Abstract
The inhibitory receptor programmed death 1 (PD-1) is upregulated on antigen-specific CD8+ T cells during persistent viral infections. Interaction with PD-1 ligand 1 (PD-L1) contributes to functional exhaustion of responding T cells and may limit immunopathology during infection. PD-L1 is expressed on both hematopoietic and nonhematopoietic cells in tissues. However, the exact roles of PD-L1 on hematopoietic versus nonhematopoietic cells in modulating immune responses are unclear. Here we used bone marrow chimeric mice to examine the effects of PD-L1 deficiency in hematopoietic or nonhematopoietic cells during lymphocytic choriomeningitis virus clone 13 (LCMV CL-13) infection. We found that PD-L1 expression on hematopoietic cells inhibited CD8+ T cell numbers and function after LCMV CL-13 infection. In contrast, PD-L1 expression on nonhematopoietic cells limited viral clearance and immunopathology in infected tissues. Together, these data demonstrate that there are distinct roles for PD-L1 on hematopoietic and nonhematopoietic cells in regulating CD8+ T cell responses and viral clearance during chronic viral infection.
Introduction
We recently identified a major inhibitory pathway consisting of the inhibitory receptor programmed death 1 (PD-1) and its ligand PD-1 ligand 1 (PD-L1) involved in T cell exhaustion during chronic lymphocytic choriomeningitis virus (LCMV) infection (1). PD-1 is upregulated significantly on CTL during chronic infection and regulates T cell expansion and function. PD-L1 is widely expressed on cells such as macrophages, DCs, T and B cells, Kupffer cells in the liver, and β cells in the pancreas as well as many others and is upregulated in response to inflammatory stimuli (2). The level of PD-L1 expression and magnitude of upregulation in response to stimuli can differ on different cell types and may be important for controlling the function of lymphocytes expressing PD-1. We have found that PD-L1 is expressed on lymphoid stromal cells, upregulated during LCMV CL-13 infection (3), and important for reducing immunopathology in the infected lymphoid stromal cells.
The expression of PD-L1 by many cell types in multiple tissues suggests that this ligand may have important roles in inhibiting immune responses in both lymphoid and nonlymphoid organs. The functions of PD-1/PD-L1 interactions in controlling T cell activation versus T cell effector functions are only beginning to be understood. Studies have demonstrated that PD-L1 on APCs can control peripheral tolerance and anergy (4, 5). Interestingly, PD-L1 expression on tissue parenchyma can protect against autoimmune pathology in diabetes models (6). The PD-1/PD-L1 pathway plays an important role in influencing T cell dysfunction and viral clearance during chronic viral infection (2, 7) and may represent a useful therapeutic approach for the treatment of persistent infections. However, a greater understanding of the cellular sources of PD-L1 inhibitory signals will prove beneficial to predicting a clinical outcome in patients with persistent infections.
In this study, we evaluated the function of PD-L1 expression on hematopoietic versus nonhematopoietic cells during chronic LCMV infection. Using BM chimeric mice, we demonstrate that PD-L1 on BM cells inhibited CD8+ T cell responses and negatively impacted T cell function. In contrast, expression of PD-L1 on non-BM–derived cells reduced viral clearance in tissues. Ultimately, however, PD-L1 expression on both compartments was critical for preventing lethal immunopathology. Together, these studies identify distinct roles for PD-L1 on hematopoietic versus parenchymal cells in regulating chronic viral infection: PD-L1 on BM cells regulates expansion of T cell numbers early during infection, whereas PD-L1 on parenchymal cells controls viral clearance and immune-mediated tissue damage.
Results
PD-L1 expression on hematopoietic cells regulates CTL responses during viral infection.
Functional exhaustion and deletion of responding T cells occur during infection of mice with LCMV CL-13. Cytotoxic T cells progressively lose the ability to produce the cytokines IL-2, TNF-α, and IFN-γ as they become exhausted (8–10). The PD-1/PD-L1 pathway has an important role in regulating T cell exhaustion, since blocking this interaction leads to enhanced CTL number and function in chronically infected individuals (1, 11–13). PD-L1 is expressed by BM-derived cells to varying degrees in uninfected mice (Supplemental Figure 1; supplemental material available online with this article; doi: 10.1172/JCI40040DS1). PD-L1 expression is rapidly upregulated on B and T lymphocytes, NK cells, DCs, macrophages, and granulocytes within 1 day after infection with LCMV CL-13 and remains increased for at least 1 week. Interestingly, DCs expressed very high levels of PD-L1 within 1 day of infection, suggesting that this expression may be important for controlling immune responses.
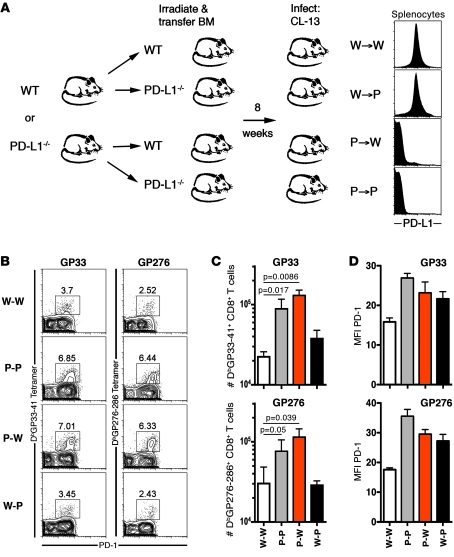
To better understand the source of PD-L1 signals for T cells, in particular the roles of PD-L1 expression by hematopoietic cells (such as DCs and macrophages) and nonhematopoietic cells (such as stromal cells and other supportive cells in the tissues) during infection, we generated mice lacking PD-L1 on either BM and/or non-BM–derived cells. These BM chimeric mice were made by lethally irradiating WT C57BL/6 mice or Pdl1–/– mice and reconstituting them with either WT or Pdl1–/– BM (Figure 1A). The resulting chimeras consisted of 4 groups: WT BM into WT recipients (W-W), WT into Pdl1–/– (W-P), Pdl1–/– into WT (P-W), and Pdl1–/– into Pdl1–/– (P-P). Expression of PD-L1, or lack thereof, was confirmed on PBMCs and splenocytes 8 weeks after reconstitution (Figure 1A).
Figure 1. PD-L1 expression on hematopoietic cells inhibits virus-specific CD8+ T cell responses.
(A) Lethally irradiated Pdl1–/– or WT mice were reconstituted with Pdl1–/– BM (P-P or P-W) or WT BM (W-P or W-W) and infected with LCMV CL-13 8 weeks later. Expression of PD-L1 on splenic hematopoietic cells was confirmed by flow cytometry. (B) GP33- and GP276-specific responses in the spleen 8 days after CL-13 infection were measured using MHC class I tetramers. Numbers above gates represent the percentage of CD8+ T cells staining positive for GP33 tetramer and PD-1. (C) The numbers of tetramer-positive cells in the spleen of chimeric mice 8 days after infection. (D) Expression of PD-1 on tetramer-positive cells from the spleen. n = 4–8 mice per group. Mean + SEM of data from 1 of 4 representative experiments are shown.
We examined antigen-specific T cell responses in the spleen 8 days after infection with LCMV CL-13 (Figure 1, B and C). LCMV DbGP33-41–specific and DbGP276-286–specific CD8+ T cell populations were significantly increased in chimeric mice lacking PD-L1 on BM-derived cells (P-P and P-W mice). However, mice with PD-L1–sufficient BM had fewer antigen-specific CD8+ T cells, irrespective of whether PD-L1 was expressed on nonhematopoietic cells (W-W and W-P). Numbers of annexin V+ DbGP33–specific and DbGP276–specific CD8+ T cells were slightly decreased in Pdl1–/– mice (Supplemental Figure 2), suggesting that increased survival might also contribute to increased responses in the absence of PD-L1 signals, in addition to increased proliferation. Together, these data show that PD-L1 expression on hematopoietic cells, and not on nonhematopoietic cells, significantly influenced CD8+ T cell responses soon after infection. Interestingly, antigen-specific T cells also expressed increased levels of PD-1 when PD-L1 was lacking on either BM- or non-BM–derived cells (Figure 1D).
PD-L1 expression on hematopoietic cells influences CTL function during chronic viral infection.
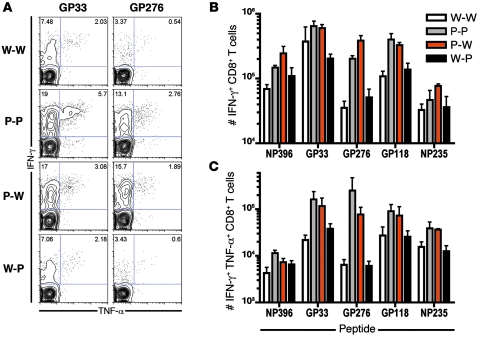
Since blockade of the PD-1/PD-L1 pathway enhances CTL function during chronic infection (1, 11–13), we next determined whether CTL primed in mice lacking expression of PD-L1 on either BM-derived cells or parenchymal cells were capable of producing cytokines upon antigen stimulation. Mice lacking PD-L1 on BM-derived cells (P-P and P-W) demonstrated an increase in GP33- and GP276-specific CD8+ T cells producing IFN-γ after infection (Figure 2A). Moreover, increased numbers of IFN-γ+ virus–specific T cells responding to multiple epitopes were induced in these mice in comparison with mice expressing PD-L1 on hematopoietic cells (W-W and W-P; Figure 2B). Mice lacking PD-L1 on BM-derived cells also demonstrated an increase in polyfunctional CD8+ T cells capable of producing both IFN-γ and TNF-α after stimulation (Figure 2, A and C). In contrast, mice lacking PD-L1 expression on nonhematopoietic cells (W-P) displayed no such increase in antigen-specific IFN-γ+TNF-α+CD8+ T cells after CL-13 infection when compared with W-W mice. Together, these data demonstrate that PD-L1 expression on BM-derived cells regulates T cell numbers and function early during viral infection. These results also indicate that PD-L1 expression on nonhematopoietic cells does not significantly influence the induction of virus-specific T cell responses during chronic infection.
Figure 2. PD-L1 expression on hematopoietic cells influences CTL function.
(A) IFN-γ and TNF-α production by GP33- and GP276-specific CD8+ T cells in the spleen 8 days after infection. Numbers in the upper quadrants represent the proportion of total CD8+ T cells expressing IFN-γ (left) or IFN-γ and TNF-α (right) after GP33 or GP276 stimulation. (B) The number of IFN-γ+ and (C) IFN-γ+TNF-α+ cells responding to NP396, GP33, GP276, GP118, and NP235 epitopes. n = 4–8 mice per group. Mean + SEM of data from 1 of 4 representative experiments are shown.
Increased T cell responses in nonlymphoid tissues in mice lacking PD-L1 expression on hematopoietic cells.
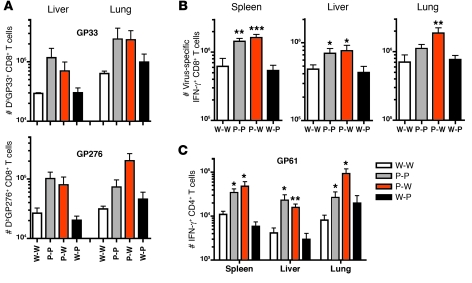
Chronic LCMV infection involves systemic dissemination of virus to lymphoid (both primary and secondary) and nonlymphoid tissues. Effector T lymphocytes are found in all tissues, though the relative distribution of cells and their function can vary depending on the tissue and time since infection (8). We examined DbGP33-41–specific and DbGP276-286–specific CD8+ T cell populations in the liver and lung in chimeric mice 8 days after LCMV CL-13 infection. Mice lacking PD-L1 expression on BM-derived cells displayed increased numbers of antigen-specific CTL in the nonlymphoid tissues (Figure 3A). The liver and lungs from chimeric mice lacking PD-L1 on BM cells (P-P and P-W) demonstrated a significant increase in the number of IFN-γ+ virus–specific CD8+ T cells responding to multiple epitopes, similar to that observed in the spleen (Figure 3B). We also observed increased numbers of polyfunctional IFN-γ+TNF-α+CD8+ T cells in the liver and lungs of P-P and P-W mice after CL-13 infection (not shown). In contrast, no significant increase in virus-specific cells was observed in mice that only lacked PD-L1 expression on parenchymal cells (W-P).
Figure 3. PD-L1 expression on hematopoietic cells controls CD8+ and CD4+ T cell responses in lymphoid and nonlymphoid tissues.
(A) GP33- and GP276-specific responses in the liver and lungs 8 days after CL-13 infection were measured using MHC class I tetramers. (B) The number of CD8+ IFN-γ+ cells responding to 6 LCMV epitopes (GP33, GP276, NP396, NP235, NP205, and GP118) in the spleen, liver, and lung 8 days after infection. Responses to individual epitopes were obtained for each sample and then pooled for display. (C) The number of CD4+IFN-γ+GP61–specific T cells in the spleen, liver, and lungs 8 days after LCMV CL-13 infection. n = 3–4 mice per group. Mean + SEM of data from 1 of 3 representative experiments are shown. *P < 0.05; **P = 0.01; ***P < 0.005.
We also examined virus-specific CD4+ T cell responses in the spleen, liver, and lungs. LCMV GP61-80–specific CD4+ T cells producing IFN-γ were significantly increased in all tissues in mice lacking PD-L1 on hematopoietic cells (P-P and P-W), yet not in W-P mice (Figure 3C). Together, these data demonstrate that CD8+ and CD4+ effector T cells are increased in both lymphoid and nonlymphoid tissues in mice deficient in PD-L1 on BM-derived cells. Thus, T cell numbers and function are controlled by BM-derived PD-L1 but not nonhematopoietic PD-L1 during chronic LCMV infection.
PD-L1 expression on nonhematopoietic cells controls viral clearance and tissue immunopathology.
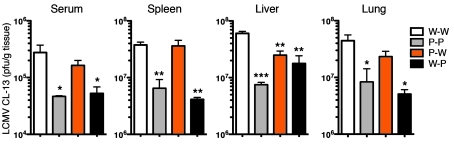
The preceding experiments show that the pool of antigen-specific T cells in LCMV-infected mice lacking PD-L1 expression on BM-derived cells was significantly increased as compared with WT mice or mice lacking PD-L1 expression only on nonhematopoietic cells. Since PD-1 signals can influence effector T cell responses in tissues such as the liver (14), we wanted to ascertain whether PD-L1 expression on either the BM-derived or parenchymal cells controlled the clearance of virus in different organs. We measured viral titers in the blood, spleen, liver, and lungs. Viral titers in the blood and tissues of mice lacking PD-L1 on all cells (P-P) were reduced in comparison to those in normal (W-W) mice (Figure 4). Surprisingly, mice with PD-L1–deficient BM and PD-L1–sufficient parenchyma (P-W) did not display enhanced viral clearance in the blood and many tissues, despite increased numbers of functional CTL in these tissues (see Figures 1–3). These mice, however, did display improved viral clearance in the liver in comparison with W-W mice. In contrast, mice lacking PD-L1 expression on parenchymal cells (W-P) demonstrated significantly greater viral clearance in the blood and all tissues examined, similar to that observed in P-P mice. Thus, despite displaying virus-specific T cell numbers similar to that in W-W mice, W-P mice were able to clear virus more efficiently. These data demonstrate that PD-L1 on parenchymal cells significantly influenced viral clearance from the tissues during LCMV CL-13 infection.
Figure 4. PD-L1 expression on nonhematopoietic cells influences viral clearance.
Viral titers in the blood (serum), spleen, liver, and lungs 8 days after LCMV CL-13 infection. n = 3–4 mice per group. Mean + SEM of data from 1 of 4 representative experiments are shown. *P ≤ 0.05; **P ≤ 0.005; ***P < 0.0001.
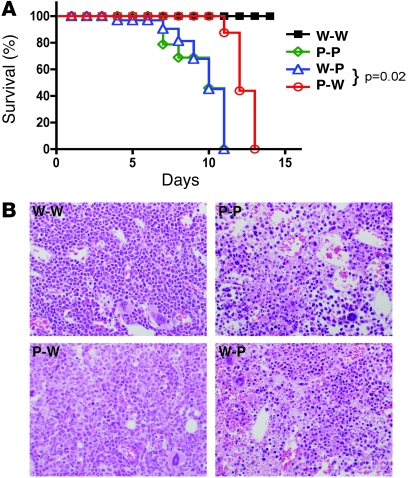
To further understand the biological significance of BM versus non-BM–derived PD-L1 signals, we compared the survival of groups of chimeric mice after CL-13 infection. Mice lacking PD-L1 on non-BM cells (P-P and W-P) demonstrated early mortality after infection (Figure 5A). These mice succumbed to infection after approximately 7–10 days. This correlated with enhanced viral clearance in these mice. Interestingly, mice lacking PD-L1 on hematopoietic cells (P-W) also succumbed to infection, although they were consistently found to survive a few days longer than P-P or W-P mice (11–13 days after infection). This correlated with reduced viral clearance in these mice (see Figure 4), potentially reflecting less severe immune damage to the tissues than in mice lacking PD-L1 on nonhematopoietic cells. Mice lacking PD-L1 expression also displayed higher levels of the cytokines IL-6 and TNF-α in the circulation compared with W-W mice (data not shown), which may have contributed to their demise. These cytokines were found to be slightly higher in the serum of mice lacking PD-L1 expression on parenchymal cells when compared with P-W mice. Together, these results suggest that the inhibitory PD-1/PD-L1 pathway may be critical for controlling virus-induced immunopathology.
Figure 5. PD-L1 expression on nonhematopoietic cells influences survival and immunopathology after infection.
(A) Chimeric mice were infected with LCMV CL-13 and survival monitored daily. n = 7–10 mice; 1 representative experiment of 3 is shown. (B) Severe pathology in the BM compartment in chimeric mice lacking PD-L1 on nonhematopoietic cells. Representative photomicrographs of BM from CL-13–infected chimeric mice 8 days after infection. Pdl1–/– mice reconstituted with WT or Pdl1–/– BM (P-P or W-P) demonstrated significant necrosis of the BM compartment in contrast with W-W and P-W groups, which did not show necrosis. Original magnification, ×400.
To further examine how PD-L1 expression on hematopoietic or nonhematopoietic cells influenced immunopathology during LCMV CL-13 infection, we examined tissues from chimeric mice after infection. No significant histopathological changes were observed in the heart or kidney 8 days after LCMV CL-13 infection. Examination of the central nervous system also revealed no significant differences among groups, and there was no meningeal inflammation. Spleens from all groups of mice were highly activated, with expanded white-pulp regions and apoptotic cells. This was most striking in mice lacking PD-L1 on both hematopoietic and nonhematopoietic cells. By immunohistochemistry, there was secondary follicle formation in all groups and no significant differences in the numbers or distribution of T cells, B cells, and macrophages. There was a slight yet noticeable increase in apoptosis in the thymus of P-P and W-P mice compared with WT recipients.
The lungs of W-W chimeric mice displayed minimal histopathological changes, and this was only slightly increased in P-W mice after infection. However, P-P and W-P mice showed increased interstitial inflammation with some alveolar edema in the lungs of 25% of the W-P mice and 100% of P-P mice. The livers from all groups showed changes typically seen in viral hepatitis. There was scattered inflammation affecting both the portal areas and the hepatic lobules, as well as significant hepatocellular necrosis in all groups with dying hepatocytes identified singly and occasionally in small clusters scattered throughout the liver. The hepatocellular necrosis was greatest in chimeric mice lacking PD-L1 on nonhematopoietic cells (W-P). Immunohistochemical staining revealed that the inflammation in the portal areas and hepatic lobules consisted of both T and B cells. Measurement of liver enzymes (serum aspartate aminotransferase [AST]; and alanine aminotransferase [ALT]) demonstrated that acute hepatocyte damage was increased in mice lacking PD-L1 on BM cells (Supplemental Figure 3). In comparison, serum bilirubin levels were increased in mice lacking PD-L1 on non-BM cells. The similar viral control (Figure 4) yet differential immunopathology in the livers of W-P and P-W mice suggests that PD-L1 on both the BM- and non-BM–derived cells in the liver plays a significant role in disease during chronic viral infection.
BM from mice after CL-13 infection also showed pronounced changes. Both groups of mice with PD-L1–sufficient parenchyma (W-W and P-W) displayed hypercellularity in the BM compartment, with a predominance of myeloid cells consistent with a systemic inflammatory response (Figure 5B). In contrast, mice lacking expression of PD-L1 on all cells (P-P) showed extensive necrosis of the BM, and the accelerated mortality in P-P mice may relate, in part, to the extensive marrow necrosis seen in this group. Mice lacking PD-L1 only on parenchymal cells (W-P) also displayed considerable necrosis of hematopoietic elements in the marrow. However, no marrow necrosis was seen in any of the W-W or P-W groups, suggesting that PD-L1 expression on nonhematopoietic cells is protective of fulminant marrow failure in LCMV CL-13 infection. In conclusion, our data show that expression of PD-L1 on nonhematopoietic cells is critical for controlling viral clearance and immunopathological damage during LCMV CL-13 infection, while expression on BM-derived cells controls the priming of T cell responses.
Discussion
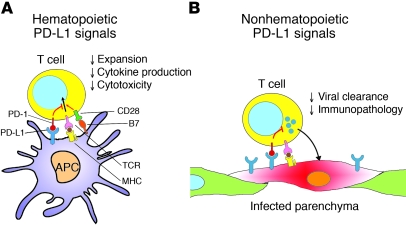
PD-1/PD-L1 interactions have been implicated in controlling tolerance, autoimmunity, and immune responses to a number of infectious diseases (2). We show here that there are differential roles for PD-L1 on BM- versus non-BM–derived cells during chronic LCMV infection. PD-L1 expression on hematopoietic cells inhibited the induction of antigen-specific CD4+ and CD8+ T cells responding to LCMV and reduced the number of polyfunctional IFN-γ+ and TNF-α+ virus–specific CD8+ T cells. We observed a small decrease in annexin V expression on CD8+ T cells in Pdl1–/– mice, suggesting that decreased apoptosis, as well as increased proliferation, may contribute to increased responses observed at day 8 when PD-L1 is lacking on BM cells. In contrast, PD-L1 expression on non-BM cells did not appear to influence early CTL numbers and function, since mice lacking PD-L1 on parenchymal cells generated similar responses to WT mice. Yet, we show that PD-L1 expression on nonhematopoietic cells in lymphoid and nonlymphoid tissues significantly influenced viral clearance. Together, these data point to distinct functions for PD-L1 on BM- and non-BM–derived cells (Figure 6) and suggest a mechanism by which PD-L1 in both compartments cooperates to control T cell priming and immune effector function during viral infection.
Figure 6. Model comparing roles of hematopoietic and nonhematopoietic cells in regulating T cell responses via the PD-1/PD-L1 pathway during infection.
(A) BM-derived cells, such as APCs, upregulate PD-L1 after infection and interact with PD-1+ antigen–specific T cells, negatively regulating cell expansion and cytokine production. Interactions between CD28 on T cells and B7 (CD80 or CD86) on APC costimulates antigen-specific TCR-MHC interactions and can be inhibited by concurrent PD-1/PD-L1 interactions. PD-L1 on T cells may also interact with CD80 on APCs to deliver inhibitory signals (not shown). (B) PD-L1 is upregulated on infected nonhematopoietic cells in tissues and regulates viral clearance and immunopathology during infection.
PD-L1 is expressed on numerous cell types, including lymphocytes, macrophages, DCs (see Supplemental Figure 1), and many parenchymal cells (2). It is interesting that expression of PD-L1 on BM-derived cells specifically reduced CTL responses, whereas expression on non-BM cells did not. This suggests that PD-L1 expression on APC and T cells may be instrumental in influencing T cell priming after infection. PD-L1 is constitutively expressed on DCs, and we show that its expression is markedly increased soon after chronic LCMV infection. This inhibitory pathway, in combination with high levels of antigen and inflammation very early after LCMV CL-13 infection, limits CTL priming and function. The altered tropism of LCMV CL-13, which infects DC (15) as well as fibroblastic reticular stromal cells in the lymphoid tissues (3), likely has a significant impact on the local environment in which virus-specific T cells are primed during chronic infection. Expression of PD-1 by other cells such as macrophages suggests that the PD-1/PD-L1 pathway also may control pathology by inhibiting many aspects of the immune response. Indeed, we observed increased numbers of TNF-α+ macrophages and systemic IL-6 in PD-L1–deficient mice (our unpublished observations), which may contribute to the lethal inflammatory response in these mice. Furthermore, the identification of PD-L1 and B7-1 (CD80) as a receptor-ligand pair and the fact that ligation of PD-L1 on T cells can inhibit T cell responses indicate that PD-L1 on the T cell as well as the APC may participate in delivering inhibitory signals during this early priming phase (16).
Although we observed enhanced responses by T cells responding to different viral epitopes in mice lacking BM-derived PD-L1, this did not result in improved clearance of virus from these mice, suggesting that the size of the T cell response is not always predictive of disease. The location and timing of inhibitory signals also can influence viral clearance versus persistence, as well as immunopathology. Epitope-specific T cells also may be differentially affected by PD-L1–mediated inhibitory signals, as evidenced by our findings that epitope-specific T cell responses were increased to different degrees in mice lacking BM-derived PD-L1 expression compared with WT mice (see Figure 2). For example, GP276-specific T cells were increased in P-W mice up to 12-fold over that in W-W mice, whereas GP33-specific responses were only 2- to 3-fold greater. Antigen levels, the relative abundance of individual epitopes presented on the surface of infected cells, as well as affinity for MHC molecules can differ between different epitopes and in different tissues (8). Antigen is a direct cause of T cell exhaustion during chronic viral infection (17). The antigenic history of responding T cell populations can influence PD-1 expression and, as a consequence, the functional effects of inhibition by PD-L1. Indeed, blockade of the PD-1 pathway in mice selectively restores the function of a subset of exhausted T cells (18), demonstrating that numerous factors can influence the function and responsiveness of antigen-specific T cells during chronic infection.
Chimeric mice lacking expression of PD-L1 on BM-derived cells did not demonstrate enhanced viral clearance when PD-L1 was present on non-BM cells in the tissues, suggesting that PD-L1 on non-BM cells can inhibit effector functions. Interestingly, PD-L1 expression on hematopoietic cells did appear to influence viral clearance and immunopathology in the liver. This may be similar to that observed in the liver after adenovirus infection (14), possibly via the involvement of Kupffer cells. Our data further suggest that PD-L1 expression on non-BM–derived cells, which are a significant target of CL-13 infection, likely influences the killing of infected non-BM–derived cells in the tissues. PD-L1 on non-BM cells appeared critical for preventing necrosis of hematopoietic precursors in the BM. LCMV infects stromal cells in the BM and causes pancytopenia, a process that involves IFN-α/β and CD8+ T cell–derived IFN-γ and TNF-α (19, 20). We find that PD-L1 expression on the non-BM–derived cells was important for reducing severe immunopathology, and lack of PD-L1 expression may have contributed to the more rapid mortality observed in these mice. It is also possible that the decrease in available mature BM cells due to necrosis contributed to immunopathology in mice lacking PD-L1 on parenchymal cells. Although the P-P and W-P chimeric mice did not show increased CTL responses, it is possible that PD-L1 signals also play a role in influencing CTL function in the tissues later during chronic infection after sustained interactions between T cells and PD-L1–expressing parenchymal cells. However, early lethality in the P-P and W-P chimeric mice prevented us from examining these potential interactions.
Maintaining the balance between CTL responses that clear virus from infected cells and preventing excessive tissue damage may involve different types of interactions. Tregs have been implicated in the suppression of CTL responses during infection (21). We observed similar numbers of Tregs in the spleen and blood and slightly elevated numbers in the liver of Pdl1–/– mice after LCMV CL-13 infection (Supplemental Figure 4). As Pdl1–/– mice are less capable of limiting T cell responses, the increased numbers of Treg cells may reflect an expansion of preexisting Tregs upon inflammatory insult. In addition, the absence of PD-L1 may also affect the suppressive capacity of Tregs. Recent work suggests that induced Tregs may rely upon PD-L1 expression for their generation and function (22). However, it is not known whether these cells are induced and play roles during chronic infections. There are data to suggest that PD-L1 blockade can enhance the function of Tregs obtained from individuals chronically infected with HCV (23). Multiple populations of Tregs exist, and it is not yet clear whether PD-1/PD-L1 interactions differentially control different Treg subtypes (e.g., natural Treg, iTreg, Tr1, Th3 cells). Further studies will be important to better understand the interactions and roles of Tregs in the tissues during chronic viral infections and how PD-1/PD-L1 interactions control Treg and effector T cell dynamics during chronic viral infections. However, due to the rapid demise of Pdl1–/– mice and their rapid development of immunopathology, these important questions may be better addressed in the chronic LCMV model using PD-1 or PD-L1 blockade.
Together, our findings demonstrate distinct roles for PD-L1 on BM- versus non-BM–derived cells in the control of T cell priming and function during a chronic viral infection (Figure 6). It is clear that the PD-1/PD-L1 pathway is critical during the priming and effector stages of the immune response after CL-13 infection, since mice succumbed to infection when expression was lacking on either compartment. Interestingly, mice were observed to die sooner after infection when PD-L1 was absent on parenchymal cells, demonstrating the importance of this pathway in reducing tissue pathology. Histopathological changes were more pronounced in some tissues such as the BM, lungs, and liver in mice lacking parenchyma PD-L1, suggesting that PD-L1 on non-BM cells predominates in protecting against immune-mediated tissue damage and may play different roles during the effector phase of CTL responses in different microenvironments.
In conclusion, we demonstrate distinct roles for PD-L1 expression on hematopoietic and nonhematopoietic cells in influencing T cell responses and viral clearance, respectively, during chronic LCMV CL-13 infection. These observations provide a better understanding of the mechanisms of immune function and inhibition by the PD-1/PD-L1 pathway during persisting infections and may assist in the design of appropriate therapeutic interventions that target this inhibitory pathway.
Methods
Mice, chimeras, and infections.
Six-week-old female C57BL/6 mice were purchased from Jackson Laboratory. Pdl1–/– mice (24) were housed and maintained in our animal facility. BM chimeras were produced by irradiation of recipient mice with 2 doses of 550 cGr, 3 hours apart, followed by reconstitution with 5 × 106 BM cells from C57BL/6 or Pdl1–/– mice as required. Mice were allowed to reconstitute for 8 weeks. For infection, mice received 2 × 106 PFU of LCMV CL-13 intravenously. Titers of virus from serum or homogenized tissue samples were determined by plaque assay on Vero cells as previously described (25). Mice were maintained and used according to institutional and NIH guidelines in a pathogen-free facility. All animal studies were approved by the IACUC of Emory University.
Cell isolation, antibodies, and staining.
For analysis of splenic BM-derived subsets, pieces of spleen were treated with 1 mg/ml collagenase type II (Worthington Biochemicals) and 1 mg/ml bovine pancreatic DNase I (Sigma-Aldrich) for 30 minutes at 37°C with gentle mixing. For the last 5 minutes of incubation, 10 mM EDTA was added to disrupt T-DC complexes. Cells were then passed through a 70-μM nylon mesh using a syringe plunger. Single-cell suspensions were stained with anti-CD8α (53-6.7), anti–PD-1 (J43), anti–PD-L1 (MIH5), anti–IFN-γ (XMG1.2), anti–TNF-α (MP6-XT22), anti-CD3 (145-2C11), anti-CD19 (ID3), anti-B220 (RA3-6B2), anti-NK1.1 (PK136), anti-CD49b (DX5), anti-CD11c (HL3), anti-CD11b (M1/70), anti-F4/80 (BM8), and anti–Gr-1 (RB6-8C5) antibodies (BD Biosciences). Intracellular staining for IFN-γ or TNF-α after 5 hours in vitro stimulation with 0.1 μg/ml of either GP33, GP276, NP396, NP235, or GP118 peptide was performed using the Cytofix/Cytoperm kit according to the manufacturer’s instructions (BD Biosciences). No peptide controls were used to confirm specificity of cytokine production. MHC class I tetramers were made and used as described previously (26).
Serum cytokine and liver enzyme analysis.
Serum levels of aspartate aminotransferase, alanine aminotransferase, and total bilirubin were measured colorimetrically on an Olympus AU640 automated analyzer (Olympus America) according to manufacturer’s instructions in the CLIA-certified Clinical Chemistry Laboratory at Brigham and Women’s Hospital. Values above assay range were diluted prior to remeasurement. Analysis of cytokines in serum was performed using a cytometric bead array mouse inflammation kit (BD Biosciences) according to the manufacturer’s instructions. Samples were analyzed using a BD FACSCalibur.
Histopathology and immunohistochemistry.
Organs were fixed in 10% phosphate-buffered formalin, dehydrated, and embedded in paraffin. Legs were decalcified briefly after fixation and prior to tissue processing. Five-micrometer paraffin tissue sections were stained with H&E for microscopic analysis. All slides were examined by at least one pathologist in a blinded fashion.
Sections of multiple liver lobes were evaluated under light microscopy and assessed for severity and activity of hepatitis. Portal inflammation and lobular inflammation were scored semiquantitatively in accordance with an established scoring system for viral hepatitis (27). Briefly, portal inflammation was scored from 0 (no portal inflammation) to 4 (severe portal inflammation and interface hepatitis); lobular inflammation was also scored from 0 (no inflammation) to 4 (severe inflammation with marked hepatocellular damage). Necrotic hepatocytes were also enumerated and averaged over 20 medium-magnification (×200) fields. The percentage of portal tracts with infiltrates or interface hepatitis (piecemeal necrosis) was also quantified.
For immunohistochemistry of formalin-fixed, paraffin-embedded sections, 5-micrometer sections were mounted on charged slides (Superfrost Plus; Fisher Scientific), baked for 1 hour at 60°C, deparaffinized, rehydrated, and subjected to pressure cooker antigen retrieval in a Pascal pressurized heating chamber (Dako) either in pH 6 citrate buffer (Dako) for B220 and Mac-2 or in pH 8 EDTA (Zymed). After blocking endogenous peroxidase (Dako), tissue sections were incubated with a rat monoclonal antibody to B220/CD45R (clone RA3-6B2; BD Biosciences), a rat monoclonal antibody to Mac-2/Galectin-3 (clone M3/38; eBioscience), or a rabbit polyclonal antibody to CD3 (Cell Marque) in Antibody Diluent (Dako) for 1 hour at room temperature. Sections were then incubated with unconjugated rabbit anti-rat IgG (Vector Laboratories) for 30 minutes (for the anti-B220 and anti–Mac-2 sections) and subsequently incubated for 40 minutes with horseradish peroxidase–conjugated rabbit-specific Envision polymer (Dako). Sections were developed using 3,3′-diaminobenzidine (Vector) followed by immersion in DAB Enhancer (Zymed) and counterstaining with hematoxylin. Photomicrographs were taken with a mounted digital camera (Olympus DP71) driven by Olympus DP Controller software. Images were prepared using Adobe Photoshop and Illustrator CS3 (Adobe Systems Inc.).
Statistics.
Statistical analysis was performed with 2-tailed unpaired Student’s t tests using Prism (GraphPad) software.
Supplementary Material
Acknowledgments
We would like to thank Neal Lindeman and Maria LeBruto in the Department of Pathology at Brigham and Women’s Hospital for assistance and expertise with liver function tests, and Loise Francisco for helpful discussions about regulatory T cell studies. This work was supported by grants from the NIH (AI56299 to A.H. Sharpe and R. Ahmed; AI30048 and AI04464409 to R. Ahmed; T32 HL007627 to Brigham and Women’s Hospital for V.K. Vanguri), and grants from the Gates Foundation Grand Challenges in Global Health to R. Ahmed, and the Korean Health Technology R&D project, Ministry for Health, Welfare & Family Affairs, Republic of Korea (A091204 to S.-J. Ha).
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Citation for this article: J Clin Invest. 2010;120(7):2508–2515. doi:10.1172/JCI40040.
Scott N. Mueller’s present address is: Department of Microbiology and Immunology, The University of Melbourne, Parkville, Victoria, Australia.
Sang-Jun Ha’s present address is: Department of Biochemistry, Yonsei University, Seodaemun-gu, Seoul, Korea.
Mary E. Keir’s present address is: ITGR Biomarkers Group, Genentech Inc., South San Franciso, California, USA.
References
- 1.Barber DL, et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2006;439(7077):682–687. doi: 10.1038/nature04444. [DOI] [PubMed] [Google Scholar]
- 2.Sharpe AH, Wherry EJ, Ahmed R, Freeman GJ. The function of programmed cell death 1 and its ligands in regulating autoimmunity and infection. Nat Immunol. 2007;8(3):239–245. doi: 10.1038/ni1443. [DOI] [PubMed] [Google Scholar]
- 3.Mueller SN, et al. Viral targeting of fibroblastic reticular cells contributes to immunosuppression and persistence during chronic infection. Proc Natl Acad Sci U S A. 2007;104(39):15430–15435. doi: 10.1073/pnas.0702579104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Probst HC, McCoy K, Okazaki T, Honjo T, van den Broek M. Resting dendritic cells induce peripheral CD8+ T cell tolerance through PD-1 and CTLA-4. Nat Immunol. 2005;6(3):280–286. doi: 10.1038/ni1165. [DOI] [PubMed] [Google Scholar]
- 5.Tsushima F, et al. Interaction between B7-H1 and PD-1 determines initiation and reversal of T-cell anergy. Blood. 2007;110(1):180–185. doi: 10.1182/blood-2006-11-060087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Keir ME, et al. Tissue expression of PD-L1 mediates peripheral T cell tolerance. J Exp Med. 2006;203(4):883–895. doi: 10.1084/jem.20051776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shin H, Wherry EJ. CD8 T cell dysfunction during chronic viral infection. Curr Opin Immunol. 2007;19(4):408–415. doi: 10.1016/j.coi.2007.06.004. [DOI] [PubMed] [Google Scholar]
- 8.Wherry EJ, Blattman JN, Murali-Krishna K, van der Most R, Ahmed R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J Virol. 2003;77(8):4911–4927. doi: 10.1128/JVI.77.8.4911-4927.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zajac AJ, et al. Viral immune evasion due to persistence of activated T cells without effector function. J Exp Med. 1998;188(12):2205–2213. doi: 10.1084/jem.188.12.2205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gallimore A, et al. Induction and exhaustion of lymphocytic choriomeningitis virus-specific cytotoxic T lymphocytes visualized using soluble tetrameric major histocompatibility complex class I-peptide complexes. J Exp Med. 1998;187(9):1383–1393. doi: 10.1084/jem.187.9.1383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Day CL, et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature. 2006;443(7109):350–354. doi: 10.1038/nature05115. [DOI] [PubMed] [Google Scholar]
- 12.Trautmann L, et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat Med. 2006;12(10):1198–1202. doi: 10.1038/nm1482. [DOI] [PubMed] [Google Scholar]
- 13.Petrovas C, et al. PD-1 is a regulator of virus-specific CD8+ T cell survival in HIV infection. J Exp Med. 2006;203(10):2281–2292. doi: 10.1084/jem.20061496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Iwai Y, Terawaki S, Ikegawa M, Okazaki T, Honjo T. PD-1 inhibits antiviral immunity at the effector phase in the liver. J Exp Med. 2003;198(1):39–50. doi: 10.1084/jem.20022235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sevilla N, et al. Immunosuppression and resultant viral persistence by specific viral targeting of dendritic cells. J Exp Med. 2000;192(9):1249–1260. doi: 10.1084/jem.192.9.1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ. Programmed death-1 ligand 1 interacts specifically with the B7-1 costimulatory molecule to inhibit T cell responses. Immunity. 2007;27(1):111–122. doi: 10.1016/j.immuni.2007.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mueller SN, Ahmed R. High antigen levels are the cause of T cell exhaustion during chronic viral infection. Proc Natl Acad Sci U S A. 2009;106(21):8623–8628. doi: 10.1073/pnas.0809818106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Blackburn SD, Shin H, Freeman GJ, Wherry EJ. Selective expansion of a subset of exhausted CD8 T cells by alphaPD-L1 blockade. Proc Natl Acad Sci U S A. 2008;105(39):15016–15021. doi: 10.1073/pnas.0801497105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Binder D, Fehr J, Hengartner H, Zinkernagel RM. Virus-induced transient bone marrow aplasia: major role of interferon-alpha/beta during acute infection with the noncytopathic lymphocytic choriomeningitis virus. J Exp Med. 1997;185(3):517–530. doi: 10.1084/jem.185.3.517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Binder D, et al. Aplastic anemia rescued by exhaustion of cytokine-secreting CD8+ T cells in persistent infection with lymphocytic choriomeningitis virus. J Exp Med. 1998;187(11):1903–1920. doi: 10.1084/jem.187.11.1903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Keynan Y, Card CM, McLaren PJ, Dawood MR, Kasper K, Fowke KR. The role of regulatory T cells in chronic and acute viral infections. Clin Infect Dis. 2008;46(7):1046–1052. doi: 10.1086/529379. [DOI] [PubMed] [Google Scholar]
- 22.Francisco LM, et al. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J Exp Med. 2009;206(13):3015–3029. doi: 10.1084/jem.20090847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Franceschini D, et al. PD-L1 negatively regulates CD4+CD25+Foxp3+ Tregs by limiting STAT-5 phosphorylation in patients chronically infected with HCV. J Clin Invest. 2009;119(3):551–564. doi: 10.1172/JCI36604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Latchman YE, et al. PD-L1-deficient mice show that PD-L1 on T cells, antigen-presenting cells, and host tissues negatively regulates T cells. Proc Natl Acad Sci U S A. 2004;101(29):10691–10696. doi: 10.1073/pnas.0307252101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ahmed R, Salmi A, Butler LD, Chiller JM, Oldstone MB. Selection of genetic variants of lymphocytic choriomeningitis virus in spleens of persistently infected mice. Role in suppression of cytotoxic T lymphocyte response and viral persistence. J Exp Med. 1984;160(2):521–540. doi: 10.1084/jem.160.2.521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Murali-Krishna K, et al. Counting antigen-specific CD8 T cells: a reevaluation of bystander activation during viral infection. Immunity. 1998;8(2):177–187. doi: 10.1016/S1074-7613(00)80470-7. [DOI] [PubMed] [Google Scholar]
- 27.Batts KP, Ludwig J. Chronic hepatitis. An update on terminology and reporting. Am J Surg Pathol. 1995;19(12):1409–1417. doi: 10.1097/00000478-199512000-00007. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.