PCR |
|||
| Positive | Negative | Positive | Negative | |
| Ulcerative (n = 71) | ||||
| FNA | 7 (9.9) | 64 (90.1) | 56 (78.9) | 15 (21.1) |
| Swabbing | 36 (50.7) | 35 (49.3) | 68 (95.8) | 3 (4.2) |
| Nonulcerative, FNA (n = 64) | 16 (25.0) | 48 (75.0) | 57 (89.0) | 7 (11.0) |
DSE, direct smear examination.
FIG. 1.
Pain assessment during swabbing and FNA.
Second, 64 patients were recruited to evaluate the diagnostic sensitivity of FNA from nonulcerative lesions. FNA was used to collect a unique sample from the estimated center of the lesion. We detected AFB in 25% of these samples (16/64 samples) through direct smear examination after Ziehl-Neelsen staining. PCR was positive for M. ulcerans DNA in 89% of the samples (57/64) (Table 1). Therefore, the sensitivity of PCR in FNA samples from nonulcerative forms was 89%. Seven cases were initially negative for M. ulcerans DNA detection. Repeating the sampling a few days later resulted in positive PCR results for four of these seven patients; the remaining three patients were diagnosed with lymphoma through differential diagnosis. Therefore, the sensitivity of the diagnosis from FNA samples was calculated to be 93.4% (57/61). The rate of positive smears from ulcerative and nonulcerative lesions in our study was lower than those obtained with other sampling methods (1, 8, 10). Thus, M. ulcerans DNA not associated with bacilli may be detected in aspirate liquid. M. ulcerans secretes vesicles containing mycolactone (7). Indeed, mass spectrometry analysis demonstrated the presence of mycolactone A/B in FNA liquid in samples collected from confirmed (PCR) Buruli ulcer patients. Moreover, we demonstrated that the vesicles contain M. ulcerans DNA (unpublished results). Therefore, we hypothesize that many vesicles and few bacilli (which are localized in tissues) are collected during FNA, explaining the high rate of positive PCR results compared to the low rate of positive smears.
To conclude, FNA is a simple, fast, accurate, painless, and inexpensive method of sampling which may be used for diagnosing M. ulcerans infection by PCR, particularly in patients presenting early-stage nonulcerative lesions.
Acknowledgments
This work was supported by the Fondation Française Raoul Follereau, the Institut National de la Santé et de la Recherche Médicale (INSERM), the European Community (FEDER 10250), Angers Loire Métropole, Région Pays de Loire, and Pasteur Network (PTR 212).
Footnotes
Published ahead of print on 7 April 2010.
REFERENCES
- 1.Affolabi, D., H. Bankole, A. Ablordey, J. Hounnouga, P. Koutchakpo, G. Sopoh, J. Aguiar, A. Dossou, R. C. Johnson, S. Anagonou, and F. Portaels. 2008. Effects of grinding surgical tissue specimens and smear staining methods on Buruli ulcer microscopic diagnosis. Trop. Med. Int. Health 13:187-190. [DOI] [PubMed] [Google Scholar]
- 2.Asiedu, K., R. Sherpbier, and M. C. Raviglione. 2000. Buruli ulcer Mycobacterium ulcerans infection. Global Buruli Ulcer Initiative. World Health Organization, Geneva, Switzerland.
- 3.Chauty, A., M. F. Ardant, A. Adeye, H. Euverte, A. Guedenon, C. Johnson, J. Aubry, E. Nuermberger, and J. Grosset. 2007. Promising clinical efficacy of streptomycin-rifampin combination for treatment of Buruli ulcer (Mycobacterium ulcerans disease). Antimicrob. Agents Chemother. 51:4029-4035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Eddyani, M., A. G. Fraga, F. Schmitt, C. Uwizeye, K. Fissette, C. Johnson, J. Aguiar, G. Sopoh, Y. Barogui, W. M. Meyers, J. Pedrosa, and F. Portaels. 2009. Fine-needle aspiration, an efficient sampling technique for bacteriological diagnosis of nonulcerative Buruli ulcer. J. Clin. Microbiol. 47:1700-1704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Etuaful, S., B. Carbonnelle, J. Grosset, S. Lucas, C. Horsfield, R. Phillips, M. Evans, D. Ofori-Adjei, E. Klustse, J. Owusu-Boateng, G. K. Amedofu, P. Awuah, E. Ampadu, G. Amofah, K. Asiedu, and M. Wansbrough-Jones. 2005. Efficacy of the combination rifampin-streptomycin in preventing growth of Mycobacterium ulcerans in early lesions of Buruli ulcer in humans. Antimicrob. Agents Chemother. 49:3182-3186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Johnson, P. D., T. Stinear, P. L. Small, G. Pluschke, R. W. Merritt, F. Portaels, K. Huygen, J. A. Hayman, and K. Asiedu. 2005. Buruli ulcer (M. ulcerans infection): new insights, new hope for disease control. PLoS Med. 2:e108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Marsollier, L., P. Brodin, M. Jackson, J. Kordulakova, P. Tafelmeyer, E. Carbonnelle, J. Aubry, G. Milon, P. Legras, J. P. Andre, C. Leroy, J. Cottin, M. L. Guillou, G. Reysset, and S. T. Cole. 2007. Impact of Mycobacterium ulcerans biofilm on transmissibility to ecological niches and Buruli ulcer pathogenesis. PLoS Pathog. 3:e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mensah-Quainoo, E., D. Yeboah-Manu, C. Asebi, F. Patafuor, D. Ofori-Adjei, T. Junghanss, and G. Pluschke. 2008. Diagnosis of Mycobacterium ulcerans infection (Buruli ulcer) at a treatment centre in Ghana: a retrospective analysis of laboratory results of clinically diagnosed cases. Trop. Med. Int. Health 13:191-198. [DOI] [PubMed] [Google Scholar]
- 9.Molyneux, D. H., P. J. Hotez, and A. Fenwick. 2005. “Rapid-impact interventions”: how a policy of integrated control for Africa's neglected tropical diseases could benefit the poor. PLoS Med. 2:e336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Phillips, R., C. Horsfield, S. Kuijper, A. Lartey, I. Tetteh, S. Etuaful, B. Nyamekye, P. Awuah, K. M. Nyarko, F. Osei-Sarpong, S. Lucas, A. H. Kolk, and M. Wansbrough-Jones. 2005. Sensitivity of PCR targeting the IS2404 insertion sequence of Mycobacterium ulcerans in an assay using punch biopsy specimens for diagnosis of Buruli ulcer. J. Clin. Microbiol. 43:3650-3656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Phillips, R. O., F. S. Sarfo, F. Osei-Sarpong, A. Boateng, I. Tetteh, A. Lartey, E. Adentwe, W. Opare, K. B. Asiedu, and M. Wansbrough-Jones. 2009. Sensitivity of PCR targeting Mycobacterium ulcerans by use of fine-needle aspirates for diagnosis of Buruli ulcer. J. Clin. Microbiol. 47:924-926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Portaels, F., P. Johnson, and W. M. Meyers. 2001. Buruli ulcer—diagnosis of M. ulcerans disease. World Health Organization, Geneva, Switzerland.
- 13.Portaels, F., M. T. Silva, and W. M. Meyers. 2009. Buruli ulcer. Clin. Dermatol. 27:291-305. [DOI] [PubMed] [Google Scholar]
- 14.Wansbrough-Jones, M., and R. Phillips. 2006. Buruli ulcer: emerging from obscurity. Lancet 367:1849-1858. [DOI] [PubMed] [Google Scholar]
- 15.Warden, V., A. C. Hurley, and L. Volicer. 2003. Development and psychometric evaluation of the Pain Assessment in Advanced Dementia (PAINAD) scale. J. Am. Med. Dir. Assoc. 4:9-15. [DOI] [PubMed] [Google Scholar]