Abstract
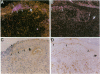
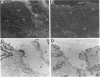
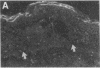
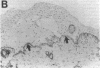
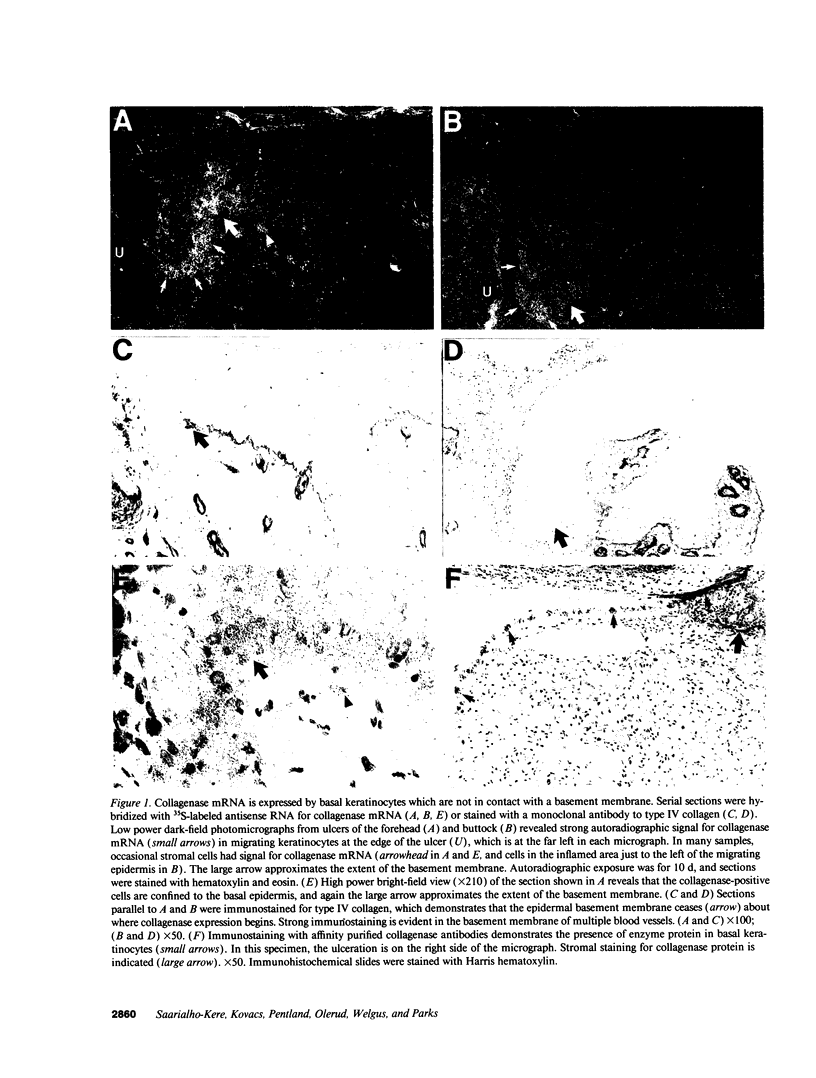
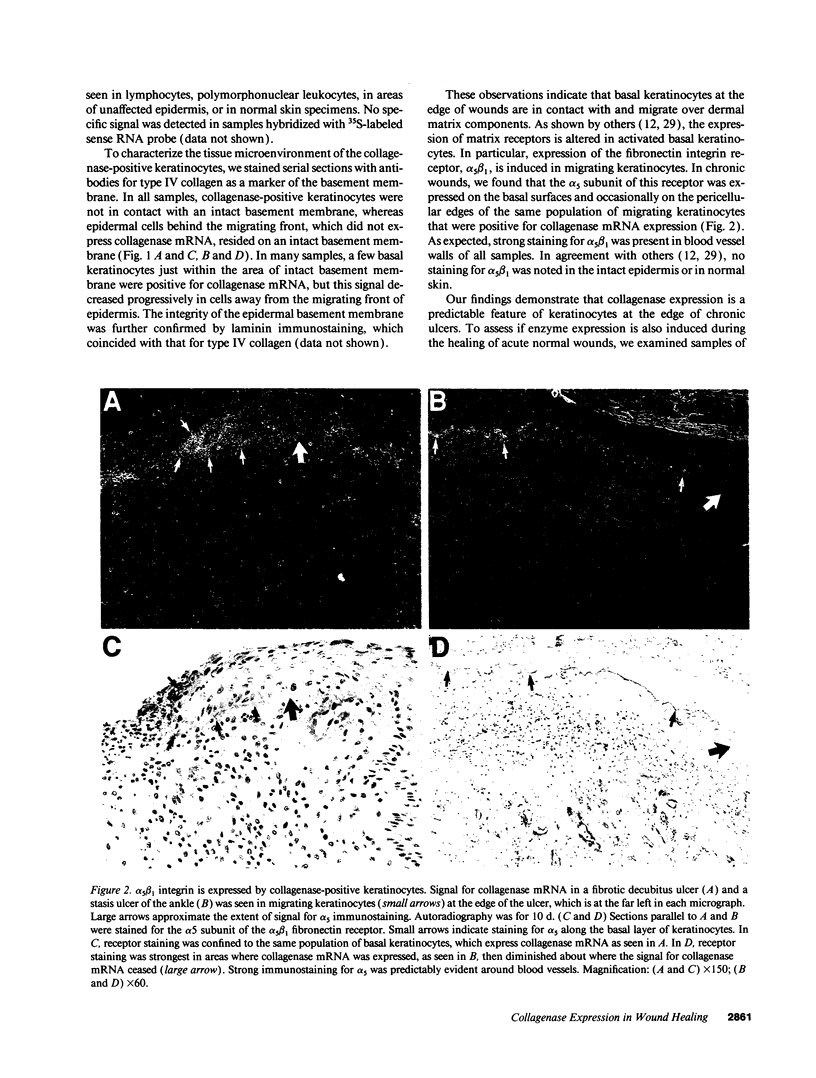
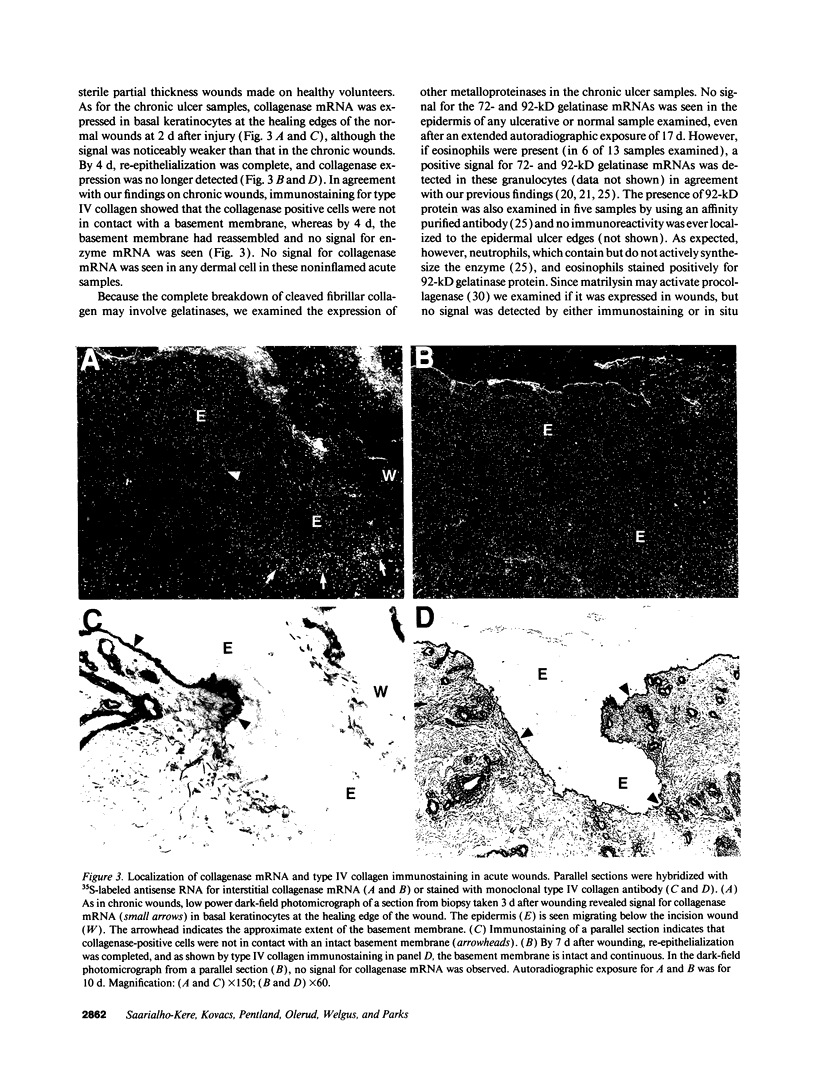
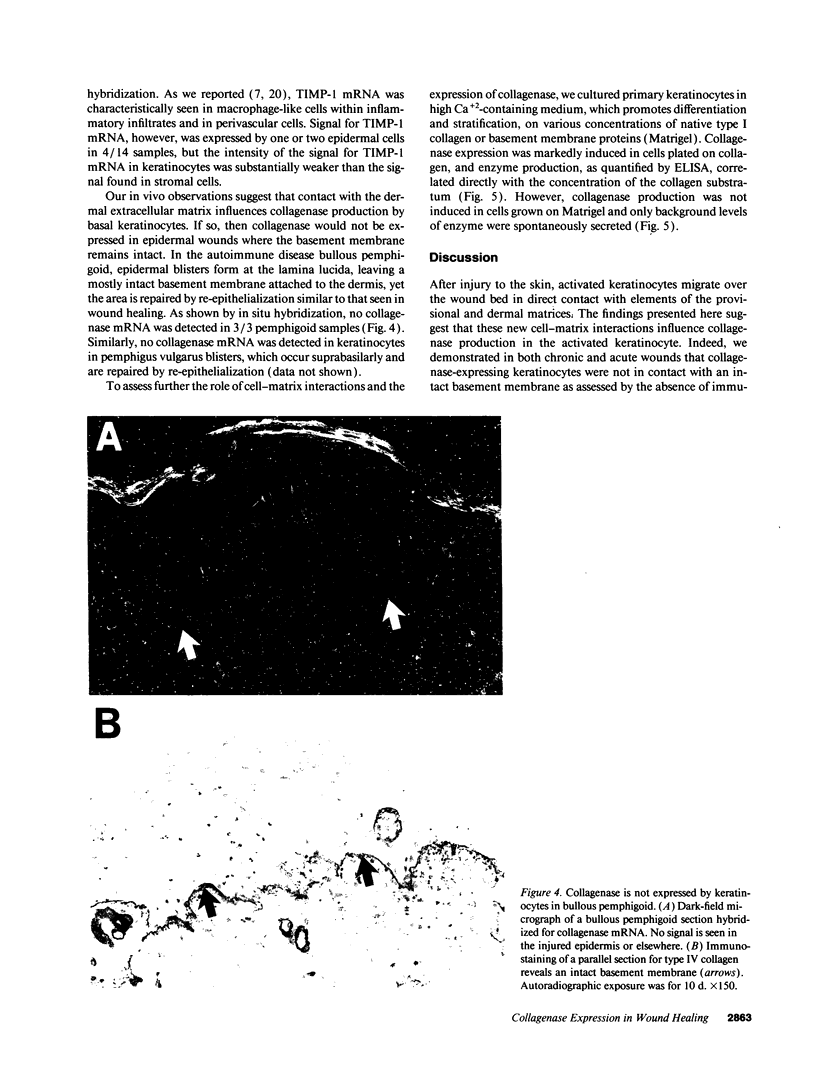
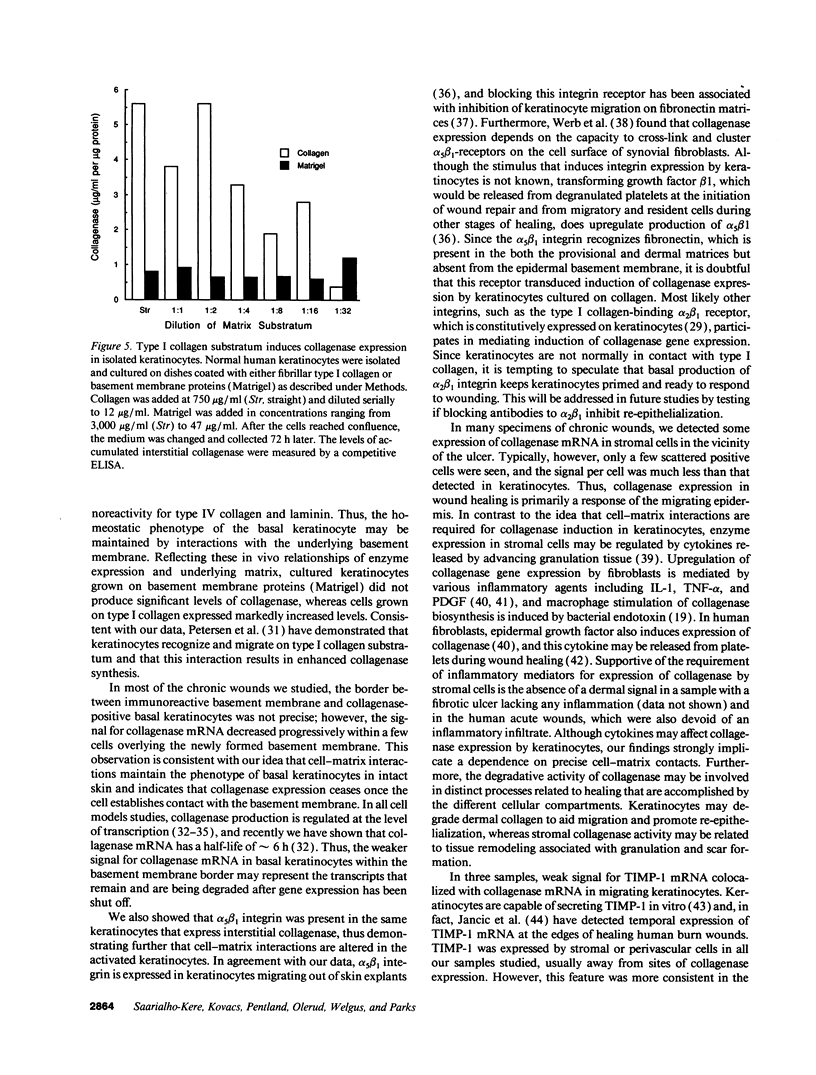
We reported that interstitial collagenase is produced by keratinocytes at the edge of ulcers in pyogenic granuloma, and in this report, we assessed if production of this metalloproteinase is a common feature of the epidermal response in a variety of wounds. In all samples of chronic ulcers, regardless of etiology, and in incision wounds, collagenase mRNA, localized by in situ hybridization, was prominently expressed by basal keratinocytes bordering the sites of active re-epithelialization indicating that collagenolytic activity is a characteristic response of the epidermis to wounding. No expression of mRNAs for 72- and 92-kD gelatinases or matrilysin was seen in keratinocytes, and no signal for any metalloproteinase was detected in normal epidermis. Immunostaining for type IV collagen showed that collagenase-positive keratinocytes were not in contact with an intact basement membrane and, unlike normal keratinocytes, expressed alpha 5 beta 1 receptors. These observations suggest that cell-matrix interactions influence collagenase expression by epidermal cells. Indeed, as determined by ELISA, primary cultures of human keratinocytes grown on basement membrane proteins (Matrigel; Collaborative Research Inc., Bedford, MA) did not express significant levels of collagenase, whereas cells grown on type I collagen produced markedly increased levels. These results suggest that migrating keratinocytes actively involved in re-epithelialization acquire a collagenolytic phenotype upon contact with the dermal matrix.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Angel P., Karin M. Specific members of the Jun protein family regulate collagenase expression in response to various extracellular stimuli. Matrix Suppl. 1992;1:156–164. [PubMed] [Google Scholar]
- Auble D. T., Brinckerhoff C. E. The AP-1 sequence is necessary but not sufficient for phorbol induction of collagenase in fibroblasts. Biochemistry. 1991 May 7;30(18):4629–4635. doi: 10.1021/bi00232a039. [DOI] [PubMed] [Google Scholar]
- Busiek D. F., Ross F. P., McDonnell S., Murphy G., Matrisian L. M., Welgus H. G. The matrix metalloprotease matrilysin (PUMP) is expressed in developing human mononuclear phagocytes. J Biol Chem. 1992 May 5;267(13):9087–9092. [PubMed] [Google Scholar]
- Carmichael D. F., Sommer A., Thompson R. C., Anderson D. C., Smith C. G., Welgus H. G., Stricklin G. P. Primary structure and cDNA cloning of human fibroblast collagenase inhibitor. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2407–2411. doi: 10.1073/pnas.83.8.2407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark R. A., Lanigan J. M., DellaPelle P., Manseau E., Dvorak H. F., Colvin R. B. Fibronectin and fibrin provide a provisional matrix for epidermal cell migration during wound reepithelialization. J Invest Dermatol. 1982 Nov;79(5):264–269. doi: 10.1111/1523-1747.ep12500075. [DOI] [PubMed] [Google Scholar]
- Collier I. E., Wilhelm S. M., Eisen A. Z., Marmer B. L., Grant G. A., Seltzer J. L., Kronberger A., He C. S., Bauer E. A., Goldberg G. I. H-ras oncogene-transformed human bronchial epithelial cells (TBE-1) secrete a single metalloprotease capable of degrading basement membrane collagen. J Biol Chem. 1988 May 15;263(14):6579–6587. [PubMed] [Google Scholar]
- Cooper T. W., Bauer E. A., Eisen A. Z. Enzyme-linked immunosorbent assay for human skin collagenase. Coll Relat Res. 1983 May;3(3):205–215. doi: 10.1016/s0174-173x(83)80004-1. [DOI] [PubMed] [Google Scholar]
- Docherty A. J., O'Connell J., Crabbe T., Angal S., Murphy G. The matrix metalloproteinases and their natural inhibitors: prospects for treating degenerative tissue diseases. Trends Biotechnol. 1992 Jun;10(6):200–207. doi: 10.1016/0167-7799(92)90214-g. [DOI] [PubMed] [Google Scholar]
- Edwards D. R., Murphy G., Reynolds J. J., Whitham S. E., Docherty A. J., Angel P., Heath J. K. Transforming growth factor beta modulates the expression of collagenase and metalloproteinase inhibitor. EMBO J. 1987 Jul;6(7):1899–1904. doi: 10.1002/j.1460-2075.1987.tb02449.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fini M. E., Girard M. T., Matsubara M. Collagenolytic/gelatinolytic enzymes in corneal wound healing. Acta Ophthalmol Suppl. 1992;(202):26–33. doi: 10.1111/j.1755-3768.1992.tb02165.x. [DOI] [PubMed] [Google Scholar]
- Goldberg G. I., Wilhelm S. M., Kronberger A., Bauer E. A., Grant G. A., Eisen A. Z. Human fibroblast collagenase. Complete primary structure and homology to an oncogene transformation-induced rat protein. J Biol Chem. 1986 May 15;261(14):6600–6605. [PubMed] [Google Scholar]
- Guo M., Kim L. T., Akiyama S. K., Gralnick H. R., Yamada K. M., Grinnell F. Altered processing of integrin receptors during keratinocyte activation. Exp Cell Res. 1991 Aug;195(2):315–322. doi: 10.1016/0014-4827(91)90379-9. [DOI] [PubMed] [Google Scholar]
- Hertle M. D., Adams J. C., Watt F. M. Integrin expression during human epidermal development in vivo and in vitro. Development. 1991 May;112(1):193–206. doi: 10.1242/dev.112.1.193. [DOI] [PubMed] [Google Scholar]
- Hibbs M. S., Hasty K. A., Seyer J. M., Kang A. H., Mainardi C. L. Biochemical and immunological characterization of the secreted forms of human neutrophil gelatinase. J Biol Chem. 1985 Feb 25;260(4):2493–2500. [PubMed] [Google Scholar]
- Kim J. P., Zhang K., Kramer R. H., Schall T. J., Woodley D. T. Integrin receptors and RGD sequences in human keratinocyte migration: unique anti-migratory function of alpha 3 beta 1 epiligrin receptor. J Invest Dermatol. 1992 May;98(5):764–770. doi: 10.1111/1523-1747.ep12499947. [DOI] [PubMed] [Google Scholar]
- Marcotte P. A., Kozan I. M., Dorwin S. A., Ryan J. M. The matrix metalloproteinase pump-1 catalyzes formation of low molecular weight (pro)urokinase in cultures of normal human kidney cells. J Biol Chem. 1992 Jul 15;267(20):13803–13806. [PubMed] [Google Scholar]
- Marinkovich M. P., Lunstrum G. P., Keene D. R., Burgeson R. E. The dermal-epidermal junction of human skin contains a novel laminin variant. J Cell Biol. 1992 Nov;119(3):695–703. doi: 10.1083/jcb.119.3.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matrisian L. M. Metalloproteinases and their inhibitors in matrix remodeling. Trends Genet. 1990 Apr;6(4):121–125. doi: 10.1016/0168-9525(90)90126-q. [DOI] [PubMed] [Google Scholar]
- Matrisian L. M. The matrix-degrading metalloproteinases. Bioessays. 1992 Jul;14(7):455–463. doi: 10.1002/bies.950140705. [DOI] [PubMed] [Google Scholar]
- McKay I. A., Leigh I. M. Epidermal cytokines and their roles in cutaneous wound healing. Br J Dermatol. 1991 Jun;124(6):513–518. doi: 10.1111/j.1365-2133.1991.tb04942.x. [DOI] [PubMed] [Google Scholar]
- Murphy G., Cockett M. I., Ward R. V., Docherty A. J. Matrix metalloproteinase degradation of elastin, type IV collagen and proteoglycan. A quantitative comparison of the activities of 95 kDa and 72 kDa gelatinases, stromelysins-1 and -2 and punctuated metalloproteinase (PUMP). Biochem J. 1991 Jul 1;277(Pt 1):277–279. doi: 10.1042/bj2770277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olerud J. E., Gown A. M., Bickenbach J., Dale B., Odland G. F. An assessment of human epidermal repair in elderly normal subjects using immunohistochemical methods. J Invest Dermatol. 1988 Jun;90(6):845–850. doi: 10.1111/1523-1747.ep12462083. [DOI] [PubMed] [Google Scholar]
- Pellegrini G., De Luca M., Orecchia G., Balzac F., Cremona O., Savoia P., Cancedda R., Marchisio P. C. Expression, topography, and function of integrin receptors are severely altered in keratinocytes from involved and uninvolved psoriatic skin. J Clin Invest. 1992 Jun;89(6):1783–1795. doi: 10.1172/JCI115782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pentland A. P., Mahoney M., Jacobs S. C., Holtzman M. J. Enhanced prostaglandin synthesis after ultraviolet injury is mediated by endogenous histamine stimulation. A mechanism for irradiation erythema. J Clin Invest. 1990 Aug;86(2):566–574. doi: 10.1172/JCI114746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen M. J., Woodley D. T., Stricklin G. P., O'Keefe E. J. Synthesis and regulation of keratinocyte collagenase. Matrix Suppl. 1992;1:192–197. [PubMed] [Google Scholar]
- Porras-Reyes B. H., Blair H. C., Jeffrey J. J., Mustoe T. A. Collagenase production at the border of granulation tissue in a healing wound: macrophage and mesenchymal collagenase production in vivo. Connect Tissue Res. 1991;27(1):63–71. doi: 10.3109/03008209109006995. [DOI] [PubMed] [Google Scholar]
- Prosser I. W., Stenmark K. R., Suthar M., Crouch E. C., Mecham R. P., Parks W. C. Regional heterogeneity of elastin and collagen gene expression in intralobar arteries in response to hypoxic pulmonary hypertension as demonstrated by in situ hybridization. Am J Pathol. 1989 Dec;135(6):1073–1088. [PMC free article] [PubMed] [Google Scholar]
- Roman J., McDonald J. A. Expression of fibronectin, the integrin alpha 5, and alpha-smooth muscle actin in heart and lung development. Am J Respir Cell Mol Biol. 1992 May;6(5):472–480. doi: 10.1165/ajrcmb/6.5.472. [DOI] [PubMed] [Google Scholar]
- Saarialho-Kere U. K., Chang E. S., Welgus H. G., Parks W. C. Distinct localization of collagenase and tissue inhibitor of metalloproteinases expression in wound healing associated with ulcerative pyogenic granuloma. J Clin Invest. 1992 Nov;90(5):1952–1957. doi: 10.1172/JCI116073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saarialho-Kere U. K., Chang E. S., Welgus H. G., Parks W. C. Expression of interstitial collagenase, 92-kDa gelatinase, and tissue inhibitor of metalloproteinases-1 in granuloma annulare and necrobiosis lipoidica diabeticorum. J Invest Dermatol. 1993 Mar;100(3):335–342. doi: 10.1111/1523-1747.ep12470032. [DOI] [PubMed] [Google Scholar]
- Saarialho-Kere U. K., Welgus H. G., Parks W. C. Distinct mechanisms regulate interstitial collagenase and 92-kDa gelatinase expression in human monocytic-like cells exposed to bacterial endotoxin. J Biol Chem. 1993 Aug 15;268(23):17354–17361. [PubMed] [Google Scholar]
- Salo T., Lyons J. G., Rahemtulla F., Birkedal-Hansen H., Larjava H. Transforming growth factor-beta 1 up-regulates type IV collagenase expression in cultured human keratinocytes. J Biol Chem. 1991 Jun 25;266(18):11436–11441. [PubMed] [Google Scholar]
- Sarret Y., Woodley D. T., Goldberg G. S., Kronberger A., Wynn K. C. Constitutive synthesis of a 92-kDa keratinocyte-derived type IV collagenase is enhanced by type I collagen and decreased by type IV collagen matrices. J Invest Dermatol. 1992 Dec;99(6):836–841. doi: 10.1111/1523-1747.ep12614800. [DOI] [PubMed] [Google Scholar]
- Senior R. M., Griffin G. L., Fliszar C. J., Shapiro S. D., Goldberg G. I., Welgus H. G. Human 92- and 72-kilodalton type IV collagenases are elastases. J Biol Chem. 1991 Apr 25;266(12):7870–7875. [PubMed] [Google Scholar]
- Shapiro S. D., Doyle G. A., Ley T. J., Parks W. C., Welgus H. G. Molecular mechanisms regulating the production of collagenase and TIMP in U937 cells: evidence for involvement of delayed transcriptional activation and enhanced mRNA stability. Biochemistry. 1993 Apr 27;32(16):4286–4292. doi: 10.1021/bi00067a018. [DOI] [PubMed] [Google Scholar]
- Ståhle-Bäckdahl M., Parks W. C. 92-kd gelatinase is actively expressed by eosinophils and stored by neutrophils in squamous cell carcinoma. Am J Pathol. 1993 Apr;142(4):995–1000. [PMC free article] [PubMed] [Google Scholar]
- Ståhle-Bäckdahl M., Sudbeck B. D., Eisen A. Z., Welgus H. G., Parks W. C. Expression of 92-kDa type IV collagenase mRNA by eosinophils associated with basal cell carcinoma. J Invest Dermatol. 1992 Oct;99(4):497–503. doi: 10.1111/1523-1747.ep12616171. [DOI] [PubMed] [Google Scholar]
- Sudbeck B. D., Jeffrey J. J., Welgus H. G., Mecham R. P., McCourt D., Parks W. C. Purification and characterization of bovine interstitial collagenase and tissue inhibitor of metalloproteinases. Arch Biochem Biophys. 1992 Mar;293(2):370–376. doi: 10.1016/0003-9861(92)90408-o. [DOI] [PubMed] [Google Scholar]
- Unemori E. N., Hibbs M. S., Amento E. P. Constitutive expression of a 92-kD gelatinase (type V collagenase) by rheumatoid synovial fibroblasts and its induction in normal human fibroblasts by inflammatory cytokines. J Clin Invest. 1991 Nov;88(5):1656–1662. doi: 10.1172/JCI115480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welgus H. G., Fliszar C. J., Seltzer J. L., Schmid T. M., Jeffrey J. J. Differential susceptibility of type X collagen to cleavage by two mammalian interstitial collagenases and 72-kDa type IV collagenase. J Biol Chem. 1990 Aug 15;265(23):13521–13527. [PubMed] [Google Scholar]
- Welgus H. G., Jeffrey J. J., Eisen A. Z. The collagen substrate specificity of human skin fibroblast collagenase. J Biol Chem. 1981 Sep 25;256(18):9511–9515. [PubMed] [Google Scholar]
- Welgus H. G., Stricklin G. P. Human skin fibroblast collagenase inhibitor. Comparative studies in human connective tissues, serum, and amniotic fluid. J Biol Chem. 1983 Oct 25;258(20):12259–12264. [PubMed] [Google Scholar]
- Werb Z., Tremble P. M., Behrendtsen O., Crowley E., Damsky C. H. Signal transduction through the fibronectin receptor induces collagenase and stromelysin gene expression. J Cell Biol. 1989 Aug;109(2):877–889. doi: 10.1083/jcb.109.2.877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilhelm S. M., Collier I. E., Marmer B. L., Eisen A. Z., Grant G. A., Goldberg G. I. SV40-transformed human lung fibroblasts secrete a 92-kDa type IV collagenase which is identical to that secreted by normal human macrophages. J Biol Chem. 1989 Oct 15;264(29):17213–17221. [PubMed] [Google Scholar]
- Woodley D. T., Bachmann P. M., O'Keefe E. J. Laminin inhibits human keratinocyte migration. J Cell Physiol. 1988 Jul;136(1):140–146. doi: 10.1002/jcp.1041360118. [DOI] [PubMed] [Google Scholar]
- Woodley D. T., Kalebec T., Banes A. J., Link W., Prunieras M., Liotta L. Adult human keratinocytes migrating over nonviable dermal collagen produce collagenolytic enzymes that degrade type I and type IV collagen. J Invest Dermatol. 1986 Apr;86(4):418–423. doi: 10.1111/1523-1747.ep12285689. [DOI] [PubMed] [Google Scholar]