Abstract

A uniquely stable acyl potassium trifluoroborate, potassium (2-phenylacetyl) trifluoroborate, has been synthesized and isolated. In the presence of an activating Lewis acid, this reagent reacts with azides to form amides in good yields.
As a functional class, boronic acids display wide utility in organic synthesis,1 participating in a broad range of reactions including Suzuki–Miyaura cross-coupling2,3 as well as borono-Mannich4 and rhodium-catalyzed 1,4-addition5 reactions. Nevertheless, the empty p-orbital of tricoordinate boronic acids leaves them susceptible to oxidation and protodeboronation and thereby limits their functional group compatibility when carried through multiple synthetic steps, as well as their utility as bench-stable reagents.6 In recent years, these laboratories have focused on the synthesis and reactivity of potassium organotrifluoroborates as an alternative to boronic acids.7 Because of their tetracoordinate nature, trifluoroborates are less susceptible to decomposition, enabling the synthesis of potassium vinyl trifluoroborate,8 as well as alkynyl-9,10 and 2-substituted heteroaryltrifluoroborates,11 the boronic acid derivatives of which are quite often unstable.
We report herein the synthesis of a stable potassium acyltrifluoroborate and its reaction with azides to form amides. Currently, there is only one reported example of a fully characterized and isolated acyl boron species (Figure 1, 1);12 however, no subsequent reactivity for this acyl boron has been described. Acyl boron species have been hypothesized to form transiently, but are subject to rapid rearrangement and oxidation to tertiary alcohols or ketones.13
Figure 1.
Related acyl boron and acyl boron equivalents.
Our synthesis of an acyltrifluoroborate avoids destructive rearrangement by conducting the carbonyl formation simultaneously with that of the trifluoroborate via direct deprotonation of (E)-(2-methoxyvinyl)benzene 5 with tert-butyllithium (route A, Scheme 1), followed by quenching with triisopropyl borate and aqueous KHF2, a method similar to the one employed by Matteson14 in the synthesis of (S)-pinanediol (1-methoxyvinyl)boronate (2). Alternatively, we were able to access the same intermediate enol ether ‘ate’ complex 6 (route B, Scheme 1) via metalation of dimethoxy acetal 8, an approach outlined by Venturello et al.15 in the synthesis of styrylboronic ester 3 (Figure 1). In contrast to the recently reported16 synthesis of 1-trifluoroborato 2,2-difluoro enol ether 4 (Figure 1), treatment of intermediate 6 with aqueous KHF2 led directly to acyltrifluoroborate salt 7, as opposed to a trifluoroborato enol ether.
Scheme 1. Preparation of Acyltrifluoroborate 7.
Disappointedly, acyltrifluoroborate 7 not only persisted as the sole example accessible through this method, but also failed to serve as a viable Suzuki–Miyaura cross-coupling partner in our hands, even under anhydrous conditions with aryl diazonium salt electrophiles. Nevertheless, we remained intrigued by the possibility of a reactive, isolable acyl boron species, and to study the fundamental reactivity of 7 we turned our attention to other known reactions of organotrifluoroborates.
Matteson and coworkers17 have demonstrated that Lewis acid-activated trifluoroborates are viable precursors to intermediate dihaloboranes, which react with azides to form secondary amines. The same protocol applied to acyltrifluoroborates would be expected to provide a novel approach to amides. To pursue this goal, we initiated an investigation to realize this intriguing transformation. Initial studies began with an exploration of various Lewis acids/fluorophiles that were expected to promote the reaction. In our hands, SiCl4, chlorotrimethylsilane, Sc(OTf)3, and BF3•OEt2 were effective in promoting the desired reaction between acyltrifluoroborate 7 and azides, but in poor conversions. In contrast, the use of tetrafluoroboric acid diethyl etherate resulted in reproducible, moderate yields of the desired amides (Table 1).
Table 1. Reaction of Acyltrifluoroborate 7 with Azides (9).
 | |||||
|---|---|---|---|---|---|
| entry | azide | azide (equiv) | time (h) | isolated yield (%)a | |
| 1 | 9a |  |
1 | 4 | 75 |
| 2 | 9b |  |
1 | 1 | 70 |
| 3 | 9c |  |
1 | 1 | 67 |
| 4 | 9d |  |
1 | 3 | 74 |
| 5 | 9e | 2 | 2 | 68 | |
| 6 | 9f | 2 | 1 | 72 | |
| 7 | 9g |  |
2 | 1 | <5 |
| 8 | 9h | 1.5 | 2 | <10 | |
Reaction conditions: 7 (0.3 mmol), 9 (0.3 mmol), HBF4·OEt2 (2 equiv), CH3CN (1.5 mL), 0 °C to rt.
Although the reaction conditions were tolerant of simple alkyl azides (entries 1–4), the method unfortunately lacks broad substrate scope. A few functional groups, such as esters (entry 5) and nitriles (entry 6) were tolerated, but aryl azides (entry 7) and azides containing alkenes (entry 8) were so badly decomposed that only trace amounts of the corresponding amides could be isolated. Additionally, no acylsulfonamide products could be isolated from the reaction of trifluoroborate salt 7 and sulfonyl azides.
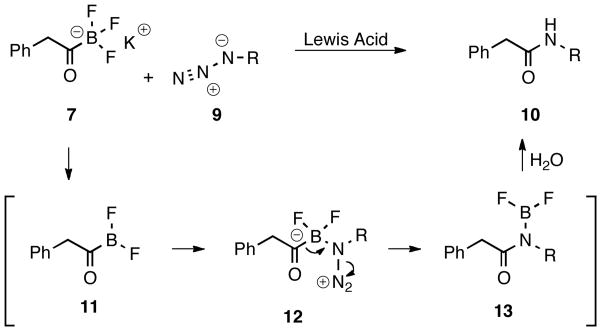
Similar to the reaction of alkyldichloroboranes and azides reported by Brown et al.,18 the reaction may proceed through reversible coordination of the azide 9 with the dihaloborane 11. Concurrent migration of the acyl group from boron to nitrogen and loss of dinitrogen would afford intermediate 13, hydrolysis of which would produce amides of type 10.
In summary, we have developed a bench-stable acyltrifluoroborate salt 7 that is capable of reacting as a stable acyl anion equivalent. Alkyl azides were compatible with the Lewis acid activated acyl boron species; however, aryl, sulfonyl, and alkenyl-containing azides were not.
Experimental Section
Synthesis of Acyltrifluoroborate 7
Route A: To a stirred solution of β-methoxystyrene (3 mL, 22.4 mmol, 3 equiv) in THF (25 mL) at −78 °C was added tert-butyllithium (6.6 mL, 1.7 M in pentane, 11.2 mmol, 1.5 equiv) dropwise. The reaction mixture was allowed to stir for 80 min at −78 °C, at which point it was warmed to 0 °C and stirred for an additional 45 min. After cooling the reaction mixture to −78 °C, triisopropyl borate (1.73 mL, 7.5 mmol) was added dropwise, and the reaction was allowed to stir at −78 °C for 30 min before being warmed to room temperature. Stirring was continued for 2 h, at which point saturated aq KHF2 (17 mL, ∼4.5 M, 76 mmol) was slowly added. The reaction mixture was stirred vigorously overnight, and the solvents were removed in vacuo. The resulting mixture of solids was dried under high vacuum overnight, then subjected to extraction using acetone (3 × 20 mL). The acetone extracts were concentrated in vacuo, and Et2O (50 mL) was added to precipitate the trifluoroborate product. Filtration gave 7 (0.53 g, 31%) as white crystals: mp > 250 °C; νmax(KBr)/cm-1 3400, 1661, 1319, 1001; 1H NMR (300 MHz, acetone-d6) δ 7.15–7.26 (m, 2H), 7.04–7.14 (m, 3H), 3.71 (s, 2H); 13C NMR (125 MHz, acetone-d6) δ 138.0, 130.9, 128.4, 126.1, 51.6; 19F NMR (470 MHz, acetone-d6) δ −151.32; 11B NMR (128 MHz, acetone-d6) δ −1.88; HRMS (ESI-TOF) calcd. for C8H7OBF3− [M–K]− 187.0542, found 187.0540.
Representative Amide Synthesis: N-Benzyl-2-phenyl-acetamide (10a)
An oven-dried 2–5 mL microwave vial was charged with trifluoroborate salt 7 (0.068 g, 0.30 mmol), capped with a rubber septum, and evacuated. After backfilling with N2, this process was repeated twice more. To the microwave vial was added anhydrous CH3CN (1.5 mL) and benzyl azide (9a) (0.040 g, 0.3 mmol). The reaction mixture was cooled in an ice/water bath before slowly adding tetrafluoroboric acid diethyl etherate (0.082 mL, 1.19 g/mL, 0.6 mmol) via a PTFE needle. The reaction mixture was allowed to warm to room temperature and stirred for 4 h. The reaction was quenched with H2O (1.5 mL) and the aqueous layer was extracted with EtOAc (2×2 mL). The combined organic layers were washed with brine (1 mL), dried (Na2SO4), filtered, and concentrated in vacuo. Purification by flash column chromatography, eluting with 7:3 hexanes:EtOAc with 0.1% Et3N afforded 10a (0.051 g, 75%) as a white solid: mp 118–119 °C; (lit.19 119 °C); Rf = 0.2 (silica gel, hexanes:EtOAc 7:3); 1H NMR (500 MHz, CDCl3) δ 7.31 (t, J = 7.2 Hz, 2H), 7.28–7.18 (m, 6H), 7.14 (d, J = 7.0, 2H) 5.71 (bs, 1H), 4.37 (d, J = 5.8, 2H), 3.58 (s, 2H); 13C NMR (125 MHz, CDCl3) δ 171.0, 138.3, 135.0, 129.6, 129.2, 128.8, 127.63, 127.55, 127.50, 43.9, 43.7; HRMS (ESI-TOF): calcd for C15H16NO+ [M+H]+: 226.1232; found: 226.1238.
Supplementary Material
Scheme 2. Plausible Reaction Mechanism.
Acknowledgments
This research was supported by a National Priorities Research Program (NPRP) grant from the Qatar National Research Fund (Grant no. 08-035-1-008), the NIH (R01 GM-081376), and Merck Research Laboratories. Dr. Rakesh Kohli (University of Pennsylvania) is acknowledged for obtaining HRMS data.
Footnotes
Supporting Information Available: Experimental procedures, characterization data, and NMR spectra for all compounds. This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Hall DG. Boronic Acids: Preparation and Applications in Organic Synthesis and Medicine. Wiley-VCH; New York: 2005. [Google Scholar]
- 2.Miyaura N. In: Metal-Catalyzed Cross-Coupling Reactions. Diederich F, de Meijere A, editors. Chapter 2 Wiley-VCH; New York: 2004. [Google Scholar]
- 3.Miyaura N, Suzuki A. Chem Rev. 1995;95:2457–2483. [Google Scholar]
- 4.McReynolds MD, Hanson PR. Chemtracts. 2001;14:796–801. [Google Scholar]
- 5.Hayashi T, Yamasaki K. Chem Rev. 2003;103:2829–2844. doi: 10.1021/cr020022z. [DOI] [PubMed] [Google Scholar]
- 6.Thomas SE. Organic Synthesis: The Roles of Boron and Silicon. Oxford University Press; New York: 1991. pp. 1–46. [Google Scholar]
- 7.Molander GA, Ellis NM. Acc Chem Res. 2007;40:275–286. doi: 10.1021/ar050199q. [DOI] [PubMed] [Google Scholar]
- 8.Darses S, Michaud G, Genet JP. Tetrahedron Lett. 1998;39:5045–5048. [Google Scholar]
- 9.Molander GA, Katona BW, Machfouhi F. J Org Chem. 2002;67:8416–8423. doi: 10.1021/jo0262356. [DOI] [PubMed] [Google Scholar]
- 10.Darses S, Michaud G, Genet JP. Eur J Org Chem. 1999:1875–1883. [Google Scholar]
- 11.Molander GA, Canturk B, Kennedy LE. J Org Chem. 2009;74:973–980. doi: 10.1021/jo802590b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yamashita M, Suzuki Y, Segawa Y, Nozaki K. J Am Chem Soc. 2007;129:9570–9571. doi: 10.1021/ja073037t. [DOI] [PubMed] [Google Scholar]
- 13.Kabalka GW, Gotsic JT, Pace RD, Li NS. Organometallics. 1994;13:5163–5165. [Google Scholar]
- 14.Matteson DS, Beedle EC. Heteroat Chem. 1990;1:135–140. [Google Scholar]
- 15.Tivola PB, Deagostino A, Prandi C, Venturello P. Org Lett. 2002;4:1275–1277. doi: 10.1021/ol0255817. [DOI] [PubMed] [Google Scholar]
- 16.Katz JD, Lapointe BT, Dinsmore CJ. J Org Chem. 2009;74:8866–8869. doi: 10.1021/jo901789b. [DOI] [PubMed] [Google Scholar]
- 17.Matteson DS, Kim GY. Org Lett. 2002;4:2153–2155. doi: 10.1021/ol025973d. [DOI] [PubMed] [Google Scholar]
- 18.Brown HC, Midland MM, Levy AB. J Am Chem Soc. 1973;95:2394–2396. [Google Scholar]
- 19.Sabot C, Kumar KA, Meunier S, Mioskowski C. Tetrahedron Lett. 2007;48:3863–3866. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.