Abstract
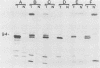
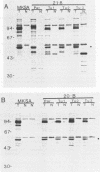
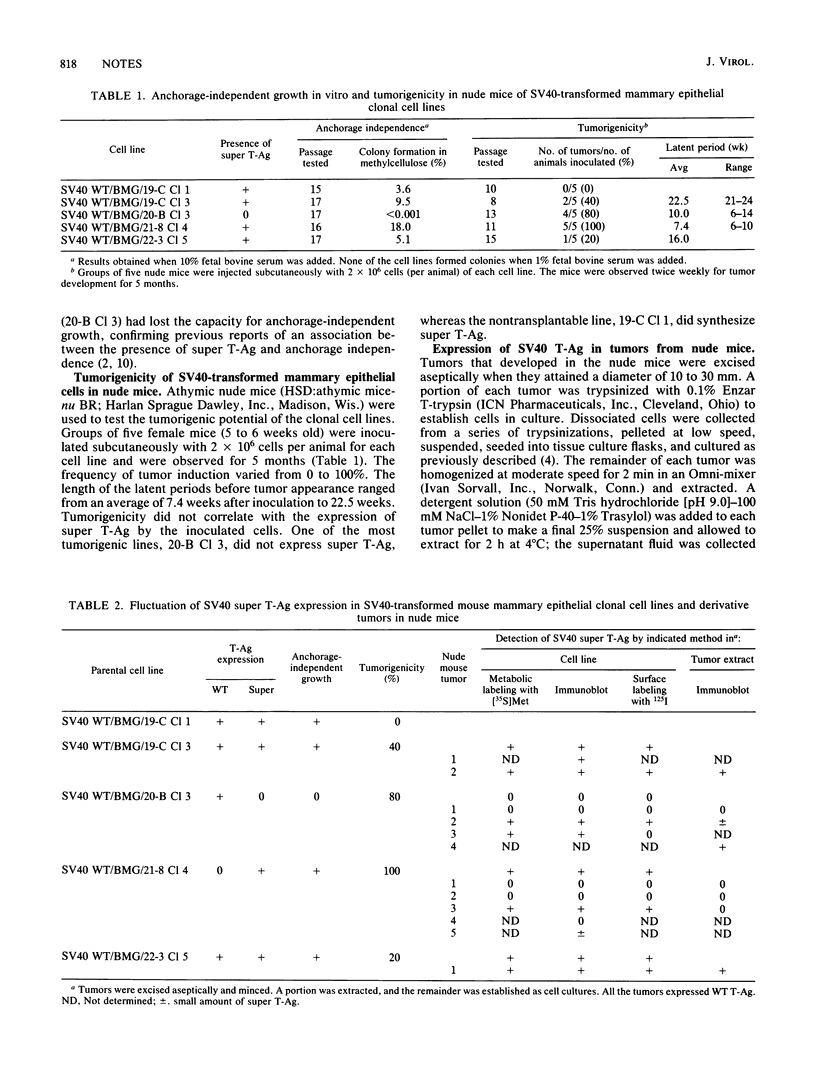
Higher-molecular-weight forms of the simian virus 40 (SV40) large tumor antigen (T-Ag), designated super T-Ag, are commonly found in SV40-transformed rodent cells. We examined the potential role of super T-Ag in neoplastic progression by using a series of clonal SV40-transformed mouse mammary epithelial cell lines. We confirmed an association between the presence of super T-Ag and cellular anchorage-independent growth in methylcellulose. However, tumorigenicity in nude mice did not correlate with the expression of super T-Ag. In the tumors that developed in nude mice, super T-Ag expression fluctuated almost randomly. Cell surface iodination showed that super T-Ag molecules were transported to the epithelial cell surface. The biological functions of super T-Ag remain obscure, but it is clear that it is not important for tumorigenicity by SV40-transformed mouse mammary epithelial cells. Super T-Ag may be most important as a marker of genomic rearrangements by the resident viral genes in transformed cells.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bender M. A., Brockman W. W. Rearrangement of integrated viral DNA sequences in mouse cells transformed by simian virus 40. J Virol. 1981 Jun;38(3):872–879. doi: 10.1128/jvi.38.3.872-879.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blanck G., Chen S., Pollack R. Integration, loss, and reacquisition of defective viral DNA in SV40-transformed mouse cell lines. Virology. 1983 Apr 30;126(2):413–428. doi: 10.1016/s0042-6822(83)80001-4. [DOI] [PubMed] [Google Scholar]
- Butel J. S., Wong C., Medina D. Transformation of mouse mammary epithelial cells by papovavirus SV40. Exp Mol Pathol. 1984 Feb;40(1):79–108. doi: 10.1016/0014-4800(84)90068-6. [DOI] [PubMed] [Google Scholar]
- Chandrasekaran K., Winterbourne D. J., Luborsky S. W., Mora P. T. Surface proteins of simian-virus-40-transformed cells. Int J Cancer. 1981 Mar 15;27(3):397–407. doi: 10.1002/ijc.2910270320. [DOI] [PubMed] [Google Scholar]
- Chang C., Simmons D. T., Martin M. A., Mora P. T. Identification and partial characterization of new antigens from simian virus 40-transformed mouse cells. J Virol. 1979 Aug;31(2):463–471. doi: 10.1128/jvi.31.2.463-471.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaudry F., Belsham G. J., Smith A. E. Biochemical properties of the 145,000-dalton super-T antigen from simian virus 40-transformed BALB/c 3T3 clone 20 cells. J Virol. 1983 Mar;45(3):1098–1106. doi: 10.1128/jvi.45.3.1098-1106.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S., Blanck G., Pollack R. Reacquisition of a functional early region by a mouse transformant containing only defective simian virus 40 DNA. Mol Cell Biol. 1984 Apr;4(4):666–670. doi: 10.1128/mcb.4.4.666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S., Grass D. S., Blanck G., Hoganson N., Manley J. L., Pollack R. E. A functional simian virus 40 origin of replication is required for the generation of a super T antigen with a molecular weight of 100,000 in transformed mouse cells. J Virol. 1983 Nov;48(2):492–502. doi: 10.1128/jvi.48.2.492-502.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S., Verderame M., Lo A., Pollack R. Nonlytic simian virus 40-specific 100K phosphoprotein is associated with anchorage-independent growth in simian virus 40-transformed and revertant mouse cell lines. Mol Cell Biol. 1981 Nov;1(11):994–1006. doi: 10.1128/mcb.1.11.994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clayton C. E., Lovett M., Rigby P. W. Functional analysis of a simian virus 40 super T-antigen. J Virol. 1982 Dec;44(3):974–982. doi: 10.1128/jvi.44.3.974-982.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clayton C. E., Rigby P. W. Cloning and characterization of the integrated viral DNA from three lines of SV40-transformed mouse cells. Cell. 1981 Aug;25(2):547–559. doi: 10.1016/0092-8674(81)90073-8. [DOI] [PubMed] [Google Scholar]
- Deppert W., Hanke K., Henning R. Simian virus 40 T-antigen-related cell surface antigen: serological demonstration on simian virus 40-transformed monolayer cells in situ. J Virol. 1980 Aug;35(2):505–518. doi: 10.1128/jvi.35.2.505-518.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiscott J., Murphy D., Defendi V. Amplification and rearrangement of integrated SV40 DNA sequences accompany the selection of anchorage-independent transformed mouse cells. Cell. 1980 Nov;22(2 Pt 2):535–543. doi: 10.1016/0092-8674(80)90363-3. [DOI] [PubMed] [Google Scholar]
- Kit S., Kurimura T., Dubbs D. R. Transplantable mouse tumor line induced by injection of SV40-transformed mouse kidney cells. Int J Cancer. 1969 Jul 15;4(4):384–392. doi: 10.1002/ijc.2910040403. [DOI] [PubMed] [Google Scholar]
- Kress M., May E., Cassingena R., May P. Simian virus 40-transformed cells express new species of proteins precipitable by anti-simian virus 40 tumor serum. J Virol. 1979 Aug;31(2):472–483. doi: 10.1128/jvi.31.2.472-483.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai C. J., Nathans D. A map of temperature-sensitive mutants of simian virus 40. Virology. 1975 Jul;66(1):70–81. doi: 10.1016/0042-6822(75)90179-8. [DOI] [PubMed] [Google Scholar]
- Lanford R. E., Butel J. S. Antigenic relationship of SV40 early proteins to purified large T polypeptide. Virology. 1979 Sep;97(2):295–306. doi: 10.1016/0042-6822(79)90341-6. [DOI] [PubMed] [Google Scholar]
- Levitt A., Chen S., Blanck G., George D., Pollack R. E. Two integrated partial repeats of simian virus 40 together code for a super-T antigen. Mol Cell Biol. 1985 Apr;5(4):742–750. doi: 10.1128/mcb.5.4.742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovett M., Clayton C. E., Murphy D., Rigby P. W., Smith A. E., Chaudry F. Structure and synthesis of a simian virus 40 super T-antigen. J Virol. 1982 Dec;44(3):963–973. doi: 10.1128/jvi.44.3.963-973.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May E., Jeltsch J. M., Gannon F. Characterization of a gene encoding a 115 K super T antigen expressed by a SV40-transformed rat cell line. Nucleic Acids Res. 1981 Aug 25;9(16):4111–4128. doi: 10.1093/nar/9.16.4111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May E., Kress M., Daya-Grosjean L., Monier R., May P. Mapping of the viral mRNA encoding a super-T antigen of 115,000 daltons expressed in simian virus 40-transformed rat cell lines. J Virol. 1981 Jan;37(1):24–35. doi: 10.1128/jvi.37.1.24-35.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May E., Lasne C., Prives C., Borde J., May P. Study of the functional activities concomitantly retained by the 115,000 Mr super T antigen, an evolutionary variant of simian virus 40 large T antigen expressed in transformed rat cells. J Virol. 1983 Mar;45(3):901–913. doi: 10.1128/jvi.45.3.901-913.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May P., Resche-Rigon M., Borde J., Breugnot C., May E. Conversion through homologous recombination of the gene encoding Simian virus 40 115,000-molecular-weight super T antigen to a gene encoding a normal-size large T antigen variant. Mol Cell Biol. 1984 Jun;4(6):1141–1151. doi: 10.1128/mcb.4.6.1141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mounts P., Kelly T. J., Jr Rearrangements of host and viral DNA in mouse cells transformed by simian virus 40. J Mol Biol. 1984 Aug 15;177(3):431–460. doi: 10.1016/0022-2836(84)90294-8. [DOI] [PubMed] [Google Scholar]
- Sager R., Anisowicz A., Howell N. Genomic rearrangements in a mouse cell line containing integrated SV40 DNA. Cell. 1981 Jan;23(1):41–50. doi: 10.1016/0092-8674(81)90268-3. [DOI] [PubMed] [Google Scholar]
- Santos M., Butel J. S. Antigenic structure of simian virus 40 large tumor antigen and association with cellular protein p53 on the surfaces of simian virus 40-infected and -transformed cells. J Virol. 1984 Aug;51(2):376–383. doi: 10.1128/jvi.51.2.376-383.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos M., Butel J. S. Association of SV40 large tumor antigen and cellular proteins on the surface of SV40-transformed mouse cells. Virology. 1982 Jul 15;120(1):1–17. doi: 10.1016/0042-6822(82)90002-2. [DOI] [PubMed] [Google Scholar]
- Santos M., Butel J. S. Surface T-antigen expression in simian virus 40-transformed mouse cells: correlation with cell growth rate. Mol Cell Biol. 1985 May;5(5):1051–1057. doi: 10.1128/mcb.5.5.1051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slagle B. L., Lanford R. E., Medina D., Butel J. S. Expression of mammary tumor virus proteins in preneoplastic outgrowth lines and mammary tumors of BALB/cV mice. Cancer Res. 1984 May;44(5):2155–2162. [PubMed] [Google Scholar]
- Smith A. E., Smith R., Paucha E. Characterization of different tumor antigens present in cells transformed by simian virus 40. Cell. 1979 Oct;18(2):335–346. doi: 10.1016/0092-8674(79)90053-9. [DOI] [PubMed] [Google Scholar]
- Soule H. R., Butel J. S. Subcellular Localization of simian virus 40 large tumor antigen. J Virol. 1979 May;30(2):523–532. doi: 10.1128/jvi.30.2.523-532.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soule H. R., Lanford R. E., Butel J. S. Detection of simian virus 40 surface-associated large tumor antigen by enzyme-catalyzed radioiodination. Int J Cancer. 1982 Mar 15;29(3):337–344. doi: 10.1002/ijc.2910290318. [DOI] [PubMed] [Google Scholar]
- Verderame M. F., Kohtz D. S., Pollack R. E. 94,000- and 100,000-molecular-weight simian virus 40 T-antigens are associated with the nuclear matrix in transformed and revertant mouse cells. J Virol. 1983 May;46(2):575–583. doi: 10.1128/jvi.46.2.575-583.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson V. G., Williams R. L. Origin binding by a 100,000-dalton super-T antigen from SVT2 cells. J Virol. 1985 Oct;56(1):102–109. doi: 10.1128/jvi.56.1.102-109.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]