Abstract
Cyclooxygenase-2 (COX-2) enzyme has been involved in the tumorigenesis and in the progression of colorectal cancer (CRC). The use of traditional nonsteroidal anti-inflammatory drugs (NSAIDs) or selective COX-2 inhibitors has been proposed for the prevention and the treatment of this relevant neoplastic disease. In the light of an innovative alternative to these pharmacological approaches, we review here the possible strategies to achieve a strong and selective inhibition of COX-2 enzyme by using the mechanism of RNA Interference (RNAi) targeted against its mRNA. Anti-COX-2 siRNA molecules (siCOX-2) can be generated in CRC cells from short hairpin RNA (shRNA) precursors, delivered in vitro by a retroviral expression system, and induce a significant and stable silencing of overexpressed COX-2 in human colon cancer cells. As a safer alternative to viral approach, nonpathogenic bacteria (E. coli) can be engineered to invade eukaryotic cells and to generate siCOX-2 molecules in cancer cells. Moreover, the involvement of miRNAs in COX-2 posttranscriptional regulation opens up the possibility to exploit an endogenous silencing mechanism to knockdown overexpressed COX-2. Thus, these recent strategies disclose new challenging perspectives for the development of clinically compatible siRNA or miRNA capable of selectively inhibiting COX-2 enzyme.
1. Introduction
1.1. COX-2 and Cancer
Several studies have supported the involvement of prostanoids in the pathogenesis of cancer. In vitro studies have demonstrated that growth factors, tumor promoters, and oncogenes induce prostanoids synthesis [1]. In vivo, the metabolism of arachidonic acid, via the COX pathway, has been found to be enhanced in various human tumors and it is now widely accepted that this is due to the induction of COX-2 enzyme [2]. A number of theories have been proposed to explain the role of tumor-derived prostanoids in promotion of angiogenesis, induction of tumor cell proliferation, suppression of immune response, and protection against apoptosis [3–5]. The expression of COX-1 and -2 in tumor tissues biopsies collected from various cancers has been analyzed. In particular, early studies by Eberhart et al. [6] and Sano et al. [7] showed that COX-2 is overexpressed in 80% of colorectal cancer tissues. In these transformed tissues, COX-2 enzyme results to be overexpressed in epithelial cells, inflammatory cells, as well as in stromal cells. In contrast, COX-2 expression is very low in adjacent normal tissues. COX-1 isoenzyme is expressed at the same basal level in both normal and tumor tissues. These findings have been confirmed analyzing many tumors including pancreas, skin, gastric, bladder, lung, head, and neck cancers [8], suggesting that COX-2, but not COX-1, may play a pivotal role in tumor formation and growth. More recent studies confirmed that the upregulated expression of COX-2, probably induced by carcinogenic stimuli or other tumor promoters, is an important contributor to tumorigenesis [9]. It is now clear that the tumorigenesis involves COX-2 overexpression in most cases, even if the molecular mechanisms responsible for this overexpression have not been completely understood. Different in vitro studies support the idea that COX-2 overexpression inhibits apoptosis and promotes tumor angiogenesis [2, 9, 10]. In fact, COX-2 overexpression in tissues seems to be crucial to favor the development of new vasculature supporting tumor growth and metastatization.
Following the development of animal models of tumorigenesis it has been demonstrated that NSAIDs strongly inhibit colon and breast tumors in rodents [11], further suggesting that products of the COX pathways may participate in carcinogen-induced tumorigenesis. The cancer chemopreventive activity of NSAIDs has been supported by epidemiological studies performed on humans taking aspirin and other NSAIDs on a regular basis. These studies demonstrate that incidence of various cancers, including colon, intestinal, gastric, breast, and bladder cancers, is reduced up to 40–50% [8]. Aspirin, which is a COX-1 and COX-2 inhibitor, has been shown to be moderately effective in preventing sporadic colorectal adenomas in patients with a familiar history of these tumors [12]. Epidemiological studies suggest that aspirin, especially if used in high doses for more than 10 years, is effective in reducing the incidence of colonic adenoma and CRC, even if careful consideration should be devoted to the possible harms of such a practice [13]. Celecoxib and rofecoxib, COX-2 selective drugs, have also been shown to reduce adenomas' incidence and to induce tumor regression in patients with familial polyposis [14, 15]. Duodenal adenomas, which are otherwise untreatable, show some reduction by treatment with celecoxib [14]. The antineoplastic activity of NSAIDs has been extensively reviewed elsewhere [16]. Recent studies demonstrate that COX-2 blocking agents have strong potential for the chemoprevention of breast, prostate, colon, and lung cancers [17] and that multifactorial molecular events may be involved in such activity. Among them, the induction of apoptosis appears to be an essential factor in explaining the ability of these drugs to cause tumor regression [18]. Clinical trials have confirmed that NSAIDs, especially selective COX-2 inhibitors (coxibs), effectively prevent colorectal adenoma formation. On the basis of available data, COX-2 inhibitors are likely to be highly effective cancer chemopreventive agents. Yet, they also have substantial side-effects that currently limit their routine use [19]. The placebo-controlled trials APC (Adenoma Prevention with Celecoxib), PreSAP (Prevention of Spontaneous Adenomatous Polyps), and APPROVe (Adenomatous Polyp Prevention on Vioxx) of celecoxib and rofecoxib were stopped early because interim safety results indicated an increased cardiovascular risk in participants receiving active medication [20–22]. Since the COX-2 isoform is one of the major sources of endothelium-derived prostacyclin (PGI2) under physiological conditions, it has been hypothesized that selective blockade of COX-2 might impair endothelial function and predispose patients to cardiovascular disease [23, 24]. Moreover, the cardiovascular toxicity of rofecoxib could be increased by its capability to directly inhibit prostacyclin synthase activity, as demonstrated by our group [25].
2. COX-2 Silencing Mediated by siRNA
In the last decade, RNA Interference (RNAi) has rapidly become an innovative and elective tool for studying genes function. Based on the works of Fire and Mello in 1998 on Caenorhabditis elegans [26], Hammond et al. and Zamore et al. in 2000 on Drosophila cells extracts [27, 28], and Tuschl and colleagues on mammalian cells [29], small interfering RNAs (siRNAs), 21–23 nt dsRNAs molecules with 2-3 nt overhangs on their 3′-ends, were defined as the effectors of the RNAi pathway. They are capable of binding to homologous target mRNAs leading to cleavage of the transcript near the centre of the pairing sequence. To date, the RNAi pathway has been almost totally defined. Synthetic mature siRNAs can be transfected into cells or generated both in transient and stable manner from longer dsRNA precursors or from ssRNA molecules containing a complementary dsRNA domain called short hairpin RNAs (shRNAs). Active siRNAs are processed into cytoplasm by the RNase III endoribonuclease Dicer [30] and mRNA cleavage is mediated by a ribonucleoprotein complex, known as the RNA-induced silencing complex (RISC), after incorporation of the “guide” strand of the siRNA duplex. RISC contains one of the eight known Argonaute proteins in humans, Ago2, characterized by an RNA-binding domain (PAZ domain) and an RNase H-like domain (PIWI domain) [31].
RNAi technology has been successfully used to silence COX-2 protein in different in vitro models (Table 1). The use of innovative RNAi-based techniques has enabled researchers to better study the molecular and phenotypical loss of function of Cox-2 gene by performing experiments based on a strong COX-2 silencing deprived of aspecific effects. In 2003, Denkert and collaborators tested for the first time the efficacy of an anti-COX-2 siRNA (siCOX-2) on OVCAR-3 cells derived from human ovarian carcinoma [32]. A comparison with the COX-2 inhibitory drug NS-398 highlighted a different effect of siCOX-2 due to its highly specific mechanism of action. Even though COX-2 protein levels resulted significantly reduced in both cases after Il-1β stimulation, only NS-398 treatment induced a G0/G1 cell cycle arrest in OVCAR-3 cells. This effect could be due to the action of NS-398 on other cellular targets involved in cell proliferation, confirming the lack of specificity of NSAIDs in COX-2 blocking. Recent works by Charames and Bapat [33] and Kobayashi et al. [34] showed an efficient COX-2 knockdown mediated by siRNAs in HT-29 human colon cancer cells and bovine Cumulus-Granulosa (CG) cells, respectively. Based on their results on cell apoptosis [33], Charames and Bapat have confirmed a COX-2-independent mechanism of action of NSAIDs, previously described by several research groups [35–38]. From these studies, it results clear that RNAi, compared with NSAIDs, is a more powerful and selective tool for studying in vitro the functional role of COX-2.
Table 1.
COX-2 silencing mediated by RNAi.
| Study | Model | RNAi-silencing | Effects | Ref |
|---|---|---|---|---|
| Denkert et al. 2003 | OVCAR-3 cells (human ovarian carcinoma) | siCOX-2 (short term expression) | - COX-2 protein silencing | |
| - Reduction of PGE2 levels | [32] | |||
| - No effects on cell proliferation | ||||
| Strillacci et al. 2006 | HUVECs (human umbilical vein endothelial cells) | siCOX-2 (short term expression) | - COX-2 protein silencing | |
| - Reduction of 6-keto- PGF2α levels | ||||
| - Reduction of capillary-like tubular | [39] | |||
| structures on 3D collagen gel | ||||
| - No effects on interferon system | ||||
| Strillacci et al. 2006 | HT-29 cells (human colon carcinoma) | shCOX-2 (stable expression) | - COX-2 protein and mRNA silencing | |
| - Reduction of PGE2 levels | ||||
| - No effects on interferon system | [39] | |||
| - No effects on cell proliferation | ||||
| - Impairment of malignant behavior | ||||
| Charames and Bapat 2006 | HT-29 cells (human colon carcinoma) | siCOX-2 (short term expression) | - COX-2 protein and mRNA silencing | [33] |
| - No effects on cell apoptosis | ||||
| Kobayashi et al. 2007 | CG cells (bovine Cumulus Granulosa) | siCOX-2 (short-term expression) | - COX-2 mRNA silencing | [34] |
| - Reduction of PGF2α levels | ||||
| Wang et al. 2008 | Hep-2 cells (human laryngeal carcinoma) | shCOX-2 (stable expression) | - COX-2 protein and mRNA silencing | |
| - Inhibition of proliferation | ||||
| - Impairment of malignant behavior | [40] | |||
| - Inhibition of in vivo growth | ||||
| - Enhanced chemosensitivity in vitro and in vivo | ||||
| Sansone et al. 2008 | HT-29 cells (human colon carcinoma) | shCOX-2 (stable expression) | - COX-2 protein and mRNA silencing | |
| - Inhibition of Erk phosphorilation | ||||
| - Inhibition of CA-IX expression | ||||
| - Inhibition of cell invasion | [41] | |||
| - Inhibition of MMP-2 activation | ||||
| - No effects on cell death | ||||
| - Inhibition of hypoxic survival | ||||
However, RNAi-mediated COX-2 silencing proved to be highly effective using anti-COX-2 shRNAs (shCOX-2). In 2006, our group published a paper in which an in vitro strategy to stably knockdown COX-2 in colon cancer cells (HT-29) was described [39]. Firstly, we tested different sequences of siCOX-2 in HUVE (human umbilical vein endothelial) cells. Among these, one siCOX-2 resulted to be more effective in silencing COX-2 protein after PMA transcriptional induction and led to a reduction of PGI2 levels and to the impairment of the ability of HUVE cells to organize capillary-like tubular structures in 3D gel. Furthermore, the active siCOX-2 sequence (5′-aactgctcaacaccggaattt-3′) was used to design a shCOX-2 in order to silence COX-2 in HT-29 colon cancer cells in a long-lasting manner. The stable expression of shCOX-2 in HT-29 (HT-29shCOX-2) induced a strong COX-2 silencing at protein, mRNA, and product (PGE2) levels, devoid of toxic effects (no activation of the interferon system). Phenotypically, COX-2 stable knockdown did not affect cell proliferation and cell cycle distribution but it strongly impaired the malignant behavior of HT-29 colon cancer cells in vitro. In fact, the invasiveness of HT-29shCOX-2 cells was reduced, as well as their ability to form colonies in soft-agar. This novel approach for COX-2 silencing is very promising and its effectiveness has been further confirmed by the work of Wang and collaborators [40] who performed a stable silencing of COX-2 protein mediated by shRNA in human laryngeal carcinoma Hep-2 cells. In this model, COX-2 silencing induced a reduction of proliferation and invasiveness, coupled with increased apoptosis. Moreover, a reduced tumorigenesis was demonstrated with Hep-2shCOX-2 xenografts in nude mice. Finally, our HT-29shCOX-2 model was successfully used to better elucidate the role of COX-2 in the hypoxic environment of colon cancer. In particular, we recently demonstrated an important interplay between COX-2 and carbonic anhydrase-IX (CA-IX, an enzyme controlling cellular pH) that promotes the hypoxic survival and invasiveness of colon cancer cells [41].
3. Transkingdom RNAi and Enhanced COX-2 Silencing
Several years ago, it was demonstrated that systemic gene silencing occurs in the nematode Caenorhabditis elegans after ingestion of Escherichia coli engineered to produce interfering RNAs, thus suggesting that RNAi-mediated gene silencing between species or kingdoms might be possible [42, 43]. Bacteria engineered to produce shRNAs can induce transkingdom RNAi (tkRNAi) in vitro and in vivo also in mammalian systems. A successful transfer of shRNAs into mammalian cells can be obtained using nonpathogenic E. coli transformed with a plasmid containing an expression cassette for shRNA and Inv/HlyA genes. In particular, Inv and HlyA encode the two bacterial factors (invasin and listeriolysin-O, resp.) responsible for the efficient transfer of shRNA from bacteria to mammalian cells. In 2006, Xiang et al. applied for the first time this new technology to silence β-1 catenin gene (CTNNB1) in human cells [44]. Firstly, they assessed the efficacy of tkRNAi against β-catenin in vitro on SW-480 colon carcinoma cell line. Then, β-catenin silencing tkRNAi-mediated was achieved also in vivo, in normal mouse intestinal epithelium and in xenografts of human colon cancer cells in mice. Based on these results, tkRNAi in mammals could represent a powerful and innovative strategy for functional genomics studies and for the development of clinically compatible RNAi-based therapies. In 2007, Cequent Pharmaceuticals (Cambridge, MA, USA) started to develop and validate new therapies for familial adenomatous polyposis (FAP) and CEQ501, the first tkRNAi-based drug, is currently in advanced preclinical trials [45].
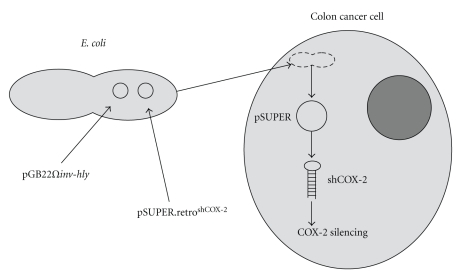
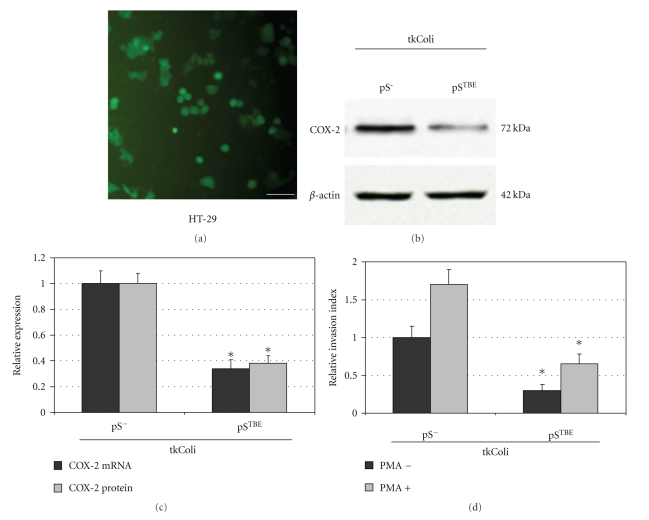
In our laboratory, we have developed a tkRNAi-based strategy to efficaciously silence COX-2 in colon cancer cells. We tried to combine our knowledge on COX-2 knockdown mediated by RNAi with this transkingdom strategy (Figure 1). Infection of HCA-7 colon cancer cells with tkColishCOX-2 determined a significant reduction of COX-2 mRNA and protein levels in this colon cancer cell lines (Figure 2). Moreover, COX-2 silencing mediated by tkRNAi induced a significant decrease in cell invasiveness in HT-29 and HCA-7 cell lines (Figure 2). We consider these data to be very promising and, as a consequence, we are trying to improve this strategy in order to obtain an enhanced and highly specific COX-2 silencing in colon cancer cells, in the light of compatible in vivo applications.
Figure 1.
COX-2 silencing mediated by transkingdom RNAi. The cotransformation of E. coli with both plasmids pGB2Ωinv-hly and pSUPER.retroshCOX-2 enables bacteria to induce the transkingdom RNAi phenomenon in colon cancer cells. Following the expression of the bacterial proteins invasin and listeriolysin-O, engineered E. coli strains are able to permeate human cells and release the plasmid DNA content. Anti-COX-2 shRNAs are then transcribed by the cellular machinery, resulting in an efficient COX-2 silencing.
Figure 2.
Invasive tkColi infect CRC HT-29 cells and promote high COX-2 silencing associated with a reduced invasive behavior. E. coli was cotransformed with pGB2-Ω-inv-hly plasmid and pSUPER.retro vectors to obtain E. coli invasive strains carrying the shCOX-2 expression vector tkColi-pSTBE (in which shCOX-2 expression is controlled by TBE promoter carrying Tcf Binding Elements). The negative control was tkColi-pS-(not expressing shCOX2 but containing the original empty pSUPER.retro vector). GFP protein expression (a) was used to evaluate the efficiency of tkColi infection of HT-29 cells (bar = 30 μm). 72 hours after the infection, the efficiency of infection was higher than 75%. The expressions of COX-2 protein and COX-2 mRNA were analyzed in HT-29 cells, 72 hours after tkColi infection, by Western blot and real-time PCR (c). COX-2 protein and COX-2 mRNA expressions were normalized against β-actin protein and GUSB (β-glucuronidase) mRNA levels, respectively. Relative expression of COX-2 protein and COX-2 mRNA refers to tkColi-pS-infected sample. The invasive behavior of tkColi infected CRC HT-29 cells was evaluated by using Boyden chambers and 8-μm polycarbonate membranes coated with Matrigel. Samples were tested in the absence (dark bars) and in the presence (light bars) of PMA 40 nM. Relative invasion index refers to HT-29 cells infected with tkColi-pS-, not treated with PMA. Data reported in (b)–(d) represent the mean ± SEM of three independent experiments; *P < .01.
4. Posttranscriptional Cox-2 RegulationMediated by MicroRNAs
MicroRNAs (miRNAs) have been found to be strongly implicated in the control of gene expression and it is believed that up to 30% of human genes are regulated by miRNAs [46]. To date, hundreds of miRNAs have been isolated in mammalians and their sequences are listed in the official miRNA database (miRBase, http://microrna.sanger.ac.uk/index.shtml) [47]. Mature miRNA molecules (RNA duplexes of 19–24 nucleotides length with 2-nt overhangs in 3′-ends) are produced by the cellular machinery after the enzymatic cleavage of longer precursors (pri- and pre-miRNAs) [48, 49]. Following the incorporation into the RNA-induced silencing complex (RISC), the pairing of miRNAs (as single-strand molecules) with target mRNAs carrying a partially complementary sequence in the 3′-untranslated region (3′-UTR) causes the translational repression and/or degradation of the messengers, resulting in the silencing of target genes [50].
miRNAs can regulate cellular genes involved in proliferation, in differentiation, or in apoptosis, and alterations of their expression have been found in various human tumors [51], including colon cancer [52]. In fact, data from recent literature clearly show that disturbances of miRNAs expression levels have detrimental effects on cell physiology and may be directly implicated in carcinogenic processes. These miRNAs, whose mutation or misexpression correlates with various human cancers, are defined “oncomiRs” and they can function as tumor suppressors or oncogenes [53]. Examples of oncomiRs are miRNAs from the miR-17-92 family, miR-155 (overexpressed in chronic lymphocytic leukemia, CLL), miR-106a, miR-21, miR-221, miR-372, miR-373, and many others. MiR-15 and miR-16, let-7 family members, miR-143, and miR-145 have been defined tumor suppressor miRNAs since they are downregulated in some malignancies, including colon cancer [54, 55].
Many intracellular pathways contribute to the induction of COX-2 protein expression, both at transcriptional (see below) and post-transcriptional level. With regards to COX-2 post-transcriptional regulation, it has been widely demonstrated that COX-2 mRNA shows an increased stability due to a mechanism that involves AU-rich regions in its 3′-UTR [56]. Moreover, novel data from literature have shown a direct link between COX-2 expression and miRNA-mediated translational silencing (Table 2). Dey and collaborators firstly described an inverse relationship between COX-2 and miR-101a/miR-199a expression in mice. In particular, they have shown that low levels of miR-101a and miR-199a associate with high COX-2 expression during mouse embryo implantation [57] and during mouse endometrial carcinogenesis [58]. Since COX-2 overexpression is involved in human CRC tumorigenesis, miR-101 and/or miR-199a could act as tumor suppressor miRNAs in CRC. This hypothesis has been confirmed in our laboratory. We recently reported the inverse correlation between COX-2 and miR-101 expression in colon cancer cell lines and we demonstrated in vitro the direct inhibition of COX-2 mRNA translation mediated by miR-101. This correlation was supported also by data collected ex vivo, in which colon cancer tissues and liver metastases derived from CRC patients were analyzed [59]. These findings provide a novel molecular insight in the modulation of COX-2 at post-transcriptional level by miR-101 and strengthen the thesis that miRNAs are highly implicated in the control of gene expression. An impairment of miR-101 levels could represent one of the leading causes of COX-2 overexpression in colon cancer cells and, in our opinion, a novel CRC therapy could be based on COX-2 silencing mediated by miR-101 molecule.
Table 2.
Anti-COX-2 miRNAs.
| Study | miRNAs | Model | Function | Ref |
|---|---|---|---|---|
| Chakrabarty et al. 2007 | mmu-miR 199a* mmu-miR-101a | mouse | - Correlate inversely with COX-2 protein | [57] |
| during embryo implantation | ||||
| - Regulate COX-2 protein expression | ||||
| Daikoku et al. 2008 | mmu-miR-199a* mmu-miR-101a | mouse | - Correlate inversely with COX-2 protein | [58] |
| in endometrial cancer cells | ||||
| Strillacci et al. 2008 | hsa-miR-101 | homo | - Correlates inversely with COX-2 protein | [59] |
| in colon cancer cells | ||||
| - Regulates COX-2 protein expression | ||||
| (translational repression) | ||||
| Shanmugam et al. 2008 | hsa-miR-16 | homo | - Promotes COX-2 mRNA degradation in | [60] |
| THP-1 monocytic cells | ||||
Finally, it has been shown that also miR-16 plays a role in COX-2 mRNA destabilization and promotes its degradation in human THP-1 monocytic cells [60].
5. Transcriptional-Based COX-2 Inhibition
The control of Cox-2 gene transcription can be mediated by various transcription factors such as NF-κB [61], C/EBPβ [62], CREB [63], NFAT [64], AP-1 [65], and PPAR [66], and it has been demostrated that also HIF-1α factor induces COX-2 overexpression in hypoxic condition [67]. A transcriptional-based COX-2 inhibition could be developed by using selective inhibitors of these transcriptional factors. In particular, C/EBPβ seems to have a pivotal role in COX-2 transcriptional activation by proinflammatory mediators [62, 68, 69] and in cancer cells [70, 71]. Moreover, it has been demonstrated that salicylate at pharmacological concentration inhibits COX-2 expression (50%) by blocking C/EBPβ phosphorylation and activation via p90 ribosomal S6 kinase 1/2 (RSK1/2) [72]. Based on these pieces of evidence, Wu and colleagues have proposed that the inhibition of RSK1/2 or C/EBPβ expression by specific siRNAs and/or the inhibition of RSK1/2 activity by pharmacological approaches may have therapeutic potential in reducing COX-2 overexpression in human pathologies (e.g., inflammation, cancer) without affecting the physiological function of COX-2 enzyme. However, transcriptional-based COX-2 inhibition could lack specificity since transcriptional factors are involved in the control of a wide number of genes and RSK1/2 is involved in phosphorylation of other proteins that may have a role in cell physiology [73].
6. Conclusion
Considering the recent literature regarding the application of RNAi-based strategies to modulate gene expression and the study of miRNAs-mediated COX-2 silencing, new COX-2 selective inhibitors based on siRNA or miRNA molecules could be developed. In particular, we suggest that anti-COX-2 siRNAs (siCOX-2) and/or miRNAs (miR-101, miR-199a) may represent innovative tools for COX-2 silencing, especially in CRC therapy. To date, selective COX-2 inhibitors (coxibs) have been tested for CRC treatment but, despite the efficacy in COX-2 blockade, an increased cardiovascular risk has been observed in chronically treated patients. In order to prevent adverse side-effects, tissue-specific expression, and targeting of siCOX-2, miR-101 or miR-199a could lead to a strong and highly specific COX-2 silencing deprived of secondary effects on other targets (e.g., PGI2 synthase) or other tissues (e.g., cardiovascular system).
Many research groups and companies are trying to develop completely new strategies based on miRNAs for an in vivo therapeutic application. Even though miRNAs expression patterns in human cells are not completely understood, the possibility of their modulation represents an exciting challenge for targeting COX-2 activity in vivo. However, clinical trials based on therapeutic small RNAs are still far. The efficiency of siRNAs and miRNAs delivery into target cells, using a nonviral delivery system, is still inadequate. To overcome these barriers, a wide number of modified small RNAs and functional carriers have been developed as an alternative strategy to viral vectors which cannot be considered safe vehicles for therapeutic application in humans. In vivo, the efficacy of delivered siRNAs depends on different factors such as pharmacokinetics, resistance to exonuclease digestion, or maintenance of duplexes integrity. Locked nucleic acids (LNAs), siRNAs with a partial phosphorothioate backbone, or siRNAs with the inclusion of 2′-O-methyl dinucleotides at the 3′-end of the antisense strand have shown enhanced stability. Improvements on the pharmacodynamics of siRNAs were obtained as well, using conjugated-siRNAs (e.g., cholesterol-conjugated) [74] or protecting siRNAs from the extracellular environment with polymer- or lipid-based delivery systems (e.g., cationic polyethylenimines or lipid-polyethylene glycol mixtures) [75, 76]. Cationic liposome-mediated delivery of anti-COX-2 siRNA was recently used in vivo [77] and resulted in an efficient downregulation of COX-2 in tumor cells. Furthermore, various types of synthetic carrier systems for small RNAs delivery have been developed to target tumor tissues preferentially through the blood stream, such as polyplexes [78]. In our laboratories, biocompatible polymeric micelles have shown to be nontoxic carriers capable to efficiently deliver small RNA molecules into mammalian cells cytosol (Benaglia M. and Spisni E, Ms. Submitted). The possibility to modify their shell, in order to obtain a more selective cellular targeting, has been documented by many authors [77, 79], opening up the perspective to deliver in vivo small RNA molecules in a specific and efficient manner.
Nonpathogenic bacteria, engineered to induce tkRNAi against COX-2 in CRC cells (tkColishCOX-2), could represent an innovative and potent strategy to achieve a strong COX-2 silencing directly in the intestinal mucosa of the patients affected by CRC, with a relatively high specificity associated with low systemic effects. Moreover, this approach directed against other cellular targets, such as tumor necrosis factor alpha (TNFα), could be also very promising for the treatment of other inflammatory bowel diseases (e.g., ulcerative colitis, Crohn's disease).
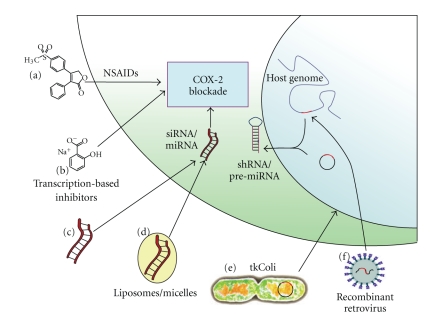
Important remarks could be made about the use of siRNAs instead of miRNAs for an anti-COX-2 therapeutic application. In general, both siRNAs and miRNAs can induce effective gene silencing by a similar mechanism of action [80] and the effective delivery of their mature molecules can be improved by using the same techniques. However, siRNAs and miRNAs have a different specificity in silencing genes and they can induce a different toxicity when transiently or constitutively expressed directly in mammalian cells. We cannot completely exclude that siCOX-2 could generate off-target effects in treated cells, silencing other mRNAs that have partial complementarity [81]. Similarily, COX-2 inhibition mediated by miR-101 or miR-199a could interfere with the expression of other target genes [82–85]. Surely, an miR-101/199a control of COX-2 expression could represent a more suitable approach for the treatment of human pathologies characterized by both COX-2 overexpression and miR-101 downregulation (e.g., colon cancer, endometrial serous adenocarcinoma) [59, 86]. Even though an shRNA/siRNA-mediated gene silencing could result more effective [87], a constitutive expression of shRNAs could lead to a lower specificity of action and a higher level of toxicity. This may be due to the saturation of the cellular machinery deputed to endogenous miRNAs processing and exportation [88] causing the induction of nonsequence-specific silencing pathway (e.g., interferon system activation) which is not observed when using miRNAs [89, 90]. In the light of this evidence, it is likely that miRNA-based strategies for COX-2 inhibition may be a more appropriate alternative for the treatment of human pathologies associated to COX-2 overexpression. These different strategies for COX-2 blockade are summarized in Figure 3.
Figure 3.
The different strategies for COX-2 blockade. (a) COX-2 selective NSAIDs (e.g., celecoxib, rofecoxib, etoricoxib) represent to date the only drugs marketed for COX-2 selective inhibition, even if rofecoxib has been withdrawn due to its toxicity; (b) inhibitors of transcriptional factors expression and/or activity (e.g., C/EBPβ transactivator) may block COX-2 overexpression associated to inflammation or cancer; (c) anti-COX-2 siRNAs (siCOX-2) or miRNAs (e.g., miR-101) with structural modifications that improve their stability may block COX-2 expression after intravenous administration; (d) polymer- or lipid-based delivery systems protect siRNAs or miRNA from the extracellular environment and, modifying their shell, a more selective cellular targeting can be obtained; (e) nonpathogenic bacteria (E. coli) can be engineered in order to invade target cells and induce RNAi against COX-2 (transkingdom RNAi, tkRNAi) by releasing DNA plasmids that express siCOX-2 or miR-101 precursors; (f) anti-COX-2 shRNA (shCOX-2) or pre-miRNA expression cassettes can be transduced into target cells genome by the use of recombinant retroviruses, allowing a stable COX-2 silencing into mammalian cells.
Finally, since transactivators such as C/EBPβ are strongly implicated in COX-2 induction at transcriptional level, we suggest that inhibition of C/EBPβ expression or activity by the use of RNAi or other molecular compounds may reduce COX-2 overexpression associated to human pathologies such as inflammation or colon cancer.
Acknowledgments
The present work was supported by grants from MIUR (FIRB 2003 RBNE03FMCJ to V. Tomasi and PRIN 2008MT34AP to E. Spisni). The authors also thank Dr. Catherine Grillot-Courvalin (Unité des Agents Antibactériens, Institut Pasteur, Paris) for providing them pGB2-Ω-inv-hly plasmid. A. Strillacci and C. Griffoni contributed equally to this work.
References
- 1.Levine L. Arachidonic acid transformation and tumor production. Advances in Cancer Research. 1981;35:49–79. doi: 10.1016/s0065-230x(08)60908-2. [DOI] [PubMed] [Google Scholar]
- 2.DuBois RN, Abramson SB, Crofford L, et al. Cyclooxygenase in biology and disease. FASEB Journal. 1998;12(12):1063–1073. [PubMed] [Google Scholar]
- 3.Wang D, DuBois RN. Prostaglandins and cancer. Gut. 2006;55(1):115–122. doi: 10.1136/gut.2004.047100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Iniguez MA, Rodriguez A, Volpert OV, Fresno M, Redondo JM. Cyclooxygenase-2: a therapeutic target in angiogenesis. Trends in Molecular Medicine. 2003;9(2):73–78. doi: 10.1016/s1471-4914(02)00011-4. [DOI] [PubMed] [Google Scholar]
- 5.Greenhough A, Smartt HJM, Moore AE, et al. The COX-2/PGE2 pathway: key roles in the hallmarks of cancer and adaptation to the tumour microenvironment. Carcinogenesis. 2009;30(3):377–386. doi: 10.1093/carcin/bgp014. [DOI] [PubMed] [Google Scholar]
- 6.Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S, DuBois RN. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology. 1994;107(4):1183–1188. doi: 10.1016/0016-5085(94)90246-1. [DOI] [PubMed] [Google Scholar]
- 7.Sano H, Kawahito Y, Wilder RL, et al. Expression of cyclooxygenase-1 and -2 in human colorectal cancer. Cancer Research. 1995;55(17):3785–3789. [PubMed] [Google Scholar]
- 8.Thun MJ, Henley SJ, Patrono C. Nonsteroidal anti-inflammatory drugs as anticancer agents: mechanistic, pharmacologic, and clinical issues. Journal of the National Cancer Institute. 2002;94(4):252–266. doi: 10.1093/jnci/94.4.252. [DOI] [PubMed] [Google Scholar]
- 9.Liu CH, Chang S-H, Narko K, et al. Overexpression of cyclooxygenase-2 is sufficient to induce tumorigenesis in transgenic mice. Journal of Biological Chemistry. 2001;276(21):18563–18569. doi: 10.1074/jbc.M010787200. [DOI] [PubMed] [Google Scholar]
- 10.Tsujii M, Kawano S, Tsuji S, Sawaoka H, Hori M, DuBois RN. Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell. 1998;93(5):705–716. doi: 10.1016/s0092-8674(00)81433-6. [DOI] [PubMed] [Google Scholar]
- 11.Reddy RS, Maruyama H, Kelloff G. Dose-related inhibition of colon carcinogenesis by dietary piroxicam, a nonsteroidal antiinflammatory drug, during different stages of rat colon tumor development. Cancer Research. 1987;47(20):5340–5346. [PubMed] [Google Scholar]
- 12.Sandler RS, Halabi S, Baron JA, et al. A randomized trial of aspirin to prevent colorectal adenomas in patients with previous colorectal cancer. The New England Journal of Medicine. 2003;348(10):883–890. doi: 10.1056/NEJMoa021633. [DOI] [PubMed] [Google Scholar]
- 13.Dubé C, Rostom A, Lewin G, et al. The use of aspirin for primary prevention of colorectal cancer: a systematic review prepared for the U.S. Preventive Services Task Force. Annals of Internal Medicine. 2007;146:365–375. doi: 10.7326/0003-4819-146-5-200703060-00009. [DOI] [PubMed] [Google Scholar]
- 14.Phillips RKS, Wallace MH, Lynch PM, et al. A randomised, double blind, placebo controlled study of celecoxib, a selective cyclooxygenase 2 inhibitor, on duodenal polyposis familial adenomatous polyposis. Gut. 2002;50(6):857–860. doi: 10.1136/gut.50.6.857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Higuchi T, Iwama T, Yoshinaga K, Toyooka M, Taketo MM, Sugihara K. A randomized, double-blind, placebo-controlled trial of the effects of rofecoxib, a selective cyclooxygenase-2 inhibitor, on rectal polyps in familial adenomatous polyposis patients. Clinical Cancer Research. 2003;9(13):4756–4760. [PubMed] [Google Scholar]
- 16.Dempke W, Rie C, Grothey A, Schmoll H-J. Cyclooxygenase-2: a novel target for cancer chemotherapy. Journal of Cancer Research and Clinical Oncology. 2001;127(7):411–417. doi: 10.1007/s004320000225. [DOI] [PubMed] [Google Scholar]
- 17.Harris RE, Beebe-Donk J, Alshafie GA. Cancer chemoprevention by cyclooxygenase 2 (COX-2) blockade: results of case control studies. Sub-Cellular Biochemistry. 2007;42:193–212. doi: 10.1007/1-4020-5688-5_9. [DOI] [PubMed] [Google Scholar]
- 18.Jana NR. NSAIDs and apoptosis. Cellular and Molecular Life Sciences. 2008;65(9):1295–1301. doi: 10.1007/s00018-008-7511-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bertagnolli MM. Chemoprevention of colorectal cancer with cyclooxygenase-2 inhibitors: two steps forward, one step back. Lancet Oncology. 2007;8(5):439–443. doi: 10.1016/S1470-2045(07)70139-0. [DOI] [PubMed] [Google Scholar]
- 20.Solomon SD, McMurray JJV, Pfeffer MA, et al. Cardiovascular risk associated with celecoxib in a clinical trial for colorectal adenoma prevention. The New England Journal of Medicine. 2005;352(11):1071–1080. doi: 10.1056/NEJMoa050405. [DOI] [PubMed] [Google Scholar]
- 21.Psaty BM, Potter JD. Risks and benefits of celecoxib to prevent recurrent adenomas. The New England Journal of Medicine. 2006;355(9):950–952. doi: 10.1056/NEJMe068158. [DOI] [PubMed] [Google Scholar]
- 22.Bresalier RS, Sandler RS, Quan H, et al. Cardiovascular events associated with rofecoxib in a colorectal adenoma chemoprevention trial. The New England Journal of Medicine. 2005;352(11):1092–1102. doi: 10.1056/NEJMoa050493. [DOI] [PubMed] [Google Scholar]
- 23.Weir MR, Sperling RS, Reicin A, Gertz BJ. Selective COX-2 inhibition and cardiovascular effects: a review of the rofecoxib development program. American Heart Journal. 2003;146(4):591–604. doi: 10.1016/S0002-8703(03)00398-3. [DOI] [PubMed] [Google Scholar]
- 24.FitzGerald GA. Coxibs and cardiovascular disease. The New England Journal of Medicine. 2004;351(17):1709–1711. doi: 10.1056/NEJMp048288. [DOI] [PubMed] [Google Scholar]
- 25.Griffoni C, Spisni E, Strillacci A, Toni M, Bachschmid MM, Tomasi V. Selective inhibition of prostacyclin synthase activity by rofecoxib. Journal of Cellular and Molecular Medicine. 2007;11(2):327–338. doi: 10.1111/j.1582-4934.2007.00021.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC. Potent and specific genetic interference by double-stranded RNA in caenorhabditis elegans. Nature. 1998;391(6669):806–811. doi: 10.1038/35888. [DOI] [PubMed] [Google Scholar]
- 27.Hammond SM, Bernstein E, Beach D, Hannon GJ. An RNA-directed nuclease mediates post-transcriptional gene silencing in Drosophila cells. Nature. 2000;404(6775):293–296. doi: 10.1038/35005107. [DOI] [PubMed] [Google Scholar]
- 28.Zamore PD, Tuschl T, Sharp PA, Bartel DP. RNAi: double-stranded RNA directs the ATP-dependent cleavage of mRNA at 21 to 23 nucleotide intervals. Cell. 2000;101(1):25–33. doi: 10.1016/S0092-8674(00)80620-0. [DOI] [PubMed] [Google Scholar]
- 29.Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001;411(6836):494–498. doi: 10.1038/35078107. [DOI] [PubMed] [Google Scholar]
- 30.Bernstein E, Caudy AA, Hammond SM, Hannon GJ. Role for a bidentate ribonuclease in the initiation step of RNA interference. Nature. 2001;409(6818):363–366. doi: 10.1038/35053110. [DOI] [PubMed] [Google Scholar]
- 31.Liu J, Carmell MA, Rivas FV, et al. Argonaute2 is the catalytic engine of mammalian RNAi. Science. 2004;305(5689):1437–1441. doi: 10.1126/science.1102513. [DOI] [PubMed] [Google Scholar]
- 32.Denkert C, Fürstenberg A, Daniel PT, et al. Induction of G0/G1 cell cycle arrest in ovarian carcinoma cells by the anti-inflammatory drug NS-398, but not by COX-2-specific RNA interference. Oncogene. 2003;22(54):8653–8661. doi: 10.1038/sj.onc.1206920. [DOI] [PubMed] [Google Scholar]
- 33.Charames GS, Bapat B. Cyclooxygenase-2 knockdown by RNA interference in colon cancer. International Journal of Oncology. 2006;28(2):543–549. [PubMed] [Google Scholar]
- 34.Kobayashi S-I, Sakatani M, Kobayashi S, Okuda K, Takahashi M. Gene silencing of cyclooxygenase-2 mRNA by RNA interference in bovine cumulus-granulosa cells. Journal of Reproduction and Development. 2007;53(6):1305–1311. doi: 10.1262/jrd.19050. [DOI] [PubMed] [Google Scholar]
- 35.Bozza PT, Payne JL, Morham SG, Langenbach R, Smithies O, Weller PF. Leukocyte lipid body formation and eicosanoid generation: cyclooxygenase-independent inhibition by aspirin. Proceedings of the National Academy of Sciences of the United States of America. 1996;93(20):11091–11096. doi: 10.1073/pnas.93.20.11091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Grilli M, Pizzi M, Memo M, Spano P. Neuroprotection by aspirin and sodium salicylate through blockade of NF-kappaB activation. Science. 1996;274(5291):1383–1385. doi: 10.1126/science.274.5291.1383. [DOI] [PubMed] [Google Scholar]
- 37.Elder DJE, Halton DE, Hague A, Paraskeva C. Induction of apoptotic cell death in human colorectal carcinoma cell lines by a cyclooxygenase-2 (COX-2)-selective nonsteroidal anti-inflammatory drug: independence from COX-2 protein expression. Clinical Cancer Research. 1997;3(10):1679–1683. [PubMed] [Google Scholar]
- 38.Lim JTE, Piazza GA, Han EK-H, et al. Sulindac derivatives inhibit growth and induce apoptosis in human prostate cancer cell lines. Biochemical Pharmacology. 1999;58(7):1097–1107. doi: 10.1016/s0006-2952(99)00200-2. [DOI] [PubMed] [Google Scholar]
- 39.Strillacci A, Griffoni C, Spisni E, Manara MC, Tomasi V. RNA interference as a key to knockdown overexpressed cyclooxygenase-2 gene in tumour cells. British Journal of Cancer. 2006;94(9):1300–1310. doi: 10.1038/sj.bjc.6603094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang R, Wang X, Lin F, Gao P, Dong K, Zhang H-Z. shRNA-targeted cyclooxygenase (COX)-2 inhibits proliferation, reduces invasion and enhances chemosensitivity in laryngeal carcinoma cells. Molecular and Cellular Biochemistry. 2008;317(1-2):179–188. doi: 10.1007/s11010-008-9847-9. [DOI] [PubMed] [Google Scholar]
- 41.Sansone P, Piazzi G, Paterini P, et al. Cyclooxygenase-2/carbonic anhydrase-IX up-regulation promotes invasive potential and hypoxia survival in colorectal cancer cells. Journal of Cellular and Molecular Medicine. 2009;13(9b):3876–3887. doi: 10.1111/j.1582-4934.2008.00580.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Timmons L, Fire A. Specific interference by ingested dsRNA. Nature. 1998;395(6705):p. 854. doi: 10.1038/27579. [DOI] [PubMed] [Google Scholar]
- 43.May RC, Plasterk RHA. RNA interference spreading in C. elegans. Methods in Enzymology. 2005;392:308–315. doi: 10.1016/S0076-6879(04)92018-6. [DOI] [PubMed] [Google Scholar]
- 44.Xiang S, Fruehauf J, Li CJ. Short hairpin RNA—expressing bacteria elicit RNA interference in mammals. Nature Biotechnology. 2006;24(6):697–702. doi: 10.1038/nbt1211. [DOI] [PubMed] [Google Scholar]
- 45.Keetes AC, Fruehauf JH, Xiang S, Parker PD, Li CJ. Cequent pharmaceuticals, Inc.: the biological pitcher for RNAi therapeutics. Pharmacogenomics. 2007;8(7):867–871. doi: 10.2217/14622416.8.7.867. [DOI] [PubMed] [Google Scholar]
- 46.Rajewsky N. microRNA target predictions in animals. Nature Genetics. 2006;38(1):S8–S13. doi: 10.1038/ng1798. [DOI] [PubMed] [Google Scholar]
- 47.Griffiths-Jones S, Saini HK, van Dongen S, Enright AJ. miRBase: tools for microRNA genomics. Nucleic Acids Research. 2008;36(1):D154–D158. doi: 10.1093/nar/gkm952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lee Y, Ahn C, Han J, et al. The nuclear RNase III Drosha initiates microRNA processing. Nature. 2003;425(6956):415–419. doi: 10.1038/nature01957. [DOI] [PubMed] [Google Scholar]
- 49.Hutvágner G, McLachlan J, Pasquinelli AE, Bálint E, Tuschl T, Zamore PD. A cellular function for the RNA-interference enzyme dicer in the maturation of the let-7 small temporal RNA. Science. 2001;293(5531):834–838. doi: 10.1126/science.1062961. [DOI] [PubMed] [Google Scholar]
- 50.Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116(2):281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 51.Lu J, Getz G, Miska EA, et al. MicroRNA expression profiles classify human cancers. Nature. 2005;435(7043):834–838. doi: 10.1038/nature03702. [DOI] [PubMed] [Google Scholar]
- 52.Schetter AJ, Leung SY, Sohn JJ, et al. MicroRNA expression profiles associated with prognosis and therapeutic outcome in colon adenocarcinoma. Journal of the American Medical Association. 2008;299(4):425–436. doi: 10.1001/jama.299.4.425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cho WCS. OncomiRs: the discovery and progress of microRNAs in cancers. Molecular Cancer. 2007;6, article 60 doi: 10.1186/1476-4598-6-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gartel AL, Kandel ES. miRNAs: little known mediators of oncogenesis. Seminars in Cancer Biology. 2008;18(2):103–110. doi: 10.1016/j.semcancer.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 55.Akao Y, Nakagawa Y, Naoe T. MicroRNAs 143 and 145 are possible common onco-microRNAs in human cancers. Oncology Reports. 2006;16(4):845–850. [PubMed] [Google Scholar]
- 56.Dixon DA, Tolley ND, King PH, et al. Altered expression of the mRNA stability factor HuR promotes cyclooxygenase-2 expression in colon cancer cells. Journal of Clinical Investigation. 2001;108(11):1657–1665. doi: 10.1172/JCI12973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chakrabarty A, Tranguch S, Daikoku T, Jensen K, Furneaux H, Dey SK. MicroRNA regulation of cyclooxygenase-2 during embryo implantation. Proceedings of the National Academy of Sciences of the United States of America. 2007;104(38):15144–15149. doi: 10.1073/pnas.0705917104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Daikoku T, Hirota Y, Tranguch S, et al. Conditional loss of uterine Pten unfailingly and rapidly induces endometrial cancer in mice. Cancer Research. 2008;68(14):5619–5627. doi: 10.1158/0008-5472.CAN-08-1274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Strillacci A, Griffoni C, Sansone P, et al. MiR-101 downregulation is involved in cyclooxygenase-2 overexpression in human colon cancer cells. Experimental Cell Research. 2009;315(8):1439–1447. doi: 10.1016/j.yexcr.2008.12.010. [DOI] [PubMed] [Google Scholar]
- 60.Shanmugam N, Reddy MA, Natarajan R. Distinct roles of heterogeneous nuclear ribonuclear protein K and microRNA-16 in cyclooxygenase-2 RNA stability induced by S100b, a ligand of the receptor for advanced glycation end products. Journal of Biological Chemistry. 2008;283(52):36221–36233. doi: 10.1074/jbc.M806322200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yamamoto K, Arakawa T, Ueda N, Yamamoto S. Transcriptional roles of nuclear factor kappa B and nuclear factor-interleukin-6 in the tumour necrosis factor alpha-dependent induction of cyclooxygenase-2 in MC3T3-E1 cells. Journal of Biological Chemistry. 1995;270(52):31315–31320. doi: 10.1074/jbc.270.52.31315. [DOI] [PubMed] [Google Scholar]
- 62.Wu KK, Liou J-Y, Cieslik K. Transcriptional Control of COX-2 via C/EBPbeta. Arteriosclerosis, Thrombosis, and Vascular Biology. 2005;25(4):679–685. doi: 10.1161/01.ATV.0000157899.35660.61. [DOI] [PubMed] [Google Scholar]
- 63.Chen J-C, Huang K-C, Wingerd B, Wu W-T, Lin W-W. HMG-CoA reductase inhibitors induce COX-2 gene expression in murine macrophages: role of MAPK cascades and promoter elements for CREB and C/EBPβ. Experimental Cell Research. 2004;301(2):305–319. doi: 10.1016/j.yexcr.2004.05.039. [DOI] [PubMed] [Google Scholar]
- 64.Duque J, Fresno M, Iñiguez MA. Expression and function of the nuclear factor of activated T cells in colon carcinoma cells: involvement in the regulation of cyclooxygenase-2. Journal of Biological Chemistry. 2005;280(10):8686–8693. doi: 10.1074/jbc.M413076200. [DOI] [PubMed] [Google Scholar]
- 65.Grau R, Iñiguez MA, Fresno M. Inhibition of activator protein 1 activation, vascular endothelial growth factor, and cyclooxygenase-2 expression by 15-deoxy-delta12,14-prostaglandin J2 in colon carcinoma cells: evidence for a redox-sensitive peroxisome proliferator-activated receptor-gamma-independent mechanism. Cancer Research. 2004;64(15):5162–5171. doi: 10.1158/0008-5472.CAN-04-0849. [DOI] [PubMed] [Google Scholar]
- 66.Ikawa H, Kameda H, Kamitani H, et al. Effect of PPAR activators on cytokine-stimulated cyclooxygenase-2 expression in human colorectal carcinoma cells. Experimental Cell Research. 2001;267(1):73–80. doi: 10.1006/excr.2001.5233. [DOI] [PubMed] [Google Scholar]
- 67.Kaidi A, Qualtrough D, Williams AC, Paraskeva C. Direct transcriptional up-regulation of cyclooxygenase-2 by hypoxia-inducible factor (HIF)-1 promotes colorectal tumor cell survival and enhances HIF-1 transcriptional activity during hypoxia. Cancer Research. 2006;66(13):6683–6691. doi: 10.1158/0008-5472.CAN-06-0425. [DOI] [PubMed] [Google Scholar]
- 68.Inoue H, Yokoyama C, Hara S, Tone Y, Tanabe T. Transcriptional regulation of human prostaglandin-endoperoxide synthase-2 gene by lipopolysaccharide and phorbol ester in vascular endothelial cells. Involvement of both nuclear factor for interleukin-6 expression site and cAMP response element. Journal of Biological Chemistry. 1995;270(42):24965–24971. doi: 10.1074/jbc.270.42.24965. [DOI] [PubMed] [Google Scholar]
- 69.Thomas B, Berenbaum F, Humbert L, et al. Critical role of C/EBPdelta and C/EBPbeta factors in the stimulation of the cyclooxygenase-2 gene transcription by interleukin-1beta in articular chondrocytes. European Journal of Biochemistry. 2000;267(23):6798–6809. doi: 10.1046/j.1432-1033.2000.01778.x. [DOI] [PubMed] [Google Scholar]
- 70.Kim Y, Fischer SM. Transcriptional regulation of cyclooxygenase-2 in mouse skin carcinoma cells: regulatory role of CCAAT/enhancer-binding proteins in the differential expression of cyclooxygenase-2 in normal and neoplastic tissues. Journal of Biological Chemistry. 1998;273(42):27686–27694. doi: 10.1074/jbc.273.42.27686. [DOI] [PubMed] [Google Scholar]
- 71.Shao J, Sheng H, Inoue H, Morrow JD, DuBois RN. Regulation of constitutive cyclooxygenase-2 expression in colon carcinoma cells. Journal of Biological Chemistry. 2000;275(43):33951–33956. doi: 10.1074/jbc.M002324200. [DOI] [PubMed] [Google Scholar]
- 72.Saunders MA, Sansores-Garcia L, Gilroy DW, Wu KK. Selective suppression of CCAAT/enhancer-binding protein beta binding and cyclooxygenase-2 promoter activity by sodium salicylate in quiescent human fibroblasts. Journal of Biological Chemistry. 2001;276(22):18897–18904. doi: 10.1074/jbc.M011147200. [DOI] [PubMed] [Google Scholar]
- 73.Xing J, Ginty DD, Greenberg ME. Coupling of the RAS-MAPK pathway to gene activation by RSK2, a growth factor-regulated CREB kinase. Science. 1996;273(5277):959–963. doi: 10.1126/science.273.5277.959. [DOI] [PubMed] [Google Scholar]
- 74.Soutschek J, Akinc A, Bramlage B, et al. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature. 2004;432(7014):173–178. doi: 10.1038/nature03121. [DOI] [PubMed] [Google Scholar]
- 75.Thomas M, Lu JJ, Ge Q, et al. Full deacylation of polyethylenimine dramatically boosts its gene delivery efficiency and specificity to mouse lung. Proceedings of the National Academy of Sciences of the United States of America. 2005;102:5679–5684. doi: 10.1073/pnas.0502067102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kim SH, Jeong JH, Lee SH, Kim SW, Park TG. PEG conjugated VEGF siRNA for anti-angiogenic gene therapy. Journal of Controlled Release. 2006;116(2):123–129. doi: 10.1016/j.jconrel.2006.05.023. [DOI] [PubMed] [Google Scholar]
- 77.Mikhaylova M, Stasinopoulos I, Kato Y, Artemov D, Bhujwalla ZM. Imaging of cationic multifunctional liposome-mediated delivery of COX-2 siRNA. Cancer Gene Therapy. 2009;16(3):217–226. doi: 10.1038/cgt.2008.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hatakeyama H, Ito E, Akita H, et al. A pH-sensitive fusogenic peptide facilitates endosomal escape and greatly enhances the gene silencing of siRNA-containing nanoparticles in vitro and in vivo. Journal of Controlled Release. 2009;139(2):127–132. doi: 10.1016/j.jconrel.2009.06.008. [DOI] [PubMed] [Google Scholar]
- 79.Toublan FJ-J, Boppart S, Suslick KS. Tumor targeting by surface-modified protein microspheres. Journal of the American Chemical Society. 2006;128(11):3472–3473. doi: 10.1021/ja0544455. [DOI] [PubMed] [Google Scholar]
- 80.Zeng Y, Yi R, Cullen BR. MicroRNAs and small interfering RNAs can inhibit mRNA expression by similar mechanisms. Proceedings of the National Academy of Sciences of the United States of America. 2003;100(17):9779–9784. doi: 10.1073/pnas.1630797100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Jackson AL, Bartz SR, Schelter J, et al. Expression profiling reveals off-target gene regulation by RNAi. Nature Biotechnology. 2003;21(6):635–637. doi: 10.1038/nbt831. [DOI] [PubMed] [Google Scholar]
- 82.Lee Y, Samaco RC, Gatchel JR, Thaller C, Orr HT, Zoghbi HY. miR-19, miR-101 and miR-130 co-regulate ATXN1 levels to potentially modulate SCA1 pathogenesis. Nature Neuroscience. 2008;11(10):1137–1139. doi: 10.1038/nn.2183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Friedman JM, Liang G, Liu C-C, et al. The putative tumor suppressor microRNA-101 modulates the cancer epigenome by repressing the polycomb group protein EZH2. Cancer Research. 2009;69(6):2623–2629. doi: 10.1158/0008-5472.CAN-08-3114. [DOI] [PubMed] [Google Scholar]
- 84.Kim S, Ui JL, Mi NK, et al. MicroRNA miR-199a∗ regulates the MET proto-oncogene and the downstream extracellular signal-regulated kinase 2 (ERK2) Journal of Biological Chemistry. 2008;283(26):18158–18166. doi: 10.1074/jbc.M800186200. [DOI] [PubMed] [Google Scholar]
- 85.Chen R, Alvero AB, Silasi DA, et al. Regulation of IKKbeta by miR-199a affects NF-kappaB activity in ovarian cancer cells. Oncogene. 2008;27(34):4712–4723. doi: 10.1038/onc.2008.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hiroki E, Akahira J-I, Suzuki F, et al. Changes in microRNA expression levels correlate with clinicopathological features and prognoses in endometrial serous adenocarcinomas. Cancer Science. 2010;101(1):241–249. doi: 10.1111/j.1349-7006.2009.01385.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Boudreau RL, Monteys AM, Davidson BL. Minimizing variables among hairpin-based RNAi vectors reveals the potency of shRNAs. RNA. 2008;14(9):1834–1844. doi: 10.1261/rna.1062908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Grimm D, Streetz KL, Jopling CL, et al. Fatality in mice due to oversaturation of cellular microRNA/short hairpin RNA pathways. Nature. 2006;441(7092):537–541. doi: 10.1038/nature04791. [DOI] [PubMed] [Google Scholar]
- 89.Bridge AJ, Pebernard S, Ducraux A, Nicoulaz A-L, Iggo R. Induction of an interferon response by RNAi vectors in mammalian cells. Nature Genetics. 2003;34(3):263–264. doi: 10.1038/ng1173. [DOI] [PubMed] [Google Scholar]
- 90.Judge AD, Sood V, Shaw JR, Fang D, McClintock K, MacLachlan I. Sequence-dependent stimulation of the mammalian innate immune response by synthetic siRNA. Nature Biotechnology. 2005;23(4):457–462. doi: 10.1038/nbt1081. [DOI] [PubMed] [Google Scholar]