Abstract
Glycine receptors (GlyRs) are recognized as the primary mediators of neuronal inhibition in the spinal cord, brain stem and higher brain regions known to be sensitive to ethanol. Building evidence supports the notion that ethanol acting on GlyRs causes at least a subset of its behavioral effects and may be involved in modulating ethanol intake. For over two decades, GlyRs have been studied at the molecular level as targets for ethanol action. Despite the advances in understanding the effects of ethanol in vivo and in vitro, the precise molecular sites and mechanisms of action for ethanol in ligand-gated ion channels in general, and in GlyRs specifically, are just now starting to become understood. The present review focuses on advances in our knowledge produced by using molecular biology, pressure antagonism, electrophysiology and molecular modeling strategies over the last two decades to probe, identify and model the initial molecular sites and mechanisms of ethanol action in GlyRs. The molecular targets on the GlyR are covered on a global perspective, which includes the intracellular, transmembrane and extracellular domains. The latter has received increasing attention in recent years. Recent molecular models of the sites of ethanol action in GlyRs and their implications to our understanding of possible mechanism of ethanol action and novel targets for drug development in GlyRs are discussed.
Keywords: Glycine, Ethanol, Targets, Electrophysiology, Molecular Models, GLIC
1. INTRODUCTION
A. Background
Alcohol abuse represents a significant problem in our society, affecting about 17.6 million people in the United States alone (Volpicelli, 2001;Grant et al., 2004). The economic costs of alcohol-related disorders in the United States are estimated to exceed 185 billion dollars per year (Grant et al., 2004), compared to the 730 million dollars spent on alcohol-related research worldwide (Rajendram and Preedy, 2005). Consumer expenditure on alcohol in the United States in 2001 was 128.6 billion dollars (Foster et al., 2006). The effects of alcohol abuse and dependence result in significant loss of workplace productivity, increased accidents, increased risk for hypertension, cardiomyopathy, obesity, and liver disease. This in turn leads to over 100,000 deaths and costing nearly 200 billion dollars per year (Grant et al., 2004;Harwood, 2000). Despite the wide consumption and the issues associated with the excessive intake of alcoholic beverages, the mechanisms of ethanol action that cause and modulate its behavioral effects are still poorly understood.
Historically, alcohols and anesthetics were believed to act by perturbing the lipid membrane. Over 100 years ago, H.H. Meyer (Meyer, 1899;Meyer, 1901) and C.E. Overton (Overton, 1901) independently found that the potency of alcohols and general anesthetics was proportional to their partition coefficient between the aqueous phase and the oil phase (water and olive oil, respectively) (Roth, 1979;Heimburg and Jackson, 2007). Structural diversity among anesthetic agents lent support to the concept of a nonspecific, lipid target and mechanism of action for all anesthetics (Trudell et al., 1973b;Trudell, 1977). Meyer refined the Meyer-Overton rule (Meyer, 1937) by proposing that anesthesia occurs when any chemically indifferent substance attains a certain concentration within the lipid bilayer of the cell (Roth, 1979). L.J. Mullins supported the notion that anesthetic potency is related to the concentration in the membrane, but suggested that the volume occupied by the anesthetic is also important for anesthesia (Mullins and Gaffey, 1954;Roth, 1979). He predicted that anesthesia occurs when a critical volume fraction of anesthetic exists within the membrane phase. Mullins suggested that the permeability of ions would be depressed once a critical volume of anesthetic within the membrane was reached, thus resulting in a loss of excitability. Later work extended Mullins’ theory to suggest that adsorption of an anesthetic would expand the membrane, thereby providing a basis for the mechanism of anesthetic action (Lever et al., 1971;Miller et al., 1973;Halsey, 1982).
The critical volume hypothesis states that "anesthesia occurs when the volume of a hydrophobic region is caused to expand beyond a certain critical amount by the adsorption of molecules of an (inert) substance. If the volume of this hydrophobic region can be restored by changes of temperature or pressure, then the anesthesia will be removed” (Mullins and Gaffey, 1954). This notion was supported by studies showing that exposure to high pressures (>100 ATA) could reverse anesthetic effects of ethanol and other general anesthetic agents (Johnson et al., 1942;Johnson and Flagler, 1950;Lever et al., 1971;Miller and Wilson, 1978;Halsey et al., 1978;Halsey and Wardley-Smith, 1975;Trudell et al., 1973a;Chin et al., 1976;Galla and Trudell, 1980), as well as by more recent studies demonstrating a relationship between body temperature and sensitivity to ethanol at behavioral and biochemical levels (Malcolm and Alkana, 1981;Malcolm and Alkana, 1983;Alkana et al., 1985;Alkana et al., 1988;Finn et al., 1990;Finn et al., 1991).
Similarly, the observation that alcohols and anesthetics protected erythrocytes from hemolysis led to the suggestion that this protection was due to an expansion of the erythrocyte membrane (Seeman and Roth, 1972). These authors suggested that the membrane was expanded isotropically in three dimensions; leading to the proposal that the membrane expansion caused by alcohols and anesthetics was ten-fold the actual volume of those molecules (Seeman, 1972). For several years, this proposal presented a conundrum for molecular mechanisms of intoxication. The realization that a membrane does not expand isotropically, but rather the membrane thins when the surface area expands (Trudell, 1977), resolved this controversy. The latter result suggested that the membrane volume in the presence of alcohols or anesthetics would be only slightly greater than the combined molecular volumes of the ligands plus the original membrane. This concept was shown experimentally by measuring how pressure could exclude small spin-labeled molecules (Trudell et al., 1973a). This notion was codified as a “mean excess volume hypothesis” (Ueda and Mashimo, 1982;Mori et al., 1984). The important point of the “mean excess volume” hypothesis is that, in order to have a response reversed by pressure, the sum of the molar volumes of the agent (alcohol or anesthetic) plus the target (membrane or protein) must be less than the total volume of the new mixed system. This “excess volume” would come about by creating disorder in the system; for example, poorer packing of water molecules in protein cavities or creation of small cavities at the protein-lipid interfaces. Then pressure would act to restore the system to a more ordered lower-volume state. However, the properties of bulk phospholipid membranes were only slightly modified at the pressures that reversed anesthesia (approximately 100 ATA) (Mastrangelo et al., 1978;Mastrangelo et al., 1979;Galla and Trudell, 1980;Galla and Trudell, 1981). As a result, many studies investigated the much more sensitive lateral phase separations in membranes (Trudell, 1977;Galla and Trudell, 1980;Galla and Trudell, 1981).
Convincing evidence indicates that alcohols and anesthetics act on proteins (either within or independent of the membrane) and that membrane perturbation alone is not sufficient to cause anesthesia (Hunt, 1985;Deitrich et al., 1989). The effects of alcohols on membrane disorder are generally measurable only at concentrations well above the pharmacological range (Goldstein, 1984). For example, at concentrations associated with anesthesia, there would be only 1 ethanol molecule per approximately 200 lipid molecules. Furthermore, the effects of intoxicating concentrations of alcohols on membrane order can be mimicked by an increase in temperature of just a few tenths of a degree Celsius (Pang et al., 1980;Franks and Lieb, 1982), which clearly does not produce behavioral signs of alcohol intoxication.
In now classic experiments, Franks and Lieb (Franks and Lieb, 1984) found that general anesthetics inhibit the light-emitting firefly luciferase reaction (a lipid-free protein preparation) and suggested that anesthetics “act by competing with endogenous ligands for binding to specific receptors.” Subsequent work found that highly purified optical isomers of the inhalational general anesthetic isoflurane, which are equally effective at disrupting lipid bilayers, exhibited clear stereoselectivity in their effects on particularly sensitive ion channels (Franks and Lieb, 1991;Jones and Harrison, 1993). Moreover, there was a strong correlation between the stereoselective effects of isoflurane on these ion channels and the potency of the anesthetic isomers in vivo (Harris et al., 1992;Lysko et al., 1994).
Collectively, these findings demonstrate that the lipid bilayer cannot account for all of the alcohol and anesthetic effects in the CNS and that more specific sites of action (such as membrane proteins) also play an important role. In spite of the bodies of evidence implicating lipid membranes and proteins, alcohols and anesthetics could have a multitude of targets, including both head and tail groups of lipids, membrane proteins, and/or the annular lipids of proteins (Franks and Lieb, 1987a;Franks and Lieb, 1987b). It is unlikely that any one of these targets can account for all of the alcohol and anesthetic effects in the CNS. More recent research has increased understanding of the interactions at each of these targets.
B. Glycine Receptors—Targets for Ethanol Action
Ligand-gated ion channels (LGICs) have received considerable attention as putative sites of ethanol action that cause its behavioral effects (Deitrich et al., 1989;Harris, 1999;Mihic et al., 1997;Ye et al., 1998;Zhou and Lovinger, 1996;Cardoso et al., 1999;Davies and Alkana, 2001;Molander and Söderpalm, 2005;Molander et al., 2007;Rewal et al., 2009). Research in this area has focused on investigating ethanol’s effects on two large superfamilies of LGICs: 1) The cys-loop superfamily of LGICs (Ortells and Lunt, 1995;Karlin, 2002) whose members include nicotinic acetylcholine (nACh), 5-hydroxytryptamine3 (5HT3), γ-aminobutyric acid type-A (GABAA) or type-C (GABAC), and glycine receptors (GlyRs) (Mihic and Harris, 1996;Zhou and Lovinger, 1996;Grant, 1995;Cardoso et al., 1999;Davies et al., 2002;Davies et al., 2004;Crawford et al., 2007;Perkins et al., 2008) and 2) The glutamate superfamily of LGICs including Nmethyl D-aspartate (NMDA), α -amino-3-hydroxyisoxazolepropionic acid (AMPA) and kainate receptors (Monaghan et al., 1989;Sommer and Seeburg, 1992). These studies found that pharmacologically relevant concentrations of ethanol potentiate (i.e. allosterically increase or positively modulate) the action of GABA and glycine on GABAARs and GlyRs respectively. On the other hand, ethanol generally inhibits (i.e., allosterically decrease; negatively modulate) the actions of glutamate on its receptors (Criswell et al., 1993;Deitrich et al., 1989;Lin et al., 1993;Franks and Lieb, 1994;Weight et al., 1992;Mihic et al., 1997;Ye et al., 1998). Other cys-loop receptors such as GABAC, nACh, and 5-HT3Rs (Mihic and Harris, 1996;Zhou and Lovinger, 1996;Grant, 1995;Cardoso et al., 1999;Davies et al., 2006) as well as the ATP-gated purinergic (P2X) superfamily (Weight et al., 1999;Xiong et al., 1999;Davies et al., 2002;Davies et al., 2005;Asatryan et al., 2008;Popova et al., 2010) have also been shown to be sensitive to the effects of ethanol.
GlyRs are recognized as the primary mediators of neuronal inhibition in the spinal cord. In addition, they mediate inhibition in the brain stem and higher brain regions known to be sensitive to ethanol (Lynch, 2004;Molander and Söderpalm, 2005;Molander et al., 2007). In addition to alcohol, GlyRs are modulated by a variety of other agents including zinc, picrotoxin, strychnine and ivermectin (Bloomenthal et al., 1994;Laube et al., 1995;Shan et al., 2001;Webb and Lynch, 2007;Yang et al., 2007;McCracken et al., 2010). Functional GlyRs consist of five subunits which come together to form a single chloride ion channel. GlyR subunits cloned to date include four a subunits (1 to 4) and one β subunit, which provide for significant diversity (Lynch, 2004). These receptors undergo a developmental switch from α2 to α1β by around postnatal day 20 in rat (Watanabe and Akagi, 1995). Adult GlyRs are found in a 2:3 αβ stoichiometry (Webb and Lynch, 2007) however, most of the information obtained regarding the molecular structure and function of GlyRs, including ethanol studies, have used homomeric α1 GlyRs.
The significance of glycinergic neurons in defined neuronal circuits in the brain has not been sufficiently investigated. This is a matter of relevance with regards to functional interpretation of the roles of GlyRs in vivo because the distribution of GlyR subunits extends to areas in addition to glycinergic axon terminals (Araki et al., 1988;Malosio et al., 1991). To address this paucity of information regarding the precise innervation of GlyRs in neurons, a recent study by Zeilhofer and colleagues (Zeilhofer et al., 2005) generated bacterial artificial chromosome (BAC) transgenic mice which expressed enhanced green fluorescent protein (EGFP). The EGFP in these mice was conditionally activated under the control of the neuronal glycine transporter 2 isoform (GlyT2) gene (Liu et al., 1993), a reliable marker for glycinergic neurons in the CNS (Poyatos et al., 1997;Ponce et al., 1998). Using these transgenic animals, the authors were able to visualize the intensely fluorescent neurons expressing GlyT2-EGFP in great detail. This approach revealed an abundance of neurons exhibiting glycinergic signals (indicated by fluorescent innervation) in the hypothalamus, intralaminar nuclei of the thalamus and basal forebrain. In contrast, there was less abundance of positive fibers in the olfactory bulb, striatum, neocortex, hippocampus and amygdala. The authors reported a lack of GlyT2-EGFP-positive cell bodies in the forebrain. The authors also carried out double immunofluorescence staining between EGFP and GlyT1, the glycine transporter specific for glia, and reported that there was distinct separation between the two markers at almost all levels of the neuraxis (Zeilhofer et al., 2005). Overall, these findings from Zeilhofer and colleagues (Zeilhofer et al., 2005), point out that the distribution of glycinergic axon terminals is less pervasive than glycine receptors. This finding has large implications for glycinergic signaling in the CNS, independent of the traditional, activity-dependent, synaptic release of glycine, especially when considering the actions of modulators such as alcohol and their behavioral effects mediated via GlyRs.
Building evidence consistently supports the notion that ethanol acting on GlyRs causes at least a subset of its behavioral effects and may be involved in modulating ethanol intake. GlyRs have been identified in key alcohol sensitive brain regions and play a critical role in modulating the excitability of neurons associated with sensory information and motor control of respiration (Rajendra et al., 1997;Betz, 1991;Lynch, 2004;Webb and Lynch, 2007). These functions are significantly altered during ethanol intoxication. In addition, glycine and D-serine (a glycine pre-cursor) were shown to enhance ethanol-induced loss of righting reflex in mice (Williams et al., 1995). The effect of glycine on the loss of righting reflex was blocked by strychnine, suggesting that glycine enhances the action of ethanol by acting on strychnine-sensitive GlyRs. This notion is consistent with other work showing that GlyRs mediate part of the immobility produced by anesthetics (Quinlan et al., 2002;Zhang et al., 2001;Zhang et al., 2003). This conclusion is further supported by studies in which transgenic expression of S276Q mutant α1GlyR subunits decreased sensitivity to ethanol-induced loss of righting reflex, motor incoordination (rotarod) and inhibition of strychnine-induced seizures (Findlay et al., 2002). Moreover, recent studies using microdialysis infusions of glycine or strychnine into the nucleus accumbens suggest that GlyRs are important for regulating voluntary ethanol intake and may act as an entrance point into the brain reward system (Molander and Söderpalm, 2005;Molander et al., 2007;Ericson et al., 2009). In addition, studies suggest that Alko Alcohol/Non-Alcohol rats are differently disposed to ethanol consumption due to the arrangement or placement and/or compositions of their GlyRs (Jonsson et al., 2009). This work found a strong positive correlation between α1 subunit expression in the nucleus accumbens and increased ethanol intake Alko Alcohol rats. Along the same lines, recent studies using α1 glycine null mutant mice suggests that the α1 subunit may be involved in ethanol consumption (Blednov and Harris, personal communication).
Electrophysiological studies of GlyRs further support a role for GlyRs in mediating the effects of ethanol on the brain. Behaviorally relevant concentrations of ethanol positively modulate GlyR function measured in synaptoneurosomes of whole-rat brain (Engblom and Åkerman, 1991), embryonic spinal neurons of mouse and chick (Celentano et al., 1988;Aguayo and Pancetti, 1994;Tapia et al., 1998;Ziskind-Conhaim et al., 2003), freshly dissociated rat neurons (Ye et al., 2001a;Ye et al., 2001b;Ye et al., 2002;Tao and Ye, 2002;Jiang and Ye, 2003;McCool et al., 2003) and brain slice preparations (Eggers et al., 2000;Eggers and Berger, 2004;Sebe et al., 2003). In addition, ethanol reliably and robustly potentiates human recombinant α1 and α2 GlyRs measured electrophysiologically (Mascia et al., 1996a;Mascia et al., 1996b;Mihic et al., 1997;Valenzuela et al., 1998;Yamakura and Harris, 2000;Davies et al., 2003;Davies et al., 2004;Crawford et al., 2007;Perkins et al., 2008;Perkins et al., 2009). Recent work by Mihic and colleagues examined the effects of ethanol on outside-out patches pulled from Xenopus laevis oocytes expressing α1 GlyRs (Welsh et al., 2009). The authors reported that ethanol potentiates GlyR function by increasing burst durations and increasing the number of channel openings per burst. They reported minimal effects of alcohol on open and closed dwell times and likelihood. Similarly, the percentage of open time within bursts was not affected. Their kinetic modeling analyses suggest that ethanol increases burst durations by decreasing the rate of glycine unbinding (Welsh et al., 2009).
Therefore, in vitro, pharmacological, molecular, electrophysiological and behavioral evidence indicates that ethanol-induced modulation of strychnine-sensitive GlyRs plays a key role in causing behavioral effects of ethanol and may influence ethanol intake through brain reward pathways.
C. Strategies for Identifying Sites and Mechanisms of Ethanol Action
Despite the advances in understanding the effects of ethanol in vivo and in vitro described above, the precise molecular sites and mechanisms of action for ethanol in ligand-gated ion channels in general, and in GlyRs specifically, are just now starting to become understood.
Part of the difficulty in identifying the sites of ethanol action, and in determining their roles in causing the effects of ethanol, reflects the physical-chemical nature of ethanol’s mechanism of action. This physical-chemical, non-structure based, low affinity mechanism limits the ability to use standard pharmacological approaches to answer these questions. Findings over the last fifteen years suggesting that ethanol acts by “binding” to pockets (Franks and Lieb, 1994;Mihic et al., 1997;Franks and Lieb, 1997;Ye et al., 1998;Wick et al., 1998;Mascia et al., 2000;Crawford et al., 2007) seem to have blurred the mechanistic distinction between intoxicant-anesthetics, such as ethanol, and other psychoactive drugs.
However, the primary determinant of intoxicant-anesthetic potency remains hydrophobicity, not molecular structure. Furthermore, the millimolar ethanol concentrations required for its biological actions are inconsistent with high affinity sites of action and suggest that ethanol acts simultaneously by the same mechanism on different types of initial sites (Deitrich et al., 1989;Davies et al., 2004). Hence, ethanol’s physical-chemical mechanism of action is fundamentally different from the selective, high affinity binding mechanism that is known to initiate the behavioral effects of most psychoactive drugs (Dunn et al., 1999). Therefore, we refer to the sites of ethanol action as “action sites” and “action pockets” to distinguish them from classical binding sites and to incorporate findings suggesting that there likely are multiple ethanol targets within one action pocket.
This atypical mechanism, and resultant lack of high affinity and pharmacological specificity, precludes the classical approach of using ethanol receptor antagonists to identify the sites and mechanisms of ethanol action and to establish cause-effect relationships (Lister and Nutt, 1987;Deitrich et al., 1989;Little, 1991;Davies and Alkana, 2001). Presented next are several strategies which have been used to help fill this void.
(i) Molecular Biology
Techniques involving molecular manipulation, such as mutagenesis and chimeric strategies, have become a classical approach to study the effect of protein structure on function and response to drugs. In the GlyR, this strategy has been used with numerous assays, such as electrophysiology and biochemistry, to investigate the role that particular amino acid residues play in the effects of ethanol. Such molecular manipulations of receptor structure can be used alone or in combination with reagents that covalently bind to cysteine substituted residues and mimic the effect of “binding” small alcohol molecules. This technique is based on the premise that these covalently bound alcohol-like anesthetics [e.g.; propanethiol or propyl methanethiosulfonate (PMTS)] will change the reversible effect of the agent to an irreversible effect only if the actions of the anesthetic result from acting at this site (Mascia et al., 2000;Crawford et al., 2007).
(ii) Pressure Antagonism of Ethanol
A second approach uses increased atmospheric pressure (pressure, hyperbaric exposure), in lieu of a traditional pharmacological antagonist, to help identify the sites and mechanisms of ethanol action. Studies show that exposure to 4–12 times normal atmospheric pressure (ATA) is a direct ethanol antagonist that blocks and reverses a broad spectrum of ethanol’s acute and chronic behavioral effects (Alkana and Malcolm, 1981;Alkana and Malcolm, 1982a;Malcolm and Alkana, 1982;Alkana et al., 1992;Nielsen et al., 1987;Bejanian et al., 1993;Davies et al., 1994;Davies et al., 1999;Davies and Alkana, 2001), as well as its effects at the biochemical (Davies and Alkana, 1998;Davies et al., 1999;Davies and Alkana, 2001) and molecular (Davies et al., 2003;Davies et al., 2004;Perkins et al., 2008) levels.
Pressure at this low level antagonizes ethanol without causing changes in behavior or baseline receptor function that called into question the specificity and utility of the higher pressures (>100 ATA) needed for pressure reversal of anesthesia (Hunter and Bennett, 1974;Halsey et al., 1970;Brauer et al., 1979;Miller, 1977;Wann and MacDonald, 1988). These studies at 12 ATA and lower demonstrate that pressure antagonism meets the key criteria for a direct mechanism (Syapin et al., 1988;Bejanian et al., 1993;Davies et al., 1994;Syapin et al., 1996;Davies et al., 1999;Davies and Alkana, 2001;Malcolm and Alkana, 1982;Alkana and Malcolm, 1982b;Alkana and Malcolm, 1982a;Davies et al., 2003) and selectivity (Alkana et al., 1995;Davies and Alkana, 1998;Davies et al., 1996;Davies and Alkana, 1998;Davies et al., 2001;Davies et al., 1999;Davies et al., 2003), necessary for it to be used in a manner analogous to a traditional pharmacological antagonist to help identify initial molecular sites of ethanol action and to test cause-effect relationships (Davies and Alkana, 2001). From this perspective, the sites of pressure antagonism are the same as the sites of ethanol action. Hence, mutation of a site that alters the sensitivity of a receptor to ethanol should also alter its sensitivity to pressure antagonism, if the site is an initial site of ethanol action. Therefore, knowledge regarding the sites and mechanism(s) of pressure antagonism of ethanol in LGICs can identify new sites and mechanisms of ethanol action, which in turn can serve as new targets for drug development.
(iii) Molecular Modeling
The lack of a crystal structure greatly complicates traditional structure-function and or docking analyses of agonist and ethanol action in the GlyR. As a result, molecular modeling that incorporates evidence generated from the strategies outlined above is used to help define the sites of ethanol action in GlyRs, to suggest structure-function relationships and to identify possible sites of molecular interaction that mediate ethanol actions. Molecular models of GlyR and GABAAR are typically built by combining multiple X-ray structures from unrelated organisms to create a suitable template for homology modeling (Trudell and Bertaccini 2004). These models have been validated by intermediate resolution cryo-electron microscopy structures of torpedo nAChR (PDB ID 2BG9) (Unwin 2005). This approach has already led to important advances.
Developing models that define the physical-chemical properties of the sites and mechanisms by which ethanol acts, that determine the properties that constitute its activation pockets, and that control ethanol sensitivity are key steps for developing agents to block, modulate and/or mimic the actions of ethanol at these sites.
Together, molecular, hyperbaric and modeling strategies provide complementary approaches for identifying, testing and depicting possible sites and mechanism(s) of ethanol action and antagonism, as well as sites that control ethanol sensitivity in a variety of receptors. The present review now focuses on advances in our knowledge produced by using these strategies over the last two decades to probe, identify and model the initial molecular sites and mechanisms of ethanol action in GlyRs.
2. REGION-SPECIFIC TARGETS AND MECHANISMS OF ETHANOL ACTION
The majority of studies that explored the sites and mechanisms of action of ethanol in GlyRs have focused on putative targets in the transmembrane (TM) and extracellular (EC) domains. However, a number of studies suggest that the intracellular domain may also be involved as a target and or mediator of the actions of ethanol in GlyRs. With this in mind, the following review primarily focuses on the TM and EC domains, but also includes a brief presentation regarding recent investigations focusing on the role of the intracellular domain.
A. INTRACELLULAR DOMAIN
Recent work found that multiple signal transduction pathways in the intracellular domain of GlyRs were modulated by pharmacologically relevant concentrations of ethanol (Morrow et al., 2004;Ron and Jurd, 2005). Additional studies suggested discrete molecular determinants of ethanol sensitivity within the large intracellular loop of this receptor. This later work focused on intracellular signaling in the GlyR initiated by PKA, PKC and Gβγ dimmers (Legendre, 2001;Lynch, 2004;Harvey et al., 2004). Further work revealed that α1 GlyRs are modulated by G proteins through Gβγ heterodimers (Yevenes et al., 2006). Interestingly, recent studies have shown that the degree of GlyR-Gβγ functional interaction is critical for ethanol-induced potentiation on the glycine-activated current (Yevenes et al., 2008). Subsequent related work found that ethanol effects on the GlyR could be blocked by a peptide that interferes with Gβγ binding (Guzman et al., 2009). Taken together, this work suggests that intracellular signaling elements, such as G proteins play a role in controlling ethanol sensitivity and possibly initiating ethanol action in GlyRs.
B. TRANSMEMBRANE DOMAIN
Most early studies investigating the initial targets for ethanol action in GlyRs focused on the TM region, which comprises the channel pore. Initial investigations into the molecular targets for ethanol within cys-loop receptors utilized two receptors: α1 GlyRs and ρ1 GABAARs (Mihic et al., 1997). Both receptors form functional homomers and have qualitatively different responses to ethanol (α1 GlyRs are potentiated by ethanol and ρ1 GABAARs are inhibited by ethanol). The authors developed chimeras of these receptors and used these chimeras as a tool to begin to search for potential important regions of the receptor for the action of ethanol. This was accomplished by testing various mutant receptors containing various segments of the protein and determining which portion of the receptor could be spliced into the corresponding location of the other receptor to change the ethanol response. This study identified a 45 amino acid residue region containing TM2 and TM3 that appeared to be both necessary and sufficient for ethanol modulation of receptor function. Sequence alignment comparison of α1 GlyRs and ρ1 GABAARs revealed twelve differences at homologous residues within this 45 amino acid residue region. Replacing the amino acids at these positions in α1 GlyRs with the respective residues of the ρ1 GABAAR identified specific TM2 and TM3 residues (S267 and A288 in the α1 GlyR) that are critical for allosteric modulation of these receptors by alcohols and volatile anesthetics.
Subsequent studies found that systematic replacement of the serine at position 267 with the other nineteen amino acids altered ethanol potentiation of GlyR function (Ye et al., 1998). Moreover, this work revealed an inverse correlation between molecular volume of the amino acid substitution and the effect of ethanol, with a crossover from ethanol potentiation to inhibition beginning at residues equal to or larger in volume than isoleucine. Potentiation by alcohols and anesthetics was also altered by mutations at I229 in TM1, A288 in TM3, and W407 and Y410 in TM4 (Greenblatt and Meng, 1999;Jenkins et al., 2001;Yamakura et al., 1999;Lobo et al., 2004a;Lobo et al., 2006). Together, these results demonstrate that the amino acid residues in TM domains can play a crucial role in determining the functional consequences of allosteric modulation of the GlyR by alcohols and anesthetics.
Further investigations used the n-chain alcohol cutoff to test the possibility that ethanol has a finite site of action in the TM domain. The cutoff refers to the failure to increase potency as a function of increasing the n-chain alcohol length and is thought to occur when the molecular volume of the alcohol exceeds the finite volume of a putative alcohol pocket (Wick et al., 1998;Pringle et al., 1981;Alifimoff et al., 1989). Prior work had established that different ligand-gated ion channels exhibit different cutoff points (the carbon chain length for GlyRs and GABAARs = 10–12; GluRs, AMPARs and KARs = 7; ρ1 GABAARs = 7 and P2XRs = 3) (Dildy-Mayfield et al., 1996;Mascia et al., 1996a;Mihic and Harris, 1996;Li et al., 1994), supporting the existence of alcohol “binding” pockets of variable size on these membrane proteins (Wick et al., 1998). Therefore, to test the hypothesis that the TM domains of α1 GlyRs and ρ1 GABAARs could form an alcohol binding pocket, mutations were performed on TM2 residues and the alcohol cutoff was observed. Substituting larger residues into position 267 of the α1 GlyR significantly decreased the alcohol cutoff, whereas mutation of the homologous position in the ρ1 GABAAR to smaller residues increased the cutoff. These results support the existence of alcohol action/”binding” pockets within each of these receptors and suggest that the amino acid residues present within the TM domains can control the size of the alcoholaction cavity.
Subsequent experiments tested the hypothesis that amino acids S267 in TM2 and A288 in TM3 in the α1 GlyR represent critical action/”binding” sites for alcohols and anesthetics by using cysteine mutagenesis and anesthetic-like thiol or methanethiosulfonate (MTS) reagents (Mascia et al., 2000). The authors suggested that the creation of a disulfide bond following covalent binding at a single site would change the reversible potentiating effect of the agent to an irreversible effect if the actions of the alcohol or anesthetic result from binding at this site. Consistent with their hypothesis, propanethiol or propyl methanethiosulfonate (PMTS) exposure to α1 S267C GlyRs caused the expected change. Furthermore, PMTS binding to position 267 abolished the potentiating effects of enflurane, isoflurane and octanol suggesting competition for a single binding site among PMTS and alcohols and anesthetics. When in the resting state of the receptor, neither propanethiol nor PMTS were able to irreversibly enhance the function of the A288C TM3 mutant or prevent action of isoflurane on these receptors. In addition, ethanol, in the absence of agonist, causes a conformational change in the receptor that can change accessibility of TM residues (Jung et al., 2005;Jung and Harris, 2006). Finally, PMTS occupancy of a single anesthetic binding pocket within the GlyR pentamer is sufficient to enhance receptor function (Roberts et al., 2005). Taken together, these studies support the notion that TM position 267 is capable of initiating the actions of ethanol.
Recent structure-function studies of the GlyR using MTS reagents identified other positions that may be involved in producing the actions of ethanol and showed the orientation of these amino acids and their importance in channel function. These results have been reported in several original articles and a review article (Lobo et al., 2004a;Lobo et al., 2004b;Lobo and Harris, 2005;Lobo et al., 2006;Lobo et al., 2008). Amino acids in TM1 (I229C), TM2 (M263C, S267C, S270C), and TM3 (A288C) were identified as being accessible to PMTS and it was proposed that these residues define a cavity (Lobo et al., 2004a). Furthermore, larger MTS reagents were used to determine if the volume of this cavity is different in the absence (resting state) and presence of glycine, and concluded that channel gating changes the conformation of this region, providing a mechanism by which occupation of this cavity by alcohols might alter channel function (Lobo et al., 2004a). Subsequent work tested a series of cysteine mutants in TM4 and found that W407C, I409C, Y410C, and K411C showed evidence of reaction with propyl MTS, and mutations at these sites eliminated actions of ethanol on the GlyR (Lobo et al., 2006).
These observations raised the critical question of whether these amino acids in TM 1, 2, 3 and 4 all face a common space or cavity. Because there is no detailed (e.g., X-ray diffraction) structural information for the GlyR, cysteines were introduced into two of these positions to determine if they were sufficiently close in space to allow crosslinking. These cross linking studies provided evidence for cross-linking of the TM2,3 regions (S267C-A288C) and the TM1,3 regions (I229C-A288C) (Lobo et al., 2004b;Lobo et al., 2008).
Taken together, these results are consistent with the notion that LGICs are dynamic proteins during alcohol modulation and channel gating. Collectively, the findings support the hypothesis that the TM domains contribute to an alcohol action/”binding” pocket and that specific residues play a role in causing alcohol modulation of GlyR function.
C. EXTRACELLULAR DOMAIN
The extracellular domain of GlyRs and GABAARs is recognized as the site of agonist binding and receptor activation (Kash et al., 2003;Kash et al., 2004b;Chakrapani et al., 2004;Crawford et al., 2008). The EC domain also contains the sites for binding of allosteric modulators like Zn2+ and benzodiazepines (Sigel and Buhr, 1997;Mhatre and Ticku, 1989;Kucken et al., 2000;Wieland et al., 1992;Barberis et al., 2002;Bloomenthal et al., 1994). A recent study suggests that the synergistic interaction between zinc ions and ethanol might involve the extracellular domain (McCracken et al., 2010), as this region is thought to contain the most likely high- and intermediate affinity binding sites for zinc (Bloomenthal et al., 1994;Laube et al., 1995). Binding to the benzodiazepine site in GABAARs is known to cause both positive and negative (inverse) agonist modulation. Recent studies suggest that the benzodiazepine inverse agonist Ro15-4513 antagonizes alcohol by binding to a non-benzodiazepine binding site in the EC domain (Mehta and Ticku, 1989;Wallner et al., 2006). Moreover, hyperbaric studies suggest that pressure antagonizes ethanol-induced chloride uptake by uncoupling a benzodiazepinelike allosteric modulation of the receptor (Davies and Alkana, 2003). Taken together, these findings provide several lines of indirect evidence suggesting that alcohol may have sites of allosteric action in the EC domain of GlyRs and GABAARs.
Recent studies using recombinant expression systems provide more direct evidence that ethanol also acts on the extracellular domain in GlyRs. The initial suggestion by Harris and colleagues came from recombinant studies of GlyRs which demonstrated that α1 GlyRs were more sensitive to ethanol than α2 GlyRs (Mascia et al., 1996b). These studies found that an alanine to serine exchange at position 52 (A52S), located at the beginning of extracellular domain Loop 2 (Brejc et al., 2001) and one of the residues that differs between α1 and α2 GlyRs, decreased the ethanol sensitivity of the α1 GlyR to the point where the ethanol response resembled that of the α2 GlyR. Overall, the findings implicated residues in the extracellular domain, particularly position 52, as possible sites of ethanol action and/or modulation.
Subsequent studies by Davies et al used a novel ethanol antagonist, increased atmospheric pressure, to probe the role of extracellular domain position 52 in ethanol action. This work found that pressure antagonized ethanol in wildtype (WT) α1GlyRS, but did not antagonize ethanol in mutant A52S GlyRs (Davies et al., 2003;Davies et al., 2004). Moreover, they found that pressure did not antagonize ethanol in α2 GlyRs, which have a threonine at the homologous position (T59) to position 52 in α1GlyRs. Interestingly, the converse experiment, substituting alanine for threonine at position 59 in α2 GlyRs, increased ethanol sensitivity and conferred sensitivity to pressure antagonism of ethanol, thereby making α2GlyRs more α1-like (Perkins et al., 2008). This ability of a single point mutation to alter sensitivity to both ethanol and pressure antagonism provided key evidence that extracellular domain position 52 is a site of ethanol action.
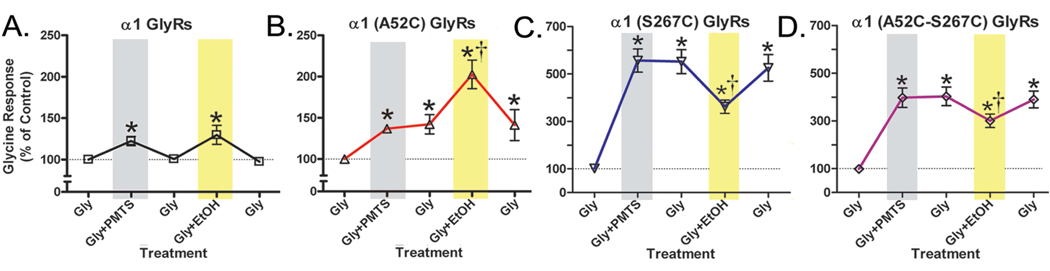
Studies by Crawford et al tested the hypothesis that position 52 is an initial target for ethanol action in α1GlyRs and began to study possible relationships between position 52 in the EC domain and position 267 in the TM domain (Crawford et al., 2007). They accomplished this by investigating the effects of cysteine substitutions at positions 52 and position 267 on the responses of α1GlyRs to n-alcohols and PMTS. Based on the earlier studies in the TM domain described above (Mascia et al., 2000;Borghese et al., 2003), the authors predicted that PMTS would cause irreversible potentiation in A52C GlyRs when bound to the substituted cysteine at the site, if the site is capable of causing alcohol-like receptor modulation. As predicted, PMTS caused irreversible potentiation in A52C, but not WT GlyRs (Fig. 1A). Taken with prior studies at positions 267, these findings support the hypothesis that both positions 52 and 267 are initial sites of ethanol action in α1GlyRs.
Fig. 1. PMTS binding to cysteines substituted at position 52 and/or 267 reveals position-specific negative and positive modulation by ethanol in α1GlyRs.
Mean ± SEM percent control glycine response for (A.) WT and (B. – D.) mutant α1GlyRs (n = 4 – 9). Shaded boxes indicate GlyR responses in the presence of PMTS (grey) or 100 mM ethanol (yellow). (* p < 0.05 vs. control glycine EC10 response, † p < 0.05 versus residual effect of PMTS). Fig. from Crawford et al. 2007 (Crawford et al., 2007).
Further studies used sequential application of PMTS followed by ethanol in cysteine-substituted α1GlyRs to isolate the effects of ethanol on positions 52 and/or 267 (Crawford et al., 2007). Crawford and colleagues reasoned that PMTS would bind to and saturate the cysteine-substituted residues, and would thus prevent further modulation through these bound sites. This is analogous to a pharmacological antagonist binding to and blocking the action of the agonist on the site. The results, summarized in Fig 1, show that ethanol reversibly potentiated (positively modulated) α1GlyRs when position 52 is occupied by PMTS (Fig 1B), that ethanol negatively modulated α1GlyR when position 267 is occupied by PMTS (Fig 1C) and that ethanol produced a small degree of negative modulation when positions 52 and 267 are both occupied by PMTS (Fig 1D). These findings led the authors to conclude that: 1) Positions 52 and 267 are sites of ethanol action that are capable of causing ethanol effects when the other is blocked; 2) Ethanol causes qualitatively different effects depending on its site of action in the GlyR--ethanol acting on position 52 produces negative modulation, whereas ethanol produces positive modulation when it acts on position 267 and 3) Positions 52 and 267 are not the only targets for ethanol in α1GlyRs based on the residual effect of ethanol in the double mutant. Collectively, these findings provided evidence that ethanol’s effect on α1GlyRs represents the summation of its actions on different targets in the EC and TM domains.
Studies from different laboratories come together to provide insight into whether the sites of action for ethanol in the EC and TM domains of α1GlyRs are part of the same alcohol action pocket. As described above, Harris and colleagues found that WT α1GlyRs have a cutoff between decanol and dodecanol and that exposing S267C mutant α1GlyRs, but not WT GlyRs, to PMTS reduced the cutoff to below octanol (Mascia et al., 1996a;Mascia et al., 2000). Subsequent studies by Crawford et al showed that exposing A52C α1GlyRs to PMTS reduced the cutoff to between octanol and decanol (Crawford et al., 2007). Whereas, exposing the double mutant (A52C-S267C) GlyRs to PMTS appeared to reduce the cutoff to below hexanol. Since PMTS changed the cutoff in both A52C and S267C GlyRs, these findings led Crawford et al to conclude that neither position 52 nor 267 belong to a separate pocket that can independently account for the WT cutoff. Therefore, these findings supported the notion that position 52 in Loop 2 of the EC domain and position 267 in the TM domain are part of the same alcohol action pocket (Crawford et al., 2007) and led to a new molecular model of an alcohol pocket spanning from position 52 in the extracellular domain to position 267 in the TM domain (See Fig 3 in the next section on molecular models).
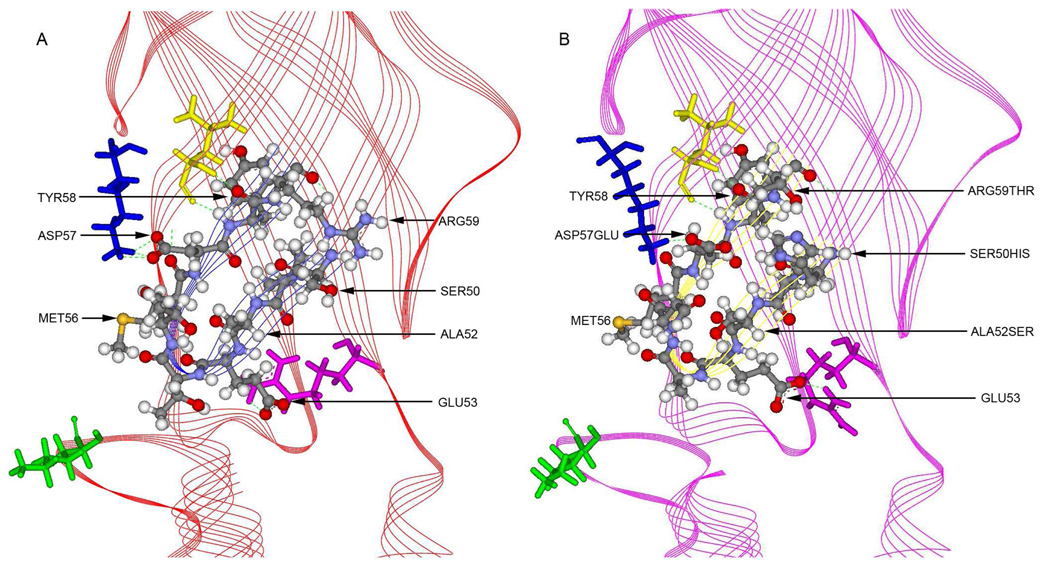
Fig. 3.
(A) Loop 2 in a single GlyR WT subunit. Residues interacting with Loop 2 are rendered as colored stick models: Lys104, blue; Leu136, yellow; Arg218, pink, Lys276, green; (B) Delta Loop 2 in a single GlyR subunit. Residues interacting with Loop 2 are rendered as colored stick models: Lys104, blue; Leu136, yellow; Arg218, pink, Lys276, green (Figure modified from Perkins et al. 2009) (Perkins et al., 2009).
More recent studies investigated the relationships between the physical-chemical properties of the residue at position 52 in α1GlyRs and the impact of these properties on the sensitivity of the receptor to ethanol. This work was based, in part, on the A52S mutant GlyRs studies described above that suggested a role for polarity of the residues at position 52 in α1 GlyRs, and the homologous position 59 in α2 GlyRs, in determining the sensitivity of the respective receptors to ethanol (Mascia et al., 1996b;Davies et al., 2004). To accomplish this, Perkins et al. systematically varied the molecular weight, volume, polarity and hydrophobicity of the residue at position 52 and determined the effect of these changes on ethanol sensitivity (Perkins et al., 2008). They found that increasing the polarity of the residue at position 52 significantly reduced sensitivity to ethanol compared to WT. Correlation analyses indicated that polarity explained approximately 88% of the variability in the sensitivity of α1GlyRs to ethanol, while hydrophobicity accounted for approximately 69% of the variability. This correlation was independent of changes in the glycine EC50. Molecular volume and weight of the residues at position 52 did not appear to affect the sensitivity of α1 GlyRs to ethanol.
The authors pointed out that their findings with polarity at position 52 in the extracellular domain (Perkins et al., 2008) differed from previous studies by others in the TM domain (Ye et al., 1998). The latter showed a role for molecular volume, but not polarity, of the residue at position 267 in controlling ethanol sensitivity in α1GlyRs. Based on these differences, Perkins and colleagues concluded that different physical-chemical properties might influence ethanol action, respectively, in the EC and TM domains.
The findings described above demonstrate that the physical-chemical properties of the residue at position 52 can influence the sensitivity of α1GlyRs to ethanol. Recent studies brought attention to other positions in Loop 2 (Crawford et al., 2008). Cysteine point mutations at positions 50–60 (Loop 2) in α1 GlyRs can significantly alter glycine sensitivity. Subsequent work reported that cysteine substitutions at several positions in Loop 2 changed the receptor’s sensitivity to ethanol (Crawford et al., 2006). Moreover, application of PMTS to these cysteine mutants produced irreversible modulation in some of the mutant receptors (Crawford et al., 2006). These findings suggest that other residues in Loop 2 also play a role in initiating and or modulating the actions of ethanol on α1 GlyRs.
Collectively, the demonstrations that the physical-chemical properties of the amino acid at position 52 alter the sensitivity of α1GlyRs to ethanol and that there are multiple positions in Loop 2 that can also affect ethanol sensitivity suggested that the structure of Loop 2 may play an important role in determining the ethanol sensitivity of α1GlyRs, and other receptors (Perkins et al., 2008). To test this possibility, the authors identified WT receptors in GlyRs and GABAARs that had marked differences in ethanol sensitivity and differed in their Loop 2 structures. Based on prior studies, delta (δ) subunit-containing extrasynaptic GABAARs were chosen as the highly ethanol sensitive representative. This choice was based on prior work, although somewhat controversial (Borghese et al., 2006;Borghese and Harris, 2007) that delta (δ) subunit-containing extrasynaptic GABAARs are sensitive to low ethanol concentrations (3–30 mM) in a variety of preparations (Wallner et al., 2003;Wei et al., 2004;Liang et al., 2006;Hanchar et al., 2005;Hanchar et al., 2006;Wallner et al., 2006;Fleming et al., 2007;Glykys et al., 2007;Mody et al., 2007;Santhakumar et al., 2007). To represent receptors that had less sensitivity to ethanol in recombinant preparations, the authors selected gamma (γ) subunit-containing synaptic GABAARs which are insensitive to low (1–30) ethanol concentrations (White et al., 1990;Weiner et al., 1997) and GlyRs which are also are not sensitive to ethanol concentrations below 10mM (Davies et al., 2003;Mihic et al., 1997;Perkins et al., 2008). A comparison of the Loop 2 sequences of GABAAR δ, GABAAR γ and GlyR α1 subunits revealed differences in the residues at homologous Loop 2 positions in these receptors that could underlie their differences in ethanol sensitivity. Based on this information, the authors predicted that mutating Loop 2 of α1GlyRs to the homologous sequence in Loop 2 in δ GABAARs, would increase the ethanol sensitivity of the mutant α1 GlyRs, if the Loop 2 δ sequence played a key role in determining ethanol sensitivity in these receptors (Perkins et al., 2009).
Perkins and colleagues tested this notion by developing mutant α1GlyRs, in which multiple mutations were performed that replaced the Loop 2 region of the GlyR α1 subunit (positions 50–59) with the homologous residues from the GABAAR δ subunit (positions 43–52) (Perkins et al., 2009). This mutant was designated as α1 δL2GlyR. In addition, mutations were performed that replaced the Loop 2 region of the GlyR α1 subunit (positions 50–59) with the homologous residues from the GABAAR γ subunit (positions 64–73). This mutant was designated as α1 γL2GlyR. As predicted, substituting the δL2 in α1GlyRs reduced the threshold for ethanol response from 30 mM in WT to 1 mM or less in the mutant. In addition, the mutation significantly increased the degree of ethanol potentiation at the low concentrations tested (1–30mM). Substituting the γL2 sequence did not affect the threshold or magnitude of ethanol response in this mutant GlyR. Parallel studies found that substituting Loop 2 of the γ subunit of α1β2γ2 GABAARs with δL2 markedly decreased the ethanol threshold and increased ethanol response of these GABAARs without altering receptor sensitivity to GABA or changing other aspects of receptor function (Perkins et al., 2009).
These findings with δL2 mutations in GlyR and GABAAR support the hypothesis that the structure of Loop 2 can markedly change the threshold and magnitude of the response to ethanol in GlyRs. The results also demonstrate that this phenomenon extends to at least one other cys-loop receptor (GABAAR). This work provided the basis for developing new molecular models of the GlyR threaded on the X-ray structure of the prokaryotic Gloeobacter violaceus pentameric ligand-gated ion channel homologue (GLIC) (Bocquet et al., 2007;Bocquet et al., 2009) presented in the next section of this review (Fig 3).
Collectively, these findings establish that Loop 2 in the EC domain contains multiple sites of action for ethanol and that the effects of ethanol on GlyR function reflect the summation of its actions on sites in the intracellular, EC and TM domains. Moreover, these findings also establish that the structure of Loop 2 in the extracellular domain can have a marked influence on ethanol sensitivity.
3. DISCUSSSION: MOLECULAR MODELS AND MECHANISMS OF ETHANOL ACTION IN GLYCINE RECEPTORS
The lack of a crystal structure precludes traditional structure-function and/or docking analyses of agonist and ethanol action in the GlyR. This disadvantage has forced the use of molecular modeling, which incorporates experimentally obtained data to help define the sites of and possible mechanisms for ethanol action in GlyRs.
Although this review focuses on the molecular targets for ethanol actions in GlyRs and uses results from recent models to explain possible mechanisms, it would not be complete without mention of the lipid component of nerve membranes as a site of action. The influence of the Meyer-Overton correlation of solubility in olive oil with potency of alcohols and anesthetics (Meyer, 1899;Overton, 1901) suggested the possible influence of alcohol on membrane structure. Studies have previously investigated the effect of alcohol (Mastrangelo et al., 1979) and inhalational anesthetics (Trudell et al., 1973b) on membrane fluidity and protein-lipid interactions (Trudell, 1977;Huang et al., 1995). There is clear evidence that membrane properties modulate the function of integral membrane proteins (Baldwin and Hubbell, 1985;Lee, 2009). However, as recently reviewed (Hemmings, Jr. et al., 2005;Lobo and Harris, 2005;Harris et al., 2008), the ability of single site-directed mutations in LGICs to modulate potentiation by alcohol directs our focus to these protein sites.
A. Molecular Models of Ethanol’s Sites of Action
Molecular modeling in GlyRs has come a long way and yet, a cohesive explanation for the mechanisms of ethanol action in GlyRs is far from clear. The new nAChR structure (PDB ID 2BG9) (Unwin, 2005) has been used extensively as a template for molecular modeling of GlyR and GABAAR (Bertaccini et al., 2005;Arias et al., 2006;Lobo et al., 2006;Bertaccini et al., 2007a;Bertaccini et al., 2007b;Crawford et al., 2007;Lobo et al., 2008;Trudell et al., 2008). However, questions remain about the exact position of many amino acid residues in the template as well as the alignment of the template with GlyR and GABAAR (Bertaccini and Trudell, 2002;Ernst et al., 2005). Many of these issues were resolved in a series of modeling and mutagenesis experiments (Jung et al., 2005;Lobo and Harris, 2005;Jung and Harris, 2006;Lobo et al., 2006;Crawford et al., 2007;Lobo et al., 2008;Crawford et al., 2008;Harris et al., 2008;Perkins et al., 2008;Trudell et al., 2008;Perkins et al., 2009).
These initial models showed that the coupling between the extracellular (ligand binding) and TM domains is mediated by a single peptide link plus van der Waals and electrostatic interactions between Loops 2 and 7 from the ligand-binding domain and the TM2-3 loop in the TM domain (Kash et al., 2003;Kash et al., 2004a). Nevertheless, this coupling is sufficiently strong to convey the energy of agonist binding over 45Å to the gate in the ion pore (Corringer et al., 2000;Karlin, 2002;Unwin, 2005;Purohit and Auerbach, 2007). There is reduced electron density at the ligand-binding and transmembrane interface in the cryo-electron micrographs of nAChR (Unwin, 2005). This low packing density is substantiated by experiments because this region is accessible to MTS reagents, including the TM2-TM3 Loop (Lynch et al., 2001;Bera et al., 2002;Goren et al., 2004) and a putative water-filled cavity that includes the extracellular one third of each subunit (Williams and Akabas, 1999;Mascia et al., 2000). Reactivity of site-directed cysteine substitutions with alkyl-MTS reagents in the interfacial region implies that the cysteine thiol groups are ionized and, therefore, that the region is solvated by water (Bera et al., 2002).
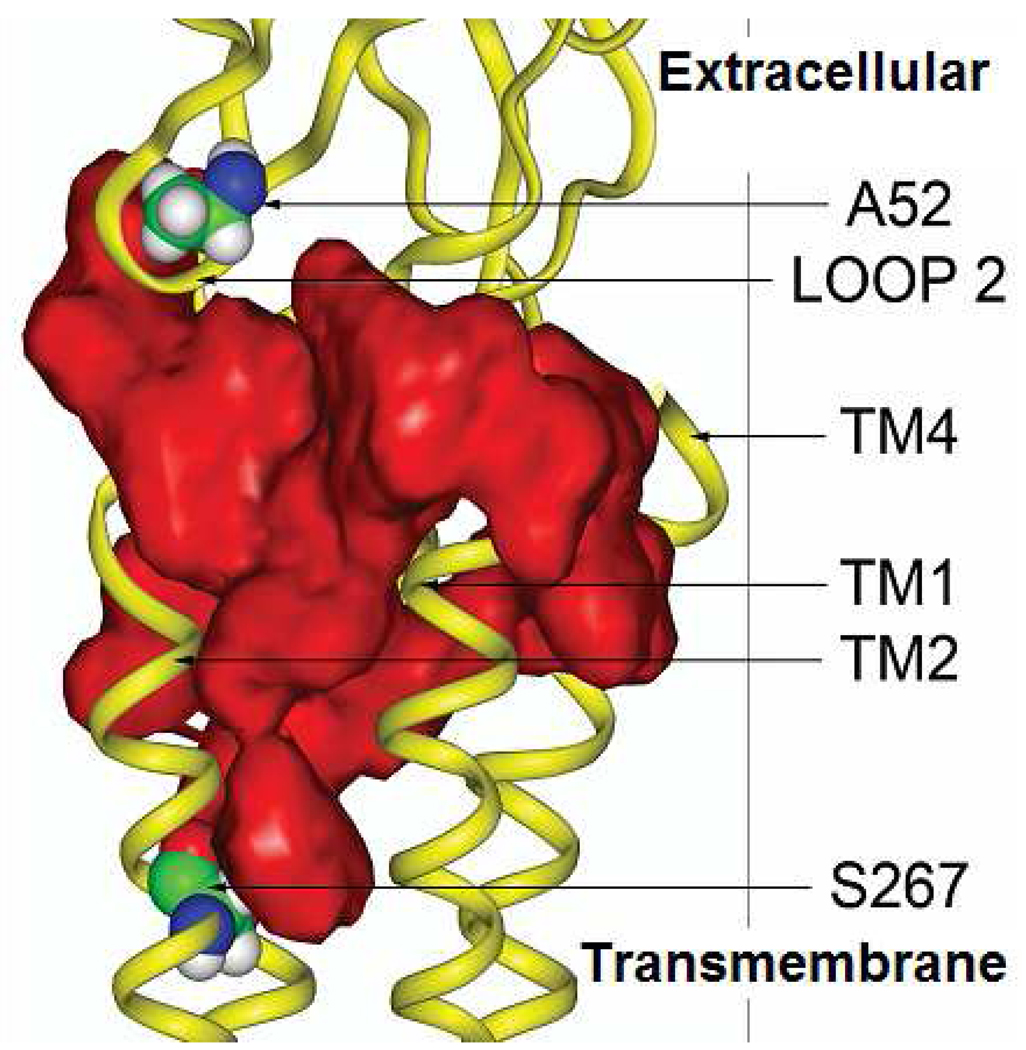
Recent findings in the extracellular and TM domains led to a new molecular model of a potential alcohol action pocket in GlyRs (Crawford et al., 2007) (Fig 2). These findings, described in the prior section, indicate that position 52 in the extracellular domain and position 267 in the TM domain are both capable of initiating the actions of ethanol and that they are both part of the same alcohol action pocket. The model revealed a cavity that extends approximately 28Å from the Cα atoms of A52 to S267 that could function as this alcohol action pocket. The proposed pocket could hold several ethanol molecules and fewer larger alcohols. The estimated distance between position 52 and 267 precludes action by one ethanol molecule at both sites simultaneously, and suggests that at low concentrations, ethanol can enter and move in the pocket without touching one of these action sites. As the concentration of ethanol increases, the probability that ethanol molecule(s) will be acting on one or more of these sites at a given moment increases. The net response to ethanol on receptor function will represent the summation of the actions of ethanol on these targets. This model gives the first depiction of an alcohol action pocket in the EC-TM interface. Further study is necessary to investigate the model, to map the role of other EC domain residues (within and outside of Loop 2) in the actions of ethanol, to discover if other alcohol action pockets exist, and to investigate whether the present findings generalize to other LGICs.
Fig. 2. Zoom view of a molecular model of a α1GlyR subunit with the alcohol pocket highlighted.
The backbone atoms of one α1GlyR subunit are shown as a yellow ribbon. The largest cavity found by the Binding Site Analysis module of Insight 2005L is shown in red. The Cα atoms of A52 and S267 are separated by approximately 28Å. The interconnected sections of the pocket are large enough to accommodate ethanol and allow its passage between these regions. The ability of the A52C-S267C double mutant to gate currents and bind PMTS is consistent with there being enough distance between positions 52 and 267 to prevent spontaneous disulfide linkage between the cysteine substitutions. Figure modified from Crawford et al. 2007 (Crawford et al., 2007).
B. Molecular Mechanisms of Ethanol Action
The physical chemical mechanisms by which the actions of ethanol on its targets are transduced into changes in the receptor’s response are not well understood. Recent advances in molecular models have begun to provide new insights.
Physical evidence to date suggests alcohol binding to be weak and involve rapid on- and off-rates. The high Kd of alcohol (approximately 100 mM) requires that the binding energies to sites of action are small. As a result, it is not expected that the binding event could cause an "induced fit" in a protein site. Rather, it is likely that these molecules bind adventitiously to pre-existing cavities or sites (Cheng et al., 2008;Harris et al., 2008). These small molecules could affect function if there is equilibrium between the conformations of resting channels and those in the open/desensitized state (Schwartz and Hubbell, 2008). Binding of small ligands could change that equilibrium. One could further hypothesize that the ligand binds to most or all conformations of the receptor with varying affinities, but that the shapes or volumes of the binding sites differ between conformations. In that case, a given ligand could fit a site in one conformation better than another and provide more stabilization to that conformation.
This suggestion leads to the possibility that, on a molecular level, a site-directed mutation could switch which conformation is most stabilized by a particular ligand. In fact, there are now several examples in which single mutations change the relative potency of alcohols and inhalational anesthetics (Greenblatt and Meng, 1999;Greenblatt and Meng, 2001;Jenkins et al., 2001;Raines et al., 2001;Scheller and Forman, 2001;Wenningmann et al., 2001;Davies et al., 2004;Woodward et al., 2004;Perkins et al., 2008). Models of alcohol binding sites include internal cavities in proteins (Rashin et al., 1986;Eriksson et al., 1992;Quillin et al., 2000). Adventitious binding of solvents to internal cavities in mutants of T4 lysozyme (Blaber et al., 1995;Morton et al., 1995;Morton and Matthews, 1995) also provides a framework for this model.
The possibility that mutations can alter the sensitivity to ethanol is strongly supported by the recent demonstration by Perkins and colleagues (Perkins et al., 2009) that changes in the structure of Loop 2 can produce 10 fold increases in the sensitivity of GlyR and GABAAR described in the prior section. This finding led to new models of the GlyR that suggest possible physical chemical mechanisms by which ethanol can modulate the receptor’s response to agonist (Fig 3) (Perkins et al., 2009). As described by the authors, these models were developed using the high resolution template provided by the recent publication of an X-ray crystal structure of a prokaryotic ligand-gated ion channel homologue from Gloeobacter violaceus (GLIC, PDB ID 3EAM)(Bocquet et al., 2009;Hilf and Dutzler, 2009). The new GlyR models were built by threading the primary sequence WT and Loop 2 mutant GlyRs onto the GLIC template using the Modeler module of Discovery Studio (DS 2.1; Accelrys, Inc, San Diego, CA) as previously described (Bertaccini et al., 2005;Crawford et al., 2007;Crawford et al., 2008;Perkins et al., 2009).
The authors propose that the resultant models offer a mechanistic explanation for ethanol’s effects on the GlyR based on changes in Loop 2 structure (Perkins et al., 2009). This mechanism is revealed by juxtaposing the models derived from threading the WT versus the δL2 GlyR sequences onto GLIC. The change in conformation as a result of the δL2 substitution in mutant GlyRs alters the manner in which Arg218 (pre-TM1) interacts with Glu53 (Loop 2) with a much different form of salt bridge. The delocalized charge of the three partially positive nitrogen atoms (N-H+ groups) at the guanidinium end of the Arg sidechain allows it to form a salt bridge with the Glu carboxyl group either straight-on (the longest net distance) or at the side of the arginine side chain (shorter net distance and not linear). The result of the δ L2 mutation is to form the more distorted side-on salt bridge in the modeling. This change causes a small distortion of the pre-TM1 segment compared to the WT GlyR. Moreover, the δL2 mutant GlyR has an Asp residue at position 57 in place of the Glu found in WT. As expected, the substitution of Asp57 with Glu results in a shift of the salt bridge with Lys104 to compensate for the increased length of the Glu side chain. The side chain of Lys276 extends out from the TM2-3 linker to make contact with the conserved Glu53 in Loop 2, forming an inter-subunit salt bridge. It is possible that the solvation/desolvation of this salt bridge is important for the structural rearrangements that accompany the gating transition (Honig and Hubbell W.L., 1984). Moreover, the salt bridge between Arg218 and Glu53 has a different conformation in the δL2 mutant GlyR. The altered length of this salt bridge may contribute to the differences in sensitivity to glycine and ethanol of the δ L2 mutant GlyR. The partial negative charges on Glu53, at the tip of the beta turn in Loop 2, are shared between Arg218 and Lys276. The salt bridge described above had been previously modeled within a single subunit (Kash et al., 2003;Kash et al., 2004a;Kash et al., 2004b). In this most recent model of the GlyR by Perkins and coworkers (Perkins et al., 2009), the salt bridge was flexible enough to extend across the inter-subunit space to contact the adjacent subunit.
Perkins and colleagues proposed that these complicated electrostatic and steric interactions might be especially sensitive to, and altered by, the presence of ethanol molecules in the adjacent cavity. These findings exploring the role of Loop 2 and the δ GABA sequence, exemplify how increasing our knowledge regarding the structures that can modulate ethanol sensitivity can increase our understanding of the targets for ethanol and structure-function relationships.
The authors further point out that it is unlikely that these are just static changes. Rather, they would change the ensemble of conformations that may occur during gating, and may be affected by the presence of alcohol molecules, which could alter ethanol sensitivity. If valid, this suggests that these dynamic movements are involved in causing and/or transducing the action of ethanol in Loop 2. Despite the low homology between Loop 2 residues in α1GlyR and δGABAAR, the global structure of the beta turn is conserved in the chimera, illustrating the importance of structural homology across the cys-loop superfamily. This suggests that insights provided by the current model may generalize to GABAARs and other members of the superfamily.
Taken together, the development of models that define the physical-chemical properties of the sites and mechanisms by which ethanol acts, that determine the properties that constitute its activation pockets, and that control ethanol sensitivity are key steps for developing agents to block, modulate and/or mimic the actions of ethanol at these sites.
The present review incorporates evidence regarding the actions of ethanol on GlyRs from a global perspective with its inclusion of findings from the intracellular, transmembrane and extracellular regions. Overall, the material reviewed summarizes advances that have increased our knowledge regarding the structures that initiate, transduce, modulate and antagonize the actions of ethanol on GlyRs and, likely, other LGICs. These findings have changed the thinking and models about the sites and mechanisms of ethanol action and contribute to the long-term goal of the field, which is to identify novel targets for medications development to prevent and treat alcohol abuse and dependence.
Acknowledgments
We would like to thank Dr. Luis Aguayo and Dr. Adron Harris for their input. This work was supported in part by research grants NIH/NIAAA F31 AA017569 (D.I.P), AA03972 (R.L.A.), AA013890 and AA013922 (D.L.D.), and AA013378 (J.R.T.), and the USC School of Pharmacy. This work was conducted as partial fulfillment of the requirements for the PhD degree in Molecular Pharmacology & Toxicology, University of Southern California (D.I.P.).
ABBREVIATIONS
- 5HT3R
5-hydroxytryptamine3 receptor
- (ATA)
atmospheric pressure
- (EGFP)
enhanced green fluorescent protein
- (GABAAR)
Gamma-aminobutyric acid type-A receptor
- (GLIC)
Gloeobacter violaceus pentameric ligand-gated ion channel homologue
- (GlyR)
glycine receptor
- (GlyT2)
glycine transporter 2
- (LGIC)
ligand-gated ion channel
- (MTS)
methanethiosulfonate
- (nAChR)
nicotinic acetylcholine receptor
- (PMTS)
propyl methanethiosulfonate
- (TM)
transmembrane
- (WT)
wild type
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Reference List
- Aguayo LG, Pancetti FC. Ethanol Modulation of the γ-Aminobutyric AcidA and Glycine-Activated Cl− Current in Cultured Mouse Neurons. J Pharmacol Exp Ther. 1994;270:61–69. [PubMed] [Google Scholar]
- Alifimoff JK, Firestone LL, Miller KW. Anaesthetic Potencies of Primary Alkanols: Implications for the Molecular Dimensions of the Anaesthetic Site. Br J Pharmacol. 1989;96:9–16. doi: 10.1111/j.1476-5381.1989.tb11777.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alkana RL, Boone DC, Finn DA. Temperature Dependence of Ethanol Depression: Linear Models in Male and Female Mice. Pharmacol Biochem Behav. 1985;23:309–316. doi: 10.1016/0091-3057(85)90575-1. [DOI] [PubMed] [Google Scholar]
- Alkana RL, Davies DL, Morland J, Parker ES, Bejanian M. Low Level Hyperbaric Exposure Antagonizes Locomotor Effects of Ethanol and N-Propanol but Not Morphine in C57BL Mice. Alcohol Clin Exp Res. 1995;19:693–700. doi: 10.1111/j.1530-0277.1995.tb01569.x. [DOI] [PubMed] [Google Scholar]
- Alkana RL, Finn DA, Bejanian M, Crabbe JC. Genetically Determined Differences in Ethanol Sensitivity Influenced by Body Temperature During Intoxication. Life Sci. 1988;43:1973–1982. doi: 10.1016/0024-3205(88)90570-x. [DOI] [PubMed] [Google Scholar]
- Alkana RL, Finn DA, Jones BL, Kobayashi LS, Babbini M, Bejanian M, Syapin PJ. Genetically Determined Differences in the Antagonistic Effect of Pressure on Ethanol-Induced Loss of Righting Reflex in Mice. Alcohol Clin Exp Res. 1992;16:17–22. doi: 10.1111/j.1530-0277.1992.tb00629.x. [DOI] [PubMed] [Google Scholar]
- Alkana RL, Malcolm RD. Low-Level Hyperbaric Ethanol Antagonism in Mice. Dose and Pressure Response. Pharmacology. 1981;22:199–208. doi: 10.1159/000137491. [DOI] [PubMed] [Google Scholar]
- Alkana RL, Malcolm RD. Hyperbaric Ethanol Antagonism in Mice: Studies on Oxygen, Nitrogen, Strain and Sex. Psychopharmacology. 1982a;77:11–16. doi: 10.1007/BF00436093. [DOI] [PubMed] [Google Scholar]
- Alkana RL, Malcolm RD. Hyperbaric Ethanol Antagonism in Mice: Time Course. Subst Alcohol Actions Misuse. 1982b;3:41–46. [PubMed] [Google Scholar]
- Araki T, Yamano M, Murakami T, Wanaka A, Betz H, Tohyama M. Localization of Glycine Receptors in the Rat Central Nervous System: an Immunocytochemical Analysis Using Monoclonal Antibody. Neuroscience. 1988;25:613–624. doi: 10.1016/0306-4522(88)90263-1. [DOI] [PubMed] [Google Scholar]
- Arias HR, Bhumireddy P, Spitzmaul G, Trudell JR, Bouzat C. Molecular Mechanisms and Binding Site Location for the Noncompetitive Antagonist Crystal Violet on Nicotinic Acetylcholine Receptors. Biochemistry. 2006;45:2014–2026. doi: 10.1021/bi051752e. [DOI] [PubMed] [Google Scholar]
- Asatryan L, Popova M, Woodward JJ, King BF, Alkana RL, Davies DL. Roles of Ectodomain and Transmembrane Regions in Ethanol and Agonist Action in Purinergic P2X2 and P2X3 Receptors. Neuropharmacology. 2008;55:835–843. doi: 10.1016/j.neuropharm.2008.06.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baldwin PA, Hubbell WL. Effects of Lipid Environment on the Light-Induced Conformational Changes of Rhodopsin. 1. Absence of Metarhodopsin II Production in Dimyristoylphosphatidylcholine Recombinant Membranes. Biochemistry. 1985;24:2624–2632. doi: 10.1021/bi00332a006. [DOI] [PubMed] [Google Scholar]
- Barberis A, Petrini EM, Cherubini E, Mozrzymas JW. Allosteric Interaction of Zinc With Recombinant α1β2γ2 and α1β2 GABAA Receptors. Neuropharmacology. 2002;43:607–618. doi: 10.1016/s0028-3908(02)00109-0. [DOI] [PubMed] [Google Scholar]
- Bejanian M, Jones BL, Alkana RL. Low-Level Hyperbaric Antagonism of Ethanol-Induced Locomotor Depression in C57BL/6J Mice: Dose Response. Alcohol Clin Exp Res. 1993;17:935–939. doi: 10.1111/j.1530-0277.1993.tb05644.x. [DOI] [PubMed] [Google Scholar]
- Bera AK, Chatav M, Akabas MH. GABAA Receptor M2-M3 Loop Secondary Structure and Changes in Accessibility During Channel Gating. J Biol Chem. 2002;277:43002–43010. doi: 10.1074/jbc.M206321200. [DOI] [PubMed] [Google Scholar]
- Bertaccini E, Trudell JR. Predicting the Transmembrane Secondary Structure of Ligand-Gated Ion Channels. Protein Eng. 2002;15:443–453. doi: 10.1093/protein/15.6.443. [DOI] [PubMed] [Google Scholar]
- Bertaccini EJ, Shapiro J, Brutlag DL, Trudell JR. Homology Modeling of a Human Glycine Alpha 1 Receptor Reveals a Plausible Anesthetic Binding Site. J Chem Inf Model. 2005;45:128–135. doi: 10.1021/ci0497399. [DOI] [PubMed] [Google Scholar]
- Bertaccini EJ, Trudell JR, Franks NP. The Common Chemical Motifs Within Anesthetic Binding Sites. Anesth Analg. 2007a;104:318–324. doi: 10.1213/01.ane.0000253029.67331.8d. [DOI] [PubMed] [Google Scholar]
- Bertaccini EJ, Trudell JR, Lindahl E. Normal-Mode Analysis of the Glycine Alpha1 Receptor by Three Separate Methods. J Chem Inf Model. 2007b;47:1572–1579. doi: 10.1021/ci600566j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Betz H. Glycine Receptors: Heterogeneous and Widespread in the Mammalian Brain. TINS. 1991;14:458–461. doi: 10.1016/0166-2236(91)90045-v. [DOI] [PubMed] [Google Scholar]
- Blaber M, Baase WA, Glassner N, Matthews BW. Alanine Scanning Mutagenesis of the Alpha-Helix 115–123 of Phage T4 Lysozyme: Effects on Structure, Stability and the Binding of Solvent. J Mol Biol. 1995;246:317–330. doi: 10.1006/jmbi.1994.0087. [DOI] [PubMed] [Google Scholar]
- Bloomenthal AB, Goldwater E, Pritchett DB, Harrison NL. Biphasic Modulation of the Strychnine-Sensitive Glycine Receptor by Zn2+ Mol Pharmacol. 1994;46:1156–1159. [PubMed] [Google Scholar]
- Bocquet N, Nury HBM, Le Poupon C, Changeux J, Delarue M, Corringer P. X-Ray Structure of a Pentameric Ligand-Gated Ion Channel in an Apparently Open Conformation. Nature. 2009;457:111–114. doi: 10.1038/nature07462. [DOI] [PubMed] [Google Scholar]
- Bocquet N, Prado de Carvalho L, Cartaud J, Neyton J, Le Poupon C, Taly A, Grutter T, Changeux J-P, Corringer P-J. A Prokaryotic Proton-Gated Ion Channel From the Nicotinic Acetylcholine Receptor Family. Nature. 2007;445:116–119. doi: 10.1038/nature05371. [DOI] [PubMed] [Google Scholar]
- Borghese CM, Harris RA. Studies of Ethanol Actions on Recombinant DContaining Gamma-Aminobutyric Acid Type A Receptors Yield Contradictory Results. Alcohol. 2007;41:155–162. doi: 10.1016/j.alcohol.2007.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghese CM, Henderson LA, Bleck V, Trudell JR, Harris RA. Sites of Excitatory and Inhibitory Actions of Alcohols on Neuronal α2β4 Nicotinic Acetylcholine Receptors. J Pharmacol Exp Ther. 2003;307:42–52. doi: 10.1124/jpet.102.053710. [DOI] [PubMed] [Google Scholar]
- Borghese CM, Storustovu S, Ebert B, Herd MB, Belelli D, Lambert JJ, Marshall G, Wafford KA, Harris RA. The δ Subunit of γ -Aminobutyric Acid Type A Receptors Does Not Confer Sensitivity to Low Concentrations of Ethanol. J Pharmacol Exp Ther. 2006;316:1360–1368. doi: 10.1124/jpet.105.092452. [DOI] [PubMed] [Google Scholar]
- Brauer RW, Mansfield WM, Jr, Beaver RW, Gillen HW. Stages in Development of High-Pressure Neurological Syndrome in the Mouse. J Appl Physiol. 1979;46:756–764. doi: 10.1152/jappl.1979.46.4.756. [DOI] [PubMed] [Google Scholar]
- Brejc K, van Dijk WJ, Klaassen RV, Schuurmans M, van der Oost J, Smit AB, Sixma TK. Crystal Structure of an ACh-Binding Protein Reveals the Ligand-Binding Domain of Nicotinic Receptors. Nature. 2001;411:269–276. doi: 10.1038/35077011. [DOI] [PubMed] [Google Scholar]
- Cardoso RA, Brozowski SJ, Chavez-Noriega LE, Harpold M, Valenzuela CF, Harris RA. Effects of Ethanol on Recombinant Human Neuronal Nicotinic Acetylcholine Receptors Expressed in Xenopus Oocytes. J Pharmacol Exp Ther. 1999;289:774–780. [PubMed] [Google Scholar]
- Celentano JJ, Gibbs TT, Farb DH. Ethanol Potentiates GABA- and Glycine-Induced Chloride Currents in Chick Spinal Cord Neurons. Brain Res. 1988;455:377–380. doi: 10.1016/0006-8993(88)90098-4. [DOI] [PubMed] [Google Scholar]
- Chakrapani S, Bailey TD, Auerbach A. Gating Dynamics of the Acetylcholine Receptor Extracellular Domain. J Gen Physiol. 2004;123:341–356. doi: 10.1085/jgp.200309004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng MH, Coalson RD, Cascio M. Molecular Dynamics Simulations of Ethanol Binding to the Transmembrane Domain of the Glycine Receptor: Implications for the Channel Potentiation Mechanism. Proteins. 2008;71:972–981. doi: 10.1002/prot.21784. [DOI] [PubMed] [Google Scholar]
- Chin JH, Trudell JR, Cohen EN. The Compression-Ordering and Solubility-Disordering Effects of High Pressure Gases on Phospholipid Bilayers. Life Sci. 1976;18:489–498. doi: 10.1016/0024-3205(76)90326-x. [DOI] [PubMed] [Google Scholar]
- Corringer PJ, Le Novere N, Changeux JP. Nicotinic Receptors at the Amino Acid Level. Annu Rev Pharmacol Toxicol. 2000;40:431–458. doi: 10.1146/annurev.pharmtox.40.1.431. [DOI] [PubMed] [Google Scholar]
- Crawford DK, Shen Y, Trudell JR, Bertaccini EJ, Davies DL, Alkana RL. 2006 Abstract Viewer/Itinerary Planner. Washington, DC: Society for Neuroscience; 2006. Cysteine point mutations at positions 50–60 (loop 2) in alpha 1 glycine receptors can significantly alter glycine and ethanol sensitivity. 2006. [Google Scholar]
- Crawford DK, Trudell JR, Bertaccini E, Li KX, Davies DL, Alkana RL. Evidence That Ethanol Acts on a Target in Loop 2 of the Extracellular Domain of Alpha1 Glycine Receptors. J Neurochem. 2007;102:2097–2109. doi: 10.1111/j.1471-4159.2007.04680.x. [DOI] [PubMed] [Google Scholar]
- Crawford DK, Perkins DI, Trudell JR, Bertaccini EJ, Davies DL, Alkana RL. Roles for Loop 2 Residues of {Alpha}1 Glycine Receptors in Agonist Activation. J Biol Chem. 2008;283:27698–27706. doi: 10.1074/jbc.M802384200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Criswell HE, Simson PE, Duncan GE, McCown TJ, Herbert JS, Morrow AL, Breese GR. Molecular Basis for Regionally Specific Action of Ethanol on γ-Aminobutyric AcidA Receptors: Generalization to Other Ligand-Gated Ion Channels. J Pharmacol Exp Ther. 1993;267(1):522–537. [PubMed] [Google Scholar]
- Davies DL, Alkana RL. Direct Antagonism of Ethanol's Effects on GABAA Receptors by Increased Atmospheric Pressure. Alcohol Clin Exp Res. 1998;22:1689–1697. [PubMed] [Google Scholar]
- Davies DL, Alkana RL. Direct Evidence for a Cause Effect Link Between Ethanol Potentiation of GABAA Receptor Function and Intoxication From Hyperbaric Studies in C57, LS and SS Mice. Alcohol Clin Exp Res. 2001;25:1098–1106. [PubMed] [Google Scholar]
- Davies DL, Alkana RL. Benzodiazepine Agonist and Inverse Agonist Coupling in GABAA Receptors Antagonized by Increased Atmospheric Pressure. Eur J Pharmacol. 2003;469:37–45. doi: 10.1016/s0014-2999(03)01733-3. [DOI] [PubMed] [Google Scholar]
- Davies DL, Asatryan L, Kuo SF, Woodward JJ, King BF, Alkana RL, Xiao C, Ye JH, Sun H, Zhang L, Hu X-Q, Hayrapetyan V, Lovinger DM, Machu TK. Effects of Ethanol on Adenosine 5'-Triphosphate-Gated Purinergic and 5-Hydroxytryptamine3 Receptors. Alcohol Clin Exp Res. 2006;30:1–9. doi: 10.1111/j.1530-0277.2006.00023.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies DL, Bejanian M, Parker ES, Morland J, Bolger MB, Brinton RD, Alkana RL. Low-Level Hyperbaric Antagonism of Diazepam's Locomotor Depressant and Anticonvulsant Properties in Mice. J Pharmacol Exp Ther. 1996;276:667–675. [PubMed] [Google Scholar]
- Davies DL, Bolger MB, Brinton RD, Finn DA, Alkana RL. In Vivo and in Vitro Hyperbaric Studies in Mice Suggest Novel Sites of Action for Ethanol. Psychopharmacology. 1999;141:339–350. doi: 10.1007/s002130050843. [DOI] [PubMed] [Google Scholar]
- Davies DL, Crawford DK, Trudell JR, Mihic SJ, Alkana RL. Multiple Sites of Ethanol Action in α1 and α2 Glycine Receptors Suggested by Sensitivity to Pressure Antagonism. J Neurochem. 2004;89:1175–1185. doi: 10.1111/j.1471-4159.2004.02390.x. [DOI] [PubMed] [Google Scholar]
- Davies DL, Kochegarov AA, Kuo ST, Kulkarni AA, Woodward JJ, King BF, Alkana RL. Ethanol Differentially Affects ATP-Gated P2X(3) and P2X(4) Receptor Subtypes Expressed in Xenopus Oocytes. Neuropharmacology. 2005;49:243–253. doi: 10.1016/j.neuropharm.2005.03.015. [DOI] [PubMed] [Google Scholar]
- Davies DL, Machu TK, Guo Y, Alkana RL. Ethanol Sensitivity in ATP-Gated P2X Receptors Is Subunit Dependent. Alcohol Clin Exp Res. 2002;26:773–778. [PubMed] [Google Scholar]
- Davies DL, McCauley LD, Bolger MB, Alkana RL. Pressure Sensitive and Insensitive Coupling in γ-Aminobutyric AcidA Receptors. Psychopharmacology. 2001;157:401–410. doi: 10.1007/s002130100825. [DOI] [PubMed] [Google Scholar]
- Davies DL, Morland J, Jones BL, Alkana RL. Low-Level Hyperbaric Antagonism of Ethanol's Anticonvulsant Property in C57BL/6J Mice. Alcohol Clin Exp Res. 1994;18:1190–1195. doi: 10.1111/j.1530-0277.1994.tb00103.x. [DOI] [PubMed] [Google Scholar]
- Davies DL, Trudell JR, Mihic SJ, Crawford DK, Alkana RL. Ethanol Potentiation of Glycine Receptors Expressed in Xenopus Oocytes Antagonized by Increased Atmospheric Pressure. Alcohol Clin Exp Res. 2003;27:1–13. doi: 10.1097/01.ALC.0000065722.31109.A1. [DOI] [PubMed] [Google Scholar]
- Deitrich R, Dunwiddie T, Harris RA, Erwin VG. Mechanism of Action of Ethanol: Initial Central Nervous System Actions. Pharmacol Rev. 1989;41:489–537. [PubMed] [Google Scholar]
- Dildy-Mayfield JE, Mihic SJ, Liu Y, Deitrich RA, Harris RA. Actions of Long Chain Alcohols on GABAA and Glutamate Receptors: Relation to in Vivo Effects. Br J Pharmacol. 1996;118:378–384. doi: 10.1111/j.1476-5381.1996.tb15413.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunn SMJ, Davies M, Muntoni AL, Lambert JJ. Mutagenesis of the Rat α1 Subunit of the γ-Aminobutyric AcidA Receptor Reveals the Importance of Residue 101 in Determining the Allosteric Effects of Benzodiazepine Site Ligands. Mol Pharmacol. 1999;56:768–774. [PubMed] [Google Scholar]
- Eggers ED, Berger AJ. Mechanisms for the Modulation of Native Glycine Receptor Channels by Ethanol. J Neurophysiol. 2004;91:2685–2695. doi: 10.1152/jn.00907.2003. [DOI] [PubMed] [Google Scholar]
- Eggers ED, O'Brien JA, Berger AJ. Developmental Changes in the Modulation of Synaptic Glycine Receptors by Ethanol. J Neurophysiol. 2000;84:2409–2416. doi: 10.1152/jn.2000.84.5.2409. [DOI] [PubMed] [Google Scholar]
- Engblom AC, Åkerman KE. Effect of Ethanol on γ-Aminobutyric Acid and Glycine Receptor-Coupled Cl− Fluxes in Rat Brain Synaptoneurosomes. J Neurochem. 1991;57:384–390. doi: 10.1111/j.1471-4159.1991.tb03764.x. [DOI] [PubMed] [Google Scholar]
- Ericson M, Clarke RB, Chau P, Adermark L, Soderpalm B. Beta-Alanine Elevates Dopamine Levels in the Rat Nucleus Accumbens: Antagonism by Strychnine. Amino Acids. 2009;38(4):1051–1055. doi: 10.1007/s00726-009-0313-0. [DOI] [PubMed] [Google Scholar]
- Eriksson AE, Baase WA, Wozniak JA, Matthews BW. A Cavity-Containing Mutant of T4 Lysozyme Is Stabilized by Buried Benzene. Nature. 1992;355:371–373. doi: 10.1038/355371a0. [DOI] [PubMed] [Google Scholar]
- Ernst M, Bruckner S, Boresch S, Sieghart W. Comparative Models of GABAA Receptor Extracellular and Transmembrane Domains: Important Insights in Pharmacology and Function. Mol Pharmacol. 2005;68:1291–1300. doi: 10.1124/mol.105.015982. [DOI] [PubMed] [Google Scholar]
- Findlay GS, Wick MJ, Mascia MP, Wallace D, Miller GW, Harris RA, Blednov YA. Transgenic Expression of a Mutant Glycine Receptor Decreases Alcohol Sensitivity of Mice. J Pharmacol Exp Ther. 2002;300:526–534. doi: 10.1124/jpet.300.2.526. [DOI] [PubMed] [Google Scholar]
- Finn DA, Bejanian M, Jones BL, McGivern RF, Syapin PJ, Crabbe JC, Alkana RL. Body Temperature Differentially Affects Ethanol Sensitivity in Both Inbred Strains and Selected Lines of Mice. J Pharmacol Exp Ther. 1990;253:1229–1235. [PubMed] [Google Scholar]
- Finn DA, Gee KW, Syapin PJ, Bejanian M, Alkana RL. Effects of temperature and ethanol on GABA-stimulated chloride uptake in LS and C57 mice. Soc.Neurosci.Abstr. 1991;17:1425. [Google Scholar]
- Fleming R, Wilson W, Swartzwelder H. Magnitude and Ethanol Sensitivity of Tonic GABAA Receptor-Mediated Inhibition in Dentate Gyrus Changes From Adolescence to Adulthood. J Neurophysiol. 2007;97:3806–3811. doi: 10.1152/jn.00101.2007. [DOI] [PubMed] [Google Scholar]
- Foster S, Vaughan R, Foster W, Califano J., Jr Estimate of the Commercial Value of Underage Drinking and Adult Abusive and Dependent Drinking to the Alcohol Industry. Arch Pediatr Adolesc Med. 2006;160:473–478. doi: 10.1001/archpedi.160.5.473. [DOI] [PubMed] [Google Scholar]
- Franks NP, Lieb WR. Molecular Mechanisms of General Anaesthesia. Nature. 1982;300:487–493. doi: 10.1038/300487a0. [DOI] [PubMed] [Google Scholar]
- Franks NP, Lieb WR. Do General Anaesthetics Act by Competitive Binding to Specific Receptors? Nature. 1984;310:599–601. doi: 10.1038/310599a0. [DOI] [PubMed] [Google Scholar]
- Franks NP, Lieb WR. Are the Biological Effects of Ethanol Due to Primary Interactions With Lipids or With Proteins? Alcohol Alcohol Suppl. 1987a;1:139–145. [PubMed] [Google Scholar]
- Franks NP, Lieb WR. Neuron Membranes: Anaesthetics on the Mind [News] Nature. 1987b;328:113–114. doi: 10.1038/328113a0. [DOI] [PubMed] [Google Scholar]
- Franks NP, Lieb WR. Selective Effects of Volatile General Anesthetics on Identified Neurons. Ann N Y Acad Sci. 1991;625:54–70. doi: 10.1111/j.1749-6632.1991.tb33829.x. [DOI] [PubMed] [Google Scholar]
- Franks NP, Lieb WR. Molecular and Cellular Mechanisms of General Anaesthesia. Nature. 1994;367:607–614. doi: 10.1038/367607a0. [DOI] [PubMed] [Google Scholar]
- Franks NP, Lieb WR. Inhibitory Synapses: Anaesthetics Set Their Sites on Ion Channels. Nature. 1997;389:334–335. doi: 10.1038/38614. [DOI] [PubMed] [Google Scholar]
- Galla H-J, Trudell JR. Asymmetric Antagonistic Effects of an Inhalation Anesthetic and High Pressure on the Phase Transition Temperature of Dipalmitoyl Phosphatidic Acid Bilayers. Biochim Biophys Acta. 1980;599:336–340. doi: 10.1016/0005-2736(80)90080-2. [DOI] [PubMed] [Google Scholar]
- Galla HJ, Trudell JR. Perturbations of Peptide-Induced Lateral Phase Separations in Phosphatidic Acid Bilayers by the Inhalation Anesthetic Methoxyflurane. Mol Pharmacol. 1981;19:432–437. [PubMed] [Google Scholar]
- Glykys J, Peng Z, Chandra D, Homanics GE, Houser CR, Mody I. A New Naturally Occurring GABA(A) Receptor Subunit Partnership With High Sensitivity to Ethanol. Nat Neurosci. 2007;10:40–48. doi: 10.1038/nn1813. [DOI] [PubMed] [Google Scholar]
- Goldstein DB. The Effects of Drugs on Membrane Fluidity. Ann Rev Pharmacol Toxicol. 1984;24:43–64. doi: 10.1146/annurev.pa.24.040184.000355. [DOI] [PubMed] [Google Scholar]
- Goren EN, Reeves DC, Akabas MH. Loose Protein Packing Around the Extracellular Half of the GABA(A) Receptor Beta1 Subunit M2 Channel-Lining Segment. J Biol Chem. 2004;279:11198–11205. doi: 10.1074/jbc.M314050200. [DOI] [PubMed] [Google Scholar]
- Grant BF, Dawson DA, Stinson FS, Chou P, Dufour MC, Pickering RP. The 12-Month Prevalence and Trends in DSM-IV Alcohol Abuse and Dependence: United States, 1991–1992 and 2001–2002. Drug Alcohol Depend. 2004;74:223–234. doi: 10.1016/j.drugalcdep.2004.02.004. [DOI] [PubMed] [Google Scholar]
- Grant KA. The Role of 5-HT3 Receptors in Drug Dependence. Drug and Alcohol Dependence. 1995;38:155–171. doi: 10.1016/0376-8716(95)01120-n. [DOI] [PubMed] [Google Scholar]
- Greenblatt EP, Meng X. A critical amino acid for halothane action. Anesthesiology. 1999;91:A807. [Google Scholar]
- Greenblatt EP, Meng X. Divergence of Volatile Anesthetic Effects in Inhibitory Neurotransmitter Receptors. Anesthesiology. 2001;94:1026–1033. doi: 10.1097/00000542-200106000-00017. [DOI] [PubMed] [Google Scholar]
- Guzman L, Moraga-Cid G, Avila A, Figueroa M, Yevenes G, Fuentealba J, Aguayo L. Blockade of Ethanol-Induced Potentiation of Glycine Receptors by a Peptide That Interferes With Gbetagamma Binding. J Pharmacol Exp Ther. 2009;331:933–939. doi: 10.1124/jpet.109.160440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halsey MJ. Effects of High Pressure on the Central Nervous System. Physiol Rev. 1982;62:1341–1377. doi: 10.1152/physrev.1982.62.4.1341. [DOI] [PubMed] [Google Scholar]
- Halsey MJ, Eger EI, Kent DW, Warne PJ. High pressure studies of anesthesia. In: Fink BR, editor. Progress in Anesthesiology. New York: Raven Press; 1970. pp. 353–361. [Google Scholar]
- Halsey MJ, Wardley-Smith B. Pressure Reversal of Narcosis Produced by Anesthetics, Narcotics and Tranquilizers. Nature. 1975;257:811–813. doi: 10.1038/257811a0. [DOI] [PubMed] [Google Scholar]
- Halsey MJ, Wardley-Smith B, Green CJ. Pressure Reversal of General Anaesthesia - A Multi-Site Expansion Hypothesis. Br J Anesth. 1978;50:1091–1097. doi: 10.1093/bja/50.11.1091. [DOI] [PubMed] [Google Scholar]
- Hanchar HJ, Chutsrinopkun P, Meera P, Supavilai P, Sieghart W, Wallner M, Olsen RW. Ethanol Potently and Competitively Inhibits Binding of the Alcohol Antagonist Ro15-4513 to Alpha4/6beta3delta GABAA Receptors. Proc Natl Acad Sci U S A. 2006;103:8546–8551. doi: 10.1073/pnas.0509903103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanchar HJ, Dodson PD, Olsen RW, Otis TM, Wallner M. Alcohol-Induced Motor Impairment Caused by Increased Extrasynaptic GABA(A) Receptor Activity. Nat Neurosci. 2005;8:339–345. doi: 10.1038/nn1398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris RA. Ethanol Actions on Multiple Ion Channels: Which Are Important? Alcohol Clin Exp Res. 1999;23:1563–1570. [PubMed] [Google Scholar]
- Harris RA, Brodie MS, Dunwiddie TV. Possible Substrates of Ethanol Reinforcement: GABA and Dopamine. Ann N Y Acad Sci. 1992;654:61–69. doi: 10.1111/j.1749-6632.1992.tb25956.x. [DOI] [PubMed] [Google Scholar]
- Harris RA, Trudell JR, Mihic SJ. Ethanol's Molecular Targets. Sci Signal. 2008;1:re7. doi: 10.1126/scisignal.128re7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey R, Depner U, Wässle H, Ahmadi S, Heindl C, Reinold H, Smart T, Harvey K, Schütz B, Abo-Salem O, Zimmer A, Poisbeau P, Welzl H, Wolfer D, Betz H, Zeilhofer H, Müller U. GlyR Alpha3: an Essential Target for Spinal PGE2-Mediated Inflammatory Pain Sensitization. Science. 2004;304:884–887. doi: 10.1126/science.1094925. [DOI] [PubMed] [Google Scholar]
- Harwood HJ. The Lewin Group. Contract No. N01-AA-7-1010. National Institute on Alcohol Abuse and Alcoholism; 2000. Updating estimates of the economic costs of alcohol abuse in the United States: Estimates, update methods, and data. [Google Scholar]
- Heimburg T, Jackson AD. The Thermodynamics of General Anesthesia. Biophys J. 2007;92:3159–3165. doi: 10.1529/biophysj.106.099754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemmings HC, Jr, Akabas MH, Goldstein PA, Trudell JR, Orser BA, Harrison NL. Emerging Molecular Mechanisms of General Anesthetic Action. Trends Pharmacol Sci. 2005;26:503–510. doi: 10.1016/j.tips.2005.08.006. [DOI] [PubMed] [Google Scholar]
- Hilf RJ, Dutzler R. Structure of a Potentially Open State of a Proton-Activated Pentameric Ligand-Gated Ion Channel. Nature. 2009;457:115–118. doi: 10.1038/nature07461. [DOI] [PubMed] [Google Scholar]
- Honig B, Hubbell WL. Stability of "Salt Bridges" in Membrane Proteins. Proc Natl Acad Sci U S A. 1984;81:5412–5416. doi: 10.1073/pnas.81.17.5412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang P, Bertaccini E, Loew GH. Molecular Dynamics Simulation of Anesthetic-Phospholipid Bilayer Interactions. J Biomol Struct Dyn. 1995;12:725–754. doi: 10.1080/07391102.1995.10508773. [DOI] [PubMed] [Google Scholar]
- Hunt WA. Alcohol and Biological Membranes. New York: Guilford Press; 1985. [Google Scholar]
- Hunter WL, Bennett PB. The Causes, Mechanisms and Prevention of the High Pressure Nervous Syndrome. Undersea Biomed Res. 1974;1:1–28. [PubMed] [Google Scholar]
- Jenkins A, Greenblatt EP, Faulkner HJ, Bertaccini E, Light A, Lin A, Andreasen A, Viner A, Trudell JR, Harrison NL. Evidence for a Common Binding Cavity for Three General Anesthetics Within the GABAA Receptor. J Neurosci. 2001;21:RC136. doi: 10.1523/JNEUROSCI.21-06-j0002.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang ZL, Ye JH. Protein Kinase C Epsilon Is Involved in Ethanol Potentiation of Glycine-Gated Cl− Current in Rat Neurons of Ventral Tegmental Area. Neuropharmacology. 2003;44:493–502. doi: 10.1016/s0028-3908(02)00409-4. [DOI] [PubMed] [Google Scholar]
- Johnson FH, Brown DE, Marsland DA. Pressure Reversal of the Action of Certain Narcotics. J Cell Comp Physiol. 1942;20:269–276. [Google Scholar]
- Johnson FH, Flagler EA. Hydrostatic Pressure Reversal of Narcosis in Tadpoles. Science. 1950;112:91–92. doi: 10.1126/science.112.2899.91-a. [DOI] [PubMed] [Google Scholar]
- Jones MV, Harrison NL. Effects of Volatile Anesthetics on the Kinetics of Inhibitory Postsynaptic Currents in Cultured Rat Hippocampal Neurons. J Neurophysiol. 1993;70:1339–1349. doi: 10.1152/jn.1993.70.4.1339. [DOI] [PubMed] [Google Scholar]
- Jonsson S, Kerekes N, Hyytia P, Ericson M, Soderpalm B. Glycine Receptor Expression in the Forebrain of Male AA/ANA Rats. Brain Res. 2009;1305 Suppl:S27–S36. doi: 10.1016/j.brainres.2009.09.053. [DOI] [PubMed] [Google Scholar]
- Jung S, Akabas MH, Harris RA. Functional and Structural Analysis of the GABAA Receptor Alpha 1 Subunit During Channel Gating and Alcohol Modulation. J Biol Chem. 2005;280:308–316. doi: 10.1074/jbc.M409871200. [DOI] [PubMed] [Google Scholar]
- Jung S, Harris RA. Sites in TM2 and 3 Are Critical for Alcohol-Induced Conformational Changes in GABA Receptors. J Neurochem. 2006;96:885–892. doi: 10.1111/j.1471-4159.2005.03617.x. [DOI] [PubMed] [Google Scholar]
- Karlin A. Emerging Structure of the Nicotinic Acetylcholine Receptors. Nat Reviews, Neurosci. 2002;3 doi: 10.1038/nrn731. 102-1. [DOI] [PubMed] [Google Scholar]
- Kash TL, Dizon MJ, Trudell JR, Harrison NL. Charged Residues in the Beta2 Subunit Involved in GABAA Receptor Activation. J Biol Chem. 2004a;279:4887–4893. doi: 10.1074/jbc.M311441200. [DOI] [PubMed] [Google Scholar]
- Kash TL, Jenkins A, Kelley JC, Trudell JR, Harrison NL. Coupling of Agonist Binding to Channel Gating in the GABAA Receptor. Nature. 2003;421:272–275. doi: 10.1038/nature01280. [DOI] [PubMed] [Google Scholar]
- Kash TL, Trudell JR, Harrison NL. Evaluation of a Proposed Mechanism of Ligand-Gated Ion Channel Activation in the GABAA and Glycine Receptors. Neurosci Lett. 2004b;371:230–234. doi: 10.1016/j.neulet.2004.09.002. [DOI] [PubMed] [Google Scholar]
- Kucken AM, Wagner DA, Ward PR, Teissere JA, Boileau AJ, Czajkowski C. Identification of Benzodiazepine Binding Site Residues in the γ2 Subuit of the γ-Aminobutyric AcidA Receptor. Mol Pharmacol. 2000;57:932–939. [PubMed] [Google Scholar]
- Laube B, Kuhse J, Rundström N, Kirsch J, Schmieden V, Betz H. Modulation by Zinc Ions of Native Rat and Recombinant Human Inhibitory Glycine Receptors. J Physiol (Lond) 1995;483:613–619. doi: 10.1113/jphysiol.1995.sp020610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee AG. Structural Biology: Highly Charged Meetings. Nature. 2009;462:420–421. doi: 10.1038/462420a. [DOI] [PubMed] [Google Scholar]
- Legendre P. The Glycinergic Inhibitory Synapse. Cell Mol Life Sci. 2001;58:760–793. doi: 10.1007/PL00000899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lever MJ, Miller KW, Paton WD, Smith EB. Pressure Reversal of Anaesthesia. Nature. 1971;231:368–371. doi: 10.1038/231368a0. [DOI] [PubMed] [Google Scholar]
- Li C, Peoples RW, Weight FF. Alcohol Action on a Neuronal Membrane Receptor: Evidence for a Direct Interaction With the Receptor Protein. Proc Natl Acad Sci U S A. 1994;91:8200–8204. doi: 10.1073/pnas.91.17.8200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang J, Zhang N, Cagetti E, Houser C, Olsen R, Spigelman I. Chronic Intermittent Ethanol-Induced Switch of Ethanol Actions From Extrasynaptic to Synaptic Hippocampal GABAA Receptors. J Neurosci. 2006;26:1749–1758. doi: 10.1523/JNEUROSCI.4702-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin L-H, Whiting P, Harris RA. Molecular Determinants of General Anesthetic Action: Role of GABAA Receptor Structure. J Neurochem. 1993;60(4):1548–1553. doi: 10.1111/j.1471-4159.1993.tb03320.x. [DOI] [PubMed] [Google Scholar]
- Lister RG, Nutt DJ. Is Ro 15-4513 a Specific Alcohol Antagonist? TINS. 1987;10:223–225. [Google Scholar]
- Little HJ. Mechanisms That May Underlie the Behavioural Effects of Ethanol. Prog Neurobiol. 1991;36:171–194. doi: 10.1016/0301-0082(91)90029-z. [DOI] [PubMed] [Google Scholar]
- Liu QR, Lopez-Corcuera B, Mandiyan S, Nelson H, Nelson N. Cloning and Expression of a Spinal Cord- and Brain-Specific Glycine Transporter With Novel Structural Features. J Biol Chem. 1993;268:22802–22808. [PubMed] [Google Scholar]
- Lobo IA, Harris RA. Sites of Alcohol and Volatile Anesthetic Action on Glycine Receptors. Int Rev Neurobiol. 2005;65:53–87. doi: 10.1016/S0074-7742(04)65003-3. [DOI] [PubMed] [Google Scholar]
- Lobo IA, Harris RA, Trudell JR. Cross-Linking of Sites Involved With Alcohol Action Between Transmembrane Segments 1 and 3 of the Glycine Receptor Following Activation. J Neurochem. 2008;104:1649–1662. doi: 10.1111/j.1471-4159.2007.05090.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lobo IA, Mascia MP, Trudell JR, Harris RA. Channel Gating of the Glycine Receptor Changes Accessibility to Residues Implicated in Receptor Potentiation by Alcohols and Anesthetics. J Biol Chem. 2004a;279:33919–33927. doi: 10.1074/jbc.M313941200. [DOI] [PubMed] [Google Scholar]
- Lobo IA, Trudell JR, Harris RA. Cross-Linking of Glycine Receptor Transmembrane Segments Two and Three Alters Coupling of Ligand Binding With Channel Opening. J Neurochem. 2004b;90:962–969. doi: 10.1111/j.1471-4159.2004.02561.x. [DOI] [PubMed] [Google Scholar]
- Lobo IA, Trudell JR, Harris RA. Accessibility to Residues in Transmembrane Segment Four of the Glycine Receptor. Neuropharmacology. 2006;50:174–181. doi: 10.1016/j.neuropharm.2005.08.017. [DOI] [PubMed] [Google Scholar]
- Lynch JW. Molecular Structure and Function of the Glycine Receptor Chloride Channel. Physiol Rev. 2004;84:1051–1095. doi: 10.1152/physrev.00042.2003. [DOI] [PubMed] [Google Scholar]
- Lynch JW, Han N-L, Haddrill JL, Pierce KD, Schofield PR. The Surface Accessibility of the Glycine Receptor M2-M3 Loop Is Increased in the Channel Open State. J Neurosci. 2001;21:2589–2599. doi: 10.1523/JNEUROSCI.21-08-02589.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lysko GS, Robinson JL, Casto R, Ferrone RA. The Stereospecific Effects of Isoflurane Isomers in Vivo. Eur J Pharmacol. 1994;263:25–29. doi: 10.1016/0014-2999(94)90519-3. [DOI] [PubMed] [Google Scholar]
- Malcolm RD, Alkana RL. Temperature Dependence of Ethanol Depression in Mice. J Pharmacol Exp Ther. 1981;217:770–775. [PubMed] [Google Scholar]
- Malcolm RD, Alkana RL. Hyperbaric Ethanol Antagonism: Role of Temperature, Blood and Brain Ethanol Concentrations. Pharmacol Biochem Behav. 1982;16:341–346. doi: 10.1016/0091-3057(82)90169-1. [DOI] [PubMed] [Google Scholar]
- Malcolm RD, Alkana RL. Temperature Dependence of Ethanol Lethality in Mice. J Pharm Pharmacol. 1983;35:306–311. doi: 10.1111/j.2042-7158.1983.tb02939.x. [DOI] [PubMed] [Google Scholar]
- Malosio ML, Marquèze-Pouey B, Kuhse J, Betz H. Widespread Expression of Glycine Receptor Subunit MRNAs in the Adult and Developing Rat Brain. EMBO J. 1991;10:2401–2409. doi: 10.1002/j.1460-2075.1991.tb07779.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mascia MP, Machu TL, Harris RA. Enhancement of Homomeric Glycine Receptor Function by Long-Chain Alcohols and Anaesthetics. Br J Pharmacol. 1996a;119:1331–1336. doi: 10.1111/j.1476-5381.1996.tb16042.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mascia MP, Mihic SJ, Valenzuela CF, Schofield PR, Harris RA. A Single Amino Acid Determines Differences in Ethanol Actions on Strychnine-Sensitive Glycine Receptors. Mol Pharmacol. 1996b;50:402–406. [PubMed] [Google Scholar]
- Mascia MP, Trudell JR, Harris RA. Specific Binding Sites for Alcohols and Anesthetics on Ligand-Gated Ion Channels. Proc Natl Acad Sci USA. 2000;97:9305–9310. doi: 10.1073/pnas.160128797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mastrangelo CJ, Kendig JJ, Trudell JR, Cohen EN. Nerve Membrane Lipid Fluidity: Opposing Effects of High Pressure and Ethanol. Undersea Biomed Res. 1979;6:47–53. [PubMed] [Google Scholar]
- Mastrangelo CJ, Trudell JR, Cohen EN. Antagonism of Membrane Compression Effects by High Pressure Gas Mixtures in a Phospholipid Bilayer System. Life Sci. 1978;22:239–244. doi: 10.1016/0024-3205(78)90524-6. [DOI] [PubMed] [Google Scholar]
- McCool BA, Frye GD, Pulido MD, Botting SK. Effects of Chronic Ethanol Consumption on Rat GABAA and Strychnine-Sensitive Glycine Receptors Expressed by Lateral/Basolateral Amygdala Neurons. Brain Res. 2003;963:165–177. doi: 10.1016/s0006-8993(02)03966-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCracken LM, Trudell JR, Goldstein BE, Harris RA, Mihic SJ. Zinc Enhances Ethanol Modulation of the Alpha1 Glycine Receptor. Neuropharmacology. 2010;58:676–681. doi: 10.1016/j.neuropharm.2009.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehta AK, Ticku MK. Chronic Ethanol Treatment Alters the Behavioral Effects of Ro 15-4513, a Partially Negative Ligand for Benzodiazepine Binding Sites. Brain Research. 1989;489:93–100. doi: 10.1016/0006-8993(89)90011-5. [DOI] [PubMed] [Google Scholar]
- Meyer HH. Zur Theorie Der Alkoholnarkose. Arch Exp Path Pharmacol. 1899;42:109–118. [Google Scholar]
- Meyer HH. Zur Theorie Dir Alkohonarkose. Der Einfluss Wechselnder Temmperatur Auf Wirkaunsstarke Und Theilungskoefficient Der Narcotica. Arch Exp Path Pharmacol. 1901;56:338–346. [Google Scholar]
- Meyer K. Contributions to the Theory of Narcosis. Trans Faraday Soc. 1937;33:1068. [Google Scholar]
- Mhatre M, Ticku MK. Chronic Ethanol Treatment Selectively Increases the Binding of Inverse Agonists for Benzodiazepine Binding Sites in Cultured Spinal Cord Neurons. J Pharmacol Exp Ther. 1989;251:164–168. [PubMed] [Google Scholar]
- Mihic SJ, Harris RA. Inhibition of ρ1 Receptor GABAergic Currents by Alcohols and Volatile Anesthetics. J Pharmacol Exp Ther. 1996;277:411–416. [PubMed] [Google Scholar]
- Mihic SJ, Ye Q, Wick MJ, Koltchine VV, Krasowski MD, Finn SE, Mascia MP, Valenzuela CF, Hanson KK, Greenblatt EP, Harris RA, Harrison NL. Sites of Alcohol and Volatile Anaesthetic Action on GABAA and Glycine Receptors. Nature. 1997;389:385–389. doi: 10.1038/38738. [DOI] [PubMed] [Google Scholar]
- Miller KW. The Opposing Physiological Effects of High Pressures and Inert Gases. Fed Proc. 1977;36:1663–1667. [PubMed] [Google Scholar]
- Miller KW, Paton WDM, Smith RA, Smith EB. The Pressure Reversal of Anaesthesia and the Critical Volume Hypothesis. Mol Pharmacol. 1973;9:131–143. [PubMed] [Google Scholar]
- Miller KW, Wilson MW. The Pressure Reversal of a Variety of Anesthetic Agents in Mice. Anesthesiology. 1978;48:104–110. doi: 10.1097/00000542-197802000-00005. [DOI] [PubMed] [Google Scholar]
- Mody I, Glykys J, Wei W. A New Meaning for "Gin & Tonic": Tonic Inhibition As the Target for Ethanol Action in the Brain. Alcohol. 2007;41:145–153. doi: 10.1016/j.alcohol.2007.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molander A, Lidö HH, Löf E, Ericson M, Söderpalm B. The Glycine Reuptake Inhibitor Org 25935 Decreases Ethanol Intake and Preference in Male Wistar Rats. Alcohol Alcohol. 2007;42:11–18. doi: 10.1093/alcalc/agl085. [DOI] [PubMed] [Google Scholar]
- Molander A, Söderpalm B. Accumbal Strychnine-Sensitive Glycine Receptors: an Access Point for Ethanol to the Brain Reward System. Alcohol Clin Exp Res. 2005;29:27–37. doi: 10.1097/01.alc.0000150012.09608.81. [DOI] [PubMed] [Google Scholar]
- Monaghan DT, Bridges RJ, Cotman CW. The Excitatory Amino Acid Receptors: Their Classes, Pharmacology, and Distinct Properties in the Function of the Central Nervous System. Annu Rev Pharmacol Toxicol. 1989;29:365–402. doi: 10.1146/annurev.pa.29.040189.002053. [DOI] [PubMed] [Google Scholar]
- Mori T, Matubayasi N, Ueda I. Membrane Expansion and Inhalation Anesthetics. Mean Excess Volume Hypothesis. Mol Pharmacol. 1984;25:123–130. [PubMed] [Google Scholar]
- Morrow A, Ferrani-Kile K, Davis M, Shumilla J, Kumar S, Maldve R, Pandey S. Ethanol Effects on Cell Signaling Mechanisms. Alcohol Clin Exp Res. 2004;28:217–227. doi: 10.1097/01.alc.0000113439.97498.ac. [DOI] [PubMed] [Google Scholar]
- Morton A, Baase WA, Matthews BW. Energetic Origins of Specificity of Ligand Binding in an Interior Nonpolar Cavity of T4 Lysozyme. Biochemistry. 1995;34:8564–8575. doi: 10.1021/bi00027a006. [DOI] [PubMed] [Google Scholar]
- Morton A, Matthews BW. Specificity of Ligand Binding in a Buried Nonpolar Cavity of T4 Lysozyme: Linkage of Dynamics and Structural Plasticity. Biochemistry. 1995;34:8576–8588. doi: 10.1021/bi00027a007. [DOI] [PubMed] [Google Scholar]
- Mullins LJ, Gaffey CT. The Depolarization of Nerve by Organic Compounds. Proc Soc Exp Biol Med. 1954;85:144–149. doi: 10.3181/00379727-85-20812. [DOI] [PubMed] [Google Scholar]
- Nielsen ST, Beninati L, Chang J. REV 5901 and Ly 171,883 Protect Rat Gastric Mucosa Against Ethanol-Induced Damage. Agents Actions. 1987 Aug 21;:320–322. doi: 10.1007/BF01966503. [DOI] [PubMed] [Google Scholar]
- Ortells MO, Lunt GG. Evolutionary History of the Ligand-Gated Ion-Channel Superfamily of Receptors. Trends Neurosci. 1995;18:121–127. doi: 10.1016/0166-2236(95)93887-4. [DOI] [PubMed] [Google Scholar]
- Overton C. Studien Über Die Narkose Zugleich Ein Beitrag Zur Allgemeinen Pharmakologie. Gustav Fischer, Jena, Switzerland. 1901 [Google Scholar]
- Pang KY, Braswell LM, Chang L, Sommer TJ, Miller KW. The Perturbation of Lipid Bilayers by General Anesthetics: a Quantitative Test of the Disordered Lipid Hypothesis. Mol Pharmacol. 1980;18:84–90. [PubMed] [Google Scholar]
- Perkins DI, Trudell JR, Crawford DK, Alkana RL, Davies D. Targets for Ethanol Action and Antagonism in Loop 2 of the Extracellular Domain of Glycine Receptors. J Neurochem. 2008;106:1337–1349. doi: 10.1111/j.1471-4159.2008.05476.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perkins DI, Trudell JR, Crawford DK, Asatryan L, Alkana RL, Davies DL. Loop 2 Structure in Glycine and GABAA Receptors Plays a Key Role in Determining Ethanol Sensitivity. J Biol Chem. 2009;284:27304–27314. doi: 10.1074/jbc.M109.023598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ponce J, Poyatos I, Aragon C, Gimenez C, Zafra F. Characterization of the 5' Region of the Rat Brain Glycine Transporter GlyT2 Gene: Identification of a Novel Isoform. Neurosci Lett. 1998;242:25–28. doi: 10.1016/s0304-3940(98)00037-8. [DOI] [PubMed] [Google Scholar]
- Popova M, Asatryan L, Ostrovskaya O, Wyatt RL, Li K, Alkana RL, Davies DL. A Point Mutation in the Ectodomain-Transmembrane 2 Interface Eliminates the Inhibitory Effects of Ethanol in P2X4 Receptors. J Neurochem. 2010;112:307–317. doi: 10.1111/j.1471-4159.2009.06460.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poyatos I, Ponce J, Aragon C, Gimenez C, Zafra F. The Glycine Transporter GLYT2 Is a Reliable Marker for Glycine-Immunoreactive Neurons. Brain Res Mol Brain Res. 1997;49:63–70. doi: 10.1016/s0169-328x(97)00124-1. [DOI] [PubMed] [Google Scholar]
- Pringle MJ, Brown KB, Miller KW. Can the Lipid Theories of Anesthesia Account for the Cutoff in Anesthetic Potency in Homologous Series of Alcohols? Mol Pharmacol. 1981;19:49–55. [PubMed] [Google Scholar]
- Purohit P, Auerbach A. Acetylcholine Receptor Gating: Movement in the Alpha-Subunit Extracellular Domain. J Gen Physiology. 2007;130:569–579. doi: 10.1085/jgp.200709858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quillin ML, Breyer WA, Griswold IJ, Matthews BW. Size Versus Polarizability in Protein-Ligand Interactions: Binding of Noble Gases Within Engineered Cavities in Phage T4 Lysozyme. J Mol Biol. 2000;302:955–977. doi: 10.1006/jmbi.2000.4063. [DOI] [PubMed] [Google Scholar]
- Quinlan JJ, Ferguson C, Jester K, Firestone LL, Homanics GE. Mice With Glycine Receptor Subunit Mutations Are Both Sensitive and Resistant to Volatile Anesthetics. Anesth Analg. 2002;95:578–582. doi: 10.1097/00000539-200209000-00016. table. [DOI] [PubMed] [Google Scholar]
- Raines DE, Claycomb RJ, Scheller M, Forman SA. Nonhalogenated Alkane Anesthetics Fail to Potentiate Agonist Actions on Two Ligand-Gated Ion Channels. Anesthesiology. 2001;95:470–477. doi: 10.1097/00000542-200108000-00032. [DOI] [PubMed] [Google Scholar]
- Rajendra S, Lynch JW, Schofield PR. The Glycine Receptor. Pharmacol Ther. 1997;73:121–146. doi: 10.1016/s0163-7258(96)00163-5. [DOI] [PubMed] [Google Scholar]
- Rajendram R, Preedy VR. Effect of Alcohol Consumption on the Gut. Digestive Diseases. 2005;23:214–221. doi: 10.1159/000090168. [DOI] [PubMed] [Google Scholar]
- Rashin AA, Iofin M, Honig B. Internal Cavities and Buried Waters in Globular Proteins. Biochemistry. 1986;25:3619–3625. doi: 10.1021/bi00360a021. [DOI] [PubMed] [Google Scholar]
- Rewal M, Jurd R, Gill TM, He DY, Ron D, Janak PH. Alpha4-Containing GABAA Receptors in the Nucleus Accumbens Mediate Moderate Intake of Alcohol. J Neurosci. 2009;29:543–549. doi: 10.1523/JNEUROSCI.3199-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts MT, Phelan R, Erlichman BS, Pillai RN, Ma L, Lopreato GF, Mihic SJ. Occupancy of a Single Anesthetic Binding Pocket Is Sufficient to Enhance Glycine Receptor Function. J Biol Chem. 2005;281:3305–3311. doi: 10.1074/jbc.M502000200. [DOI] [PubMed] [Google Scholar]
- Ron D, Jurd R. The "Ups and Downs" of Signaling Cascades in Addiction. Sci STKE. 2005:309. doi: 10.1126/stke.3092005re14. [DOI] [PubMed] [Google Scholar]
- Roth SH. Physical Mechanisms of Anesthesia. Annu Rev Pharmacol Toxicol. 1979;19:159–178. doi: 10.1146/annurev.pa.19.040179.001111. [DOI] [PubMed] [Google Scholar]
- Santhakumar V, Wallner M, Otis T. Ethanol Acts Directly on Extrasynaptic Subtypes of GABAA Receptors to Increase Tonic Inhibition. Alcohol. 2007;41:211–221. doi: 10.1016/j.alcohol.2007.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheller M, Forman SA. The Gamma Subunit Determines Whether Anesthetic-Induced Leftward Shift Is Altered by a Mutation at Alpha1 S270 in α1β2γ2L GABA(A) Receptors. Anesthesiology. 2001;95:123–131. doi: 10.1097/00000542-200107000-00022. [DOI] [PubMed] [Google Scholar]
- Schwartz TW, Hubbell WL. Structural Biology: A Moving Story of Receptors. Nature. 2008;455:473–474. doi: 10.1038/455473a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sebe JY, Eggers ED, Berger AJ. Differential Effects of Ethanol on GABA(A) and Glycine Receptor-Mediated Synaptic Currents in Brain Stem Motoneurons. J Neurophysiol. 2003;90:870–875. doi: 10.1152/jn.00119.2003. [DOI] [PubMed] [Google Scholar]
- Seeman P. The Membrane Actions of Anesthetics and Tranquilizers. Pharmacol Rev. 1972;24:583–633. [PubMed] [Google Scholar]
- Seeman P, Roth S. General Anesthetics Expand Cell Membranes at Surgical Concentrations. Biochim Biophys Acta. 1972;255:171–177. doi: 10.1016/0005-2736(72)90019-3. [DOI] [PubMed] [Google Scholar]
- Shan Q, Haddrill JL, Lynch JW. Ivermectin, an Unconventional Agonist of the Glycine Receptor Chloride Channel. J Biol Chem. 2001;276:12556–12564. doi: 10.1074/jbc.M011264200. [DOI] [PubMed] [Google Scholar]
- Sigel E, Buhr A. The Benzodiazepine Binding Site of GABAA Receptors. TiPS. 1997;18:425–429. doi: 10.1016/s0165-6147(97)01118-8. [DOI] [PubMed] [Google Scholar]
- Sommer B, Seeburg P. Glutamate Receptor Channels: Novel Properties and New Clones. Trends Pharmacol Sci. 1992;13:291–296. doi: 10.1016/0165-6147(92)90088-n. [DOI] [PubMed] [Google Scholar]
- Syapin PJ, Chen J, Finn DA, Alkana RL. Antagonism of Ethanol-Induced Depression of Mouse Locomotor Activity by Hyperbaric Exposure. Life Sci. 1988;43:2221–2229. doi: 10.1016/0024-3205(88)90415-8. [DOI] [PubMed] [Google Scholar]
- Syapin PJ, Jones BL, Finn DA, Davies DL, Alkana RL. Effect of 12 Atmospheres Helium-Oxygen on the Response of Mice to Convulsant Drugs. Undersea and Hyperbaric Medicine. 1996;23:35–41. [PubMed] [Google Scholar]
- Tao L, Ye JH. Protein Kinase C Modulation of Ethanol Inhibition of Glycine-Activated Currnt in Dissociated Neurons of Rat Ventral Tegmental Area. J Pharmacol Exp Ther. 2002;300:967–975. doi: 10.1124/jpet.300.3.967. [DOI] [PubMed] [Google Scholar]
- Tapia JC, Aguilar LF, Sotomayor CP, Aguayo LG. Ethanol Affects the Function of a Neurotransmitter Receptor Protein Without Altering the Membrane Lipid Phase. Eur J Pharmacol. 1998;354:239–244. doi: 10.1016/s0014-2999(98)00445-2. [DOI] [PubMed] [Google Scholar]
- Trudell JR. The Membrane Volume Occupied by Anesthetic Molecules: a Reinterpretation of the Erythrocyte Expansion Data. Biochim Biophys Acta. 1977;470:509–510. doi: 10.1016/0005-2736(77)90143-2. [DOI] [PubMed] [Google Scholar]
- Trudell JR, Hubbell WL, Cohen EN. Pressure Reversal of Inhalation Anesthetic-Induced Disorder in Spin-Labeled Phospholipid Vesicles. Biochim Biophys Acta. 1973a;291:328–334. doi: 10.1016/s0005-2736(73)80001-x. [DOI] [PubMed] [Google Scholar]
- Trudell JR, Yue ME, Bertaccini EJ, Jenkins A, Harrison NL. Molecular Modeling and Mutagenesis Reveals a Tetradentate Binding Site for Zn2+ in GABA(A) Alphabeta Receptors and Provides a Structural Basis for the Modulating Effect of the Gamma Subunit. J Chem Inf Model. 2008;48:344–349. doi: 10.1021/ci700324a. [DOI] [PubMed] [Google Scholar]
- Trudell J, Hubbell W, Cohen E. The Effect of Two Inhalation Anesthetics on the Order of Spin-Labeled Phospholipid Vesicles. Biochim Biophys Acta. 1973b;291:321–327. doi: 10.1016/0005-2736(73)90485-9. [DOI] [PubMed] [Google Scholar]
- Ueda I, Mashimo T. Anesthetics Expand Partial Molal Volume of Lipid-Free Protein Dissolved in Water: Electrostriction Hypothesis. Physiol Chem Phys. 1982;14:157–164. [PubMed] [Google Scholar]
- Unwin N. Refined Structure of the Nicotinic Acetylcholine Receptor at 4Å Resolution. J Mol Biol. 2005;346:967–989. doi: 10.1016/j.jmb.2004.12.031. [DOI] [PubMed] [Google Scholar]
- Valenzuela CF, Cardoso RA, Wick MJ, Weiner JL, Dunwiddie TV, Harris RA. Effects of Ethanol on Recombinant Glycine Receptors Expressed in Mammalian Cell Lines. Alcohol Clin Exp Res. 1998;22:1132–1136. [PubMed] [Google Scholar]
- Volpicelli JR. Alcohol Abuse and Alcoholism: an Overview. J Clin Psychiatry. 2001;62:4–10. [PubMed] [Google Scholar]
- Wallner M, Hanchar HJ, Olsen RW. Ethanol Enhances Alpha 4 Beta 3 Delta and Alpha 6 Beta 3 Delta Gamma-Aminobutyric Acid Type A Receptors at Low Concentrations Known to Affect Humans. Proc Natl Acad Sci U S A. 2003;100:15218–15223. doi: 10.1073/pnas.2435171100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallner M, Hanchar HJ, Olsen RW. Low-Dose Alcohol Actions on Alpha4beta3delta GABAA Receptors Are Reversed by the Behavioral Alcohol Antagonist Ro15-4513. Proc Natl Acad Sci U S A. 2006;103:8540–8545. doi: 10.1073/pnas.0600194103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wann KT, MacDonald AG. Actions and Interactions of High Pressure and General Anesthetics. Prog Neurobiol. 1988;30:271–307. doi: 10.1016/0301-0082(88)90025-1. [DOI] [PubMed] [Google Scholar]
- Watanabe E, Akagi H. Distribution Patterns of MRNAs Encoding Glycine Receptor Channels in the Developing Rat Spinal Cord. Neurosci Res. 1995;23:377–382. doi: 10.1016/0168-0102(95)00972-V. [DOI] [PubMed] [Google Scholar]
- Webb T, Lynch J. Molecular Pharmacology of the Glycine Receptor Chloride Channel. Curr Pharm Des. 2007;13:2350–2367. doi: 10.2174/138161207781368693. [DOI] [PubMed] [Google Scholar]
- Wei W, Faria L, Mody I. Low Ethanol Concentrations Selectively Augment the Tonic Inhibition Mediated by Delta Subunit-Containing GABAA Receptors in Hippocampal Neurons. J Neurosci. 2004;24:8379–8382. doi: 10.1523/JNEUROSCI.2040-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weight FF, Aguayo LG, White G, Lovinger DM, Peoples RW. GABA- and Glutamate-Gated Ion Channels As Molecular Sites of Alcohol and Anesthetic Action. Adv Biochem Psychopharmacol. 1992;47:335–347. [PubMed] [Google Scholar]
- Weight FF, Li C, Peoples RW. Alcohol Action on Membrane Ion Channels Gated by Extracellular ATP (P2X Receptors) Neurochem Int. 1999;35:143–152. doi: 10.1016/s0197-0186(99)00056-x. [DOI] [PubMed] [Google Scholar]
- Weiner JL, Gu C, Dunwiddie TV. Differential Ethanol Sensitivity of Subpopulations of GABAA Synapses Onto Rat CA1 Pyramidal Neurons. J Neurophys. 1997;77:1306–1312. doi: 10.1152/jn.1997.77.3.1306. [DOI] [PubMed] [Google Scholar]
- Welsh BT, Goldstein BE, Mihic SJ. Single-Channel Analysis of Ethanol Enhancement of Glycine Receptor Function. J Pharmacol Exp Ther. 2009;330:198–205. doi: 10.1124/jpet.109.154344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wenningmann I, Barann M, Vidal AM, Dilger JP. The Effects of Isoflurane on Acetylcholine Receptor Channels: 3. Effects of Conservative Polar-to-Nonpolar Mutations Within the Channel Pore. Mol Pharmacol. 2001;60:584–594. [PubMed] [Google Scholar]
- White G, Lovinger DM, Weight FF. Ethanol Inhibits NMDA-Activated Current but Does Not Alter GABA- Activated Current in an Isolated Adult Mammalian Neuron. Brain Res. 1990;507:332–336. doi: 10.1016/0006-8993(90)90292-j. [DOI] [PubMed] [Google Scholar]
- Wick MJ, Mihic SJ, Ueno S, Mascia MP, Trudell JR, Brozowski SJ, Ye Q, Harrison NL, Harris RA. Mutations of γ-Aminobutyric Acid and Glycine Receptors Change Alcohol Cutoff: Evidence for an Alcohol Receptor? Proc Natl Acad Sci U S A. 1998;95:6504–6509. doi: 10.1073/pnas.95.11.6504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wieland HA, Lüddens H, Seeburg PH. A Single Histadine in GABAA Receptors Is Essential for Benzodiazepine Agonist Binding. J Biol Chem. 1992;267(3):1426–1429. [PubMed] [Google Scholar]
- Williams DB, Akabas MH. GABA Increases the Water-Accessibility of M3 Membrane-Spanning Residues in GABA-A Receptors. Biophys J. 1999;77:2563–2574. doi: 10.1016/s0006-3495(99)77091-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams KL, Ferko AP, Barbieri EJ, Di Gregorio GJ. Glycine Enhances the Central Depressant Properties of Ethanol in Mice. Pharmacol Biochem Behav. 1995;50:199–205. doi: 10.1016/0091-3057(94)00288-t. [DOI] [PubMed] [Google Scholar]
- Woodward JJ, Nowak M, Davies DL. Effects of the Abused Solvent Toluene on Recombinant P2X Receptors Expressed in HEK293 Cells. Molecular Brain Research. 2004;125:86–95. doi: 10.1016/j.molbrainres.2004.03.005. [DOI] [PubMed] [Google Scholar]
- Xiong KM, Li C, Weight FF. Differential Ethanol Sensitivity of P2X4 and P2X4/P2X6 Receptors Expressed in Xenopus Oocytes. Alcohol Clin Exp Res. 1999;23:8A. [Google Scholar]
- Yamakura T, Harris RA. Effects of Gaseous Anesthetics Nitrous Oxide and Xenon on Ligand-Gated Ion Channels. Comparison With Isoflurane and Ethanol. Anesthesiology. 2000;93:1095–1101. doi: 10.1097/00000542-200010000-00034. [DOI] [PubMed] [Google Scholar]
- Yamakura T, Mihic SJ, Harris RA. Amino Acid Volume and Hydropathy of a Transmembrane Site Determine Glycine and Anesthetic Sensitivity of Glycine Receptors. J Biol Chem. 1999;274:23006–23012. doi: 10.1074/jbc.274.33.23006. [DOI] [PubMed] [Google Scholar]
- Yang Z, Cromer BA, Harvey RJ, Parker MW, Lynch JW. A Proposed Structural Basis for Picrotoxinin and Picrotin Binding in the Glycine Receptor Pore. Journal of Neurochemistry. 2007;103:580–589. doi: 10.1111/j.1471-4159.2007.04850.x. [DOI] [PubMed] [Google Scholar]
- Ye JH, Tao L, Krnjevic K, McArdle JJ. Ethanol Inhibition of Glycine-Activated Responses in Neurons of Ventral Tegmental Area of Neonatal Rats. J Neurophysiol. 2001a;86:2426–2434. doi: 10.1152/jn.2001.86.5.2426. [DOI] [PubMed] [Google Scholar]
- Ye JH, Tao L, Krnjevic K, McArdle JJ. Decay of Ethanol-Induced Suppression of Glycine-Activated Current of Ventral Tegmental Area Neurons. Neuropharmacology. 2002;43:788–798. doi: 10.1016/s0028-3908(02)00179-x. [DOI] [PubMed] [Google Scholar]
- Ye JH, Tao L, Ren J, Schaefer R, Krnjevic K, Liu PL, Schiller DA, McArdle JJ. Ethanol Potentiation of Glycine-Induced Responses in Dissociated Neurons of Rat Ventral Tegmental Area. J Pharmacol Exp Ther. 2001b;296:77–83. [PubMed] [Google Scholar]
- Ye Q, Koltchine VV, Mihic SJ, Mascia MP, Wick MJ, Finn SE, Harrison NL, Harris RA. Enhancement of Glycine Receptor Function by Ethanol Is Inversely Correlated With Molecular Volume at Position alpha267. J Biol Chem. 1998;273(6):3314–3319. doi: 10.1074/jbc.273.6.3314. [DOI] [PubMed] [Google Scholar]
- Yevenes GE, Moraga-Cid G, Guzmán L, Haeger S, Oliveira L, Olate J, Schmalzing G, Aguayo LG. Molecular Determinants for G Protein Betagamma Modulation of Ionotropic Glycine Receptors. J Biol Chem. 2006;281:39300–39307. doi: 10.1074/jbc.M608272200. [DOI] [PubMed] [Google Scholar]
- Yevenes G, Moraga-Cid G, Peoples R, Schmalzing G, Aguayo LG. A Selective G Betagamma-Linked Intracellular Mechanism for Modulation of a Ligand-Gated Ion Channel by Ethanol. Proc Natl Acad Sci U S A. 2008;105:20523–20528. doi: 10.1073/pnas.0806257105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeilhofer HU, Studler B, Arabadzisz D, Schweizer C, Ahmadi S, Layh B, Bosl MR, Fritschy JM. Glycinergic Neurons Expressing Enhanced Green Fluorescent Protein in Bacterial Artificial Chromosome Transgenic Mice. J Comp Neurol. 2005;482:123–141. doi: 10.1002/cne.20349. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Laster MJ, Hara K, Harris RA, Eger EI, Stabernack CR, Sonner JM. Glycine Receptors Mediate Part of the Immobility Produced by Inhaled Anesthetics. Anesth Analg. 2003;96:97–101. doi: 10.1097/00000539-200301000-00021. table. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Wu S, Eger EI, Sonner JM. Neither GABA(A) nor Strychnine-Sensitive Glycine Receptors Are the Sole Mediators of MAC for Isoflurane. Anesth Analg. 2001;92:123–127. doi: 10.1097/00000539-200101000-00024. [DOI] [PubMed] [Google Scholar]
- Zhou Q, Lovinger DM. Pharmacologic Characteristics of Potentiation of 5-HT3 Receptors by Alcohols and Diethyl Ether in NCB-20 Neuroblastoma Cells. J Pharmacol Exp Ther. 1996;278:732–740. [PubMed] [Google Scholar]
- Ziskind-Conhaim L, Gao BX, Hinckley C. Ethanol Dual Modulatory Actions on Spontaneous Postsynaptic Currents in Spinal Motoneurons. J Neurophysiol. 2003;89:806–813. doi: 10.1152/jn.00614.2002. [DOI] [PubMed] [Google Scholar]