Abstract
In this review we discuss the implications for ventilatory control of newer evidence suggesting that central and peripheral chemoreceptors are not functionally separate but rather that they are dependent upon one another such that the sensitivity of the medullary chemoreceptors is critically determined by input from the carotid body chemoreceptors and vice versa i.e., they are interdependent. We examine potential interactions of the interdependent central and carotid body (CB) chemoreceptors with other ventilatory-related inputs such as central hypoxia, lung stretch, and exercise. The limitations of current approaches addressing this question are discussed and future studies are suggested.
1. Introduction
Traditionally, since the time of Heymans and Comroe, a key area of interest in the control of breathing has been the relative influence of peripheral vs. medullary chemoreceptors in the ventilatory response to hypercapnia, hypocapnia, and hypoxia. The peripheral and central chemoreceptors were usually viewed as independent entities responding solely to the [H+], PCO2 and PO2 in their environments, their outputs being summated in a fixed way (Fig. 1). In this review we discuss the implications of newer evidence suggesting that these two sets of receptors are not functionally separate but rather that they are dependent upon one another such that the sensitivity of the medullary chemoreceptors is critically determined by input from the peripheral chemoreceptors and possibly other breathing-related reflex afferents as well i.e., they are interdependent. There is also evidence showing the converse i.e., the degree of central stimulation can affect the sensitivity of the peripheral chemoreceptors.
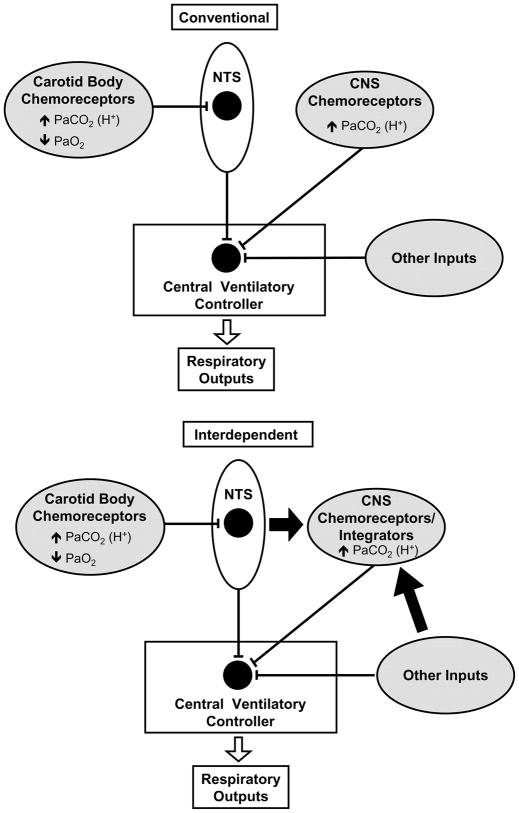
Figure 1.
Schematic representation of the conventional (top) and interdependent (bottom) models of ventilatory control. Proposed new pathways of the interdependent model are indicated by the bold arrows. Note that in the interdependent model carotid body inputs via the NTS have the potential to change the gain of the CNS chemoreceptors/integrators. “Other Inputs”, such as from central hypoxia sensors, may also have this capability. Also note that communication between the CNS chemoreceptors/integrators and the NTS and Other Inputs could be bidirectional offering the potential for even more complex interactions.
2. Models of Central/Peripheral Interdependence
Since the late 1960s the region of the ventrolateral medulla termed “area S” or “intermediate area” has been suspected of having integrative properties at least for chemoafferent and airway rapidly adapting receptor signals involved in the control of breathing (Schlaefke et al. 1967; Cherniack et al. 1979; Millhorn et al. 1984; Millhorn et al. 1986). Within the past decade more direct neuroanatomical and neurophysiological evidence from reduced preparations has given rise to the idea that carotid body chemoreceptor inputs, central chemoreceptor inputs from several distributed sites (Nattie et al. 2006; Dias et al. 2008; Nattie et al. 2009), and possibly many other ventilatory-related reflex inputs impinge on ventilatory control centers in the CNS which provides the potential for the various inputs to interact in complex ways. Perhaps the most comprehensive model put forward to date posits that these inputs converge primarily on a putative chemosensitive/integrating region in the retrotrapezoid nucleus (RTN) characterized by chemosensitive glutamatergic interneurons which strongly express Phox2b (Smith et al. 1989; Mulkey et al. 2004; Guyenet et al. 2005b; Rosin et al. 2006; Stornetta et al. 2006; Takakura et al. 2006; Moreira et al. 2007; Guyenet 2008; Guyenet et al. 2008a; Guyenet et al. 2008b). Phox2b has been shown to mark an “uninterrupted chain of sensors and neurons involved in the integration of peripheral and central chemoreception. This circuit includes the carotid bodies, chemoreceptor afferents, chemoresponsive NTS projections to the VLM, and RTN central chemoreceptors.” (Dauger et al. 2003; Stornetta et al. 2006). Other structures such as the NTS, the lateral parabrachial nucleus (LPBN), the raphé, and the dorso-lateral pontine respiratory group are probably also involved in integrative and chemoreceptive pathways (Mizusawa et al. 1995; Takakura et al. 2006; Wang, 2001 #3534; Nuding et al. 2009; Song et al. 2009) (Fig. 1).
Thus, it seems plausible that the carotid body chemoreceptors and other breathing-related afferents could modulate the sensitivity/integrating properties of the chemosensitive RTN neurons, and possibly other integrative/chemosensitive areas in the CNS as well, which in turn modulate the output of the respiratory controller. This neural link between ventilatory afferents and putative central chemoreceptors/integrators offers an additional theoretical framework that may help explain many aspects of peripheral/central chemoreceptor interactions as well as interactions with non-chemical afferent inputs and provides the basis for much of this review.
3. The Nature of Interdependence Is Controversial
The concept of interaction between central and peripheral chemoreceptors (as opposed to the well-known CO2/hypoxia interaction at the carotid body chemoreceptors) remains controversial. Although known for many years, it has perhaps not been widely appreciated that any interaction other than simple additive interaction (i.e., no change in slope of the ventilatory response) requires some sort of modulation of one set of receptors by the other (e.g., (Adams et al. 1982)). Thus, hypoadditive interaction or hyperadditive interaction would require what we have termed “interdependence”. Even simple additive interaction may employ interdependence although it would not be required.
3.1 Evidence from Reduced Preparations
A number of earlier studies undertaken in reduced preparations have shown either hyperaddition (Loeschcke et al. 1963; Lahiri et al. 1975a; Adams et al. 1978; Heeringa et al. 1979), addition (i.e., no interaction) (Adams et al. 1982; van Beek et al. 1983), or hypoaddition (Gesell et al. 1940; Kiwull et al. 1976; Berger et al. 1978; Geise et al. 1978; Eldridge et al. 1981a; Adams et al. 1982; Day et al. 2007; Day et al. 2009) of central and peripheral hypercapnic or hypoxic stimuli. This lack of consensus is probably due to the diverse experimental approaches used, most of which required anesthesia, decerebration, carotid body denervation, vagotomy, the use of non-blood perfusates or some combination of these. It is well known that anesthetics obtund the ventilatory control system (e.g., (Faber et al. 1982; Fregosi et al. 1987) and may unmask the neural “saturation” phenomenon [e.g., (Eldridge et al. 1981a)] whereby a central neural component with two or more inputs progressively fails to increase its output proportional to the increasing inputs, although this may not apply to all types of afferents (Eldridge et al. 1982). Decerebration has been shown to disrupt normal ventilatory control (Tenney et al. 1977; Nielsen et al. 1986; Hayashi et al. 1991) which can be further complicated by the level at which the decerebration is performed.
The most common interaction observed in reduced preparations appears to be hypoaddition and this idea has received recent support from Day & Wilson (Day et al. 2007; Day et al. 2009). These authors used a sophisticated dual perfusion decerebrate rat preparation to control the environments of the carotid body chemoreceptors and central chemoreceptors independently. They believe that their techniques avoided the problem of neural saturation because their phrenic responses were linear both above and below eupneic values and the linear responses persisted over three different brainstem PCO2/pH conditions. The linearity is a significant observation because a curvilinear decrease in response is thought to be characteristic of progressive saturation (Eldridge et al. 1981a). However, as they themselves point out, there are some significant caveats to their approach including much reduced ventilatory response sensitivities relative to intact anesthetized or unanesthetized preparations. It is of interest that their data is not in agreement with that of Heeringa et al. (Heeringa et al. 1979) or van Beek et al. (van Beek et al. 1983) who used a similar preparation (isolated brain stem perfusion) and observed additive interaction.
3.2 Evidence from Unanesthetized Preparations
The controversy remains even if one examines only data from intact, unanesthetized preparations. Additive interaction has been observed in humans (Clement et al. 1992; Clement et al. 1995; St Croix et al. 1996) and goats (Daristotle et al. 1989); hyperadditive interaction has been observed in one study in humans (Robbins 1988), and hypoaddition has been observed in goats (Smith et al. 1984) and cats (Ou et al. 1976). Again, comparisons are difficult due to the diverse experimental approaches used and all have significant limitations. The human studies relied on temporal differences in the transient responses of central and peripheral chemoreceptors which, while real, are short (Smith et al. 2006) and, therefore, achieving clear and complete separation of the two sets of receptors is difficult. Moreover, if the neural circuitry described above has physiological significance, then temporal separation is not really meaningful inasmuch as CO2 and O2 changes in the environment of the carotid body (CB) chemoreceptors would have immediate effects on central responsiveness. In addition, the presence of short term potentiation of respiratory motor output (Eldridge et al. 1980; Badr et al. 1992; Georgopoulus et al. 1992; Hayashi et al. 1993) seriously confounds the assumption of complete temporal separation of central and peripheral chemoreceptors based solely on when the apparent chemical stimulus is present or absent. The awake goat study (Smith et al. 1984) was limited by possible interactive effects of increased PaCO2 sensed by the CB and the superimposed cyanide stimulus as well as the transient nature of the CB stimulus. The cat study of Ou and Tenney (Ou et al. 1976) is often cited in support of hypoaddition but they made no attempt to separate the stimuli at the central vs. peripheral chemoreceptors and they reported behavioral problems in the cats that may have affected the data. The most physiological study to date appears to be the awake goat study of Daristotle and Bisgard (Daristotle et al. 1989) using an extracorporeal CB perfusion technique.. These authors obtained systemic CO2 ventilatory response curves during CB perfusion with hypercapnic-hypoxic blood and normocapnic-normoxic blood. They concluded that central/peripheral interaction was additive since they found no statistically significant increase in the slope of the ventilatory response to CO2 when the isolated CB chemoreceptors were stimulated. Interestingly, however, there was a trend toward an increased slope. Unfortunately, they did not utilize CB inhibition so their range of CB stimuli was somewhat limited and may have obscured a hyperadditive interaction.
There is some evidence to suggest that chemosensitivity of the RTN and other chemosensitive sites in the CNS may be state dependent (Nattie et al. 2001; Nattie et al. 2002). Thus, interdependence could be modified by sleep vs. wakefulness as the gain of the central chemoreceptors could vary with state of consciousness.
In summary, we believe that newer functional and anatomical evidence provides support for a concept of interdependence between peripheral and central chemoreceptors as well as other afferent inputs relevant to ventilatory control. This interdependence of chemoreceptors may modulate not only the gain of the peripheral and central chemoreceptors to chemical stimuli but also the integrating properties of some central chemosensitive structures such as the RTN. In subsequent sections of this review, we speculate on the implications that such complex interdependence will have on long-standing key problems in ventilatory control such as the relative contributions of peripheral and central chemoreceptors to the ventilatory response to CO2, determinants of eupnea, acclimatization to chronic hypoxia, and sleep apnea.
4. Implications for the “Relative Contribution” Concept
How does the concept of interdependent chemoreceptors fit with the “60:40”contribution generally presumed for central: peripheral chemoreceptor contributions to the whole body CO2 response? The effects of bilateral carotid body denervation in unanesthetized animals have been used to partition the ventilatory response to CO2 to about a 60:40 central/peripheral split (Kiwull et al. 1972; Bisgard et al. 1976; Pan et al. 1998; Rodman et al. 2001). A hyperoxic background to the inspired CO2 – using a steady state or rebreathe technique – is often presumed to remove the carotid chemoreceptor as a significant contributor to the ventilatory response to inhaled CO2 (Read 1967) – but this is unlikely given the following findings: a) Carotid sinus nerve activity in response to CO2 is reduced but still significant in hyperoxia (Lahiri et al. 1975a); and b) Bilateral carotid body denervation in unanesthetized dogs reduces the slope of the ventilatory response to hyperoxic CO2 inhalation by 40–45% (Rodman et al. 2001).
We question whether this partitioning represents the true division of peripheral:central contributions to “whole body” hypercapnia. First, the adaptations in the central and peripheral reflex responsiveness which likely occur over time as a result of bilateral carotid body denervation will mean that the relative sensitivity of the chemoreceptors have been altered from normal (Liu et al. 2003; Hodges et al. 2005). However, even if the carotid body is isolated and its PCO2 is maintained normocapnic via extracorporeal circulation, the central:peripheral partition is still close to 60:40, although marked inter-animal variability does exist in these partitions, varying from 90:10 to 20:80 (Smith et al. 2006). More serious criticism of this presumed partitioning would be justified if peripheral stimulation via arterial hypercapnia significantly influenced the gain of the central chemoreceptor and visa versa. For example, if hyperadditive interaction is indeed present (Lahiri et al. 1975b), then the CO2-induced stimulation of the carotid chemoreceptor would significantly enhance the sensitivity of the central CO2 response, whereas hypoadditive interaction (Day et al. 2007; Day et al. 2009) would mean that an acid brain or carotid chemoreceptor would reduce the CO2 sensitivity of its peripheral or central counterpart. Either way, the interdependence of peripheral and central chemoreceptor function and sensitivities would negate the usefulness of this traditional relative partitioning exercise. Furthermore, even the attempt to quantify the relative changes in carotid chemoreceptor versus central chemoreceptor sensitivities in various disease states or treatments or other experimental conditions by, for example, using hyperoxic CO2 ventilatory response tests or isocapnic hypoxic response tests (see Ventilatory Acclimatization to Hypoxia below) would fail to take into account the potential effect of chemoreceptor interdependence. In short, in the face of true interdependence of chemoreceptor functions, the common practice of partitioning the peripheral:central responsiveness in intact or denervated preparations may provide no useful insights into the cause of the observed change(s) in ventilatory control.
5. Implications for Eupnea
Interdependence of peripheral and central chemoreceptors would suggest an important role for carotid body chemoreceptors even during quiet eupnea and some recent data supports this idea. Blain et al (Blain et al. 2009) used an unanesthetized canine model with reversibly isolated carotid bodies that were perfused from an extracorporeal gas exchanger that allowed control of the blood gas environment of the carotid bodies independently of the systemic (and, importantly, brain) circulation. They inhibited the carotid bodies with hypocapnic and hyperoxic blood which resulted in marked reductions in V̇I and systemic (and therefore brain) CO2 retention that was only partially compensated for up to 25 minutes of CB inhibition. Blain et al. interpreted these data to mean that CB chemoreceptors contributed more than one half of the total eupneic drive to breathe as a result of both the normal tonic sensory input from the CB chemoreceptors to medullary respiratory controllers and a strong modulatory effect on central chemoreceptor responsiveness to CO2. Future work could confirm this idea by assessing the effects of CB inhibition on the ventilatory response to specific central hypercapnia.
6. Implications for sleep-induced periodic breathing and apnea
With the loss of wakefulness at the onset of non-REM sleep the control of breathing becomes exquisitely dependent upon chemoreceptor influences especially on PCO2. Apneas due to loss of central respiratory motor output commonly occur following a brief ventilatory overshoot and transient hypocapnia. Controlled studies using progressive levels of mechanical ventilation to mimic the transient ventilatory overshoot have demonstrated that the apneic threshold for PaCO2 in healthy sleeping subjects resides within 3 – 5 mmHg below the eupneic PaCO2 (Skatrud et al. 1983; Meza et al. 1998). Further, if very small amounts of inspired CO2 were administered to sleeping subjects undergoing periodic breathing (e.g. in hypoxia or heart failure), breathing pattern was immediately stabilized (Berssenbrugge et al. 1983; Khayat et al. 2003). The sensitivity of this hypocapnic induced apneic threshold is not fixed; rather it can be altered via changing gains – either respiratory controller gain (ΔV̇E/ΔPaCO2) or plant gain (ΔPaCO2/ΔV̇ E) (Khoo et al. 1982; Xie et al. 2001; Nakayama et al. 2002; Dempsey 2004). Thus, the regulation of PaCO2 and control system sensitivity to PaCO2 during sleep plays a dominant role in determining central apnea and periodic breathing and – in some cases – may contribute significantly to cyclical obstructive apneas (Hudgel et al. 1987; Warner et al. 1987; Badr et al. 1995; Hudgel et al. 1998; Wellman et al. 2004).
We have attempted to determine which chemoreceptors – peripheral or central – are primarily responsible for sensing the transient hypocapnia and triggering apneas. Given that apneas occur within ~20 seconds of PaCO2 reaching the apneic threshold (following a transient ventilatory overshoot), the carotid chemoreceptors are highly suspect as primary sensors (Smith et al. 2006). Accordingly, the effects of bilateral carotid body denervation on the hypocapnia-induced apneic threshold were studied, using step-wise increases in assist-control mechanical ventilation in sleeping dogs (Smith et al. 2007). When intact they consistently became apneic and breathed periodically when PaCO2 was reduced 3–5 mmHg less than eupnea. However following carotid body denervation: a) eupneic V̇E was reduced sufficiently to increase PaCO2 8–10 mmHg above intact control; b) substantial transient reductions in PaCO2 even to more than 10 mmHg < eupnea failed to elicit apneas or periodic breathing; and c) sustained reductions of > 8–10 mmHg PaCO2 for a minute or more would occasionally elicit a prolongation of TE but these apneas occurred well beyond the 30–40 seconds required for the central chemoreceptor response (Smith et al. 2006) and were never accompanied by periodic breathing. A similar finding of essentially no hypocapnic apneic threshold for the central chemoreceptors alone was obtained in sleeping dogs whose CB chemoreceptors were isolated and maintained normocapnic and normoxic via extracorporeal perfusion while a ventilator was used to produce progressive systemic hypocapnia (Fig. 2) (Smith et al. 2007).
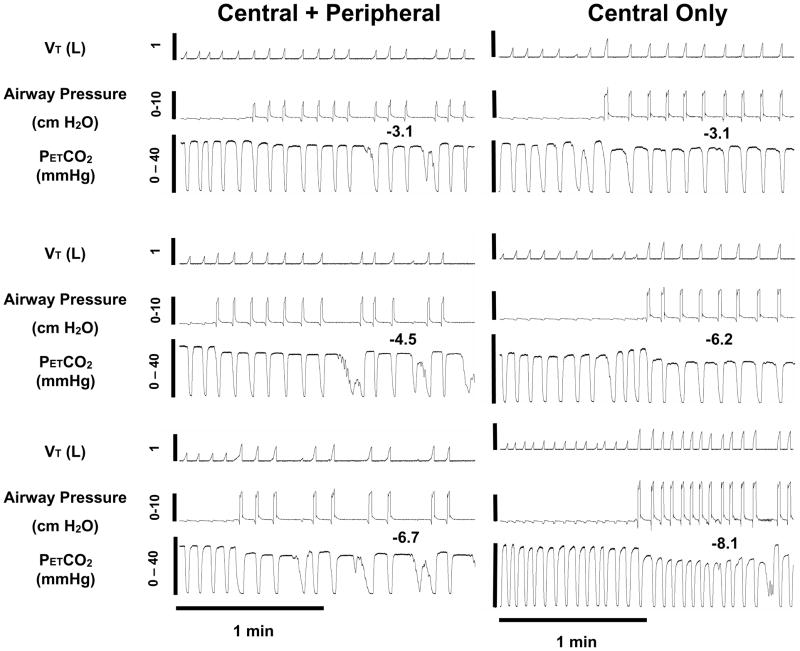
Figure 2.
Representative polygraph records showing the effect of progressive hypocapnia induced by pressure support ventilation (PSV; see Methods for details) during NREM sleep in a dog. The onset of PSV is indicated by the positive shift in airway pressure and increase in VT. Numbers in each panel = the degree of hypocapnia relative to eupnea in Torr. When both sets of chemoreceptors sensed the hypocapnia (Central+peripheral) clear periodicity was present with as little as a 3.1 Torr decrease in PETCO2 and persisted as hypocapnia became more severe. In contrast, when only central chemoreceptors sensed the hypocapnia (carotid bodies isolated and perfused with normal blood gases and pH), apneas were rare, short, delayed, and periodicity never occurred. We were unable to detect a true apneic threshold despite marked CNS hypocapnia indicating that peripheral hypocapnia is obligatory for the genesis of central apnea in sleep. (Data from (Smith et al. 2007) permission pending).
So, clearly the carotid chemoreceptors appear to be required sensors of both transient hypocapnia (to initiate apnea) as well as transient hypercapnia plus mild hypoxemia at the end of an apnea (to trigger the transient ventilatory overshoot and subsequent unstable alternating periods of ventilatory over- and undershoots). However, it is puzzling that even substantial levels of ventilator-induced CNS hypocapnia, per se, in the carotid body denervated animal did not cause apnea or periodicity – a finding that was consistent with previous work in anesthetized cats (Berkenbosch et al. 1984).
If the carotid body is solely responsible for mediating the hypocapnic induced apnea then we reasoned that hypocapnia applied solely to the isolated carotid body should cause apnea. However, this was not the case as reductions in carotid body-specific PCO2 as large as 15 mmHg caused significant reductions in VT but no appreciable change in breath timing in the sleeping dog (Smith et al. 1995). Similarly carotid body specific hyperoxia (Smith et al. 1995) as well as combinations of carotid body specific hyperoxia and extreme hypocapnia (Blain et al. 2009) caused reductions in VT – as much as 60% in the latter condition – but apnea or instability did not occur. In the awake goat, carotid body specific hypocapnia did occasionally cause some significant apneic periods but this was only after several minutes of hypocapnia, which questions whether this effect on breath timing was due exclusively to carotid chemoreceptor inhibition, per se (Daristotle et al. 1987). These relatively modest effects on breath timing of hypocapnia isolated to the carotid chemoreceptor contrast sharply with the apnea-inducing effects of combined systemic and central reductions in PaCO2 during a transient ventilatory overshoot.
Finally, there is also recent indirect evidence in normal sleeping humans supporting a significant role for central chemoreceptor hypocapnia in causing apnea. Indomethacin administration was shown to reduce the normally highly sensitive cerebral vascular response to CO2 (Xie et al. 2006). Accordingly, during a ventilatory overshoot with reduced PaCO2 the normal protective effects of a reduced cerebral blood flow in minimizing the reduction in brain PCO2 was compromised. The resultant greater than normal brain hypocapnia, by itself, was shown to sensitize the apneic threshold (i.e. narrowed the “CO2 reserve” between eupneic and apneic threshold PaCO2) and increased the propensity for ventilatory instability (Xie et al. 2009).
In total we interpret these findings to mean that while hypocapnia at the level of the carotid bodies is critical to causing apneas and periodic breathing following a transient ventilatory overshoot, additional inhibitory inputs are also required. Several potential interactive influences may be present in the intact animal during a transient ventilatory overshoot, all of which may interact with the inhibitory effects of carotid body hypocapnia. Three candidates are: a) the inhibitory feedback from lung inflation which has been shown to interact centrally with carotid chemoreceptor chemical stimuli (Mitchell et al. 1987; Bajic et al. 1994); b) the influence of CB afferent input on central CO2 responsiveness at the level of the central chemoreceptors (Guyenet 2008; Blain et al. 2009); and c) conversely, the influence of brain ECF/central chemoreceptor hypocapnia on the CO2 responsiveness of the carotid chemoreceptors. At the very least, it appears as though BOTH peripheral and central chemosensitive structures must “sense” the hypocapnia in order to cause apnea following a transient ventilatory overshoot. In turn, we postulate that the resultant cessation of respiratory rhythm would occur because of the synergystic inhibition due to chemoreceptor interdependence effects (possibly including synergistic contributions from pulmonary stretch receptors) rather than to the summed effects of peripheral and central chemoreceptor inhibition alone on rhythm generating neurons.
7. Implications for Ventilatory Responses to Acute and Chronic Hypoxia
Acute Hypoxia
Based largely on studies in anesthetized preparations that had been carotid body denervated, made hypoxic with carbon monoxide (Neubauer et al. 1985; Melton et al. 1988; Melton et al. 1992), or exposed to specific pontomedullary hypoxia (van Beek et al. 1984), it was long thought that the direct effect of hypoxia on the CNS was ventilatory depression. However, when unanesthetized carotid body denervated animals were made hypoxic most studies reported that ventilation was unchanged or increased slightly (Davenport et al. 1947; Bouverot et al. 1973; Krasney et al. 1973; Bouverot et al. 1975; Forster et al. 1976; Bowes et al. 1981; Lahiri et al. 1981; Gautier et al. 1982; Smith et al. 1986; Olson et al. 1988; Long et al. 1993). Even more noteworthy are studies in CB intact, unanesthetized animals which have shown that when carotid body afferents are isolated but intact and held normal via extracorporeal perfusion, specific CNS hypoxia has a stimulatory effect on ventilation during wakefulness (Smith et al. 1993; Engwall et al. 1994) or sleep (Curran et al. 2000) (Figure 3). Taken together, these data provide strong support for a mandatory role for carotid body afferents in the normal sensing of hypoxia by structures in the CNS and therefore a normal ventilatory response to acute and chronic hypoxia (also see Acclimatization below).
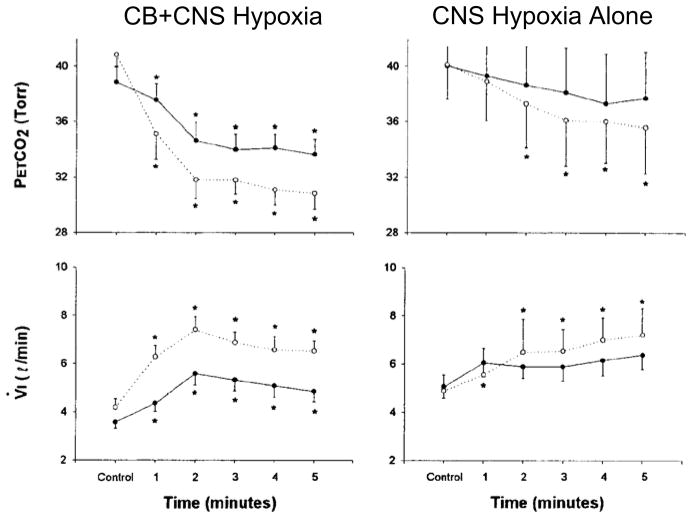
Figure 3.
Minute-by-minute means of PETCO2 & V̇I response to PaO2=52 mmHg (filled) and 38 mmHg (open) for CB+CNS (whole body) hypoxia and CNS (CBs isolated & held normal via perfusion) hypoxia alone. Note the graded ventilatory response to CNS hypoxia (proportional to stimulus) that is approximately 1/3 that of the response when both CBs and CNS can sense hypoxia. In CB+CNS hypoxia the hyperventilation was due to increases in both VT and fb; in CNS hypoxia alone the hyperventilation was due almost entirely to increased fb. VT/TI increased in both conditions. Also note similar time courses of response. In both conditions onset of response was rapid (~20 sec.). (Data from (Curran et al. 2000) permission pending).
The concept of interdependence might help explain these observations. The observation that intact but isolated and unstimulated CB chemoreceptors unmask a clear centrally-mediated ventilatory response to hypoxia suggests that there are hypoxia-sensitive neurons in the CNS that act like chemosensors for hypoxia. There are a number of studies supporting this idea of a CNS hypoxia-sensing system that participates in ventilatory and vasomotor control (for e.g., (Nolan et al. 1996; Teppema et al. 1997; Guyenet 2000; Solomon 2000; Neubauer et al. 2004)). If the putative chemosensitive integrating centers process afferent output from central hypoxia chemosensors, as they are thought to do for CB afferents (e.g., (Guyenet 2008)) then it is possible that the central hypoxic chemosensors could modulate the gain of the central chemoreceptors in a similar way. It is also possible that CB afferents could sensitize the central hypoxia sensors. If these conjectures are true, then upon normal physiological exposure to hypoxia there is a potential for a complex interaction between central chemoreceptors/integrators, CB chemoreceptors, and central hypoxia sensors that could contribute to the observed enhanced ventilatory responsiveness. To our knowledge this idea has not been tested in intact, unanesthetized animals.
7.1 Acclimatization
It is well known that ventilatory chemosensitivity to hypercapnia and hypoxia increases as acclimatization develops over hours to days of exposure to hypoxia (Rahn et al. 1953; Chiodi 1957; Forster et al. 1971; Fatemian et al. 1998; Dempsey et al. 2009 In Press). Several lines of evidence support a key role for carotid body chemoreceptors in acclimatization: 1) Hypoxia at the carotid body alone is sufficient to produce acclimatization in the unanesthetized goat (Fig. 4) (Busch et al. 1985; Bisgard et al. 1986b). 2) The time course of ventilatory acclimatization correlates well with increased activity in the carotid sinus nerve in the goat (Nielsen et al. 1988) or with increased hypoxic sensitivity of the carotid body in cats (Vizek et al. 1987) and rats (Chen et al. 2002). 3) Intact carotid bodies are required for acclimatization to occur (Bouverot et al. 1975; Forster et al. 1976; Forster et al. 1981; Fordyce et al. 1984; Smith et al. 1986; Olson et al. 1988). 4) There is an enhanced central responsiveness, as measured by increases in phrenic nerve activity, to a given electrical stimulation of the carotid sinus nerve in acclimatized rats (Dwinell et al. 1999). 5) Consistent with the latter findings are more recent observations from brain slices of acclimatized rats showing enhanced evoked excitatory inputs to NTS neurons receiving arterial chemoreceptor inputs (Zhang et al. 2009). 6) Brain hypoxia per se, despite its ability to stimulate breathing, does not produce acclimatization (Weizhen et al. 1992). Taken together, these data clearly suggest that carotid body chemoreceptor afferent information sensitizes the central processing of afferent information.
Figure 4.
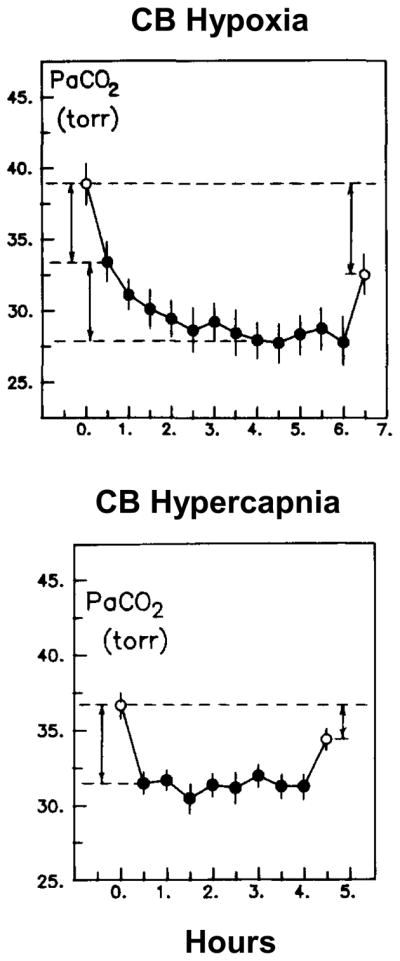
Time course of the ventilatory response to specific CB hypoxia vs. specific CB hypercapnia in the unanesthetized, CB perfused goat. Note the progressive increase in ventilation when CB hypoxia was imposed; when CB hypercapnia was imposed there was no further increase in ventilation over time after the initial increase. Also note that upon return to normoxic and normocapnic conditions CB hypoxia resulted in a persistent hyperventilation whereas CB hypercapnia did not. (Data from (Bisgard et al. 1986a) permission pending).
The foregoing arguments might also help explain the enhanced ventilatory response to exercise that has been observed upon exposure to hypoxia (Dempsey et al. 1972; Weil et al. 1972; Dempsey et al. 1977). If exercise-related inputs from the hypothalamus (“central command”) impinge on the RTN chemosensor/integrator as suggested by Guyenet then this could account for an upregulated response to a given hypothalamic input.
We have already discussed the ventilatory stimulation caused by CNS-specific acute hypoxia and the likely existence of hypoxia sensors in the CNS (see Acute Hypoxia above). To our knowledge it is not known if these CNS hypoxia sensors impinge on the chemosensitive neurons in the RTN, or if they modulate the sensitivity of RTN CO2/H+ chemosensitive neurons. Some very recent evidence suggests a role for oxygen sensing in the RTN itself mediated by Task2 channels (Gestreau et al. 2010) The answers to these questions might prove crucial in understanding the mechanisms of ventilatory acclimatization to hypoxia.
We propose that these studies, taken together, are consistent with the concept of peripheral-central interdependence in that the increased central responsiveness is likely to be a consequence of the time-dependent increase in carotid body chemoreceptor afferent output. This idea is consistent with modulation of central chemosensitive/integrative neurons and possibly central hypoxia chemosensors by increased afferent activity in the carotid sinus nerve but it remains to be determined if the RTN is the final common pathway for this sensitization/integration or if other loci such as the NTS, raphe, and pons also play a role.
7.2 Deacclimatization
After acclimatization to hypoxia has occurred, breathing does not immediately return to normal when normoxic conditions are restored. Rather, there is continued but waning time-dependent hyperventilation which can persist for 24–36 hours in humans (Dempsey et al. 1979). Upregulation of the ventilatory chemosensitivity (isocapnic hypoxia) in humans has been reported to persist for up to two weeks after normoxia was restored (Forster et al. 1971) despite normal eupneic ventilation. There is some disagreement as to whether deacclimatization utilizes the same suite of mechanisms that cause acclimatization (see review by Dempsey (Dempsey et al. 2009 In Press )). However, we propose that sensitized CB chemoreceptors (Barnard et al. 1987; Vizek et al. 1987; Nielsen et al. 1988; Chen et al. 1995; He et al. 2006; He et al. 2007) and interdependence of central and peripheral chemoreceptors are key features of the deacclimatization process.
8. Role of Chemoreceptors and Chemoreceptor Interactions in Exercise Hyperpnea
The ventilatory response to exercise is proportional to the increasing metabolic requirement in mild to moderate exercise and rises out of proportion to metabolism in heavy exercise. The dominant mechanism for this ventilatory response remains unknown, but two locomotor-related mechanisms are clearly involved, namely: a) the feedforward link of a descending drive to the spinal motor neurons driving locomotion that originate in locomotor regions of the hypothalamus and cerebellum and which occur in parallel with the engagement of cardiorespiratory medullary neurons thereby producing the immediate ventilatory response to exercise (Eldridge et al. 1981b; Eldridge et al. 1985), and b) the feedback link from type III–IV activation from contracting limb muscles to the nucleus of the solitary tract in the medulla via spinal afferents (Kalia et al. 1981).
It is reasonable to expect that the peripheral and/or medullary chemoreceptors may also be involved in exercise hyperpnea to some extent given the close matching of increased alveolar ventilation to CO2 production and the resultant regulation of arterial blood gases to within a few mmHg of resting values. However, several lines of evidence speak against a role for arterial chemoreceptors: a) Changes in potential circulating chemoreceptor stimuli such as arterial [H+], PaCO2 or PaO2 or even circulating K+ or norepinephrine or osmolarity or angiotensin II are not altered sufficiently or even in some cases in the appropriate direction to explain hyperpnea up to moderate exercise intensities. b) Bilateral denervation of the carotid chemoreceptors results in substantial hypoventilation and CO2 retention at rest and in equal amounts during steady-state exercise – although the magnitude of the ventilatory response to moderate exercise is unaffected by carotid body denervation (Mitchell et al. 1984; Pan et al. 1995). Similarly, substantial and specific alkalization of the medullary chemoreceptors achieved via cisternal perfusion in awake animals caused substantial hypoventilation at rest and exercise without altering the magnitude of the exercise-induced increase in ventilation (Smith et al. 1988). These data do not implicate peripheral or central chemoreception as significant contributors to the steady state ventilatory response to exercise. It is only during heavy intensity exercise that the potential contributions of the carotid chemoreceptors in response to increasing levels of circulating metabolites may play a significant role in the hyperventilatory response and even here the findings are controversial (Pan et al. 1995).
On the other hand, recent evidence suggests that there are functional neural connections between hypothalamic sites of origin for central command of locomotion and the central chemoreceptors located in the retrotrapazoid nucleus RTN (Fortuna et al. 2009) (see Introduction). When these hypothalamic sites were electrically or pharmacologically stimulated in anesthetized rats, CO2 sensitive neurons in the RTN increased their discharge – providing evidence for at least a functional neural pathway from a demonstrated major source of exercise hyperpnea to central chemoreceptors. However, it was also shown that the increased central command produced only an additive effect on the responsiveness of these RTN neurons and phrenic nerve activity to increases in central CO2. Accordingly, at least in this anesthetized preparation it appears that the central command stimulus would only increment the onging activity of chemosensitive neurons in the RTN.
It remains to be determined in the unanesthetized animal if synergistic – rather than purely additive – responses at the level of the RTN chemosensitive neurons may be present in response to central locomotor command. Furthermore, actual exercise itself incorporating both peripheral feedback from locomotor muscles as well as central feedforward stimuli might elicit a synergistic responsiveness at the level of the central chemoreceptors. With the established interconnections between locomotor-driven drives to breathe and central chemoreceptors, these possibilities for chemoreceptor contributions to exercise hyperpnea clearly exist. The established neural connections between carotid and RTN chemoreceptors (Takakura et al. 2006) – especially if shown to have synergistic effects on breathing in the awake, intact animal – may also contribute to the hyperpnea of “real” exercise, even if the tonic carotid chemoreceptor input is unchanged from rest to exercise. On the other hand, given the unchanged slope of the whole body ventilatory response to inspired CO2 during mild and moderate exercise in healthy humans (Asmussen et al. 1957) we would not hypothesize that this link to central chemoreceptors would be an important contribution to the hyperpneic stimulus. A more definitive testing of this hypothesis is required, using an intact, exercising animal preparation with clear separation of peripheral and central chemoreceptors in order to determine how precisely input from truly physiologic locomotor-linked stimuli, coupled with varying levels of sensory input from peripheral chemoreceptors, might impact on central CO2 sensitivity and ultimately on the hyperpneic drive to breathe.
9. Does the type of carotid body stimulus affect central/peripheral interaction?
Does the type of carotid body stimulus determine the observed interaction between carotid body chemoreceptors and central chemoreceptors? In other words, is the central/peripheral interaction always the same for a given amount of carotid sinus nerve afferent traffic or does the nature of the carotid body stimulus affect the outcome? There is a paucity of data on this topic; nevertheless there are hints that the nature of the carotid body stimulus may affect central responses and therefore determine the nature of central/peripheral interaction.
One experimental approach has been to try to detect different discharge patterns in single fibers of the carotid sinus nerve in response to normocapnic hypoxia or normoxic hypercapnia in anesthetized goats (Niu et al. 1990). The authors concluded that the chemoreceptors did not code information about arterial PO2 and PCO2 in the discharge pattern. However, these findings do not rule out differential information content in the afferent carotid sinus nerve traffic. For example, more than one type of glomus cell has been recognized morphologically (Kobayashi 1968; Morita et al. 1969; Hellstrom 1975; McDonald et al. 1975). If these cells transduce CO2 and O2 differentially, and their output is interpreted differentially in the respiratory control centers, then it is conceivable that such a response might affect central/peripheral interaction.
It is of interest that a differential transduction mechanism of this type might help explain the observation that specific carotid body hypoxia caused time-dependent ventilatory acclimatization to hypoxia but specific carotid body hypercapnia did not (Bisgard et al. 1986a). In this study Bisgard and colleagues used the isolated and perfused carotid body model in unanesthetized goats to expose the carotid bodies specifically to increased PCO2 for 4 hours. The goats hyperventilated acutely (PaCO2 decreased 5.2 mmHg) in response to the CB hypercapnia and maintained that level of hyperventilation for the 4-hour duration of the experiment but no time dependent increase in ventilation was observed. In contrast, when goats were exposed to specific CB hypoxia sufficient to match the acute hyperventilation caused by CB hypercapnia (PaCO2 decreased 5.5 mmHg) an additional, time-dependent hyperventilation (PaCO2 decreased an additional 5.6 mmHg) ensued over the 4 hour duration of the experiment.
Additional evidence consistent with differential transduction of hypoxia vs. hypercapnia has been provided by Mizusawa et al. (Mizusawa et al. 1994). They used microdialysis of the NTS in the unanesthetized, freely moving rat and to show that, at equal ventilations, hypoxia stimulated glutamate production but hypercapnia did not. Carotid body denervation eliminated the ventilatory and glutamate response to hypoxia but did not affect the ventilatory response to CO2.
More recently, Takakura et al. (Takakura et al. 2006) have shown that CO2/H+ sensitive neurons in the RTN increased their activity in response to stimulation of the carotid body chemoreceptors with hypoxia or cyanide and this increase was abolished when the carotid bodies were denervated. In contrast, increased activity of the CO2/H+ sensitive neurons in the RTN to systemic increases in PCO2 was unchanged by carotid body denervation. Again, these data are consistent with a differential effect of CO2 vs. hypoxia at the carotid body chemoreceptor but a major limitation is whether the CO2 stimulus at the carotid body in this reduced preparation was sufficient to increase activity in the carotid sinus nerve to a level comparable with the hypoxic or cyanide stimuli.
10. Future Directions
The neuroanatomical and neurophysiological evidence in favor of an integrative function for the RTN, probably in conjunction with other neural loci (e.g., NTS, pons (Kubin et al. 2006; Kline 2008)) seems compelling but we believe that the physiological significance of these pathways has not yet been established. Most of the experiments on this topic were performed in highly reduced, anesthetized preparations. In addition to the anesthesia, procedures such as decerebration, carotid body denervation, vagotomy, cooling of medullary structures, or use of tissues in vitro impose major limitations on the applicability of these results to the intact, unanesthetized animal.
The model of ventilatory control resulting from these data predicts interdependence between these inputs; however, the nature of the interdependence remains unresolved. We suggest that experiments which employ specific carotid body chemoreceptor stimulation or inhibition while independently testing responsiveness of the central control mechanisms to chemical or non- chemical stimuli are required. Ideally, these would be performed in an intact and unanesthetized preparation with all neural feedback systems intact.
Acknowledgments
Work cited from our laboratories was supported by grants from the National Heart Lung and Blood Institute of the National Institutes of Health (Smith, Dempsey, Blain: HL 50531, HL 15469. Forster: HL 25739); the American Heart Association (Smith, Dempsey, Blain) and the Department of Veterans Affairs (Forster).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
LITERATURE CITED
- Adams JM, Attinger FM, Attinger EO. Medullary and carotid chemoreceptor interaction for mild stimuli. Pflugers Arch. 1978;374:39–45. doi: 10.1007/BF00585695. [DOI] [PubMed] [Google Scholar]
- Adams JM, Severns ML. Interaction of chemoreceptor effects and its dependence on the intensity of stimuli. J Appl Physiol. 1982;52:602–606. doi: 10.1152/jappl.1982.52.3.602. [DOI] [PubMed] [Google Scholar]
- Asmussen E, Nielsen M. Ventilatory responses to CO2 during work at normal and at low oxygen tensions. Acta Physiol Scand. 1957;39:27–35. doi: 10.1111/j.1748-1716.1957.tb01406.x. [DOI] [PubMed] [Google Scholar]
- Badr MS, Skatrud JB, Dempsey JA. Determinants of poststimulus potentiation in humans during NREM sleep. J Appl Physiol. 1992;73:1958–1971. doi: 10.1152/jappl.1992.73.5.1958. [DOI] [PubMed] [Google Scholar]
- Badr MS, Toiber F, Skatrud JB, Dempsey J. Pharyngeal narrowing/occlusion during central sleep apnea. J Appl Physiol. 1995;78:1806–1815. doi: 10.1152/jappl.1995.78.5.1806. [DOI] [PubMed] [Google Scholar]
- Bajic J, Zuperku EJ, Tonkovic-Capin M, Hopp FA. Interaction between chemoreceptor and stretch receptor inputs at medullary respiratory neurons. Am J Physiol. 1994;266:R1951–R1961. doi: 10.1152/ajpregu.1994.266.6.R1951. [DOI] [PubMed] [Google Scholar]
- Barnard P, Andronikou S, Pokorski M, Smatresk N, Mokashi A, Lahiri S. Time-dependent effect of hypoxia on carotid body chemosensory function. J Appl Physiol. 1987;63:685–691. doi: 10.1152/jappl.1987.63.2.685. [DOI] [PubMed] [Google Scholar]
- Berger W, Berger K, Berndt J, Giese K. Interaction of peripheral and central respiratory drives in cats. I. Effects of sodium cyanide as a peripheral chemoreceptor stimulus at different levels of CSF pH. Pflugers Arch. 1978;374:205–210. doi: 10.1007/BF00585596. [DOI] [PubMed] [Google Scholar]
- Berkenbosch A, van Beek JH, Olievier CN, De Goede J, Quanjer PH. Central respiratory CO2 sensitivity at extreme hypocapnia. Respir Physiol. 1984;55:95–102. doi: 10.1016/0034-5687(84)90119-1. [DOI] [PubMed] [Google Scholar]
- Berssenbrugge A, Dempsey J, Iber C, Skatrud J, Wilson P. Mechanisms of hypoxia-induced periodic breathing during sleep in humans. J Physiol (London) 1983;343:507–526. doi: 10.1113/jphysiol.1983.sp014906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisgard GE, Busch MA, Daristotle L, Berssenbrugge AD, Forster HV. Carotid body hypercapnia does not elicit ventilatory acclimatization in goats. Respir Physiol. 1986a;65:113–125. doi: 10.1016/0034-5687(86)90010-1. [DOI] [PubMed] [Google Scholar]
- Bisgard GE, Busch MA, Forster HV. Ventilatory acclimatization to hypoxia is not dependent on cerebral hypocapnic alkalosis. J Appl Physiol. 1986b;60:1011–1015. doi: 10.1152/jappl.1986.60.3.1011. [DOI] [PubMed] [Google Scholar]
- Bisgard GE, Forster HV, Orr JA, Buss DD, Rawlings CA, Rasmussen B. Hypoventilation in ponies after carotid body denervation. J Appl Physiol. 1976;40:184–190. doi: 10.1152/jappl.1976.40.2.184. [DOI] [PubMed] [Google Scholar]
- Blain GM, Smith CA, Henderson KS, Dempsey JA. Contribution of the Carotid Body Chemoreceptors to Eupneic Ventilation in the Intact, Unanesthetized Dog. J Appl Physiol. 2009;106:1564–1573. doi: 10.1152/japplphysiol.91590.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouverot P, Bureau M. Ventilatory acclimatization and csf acid-base balance in carotid chemodenervated dogs at 3550 m. Pflugers Arch. 1975;361:17–23. doi: 10.1007/BF00587335. [DOI] [PubMed] [Google Scholar]
- Bouverot P, Candas V, Libert JP. Role of the arterial chemoreceptors in ventilatory adaptation to hypoxia of awake dogs and rabbits. Respir Physiol. 1973;17:209–219. doi: 10.1016/0034-5687(73)90062-5. [DOI] [PubMed] [Google Scholar]
- Bowes G, Townsend ER, Kozar LF, Bromley SM, Phillipson EA. Effect of carotid body denervation on arousal response to hypoxia in sleeping dogs. J Appl Physiol. 1981;51:40–45. doi: 10.1152/jappl.1981.51.1.40. [DOI] [PubMed] [Google Scholar]
- Busch MA, Bisgard GE, Forster HV. Ventilatory acclimatization to hypoxia is not dependent on arterial hypoxemia. J Appl Physiol. 1985;58:1874–1880. doi: 10.1152/jappl.1985.58.6.1874. [DOI] [PubMed] [Google Scholar]
- Chen J, Dinger B, Fidone SJ. Second messenger regulation of tyrosine hydroxylase gene expression in rat carotid body. Biol Signals. 1995;4:277–285. doi: 10.1159/000109453. [DOI] [PubMed] [Google Scholar]
- Chen J, He L, Dinger B, Stensaas L, Fidone S. Role of endothelin and endothelin Atype receptor in adaptation of the carotid body to chronic hypoxia. Am J Physiol Lung Cell Mol Physiol. 2002;282:L1314–1323. doi: 10.1152/ajplung.00454.2001. [DOI] [PubMed] [Google Scholar]
- Cherniack NS, von Euler C, Homma I, Kao FF. Experimentally induced Cheyne-Stokes breathing. Respir Physiol. 1979;37:185–200. doi: 10.1016/0034-5687(79)90070-7. [DOI] [PubMed] [Google Scholar]
- Chiodi H. Respiratory Adaptations to Chronic High Altitude Hypoxia. J Appl Physiol. 1957;10:81–87. doi: 10.1152/jappl.1957.10.1.81. [DOI] [PubMed] [Google Scholar]
- Clement ID, Bascom DA, Robbins PA. An assessment of central-peripheral ventilatory chemoreflex interaction in humans. Respir Physiol. 1992;88:87–100. doi: 10.1016/0034-5687(92)90031-q. [DOI] [PubMed] [Google Scholar]
- Clement ID, Pandit JJ, Bascom DA, Dorrington KL, O’Connor DF, Robbins PA. An assessment of central-peripheral ventilatory chemoreflex interaction using acid and bicarbonate infusions in humans. J Physiol (London) 1995;485:561–570. doi: 10.1113/jphysiol.1995.sp020752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curran AK, Rodman JR, Eastwood PR, Henderson KS, Dempsey JA, Smith CA. Ventilatory responses to specific CNS hypoxia in sleeping dogs. J Appl Physiol. 2000;88:1840–1852. doi: 10.1152/jappl.2000.88.5.1840. [DOI] [PubMed] [Google Scholar]
- Daristotle L, Berssenbrugge AD, Bisgard GE. Hypoxic-hypercapnic ventilatory interaction at the carotid body of awake goats. Respir Physiol. 1987;70:63–72. doi: 10.1016/s0034-5687(87)80032-4. [DOI] [PubMed] [Google Scholar]
- Daristotle L, Bisgard GE. Central-peripheral chemoreceptor ventilatory interaction in awake goats. Respir Physiol. 1989;76:383–391. doi: 10.1016/0034-5687(89)90078-9. [DOI] [PubMed] [Google Scholar]
- Dauger S, Pattyn A, Lofaso F, Gaultier C, Goridis C, Gallego J, Brunet JF. Phox2b controls the development of peripheral chemoreceptors and afferent visceral pathways. Development. 2003;130:6635–6642. doi: 10.1242/dev.00866. [DOI] [PubMed] [Google Scholar]
- Davenport HW, Brewer G, Chambers AH, Goldschmidt S. The respiratory responses to anoxemia of unanesthetized dogs with chronically denervated aortic and carotid chemoreceptors and their causes. Am J Physiol. 1947;148:406–416. doi: 10.1152/ajplegacy.1947.148.2.406. [DOI] [PubMed] [Google Scholar]
- Day TA, Wilson RJ. Brainstem PCO2 modulates phrenic responses to specific carotid body hypoxia in an in situ dual perfused rat preparation. J Physiol. 2007;578:843–857. doi: 10.1113/jphysiol.2006.119594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Day TA, Wilson RJA. A negative interaction between brainstem and peripheral respiratory chemoreceptors modulates peripheral chemoreflex magnitude. J Physiol. 2009;587:883–896. doi: 10.1113/jphysiol.2008.160689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempsey JA. Crossing the apneic threshold: causes and consequences. Exp Physiol. 2004;90:13–24. doi: 10.1113/expphysiol.2004.028985. [DOI] [PubMed] [Google Scholar]
- Dempsey JA, Forster HV, Birnbaum ML, Reddan WG, Thoden J, Grover RF, Rankin J. Control of exercise hyperpnea under varying durations of exposure to moderate hypoxia. Respir Physiol. 1972;16:213–231. doi: 10.1016/0034-5687(72)90052-7. [DOI] [PubMed] [Google Scholar]
- Dempsey JA, Forster HV, Bisgard GE, Chosy LW, Hanson PG, Kiorpes AL, Pelligrino DA. Role of cerebrospinal fluid [H+] in ventilatory deacclimatization from chronic hypoxia. J Clin Invest. 1979;64:199–205. doi: 10.1172/JCI109440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempsey JA, Gledhill N, Reddan WG, Forster HV, Hanson PG, Claremont AD. Pulmonary adaptation to exercise: effects of exercise type and duration, chronic hypoxia and physical training. Ann N Y Acad Sci. 1977;301:243–261. doi: 10.1111/j.1749-6632.1977.tb38203.x. [DOI] [PubMed] [Google Scholar]
- Dempsey JA, Powell F, Bisgard G, Blain G, Smith CA. Role of Chemoreception in Cardio-Respiratory Acclimatization to and Deacclimatization from Hypoxia. Advances in Experimental Medicine and Biology. 2009 doi: 10.1152/japplphysiol.01126.2013. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dias MB, Li A, Nattie E. Focal CO2 dialysis in raphe obscurus does not stimulate ventilation but enhances the response to focal CO2 dialysis in the retrotrapezoid nucleus. J Appl Physiol. 2008;105:83–90. doi: 10.1152/japplphysiol.00120.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dwinell MR, Powell FL. Chronic hypoxia enhances the phrenic nerve response to arterial chemoreceptor stimulation in anesthetized rats. J Appl Physiol. 1999;87:817–823. doi: 10.1152/jappl.1999.87.2.817. [DOI] [PubMed] [Google Scholar]
- Eldridge FL, et al. Input-output relationships of the central respiratory controller during peripheral muscle stimulation in cats. J Physiol (Lond) 1982;324:285–295. doi: 10.1113/jphysiol.1982.sp014113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eldridge FL, Gill-Kumar P. Central neural respiratory drive and afterdischarge. Respir Physiol. 1980;40:49–63. doi: 10.1016/0034-5687(80)90004-3. [DOI] [PubMed] [Google Scholar]
- Eldridge FL, Gill-Kumar P, Millhorn DE. Input-output relationships of central neural circuits involved in respiration in cats. J Physiol. 1981a;311:81–95. doi: 10.1113/jphysiol.1981.sp013574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eldridge FL, Millhorn DE, Kiley JP, Waldrop TG. Stimulation by central command of locomotion, respiration and circulation during exercise. Respir Physiol. 1985;59:313–337. doi: 10.1016/0034-5687(85)90136-7. [DOI] [PubMed] [Google Scholar]
- Eldridge FL, Millhorn DE, Waldrop TG. Exercise hyperpnea and locomotion: parallel activation from the hypothalamus. Science. 1981b;211:844–846. doi: 10.1126/science.7466362. [DOI] [PubMed] [Google Scholar]
- Engwall MJ, Smith CA, Dempsey JA, Bisgard GE. Ventilatory afterdischarge and central respiratory drive interactions in the awake goat. J Appl Physiol. 1994;76:416–423. doi: 10.1152/jappl.1994.76.1.416. [DOI] [PubMed] [Google Scholar]
- Faber JE, Harris PD, Wiegman DL. Anesthetic depression of microcirculation, central hemodynamics, and respiration in decerebrate rats. Am J Physiol. 1982;243:H837–843. doi: 10.1152/ajpheart.1982.243.6.H837. [DOI] [PubMed] [Google Scholar]
- Fatemian M, Robbins PA. Human ventilatory response to CO2 after 8 h of isocapnic or poikilocapnic hypoxia. J Appl Physiol. 1998;85:1922–1928. doi: 10.1152/jappl.1998.85.5.1922. [DOI] [PubMed] [Google Scholar]
- Fordyce WE, et al. Role of the carotid bodies in ventilatory acclimation to chronic hypoxia by the awake cat. Respir Physiol. 1984;58:207–221. doi: 10.1016/0034-5687(84)90149-x. [DOI] [PubMed] [Google Scholar]
- Forster HV, Bisgard GE, Klein JP. Effect of peripheral chemoreceptor denervation on acclimatization of goats during hypoxia. J Appl Physiol. 1981;50:392–398. doi: 10.1152/jappl.1981.50.2.392. [DOI] [PubMed] [Google Scholar]
- Forster HV, Bisgard GE, Rasmussen B, Orr JA, Buss DD, Manohar M. Ventilatory control in peripheral chemoreceptor-denervated ponies during chronic hypoxemia. J Appl Physiol. 1976;41:878–885. doi: 10.1152/jappl.1976.41.6.878. [DOI] [PubMed] [Google Scholar]
- Forster HV, Dempsey JA, Birnbaum ML, Reddan WG, Thoden J, Grover RF, Rankin J. Effect of chronic exposure to hypoxia on ventilatory response to CO 2 and hypoxia. J Appl Physiol. 1971;31:586–592. doi: 10.1152/jappl.1971.31.4.586. [DOI] [PubMed] [Google Scholar]
- Fortuna MG, Stornetta RL, West GH, Guyenet PG. Activation of the retrotrapezoid nucleus by posterior hypothalamic stimulation. J Physiol. 2009;587:5121–5138. doi: 10.1113/jphysiol.2009.176875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fregosi RF, Knuth SL, Ward DK, Bartlett D., Jr Hypoxia inhibits abdominal expiratory nerve activity. J Appl Physiol. 1987;63:211–220. doi: 10.1152/jappl.1987.63.1.211. [DOI] [PubMed] [Google Scholar]
- Gautier H, Bonora M. Effects of hypoxia and respiratory stimulants in conscious intact and carotid denervated cats. Bull Eur Physiopathol Respir. 1982;18:565–582. [PubMed] [Google Scholar]
- Geise K, Berndt J, Berger N. Interaction of central and peripheral drives in cats. II. Peripheral and central interaction of hypoxia and hypercapnia. Pflugers Arch. 1978;374:211–217. doi: 10.1007/BF00585597. [DOI] [PubMed] [Google Scholar]
- Georgopoulus D, Giannouli E, Tsara V, Argiropoulou P, Patakas D, Anthonisen NR. Respiratory short-term poststimulus potentiation (after-discharge) in patients with obstructive sleep apnea. Am Rev Respir Dis. 1992;146:1250–1255. doi: 10.1164/ajrccm/146.5_Pt_1.1250. [DOI] [PubMed] [Google Scholar]
- Gesell R, Lapides J, Levin M. The interaction of central and peripheral chemical control of breathing. Am J Physiol. 1940;130:155–170. [Google Scholar]
- Gestreau C, Heitzmann D, Thomas J, Dubreuil V, Bandulik S, Reichold M, Bendahhou S, Pierson P, Sterner C, Peyronnet-Roux J, Benfriha C, Tegtmeier I, Ehnes H, Georgieff M, Lesage F, Brunet JF, Goridis C, Warth R, Barhanin J. Task2 potassium channels set central respiratory CO2 and O2 sensitivity. Proc Natl Acad Sci U S A. 2010;107:2325–2330. doi: 10.1073/pnas.0910059107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guyenet PG. Neural structures that mediate sympathoexcitation during hypoxia. Respir Physiol. 2000;121:147–162. doi: 10.1016/s0034-5687(00)00125-0. [DOI] [PubMed] [Google Scholar]
- Guyenet PG. The 2008 Carl Ludwig Lecture: retrotrapezoid nucleus, CO2 homeostasis, and breathing automaticity. J Appl Physiol. 2008;105:404–416. doi: 10.1152/japplphysiol.90452.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guyenet PG, Bayliss DA, Mulkey DK, Stornetta RL, Moreira TS, Takakura AT. The retrotrapezoid nucleus and central chemoreception. Adv Exp Med Biol. 2008a;605:327–332. doi: 10.1007/978-0-387-73693-8_57. [DOI] [PubMed] [Google Scholar]
- Guyenet PG, Mulkey DK, Stornetta RL, Bayliss DA. Regulation of ventral surface chemoreceptors by the central respiratory pattern generator. J Neurosci. 2005a;25:8938–8947. doi: 10.1523/JNEUROSCI.2415-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guyenet PG, Stornetta RL, Bayliss DA. Retrotrapezoid nucleus and central chemoreception. J Physiol. 2008b;586:2043–2048. doi: 10.1113/jphysiol.2008.150870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guyenet PG, Stornetta RL, Bayliss DA, Mulkey DK. Retrotrapezoid nucleus: a litmus test for the identification of central chemoreceptors. Exp Physiol. 2005b doi: 10.1113/expphysiol.2004.029637. [DOI] [PubMed] [Google Scholar]
- Hayashi F, Coles SK, Bach KB, Mitchell GS, McCrimmon DR. Time-dependent phrenic nerve responses to carotid afferent activation: intact vs. decerebellate rats. Am J Physiol. 1993;265:R811–819. doi: 10.1152/ajpregu.1993.265.4.R811. [DOI] [PubMed] [Google Scholar]
- Hayashi F, Sinclair JD. Respiratory patterns in anesthetised rats before and after anemic decerebration. Respir Physiol. 1991;84:61–76. doi: 10.1016/0034-5687(91)90019-f. [DOI] [PubMed] [Google Scholar]
- He L, Chen J, Liu X, Dinger B, Fidone S. Enhanced nitric oxide-mediated chemoreceptor inhibition and altered cyclic GMP signaling in rat carotid body following chronic hypoxia. Am J Physiol Lung Cell Mol Physiol. 2007;293:L1463–1468. doi: 10.1152/ajplung.00249.2007. [DOI] [PubMed] [Google Scholar]
- He L, Dinger B, Gonzalez C, Obeso A, Fidone S. Function of NADPH oxidase and signaling by reactive oxygen species in rat carotid body type I cells. Adv Exp Med Biol. 2006;580:155–160. doi: 10.1007/0-387-31311-7_23. discussion 351–159. [DOI] [PubMed] [Google Scholar]
- Heeringa J, Berkenbosch A, de Goede J, Olievier CN. Relative contribution of central and peripheral chemoreceptors to the ventilatory response to CO2 during hyperoxia. Respir Physiol. 1979;37:365–379. doi: 10.1016/0034-5687(79)90082-3. [DOI] [PubMed] [Google Scholar]
- Hellstrom S. Morphometric studies of dense-cored vesicles in type I cells of rat carotid body. J Neurocytol. 1975;4:77–86. doi: 10.1007/BF01099097. [DOI] [PubMed] [Google Scholar]
- Hodges MR, Opansky C, Qian B, Davis S, Bonis JM, Krause K, Pan LG, Forster HV. Carotid body denervation alters ventilatory responses to ibotenic acid injections or focal acidosis in the medullary raphe. J Appl Physiol. 2005;98:1234–1242. doi: 10.1152/japplphysiol.01011.2004. [DOI] [PubMed] [Google Scholar]
- Hudgel DW, Chapman KR, Faulks C, Hendricks C. Changes in inspiration muscle electrical activity and upper airway resistance during periodic breathing induced by hypoxia during sleep. Am Rev Respir Dis. 1987;135:899–906. doi: 10.1164/arrd.1987.135.4.899. [DOI] [PubMed] [Google Scholar]
- Hudgel DW, Gordon EA, Thanakitcharu S, Bruce EN. Instability of ventilatory control in patients with obstructive sleep apnea. Am J Respir Crit Care Med. 1998;158:1142–1149. doi: 10.1164/ajrccm.158.4.9712105. [DOI] [PubMed] [Google Scholar]
- Kalia M, Mei SS, Kao FF. Central projections from ergoreceptors (C fibers) in muscle involved in cardiopulmonary responses to static exercise. Circ Res. 1981;48:148–162. [PubMed] [Google Scholar]
- Khayat RN, Xie A, Patel AK, Kaminski A, Skatrud JB. Cardiorespiratory effects of added dead space in patients with heart failure and central sleep apnea. Chest. 2003;123:1551–1560. doi: 10.1378/chest.123.5.1551. [DOI] [PubMed] [Google Scholar]
- Khoo MC, Kronauer RE, Strohl KP, Slutsky AS. Factors inducing periodic breathing in humans: a general model. J Appl Physiol. 1982;53:644–659. doi: 10.1152/jappl.1982.53.3.644. [DOI] [PubMed] [Google Scholar]
- Kiwull P, Wiemer W, Schone H. The role of the carotid chemoreceptors in the CO 2 -hyperpnea under hyperoxia. Pflugers Arch. 1972;336:171–186. doi: 10.1007/BF00592930. [DOI] [PubMed] [Google Scholar]
- Kiwull PH, Kiwull-Schone S, Klatt W. Interaction of central and peripheral respiratory drives: differentiation between the role of stimuli and afferents. In: Loeschcke HH, editor. Acid-Base Homeostasis of the Brain Extracellular Fluid and Respiratory Control System. Thieme; Stuttgart: 1976. pp. 146–156. [Google Scholar]
- Kline DD. Plasticity in glutamatergic NTS neurotransmission. Respir Physiol Neurobiol. 2008;164:105–111. doi: 10.1016/j.resp.2008.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi S. Fine structure of the carotid body of the dog. Arch Histol Jpn. 1968;30:95–120. doi: 10.1679/aohc1950.30.95. [DOI] [PubMed] [Google Scholar]
- Krasney JA, Magno MG, Levitzky MG, Koehler RC, Davies DG. Cardiovascular responses to arterial hypoxia in awake sinoaortic-denervated dogs. J Appl Physiol. 1973;35:733–738. doi: 10.1152/jappl.1973.35.5.733. [DOI] [PubMed] [Google Scholar]
- Kubin L, Alheid GF, Zuperku EJ, McCrimmon DR. Central pathways of pulmonary and lower airway vagal afferents. J Appl Physiol. 2006;101:618–627. doi: 10.1152/japplphysiol.00252.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lahiri S, DeLaney RG. Relationship between carotid chemoreceptor activity and ventilation in the cat. Respir Physiol. 1975a;24:267–286. doi: 10.1016/0034-5687(75)90018-3. [DOI] [PubMed] [Google Scholar]
- Lahiri S, DeLaney RG. Stimulus interaction in the responses of carotid body chemoreceptor single afferent fibers. Respir Physiol. 1975b;24:249–266. doi: 10.1016/0034-5687(75)90017-1. [DOI] [PubMed] [Google Scholar]
- Lahiri S, Edelman NH, Cherniack NS, Fishman AP. Role of carotid chemoreflex in respiratory acclimatization to hypoxemia in goat and sheep. Respir Physiol. 1981;46:367–382. doi: 10.1016/0034-5687(81)90132-8. [DOI] [PubMed] [Google Scholar]
- Liu Q, Kim J, Cinotte J, Homolka P, Wong-Riley MT. Carotid body denervation effect on cytochrome oxidase activity in pre-Botzinger complex of developing rats. J Appl Physiol. 2003;94:1115–1121. doi: 10.1152/japplphysiol.00765.2002. [DOI] [PubMed] [Google Scholar]
- Loeschcke HH, Mitchell RA, Katsaros B, Perkins JF, Jr, Konig A. Interaction of intracranial chemosensitivity with peripheral afferents to the respiratory centers. Ann N Y Acad Sci. 1963;109:651–660. doi: 10.1111/j.1749-6632.1963.tb13495.x. [DOI] [PubMed] [Google Scholar]
- Long WQ, Giesbrecht GG, Anthonisen NR. Ventilatory response to moderate hypoxia in awake chemodenervated cats. J Appl Physiol. 1993;74:805–810. doi: 10.1152/jappl.1993.74.2.805. [DOI] [PubMed] [Google Scholar]
- McDonald DM, RAM The innervation of glomus cells, ganglion cells and blood vessels in the rat carotid body: A quantitative ultrastructural analysis. J Neurocytol. 1975;4:177–230. [Google Scholar]
- Melton JE, Neubauer JA, Edelman NH. CO2 sensitivity of cat phrenic neurogram during hypoxic respiratory depression. J Appl Physiol. 1988;65:736–743. doi: 10.1152/jappl.1988.65.2.736. [DOI] [PubMed] [Google Scholar]
- Melton JE, Yu QP, Neubauer JA, Edelman NH. Modulation of respiratory responses to carotid sinus nerve stimulation by brain hypoxia. J Appl Physiol. 1992;73:2166–2171. doi: 10.1152/jappl.1992.73.5.2166. [DOI] [PubMed] [Google Scholar]
- Meza S, Mendez M, Ostrowski M, Younes M. Susceptibility to periodic breathing with assisted ventilation during sleep in normal subjects. J Appl Physiol. 1998;85:1929–1940. doi: 10.1152/jappl.1998.85.5.1929. [DOI] [PubMed] [Google Scholar]
- Millhorn DE, Eldridge FL. Role of ventrolateral medulla in regulation of respiratory and cardiovascular systems. J Appl Physiol. 1986;61:1249–1263. doi: 10.1152/jappl.1986.61.4.1249. [DOI] [PubMed] [Google Scholar]
- Millhorn DE, Kiley JP. Effect of graded cooling of intermediate areas on respiratory response to vagal input. Respir Physiol. 1984;58:51–64. doi: 10.1016/0034-5687(84)90044-6. [DOI] [PubMed] [Google Scholar]
- Mitchell GS, Selby BD. Effects of carotid denervation on interactions between lung inflation and PaCO2 in modulating phrenic activity. Respir Physiol. 1987;67:367–378. doi: 10.1016/0034-5687(87)90066-1. [DOI] [PubMed] [Google Scholar]
- Mitchell GS, Smith CA, Dempsey JA. Changes in the VI-VCO2 relationship during exercise in goats: role of carotid bodies. J Appl Physiol. 1984;57:1894–1900. doi: 10.1152/jappl.1984.57.6.1894. [DOI] [PubMed] [Google Scholar]
- Mizusawa A, Ogawa H, Kikuchi Y, Hida W, Kurosawa H, Okabe S, Takishima T, Shirato K. In vivo release of glutamate in nucleus tractus solitarii of the rat during hypoxia. J Physiol (Lond) 1994;478:55–66. doi: 10.1113/jphysiol.1994.sp020229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizusawa A, Ogawa H, Kikuchi Y, Hida W, Shirato K. Role of the parabrachial nucleus in ventilatory responses of awake rats. J Physiol (Lond) 1995;489:877–884. doi: 10.1113/jphysiol.1995.sp021100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreira TS, Takakura AC, Colombari E, West GH, Guyenet PG. Inhibitory input from slowly adapting lung stretch receptors to retrotrapezoid nucleus chemoreceptors. J Physiol. 2007;580:285–300. doi: 10.1113/jphysiol.2006.125336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morita E, Chiocchio SR, Tramezzani JH. Four types of main cells in the carotid body of the cat. J Ultrastruct Res. 1969;28:399–410. doi: 10.1016/s0022-5320(69)80029-8. [DOI] [PubMed] [Google Scholar]
- Mulkey DK, Rosin DL, West G, Takakura AC, Moreira TS, Bayliss DA, Guyenet PG. Serotonergic neurons activate chemosensitive retrotrapezoid nucleus neurons by a pH-independent mechanism. J Neurosci. 2007;27:14128–14138. doi: 10.1523/JNEUROSCI.4167-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulkey DK, Stornetta RL, Weston MC, Simmons JR, Parker A, Bayliss DA, Guyenet PG. Respiratory control by ventral surface chemoreceptor neurons in rats. Nat Neurosci. 2004;7:1360–1369. doi: 10.1038/nn1357. [DOI] [PubMed] [Google Scholar]
- Nakayama H, Smith CA, Rodman JR, Skatrud JB, Dempsey JA. Effect of ventilatory drive on carbon dioxide sensitivity below eupnea during sleep. Am J Respir Crit Care Med. 2002;165:1251–1259. doi: 10.1164/rccm.2110041. [DOI] [PubMed] [Google Scholar]
- Nattie E, Li A. Central chemoreception 2005: a brief review. Auton Neurosci. 2006;126–127:332–338. doi: 10.1016/j.autneu.2006.02.003. [DOI] [PubMed] [Google Scholar]
- Nattie EE, Li A. CO2 dialysis in the medullary raphe of the rat increases ventilation in sleep. J Appl Physiol. 2001;90:1247–1257. doi: 10.1152/jappl.2001.90.4.1247. [DOI] [PubMed] [Google Scholar]
- Nattie EE, Li A. CO2 dialysis in nucleus tractus solitarius region of rat increases ventilation in sleep and wakefulness. J Appl Physiol. 2002;92:2119–2130. doi: 10.1152/japplphysiol.01128.2001. [DOI] [PubMed] [Google Scholar]
- Nattie EE, Li A. Central chemoreception is a complex system function that involves multiple brainstem sites. J Appl Physiol. 2009;106:1464–1466. doi: 10.1152/japplphysiol.00112.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neubauer JA, Santiago TV, Posner MA, Edelman NH. Ventral medullary pH and ventilatory responses to hyperperfusion and hypoxia. J Appl Physiol. 1985;58:1659–1668. doi: 10.1152/jappl.1985.58.5.1659. [DOI] [PubMed] [Google Scholar]
- Neubauer JA, Sunderram J. Oxygen-sensing neurons in the central nervous system. J Appl Physiol. 2004;96:367–374. doi: 10.1152/japplphysiol.00831.2003. [DOI] [PubMed] [Google Scholar]
- Nielsen AM, Bisgard GE, Mitchell GS. Phrenic nerve responses to hypoxia and CO2 in decerebrate dogs. Respir Physiol. 1986;65:267–283. doi: 10.1016/0034-5687(86)90012-5. [DOI] [PubMed] [Google Scholar]
- Nielsen AM, Bisgard GE, Vidruk EH. Carotid chemoreceptor activity during acute and sustained hypoxia in goats. J Appl Physiol. 1988;65:1796–1802. doi: 10.1152/jappl.1988.65.4.1796. [DOI] [PubMed] [Google Scholar]
- Niu WZ, Engwall MJ, Bisgard GE. Two discharge patterns of carotid body chemoreceptors in the goat. J Appl Physiol. 1990;69:734–739. doi: 10.1152/jappl.1990.69.2.734. [DOI] [PubMed] [Google Scholar]
- Nolan PC, Waldrop TG. In vitro responses of VLM neurons to hypoxia after normobaric hypoxic acclimatization. Respir Physiol. 1996;105:23–33. doi: 10.1016/0034-5687(96)00033-3. [DOI] [PubMed] [Google Scholar]
- Nuding SC, Segers LS, Shannon R, O’Connor R, Morris KF, Lindsey BG. Central and peripheral chemoreceptors evoke distinct responses in simultaneously recorded neurons of the raphe-pontomedullary respiratory network. Philos Trans R Soc Lond B Biol Sci. 2009;364:2501–2516. doi: 10.1098/rstb.2009.0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olson EB, Jr, Vidruk EH, Dempsey JA. Carotid body excision significantly changes ventilatory control in awake rats. J Appl Physiol. 1988;64:666–671. doi: 10.1152/jappl.1988.64.2.666. [DOI] [PubMed] [Google Scholar]
- Ou LC, Miller MJ, Tenney SM. Hypoxia and carbon dioxide as separate and interactive depressants of ventilation. Respir Physiol. 1976;28:347–358. doi: 10.1016/0034-5687(76)90029-3. [DOI] [PubMed] [Google Scholar]
- Pan LG, Forster HV, Martino P, Strecker PJ, Beales J, Serra A, Lowry TF, Forster MM, Forster AL. Important role of carotid afferents in control of breathing. J Appl Physiol. 1998;85:1299–1306. doi: 10.1152/jappl.1998.85.4.1299. [DOI] [PubMed] [Google Scholar]
- Pan LG, Forster HV, Wurster RD, Brice AG, Lowry TF. Effect of multiple denervations on the exercise hyperpnea in awake ponies. J Appl Physiol. 1995;79:302–311. doi: 10.1152/jappl.1995.79.1.302. [DOI] [PubMed] [Google Scholar]
- Rahn H, Stroud RC, Tenney SM, Mithoefer JC. Adaptation to High Altitude: Respiratory Response to CO2 and O2. J Appl Physiol. 1953;6:158–162. doi: 10.1152/jappl.1953.6.3.158. [DOI] [PubMed] [Google Scholar]
- Read DJC. A clinical method for assessing the ventilatory response to carbon dioxide. Australas Ann Med. 1967;16:20–32. doi: 10.1111/imj.1967.16.1.20. [DOI] [PubMed] [Google Scholar]
- Robbins PA. Evidence for interaction between the contributions to ventilation from the central and peripheral chemoreceptors in man. J Physiol (London) 1988;401:503–518. doi: 10.1113/jphysiol.1988.sp017175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodman JR, Curran AK, Henderson KS, Dempsey JA, Smith CA. Carotid body denervation in dogs: eupnea and the ventilatory response to hyperoxic hypercapnia. J Appl Physiol. 2001;91:328–335. doi: 10.1152/jappl.2001.91.1.328. [DOI] [PubMed] [Google Scholar]
- Rosin DL, Chang DA, Guyenet PG. Afferent and efferent connections of the rat retrotrapezoid nucleus. J Comp Neurol. 2006;499:64–89. doi: 10.1002/cne.21105. [DOI] [PubMed] [Google Scholar]
- Schlaefke ME, Loeschcke HH. Lokalisation an der Regulation von Atmung und Kreislauf beteiligten Gebietes an der ventralen Oberfläche de Medulla oblongata durch Kälteblockade. Pflugers Arch. 1967;297:201–220. [Google Scholar]
- Skatrud JB, Dempsey JA. Interaction of sleep state and chemical stimuli in sustaining rhythmic ventilation. J Appl Physiol. 1983;55:813–822. doi: 10.1152/jappl.1983.55.3.813. [DOI] [PubMed] [Google Scholar]
- Smith CA, Bisgard GE, Nielsen AM, Daristotle L, Kressin NA, Forster HV, Dempsey JA. Carotid bodies are required for ventilatory acclimatization to chronic hypoxia. J Appl Physiol. 1986;60:1003–1010. doi: 10.1152/jappl.1986.60.3.1003. [DOI] [PubMed] [Google Scholar]
- Smith CA, Chenuel BJ, Henderson KS, Dempsey JA. The apneic threshold during non-REM sleep in dogs: sensitivity of carotid body vs. central chemoreceptors. J Appl Physiol. 2007;103:578–586. doi: 10.1152/japplphysiol.00017.2007. [DOI] [PubMed] [Google Scholar]
- Smith CA, Engwall MJ, Dempsey JA, Bisgard GE. Effects of specific carotid body and brain hypoxia on respiratory muscle control in the awake goat. J Physiol. 1993;460:623–640. doi: 10.1113/jphysiol.1993.sp019490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith CA, Jameson LC, Dempsey JA. Effects of altered CSF [H+] on ventilatory responses to exercise in the awake goat. J Appl Physiol. 1988;65:921–927. doi: 10.1152/jappl.1988.65.2.921. [DOI] [PubMed] [Google Scholar]
- Smith CA, Jameson LC, Mitchell GS, Musch TI, Dempsey JA. Central-peripheral chemoreceptor interaction in awake cerebrospinal fluid-perfused goats. J Appl Physiol. 1984;56:1541–1549. doi: 10.1152/jappl.1984.56.6.1541. [DOI] [PubMed] [Google Scholar]
- Smith CA, Rodman JR, Chenuel BJ, Henderson KS, Dempsey JA. Response Time and Sensitivity of the Ventilatory Response to CO2 in Unanesthetized Intact Dogs: Central vs. Peripheral Chemoreceptors. J Appl Physiol. 2006;100:13–19. doi: 10.1152/japplphysiol.00926.2005. [DOI] [PubMed] [Google Scholar]
- Smith CA, Saupe KW, Henderson KS, Dempsey JA. Ventilatory effects of specific carotid body hypocapnia in dogs during wakefulness and sleep. J Appl Physiol. 1995;79:689–699. doi: 10.1152/jappl.1995.79.3.689. [DOI] [PubMed] [Google Scholar]
- Smith JC, Morrison DE, Ellenberger HH, Otto MR, Feldman JL. Brainstem projections to the major respiratory neuron populations in the medulla of the cat. J Comp Neurol. 1989;281:69–96. doi: 10.1002/cne.902810107. [DOI] [PubMed] [Google Scholar]
- Solomon IC. Excitation of phrenic and sympathetic output during acute hypoxia: contribution of medullary oxygen detectors. Respir Physiol. 2000;121:101–117. doi: 10.1016/s0034-5687(00)00122-5. [DOI] [PubMed] [Google Scholar]
- Song G, Poon CS. Lateral parabrachial nucleus mediates shortening of expiration and increase of inspiratory drive during hypercapnia. Respir Physiol Neurobiol. 2009;165:9–12. doi: 10.1016/j.resp.2008.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- St Croix CM, Cunningham DA, Paterson DH. Nature of the interaction between central and peripheral chemoreceptor drives in human subjects. Can J Physiol Pharmacol. 1996;74:640–646. doi: 10.1139/cjpp-74-6-640. [DOI] [PubMed] [Google Scholar]
- Stornetta RL, Moreira TS, Takakura AC, Kang BJ, Chang DA, West GH, Brunet JF, Mulkey DK, Bayliss DA, Guyenet PG. Expression of Phox2b by brainstem neurons involved in chemosensory integration in the adult rat. J Neurosci. 2006;26:10305–10314. doi: 10.1523/JNEUROSCI.2917-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takakura AC, Moreira TS, Colombari E, West GH, Stornetta RL, Guyenet PG. Peripheral chemoreceptor inputs to retrotrapezoid nucleus (RTN) CO2-sensitive neurons in rats. J Physiol. 2006;572:503–523. doi: 10.1113/jphysiol.2005.103788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takakura AC, Moreira TS, Stornetta RL, West GH, Gwilt JM, Guyenet PG. Selective lesion of retrotrapezoid Phox2b-expressing neurons raises the apnoeic threshold in rats. J Physiol. 2008;586:2975–2991. doi: 10.1113/jphysiol.2008.153163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tenney SM, Ou LC. Ventilatory response of decorticate and decerebrate cats to hypoxia and CO2. Respiration Physiology. 1977;29:81–92. doi: 10.1016/0034-5687(77)90119-0. [DOI] [PubMed] [Google Scholar]
- Teppema LJ, Veening JG, Kranenburg A, Dahan A, Berkenbosch A, Olievier C. Expression of c-fos in the rat brainstem after exposure to hypoxia and to normoxic and hyperoxic hypercapnia. J Comp Neurol. 1997;388:169–190. doi: 10.1002/(sici)1096-9861(19971117)388:2<169::aid-cne1>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- van Beek JH, Berkenbosch A, de Goede J, Olievier CN. Influence of peripheral O2 tension on the ventilatory response to CO2 in cats. Respir Physiol. 1983;51:379–390. doi: 10.1016/0034-5687(83)90030-0. [DOI] [PubMed] [Google Scholar]
- van Beek JH, Berkenbosch A, de Goede J, Olievier CN. Effects of brain stem hypoxaemia on the regulation of breathing. Respir Physiol. 1984;57:171–188. doi: 10.1016/0034-5687(84)90091-4. [DOI] [PubMed] [Google Scholar]
- Vizek M, Pickett CK, Weil JV. Increased carotid body hypoxic sensitivity during acclimatization to hypobaric hypoxia. J Appl Physiol. 1987;63:2403–2410. doi: 10.1152/jappl.1987.63.6.2403. [DOI] [PubMed] [Google Scholar]
- Warner G, Skatrud JB, Dempsey JA. Effect of hypoxia-induced periodic breathing on upper airway obstruction during sleep. J Appl Physiol. 1987;62:2201–2211. doi: 10.1152/jappl.1987.62.6.2201. [DOI] [PubMed] [Google Scholar]
- Weil JV, Byrne-Quinn E, Sodal IE, Kline JS, McCullough RE, Grover RF, Filley GF. Ventilatory control in normal man: effects of acute exercise, chronic physical conditioning and chronic hypoxia. Chest. 1972;61(Suppl):45S–46S. doi: 10.1378/chest.61.2_supplement.45s. [DOI] [PubMed] [Google Scholar]
- Weizhen N, Engwall MJ, Daristotle L, Pizarro J, Bisgard GE. Ventilatory effects of prolonged systemic (CNS) hypoxia in awake goats. Respir Physiol. 1992;87:37–48. doi: 10.1016/0034-5687(92)90098-h. [DOI] [PubMed] [Google Scholar]
- Wellman A, Jordan AS, Malhotra A, Fogel RB, Katz ES, Schory K, Edwards JK, White DP. Ventilatory control and airway anatomy in obstructive sleep apnea. Am J Respir Crit Care Med. 2004;170:1225–1232. doi: 10.1164/rccm.200404-510OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weston MC, Stornetta RL, Guyenet PG. Glutamatergic neuronal projections from the marginal layer of the rostral ventral medulla to the respiratory centers in rats. J Comp Neurol. 2004;473:73–85. doi: 10.1002/cne.20076. [DOI] [PubMed] [Google Scholar]
- Xie A, Skatrud JB, Barczi SR, Reichmuth K, Morgan BJ, Mont S, Dempsey JA. Influence of cerebral blood flow on breathing stability. J Appl Physiol. 2009;106:850–856. doi: 10.1152/japplphysiol.90914.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie A, Skatrud JB, Dempsey JA. Effect of hypoxia on the hypopnoeic and apnoeic threshold for CO2 in sleeping humans. J Physiol. 2001;535:269–278. doi: 10.1111/j.1469-7793.2001.00269.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie A, Skatrud JB, Morgan B, Chenuel B, Khayat R, Reichmuth K, Lin J, Dempsey JA. Influence of cerebrovascular function on the hypercapnic ventilatory response in healthy humans. J Physiol. 2006;577:319–329. doi: 10.1113/jphysiol.2006.110627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, Carreno FR, Cunningham JT, Mifflin SW. Chronic sustained hypoxia enhances both evoked EPSCs and norepinephrine inhibition of glutamatergic afferent inputs in the nucleus of the solitary tract. J Neurosci. 2009;29:3093–3102. doi: 10.1523/JNEUROSCI.2648-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]