Abstract
The causes of epilepsies and epileptic seizures are multifactorial. Genetic predisposition may contribute in certain types of epilepsies and seizures, whether idiopathic or symptomatic of genetic origin. Although these are not very common, they have offered a unique opportunity to investigate the molecular mechanisms underlying epileptogenesis and ictogenesis. Among the implicated gene mutations, a number of GABAA receptor subunit mutations have been recently identified that contribute to several idiopathic epilepsies, febrile seizures, and rarely to certain types of symptomatic epilepsies, like the severe myoclonic epilepsy of infancy. Deletion of GABAA receptor genes has also been linked to Angelman syndrome. Furthermore, mutations of proteins controlling chloride homeostasis, which indirectly defines the functional consequences of GABAA signaling, have been identified. These include the chloride channel 2 (CLCN2) and the potassium chloride cotransporter KCC3. The pathogenic role of CLCN2 mutations has not been clearly demonstrated and may represent either susceptibility genes or, in certain cases, innocuous polymorphisms. KCC3 mutations have been associated with hereditary motor and sensory polyneuropathy with corpus callosum agenesis (Andermann syndrome) that often manifests with epileptic seizures. This review summarizes the recent progress in the genetic linkages of epilepsies and seizures to the above genes and discusses potential pathogenic mechanisms that contribute to the age, sex, and conditional expression of these seizures in carriers of these mutations.
Keywords: GABA receptor, Epilepsy, Mutation, Seizure, Chloride, Thalamus
Introduction
Genetic defects that manifest predominantly as seizures or epilepsies (a.k.a. “idiopathic epilepsies”) are not very common. They are known to occur in approximately 1% only of epilepsy patients. Genotype–phenotype studies and the increasing knowledge on the function of these genes have enriched our understanding of the pathophysiology of epilepsy and seizures. Most of the current linkage studies have revealed associations supporting the polygenic origin of idiopathic generalized epilepsies (IGEs). Genetic mutations seem to predispose but are not usually sufficient to cause epilepsy on their own. Among the implicated genes, are those involved in γ-aminobutyric acid (GABA) signaling, the principal inhibitory mechanism in the central nervous system. In this review, I will summarize the key components and features of the GABA signaling and describe the known channelopathies that perturb GABA signaling, either via direct effects upon GABAA receptors or through indirect effects on chloride transporters that ultimately impair GABAA signaling. Finally, I will also discuss how the functional consequences of these mutations are believed to explain the key features of the respective epileptic syndromes or seizures.
GABA signaling: basics on biology and physiology
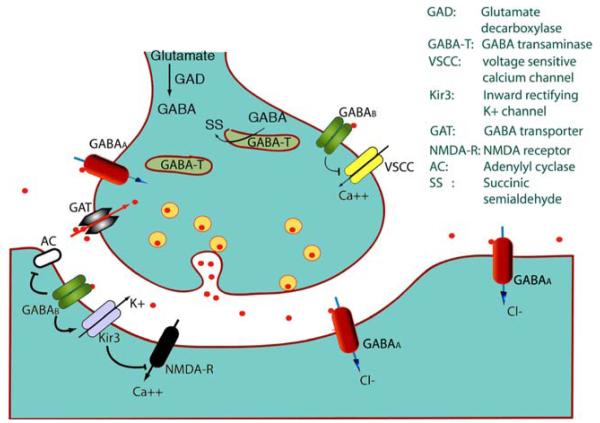
GABA is synthesized from glutamic acid, the principal excitatory neurotransmitter, through a decarboxylation reaction catalyzed by glutamic acid decarboxylase. GABA activates two major types of receptors (Fig. 1): (a) GABAA and the closely related GABAC receptors, which function as chloride channels; (b) the GABAB metabotropic receptors which are baclofen sensitive [1–3]. GABAA receptors are pentameric channels comprised of different combinations of the following subunits: α (1–6), β (1–3), γ (1–3), δ, ε, θ, π. These pentameric complexes consist of 2 α and 2 β subunits, whereas the fifth subunit can belong to any of the other classes. The exact combination of these subunits determines the pharmacologic and pharmacokinetic properties, including agonist profile, affinity for allosteric modulators or antagonists, and subcellular localization. The GABA binding site is formed by the α and β subunits; hence, two of them can be present in each receptor complex. Benzodiazepine binding sites are bordered by an α and a γ subunit and therefore again two of them, at most, can be present in each receptor. The δ subunits are usually present in extrasynaptic GABAA receptors, which are activated by ambient GABA and mediate tonic GABAA responses. The presence of α1 subunit confers sensitivity to drugs like zolpidem and accelerates the kinetics of GABAA receptor opening. GABAC receptors are bicuculline-insensitive chloride channels defined by the presence of a ρ subunit (ρ1–ρ3); however, in the recent classifications they are categorized as subtypes of GABAA receptors [3].
Fig. 1.
ABA signaling. Release of GABA through synaptic vesicles activates GABA receptor Cl– channels (GABAA or GABAC) and metabotropic GABAB receptors, which are either pre- or postsynaptic. GABAA and GABAC receptors hyperpolarize the neurons via influx of Cl–. GABAB receptors can inhibit adenyl cyclase (AC); may activate inward rectifying potassium channels (Kir), or may inhibit voltage sensitive calcium channels (VSCC). The released GABA can then be uptaken to the presynaptic compartment via GABA transporters (GAT)
Whether through phasic or tonic conductance, GABAA receptors mediate the principal inhibitory function in the brain. Their function, however, changes depending upon the specific conditions that control the chloride gradient [1, 2, 4]. When chloride accumulates intracellularly, GABAA receptor activation has depolarizing effects. This is classically the case in immature neurons, in which the expression or activity of chloride transporters that favor the import of Cl– into the cell, such as the sodium potassium chloride cotransporters (NKCCs), override the effects of chloride exporting proteins, like the potassium chloride cotransporters (KCCs). In every neuron, there is a gradual change in Cl– gradient during development, attributed to a gradual switch from NKCC to a KCC dominant state, which leads to low intracellular Cl– and allows the hyperpolarizing effects of GABAA receptors to manifest. The importance of KCCs in promoting this shift was elegantly demonstrated by Rivera et al. who showed that KCC2 activity is necessary for the appearance of hyperpolarizing GABAA signaling in hippocampal neurons [5]. KCC2 has been one of the most studied chloride cotransporters in regard to its role in GABAA receptor function and seizures, and its importance in both GABAA signaling and brain development has been well documented [1, 2, 6–9]. However, KCC2 mutations have not yet been identified in patients with epilepsy or seizures. It is possible that disruption of KCC2 may result in early mortality, similar to what has been observed in mice [10], precluding the identification of KCC2 mutation carriers. In contrast, KCCs that may have more restricted expression or may not be as essential for survival may result in an identifiable phenotype. In this review, reference will be made to KCC3 mutations that have been linked with seizure disorders.
GABAB receptors are metabotropic receptors, consisting of seven transmembrane domains. They are localized at pre-, post-, or extrasynaptic sites as functional heterodimers. Homodimers of R1 or R2 GABAB receptor subunits have been described with distinct intracellular localization and can under certain conditions be directed to the plasma membrane [11–13]. Although there is no evidence currently that homodimers are functionally active, atypical responses to GABAB receptor agonists have been described in the GABAB R2 knockout mouse [13]. By forming heterodimers, they can inhibit calcium channels (presynaptic effects) and block as a result the presynaptic release of neurotransmitters, whether these may be inhibitory (i.e. GABA) or excitatory (i.e. glutamate). In the postsynaptic, and possibly presynaptic, compartment they may also activate inward rectifying potassium channels (Kir), hyperpolarizing the neurons, and generating inhibitory postsynaptic potentials (IPSCs). Furthermore, they may inhibit adenylate cyclase postsynaptically influencing cAMP-dependent cell signaling [14].
Currently, there has been significant progress in genetic linkage studies associating seizure or epilepsy disorders with specific components of the GABA signaling pathways. In this review, I will focus on the GABA channelopathies that are linked to increased susceptibility to human epilepsies and seizures. Specifically, I will describe the human mutations of GABAA receptors, as well as on mutations of chloride transporters and channels that perturb chloride homeostasis and disrupt GABAA signaling, leading to epileptogenesis.
Epilepsy syndromes and seizures associated with GABAA receptor mutations
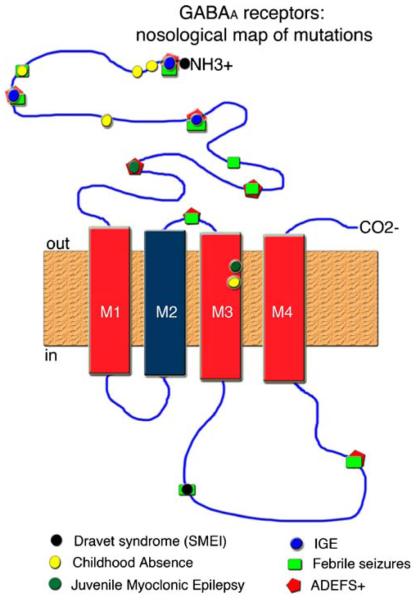
The known mutations of GABAA receptors have been linked to three types of idiopathic epilepsies: childhood absence epilepsy (CAE), autosomal dominant epilepsy with febrile seizures plus (ADEFS+), and autosomal dominant Juvenile Myoclonic Epilepsy (JME). Sporadic cases of severe myoclonic epilepsy of infancy (Dravet syndrome or SMEI) and association with febrile seizures have also been documented. Finally, deletions of chromosomal loci carrying GABAA receptor subunit genes occur in conditions manifesting with seizures, like the Angelman syndrome. These are discussed below, and the map of the known mutations on the structure of GABAA receptors is presented at Fig. 2.
Fig. 2.
Nosological mapping of GABAA receptor mutations. Distribution of the known mutations of GABAA receptors that have been linked with seizures or epilepsies. The majority of mutations are located at the extracellular segments (amino-terminal and M2–M3 linker) or the intracellular M3–M4 linker. Rarely, they involve the M3 transmembrane domain
Childhood absence epilepsy
CAE manifests in childhood (onset at 4–8 years of life) with frequent staring spells associated with bursts of 3-Hz spike and slow-wave complexes, which classically respond to blockers of the low-threshold calcium channels (T channels) like ethosuximide. Two thirds of CAE patients are girls. The first association of GABAA receptor subunits with CAE was proposed by Feucht et al. [15] who reported greater incidence of specific alleles of the β3 subunit in patients with CAE (9/48 patients) compared with normal controls (11/96 subjects). Among the genes that have been linked so far with CAE are mutations in the α1, α6, and β3GABAA receptor subunits (Table 1).
Table 1.
GABAA receptor mutations in epilepsy and seizures
| Seizure type / epilepsy |
Mutation | Type of mutation | Site of mutation | Functional effect of mutation | Reference |
|---|---|---|---|---|---|
| Autosomal dominant JME |
GABRA1 (A322D) | Missense | M3 domain | Low amplitude GABA currents reduced surface expression; increased GABA EC50 |
[56,57] |
| GABRD (R220H | Missense | Extracellular domain |
Low amplitude GABA currents; impaired trafficking? |
[46] | |
| CAE | GABRA1 (975delC; S326fs328X) |
Frameshift mutation Premature termination |
M3 domain | Nonsense-mediated mRNA decay; degradation in ER |
[16,17] |
| GABRA6 (R46W) | Missense | Extracellular domain |
No change in GABAA current | [18] | |
| GABRB3 (–897T/C polymorphism) |
Promoter region | Decreased transcription (impaired N-Oct-3 binding) |
[19] | ||
| GABRB3 (P11S; S15F; G32R) |
Missense | Extracellular domain |
Hyperglycosylation? | [20] | |
| GABRB3 | Association analysis | [15] | |||
| CAE + febrile seizures |
GABRG2 (R43Q) | Missense | Extracellular domain |
Decreased surface expression; ER retention; Altered benzodiazepine / GABA binding? |
[24,57,126] [26,27,32,34] |
| (IVS6 + 2 T<G) | Exon 6 skipping Premature termination |
M1–M2 linker |
Exon skipping, premature termination ER retention and degradation vs nonfunctional protein |
[37] | |
| ADEFS+ | GABRG2 (K289M) | Missense | M2–M3 linker (extracellular) |
Decreased GABAA current amplitude; Intact DZ potentiation |
[38] |
| GABRG2 (W390X) | Premature termination |
M3–M4 linker (intracellular) |
? | [127] | |
| GABRD (E177A) (R220C) |
Missense | Extracellular domain |
Low amplitude GABA currents impaired trafficking?; |
[46] | |
| GABRA4 (T320A) | Missense | ? Also seen in controls |
[18] | ||
| IGE, ADEFS+ febrile seizures |
GABRP (V10M) | Missense | Extracellular domain |
? | [18] |
| GABRE (Y38S; E52K) |
Missense | Extracellular domain |
? Also present in controls |
[18] | |
| SMEI ADEFS+ | GABRG2 (Q351X) | Premature termination |
M3-M4 linker (intracellular) |
ER retention | [42, 121] |
| SMEI | GABRG2 (Q40X) | Premature termination |
Extracellular domain |
ER retention | [49] |
| Febrile seizures | GABRG2(R139G) | Missense | Extracellular domain |
Increased fast phase desensitization, reduced sensitivity to diazepam |
[44] |
| GABRE (G66S) | Missense | Extracellular domain |
? | [18] | |
| Angelman syndrome | 15q11-13 deletion | Deletion, maternally inherited |
15q11-13 | Decreased expression of involved genes (may include GABRB3, GABRA5, GABRG3) |
[60] |
GABRA1 (975delC; S326fs328X)
Screening of 98 patients with IGEs (38 had CAE) identified a deletion of a single base pair at position 975 (975delC) of the α1 subunit, which was not seen in three unaffected relatives [16]. This deletion causes a frameshift and premature termination within the third transmembrane domain (M3) of the α1 subunit [16]. Transfection of wild type (α1-WT) or α1975delC constructs labeled with enhanced green fluorescent protein (EGFP) in human embryonic cells (HEK293) resulted in the retention of the truncated α1975delC subunit within the cytoplasm and endoplasmic reticulum (ER) [16]. The inability of the truncated protein to be inserted into the plasma membrane is probably attributed to the propensity of its mRNA to be degraded through the nonsense-mediated mRNA degradation pathway (NMD), as well as to the protein degradation through the ER-associated degradation pathway (ERAD) [17]. Concurring with the absence of this mutant from the cell membrane, no GABAA currents were recorded in these α1975delCβ2γ2 transfected cells [16]. Coexpression of α1975delC with α1-WT did not reveal any dominant negative effect [16].
GABRA6 (R46W)
A missense mutation of the extracellular segment of this subunit was found in a patient with CAE with atonic seizures but without any evidence of pathological alteration of GABAA currents [18].
GABRB3 (–897 T/C polymorphism)
Among 45 patients with CAE, 28 manifested a polymorphism in the promoter of the exon 1a of the GABRB3 subunit (–897 T/C; haplotype 2) that reduced the affinity for binding of the neuron-specific transcriptional activator N-Oct-3 [19]. Although haplotype two was not restricted to CAE patients (also present in 10/90 control samples), it increased the risk for CAE. Functionally, this haplotype reduces the expression of the GABRB3 subunit.
GABRB3 (P11S; A15F; G32R)
Screeningof 48families of probands with remitting pyknoleptic absence produced associations with three missense mutations at positions 11, 15 (exon 1a), and 32 (exon 2) of the β3 subunit in members of four families [20]. The probands had absence seizures that could be associated with eyelid myoclonias (P11S, S15F) or grand mal seizures (S15F) or atonic seizures and vomiting (P11S). Incidental presence of the G32R mutation was also found in two siblings of the proband who had 3–4 or 5–6 Hz spike-wave complexes in their EEG with or without prior history of febrile seizures [20]. Asymptomatic carriers were also found among relatives of the proband with the S15F mutation.
In vitro translation of the mutated GABRB3 yielded proteins with excessive glycosylation and reduced amplitudes and densities of GABAA currents in transfected cells, suggesting a decreased number of functional GABAA receptors at the plasma membrane [20]. Whether this is due to impaired trafficking or hypofunctional GABAA receptor complexes remains to be shown.
Febrile seizures and autosomal dominant epilepsy with febrile seizure plus syndromes
Seizures in the context of a febrile illness (i.e. febrile seizures) are very common in the infantile and childhood years, occurring in approximately 2–5% of children. Febrile seizures do not typically raise the risk of subsequent epilepsy, unless they occur as focal, prolonged or clusters of seizures, or when they are associated with neurodevelopmental abnormalities or a family history of epilepsy [21]. A subgroup of these families demonstrates an autosomal dominant mode of inheritance [21, 22]. Reports of cosegregation of patients with different types of idiopathic generalized or focal epilepsies and/or history of febrile seizures within the same family pedigrees emerged over the past decade, bringing onto light a number of susceptibility genes. Initially, the term Generalized Epilepsy with Febrile Seizures Plus syndrome was given as a descriptor [23]. However the term Autosomal Dominant Epilepsy with Febrile Seizure Plus Syndromes has also prevailed, as it was recognized that both focal onset and generalized afebrile seizures could manifest within these pedigrees, and the trait was inherited in an autosomal dominant mode. Although the most common gene mutations are sodium channelopathies, a number of GABAA receptor mutations have also been implicated, as described below.
Mutations linked with CAE and febrile seizures syndrome
GABRG2 (R43Q)
One of the first associations of GABRG2 mutations and IGEs was reported by Wallace et al. [24] in a large pedigree of patients with CAE and febrile seizures with or without other types of seizures. Most of the affected members carried the γ2R43Q mutation. Asymptomatic carriers were also found in older generations of this pedigree. In parallel, the same pedigree also included members who had either EEG abnormalities or other seizure types without carrying the γ2R43Q mutation. The involvement of compensatory factors that suppress the expression of seizures in selected subjects is therefore suggested, whereas the influence of genetic or epigenetic factors modifying the phenotype cannot be excluded.
A recent knockin mouse constructed to express γ2R43Q provided a proof of principle that heterozygous expression of this mutation is sufficient to induce behavioral arrests with bursts of spike-wave complexes (5–8 Hz) in the majority of mice [25]. These seizures first appeared around postnatal day 20 (equivalent to human childhood) and were suppressed by ethosuximide, similar to absence seizures [25]. The genetic background modified the probability to express these seizures, explaining the variable expression of seizures among carriers [25].
Several studies have addressed the effects of γ2R43Q mutation not only on the basic electrophysiological properties of the GABAA receptor complexes, but also upon benzodiazepine binding, receptor trafficking, and complex formation. A uniform finding is the decreased trafficking of the γ2R43Q subunit to the cell surface, due to its retention and possible degradation within the ER [25–28]. Such studies utilized coexpression with the α1β2 or α1β3 subunits. In cell-culture experiments using heterologous transfections, a dominant negative effect has also been reported. The γ2R43Q mutation reduced the surface expression of the β2 subunit [27], α1β2 complexes [29], a5 subunits [28], and re-routed the expression of γ2or pentameric α1β2γ2S or α1β2γ2SR43Q receptor complexes to perinuclear and ER sites [30]. However in embryonic cultures from γ2R43Q knockin mice, no changes in the trafficking and localization of α1 subunits were seen [25], raising the possibility that in vivo conditions may modify the dynamics of subunit interactions and trafficking. These may include post-translational modifications, compensatory expression of other subunits, or regulatory factors. Indeed, even in the same model system of γ2R43Q knockin mouse, regional differences in the effects of GABAA receptors were observed. The cortical pyramidal (layers II/III) neurons of γ2R43Q knockin mice manifested the greatest impairment in GABAA neurotransmission, whereas GABAA function in the thalamic nuclei was unaffected [25]. This generated the hypothesis that cortical hyperexcitability may underlie the pathogenesis of absence seizures in these mice. In agreement, increased cortical excitability was also seen in human patients with the γ2R43Q mutation, using transcranial magnetic stimulation [31]. Despite the significant reduction in surface availability of γ2R43Q containing GABAA receptor complexes, a small number of assembled complexes eventually escape intracellular degradation forming complexes. As a result of the decreased number of functional membrane GABAA receptors, IPSC amplitudes are smaller in most studies.
R43Q is located at the γ2/β subunit interface, in the proximity of the first high-affinity benzodiazepine binding site [27]. Although it does not seem to be directly involved in benzodiazepine or GABA binding, it may indirectly affect them, in certain model systems, possibly through conformational changes [24, 28, 29, 32, 33]. However, this has not been supported by other studies [26, 27]. Interestingly, in humans, decreased benzodiazepine receptor binding has been found with (11C)-flumazenil positron emission tomography in insular and anterior cingulated cortex [34], although this may probably reflect the reduced overall membrane expression of GABAA receptors.
Apart from altering synaptic physiology, the γ2R43Q mutation has also been proposed to influence brain plasticity [35]. Conditional suppression of this allele in knockin mice from conception till PN20 decreases the susceptibility to pentylenetetrazole seizures in adulthood, when compared to mice with life-long expression of γ2R43Q allele. A plausible proposed explanation is that disruption of the physiological effects of GABAA receptors during the sensitive development fetal and neonatal/ infantile life may compromise the developmental processes that are crucial for normal brain development and facilitate, possibly, the development of epilepsy [1, 4, 35, 36].
GABRG2 (IVS6+2T>G)
A splice donor site mutation in intron six of the GABRG2 subunit was identified in a family with CAE and febrile seizures [37]. This is predicted to cause the skipping of exon 6, creation of a premature stop codon at the junction of exons 5 and 7, and nonfunctional truncation upstream of the transmembrane domain M1.
Mutations linked with ADEFS plus syndrome
GABRG2 (K289M)
A missense mutation K289M, at the linker of M2 and M3 domains, was identified in members of a family with generalized tonic–clonic epilepsy and febrile seizures, who were therefore classified as autosomal dominant epilepsy with febrile seizure plus syndrome (ADEFS+) [38]. The K289M mutation causes significant reduction in the amplitude of the GABAA currents and shortening of the receptor open time, whereas benzodiazepine sensitivity is retained [26, 38–40]. Interestingly, phenobarbital potentiates α1β2γ2K289M receptors to a greater extent than wild-type receptors improving therefore GABAA inhibition [41].
GABRG2 (Q351X)
In a French family with ADEFS+ phenotype a missense mutation Q351X was identified [42]. Heterologous expression of GABAA receptors with this subunit did not produce any functional chloride channels due to intracellular retention of the mutated subunits. This mutation is predicted to cause premature termination between M3 and M4 domains.
GABRG2 (W390X)
The W390X nonsense mutation causes premature termination between M3 and M4 domains. It was identified in a Chinese family with several members with febrile seizures with or without other types of seizures [43].
GABRG2 (R139G)
A missense mutation at the extracellular segment has been linked with febrile seizures [44]. Functionally, the receptors had increased desensitization and reduced sensitivity to diazepam.
GABRD (E177A; R220C)
GABRD containing receptors are typically extrasynaptic and mediate tonic GABAA inhibition after being activated by ambient GABA [45]. Missense mutations E177A and R220C were identified in families with ADEFS+ or febrile seizures [46]. The electrophysiological effect of the E177A mutation is the reduction of the peak current under saturated doses of GABA without changing binding affinity; decreased open times and reduced cellular expression have been proposed as possible causes for this deficit [47].
Screening of patients with ADEFS+, IGEs or febrile seizures for mutations or polymorphisms at the known GABAA receptor subunit genes revealed rare additional missense mutations at the GABRP (V10M) and GABRE (Y38S, E52K, G66S), GABRA4 (T320A) genes. Rare occurrence in control patients was also reported for the GABRA4 (T320A) and GABRE (Y38S, E52K) mutations [18]. The functional and pathogenic significance of these mutations is currently unclear.
Severe myoclonic epilepsy of infancy (Dravet syndrome; SMEI)
SMEI or Dravet syndrome manifests in the first year of life with clusters or prolonged febrile seizures (clonic, tonic, and generalized tonic–clonic), which can be difficult to suppress and frequent evolution to other epilepsies and developmental problems [48]. The known GABAA receptor-related mutations that have been linked so far with this condition are nonsense mutations, leading to premature termination of the γ2 (Q351X or Q40X) subunits; ER retention reduced expression of these subunits in the cell surface.
GABRG2 (Q351X)
An isolated case of a patient with SMEI within an Austrian family with ADEFS+ linked to a Q351X nonsense mutation of the γ2 subunit was reported [42]. Other carriers of the same mutation had milder seizure types.
GABRG2 (Q40X)
This nonsense mutation has been reported in patients with SMEI, causing premature termination, ER retention, and reduction in the surface expression of the γ2 subunit [49]. Asymptomatic carriers were also found within this family.
Autosomal dominant juvenile myoclonic epilepsy
Juvenile myoclonic epilepsy is an autosomal dominant form of IGE that typically begins when the child approaches the teenage years. The classical seizure type is the myoclonic jerks upon awakening, whereas grand mal seizures (almost in all patients) and absence seizures (in a third of patients) are also very common [50]. JME can appear within the spectrum of ADEFS+ and therefore can be linked to the mutations discussed above. However, two specific GABAA receptor mutations have been associated specifically with JME.
GABRA1 (A322D)
The A322D mutation is located within the M3 transmembrane domain, close to a region that is thought to undergo a conformational change upon binding of agonists or allosteric modulators of GABAA receptors [51–53]. Heterologous expression of the missense mutation α1A322D in HEK293 cells produces hypofunctional GABAA receptor channels (α1A322Dβ2γ2). In transfection experiments in HEK293T cells, differences in the properties and kinetics of the assembled channels were found under conditions favoring the formation of wild-type, heterozygous, or homozygous GABAA receptors. Heterozygous channels had reduced current amplitudes yet similar activation, desensitization, and deactivation kinetics with the wildtype channels. In contrast, homozygous channels had accelerated deactivation and resensitization kinetics, significantly reduced open times and affinity for GABA, as well as very low current amplitudes [54–57]. Reduced whole-cell and cell-surface expression of the mutated channels has been proposed to explain the reduced amplitudes [57].
GABRD (R220H)
Heterozygous or homozygous δR220H mutations were found in a family with JME, as well as sporadically in control patients [46]. This mutation is present at the extracellular domain of the GABAA receptors and results in low amplitude GABAA currents, due to reduced open times and, to a lesser extent, decreased expression of the receptor onto the cell membrane [47]. However, the mutation effect upon the membrane expression of the receptors was rescued by the presence of a wild-type allele [47].
Angelman syndrome
The Angelman syndrome appears in the first year of life, with an estimated frequency of one in 15,000 births [50]. Infants with normal development in the first months of life manifest developmental delay, followed by more permanent intellectual and developmental disabilities, including speech, gait, and balance impairments, hypotonia, epileptic seizures (infantile spasms, myoclonic seizures and absences, tonic or tonic–clonic seizures, and partial seizures), episodes of arm flapping, tongue thrusting, and head drops, yet they have a happy appearance and paroxysms of laughter [50, 58]. Their EEGs often demonstrate slow, high amplitude spike- and slow-wave complexes (SWDs) or dysrhythmia (rhythmic slowing frontal or occipital or diffuse) [59]. In the majority of cases, a maternally inherited deletion of 15q11-13 locus is identified, which includes the UBE3A gene (ubiquitin-protein ligase E3A), as well as three GABAA receptor subunit genes: the GABRB3, GABRA5, and GABRG3 [60–62]. Minassian et al. correlated genotype with phenotype in patients with Angelman syndrome and proposed that patients with 15q11-13 deletion, that included the deletion of GABR genes in addition to UBE3A, follow a more severe course than patients with UBE3A mutations [63]. Patients with 15q11-13 deletion often had flexor or extensor epileptic spasms in addition to atypical absences or myoclonus [63]. In contrast, patients with UBE3A mutations have only a mild phenotype.
Although the GABRB3–/– knockout mice have additional characteristics, related to the GABA receptor deficit, such as cleft palate and decreased viability, the survivors develop the neurodevelopmental deficits and epilepsy phenotype of Angelman syndrome: neurodevelopmental impairment, stereotypical behaviors, such as running in circles, hyperactivity, impaired motor development, abnormal electrographic patterns, and seizures [64]. Seizures include myoclonic jerks and seizures, head drops, opisthotonus and tail arching, sudden drops, and wild running episodes. The brains of GABRB3–/– knockout mice also showed significant widespread reductions in the binding affinity for both direct and benzodiazepine-like GABAAergic agonists. The authors proposed that the deficit in β3 may also decrease other non-β3-containing, zolpidem-insensitive, GABAA receptors [64]. Decreased sensitivity to certain anesthetics, like midazolam and etomidate, has been described in the GABRB3–/– knockout mice [65]. An interesting sex-specific pattern of epigenetic modification of the β3gene was reported in the β3+/– heterozygotes, where decreased β3 expression was seen in all females, regardless of the parental origin of the affected allele, whereas males that demonstrated low β3 levels only in the affected allele were of maternal origin [66].
Clinical and pathophysiologic implications of GABAA receptor mutations in epilepsies
Pathophysiological role of GABAA receptors in IGEs
Most of the identified GABAA receptor mutations linked with IGEs reduce GABAAergic transmission. In IGEs, SWD generation occurs within the corticothalamic network and is intimately controlled by GABAergic neurons at multiple levels (Fig. 3) [67–69]. To explain the pathogenic role of GABAA receptor mutations, one has to speculate that they reduce GABAAergic input and therefore disinhibit the corticothalamic or nucleus reticularis thalami (nRT) neurons. Indeed, Tan et al. demonstrated that, in the γ2R43Q knockin mouse model of absence epilepsy, the mutation produces a small reduction of the GABAAergic inhibition of cortical neurons, whereas the GABAAergic input to the thalamic neurons remains intact [25]. The authors proposed that this small degree of cortical disinhibition may be pathogenetically linked to the generation of absence seizures.
Fig. 3.
Corticothalamic network and spike-wave complex generation. Panel a: The corticothalamic network is the primary network involved in the generation of spike-wave discharges (SWD) of idiopathic generalized epilepsies, like absence seizures. The thalamocortical relay neurons (TC) receive afferent sensory excitatory input and subsequently activate the cortical pyramidal neurons (layers III/IV and V/VI) and adjacent GABA interneurons (red) (step 1). This is further spread to adjacent cortical neurons generating a burst of activity that through the layer VI cortical neurons propagates back to the thalamus, activating the nucleus reticularis thalami (nRT) GABAergic neurons and local thalamic GABAergic interneurons (step 2). The nRT activation inhibits the activity of TC neurons via a fast GABAA and a slow GABAB inhibitory component (step 3) (Panel b). The hyperpolarizing effect of GABA-inhibition of TC neurons activate hyperpolarization-activated cation channels (HCN) and subsequently low-threshold activated calcium channels (T channels) that generate a calcium spike and subsequently triggers a burst of action potentials (Na+/K+ AP) that re-activates the circuit. This sequence of events is detected by the surface EEG as spike (i.e. burst of action potentials) and slow wave (i.e. hyperpolarization phase) discharges
Implications for age and sex-specific features of IGEs
IGEs demonstrate a classical, defining for each syndrome, age-specific pattern of expression. This may correspond potentially to the ages when the presence of a pathogenic mutation of the specific GABAA receptor subunits directly or indirectly (i.e. through dominant negative effects or interactions with other subunits or pathways) weakens the intrinsic defense mechanisms that control seizures. Three of the GABAA receptor subunits discussed here, α1, β3, and γ2, exhibit distinct developmental patterns of expression, based on studies in rodents, yet all three have been implicated in CAE. In the corticothalamic network, α1 and γ2 increase during the infantile and juvenile periods, whereas β3 declines [19, 70, 71]. It is therefore also possible that the ages of vulnerability to childhood absence seizures may coincide with the developmental periods when rapid changes occur in the expression of these subunits. As a result of these dynamic changes in gene expression, the brain may not be able to fully compensate for deficiencies in the affected subunit, by increasing the availability of other subunits. Resolution of absence seizures may then occur either because the biological role of the affected subunit in the corticothalamic network may, normally, diminish at older ages (as postulated for the β3 subunit [19]) or because increase in other subunits or the wild-type alleles may eventually compensate.
GABAA receptors are also key regulators of seizure susceptibility, whether these are generalized or focal, by influencing the activity of subcortical centers, like the substantia nigra, that control seizure propagation [72–74]. The immaturity of the GABA-sensitive seizure controlling subcortical networks, early in life, is an important factor for the age-related vulnerability to certain seizure types, like absence seizures. The presence of the above GABAA receptor mutations may further deteriorate their ability to curtail seizures, leading to epilepsy. Further studies are needed to elucidate the impact of these mutations in the function of these subcortical structures.
IGEs may also show preferential expression in one sex. Two thirds of CAE patients are girls whereas a trend for higher incidence in boys is seen with ADEFS+ and grand mal on awakening [75]. It is therefore not very surprising that GABAA receptors, well-known for its age- and sex-specific patterns in expression and function, are involved in the susceptibility to these syndromes [70, 76–80]. As the signature of IGEs is the SWDs on the EEG (Fig. 3), changes in GABAA receptor expression and function within the corticothalamic network, the generator of SWDs, have been a focus of attention. In a study utilizing Long–Evans rats, lower developmental expression of the α1and γ2subunits was seen in the cortex and thalamus of female rats, that correlated with higher frequency of SWDs in females following treatments that induce SWDs in these rats, i.e. perinatal administration of the cholesterol synthesis inhibitor AY9944 [70, 81]. Moreover, AY9944 reduced the expression of these subunits in the female, but not the male thalamus [70]. Such findings are in support of the hypothesis that decreased availability of these two subunits, either via loss-of-function mutations or by impaired trafficking, may increase the susceptibility to CAE, and that this vulnerability may be greatest in girls. However, the rarity of these mutations does not allow definite conclusions yet as to their contribution in the sex-specific prevalence of these disorders.
An alternative mechanism underlying the sex-specific prevalence of epilepsies linked to β3 mutations was presented by Liljelund et al. [66], who reported sex-specific imprinting of the β3 subunit in heterozygote GABRB3+/– mice. In female heterozygotes, β3 subunit expression was significantly reduced whether the affected gene was inherited by the father or the mother. In contrast, in male heterozygotes, significant reduction was noted only through maternal transmission of the affected gene. Genomic imprinting of the paternal chromosome 15 is known to also occur in human patients with Angelman syndrome. In these cases, however, paternal imprinting explains the preference for transmission through the maternally transmitted affected allele, without contributing to any sex-specific prevalence of the disease [82, 83].
Beyond the immediate consequences of impaired GABAA receptor signaling due to the above mutations, there are wider and long-term implications relating to disruption of GABA-dependent developmental processes that shape the normal and sex-appropriate brain development [1, 4, 9, 76, 84–88]. GABAA signaling controls morphogenesis, neuronal migration and differentiation, as well as synaptogenesis. Early life events that alter GABAA signaling may leave their long-term imprints on the way the brain works and responds to the environment. An elegant proof-of-principle example was offered by Chiu et al. who showed that transient suppression of the expression of the γ2R43Q, early in life, has lasting changes by decreasing susceptibility to seizures in adulthood compared to mice with constant expression of the mutant allele [35].
Fever, GABAA receptor mutations and seizures
An attractive theory as to the role of fever in unmasking the seizure vulnerability of patients with specific GABAA receptor mutations was proposed by Kang et al., based on a temperature-sensitive impairment in intracellular trafficking [89]. Specifically, a rapid increase in temperature from 37 to 40°C within 10 min impaired trafficking or decreased endocytosis of γ2R43Q subunits, but did not affect subunits with mutations not linked to febrile seizures, like the α1A322D. However, febrile seizures can often occur at the onset of a febrile illness, with minor changes in temperature, whereas in epilepsies like ADEFS+ febrile but also afebrile seizures may occur in the family pedigree. Additional factors need therefore, be invoked to explain susceptibility to febrile seizures and epileptogenesis in such instances. Possible culprits may be immune-related factors, such as cytokines, which increase excitability and excitotoxic injury [90–92]. A genetic background with deficient GABAA inhibition due to the above mutations may therefore, augment the brain excitability due to inflammatory factors and lead to seizures and subsequent epilepsy.
Other genes modulating seizure susceptibility
Chloride channel 2 (CLC-2)
Chloride channel 2 (CLC-2) is a hyperpolarization-activated, inward rectifying Cl– channel that activates at membrane potentials more negative to the reversal potential for Cl– (ECl) [93, 94]. As a result, when Cl– loading occurs through the repetitive activation of hyperpolarizing GABAA receptors, the activation of ClC-2 leads to compensatory efflux of Cl–, maintaining the intracellular Cl– concentrations low. This permits the hyperpolarizing actions of GABAA receptors. The developmental increase in CLC-2 has been proposed as one of the factors that contribute to the gradual decline in ECl in neurons and the appearance of hyperpolarizing GABAA currents [95, 96]. The localization of its gene (CLCN2) within the 3q24 chromosomal region, previously linked to IGEs, prompted the screening of patients with IGEs for mutations in CLCN2. Mutations in CLCN2 have been found in several patients with IGEs, including JME or Juvenile absence epilepsy (JAE) [97, 98] or focal epilepsy [98]. In a study of 112 patients with such epilepsies and 192 control subjects, mutations present in the epilepsy cohort only were found in 1.7% subjects [98].
CLCN2 (IVS17-3C> T)
This single nucleotide polymorphism (SNP) was found in a small IGE family, including an unaffected parent. However, there is no evidence for a pathogenic role for this mutation, as no change in splicing or expression of CLC-2 was identified [98].
CLCN2 (R688G, E718D)
The R688G and E718D missense mutations were found in a father and a son who had absence and generalized seizures or frontal lobe epilepsy, respectively [98]. The affected amino acids are located within the carboxy-terminal segment of the CLC. Based on their location, it has been proposed that they interfere with voltage gating [98]. However, this was not confirmed by subsequent studies [99].
In a different study of three families of patients with epilepsy, three additional mutations were found, that were not present among 4,700 control patients [97]. These were, however, not restricted to patients with epilepsy but were also sporadically present in their asymptomatic relatives, suggesting that they may be susceptibility mutations [97].
CLCN2 (Gins597; p.M200fsX231)
The p.M200fsX231 mutation leads to premature termination of CLC-2, rendering it nonfunctional. It was detected in a patient with JME, as well as in asymptomatic relatives [97]. Coexpression of the wild type and the M200fsX231 mutant did not reveal any dominant negative effect upon the gating of the coexpressed wild-type CLC-2 channels [99, 100].
CLCN2 (IVS2-14del11)
Atypical splicing IVS2-14del11, that predicts the deletion of 44 amino acids (del74-117) and skipping of helix B of CLC-2, was found in a patient with IGE, as well as some of his asymptomatic relatives [97]. Such a deletion should result in nonfunctional channels [100, 101]. However, expression of a minigene with the IVS2-14del11 mutation in N2a neuroblastoma cells produced both normally spliced products, as well as mRNAs bearing the expected deletion, suggesting that its pathogenicity may either not be significant or be cell-type selective [100].
CLCN2 (G715E)
A missense mutation G715E was found in two siblings with JAE or spike-wave activity in the EEG, as well as their father [97]. This mutation occurs at the cytoplasmic carboxy-terminal ending of the protein, between the two cystathionine-β-synthase domains CBS1 and CBS2 [97, 100, 101]. Although initial studies suggested that it may alter the voltage-gating properties of the channel [101], this was not confirmed [100]. Independent laboratories showed that the G715E mutant of the CLC-2 channel exhibits decreased sensitivity to intracellular adenosine nucleotides, raising the possibility that the mutant allele may impair the activation of the CLC-2 channel under conditions of energy depletion (i.e. low ATP, increased AMP levels) [100, 102]. For example, the binding affinities for ATP or AMP are reduced at least tenfold in the G715E mutant [102]. Furthermore, replacement of intracellular ATP by AMP does not accelerate the kinetics of gate opening in the G715E CLC-2 channel as it does in the wild-type channel [100]. However, it remains to be shown whether such aberrations from normal function are important enough to generate a pathogenic phenotype, at least under conditions of energy depletion.
Overall, the involvement of CLCN2 mutations in idiopathic generalized and/or focal epilepsies is unclear and may represent innocuous mutations. At best, these may be considered as susceptibility gene mutations, rather thanpathogenic, especially if one considers that most of these patients with epilepsy carry heterozygous CLCN2 mutations, which do not compromise the functional role of the wild-type CLC-2, at least under normal conditions. Whether this balance can be tipped, leading to epilepsy, in the presence of additional pathogenic or susceptibility mutations, under specific pathological conditions, or during vulnerable developmental periods when the expression of wild-type CLC-2 is still low and unable to fully compensate for haploinsufficiency states due to loss-of-function mutations, still remains to be shown. Of interest, heterozygous loss of CLC-2 had no overt behavioral or morphological consequence in mice, whereas homozygous CLCN2 knockout mice had leukoencephalopathy, blindness, auditory conduction deficits, but no evidence of spontaneous seizures or increased susceptibility to flurothyl-induced seizures [99].
Potassium chloride cotransporter 3 (KCC3; SLC12A6)
KCCs mediate the electroneutral export of Cl– with cations. They maintain the intracellular Cl– concentration low, promoting therefore, the hyperpolarizing actions of GABAA receptors. Furthermore, they decrease cell volume preventing the excessive cell swelling under hypotonic conditions or during excessive neuronal activity. They have 12 transmembrane domains [103]. A number of mutations of the KCC3 isoform have been identified and linked with a type of autosomal recessive type of hereditary motor and sensory polyneuropathy and agenesis of corpus callosum (HMSN/ ACC) or Andermann syndrome that had originally been described in French Canadians [104, 105]. Apart from these two principal features, i.e., polyneuropathy with arreflexia and agenesis of corpus callosum, associated findings may include cranial neuropathies affecting usually the 3rd or 7th nerve (blepharoptosis, facial diplegia), amyotrophy or scoliosis, dysmorphic features (hypertelorism, hypoplastic maxilla, prominent chin, narrow forehead, low hairline, large ears, open mouth with protruding fissured tongue, high arched palate, overriding first toe, long tapered fingers, syndactyly, and brachycephaly), as well as psychomotor retardation [104]. Among the neurological symptoms is also bipolar disease [106]. The clinical onset is often in the first year of life. Although sporadic reports of patients with familial agenesis of corpus callosum and infantile spasms or other types of seizures have been reported, these have not documented the presence of associated KCC3 mutations [104,107,108]. On the other hand, EEG epileptiform abnormalities without overt seizures have been documented in patients with KCC3 mutations [109]. The following are some of the mutations that have been associated with HMSN/ACC and seizures. Mapping of the known mutations on the structure of KCC3 is presented in Fig. 4.
Fig. 4.

Map of KCC3 mutations associated with HNSN/ACC. Mapping of known KCC3 mutations associated with HMSN/ACC phenotype, with (red) or without (blue) reported seizures. Most are truncation mutations that result into loss of function, due to the removal of the carboxy-terminal segment. The distal carboxy-terminal portion of KCC3 contains a creatine kinase B domain (CK-B). Interaction with CK-B through this domain appears to be important for KCC3 activation. Furthermore, a number of phosphorylation sites are removed, which are thought to mediate the hypotonia-sensitive, KCC3-mediated control of cell volume (i.e. T991; T1048)
KCC3 (c.2436delG; T813fsX813)
In a cohort of 64 French Canadian patients with HMSN/ACC [110, 111], 70% were homozygous for a truncating mutation of KCC3 that deleted the majority of the carboxy-terminal intracellular segment. Seventeen percent of these patients had infrequent seizures [111, 112] and 8.4% had epileptiform EEGs [113].
KCC3 (p.Y678LfsX41 / p.F493CfsX48)
A patient carrying two mutations of KCC3 leading to premature termination and truncations following the 6th or 12th transmembrane domains was reported with features of HMSN/ACC and seizures [104].
KCC3 (p.I301SfsX15 / p.I301SfsX15)
Homozygous truncating mutations of KCC3 at the extracellular linker of the 3rd and 4th transmembrane domain were found in a 5-yearold patient with seizures and HMSN/ACC [104].
KCC3 (1584_1585delCTinsG; F529fsX532)
This mutation truncates the protein at the 7th transmembrane domain. It cosegregated with the T813fsX813 mutation in a French Canadian patient with HMSN/ACC [112].
KCC3 (c.3031C->T; p.R1011X)
The R1011X mutation has been identified in several non-French-Canadian patients with HMSN/ACC of Turkish, Italian, African, and Dutch descent and results in the deletion of a small fragment from the carboxy-terminus of the protein [111–113]. Of the patients, 4/8 had seizures or epileptiform EEGs [113]. The truncated protein was nonfunctional when expressed in Xenopus oocytes [113].
Additional mutations have been reported with HMSN/ ACC, including in non-French Canadians, but with no clear co-morbidity with seizures. These include the R675X mutation that truncates KCC3 after the 10th transmembrane domain [112]; the missense p.G539D mutation in association with the splicing mutation c.1118+1G>A [114]; the c.619C>T, p.R207C missense mutation [104]; the E1015X mutation in a Sudanese family [113]. In contrast, no pathogenic mutations of KCC3 were identified in the screening of 16 patients with rolandic seizures and 23 patients with IGE [115].
All of the reported KCC3 pathogenic truncating mutations result in various length truncations of the carboxyterminal fragment of the protein, which has been proposed to interact with creatine kinase B (Fig. 5) [116]. Creatine kinase B interacts with KCC3 through a domain located at the last 18 amino acids of its carboxy-terminus, and this is thought to be important in providing the ATP required for the activation of KCC3-mediated Cl-transport [116]. The exact mechanisms through which ATP generation from creatine kinase B regulates KCC3 cotransport have not been clearly outlined. Speculated mechanisms for the ATP role in KCC3 activity include kinase-mediated phosphorylation and activation of KCC3 or supply of the necessary K+ for KCC3 cotransport via activated Na+/K+-ATPases [113]. However, the evidence that decrease in ATP and Mg++ availability can also result in KCC3 activation suggests that further studies on the molecular steps of KCC3 activation are needed. In addition, most of the truncations remove the phosphorylation sites (T991, T1048) that regulate KCC3 activity and cell-volume regulation in response to hypotonic conditions [117].
Fig. 5.
Pathophysiology of KCC3 truncating mutations. Under normal conditions, KCC3 mediates the electroneutral cotransport of K+ and Cl–, maintaining intracellular Cl– concentration ([Cl-]i) low. As a result, activation of GABAA receptors results in influx of Cl–, across its electrochemical gradient, and hyperpolarization. Under hypotonic conditions, dephosphorylation of KCC3 activates it and prevents cell swelling. Truncating KCC3 mutations render it inactive and unable to control [Cl-]i or cell volume, leading to increased [Cl-]i and GABAA-mediated depolarizations that promote excitability. Furthermore, the regulatory adjustment of the cell volume under hypotonic conditions or excessive neuronal activity is impaired, leading to cell swelling and potential cellular degeneration that may further contribute to excitability and epileptogenesis
In confirmation of the clinical pathogenicity of these truncating mutations, mice homozygous for KCC3 gene knockouts exhibited deficits in locomotion, paired pulse inhibition, and hypomyelination and axonal swelling in theperipheral nerves [109, 112, 118]. Neurodegeneration followed the pathological evidence of cell swelling, suggesting that impaired cell-volume regulation may underlie the cellular degeneration [118]. Although peripheral nervous system pathology was a common finding in the various knockout mice for KCC3, the central nervous system (CNS) was not always affected [109, 112]. When present, it involved the hippocampus but not the corpus callosum, a key site affected in the human phenotype, suggesting that species differences or technical factors that relate to the utilized constructs may influence outcomes [109]. Decreased seizure threshold to flurothyl seizures was reported in the KCC3–/– with CNS involvement [109]. Mechanisms that could underlie this lowered threshold to seizures may primarily relate to the cell-volume dysregulation and cell swelling, which not only can induce neurodegeneration, but also directly increases seizure susceptibility [119]. KCC3 mutations also increase intracellular Cl– and shift the GABAAergic responses to less hyperpolarizing values. However, this effect is expected to be at best modest, as the lack of KCC3 may be compensated by more potent KCCs, like KCC2 [109].
Clinico-pathogenetic implications
The identification of so many mutations in GABAA receptors or chloride transporters / channels is linked with human seizure or epilepsy disorders, and the availability of in vivo and in vitro models to test their functional significance and consequences for neuronal excitability and epilepsy. Extensive review of the molecular mechanisms underlying the pathophysiological consequences of missense or nonsense mutations of GABAA receptor subunits is presented in detail in an excellent recent review by Kang and Macdonald [120]. As listed in Table 1, many of these are missense mutations that impair GABAA receptor function either by reducing the externalization of the receptor to the plasma membrane (trafficking problems) or via altering the gating characteristics of the channel. In this category are included most of the mutations currently linked with JME, CAE, and CAE with febrile seizures syndrome, ADEFS+ syndrome, or febrile seizures (detailed in Table 1). On the other hand, nonsense GABAA receptor mutations causing premature termination of the transcript include: α1(975delC; S326fs328X) seen in CAE patients, γ2W390X observed in ADEFS+ patients, γ2Q351X seen in SMEI or ADEFS+ patients, and γ2Q40X reported in SMEI patients. All the known KCC3 mutations associated with HMSN/ACC are also premature terminations (Table 2). Nonsense GABAA receptor subunit mutations can have a more severe phenotype, as it occurs in SMEI. The aberrant transcript undergoes nonsense-mediated mRNA decay (NMD), a translation-dependent degradation process, but this process does not always remove the aberrant transcripts efficiently. Transcripts that escape NMD can be further degraded in the endoplasmic reticulum (ERAD: endoplasmic reticulum associated degradation), while a small portion may still be detected at the plasma membrane. The retention or expression of these abnormal transcripts may further disrupt the cellular physiology. The efficiency of these processes and the functional state of the final product influence the severity of the phenotype. Furthermore, mutations with dominant negative effects on the expression of other proteins or subunits may increase the severity of the disease, as has been proposed for the γ2Q351X mutation, which is also associated with Dravet syndrome [121].
Table 2.
Chloride transport and genetics of human epilepsies
| Epilepsy type | Mutation | Type of mutation | Site of mutation | Functional effect of mutation |
Reference |
|---|---|---|---|---|---|
| JME | CLCN2 (M200fsX231) | Premature termination | Loss of function | [97, 100] | |
| IGE | CLCN2 (IVS2-14del11; del74-117) |
Atypical splicing; the expected deletion of helix B not confirmed |
Pathogenicity not confirmed |
[97, 100] | |
| JAE, spike-wave EEG |
CLCN2 (G715E) | Missense | Pathogenicity of heterozygous mutations not confirmed; Alters AMP-dependent gating. |
[97, 100] | |
| HMSN/ACC (Andermann syndrome) |
KCC3 (c.2436delG; T813fsX813) |
Premature termination | Deletion of carboxy- terminal intracellular segment |
Loss of function; Impaired cell volume regulation in hypotonic conditions and increase in ECl |
[110, 111, 113] |
| KCC3 (p.Y678LfsX41 / p.F493CfsX48) |
Premature termination | Truncation at TM7 or after TM12 |
[104] | ||
| KCC3 (p.I301SfsX15 / p.I301SfsX15) |
Premature termination | Linker of TM3-TM4 | [104] | ||
| KCC3 (1584_1585delCTinsG; F529fsX532) |
Premature termination | Truncation at TM7 | [112] | ||
| KCC3 (c.3031C->T; p.R1011X) |
Premature termination | Deletion of carboxy- terminal intracellular segment |
[111–113] |
What do these mutations reveal about neuronal excitability, ictogenesis, and epileptogenesis? As summarized in Table 1, most GABAA receptor mutations reduce the efficiency of GABAAergic neurotransmission. It is currently unknown whether and when the change to hyperpolarizing GABAA signaling occurs in the human brain and how these tempos are different in brains that have had different prior life experiences (i.e. seizures, stress, fever, etc.). Whether GABAA signaling is depolarizing or hyperpolarizing, reduction of available open GABAA receptors will be expected to reduce shunting inhibition promoting the excitability of a neuron. In regard to ictogenesis, the main question is what the impact will be in a specific network as this will be the determining factor for the type of seizures that will occur. It is interesting that most of the available known mutations have been linked with generalized epilepsies associated with SWD (i.e. CAE, JME) or with hyperexcitability in the setting of a trigger (i.e. fever). Do these indicate that the corticothalamic network is more vulnerable to dysfunction of GABAA signaling or that the mutations linked with febrile seizures or ADEFS+ are simply susceptibility mutations? If the latter, why is the prevalence of other types of seizures and epilepsies, like temporal lobe epilepsy, not as high in these carriers, at least with the association studies that exist so far? Could the lower prevalence of non-SWD seizures reflect a higher degree of redundancy for such isolated GABAA receptor subunit deficits within the networks that generate them, possibly due to local compensatory factors? Or could the early development of IGEs associated with SWD prevent epileptogenesis outside the corticothalamic network, as suggested by Onat’s group [122]. Does the bias in screening patients with strong family history interfere with our ability to unravel the full extent of their involvement in the other types of seizures and epilepsies? Obviously, a seizure susceptibility gene mutation is more likely to be linked with an inherited type of seizure, if we mostly screen patients predisposed to such seizures. A strong argument against this possibility, for the γ2R43Q at least, is the recreation of only the absence seizure phenotype, and not other seizure types, in the γ2R43Q knockin mice of various backgrounds [25]. More research needs to be done to better document the answers to all these questions.
Do the described genetic mutations promote ictogenesis or epileptogenesis? In the constant presence of the original genetically determined cause of deficient GABAAergic transmission, it is hard to firmly state that the ongoing seizures are not simply a result of a constantly lowered seizure threshold due to these mutations. In the classical models of acquired epileptogenesis, we are seeking evidence that either spontaneous seizures can occur in the absence of the original trigger (as in kainic acid induced epileptogenesis) or that seizure threshold decreases with time (as in kindling paradigm). The process however of genetically determined epileptogenesis will have to be viewed as entirely different from acquired epileptogenesis. Genetically determined epileptogenesis may indeed depend more upon the constant presence of causative or contributory genetic deficits that promote excitability of a seizure network and less upon plastic changes. However, the brain, especially during development, is highly plastic and responsive to even the smallest intrinsic or exogenous changes, especially as it pertains to GABA signaling [1, 4, 9, 76, 85, 86, 123]. It is therefore not surprising that beyond their effects upon neuronal physiology and excitability, genetic mutations may also cause plastic changes in the brain, increasing the susceptibility to seizures, as shown by Chiu et al. [35]. In their studies, mice with constitutive expression of the γ2R43Q allele had lower seizure threshold to pentylenetetrazole seizures in adulthood compared with mice conditionally expressing the mutant allele only after the weaning age [35].
At present, our understanding of genotype–phenotype correlations is very limited to allow definite conclusions. The cost associated with genetic testing and the bias in screening only patients who agree to be tested and have a strong positive family history for certain types of seizures are prohibiting factors in attempts to correctly estimate the prevalence, establish more powerful genotype–phenotype correlations, as well as clarify the interactions with genetic or epigenetic modifiers that may skew such associations [124]. Hopefully, multicenter ongoing studies, as well as the advances from the experimental models of genetic epilepsies, whether conditional or constitutive, will greatly enhance our ability to better understand the underlying pathogenetic mechanisms.
How can we translate these findings to improve the clinical management of patients with genetic epilepsies? Knowing the pathogenic mutation and its functional consequences may potentially help us select or design individualized therapies. For instance, the γ2K289M mutant allele may result in increased potentiation by barbiturates, suggesting that perhaps the utilization of barbiturates in carriers of this mutation may be beneficial, if their seizure types are appropriate for this drug. The availability of drugs that read through premature termination codons may be of use in patients with such mutations. However, it will be essential to determine what will the function of the novel transcript be and what will its impact be on the overall cell physiology. As prior studies indicate [125], the answer will be different for each mutation. It will therefore be essential to screen the utility and benefit of such drugs in in vitro and in vivo models of genetic diseases before committing them to clinical trials.
Conclusions
Several mutations of the GABAA receptor subunits, the CLCN2 and KCC3 genes have been found in patients with epilepsies of seizures. At present, the pathogenic role of CLCN2 mutations is still unclear, and at best they might contribute to increased susceptibility to seizures, in the presence of additional risk factors. More solid evidence exists on the role of GABAA receptor mutations and KCC3, most of which produce a loss-of-function state due to the production of nonfunctional or hypofunctional proteins, or impair trafficking and insertion to the plasma membrane. However, these affect a minority of the population affected with the associated seizure types and epilepsy syndromes. A better understanding of genotype–phenotype of the affected patients, as well as of the pathogenetic mechanisms leading to ictogenesis and epileptogenesis in these predisposed patients will be invaluable both to improve the management and counseling of these patients and their families, as well as enhance our ability to effectively treat them. The availability of animal models with knockin mutations will be invaluable in such a task.
Acknowledgements
I would like to acknowledge the funding by NIH NINDS research grants NS20253, NS58303, and NS45243, NINDS/NICHD grant NS62947, as well as grants from People Against Childhood Epilepsy, the International Rett Syndrome Foundation, Johnson & Johnson (unrelated to this paper), and the Heffer Family Foundation that supported research in my lab.
References
- 1.Galanopoulou AS. GABA(A) receptors in normal development and seizures: friends or foes? Curr Neuropharmacol. 2008;6(1):1–20. doi: 10.2174/157015908783769653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Farrant M, Kaila K. The cellular, molecular and ionic basis of GABA(A) receptor signalling. Prog Brain Res. 2007;160:59–87. doi: 10.1016/S0079-6123(06)60005-8. [DOI] [PubMed] [Google Scholar]
- 3.Olsen RW, Sieghart W. International Union of Pharmacology. LXX. Subtypes of gamma-aminobutyric acid(A) receptors: classification on the basis of subunit composition, pharmacology, and function. Update. Pharmacol Rev. 2008;60(3):243–260. doi: 10.1124/pr.108.00505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ben-Ari Y. Excitatory actions of gaba during development: the nature of the nurture. Nat Rev Neurosci. 2002;3(9):728–739. doi: 10.1038/nrn920. [DOI] [PubMed] [Google Scholar]
- 5.Rivera C, Voipio J, Payne JA, Ruusuvuori E, Lahtinen H, Lamsa K, Pirvola U, Saarma M, Kaila K. The K+/Cl– co-transporter KCC2 renders GABA hyperpolarizing during neuronal maturation. Nature. 1999;397(6716):251–255. doi: 10.1038/16697. [DOI] [PubMed] [Google Scholar]
- 6.Huberfeld G, Wittner L, Clemenceau S, Baulac M, Kaila K, Miles RRivera C. Perturbed chloride homeostasis and GABAergic signaling in human temporal lobe epilepsy. J Neurosci. 2007;27(37):9866–9873. doi: 10.1523/JNEUROSCI.2761-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Blaesse P, Airaksinen MS, Rivera C, Kaila K. Cation-chloride cotransporters and neuronal function. Neuron. 2009;61(6):820–838. doi: 10.1016/j.neuron.2009.03.003. [DOI] [PubMed] [Google Scholar]
- 8.Li H, Khirug S, Cai C, Ludwig A, Blaesse P, Kolikova J, Afzalov R, Coleman SK, Lauri S, Airaksinen MS, Keinanen K, Khiroug L, Saarma M, Kaila K, Rivera C. KCC2 interacts with the dendritic cytoskeleton to promote spine development. Neuron. 2007;56(6):1019–1033. doi: 10.1016/j.neuron.2007.10.039. [DOI] [PubMed] [Google Scholar]
- 9.Cancedda L, Fiumelli H, Chen K, Poo MM. Excitatory GABA action is essential for morphological maturation of cortical neurons in vivo. J Neurosci. 2007;27(19):5224–5235. doi: 10.1523/JNEUROSCI.5169-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hubner CA, Stein V, Hermans-Borgmeyer I, Meyer T, Ballanyi K, Jentsch TJ. Disruption of KCC2 reveals an essential role of K–Cl cotransport already in early synaptic inhibition. Neuron. 2001;30(2):515–524. doi: 10.1016/s0896-6273(01)00297-5. [DOI] [PubMed] [Google Scholar]
- 11.Villemure JF, Adam L, Bevan NJ, Gearing K, Chenier S, Bouvier M. Subcellular distribution of GABA(B) receptor homo- and hetero-dimers. Biochem J. 2005;388(Pt 1):47–55. doi: 10.1042/BJ20041435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Balasubramanian S, Teissere JA, Raju DVHall RA. Hetero-oligomerization between GABAA and GABAB receptors regulates GABAB receptor trafficking. J Biol Chem. 2004;279(18):18840–18850. doi: 10.1074/jbc.M313470200. [DOI] [PubMed] [Google Scholar]
- 13.Gassmann M, Shaban H, Vigot R, Sansig G, Haller C, Barbieri S, Humeau Y, Schuler V, Muller M, Kinzel B, Klebs K, Schmutz M, Froestl W, Heid J, Kelly PH, Gentry C, Jaton AL, Van der Putten H, Mombereau C, Lecourtier L, Mosbacher J, Cryan JF, Fritschy JM, Luthi A, Kaupmann K, Bettler B. Redistribution of GABAB (1) protein and atypical GABAB responses in GABAB(2)-deficient mice. J Neurosci. 2004;24(27):6086–6097. doi: 10.1523/JNEUROSCI.5635-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bettler B, Tiao JY. Molecular diversity, trafficking and subcellular localization of GABAB receptors. Pharmacol Ther. 2006;110(3):533–543. doi: 10.1016/j.pharmthera.2006.03.006. [DOI] [PubMed] [Google Scholar]
- 15.Feucht M, Fuchs K, Pichlbauer E, Hornik K, Scharfetter J, Goessler R, Fureder T, Cvetkovic N, Sieghart W, Kasper S, Aschauer H. Possible association between childhood absence epilepsy and the gene encoding GABRB3. Biol Psychiatry. 1999;46(7):997–1002. doi: 10.1016/s0006-3223(99)00039-6. [DOI] [PubMed] [Google Scholar]
- 16.Maljevic S, Krampfl K, Cobilanschi J, Tilgen N, Beyer S, Weber YG, Schlesinger F, Ursu D, Melzer W, Cossette P, Bufler J, Lerche H, Heils A. A mutation in the GABA(A) receptor alpha(1)-subunit is associated with absence epilepsy. Ann Neurol. 2006;59(6):983–987. doi: 10.1002/ana.20874. [DOI] [PubMed] [Google Scholar]
- 17.Kang JQ, Shen W, Macdonald RL. Two molecular pathways (NMD and ERAD) contribute to a genetic epilepsy associated with the GABA(A) receptor GABRA1 PTC mutation, 975delC, S326fs328X. J Neurosci. 2009;29(9):2833–2844. doi: 10.1523/JNEUROSCI.4512-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dibbens LM, Harkin LA, Richards M, Hodgson BL, Clarke AL, Petrou S, Scheffer IE, Berkovic SF, Mulley JC. The role of neuronal GABA(A) receptor subunit mutations in idiopathic generalized epilepsies. Neurosci Lett. 2009;453(3):162–165. doi: 10.1016/j.neulet.2009.02.038. [DOI] [PubMed] [Google Scholar]
- 19.Urak L, Feucht M, Fathi N, Hornik K, Fuchs K. A GABRB3 promoter haplotype associated with childhood absence epilepsy impairs transcriptional activity. Hum Mol Genet. 2006;15(16):2533–2541. doi: 10.1093/hmg/ddl174. [DOI] [PubMed] [Google Scholar]
- 20.Tanaka M, Olsen RW, Medina MT, Schwartz E, Alonso ME, Duron RM, Castro-Ortega R, Martinez-Juarez IE, Pascual-Castroviejo I, Machado-Salas J, Silva R, Bailey JN, Bai D, Ochoa A, Jara-Prado A, Pineda G, Macdonald RL, Delgado-Escueta AV. Hyperglycosylation and reduced GABA currents of mutated GABRB3 polypeptide in remitting childhood absence epilepsy. Am J Hum Genet. 2008;82(6):1249–1261. doi: 10.1016/j.ajhg.2008.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shinnar S, Glauser TA. Febrile seizures. J Child Neurol. 2002;17(Suppl 1):S44–S52. doi: 10.1177/08830738020170010601. [DOI] [PubMed] [Google Scholar]
- 22.Johnson EW, Dubovsky J, Rich SS, O’Donovan CA, Orr HT, Anderson VE, Gil-Nagel A, Ahmann P, Dokken CG, Schneider DT, Weber JL. Evidence for a novel gene for familial febrile convulsions, FEB2, linked to chromosome 19p in an extended family from the Midwest. Hum Mol Genet. 1998;7(1):63–67. doi: 10.1093/hmg/7.1.63. [DOI] [PubMed] [Google Scholar]
- 23.Scheffer IE, Berkovic SF. Generalized epilepsy with febrile seizures plus. A genetic disorder with heterogeneous clinical phenotypes. Brain. 1997;120(Pt 3):479–490. doi: 10.1093/brain/120.3.479. [DOI] [PubMed] [Google Scholar]
- 24.Wallace RH, Marini C, Petrou S, Harkin LA, Bowser DN, Panchal RG, Williams DA, Sutherland GR, Mulley JC, Scheffer IE, Berkovic SF. Mutant GABA(A) receptor gamma2- subunit in childhood absence epilepsy and febrile seizures. Nat Genet. 2001;28(1):49–52. doi: 10.1038/ng0501-49. [DOI] [PubMed] [Google Scholar]
- 25.Tan HO, Reid CA, Single FN, Davies PJ, Chiu C, Murphy S, Clarke AL, Dibbens L, Krestel H, Mulley JC, Jones MV, Seeburg PH, Sakmann B, Berkovic SF, Sprengel R, Petrou S. Reduced cortical inhibition in a mouse model of familial childhood absence epilepsy. Proc Natl Acad Sci USA. 2007;104(44):17536–17541. doi: 10.1073/pnas.0708440104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bianchi MT, Song L, Zhang H, Macdonald RL. Two different mechanisms of disinhibition produced by GABAA receptor mutations linked to epilepsy in humans. J Neurosci. 2002;22(13):5321–5327. doi: 10.1523/JNEUROSCI.22-13-05321.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sancar F, Czajkowski C. A GABAA receptor mutation linked to human epilepsy (gamma2R43Q) impairs cell surface expression of alphabetagamma receptors. J Biol Chem. 2004;279(45):47034–47039. doi: 10.1074/jbc.M403388200. [DOI] [PubMed] [Google Scholar]
- 28.Eugene E, Depienne C, Baulac S, Baulac M, Fritschy JM, Le Guern E, Miles R, Poncer JC. GABA(A) receptor gamma 2 subunit mutations linked to human epileptic syndromes differentially affect phasic and tonic inhibition. J Neurosci. 2007;27(51):14108–14116. doi: 10.1523/JNEUROSCI.2618-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hales TG, Tang H, Bollan KA, Johnson SJ, King DP, McDonald NA, Cheng A, Connolly CN. The epilepsy mutation, gamma2(R43Q) disrupts a highly conserved inter-subunit contact site, perturbing the biogenesis of GABAA receptors. Mol Cell Neurosci. 2005;29(1):120–127. doi: 10.1016/j.mcn.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 30.Kang JQ, Macdonald RL. The GABAA receptor gamma2 subunit R43Q mutation linked to childhood absence epilepsy and febrile seizures causes retention of alpha1beta2gamma2S receptors in the endoplasmic reticulum. J Neurosci. 2004;24(40):8672–8677. doi: 10.1523/JNEUROSCI.2717-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fedi M, Berkovic SF, Macdonell RA, Curatolo JM, Marini C, Reutens DC. Intracortical hyperexcitability in humans with a GABAA receptor mutation. Cereb Cortex. 2008;18(3):664–669. doi: 10.1093/cercor/bhm100. [DOI] [PubMed] [Google Scholar]
- 32.Goldschen-Ohm MP, Wagner DA, Petrou S, Jones MV. An epilepsy-related region in the GABAA receptor mediates long-distance effects on GABA and benzodiazepine binding sites. Mol Pharmacol. 2009 doi: 10.1124/mol.109.058289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bowser DN, Wagner DA, Czajkowski C, Cromer BA, Parker MW, Wallace RH, Harkin LA, Mulley JC, Marini C, Berkovic SF, Williams DA, Jones MV, Petrou S. Altered kinetics and benzodiazepine sensitivity of a GABAA receptor subunit mutation [gamma 2(R43Q)] found in human epilepsy. Proc Natl Acad Sci USA. 2002;99(23):15170–15175. doi: 10.1073/pnas.212320199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fedi M, Berkovic SF, Marini C, Mulligan R, Tochon-Danguy H, Reutens DC. A GABAA receptor mutation causing generalized epilepsy reduces benzodiazepine receptor binding. Neuroimage. 2006;32(3):995–1000. doi: 10.1016/j.neuroimage.2006.05.059. [DOI] [PubMed] [Google Scholar]
- 35.Chiu C, Reid CA, Tan HO, Davies PJ, Single FN, Koukoulas I, Berkovic SF, Tan SS, Sprengel R, Jones MV, Petrou S. Developmental impact of a familial GABAA receptor epilepsy mutation. Ann Neurol. 2008;64(3):284–293. doi: 10.1002/ana.21440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Raol YH, Lund IV, Bandyopadhyay S, Zhang G, Roberts DS, Wolfe JH, Russek SJ, Brooks-Kayal AR. Enhancing GABA(A) receptor alpha 1 subunit levels in hippocampal dentate gyrus inhibits epilepsy development in an animal model of temporal lobe epilepsy. J Neurosci. 2006;26(44):11342–11346. doi: 10.1523/JNEUROSCI.3329-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kananura C, Haug K, Sander T, Runge U, Gu W, Hallmann K, Rebstock J, Heils A, Steinlein OK. A splice-site mutation in GABRG2 associated with childhood absence epilepsy and febrile convulsions. Arch Neurol. 2002;59(7):1137–1141. doi: 10.1001/archneur.59.7.1137. [DOI] [PubMed] [Google Scholar]
- 38.Baulac S, Huberfeld G, Gourfinkel-An I, Mitropoulou G, Beranger A, Prud’homme JF, Baulac M, Brice A, Bruzzone R, LeGuern E. First genetic evidence of GABA(A) receptor dysfunction in epilepsy: a mutation in the gamma2-subunit gene. Nat Genet. 2001;28(1):46–48. doi: 10.1038/ng0501-46. [DOI] [PubMed] [Google Scholar]
- 39.Ramakrishnan L, Hess GP. On the mechanism of a mutated and abnormally functioning gamma-aminobutyric acid (A) receptor linked to epilepsy. Biochemistry. 2004;43(23):7534–7540. doi: 10.1021/bi036181+. [DOI] [PubMed] [Google Scholar]
- 40.Hales TG, Deeb TZ, Tang H, Bollan KA, King DP, Johnson SJ, Connolly CN. An asymmetric contribution to gamma-aminobutyric type A receptor function of a conserved lysine within TM2-3 of alpha1, beta2, and gamma2 subunits. J Biol Chem. 2006;281(25):17034–17043. doi: 10.1074/jbc.M603599200. [DOI] [PubMed] [Google Scholar]
- 41.Krivoshein AV, Hess GP. On the mechanism of alleviation by phenobarbital of the malfunction of an epilepsy-linked GABA (A) receptor. Biochemistry. 2006;45(38):11632–11641. doi: 10.1021/bi061207t. [DOI] [PubMed] [Google Scholar]
- 42.Harkin LA, Bowser DN, Dibbens LM, Singh R, Phillips F, Wallace RH, Richards MC, Williams DA, Mulley JC, Berkovic SF, Scheffer IE, Petrou S. Truncation of the GABA(A)- receptor gamma2 subunit in a family with generalized epilepsy with febrile seizures plus. Am J Hum Genet. 2002;70(2):530–536. doi: 10.1086/338710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sun H, Zhang Y, Liang J, Liu X, Ma X, Wu H, Xu K, Qin J, Qi Y, Wu X. Gene symbol: GABRG2. Disease: generalized epilepsy with febrile seizures plus. Hum Genet. 2008;124(3):298. [PubMed] [Google Scholar]
- 44.Audenaert D, Schwartz E, Claeys KG, Claes L, Deprez L, Suls A, Van Dyck T, Lagae L, Van Broeckhoven C, Macdonald RL, De Jonghe P. A novel GABRG2 mutation associated with febrile seizures. Neurology. 2006;67(4):687–690. doi: 10.1212/01.wnl.0000230145.73496.a2. [DOI] [PubMed] [Google Scholar]
- 45.Nusser Z, Mody I. Selective modulation of tonic and phasic inhibitions in dentate gyrus granule cells. J Neurophysiol. 2002;87(5):2624–2628. doi: 10.1152/jn.2002.87.5.2624. [DOI] [PubMed] [Google Scholar]
- 46.Dibbens LM, Feng HJ, Richards MC, Harkin LA, Hodgson BL, Scott D, Jenkins M, Petrou S, Sutherland GR, Scheffer IE, Berkovic SF, Macdonald RL, Mulley JC. GABRD encoding a protein for extra- or peri-synaptic GABAA receptors is a susceptibility locus for generalized epilepsies. Hum Mol Genet. 2004;13(13):1315–1319. doi: 10.1093/hmg/ddh146. [DOI] [PubMed] [Google Scholar]
- 47.Feng HJ, Kang JQ, Song L, Dibbens L, Mulley J, Macdonald RL. Delta subunit susceptibility variants E177A and R220H associated with complex epilepsy alter channel gating and surface expression of alpha4beta2delta GABAA receptors. J Neurosci. 2006;26(5):1499–1506. doi: 10.1523/JNEUROSCI.2913-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dravet C. Les epilepsies graves de l’ enfant. Vie Med. 1978;8:543–548. [Google Scholar]
- 49.Hirose S. A new paradigm of channelopathy in epilepsy syndromes: intracellular trafficking abnormality of channel molecules. Epilepsy Res. 2006;70(Suppl 1):S206–S217. doi: 10.1016/j.eplepsyres.2005.12.007. [DOI] [PubMed] [Google Scholar]
- 50.Panayiotopoulos CP. Idiopathic generalized epilepsies. In: Panayiotopoulos CP, editor. A clinical guide to epileptic syndromes and their treatment. Springer-Verlag London Ltd; London: 2007. pp. 319–362. [Google Scholar]
- 51.Williams DB, Akabas MH. Gamma-aminobutyric acid increases the water accessibility of M3 membrane-spanning segment residues in gamma-aminobutyric acid type A receptors. Biophys J. 1999;77(5):2563–2574. doi: 10.1016/s0006-3495(99)77091-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Williams DB, Akabas MH. Benzodiazepines induce a conformational change in the region of the gamma-aminobutyric acid type A receptor alpha(1)-subunit M3 membrane-spanning segment. Mol Pharmacol. 2000;58(5):1129–1136. doi: 10.1124/mol.58.5.1129. [DOI] [PubMed] [Google Scholar]
- 53.Williams DB, Akabas MH. Structural evidence that propofol stabilizes different GABA(A) receptor states at potentiating and activating concentrations. J Neurosci. 2002;22(17):7417–7424. doi: 10.1523/JNEUROSCI.22-17-07417.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Krampfl K, Maljevic S, Cossette P, Ziegler E, Rouleau GA, Lerche H, Bufler J. Molecular analysis of the A322D mutation in the GABA receptor alpha-subunit causing juvenile myoclonic epilepsy. Eur J NeuroSci. 2005;22(1):10–20. doi: 10.1111/j.1460-9568.2005.04168.x. [DOI] [PubMed] [Google Scholar]
- 55.Fisher JL. A mutation in the GABAA receptor alpha 1 subunit linked to human epilepsy affects channel gating properties. Neuropharmacology. 2004;46(5):629–637. doi: 10.1016/j.neuropharm.2003.11.015. [DOI] [PubMed] [Google Scholar]
- 56.Cossette P, Liu L, Brisebois K, Dong H, Lortie A, Vanasse M, Saint-Hilaire JM, Carmant L, Verner A, Lu WY, Wang YT, Rouleau GA. Mutation of GABRA1 in an autosomal dominant form of juvenile myoclonic epilepsy. Nat Genet. 2002;31(2):184–189. doi: 10.1038/ng885. [DOI] [PubMed] [Google Scholar]
- 57.Macdonald RL, Gallagher MJ, Feng HJ, Kang J. GABA(A) receptor epilepsy mutations. Biochem Pharmacol. 2004;68(8):1497–1506. doi: 10.1016/j.bcp.2004.07.029. [DOI] [PubMed] [Google Scholar]
- 58.Robb SA, Pohl KR, Baraitser M, Wilson J, Brett EM. The ‘happy puppet’ syndrome of Angelman: review of the clinical features. Arch Dis Child. 1989;64(1):83–86. doi: 10.1136/adc.64.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dan B, Boyd SG. Angelman syndrome reviewed from a neurophysiological perspective. The UBE3A-GABRB3 hypothesis. Neuropediatrics. 2003;34(4):169–176. doi: 10.1055/s-2003-42213. [DOI] [PubMed] [Google Scholar]
- 60.Wagstaff J, Knoll JH, Fleming J, Kirkness EF, Martin-Gallardo A, Greenberg F, Graham JM, Jr, Menninger J, Ward D, Venter JC, et al. Localization of the gene encoding the GABAA receptor beta 3 subunit to the Angelman/Prader–Willi region of human chromosome 15. Am J Hum Genet. 1991;49(2):330–337. [PMC free article] [PubMed] [Google Scholar]
- 61.Knoll JH, Cheng SD, Lalande M. Allele specificity of DNA replication timing in the Angelman/Prader–Willi syndrome imprinted chromosomal region. Nat Genet. 1994;6(1):41–46. doi: 10.1038/ng0194-41. [DOI] [PubMed] [Google Scholar]
- 62.Hogart A, Nagarajan RP, Patzel KA, Yasui DH, Lasalle JM. 15q11-13 GABAA receptor genes are normally biallelically expressed in brain yet are subject to epigenetic dysregulation in autism-spectrum disorders. Hum Mol Genet. 2007;16(6):691–703. doi: 10.1093/hmg/ddm014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Minassian BA, DeLorey TM, Olsen RW, Philippart M, Bronstein Y, Zhang Q, Guerrini R, Van Ness P, Livet MO, Delgado-Escueta AV. Angelman syndrome: correlations between epilepsy phenotypes and genotypes. Ann Neurol. 1998;43(4):485–493. doi: 10.1002/ana.410430412. [DOI] [PubMed] [Google Scholar]
- 64.Homanics GE, DeLorey TM, Firestone LL, Quinlan JJ, Handforth A, Harrison NL, Krasowski MD, Rick CE, Korpi ER, Makela R, Brilliant MH, Hagiwara N, Ferguson C, Snyder K, Olsen RW. Mice devoid of gamma-aminobutyrate type A receptor beta3 subunit have epilepsy, cleft palate, and hypersensitive behavior. Proc Natl Acad Sci USA. 1997;94(8):4143–4148. doi: 10.1073/pnas.94.8.4143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Quinlan JJ, Homanics GE, Homanics LL. Anesthesia sensitivity in mice that lack the beta3 subunit of the gamma-aminobutyric acid type A receptor. Anesthesiology. 1998;88(3):775–780. doi: 10.1097/00000542-199803000-00030. [DOI] [PubMed] [Google Scholar]
- 66.Liljelund P, Handforth A, Homanics GE, Olsen RW. GABAA receptor beta3 subunit gene-deficient heterozygous mice show parent-of-origin and gender-related differences in beta3 subunit levels, EEG, and behavior. Brain Res Dev Brain Res. 2005;157(2):150–161. doi: 10.1016/j.devbrainres.2005.03.014. [DOI] [PubMed] [Google Scholar]
- 67.Gibbs JW, 3rd, GBCoulter DASchroder. GABAA receptor function in developing rat thalamic reticular neurons: whole cell recordings of GABA-mediated currents and modulation by clonazepam. J Neurophysiol. 1996;76(4):2568–2579. doi: 10.1152/jn.1996.76.4.2568. [DOI] [PubMed] [Google Scholar]
- 68.Tan HO, Reid CA, Chiu C, Jones MV, Petrou S. Increased thalamic inhibition in the absence seizure prone DBA/2J mouse. Epilepsia. 2008;49(5):921–925. doi: 10.1111/j.1528-1167.2008.01536.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Huguenard JR, Prince DA. Clonazepam suppresses GABAB-mediated inhibition in thalamic relay neurons through effects in nucleus reticularis. J Neurophysiol. 1994;71(6):2576–2581. doi: 10.1152/jn.1994.71.6.2576. [DOI] [PubMed] [Google Scholar]
- 70.Li H, Huguenard JR, Fisher RS. Gender and age differences in expression of GABAA receptor subunits in rat somatosensory thalamus and cortex in an absence epilepsy model. Neurobiol Dis. 2007;25(3):623–630. doi: 10.1016/j.nbd.2006.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Laurie DJ, Wisden W, Seeburg PH. The distribution of thirteen GABAA receptor subunit mRNAs in the rat brain. III. Embryonic and postnatal development. J Neurosci. 1992;12(11):4151–4172. doi: 10.1523/JNEUROSCI.12-11-04151.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sperber EF, Veliskova J, Germano IM, Friedman LK, Moshe SL. Age-dependent vulnerability to seizures. Adv Neurol. 1999;79:161–169. [PubMed] [Google Scholar]
- 73.Veliskova J, Moshe SL. Update on the role of substantia nigra pars reticulata in the regulation of seizures. Epilepsy Curr. 2006;6(3):83–87. doi: 10.1111/j.1535-7511.2006.00106.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Moshe SL, Brown LL, Kubova H, Veliskova J, Zukin RS, Sperber EF. Maturation and segregation of brain networks that modify seizures. Brain Res. 1994;665(1):141–146. doi: 10.1016/0006-8993(94)91164-9. [DOI] [PubMed] [Google Scholar]
- 75.Janz D. The idiopathic generalized epilepsies of adolescence with childhood and juvenile age of onset. Epilepsia. 1997;38(1):4–11. doi: 10.1111/j.1528-1157.1997.tb01073.x. [DOI] [PubMed] [Google Scholar]
- 76.Galanopoulou AS. Sexually dimorphic expression of KCC2 and GABA function. Epilepsy Res. 2008;80(2–3):99–113. doi: 10.1016/j.eplepsyres.2008.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Nett ST, Jorge-Rivera JC, Myers M, Clark AS, Henderson LP. Properties and sex-specific differences of GABAA receptors in neurons expressing gamma1 subunit mRNA in the preoptic area of the rat. J Neurophysiol. 1999;81(1):192–203. doi: 10.1152/jn.1999.81.1.192. [DOI] [PubMed] [Google Scholar]
- 78.Chudomel O, Herman H, Nair K, Moshe SL, Galanopoulou AS. Age-and gender-related differences in GABAA receptor-mediated postsynaptic currents in GABAergic neurons of the substantia nigra reticulata in the rat. Neuroscience. 2009;163(1):155–167. doi: 10.1016/j.neuroscience.2009.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Juptner M, Hiemke C. Sex differences in GABAA receptor binding in rat brain measured by an improved in vitro binding assay. Exp Brain Res. 1990;81(2):297–302. doi: 10.1007/BF00228119. [DOI] [PubMed] [Google Scholar]
- 80.Canonaco M, Tavolaro R, Facciolo RM, Carelli A, MCristaldi M Cagnin. Sexual dimorphism of GABAA receptor levels in subcortical brain regions of a woodland rodent (Apodemus sylvaticus) Brain Res Bull. 1996;40(3):187–194. doi: 10.1016/0361-9230(96)00034-2. [DOI] [PubMed] [Google Scholar]
- 81.Persad V, Cortez MA, Snead OC., 3rd A chronic model of atypical absence seizures: studies of developmental and gender sensitivity. Epilepsy Res. 2002;48(1–2):111–119. doi: 10.1016/s0920-1211(01)00319-9. [DOI] [PubMed] [Google Scholar]
- 82.Magenis RE, Toth-Fejel S, Allen LJ, Black M, Brown MG, Budden S, Cohen R, Friedman JM, Kalousek D, Zonana J, et al. Comparison of the 15q deletions in Prader–Willi and Angelman syndromes: specific regions, extent of deletions, parental origin, and clinical consequences. Am J Med Genet. 1990;35(3):333–349. doi: 10.1002/ajmg.1320350307. [DOI] [PubMed] [Google Scholar]
- 83.Driscoll DJ, Waters MF, Williams CA, Zori RT, Glenn CC, Avidano KM, Nicholls RD. A DNA methylation imprint, determined by the sex of the parent, distinguishes the Angelman and Prader–Willi syndromes. Genomics. 1992;13(4):917–924. doi: 10.1016/0888-7543(92)90001-9. [DOI] [PubMed] [Google Scholar]
- 84.Segovia S, del Cerro MC, Ortega E, Perez-Laso C, Rodriguez-Zafra C, Izquierdo MA, Guillamon A. Role of GABAA receptors in the organization of brain and behavioural sex differences. NeuroReport. 1996;7(15–17):2553–2557. doi: 10.1097/00001756-199611040-00030. [DOI] [PubMed] [Google Scholar]
- 85.Wang DD, Kriegstein AR. Defining the role of GABA in cortical development. J Physiol. 2009;587(Pt 9):1873–1879. doi: 10.1113/jphysiol.2008.167635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wang DD, Kriegstein AR. GABA regulates excitatory synapse formation in the neocortex via NMDA receptor activation. J Neurosci. 2008;28(21):5547–5558. doi: 10.1523/JNEUROSCI.5599-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wang DD, Kriegstein AR, Ben-Ari Y. GABA regulates stem cell proliferation before nervous system formation. Epilepsy Curr. 2008;8(5):137–139. doi: 10.1111/j.1535-7511.2008.00270.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chudotvorova I, Ivanov A, Rama S, Hubner CA, Pellegrino C, Ben-Ari Y, Medina I. Early expression of KCC2 in rat hippocampal cultures augments expression of functional GABA synapses. J Physiol. 2005;566(Pt 3):671–679. doi: 10.1113/jphysiol.2005.089821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kang JQ, Shen W, Macdonald RL. Why does fever trigger febrile seizures? GABAA receptor gamma2 subunit mutations associated with idiopathic generalized epilepsies have temperature-dependent trafficking deficiencies. J Neurosci. 2006;26(9):2590–2597. doi: 10.1523/JNEUROSCI.4243-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sankar R, Auvin S, Mazarati A, Shin D. Inflammation contributes to seizure-induced hippocampal injury in the neonatal rat brain. Acta Neurol Scand Suppl. 2007;186:16–20. [PubMed] [Google Scholar]
- 91.Auvin S, Shin D, Mazarati A, Nakagawa J, Miyamoto J, Sankar R. Inflammation exacerbates seizure-induced injury in the immature brain. Epilepsia. 2007;48(Suppl 5):27–34. doi: 10.1111/j.1528-1167.2007.01286.x. [DOI] [PubMed] [Google Scholar]
- 92.Heida JG, Teskey GC, Pittman QJ. Febrile convulsions induced by the combination of lipopolysaccharide and low-dose kainic acid enhance seizure susceptibility, not epileptogenesis, in rats. Epilepsia. 2005;46(12):1898–1905. doi: 10.1111/j.1528-1167.2005.00286.x. [DOI] [PubMed] [Google Scholar]
- 93.Thiemann A, Grunder S, Pusch M, Jentsch TJ. A chloride channel widely expressed in epithelial and non-epithelial cells. Nature. 1992;356(6364):57–60. doi: 10.1038/356057a0. [DOI] [PubMed] [Google Scholar]
- 94.Staley K. The role of an inwardly rectifying chloride conductance in postsynaptic inhibition. J Neurophysiol. 1994;72(1):273–284. doi: 10.1152/jn.1994.72.1.273. [DOI] [PubMed] [Google Scholar]
- 95.Mladinic M, Becchetti A, Didelon F, Bradbury A, Cherubini E. Low expression of the ClC-2 chloride channel during postnatal development: a mechanism for the paradoxical depolarizing action of GABA and glycine in the hippocampus. Proc Biol Sci. 1999;266(1425):1207–1213. doi: 10.1098/rspb.1999.0764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Staley K, Smith R, Schaack J, Wilcox C, Jentsch TJ. Alteration of GABAA receptor function following gene transfer of the CLC-2 chloride channel. Neuron. 1996;17(3):543–551. doi: 10.1016/s0896-6273(00)80186-5. [DOI] [PubMed] [Google Scholar]
- 97.Haug K, Warnstedt M, Alekov AK, Sander T, Ramirez A, Poser B, Maljevic S, Hebeisen S, Kubisch C, Rebstock J, Horvath S, Hallmann K, Dullinger JS, Rau B, Haverkamp F, Beyenburg S, Schulz H, Janz D, Giese B, Muller-Newen G, Propping P, Elger CE, Fahlke CLerche H. Retraction: mutations in CLCN2 encoding a voltage-gated chloride channel are associated with idiopathic generalized epilepsies. Nat Genet. 2009;41(9):1043. doi: 10.1038/ng0909-1043. [DOI] [PubMed] [Google Scholar]
- 98.D’Agostino D, Bertelli M, Gallo S, Cecchin S, Albiero E, Garofalo PG, Gambardella A, St Hilaire JM, Kwiecinski H, Andermann E, Pandolfo M. Mutations and polymorphisms of the CLCN2 gene in idiopathic epilepsy. Neurology. 2004;63(8):1500–1502. doi: 10.1212/01.wnl.0000142093.94998.1a. [DOI] [PubMed] [Google Scholar]
- 99.Blanz J, Schweizer M, Auberson M, Maier H, Muenscher A, Hubner CA, Jentsch TJ. Leukoencephalopathy upon disruption of the chloride channel ClC-2. J Neurosci. 2007;27(24):6581–6589. doi: 10.1523/JNEUROSCI.0338-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Niemeyer MI, Yusef YR, Cornejo I, Flores CA, Sepulveda FV, Cid LP. Functional evaluation of human ClC-2 chloride channel mutations associated with idiopathic generalized epilepsies. Physiol Genomics. 2004;19(1):74–83. doi: 10.1152/physiolgenomics.00070.2004. [DOI] [PubMed] [Google Scholar]
- 101.Haug K, Warnstedt M, Alekov AK, Sander T, Ramirez A, Poser B, Maljevic S, Hebeisen S, Kubisch C, Rebstock J, Horvath S, Hallmann K, Dullinger JS, Rau B, Haverkamp F, Beyenburg S, Schulz H, Janz D, Giese B, Muller-Newen G, Propping P, Elger CE, Fahlke C, Lerche H, Heils A. Mutations in CLCN2 encoding a voltage-gated chloride channel are associated with idiopathic generalized epilepsies. Nat Genet. 2003;33(4):527–532. doi: 10.1038/ng1121. [DOI] [PubMed] [Google Scholar]
- 102.Scott JW, Hawley SA, Green KA, Anis M, Stewart G, Scullion GA, Norman DG, Hardie DG. CBS domains form energysensing modules whose binding of adenosine ligands is disrupted by disease mutations. J Clin Invest. 2004;113(2):274–284. doi: 10.1172/JCI19874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Race JE, Makhlouf FN, Logue PJ, Wilson FH, Dunham PB, Holtzman EJ. Molecular cloning and functional characterization of KCC3, a new K-Cl cotransporter. Am J Physiol. 1999;277(6 Pt 1):C1210–C1219. doi: 10.1152/ajpcell.1999.277.6.C1210. [DOI] [PubMed] [Google Scholar]
- 104.Uyanik G, Elcioglu N, Penzien J, Gross C, Yilmaz Y, Olmez A, Demir E, Wahl D, Scheglmann K, Winner B, Bogdahn U, Topaloglu H, Hehr U, Winkler J. Novel truncating and missense mutations of the KCC3 gene associated with Andermann syndrome. Neurology. 2006;66(7):1044–1048. doi: 10.1212/01.wnl.0000204181.31175.8b. [DOI] [PubMed] [Google Scholar]
- 105.Andermann E, Andermann F, Joubert M, Melancon D, Karpati G, Carpenter S. Three familial midline malformation syndromes of the central nervous system: agenesis of the corpus callosum and anterior horn-cell disease; agenesis of cerebellar vermis; and atrophy of the cerebellar vermis. Birth Defects Orig Artic Ser. 1975;11(2):269–293. [PubMed] [Google Scholar]
- 106.Meyer J, Johannssen K, Freitag CM, Schraut K, Teuber I, Hahner A, Mainhardt C, Mossner R, Volz HP, Wienker TF, McKeane D, Stephan DA, Rouleau G, Reif A, Lesch KP. Rare variants of the gene encoding the potassium chloride cotransporter 3 are associated with bipolar disorder. Int J Neuropsychopharmacol. 2005;8(4):495–504. doi: 10.1017/S1461145705005821. [DOI] [PubMed] [Google Scholar]
- 107.Cao A, Cianchetti C, Signorini E, Loi M, Sanna G, De Virgiliis S. Agenesis of the corpus callosum, infantile spasms, spastic quadriplegia, microcephaly and severe mental retardation in three siblings. Clin Genet. 1977;12(5):290–296. doi: 10.1111/j.1399-0004.1977.tb00943.x. [DOI] [PubMed] [Google Scholar]
- 108.Shapira Y, Cohen T. Agenesis of the corpus callosum in two sisters. J Med Genet. 1973;10(3):266–269. doi: 10.1136/jmg.10.3.266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Boettger T, Rust MB, Maier H, Seidenbecher T, Schweizer M, Keating DJ, Faulhaber J, Ehmke H, Pfeffer C, Scheel O, Lemcke B, Horst J, Leuwer R, Pape HC, Volkl H, Hubner CA, Jentsch TJ. Loss of K–Cl co-transporter KCC3 causes deafness, neurodegeneration and reduced seizure threshold. EMBO J. 2003;22(20):5422–5434. doi: 10.1093/emboj/cdg519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mathieu J, Bedard F, Prevost C, Langevin P. Motor and sensory neuropathies with or without agenesis of the corpus callosum: a radiological study of 64 cases. Can J Neurol Sci. 1990;17(2):103–108. [PubMed] [Google Scholar]
- 111.Dupre N, Howard HC, Mathieu J, Karpati G, Vanasse M, Bouchard JP, Carpenter S, Rouleau GA. Hereditary motor and sensory neuropathy with agenesis of the corpus callosum. Ann Neurol. 2003;54(1):9–18. doi: 10.1002/ana.77777. [DOI] [PubMed] [Google Scholar]
- 112.Howard HC, Mount DB, Rochefort D, Byun N, Dupre N, Lu J, Fan X, Song L, Riviere JB, Prevost C, Horst J, Simonati A, Lemcke B, Welch R, England R, Zhan FQ, Mercado A, Siesser WB, George AL, Jr, McDonald MP, Bouchard JP, Mathieu J, Delpire E, Rouleau GA. The K–Cl cotransporter KCC3 is mutant in a severe peripheral neuropathy associated with agenesis of the corpus callosum. Nat Genet. 2002;32(3):384–392. doi: 10.1038/ng1002. [DOI] [PubMed] [Google Scholar]
- 113.Salin-Cantegrel A, Riviere JB, Dupre N, Charron FM, Shekarabi M, Karemera L, Gaspar C, Horst J, Tekin M, Deda G, Krause A, Lippert MM, Willemsen MA, Jarrar R, Lapointe JY, Rouleau GA. Distal truncation of KCC3 in non-French Canadian HMSN/ACC families. Neurology. 2007;69(13):1350–1355. doi: 10.1212/01.wnl.0000291779.35643.15. [DOI] [PubMed] [Google Scholar]
- 114.Rudnik-Schoneborn S, Hehr U, von Kalle T, Bornemann A, Winkler J, Zerres K. Andermann syndrome can be a phenocopy of hereditary motor and sensory neuropathy—report of a discordant sibship with a compound heterozygous mutation of the KCC3 gene. Neuropediatrics. 2009;40(3):129–133. doi: 10.1055/s-0029-1234084. [DOI] [PubMed] [Google Scholar]
- 115.Steinlein OK, Neubauer BA, Sander T, Song L, Stoodt J, Mount DB. Mutation analysis of the potassium chloride cotransporter KCC3 (SLC12A6) in rolandic and idiopathic generalized epilepsy. Epilepsy Res. 2001;44(2–3):191–195. doi: 10.1016/s0920-1211(01)00230-3. [DOI] [PubMed] [Google Scholar]
- 116.Salin-Cantegrel A, Shekarabi M, Holbert S, Dion P, Rochefort D, Laganiere J, Dacal S, Hince P, Karemera L, Gaspar C, Lapointe JY, Rouleau GA. HMSN/ACC truncation mutations disrupt brain-type creatine kinase-dependant activation of K+/ Cl– co-transporter 3. Hum Mol Genet. 2008;17(17):2703–2711. doi: 10.1093/hmg/ddn172. [DOI] [PubMed] [Google Scholar]
- 117.Rinehart J, Maksimova YD, Tanis JE, Stone KL, Hodson CA, Zhang J, Risinger M, Pan W, Wu D, Colangelo CM, Forbush B, Joiner CH, Gulcicek EE, Gallagher PG, Lifton RP. Sites of regulated phosphorylation that control K–Cl cotransporter activity. Cell. 2009;138(3):525–536. doi: 10.1016/j.cell.2009.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Byun N, Delpire E. Axonal and periaxonal swelling precede peripheral neurodegeneration in KCC3 knockout mice. Neurobiol Dis. 2007;28(1):39–51. doi: 10.1016/j.nbd.2007.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Schwartzkroin PA, Baraban SC, Hochman DW. Osmolarity, ionic flux, and changes in brain excitability. Epilepsy Res. 1998;32(1–2):275–285. doi: 10.1016/s0920-1211(98)00058-8. [DOI] [PubMed] [Google Scholar]
- 120.Kang JQ, Macdonald RL. Making sense of nonsense GABA(A) receptor mutations associated with genetic epilepsies. Trends Mol Med. 2009;15(9):430–438. doi: 10.1016/j.molmed.2009.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kang JQ, Shen W, Macdonald RL. The GABRG2 mutation, Q351X, associated with generalized epilepsy with febrile seizures plus, has both loss of function and dominantnegative suppression. J Neurosci. 2009;29(9):2845–2856. doi: 10.1523/JNEUROSCI.4772-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Onat FY, Aker RG, Gurbanova AA, Ates N, van Luijtelaar G. The effect of generalized absence seizures on the progression of kindling in the rat. Epilepsia. 2007;48(Suppl 5):150–156. doi: 10.1111/j.1528-1167.2007.01303.x. [DOI] [PubMed] [Google Scholar]
- 123.Hsu FC, Zhang GJ, Raol YS, Valentino RJ, Coulter DA, Brooks-Kayal AR. Repeated neonatal handling with maternal separation permanently alters hippocampal GABAA receptors and behavioral stress responses. Proc Natl Acad Sci USA. 2003;100(21):12213–12218. doi: 10.1073/pnas.2131679100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Glasscock E, Qian J, Yoo JW, Noebels JL. Masking epilepsy by combining two epilepsy genes. Nat Neurosci. 2007;10(12):1554–1558. doi: 10.1038/nn1999. [DOI] [PubMed] [Google Scholar]
- 125.Inoue K, Khajavi M, Ohyama T, Hirabayashi S, Wilson J, Reggin JD, Mancias P, Butler IJ, Wilkinson MF, Wegner M, Lupski JR. Molecular mechanism for distinct neurological phenotypes conveyed by allelic truncating mutations. Nat Genet. 2004;36(4):361–369. doi: 10.1038/ng1322. [DOI] [PubMed] [Google Scholar]
- 126.Marini C, Harkin LA, Wallace RH, Mulley JC, Scheffer IE, Berkovic SF. Childhood absence epilepsy and febrile seizures: a family with a GABA(A) receptor mutation. Brain. 2003;126(Pt 1):230–240. doi: 10.1093/brain/awg018. [DOI] [PubMed] [Google Scholar]
- 127.Sun H, Zhang Y, Liang J, Liu X, Ma X, Wu H, Xu K, Qin J, Qi Y, Wu X. SCN1A, SCN1B, and GABRG2 gene mutation analysis in Chinese families with generalized epilepsy with febrile seizures plus. J Hum Genet. 2008;53(8):769–774. doi: 10.1007/s10038-008-0306-y. [DOI] [PubMed] [Google Scholar]