Abstract
Objective
Dairy food intake has been associated with prostate cancer in previous work, but the mechanism by which this occurs is unknown. Dairy calcium may suppress circulating levels of potentially cancer-protective 1,25-hydroxyvitamin D (1,25(OH)2D). We examined the associations of dairy, milk, calcium, and vitamin D intake with plasma 1,25(OH)2D levels among 296 men (194 black, 102 non-black) enrolled in high risk program for prostate cancer from 10/96-10/07.
Methods
All participants completed diet and health history questionnaires and provided plasma samples, which were assessed for levels of 25-hydroxyvitamin D and 1,25(OH)2D. We used multivariate linear regression to examine associations with 1,25(OH)2D.
Results
After adjustment for age, race, energy intake, BMI, and alcohol intake, we observed no associations for any of our variables of interest with 1,25(OH)2D, or any meaningful differences in estimates by race or vitamin D status.
Conclusion
Our findings, in a sample including a large proportion of black participants, do not confirm previous findings showing an inverse association between calcium intake and 1,25(OH)2D levels. As such, they suggest that future work should explore other mechanisms by which dairy foods and calcium might increase prostate cancer risk.
Keywords: dairy, milk, prostate neoplasms, vitamin D
INTRODUCTION
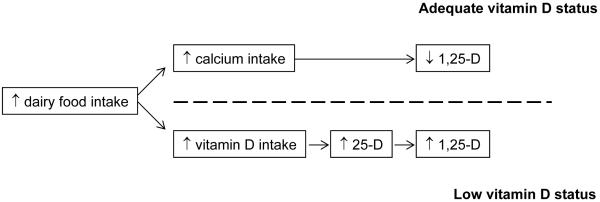
Although epidemiologic studies have fairly consistently found an increase in prostate cancer risk with intake of dairy foods (1), mechanisms by which dairy foods increase risk of prostate cancer are unknown. The hypothesis that 1,25-dihydroxyvitamin D (1,25(OH)2D) might protect against prostate cancer (2) suggests one possible mechanism: that, consumed at sufficiently high levels, calcium in dairy suppresses production and circulating levels of 1,25(OH)2D, thereby increasing risk for prostate cancer (3). Vitamin D, which is derived primarily from the sunlight-catalyzed synthesis of 7-dehydrocholesterol in the skin, is converted through hydroxylation first to 25-hydroxyvitamin D (25(OH)D), considered the better indicator for vitamin D status (4), then again by the enzyme 25-hydroxyvitamin D-1alpha-hydroxylase (1alpha-OHase) to its biologically active form, 1,25(OH)2D (4). The effect of dairy intake on 1,25(OH)2D levels may differ by vitamin D status. Whereas a high level of dairy calcium may increase risk among those with adequate vitamin D status, in vitamin D deficient men dairy foods may increase levels of 25(OH)D, the precursor substrate for conversion to 1,25(OH)2D in the prostate (4), consequently increasing availability of 1,25(OH)2D (Figure 1).
Figure 1.
Confirming associations within these pathways would have important implications for dairy food and calcium intake in prostate cancer prevention. These implications are particularly relevant to men at high risk for prostate cancer, for whom preventive measures are limited but are most needed. The objectives of this analysis were to evaluate the hypothesis that dairy food and calcium intake reduces circulating 1,25(OH)2D levels in a sample of men at high risk for prostate cancer, including a large proportion of black participants. We also hypothesized that the association of dairy food intake with 1,25(OH)2D levels would differ by vitamin D status.
MATERIALS AND METHODS
Study sample
Participants for the proposed study were men enrolled in the Fox Chase Cancer Center (FCCC) Prostate Cancer Risk Assessment Program (PRAP). PRAP was established by FCCC investigators in 1996 to offer education and preventive interventions to men at potentially high risk for prostate cancer, and to serve as a research base for studying gene-environment interactions in prostate cancer (5). PRAP participants are healthy men at high genetic risk for prostate cancer. Men are eligible for the program if they are (1) between 35 and 69 years of age, and (2) have no history of prostate cancer. In addition, non-black men are required to have at least one first-degree relative or two same-side second-degree relatives with prostate cancer; no family history of prostate cancer is required for African American men because of their already elevated risk. Recruitment strategies include referrals from prostate cancer patients at FCCC, radio and newspaper advertisements, and physician and self-referrals.
Of 702 men enrolled in PRAP between October, 1996, and October, 2007, we excluded those who were recruited through satellite locations and whose plasma samples thus could not be processed immediately after collection (N=152); who did not provide a blood sample after July, 2000, when standard procedures were established for processing and storing plasma samples in the FCCC Biosample Repository (N=116); who did not return a dietary questionnaire (N=108); who reported an infeasible energy intake (<500 kcal/day or >4400 kcal/day) (N=18); or who had a previous diagnosis of cancer (N=12), leaving 296 men available for these analyses.
The project was approved by the Institutional Review Board at the Fox Chase Cancer Center.
Data collection
Upon enrollment into PRAP, men completed a health history questionnaire and the Harvard Diet Assessment Form (DAF) (6). At initial and follow-up appointments, participants also contributed 51 mL of non-fasting blood samples for storage in the FCCC Biosample Repository. The health history questionnaire elicited information on sociodemographic characteristics, occupation, family history of prostate cancer, smoking status, physical activity, and self-reported height and weight. Occupational sun exposure was assessed based on each participant’s current occupation, which was classified into one of three categories according to presumed sunlight exposure (indoor work, combined indoor and outdoor work, outdoor work), using an index that was related to mortality from non-melanoma skin cancer in previous studies (7-9). The DAF elicited information on frequency of intake of 126 food items, and on length of use and dosage of dietary supplements, including calcium. Intake of specific nutrients such as energy, total fat, and calcium was estimated by multiplying reported frequency of intake of each food by the nutrient content for the specified portion size.
Assessment of plasma 25(OH)D and 1,25(OH)2D levels
Levels of plasma 25(OH)D and 1,25(OH)2D were determined by Heartland Assays, Inc. (Ames, IA). Levels of 25(OH)D were determined with a direct, competitive chemiluminescence immunoassay (CLIA) using the DiaSorin LIAISON platform. Levels of 1,25(OH)2D were determined using a competitive RIA procedure following extraction and subsequent purification of vitamin D metabolites from the plasma samples using C18OH cartridges.
Statistical analyses
We used linear regression models adjusting for age and race (black or non-black) to examine associations of sociodemographic, lifestyle, and dietary factors with 1,25(OH)2D levels. Intake of dairy was calculated as the total frequency of intake of nine items in the DAF: skim or low fat milk; whole milk; cream; sour cream; ice cream; yogurt; cottage or ricotta cheese; cream cheese; and other cheese. Nutrient values, estimated from dietary and supplemental sources, were log-transformed as necessary and energy-adjusted using the residual method (10). Dietary variables were modeled both as continuous variables and as categorical variables with the lowest category as the referent group. Categorical covariates were coded using dummy variables to allow for non-linear associations across categories.
Variables were included as potential confounders in multivariate models if they were significantly associated with 1,25(OH)2D levels in models including all men. Final multivariate models included 288 men with complete covariate data and adjusted for age (years), race (black, non-black), energy intake (kcal), BMI (kg/m2), and alcohol intake (0, >0-<6.5, >6.5-<26, ≥26 g/day). Models for dietary variables also adjusted for energy intake (kcals), and models for calcium and vitamin D intake were mutually adjusted for the other variable. Additional analyses with 25(OH)D as the outcome also adjusted for season of blood draw because of its strong association with 25(OH)D levels. Other variables evaluated as confounders but not included in final models were level of education, family history of prostate cancer, physical activity, total fat intake, and occupational sun exposure.
To evaluate the possibility that associations differ by vitamin D status, we compared estimates derived from models stratified on 25(OH)D level. We defined low 25(OH)D levels using a cutpoint of ≤15 ng/mL (37.5 nmol/L), which defines the lower limit of the normal range of 25(OH)D levels (11) and has been used as a criterion to define low vitamin D status in other studies (12, 13). We estimated p-values for interaction from models including all men, with dairy or calcium intake x vitamin D status (25(OH)D level ≤15 or >15 ng/mL) interaction terms. Dairy and calcium variables were analyzed as both continuous and categorical variables. We conducted similar analyses to examine potential effect modification by race (black, non-black). Because results stratified on race were not meaningfully different, we present results for all men or in subsets defined only based on 25(OH)D levels.
RESULTS
Distributions of relevant variables and their associations with 1,25(OH)2D level are shown in Table 1. The majority of our sample (66%) were black, and about half of the sample (47%) were classified as having low 25(OH)D levels. Black participants were more likely to have low vitamin D levels than were non-black participants (61% vs. 20%). Further, of 139 men with 25(OH)D level ≤15 ng/mL, 119 (86%) were black, vs. 75 (48%) of 157 men with 25(OH)D level >15 ng/mL. In age- and race-adjusted linear regression analyses, only 25(OH)D level was a significant predictor of 1,25(OH)2D level, although associations for BMI, vitamin D intake, and alcohol intake were marginally significant (Table 1). A surprisingly inverse association of outdoor occupation with 1,25(OH)2D level was non-significant and based on only ten men with occupations categorized as outdoor.
TABLE 1.
Distribution of variables among 296 participants in the Prostate Cancer Risk Assessment Program, and age- and race-adjusted associations with 1,25(OH)2D
| Variable | Beta a (p-value) | |
|---|---|---|
| Mean (±SD) age (y) | 49.9 (8.6) | <0.1 (0.52) |
|
| ||
| African American (%) | 66 | 0.9 (0.64) |
|
| ||
| Level of education (%) | ||
| high school graduate | 19 | ref |
| some college | 36 | 3.8 (0.14) |
| college graduate | 27 | 2.5 (0.37) |
| graduate degree | 18 | 1.8 (0.54) |
|
| ||
| Mean (±SD) BMI (kg/m2)b | 28.9 (5.1) | -0.3 (0.08) |
|
| ||
| Physical activity (hrs/wk) | 2.4 (2.1) | -0.3 (0.61) |
|
| ||
| Dairy food intake (times/week) | ||
| Mean (SD) | 10.5 (8.8) | 0.1 (0.50) |
| Median (1st, 3rd quartiles) | 8.4 (3.9, 14.2) | |
|
| ||
| Total milk intake (times/week) | ||
| Mean (SD) | 3.2 (4.5) | 0.2 (0.32) |
| Median (1st, 3rd quartiles) | 1.0 (0.5, 3.5) | |
|
| ||
| Whole milk intake (times/week) | ||
| Mean (SD) | 0.6 (1.5) | 0.3 (0.64) |
| Median (1st, 3rd quartiles) | 0 (0, 0.5) | |
|
| ||
| Skim/lowfat fat milk intake (times/week) | ||
| Mean (SD) | 2.6 (4.6) | 0.2 (0.4) |
| Median (1st, 3rd quartiles) | 0.5 (0, 3.0) | |
|
| ||
| Mean (SD) energy intake (kcal/day) | 1982 (742) | -1.3 (0.28) |
|
| ||
| Mean (SD) calcium intake (mg/day) | 718 (414) | 1.1 (0.27) |
|
| ||
| Mean (SD) vitamin D intake (IU /day) | 374 (312) | 0.6 (0.06) |
|
| ||
| Alcohol intake (%) | ||
| 0 g/day | 34 | ref |
| >0 - <6.5 g/day | 27 | 3.5 (0.14) |
| >6.5 - <26 g/day | 29 | 4.1 (0.09) |
| ≥26 g/day | 10 | 6.6 (0.05) |
| p-for-trend c | 0.06 | |
|
| ||
| Occupational sun exposure (%) | ||
| Indoor | 68 | ref |
| Mixed | 26 | 0.9 (0.78) |
| Outdoor | 6 | -0.9 (0.87) |
|
| ||
| Season of blood draw (%) | ||
| January - March | 28 | ref |
| April - June | 22 | 1.9 (0.47) |
| July - September | 25 | 0.2 (0.94) |
| October - December | 25 | 0.8 (0.76) |
|
| ||
| Mean (±SD) 25(OH)D (ng/mL) | 15.9 (6.8) | 0.5 (0.0005) |
|
| ||
| 25(OH)D ≤15 ng/mL (%) | 47 | -4.6 (0.02) |
|
| ||
| Mean (±SD) 1,25(OH)2D (pg/mL) | 37.2 (15.7) | -- |
Beta represents mean change in 1,25(OH)2D (pg/mL) for each 1000 kcal increm ent in energy intake, 350 mg increment in calcium intake, 100 IU increment in vitamin D intake. For all other continuous variables, beta represents mean change in outcome variable for each one-unit increment in predictor variable. For categorical variables, betas represent mean difference in outcome relative to referent category.
Due to missing data, N=288 for BmI; N=192 for physical activity; N=164 for occupational sun exposure.
Trend p-value calculated by including alcohol as an ordinal categorical variable in the model.
In unadjusted correlational analyses among the major factors of interest shown in Figure 1, dairy intake was strongly correlated with calcium intake (Table 2); it was also weakly correlated with vitamin D intake, but this was significant only when all men were included (Table 2). Vitamin D intake was correlated with 25(OH)D level, but only among men with low vitamin D status. Similarly, 25(OH)D and 1,25(OH)2D levels were correlated only among men with low vitamin D status. 1,25(OH)2D was not correlated with intake of dairy foods, calcium, or vitamin D in these analyses.
TABLE 2.
Spearman correlations among dairy food (servings/week), calcium (mg/day), and vitamin D (IU/day) intake a and levels of 25(OH)D (ng/mL) and 1,25(OH)2D (pg/mL) among all 296 participants in the Prostate Cancer Risk Assessment Program, and stratified on vitamin D status
| Dairy food | Calcium | Vitamin D | 25(OH)D | 1,25(OH)2D | |
|---|---|---|---|---|---|
| All men (N=296) | |||||
| Dairy food | 1.0 |
0.53 b (p<0.0001) |
0.15
(p=0.008) |
0.21
(p=0.0003) |
0.01 (p=0.87) |
| Calcium | 1.0 |
0.36
(p<0.0001) |
0.29
(p<0.0001) |
0.05 (p=0.39) | |
| Vitamin D | 1.0 |
0.24
(p<0.0001) |
0.10 (p=0.10) | ||
| 25(OH)D | 1.0 |
0.17
(p=0.003) |
|||
| 1,25(OH)2D | 1.0 | ||||
| 25(OH)D >15 ng/mL (N=157) | |||||
| Dairy food | 1.0 |
0.56
(p<0.0001) |
0.14 (p=0.09) |
0.003 (p=0.97) |
-0.002 (p=0.98) |
| Calcium | 1.0 |
0.31
(p<0.0001) |
0.11 (p=0.19) |
0.05 (p=0.52) |
|
| Vitamin D | 1.0 | 0.07 (p=0.39) |
0.12 (p=0.13) |
||
| 25(OH)D | 1.0 | 0.11 (p=0.17) |
|||
| 1,25(OH)2D | 1.0 | ||||
| 25(OH)D ≤15 ng/mL (N=139) | |||||
| Dairy food | 1.0 |
0.44
(p<0.0001) |
0.11 (p=0.21) |
0.09 (p=0.27) |
-0.02 (p=0.86) |
| Calcium | 1.0 |
0.37
(p<0.0001) |
0.24
(p=0.004) |
-0.001 (p=0.99) |
|
| Vitamin D | 1.0 |
0.32
(p=0.0002) |
0.01 (p=0.89) |
||
| 25(OH)D | 1.0 |
0.25
(p=0.004) |
|||
| 1,25(OH)2D | 1.0 |
Calcium and vitamin D intake were adjusted for energy intake using the residual method.
Boldface indicates p<0.05.
In multivariate linear regression analyses adjusting for BMI and alcohol intake as well as for age and race, we observed no associations of total dairy food, total milk, calcium, or vitamin D intake with 1,25(OH)2D levels in models including all men, nor we did we see any evidence of effect modification by vitamin D status (Table 3). We also saw no associations when we examined whole milk and low fat milk separately (data not shown). When we created categories to distinguish men with very high calcium intake, we still found no evidence that calcium >1500 mg/day was associated with lower 1,25(OH)2D levels, although the estimate for the highest category was based on only 16 men (data not shown).
Table 3.
Multivariate-adjusted mean 1,25(OH)2D levelsa by categories of dairy food, milk, calcium, and vitamin D intake in all men and stratified on vitamin D status
| Median | All men N=288 |
25(OH)D >15 ng/mL N=154 |
25(OH)D ≤15 ng/mL N=134 |
||||
|---|---|---|---|---|---|---|---|
| N | Mean 1,25(OH)2D (pg/mL) | N | Mean 1,25(OH)2D (pg/mL) | N | Mean 1,25(OH)2D (pg/mL) | ||
| Dairy | times/week | ||||||
| quartile 1 | 3 | 74 | 36.7 | 29 | 41.4 | 45 | 33.7 |
| quartile 2 | 7 | 70 | 36.8 | 35 | 38.4 | 35 | 35.0 |
| quartile 3 | 10 | 70 | 37.1 | 37 | 34.8 | 33 | 40.5 |
| quartile 4 | 20 | 74 | 39.0 | 53 | 41.7 | 21 | 33.9 |
| p-valueb | 0.45 | 0.40 | 0.95 | ||||
|
| |||||||
| Total milk | times/week | ||||||
| quartile 1 | 0 | 69 | 35.8 | 36 | 36.9 | 33 | 33.8 |
| quartile 2 | 0.5 | 81 | 36.8 | 36 | 41.6 | 45 | 32.7 |
| quartile 3 | 3 | 67 | 39.6 | 29 | 39.5 | 38 | 38.9 |
| quartile 4 | 7 | 71 | 38.3 | 53 | 38.4 | 18 | 37.3 |
| p-value | 0.27 | 0.49 | 0.53 | ||||
|
| |||||||
| Calcium | mg/day | ||||||
| quartile 1 | 427 | 69 | 37.0 | 29 | 38.4 | 40 | 34.9 |
| quartile 2 | 571 | 73 | 36.2 | 32 | 37.4 | 41 | 34.9 |
| quartile 3 | 725 | 72 | 38.1 | 41 | 39.4 | 31 | 36.6 |
| quartile 4 | 1033 | 74 | 38.5 | 52 | 40.8 | 22 | 35.5 |
| p-value | 0.40 | 0.41 | 0.69 | ||||
|
| |||||||
| Vitamin D | IU/day | ||||||
| quartile 1 | 116 | 71 | 36.4 | 29 | 33.4 | 42 | 38.6 |
| quartile 2 | 190 | 72 | 34.6 | 39 | 38.0 | 33 | 30.4 |
| quartile 3 | 397 | 72 | 40.3 | 37 | 43.3 | 35 | 37.0 |
| quartile 4 | 675 | 73 | 38.8 | 49 | 40.7 | 24 | 34.9 |
| p-value | 0.15 | 0.14 | 0.96 | ||||
Model adjusted for age, race, total energy intake, BMI, alcohol intake. Models for calcium and vitamin D intake also adjusted for the other as a continuous variable.
p-value for continuous variable
In exploring our hypothesized associations among dairy food intake, vitamin D intake, and levels of 25(OH)D and 1,25(OH)2D, we found that dietary vitamin D was significantly (beta=0.004, p=0.0001) associated with 25(OH)D levels after adjustment for age, race, BMI, intake of energy, alcohol, and calcium, and season of blood draw, regardless of vitamin D status (results not shown). Levels of 25(OH)D, in turn, were significantly associated with 1,25(OH)2D levels, but only among men with 25(OH)D levels ≤15 ng/mL (beta=0.98 (p=0.01) for men with 25(OH)D ≤15 ng/mL vs. beta=0.37 (p=0.28) for men with 25(OH)D >15 ng /mL). Dairy intake, however, was only weakly correlated with estimated intake of vitamin D (Table 2), and neither dairy nor vitamin D intake was associated with 1,25(OH)2D level (Table 3).
DISCUSSION
We found that dairy food and calcium intake was not associated with 1,25(OH)2D levels in our sample of men at high risk for prostate cancer. Results did not differ by vitamin D status or race, although an association of 25(OH)D with 1,25(OH)2D levels was limited to men with 25(OH)D levels ≤15 ng/mL.
A potentially suppressive effect of calcium on production and circulating levels of 1,25(OH)2D is a primary mechanism that has been suggested to explain the fairly consistent association of dairy and calcium intake with elevated prostate cancer risk. Prostate cells express 1α-hydroxylase, the enzyme that converts 25(OH)D to 1,25(OH)2D, but prostate cancer cells show a marked decrease in 1α-hydroxylase activity (14), thus possibly increasing prostatic cell dependence on circulating 1,25(OH)2D (15). In experimental studies, 1,25(OH)2D reduces cell proliferation, induces cell differentiation and apoptosis, and disturbs cell survival signals in the signaling pathway (16, 17), and these effects have been confirmed in prostate epithelial and cancer cells in animal models, (3). Despite tight regulation of circulating levels of 1,25(OH)2D, calcium intake may affect 1,25(OH)2D levels by affecting circulating calcium levels. Low calcium levels stimulate the release of parathyroid hormone, which in turn stimulates the hydroxylation of 25(OH)D to 1,25(OH)2D. High calcium levels may have the opposite effect (3).
We hypothesized that by increasing calcium intake and availability, dairy food intake would be associated with lower 1,25(OH)2D levels, particularly among men with adequate vitamin D status. Despite a strong correlation between dairy food and calcium intake (r=0.50, p<0.0001), we did not observe an inverse association for dairy food or calcium intake with 1,25(OH)2D level, even in analyses limited to men with adequate vitamin D status, or in additional analyses examining very high calcium intake. While a recent analysis conducted in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial also showed no association between calcium intake and 1,25(OH)2D levels (18), several other studies have shown an inverse correlation between the two (15, 19-26), even within the range of usual calcium intake (20-22). Nevertheless, levels of 1,25(OH)2D are known to be tightly regulated (11), and most of the studies showed relatively small changes in 1,25(OH)2D levels with large variations in calcium intake. Together, these studies suggest that an association between calcium and 1,25(OH)2D, if any, is too modest to explain the association between calcium and prostate cancer risk. Moreover, despite fairly consistent findings of an association for dairy and calcium intake with prostate cancer, even studies on prediagnostic serum vitamin D levels and prostate cancer are inconsistent (27-31). Whether suppression of 1,25(OH)2D levels is the primary mechanism by which dairy/calcium increase prostate cancer risk, therefore, remains an open question. Examining determinants of intracellular rather than circulating 1,25(OH)2D may yield more insight into prostate cancer risk, although measurement of intracellular 1,25(OH)2D in epidemiologic studies poses logistical challenges.
Among men with low vitamin D status we hypothesized that by increasing vitamin D intake dairy food intake would increase rather than decrease 1,25(OH)2D levels. The null findings in this low vitamin D status sample - mean 25(OH)D in our sample was 15.9 ng/mL, compared with 31.5 ng/mL in the Third National Health and Nutrition Examination Survey (20.9 ng/mL among black participants only) (32) - argues against this hypothesis. Although vitamin D intake in our sample was significantly associated with higher 25(OH)D levels, which was in turn associated with higher 1,25(OH)2D levels, dairy food intake was only weakly correlated with vitamin D intake, suggesting that other dietary sources of vitamin D are likely to be important. Also worth noting is that dietary vitamin D was not associated with 1,25(OH)2D level, possibly due in part to tight regulation of 1,25(OH)2D, as well as to measurement error in estimating vitamin D intake (11). Notably, previous studies have also not demonstrated a protective effect of dietary vitamin D for prostate cancer (22, 33-36).
We observed a marginally significant association of alcohol intake with 1,25(OH) 2D level. Alcohol abuse is known to disrupt bone metabolism, but the limited evidence available suggests that it would decrease rather than increase 1,25(OH)2D level (37). With no a priori reason to expect a positive association between alcohol intake and 1,25(OH) 2D, this finding warrants confirmation in other studies before further interpretation is possible.
Several limitations of this analysis merit discussion. First, our sample included men at high genetic risk for prostate cancer based on their race and/or family history. To the extent that circulating levels of 1,25(OH)2D are genetically influenced, their heritable component may overwhelm the effects of non-genetic factors such as diet. While it is unlikely that high risk men would be less responsive to dietary factors influencing vitamin D levels, we did attempt to account for genetic effects using information on men’s family history of prostate cancer but saw no association of family history with 1,25(OH)2D levels. Also, because black participants were eligible for PRAP regardless of their family history of prostate cancer, our findings have implications for black men in general. Indeed, despite our relatively small sample size, our analysis is the largest examination of these relationships among black men.
Measurement error in estimating dietary intake is another potential limitation, although one study demonstrated good validity of the questionnaire in an ethnically diverse sample (38). That dietary questionnaires were not always completed at the same time as the blood collection may have led to some error as well, although the median time between dietary questionnaire completion and blood draw for the 296 men in our analysis was only 12 days.
In summary, in this sample of men at high risk for prostate cancer, dairy, milk, and calcium intake were not associated with reduced 1,25(OH)2D levels. Among men with low vitamin D status, vitamin D intake contributed to higher 25(OH)D levels, which were in turn associated with levels of 1,25(OH)2D, but we were unable to confirm a direct association of dairy or vitamin D intake with 1,25(OH)2D levels in these men. Our findings, in a sample including a large proportion of black participants, do not confirm previous findings showing an inverse association between calcium intake and 1,25(OH)2D levels. As such, they suggest that future work should explore other mechanisms by which dairy foods and calcium might increase prostate cancer risk.
Supplementary Material
ACKNOWLEDGEMENTS
The authors thank the participants of the Prostate Cancer Risk Assessment Program at Fox Chase Cancer Center. We also thank Ms. Susan Raysor and Ms. JoEllen Weaver for their assistance in making the necessary datasets and samples available from the Prostate Cancer Risk Assessment Program; Dr. Cynthia Spittle for her assistance in facilitating transport of samples for vitamin D assays; and Dr. Mustafa Dosemeci for sharing his indices on residential and occupational sunlight exposure.
Grant support: Supported by grants R03 CA110892 and P30 CA006927 from the National Institutes of Health.
REFERENCES
- 1.Gao X, LaValley MP, Tucker KL. Prospective studies of dairy product and calcium intakes and prostate cancer risk: a meta-analysis. J Natl Cancer Inst. 2005 Dec 7;97(23):1768–77. doi: 10.1093/jnci/dji402. 2005. [DOI] [PubMed] [Google Scholar]
- 2.Schwartz GG, Hulka BS. Is vitamin D deficiency a risk factor for prostate cancer? Anticancer Res. 1990;10:1307–12. [PubMed] [Google Scholar]
- 3.Giovannucci EL. Dietary influences of 1,25(OH)2 vitamin D in relation to prostate cancer: a hypothesis. Cancer Causes Control. 1998;9:567–82. doi: 10.1023/a:1008835903714. [DOI] [PubMed] [Google Scholar]
- 4.Holick MF. Calcium and vitamin D: diagnostics and therapeutics. Clin Lab Med. 2000;20:569–90. [PubMed] [Google Scholar]
- 5.Bruner DW, Baffoe-Bonnie A, Miller S, Diefenbach M, Tricoli JV, Daly M, et al. Prostate cancer risk assessment program. A model for the early detection of prostate cancer. Oncology. 1999 Mar;13(3):325–34. discussion 37-9, 43-4 pas. [PubMed] [Google Scholar]
- 6.Willett WC, Sampson L, Browne ML, Stampfer MJ, Rosner B, Hennekens CH, et al. The use of a self-administered questionnaire to assess diet four years in the past. Am J Epidemiol. 1988;127:188–99. doi: 10.1093/oxfordjournals.aje.a114780. [DOI] [PubMed] [Google Scholar]
- 7.Freedman DM, Zahm SH, Dosemeci M. Residential and occupational exposure to sunlight and mortality from non-Hodgkin’s lymphoma: composite (threefold) case-control study. Br Med J. 1997;314:1451–5. doi: 10.1136/bmj.314.7092.1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Freedman DM, Dosemeci M, Alavanja MC. Mortality from multiple sclerosis and exposure to residential and occupational solar radiation: a case-control study based on death certificates. Occup Environ Med. 2000;57:418–21. doi: 10.1136/oem.57.6.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Freedman DM, Dosemeci M, McGlynn K. Sunlight and mortality from breast, ovarian, colon, prostate, and non-melanoma skin cancer: a composite death certificate based case-control study. Occup Environ Med. 2002;59:257–62. doi: 10.1136/oem.59.4.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Willett W, Stampfer M. Total energy intake: implications for epidemiologic analyses. Am J Epidemiol. 1986;124:17–27. doi: 10.1093/oxfordjournals.aje.a114366. [DOI] [PubMed] [Google Scholar]
- 11.Standing Committee on the Scientific Evaluation of Dietary Reference Intakes . Dietary Reference Intakes for Calcium, Phosphorous, Magnesium, Vitamin D, and Fluoride. National Academy Press; Washington, DC: 1997. [Google Scholar]
- 12.Nesby-O’Dell S, Scanlon KS, Cogswell ME, Gillespie C, Hollis BW, Looker AC, et al. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: third National Health and Nutrition Examination Survey, 1988-1994. Am J Clin Nutr. 2002;76:187–92. doi: 10.1093/ajcn/76.1.187. [DOI] [PubMed] [Google Scholar]
- 13.Jacques PF, Felson DT, Tucker KL, Mahnken B, Wilson PW, Rosenberg IH, et al. Plasma 25-hydroxyvitamin D and its determinants in an elderly population sample. Am J Clin Nutr. 1997;66:929–36. doi: 10.1093/ajcn/66.4.929. [DOI] [PubMed] [Google Scholar]
- 14.Wang L, Whitlatch LW, Flanagan JN, Holick MF, Chen TC. Vitamin D autocrine system and prostate cancer. Recent results in cancer research Fortschritte der Krebsforschung. 2003;164:223–37. doi: 10.1007/978-3-642-55580-0_16. [DOI] [PubMed] [Google Scholar]
- 15.Giovannucci E, Liu Y, Stampfer MJ, Willett WC. A prospective study of calcium intake and incident and fatal prostate cancer. Cancer Epidemiol Biomarkers Prev. 2006 Feb;15(2):203–10. doi: 10.1158/1055-9965.EPI-05-0586. [DOI] [PubMed] [Google Scholar]
- 16.Miller GJ. Vitamin D and prostate cancer: biologic interactions and clinical potentials. Cancer Metastasis Rev. 1999;17:353–60. doi: 10.1023/a:1006102124548. [DOI] [PubMed] [Google Scholar]
- 17.Johnson CS, Hershberger PA, Trump DL. Vitamin D-related therapies in prostate cancer. Cancer Metastasis Rev. 2002;21:147–58. doi: 10.1023/a:1020836226594. [DOI] [PubMed] [Google Scholar]
- 18.Ahn J, Albanes D, Peters U, Schatzkin A, Lim U, Freedman M, et al. Dairy products, calcium intake, and risk of prostate cancer in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol Biomarkers Prev. 2007 Dec;16(12):2623–30. doi: 10.1158/1055-9965.EPI-07-0601. [DOI] [PubMed] [Google Scholar]
- 19.Adams ND, Gray RW, Lemann J., Jr. The effects of oral CaCO3 loading and dietary calcium deprivation on plasma 1,25-dihydroxyvitamin D concentrations in healthy adults. J Clin Endocrinol Metab. 1979;48:1008–16. doi: 10.1210/jcem-48-6-1008. [DOI] [PubMed] [Google Scholar]
- 20.Gascon-Barre M, D’Amour P, Dufresne L, JP P. Interrelationships between circulating vitamin D metabolites in normocalciuric and hypercalciuric renal stone formers. Ann Nutr Metab. 1985;29:289–96. doi: 10.1159/000176984. [DOI] [PubMed] [Google Scholar]
- 21.Chan JM, Stampfer MJ, Ma J, H GP, Gaziano JM, Giovannucci EL. Dairy products, calcium, and prostate cancer risk in the Physicians’ Health Study. Am J Clin Nutr. 2001;74:549–54. doi: 10.1093/ajcn/74.4.549. [DOI] [PubMed] [Google Scholar]
- 22.Chan JM, Pietinen P, Virtanen M, Malila N, Tangrea J, Albanes D, et al. Diet and prostate cancer risk in a cohort of smokers, with a specific focus on calcium and phosphorous (Finland) Cancer Causes Control. 2000;11:859–67. doi: 10.1023/a:1008947201132. [DOI] [PubMed] [Google Scholar]
- 23.Martini L, Wood RJ. Relative bioavailability of calcium-rich dietary sources in the elderly. Am J Clin Nutr. 2002;76:1345–50. doi: 10.1093/ajcn/76.6.1345. [DOI] [PubMed] [Google Scholar]
- 24.Gallagher JC, Riggs BL, Eisman J, Hamstra A, Arnaud SB, DeLuca HF. Intestinal calcium absorption and serum vitamin D metabolites in normal subjects and osteoporotic patients: effect of age and dietary calcium. J Clin Invest. 1979 Sep;64(3):729–36. doi: 10.1172/JCI109516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ferrari SL, Bonjour JP, Rizzoli R. Fibroblast growth factor-23 relationship to dietary phosphate and renal phosphate handling in healthy young men. The Journal of clinical endocrinology and metabolism. 2005 Mar;90(3):1519–24. doi: 10.1210/jc.2004-1039. [DOI] [PubMed] [Google Scholar]
- 26.Baron JA, Beach M, Wallace K, Grau MV, Sandler RS, Mandel JS, et al. Risk of prostate cancer in a randomized clinical trial of calcium supplementation. Cancer Epidemiol Biomarkers Prev. 2005 Mar;14(3):586–9. doi: 10.1158/1055-9965.EPI-04-0319. [DOI] [PubMed] [Google Scholar]
- 27.Corder EH, Guess HA, Hulka BS, Friedman GD, Sadler M, Vollmer RT, et al. Vitamin D and prostate cancer: a prediagnostic study with stored sera. Cancer Epidemiol Biomarkers Prev. 1993;2:467–72. [PubMed] [Google Scholar]
- 28.Ahonen MH, Tenkanen L, Teppo L, Hakama M, Tuohimaa P. Prostate cancer risk and prediagnostic serum 25-hydroxyvitamin D levels (Finland) Cancer Causes Control. 2000;11:847–52. doi: 10.1023/a:1008923802001. [DOI] [PubMed] [Google Scholar]
- 29.Braun MM, Helzlsouer KJ, Hollis BW, Comstock GW. Prostate cancer and prediagnostic levels of serum vitamin D metabolites (Maryland, United States) Cancer Causes Control. 1995;6:235–9. doi: 10.1007/BF00051795. [DOI] [PubMed] [Google Scholar]
- 30.Gann PH, Ma J, Hennekens CH, Hollis BW, Haddad JG, Stampfer MJ. Circulating vitamin D metabolites in relation to subsequent development of prostate cancer. Cancer Epidemiol Biomarkers Prev. 1996;5:121–6. [PubMed] [Google Scholar]
- 31.Nomura AMY, Stemmermann GN, Lee J, Kolonel LN, Chen TC, Turner A, et al. Serum vitamin D metabolite levels and the subsequent development of prostate cancer (Hawaii, United States) Cancer Causes Control. 1998;9:425–32. doi: 10.1023/a:1008875819232. [DOI] [PubMed] [Google Scholar]
- 32.Zadshir A, Tareen N, Pan D, Norris K, Martins D. The prevalence of hypovitaminosis D among US adults: data from the NHANES III. Ethn Dis. 2005;15(4 Suppl 5):S5-97–101. Autumn. [PubMed] [Google Scholar]
- 33.Chan JM, Giovannucci E, Andersson SO, Yuen J, Adami HO, Wolk A. Dairy products, calcium, phosphorous, vitamin D, and risk of prostate cancer (Sweden) Cancer Causes Control. 1998;9:559–66. doi: 10.1023/a:1008823601897. [DOI] [PubMed] [Google Scholar]
- 34.Giovannucci E, Rimm EB, Wolk A, Ascherio A, Stampfer MJ, Colditz GA, et al. Calcium and fructose intake in relation to risk of prostate cancer. Cancer Res. 1998;58:442–7. [PubMed] [Google Scholar]
- 35.Kristal AR, Cohen JH, Qu P, Stanford JL. Associations of energy, fat, calcium, and vitamin D with prostate cancer risk. Cancer Epidemiol Biomarkers Prev. 2002;11:719–25. [PubMed] [Google Scholar]
- 36.Tseng M, Breslow RA, Graubard BI, Ziegler RG. Dairy, calcium, and vitamin D intakes and prostate cancer risk in the National Health and Nutrition Examination Epidemiologic Follow-up Study cohort. Am J Clin Nutr. 2005 May;81(5):1147–54. doi: 10.1093/ajcn/81.5.1147. [DOI] [PubMed] [Google Scholar]
- 37.Shankar K, Liu X, Singhal R, Chen JR, Nagarajan S, Badger TM, et al. Chronic ethanol consumption leads to disruption of vitamin D3 homeostasis associated with induction of renal 1,25 dihydroxyvitamin D3-24-hydroxylase (CYP24A1) Endocrinology. 2008 Apr;149(4):1748–56. doi: 10.1210/en.2007-0903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Subar AF, Thompson FE, Kipnis V, Midthune D, Hurwitz P, McNutt S, et al. Comparative validation of the Block, Willett, and National Cancer Institute food frequency questionnaires: the Eating at America’s Table Study. Am J Epidemiol. 2001;154:1089–99. doi: 10.1093/aje/154.12.1089. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.