Abstract
This histopathologic case-control study was designed to characterize the dynamic changes in protein expression of nuclear factor-kappa B (NF-κB)/p65 subunit, macrophage inflammatory protein-2 (MIP-2), and matrix metalloproteinase-9 (MMP-9) in postmortem brains of patients with and without intracerebral hemorrhage (ICH). Thirty-six human brains from patients with ICH and six control brains were included in this study. We found that expression levels of NF-κB/p65, MIP-2, and MMP-9 were each upregulated on the injured side of the hippocampus at times ranging from 2 hr to 5 days post-ICH. Interestingly, the expression of all three markers was also upregulated on the uninjured side of the hippocampus and in the cerebellum, although to a lesser extent. These data suggest that inflammation occurs early and persists for several days after ICH in humans and could be involved in the progression of ICH-induced secondary brain damage.
Keywords: brain damage, intracerebral hemorrhage, MIP-2, MMP-9, NF-κB/p65
1. INTRODUCTION
Intracerebral hemorrhage (ICH) is one of the most common and devastating types of stroke. ICH causes secondary brain damage through various pathways, including inflammation-related mechanisms, the local release of reactive oxygen species (ROS), and activation of proteolytic enzymes (Wang and Doré, 2007; Xi et al., 2006). Nuclear factor-kappa B (NF-κB) is exquisitely sensitive to ROS and is rapidly activated in perihematomal brain after ICH in rats (Hickenbottom et al., 1999; Wagner, 2007). Activation of NF-κB leads to rapid and coordinated induction of proteolytic enzymes and release of proinflammatory cytokines and chemokines (Wagner, 2007). Most preclinical studies have indicated that proteolytic enzyme matrix metalloproteinase (MMP)-9 has a detrimental role (Rosenberg and Navratil, 1997; Wang and Tsirka, 2005; Xue et al., 2009). In addition, several clinical studies have assessed blood MMP-9 levels in patients with acute spontaneous ICH and found that the increase in blood MMP-9 levels within 12 or 24 hr after stroke onset is associated with subsequent enlargement of the hematoma, perihematomal edema, and deterioration of neurologic function (Abilleira et al., 2003; Alvarez-Sabin et al., 2004; Castellazzi et al., 2010; Silva et al., 2005). In our own studies using a blood model of ICH in rats, we found that NF-κB activation and expression of macrophage inflammatory protein (MIP)-2 and MMP-9 were increased at 3 hr and reached a maximum at 2 days after ICH (Wu et al., 2008; Wu et al., 2009).
To date, relatively little ICH histopathologic data have been obtained from human post-mortem specimens (Wang and Doré, 2007). There are no reports regarding NF-κB or MIP-2, and only two reports on MMP-9 in human ICH brain (Rosell et al., 2006; Tejima et al., 2007). The purpose of this histopathologic case-control study was to characterize the dynamic changes of the protein expression of NF-κB/p65 subunit, MIP-2, and MMP-9 by immunohistochemistry in postmortem brains of patients with and without ICH.
2. RESULTS
2.1 Histologic examination
In the postmortem human brain tissue, normal morphology of hippocampal CA1 neurons was observed in the brain sections from the control group (Fig. 1A); few morphologic changes were observed in hippocampal CA1 neurons at 2 hr after ICH (Fig. 1B). At 24 hr, obvious histopathologic changes were observed (Fig. 1C). Some neurons were pyknotic and triangular. Some neurons exhibited nuclear swelling and incomplete cellular structures. The inflammatory cells that had infiltrated the area were mainly mononuclear cells, with some neutrophils also present. At 120 hr after ICH, hyperplastic glial cells were present.
Fig. 1.
Hematoxylin and eosin staining in the CA1 region of hippocampus. (A) Control group: non-ischemic pyramidal neurons had normal morphology; (B) 2 hr after ICH: limited neurons were pyknotic; (C) 24 hr after ICH: some neurons were triangular and pyknotic. Scale bar = 50 µm.
2.2 Expression of NF-κB/p65
Activation of NF-κB after ICH was assessed by immunohistochemistry by using an antibody specific to activated NF-κB/p65 subunit, the epitope of which binds only after IκB dissociation. In the cerebellum, NF-κB/p65 immunoreactivity was observed in cell bodies of Purkinje cells and some white matter neurons (Fig. 2). Some NF-κB/p65-positive cells were found in the granular layer. In the hippocampus, NF-κB/p65 immunoreactivity was observed in the cytoplasm of pyramidal neurons and was mild in the control group (Fig. 3). After ICH, NF-κB/p65 immunoreactivity was detected primarily in neurons and infrequently in glial cells and vasculature. It was evident that NF-κB/p65 had translocated from the cytosol to the nucleus in a few neurons. NF-κB/p65 immunoreactivity varied with time after ICH. On the injured side of the hippocampal CA1 region, the number of immunostained neurons was increased at 2–6 hr after ICH, reached a maximum at 7–24 hr after ICH (P < 0.01 compared with control group and the previous time point), decreased somewhat after that, and then increased again at 49–120 hr after ICH (Table I). On the uninjured side of the hippocampal CA1 region, the number of immunostained neurons increased at 7–24 hr after ICH (P < 0.01 compared with control group), decreased somewhat, and then reached a maximum at 49–120 hr after ICH (P < 0.01 compared with control group and the previous time point; Table I). In the cerebellum, the number of immunostained neurons reached a maximum at 7–24 hr after ICH (P < 0.01 compared with control group; Table I). More NF-κB/p65 immunoreactive neurons were observed on the injured side of the hippocampus than on the uninjured side of the hippocampus or in the cerebellum.
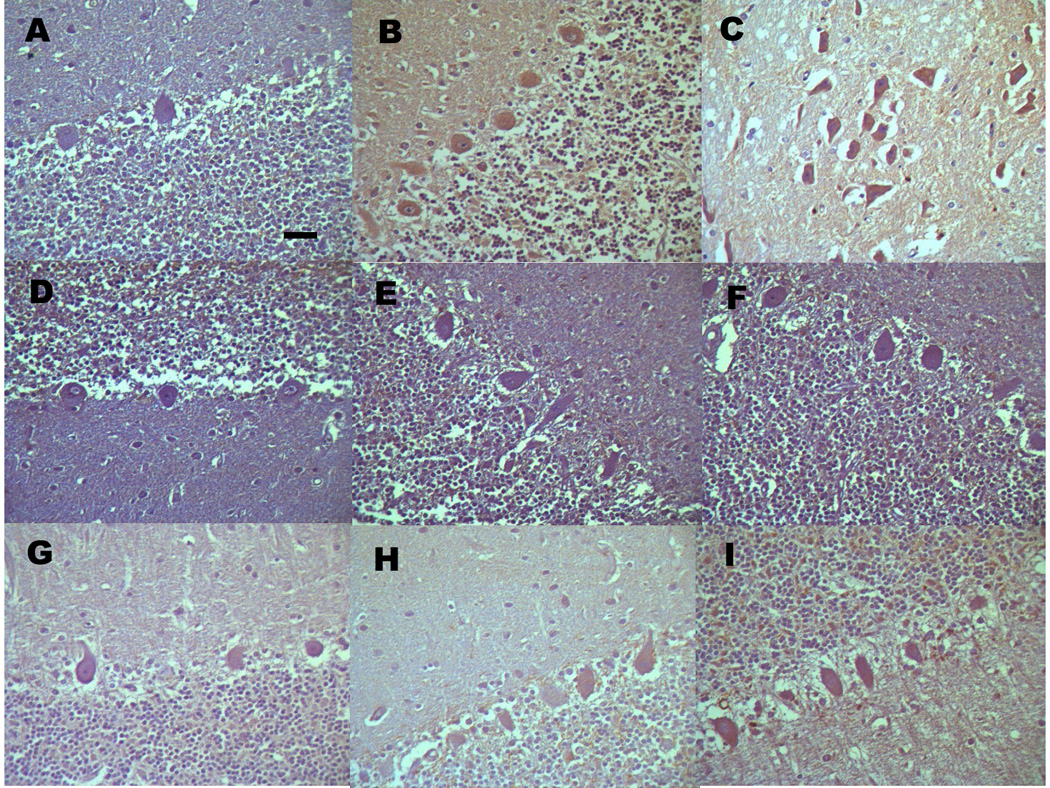
Fig. 2.
Effect of ICH on immunoreactivity of NF-κB/p65, MIP-2, and MMP-9 in the cerebellum. (A) Protein expression of NF-κB/p65 subunit in cerebellum of the control group. The immunopositive cells showed brown staining. (B) After ICH, NF-κB/p65 immunoreactivity increased and appeared in cell bodies of Purkinje cells in the cerebellum. (C) NF-κB/p65 immunoreactivity increased in some white matter neurons in the cerebellum after ICH. (D) Immunoreactivity of MIP-2 in cerebellum of the control group. (E) MIP-2 immunoreactivity was elevated in cerebellum at 2 hr after ICH. (F) MIP-2 immunoreactivity was elevated in cell bodies of Purkinje cells at 24 hr after ICH. (G) Immunoreactivity of MMP-9 in cerebellum of the control group. (H) Immunoreactivity of MMP-9 was elevated in cell bodies of Purkinje cells in the cerebellum at 2 hr after ICH. (I) Immunoreactivity of MMP-9 was elevated in the cerebellum at 24 hr after ICH. Scale bar = 50 µm.
Fig. 3.
Effect of ICH on immunoreactivity of NF-κB/p65, MIP-2, and MMP-9 in the hippocampus. (A) Protein expression of NF-κB/p65 subunit in hippocampus of the control group. NF-κB/p65 immunoreactivity was elevated on the injured side of the hippocampus at 2 hr (B) and 24 hr (C) after ICH. (D) MIP-2 immunoreactivity in hippocampus of the control group. MIP-2 immunoreactivity was elevated on the injured side of the hippocampus 2 hr (E) and 24 hr (F) after ICH. (G) Immunoreactivity of MMP-9 in hippocampus of the control group. Immunoreactivity of MMP-9 was elevated on the injured side of the hippocampus 2 hr (H) and 24 hr (I) after ICH. Scale bar = 50 µm.
Table I.
Immunoreactivity of NF-κB, MIP-2, and MMP-9 in the hippocampus and cerebellum
| Marker | Time between ICH and death |
Case number |
Number of immunopositive neurons per 40× field | ||
|---|---|---|---|---|---|
| Injured side of hippocampus |
Uninjured side of hippocampus |
Cerebellum | |||
| NF- κB | Control | 6 | 5.0±0.7 | 5.0±0.7 | 1.4±0.5 |
| 2–6 hr | 5 | 21.0±5.0** | 8.8±2.7** | 5.2±1.3** | |
| 7–24 hr | 13 | 35.6±9.5**†† | 11.8±5.3** | 6.2±1.3** | |
| 25–48 hr | 3 | 18.4±1. 7**†† | 6.2±1.5†† | 4.2±1.9* | |
| 49–120 hr | 7 | 22.2±3.7**† | 17.0±4.7**†† | 4.8±1.9** | |
| 121–240 hr | 2 | 6.6±2.3†† | 3.6±1.5†† | 4.0±2.2* | |
| MIP-2 | Control | 6 | 8.8±1.9 | 8.8±1.9 | 3.2±1.3 |
| 2–6 hr | 5 | 22.4±4.4** | 10.4±1.5 | 4.8±1.3 | |
| 7–24 hr | 13 | 15.2±4.3**†† | 11.4±1.3 | 5.2±0.8* | |
| 25–48 hr | 3 | 12.4±1.1* | 7.2±1.5†† | 3.2±0.8† | |
| 49–120 hr | 7 | 15.2±1.8** | 11.6±2.2*†† | 4.8±1.6 | |
| 121–240 hr | 2 | 10.6±1.1†† | 8.8±1.9 | 4.6±1.1 | |
| MMP-9 | Control | 6 | 9.4±4.7 | 9.4±4.7 | 4.0±2.4 |
| 2–6 hr | 5 | 19.0±5.8** | 4.4±1.7** | 6.6±1.7* | |
| 7–24 hr | 13 | 30.6±4.0**†† | 15.4±3.7**†† | 9.8±1.5**†† | |
| 25–48 hr | 3 | 19.6±3.2**†† | 8.2±3.0†† | 4.8±2.2†† | |
| 49–120 hr | 7 | 17.4±3.2** | 9.0±2.7 | 4.6±1.1 | |
| 121–240 hr | 2 | 11.2±3.1†† | 3.0±1.6**†† | 6.0±1.6 | |
Data are mean ± SD; ICH, intracerebral hemorrhage.
P < 0.05 compared with control group,
P < 0.01 compared with control group.
P < 0.05,
P < 0.01 compared with the previous time point.
2.3 Expression of MIP-2
MIP-2 immunoreactivity was mild in the brain sections from the control group. In the cerebellum, MIP-2 protein was present in cell bodies of Purkinje cells and in some large white matter neurons. Some immunoreactivity was observed in the granular layer (Fig. 2). In the hippocampus, MIP-2 immunoreactivity was observed in cell bodies and extended into the major axons of pyramidal neurons in the CA1–CA3 layers and the subiculum. Furthermore, some intense positive puncta were observed in cell bodies of pyramidal neurons (Fig. 3). Mild MIP-2 immunoreactivity was present in the cytoplasm of a few glia cells. On the injured side of the hippocampal CA1 region, the number of MIP-2 immunostained neurons reached a maximum at 2–6 hr after ICH (P < 0.01 compared with control group; Table I). On the uninjured side of the hippocampal CA1 region, the number of immunostained neurons reached a maximum at 49–120 hr after ICH (P < 0.05 compared with control group and the previous time point; Table I). In the cerebellum, the number of immunostained neurons reached a maximum at 7–24 hr after ICH (P < 0.05 compared with control group; Table I). The number of MIP-2-positive neurons on the injured side of the hippocampus was much greater than that on the uninjured side of the hippocampus or in the cerebellum after ICH.
2.4 Expression of MMP-9
MMP-9 immunoreactivity was mild in the brain sections from the control group. In the cerebellum, MMP-9 protein was expressed in cell bodies of Purkinje cells and in some white matter neurons (Fig. 2). Some immunoreactive cells were found in the granular layer. After ICH, MMP-9 immunoreactivity appeared as intense positive puncta in cell bodies of pyramidal neurons on the injured and uninjured sides of the hippocampus (Fig. 3). Faint MMP-9 immunoreactivity was observed in the cytoplasm of a few glia cells. On the injured and uninjured sides of the hippocampal CA1 region and in the cerebellum, the number of MMP-9-immunostained neurons was increased at 2–6 hr after ICH and reached a maximum at 7–24 hr after ICH (P < 0.01 compared with control group and the previous time point; Table I). The number of MMP-9-positive neurons on the injured side of the hippocampus was much greater than that on the uninjured side of the hippocampus or in the cerebellum (Table I).
3. DISCUSSION
We systematically assessed the protein expression of NF-κB/p65, MIP-2, and MMP-9 to elucidate the changes that occur after ICH in a histopathologic study of patients with and without ICH. By using immunohistochemistry, we found that NF-κB/p65, MIP-2, and MMP-9 protein expression increased on the injured side of the hippocampus over the course of 2 hr to 5 days after ICH. The expression of these three markers was also upregulated, although to a lesser extent, on the uninjured side of the hippocampus and in the cerebellum. To our knowledge, we are the first to examine the dynamic changes of NF-κB/p65, MIP-2, and MMP-9 after ICH in the human brain. Our data suggest that neuroinflammation occurs early and persists for several days after ICH in humans. In addition, neuroinflammation after ICH could affect the normal function of the entire brain, as brain regions remote from the hematoma were also affected.
Evidence suggests that ICH-induced thrombin released at protease-activated receptors could activate NF-κB (Paria et al., 2006; Thomas and Brugge, 1997). In two early studies, investigators showed that NF-κB was activated in neurons early after ICH in rats (Hickenbottom et al., 1999; Zhao et al., 2007). Consistent with their results, we revealed that NF-κB/p65 immunoreactivity in the perilesional hippocampal CA1 region increased as early as 2 hr and peaked at 7–24 hr after ICH; this increase occurred not only on the injured side of the hippocampus, but also on the uninjured side of the hippocampus and in the cerebellum. Activation of NF-κB could lead to perihematomal inflammation and cell death (Hickenbottom et al., 1999), possibly mediated by rapid induction of proteolytic enzymes and local release of proinflammatory cytokines and chemokines (Wagner, 2007). Here we demonstrated that ICH-triggered activation of NF-κB was correlated with increased immunoreactivity of chemokine MIP-2 and proteolytic enzyme MMP-9 in the human brain.
The transcription factor NF-κB is a major regulator of chemokine gene expression (Silverman and Maniatis, 2001). NF-κB binding sites are located in the transcription start site of the MIP-2 gene. Therefore, MIP-2 protein expression is dependent on and regulated by NF-κB (Driscoll, 2000; Zampetaki et al., 2004). As a chemoattractant (Diab et al., 1999), MIP-2 is an activating factor for neutrophils (Tekamp-Olson et al., 1990). Recent studies have demonstrated that MIP-2 can be expressed in neurons, astrocytes, and macrophages under various conditions (Rhodes et al., 2009; Tomita et al., 2005; Wu et al., 2009; Zampetaki et al., 2004). We have provided the first evidence that the immunoreactivity of MIP-2 is increased primarily in neurons, earlier and to a much greater extent on the injured side of the hippocampus than on the uninjured side of the hippocampus or cerebellum in the human ICH brain. Previously, we observed a similar upregulation of MIP-2 associated with increased brain edema formation in a rat blood model (Wu et al., 2009). As an important chemotactic chemokine, MIP-2 could be involved in ICH-induced secondary brain injury.
MMP-9 expression can be regulated by NF-κB, as the NF-κB response element is located in the promoter region of the MMP-9 gene (Rangaswami et al., 2006). This scenario was supported by our data, which showed that on the injured side of the hippocampus, protein expression of NF-κB/p65 and MMP-9 peaked in parallel at 7–24 hr after ICH. Protein expression of MMP-9 could also be regulated by MIP-2, as MIP-2 is one of the major neutrophil chemotactic proteins (Bell et al., 1996), and neutrophils appear to be a major source of MMP-9 after brain injury (Chen et al., 2006).
In preclinical and clinical ICH studies, MMP-9 has been associated with poor outcomes (Abilleira et al., 2003; Alvarez-Sabin et al., 2004; Castellazzi et al., 2010; Rosell et al., 2006; Silva et al., 2005; Tejima et al., 2007; Wang et al., 2003; Wang and Tsirka, 2005; Wang and Doré, 2007; Xue and Yong, 2008). In the brain, activated MMP-9 contributes to the perihematomal edema formation and neuronal death that occur after hemorrhagic stroke (Wang and Doré, 2007). Two studies of MMPs in human ICH brain (within the first 6 hr after death) showed that, compared with the contralateral hemisphere, tissue around the hemorrhage had higher MMP levels in close relationship with neurons and glial cells (Rosell et al., 2006; Tejima et al., 2007). Here, our data demonstrate that MMP-9 immunoreactivity increases in pyramidal neurons on the injured side of the hippocampus, reaching a maximum at 7–24 hr after ICH. This increase corresponds in time with the increase in blood MMP-9 levels seen in patients with acute ICH (Abilleira et al., 2003; Alvarez-Sabin et al., 2004; Castellazzi et al., 2010; Silva et al., 2005).
Our study has limitations. First, hippocampus and cerebellum were the only brain tissues that could be obtained from our brain bank. For our future studies, we intend to obtain brain tissue from the perihematomal region as well as from the uninjured side of the hippocampus and cerebellum. Second, we assessed selected inflammatory markers in patients who did not survive ICH; the time course and extent of inflammation may differ from those who survived with smaller hematoma and less brain injury. Third, we did not use a stereologic technique to quantify immunopositive cells because the apparatus is unavailable at our university. To reduce bias, immunopositive cells were quantified by two experienced neuropathologists who were blinded to the tissue identity.
In conclusion, our results provide evidence that upregulation of NF-κB/p65, MIP-2, and MMP-9 occurs very early and persists for several days after ICH in humans. In addition, neuroinflammation following ICH could affect the normal function of the entire brain. These observations support the possibility that a therapeutic strategy targeting neuroinflammation could offer new promise for ICH patients.
4. Experimental procedures
4.1 Collection of human brain tissue
All specimens were from the brain bank in the Department of Neuropathology, the First Hospital of Harbin Medical University, China. Sections of formaldehyde-fixed human brains were obtained from 30 fatal cases of ICH (age at death, 36–70 years; 20 male; 10 female; Table II). Patients with hematoma secondary to head injury, congenital or acquired coagulation abnormalities, known acute or chronic infections, inflammatory or malignant diseases, or immunosuppressive treatment were excluded from the study. Brains from six patients who had died of other non-cerebrovascular-related diseases were used as controls (mean age at death 48.5 years, 28–69 years; all males). The interval between death and autopsy was less than 24 hr. For each case, injured and uninjured sides of the hippocampus and cerebellar specimens were examined. After being dehydrated in graded ethanol and cleared in xylene, the selected brain tissues were embedded in paraffin and cut into 4-µm-thick coronal sections. Ipsilateral hippocampus was used to represent the perihematomal region, though not typically immediate perihematomal region; contralateral hippocampus and cerebellum represented the more distal peripheral region. Based on the interval between the onset of clinical symptoms and death, the specimens were divided into five groups as follows: 2–6 hr group (n=5), 7–24 hr group (n=13), 25–48 hr group (n=3), 49–120 hr group (n=7), and 121–240 hr group (n=2). This study was approved by the Ethics Committee of the hospital, and informed consent was obtained from relatives before the autopsy.
Table II.
Summary of clinical data
| Case | Age (years) | Sex | Time between ICH and patient death (hr) |
ICH lesion location |
|---|---|---|---|---|
| 1 | 58 | M | 2 | Lt Pu |
| 2 | 61 | M | 2 | Rt Pu |
| 3 | 56 | M | 3 | Po |
| 4 | 57 | M | 5 | Lt Tl,Pl |
| 5 | 58 | F | 5 | Lt Tl |
| 6 | 58 | M | 7 | Lt Bg |
| 7 | 60 | M | 9 | Po,Rt Pu |
| 8 | 70 | M | 9 | Po,Rt Pu |
| 9 | 60 | M | 9 | Po |
| 10 | 60 | M | 10 | Lt Pu,Tp |
| 11 | 70 | F | 10 | El |
| 12 | 59 | F | 10 | El |
| 13 | 61 | M | 11 | Lt Ci |
| 14 | 48 | M | 11 | Rt Pu |
| 15 | 63 | M | 12 | Po |
| 16 | 39 | M | 16 | El,Sa |
| 17 | 57 | F | 23 | Rt Pu |
| 18 | 42 | M | 24 | Lt Tp |
| 19 | 58 | M | 26 | Rt Fl,Tl,Ol |
| 20 | 81 | M | 30 | Lt Pu |
| 21 | 48 | M | 36 | Lt Fl,Tl,Ol |
| 22 | 47 | M | 72 | Po |
| 23 | 60 | F | 81 | Rt Pu |
| 24 | 84 | F | 84 | Lt Pu,Fl |
| 25 | 57 | M | 96 | Rt Pu |
| 26 | 66 | F | 96 | Rt Tp,Bs |
| 27 | 66 | F | 108 | Rt Pu |
| 28 | 36 | M | 120 | Lt Pu,Po |
| 29 | 59 | F | 130 | Po |
| 30 | 60 | F | 240 | Lt Pu,Tp |
| 31 | 28 | M | control | |
| 32 | 46 | M | control | |
| 33 | 58 | M | control | |
| 34 | 36 | M | control | |
| 35 | 54 | M | control | |
| 36 | 69 | M | control |
Age, age at death; ICH, intracerebral hemorrhage; Lt, left; Rt, right; Fl, frontal lobe; Pl, parietal lobe; Tl, temporal lobe; Ol, occipital lobe; Pu, putamen; Po, pontine; Bg, basal ganglia; Tp, thalamencephalon; El, encephalocoele; Ci, capsula interna nuclei lentiformis; Sa, subarachnoid cavity; Bs, brainstem.
4.2 Histologic Examination
Coronal brain sections from different levels of the hippocampus were dewaxed and stained with hematoxylin and eosin according to standard procedures (Wu et al., 2008). The extent of neuronal damage to hippocampus was examined under an Olympus BX51 light microscope (Olympus, Tokyo, Japan).
4.3 Immunohistochemistry
Immunohistochemistry was carried out with the following primary antibodies: goat anti-MMP-9 polyclonal antibody (1:100, Santa Cruz); goat anti-MIP-2 polyclonal antibody (1:100, Santa Cruz), and rabbit anti-NF-κB/p65 subunit polyclonal antibody (1:100, Santa Cruz). The tissue sections were dewaxed and rehydrated, rinsed with distilled water and phosphate-buffered saline (PBS), quenched with 3% H2O2, and then exposed to the primary antibodies and incubated at 4°C overnight. Sections were washed with Triton-PBS, incubated in biotinylated goat anti-rabbit IgG or rabbit anti-goat IgG (DAKO) for 1 hr at room temperature, washed again, and incubated with streptavidin-peroxidase for 30 min at room temperature. Finally, the sections were stained with diaminobenzidine-H2O2 solution, washed, dehydrated in graded ethanol, immersed in xylene, and covered with a cover slip. Control sections were processed without the primary antibodies.
4.4 Cell counting and statistical analysis
Two experienced neuropathologists blinded to the specimen group examined histologic and morphometric parameters in brain sections under light microscopy. To perform the quantification analysis of immunopositive cells, four brain sections from different levels of the hippocampus and cerebellum were randomly chosen, and positively stained cells over a microscopic field of 40× were counted in three comparable non-overlapping fields. Based on cell body diameter and morphology, only positively immunostained neurons were counted. The number of immunostained neurons in 12 locations of the hippocampal CA1 subfield or cerebellum (three fields per section and four sections per brain) were averaged and expressed as positive neurons/field. All data are reported as mean ± standard deviation (SD) of the number of positively immunostained neurons per 40× field. The statistical significance of the data was determined by analysis of variance (ANOVA) and post hoc tests with SPSS 13.0 for Windows (SPSS Inc, Chicago). Statistical significance was set at P < 0.05.
Acknowledgments
We thank Claire Levine for assistance with this manuscript.
Grant information:
This work was supported by the Natural Science Foundation of Heilongjiang Province ZJY0705 and the Foundation of the First Clinical Hospital of Harbin Medical University Y08-009 (HW); and AHA 09BGIA2080137 and NIH K01AG031926 (JW).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Abilleira S, Montaner J, Molina CA, Monasterio J, Castillo J, Alvarez-Sabin J. Matrix metalloproteinase-9 concentration after spontaneous intracerebral hemorrhage. J Neurosurg. 2003;99:65–70. doi: 10.3171/jns.2003.99.1.0065. [DOI] [PubMed] [Google Scholar]
- Alvarez-Sabin J, Delgado P, Abilleira S, Molina CA, Arenillas J, Ribo M, Santamarina E, Quintana M, Monasterio J, Montaner J. Temporal profile of matrix metalloproteinases and their inhibitors after spontaneous intracerebral hemorrhage: relationship to clinical and radiological outcome. Stroke. 2004;35:1316–1322. doi: 10.1161/01.STR.0000126827.69286.90. [DOI] [PubMed] [Google Scholar]
- Bell MD, Taub DD, Kunkel SJ, Strieter RM, Foley R, Gauldie J, Perry VH. Recombinant human adenovirus with rat MIP-2 gene insertion causes prolonged PMN recruitment to the murine brain. Eur J Neurosci. 1996;8:1803–1811. doi: 10.1111/j.1460-9568.1996.tb01324.x. [DOI] [PubMed] [Google Scholar]
- Castellazzi M, Tamborino C, De Santis G, Garofano F, Lupato A, Ramponi V, Trentini A, Casetta I, Bellini T, Fainardi E. Timing of serum active MMP-9 and MMP-2 levels in acute and subacute phases after spontaneous intracerebral hemorrhage. Acta Neurochir Suppl. 2010;106:137–140. doi: 10.1007/978-3-211-98811-4_24. [DOI] [PubMed] [Google Scholar]
- Chen Y, Fan Y, Poon KY, Achrol AS, Lawton MT, Zhu Y, McCulloch CE, Hashimoto T, Lee C, Barbaro NM, Bollen AW, Yang GY, Young WL. MMP-9 expression is associated with leukocytic but not endothelial markers in brain arteriovenous malformations. Front Biosci. 2006;11:3121–3128. doi: 10.2741/2037. [DOI] [PubMed] [Google Scholar]
- Diab A, Abdalla H, Li HL, Shi FD, Zhu J, Hojberg B, Lindquist L, Wretlind B, Bakhiet M, Link H. Neutralization of macrophage inflammatory protein 2 (MIP-2) and MIP-1alpha attenuates neutrophil recruitment in the central nervous system during experimental bacterial meningitis. Infect Immun. 1999;67:2590–2601. doi: 10.1128/iai.67.5.2590-2601.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driscoll KE. TNFalpha and MIP-2: role in particle-induced inflammation and regulation by oxidative stress. Toxicol Lett. 2000:112–113. 177–183. doi: 10.1016/s0378-4274(99)00282-9. [DOI] [PubMed] [Google Scholar]
- Hickenbottom SL, Grotta JC, Strong R, Denner LA, Aronowski J. Nuclear factor-kappaB and cell death after experimental intracerebral hemorrhage in rats. Stroke. 1999;30:2472–2477. doi: 10.1161/01.str.30.11.2472. discussion 2477–8. [DOI] [PubMed] [Google Scholar]
- Paria BC, Bair AM, Xue J, Yu Y, Malik AB, Tiruppathi C. Ca2+ influx induced by protease-activated receptor-1 activates a feed-forward mechanism of TRPC1 expression via nuclear factor-kappaB activation in endothelial cells. J Biol Chem. 2006;281:20715–20727. doi: 10.1074/jbc.M600722200. [DOI] [PubMed] [Google Scholar]
- Rangaswami H, Bulbule A, Kundu GC. Nuclear factor inducing kinase: a key regulator in osteopontin- induced MAPK/IkappaB kinase dependent NF-kappaB-mediated promatrix metalloproteinase-9 activation. Glycoconj J. 2006;23:221–232. doi: 10.1007/s10719-006-7927-1. [DOI] [PubMed] [Google Scholar]
- Rhodes JK, Sharkey J, Andrews PJ. The temporal expression, cellular localization, and inhibition of the chemokines MIP-2 and MCP-1 after traumatic brain injury in the rat. J Neurotrauma. 2009;26:507–525. doi: 10.1089/neu.2008.0686. [DOI] [PubMed] [Google Scholar]
- Rosell A, Ortega-Aznar A, Alvarez-Sabin J, Fernandez-Cadenas I, Ribo M, Molina CA, Lo EH, Montaner J. Increased brain expression of matrix metalloproteinase-9 after ischemic and hemorrhagic human stroke. Stroke. 2006;37:1399–1406. doi: 10.1161/01.STR.0000223001.06264.af. [DOI] [PubMed] [Google Scholar]
- Rosenberg GA, Navratil M. Metalloproteinase inhibition blocks edema in intracerebral hemorrhage in the rat. Neurology. 1997;48:921–926. doi: 10.1212/wnl.48.4.921. [DOI] [PubMed] [Google Scholar]
- Silva Y, Leira R, Tejada J, Lainez JM, Castillo J, Davalos A. Molecular signatures of vascular injury are associated with early growth of intracerebral hemorrhage. Stroke. 2005;36:86–91. doi: 10.1161/01.STR.0000149615.51204.0b. [DOI] [PubMed] [Google Scholar]
- Silverman N, Maniatis T. NF-kappaB signaling pathways in mammalian and insect innate immunity. Genes Dev. 2001;15:2321–2342. doi: 10.1101/gad.909001. [DOI] [PubMed] [Google Scholar]
- Tejima E, Zhao BQ, Tsuji K, Rosell A, van Leyen K, Gonzalez RG, Montaner J, Wang X, Lo EH. Astrocytic induction of matrix metalloproteinase-9 and edema in brain hemorrhage. J Cereb Blood Flow Metab. 2007;27:460–468. doi: 10.1038/sj.jcbfm.9600354. [DOI] [PubMed] [Google Scholar]
- Tekamp-Olson P, Gallegos C, Bauer D, McClain J, Sherry B, Fabre M, van Deventer S, Cerami A. Cloning and characterization of cDNAs for murine macrophage inflammatory protein 2 and its human homologues. J Exp Med. 1990;172:911–919. doi: 10.1084/jem.172.3.911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas SM, Brugge JS. Cellular functions regulated by Src family kinases. Annu Rev Cell Dev Biol. 1997;13:513–609. doi: 10.1146/annurev.cellbio.13.1.513. [DOI] [PubMed] [Google Scholar]
- Tomita M, Holman BJ, Santoro CP, Santoro TJ. Astrocyte production of the chemokine macrophage inflammatory protein-2 is inhibited by the spice principle curcumin at the level of gene transcription. J Neuroinflammation. 2005;2:8. doi: 10.1186/1742-2094-2-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner KR. Modeling intracerebral hemorrhage: glutamate, nuclear factor-kappa B signaling and cytokines. Stroke. 2007;38:753–758. doi: 10.1161/01.STR.0000255033.02904.db. [DOI] [PubMed] [Google Scholar]
- Wang J, Rogove AD, Tsirka AE, Tsirka SE. Protective role of tuftsin fragment 1–3 in an animal model of intracerebral hemorrhage. Ann Neurol. 2003;54:655–664. doi: 10.1002/ana.10750. [DOI] [PubMed] [Google Scholar]
- Wang J, Tsirka SE. Neuroprotection by inhibition of matrix metalloproteinases in a mouse model of intracerebral haemorrhage. Brain. 2005;128:1622–1633. doi: 10.1093/brain/awh489. [DOI] [PubMed] [Google Scholar]
- Wang J, Doré S. Inflammation after intracerebral hemorrhage. J Cereb Blood Flow Metab. 2007;27:894–908. doi: 10.1038/sj.jcbfm.9600403. [DOI] [PubMed] [Google Scholar]
- Wu H, Zhao R, Qi J, Cong Y, Wang D, Liu T, Gu Y, Ban X, Huang Q. The expression and the role of protease nexin-1 on brain edema after intracerebral hemorrhage. J Neurol Sci. 2008;270:172–183. doi: 10.1016/j.jns.2008.03.010. [DOI] [PubMed] [Google Scholar]
- Wu H, Cong Y, Wang D, Zhao R, Qi J. Correlation of macrophage inflammatory protein-2 expression and brain edema in rats after intracerebral hemorrhage. Int J Clin Exp Pathol. 2009;2:83–90. [PMC free article] [PubMed] [Google Scholar]
- Xi G, Keep RF, Hoff JT. Mechanisms of brain injury after intracerebral haemorrhage. Lancet Neurol. 2006;5:53–63. doi: 10.1016/S1474-4422(05)70283-0. [DOI] [PubMed] [Google Scholar]
- Xue M, Yong VW. Matrix metalloproteinases in intracerebral hemorrhage. Neurol Res. 2008;30:775–782. doi: 10.1179/174313208X341102. [DOI] [PubMed] [Google Scholar]
- Xue M, Fan Y, Liu S, Zygun DA, Demchuk A, Yong VW. Contributions of multiple proteases to neurotoxicity in a mouse model of intracerebral haemorrhage. Brain. 2009;132:26–36. doi: 10.1093/brain/awn215. [DOI] [PubMed] [Google Scholar]
- Zampetaki A, Mitsialis SA, Pfeilschifter J, Kourembanas S. Hypoxia induces macrophage inflammatory protein-2 (MIP-2) gene expression in murine macrophages via NF-kappaB: the prominent role of p42/p44 and PI3 kinase pathways. Faseb J. 2004;18:1090–1092. doi: 10.1096/fj.03-0991fje. [DOI] [PubMed] [Google Scholar]
- Zhao X, Zhang Y, Strong R, Zhang J, Grotta JC, Aronowski J. Distinct patterns of intracerebral hemorrhage-induced alterations in NF-kappaB subunit, iNOS, and COX-2 expression. J Neurochem. 2007;101:652–663. doi: 10.1111/j.1471-4159.2006.04414.x. [DOI] [PubMed] [Google Scholar]