Abstract
Tuberculosis (TB) has a worthy reputation as one of the great mimickers in medicine with a multitude of clinical pictures and variations. Noncontiguous spinal TB is described as atypical and case reports are published as rarities in the mainstream academic journals. The aim of the study was to asses the incidence and review of the management of non-contiguous spinal TB. We identified 16 cases of noncontiguous spinal TB from a single surgeon series of 98 patients, who were managed surgically between 2001 and 2006. These were diagnosed on whole spine MRI. This represents the largest series reported in literature to date and is higher than the expected incidence. Case notes and imaging were retrospectively reviewed in an attempt to ascertain if there were any parameters to differentiate this group from the rest of the TB spine population. Our incidence of noncontiguous spinal TB is 16.3%. There was a higher incidence of neurology in the noncontiguous group (75%) compared to the rest of our group (58.5%). Non-contiguous TB was not found to be a manifestation of HIV, MDR TB or of chronicity in our series. Most noncontiguous lesions were evident on plain radiology. Noncontiguous spinal TB is common in areas of high prevalence such as South Africa. Despite being frequently missed initially, noncontiguous involvement is evident on plain radiography and simply requires a higher index of suspicion. When investigating spine TB patients, simple radiology of the entire spine is mandatory. If available, a full spine sagittal MRI is extremely useful in identifying noncontiguous lesions. Treatment of noncontiguous tuberculosis is as for standard spinal TB cases in our unit with similar outcomes, but care needs to be taken in surgical planning as patients may have multiple areas of neurological compromise.
Keywords: Tuberculosis, Noncontiguous, Infection, Spinal, Multi-site
Introduction
Tuberculosis (TB), a disease that once was on the decline and almost eradicated in the first world, has made a dramatic comeback. According to the World Health Organisation, TB is the number one specific infectious cause of death worldwide [23]. It is estimated that one-third of the world’s population is infected with TB, an estimated 2 billion people.
South Africa has the world’s highest reported incidence of tuberculosis at 718/100,000 reaching 1.2% of the population in certain areas. In the Western Cape Province in 2005, there were 48,000 registered cases. There has been a steady 8–10% annual increase in cases, and a 32% re-treatment rate in 2005. In the busiest clinics in Cape Town, up to 46% of pulmonary TB cases are not confirmed bacteriologically. The financial burden of TB is estimated to cost South Africa around 250 million US dollars per year [23].
Extrapulmonary tuberculosis consists of 15–20% of these patients if you include pleural and lymphatic disease, which is most common. Skeletal TB is 10% of this, of which spinal TB accounts for approximately 50%. This gives an incidence of between 1 and 2% for osteoarticular TB and 0.5–1% for spinal TB [15].
Tuberculosis has a worthy reputation as one of the great mimickers in medicine with a multitude of clinical pictures and variations. This makes the description of the classic disease problematic, with most texts describing a typical clinical and radiological picture followed by many atypical derivatives. Although TB spondylitis frequently involves multiple adjacent spinal vertebrae, noncontiguous, remote involvement is reported as rare in literature.
Groote Schuur Hospital is a tertiary level referral centre in Cape Town, where a surprising number of our spinal TB patients were noted to have noncontiguous spinal lesions, i.e., discrete foci of spinal involvement with intervening normal vertebrae and no evidence of a connecting soft tissue abscess. This led to this review.
Materials and methods
A prospective database of all TB patients managed surgically is maintained by the senior author. Over a period of July 2001 to December 2006, 98 consecutive cases were reviewed to identify cases with noncontiguous spinal involvement; 16 cases were identified and the case notes and radiology reviewed.
Data collection
Epidemiological data and the length of history were collected. All patients had plain radiology of the spine, a chest X-ray and magnetic resonance imaging of the whole spine. The patient’s notes were reviewed for the history, presenting and follow-up neurology on the Frankel grading, type of surgery as well as laboratory results. Plain radiographs of the spine were reviewed prior to MRI scan review.
Diagnosis
The spinal diagnostic procedure was usually an open surgical biopsy with one radiologically guided percutaneous needle biopsy. Biopsies were subjected to microscopic examination for acid-fast bacilli after auramine staining and were inoculated into two culture mediums, a solid medium (Lowenstein–Jensen) and a liquid medium (MGIT fluorescent system). Specimens were cultured for a total of 6 weeks before being declared negative. Histological studies of biopsy samples were performed to look for tuberculoid granulomas, Langhans giant cells and caseous necrosis. Mantoux skin testing was performed in three adolescent children.
Management
Patient management consisted of chemotherapy, attention to nutrition supplementation and surgery in the majority of cases. The surgical procedure performed was case dependent, ranging from diagnostic to radical debridement and reconstruction. Four drug antituberculous chemotherapy (rifampicin, isoniazid, pyrazinamide and ethambutol) was initiated in hospital and continued on an outpatient basis at community clinics for a minimum of 9 months according to clinical, radiological and blood parameters. HIV-positive patients were treated for 18 months routinely during the period of this study.
Surgery
Surgery was considered on an individualised basis. The majority of the 16 patients had surgery on a single focus only. Twelve patients had anterior radical debridement, decompression and strut grafting (Hong Kong-type procedure) [10]. Five of these had a posterior fusion as an additional procedure. Three patients had posterior procedures only, viz. one biopsy, one instrumented fusion and one laminectomy. One patient had surgery at two distinct levels.
Results
A total of 16 patients with noncontiguous spinal lesions were identified in the 98 TB spine cases reviewed, yielding an incidence of 16.3%.
In the noncontiguous group, the average age was 34.4 years (12–68 years, SD 21.1) with nine females and seven males. The average ESR was 76.8 mm/h (30–106 mm/h, SD 21.2). The white cell count average was 7.7 × 109 (4.2–15.9, SD 2.94). Of the 12 patients tested for HIV, only 1 was positive. The remaining 82 patients from the series had a single spinal lesion identified. Their average age was 32.14 years (2–68 years, SD 19.98) with 47 females and 35 males. The average ESR was 74.3 mm/h (12–143 mm/h, SD 32.7). The white cell count average was 8.5 × 109 (3.8–20.2, SD 3.6). Of the 48 patients tested for HIV, 10 were positive.
Comparing the two groups for sex, age, length of history, presentation and outcome showed no significant difference. The blood parameter differences were also not significantly different; ESR P = 0.772 (Student’s t test) WCC P = 0.39 (Student’s t test).
Of the 16 cases, 12 (75%) had neurological deficit (Frankel A–D) as opposed to 48/82 (58.5%) with single focus involvement. Whilst suggestive, this trend was not statistically significant (P = 0.27), using the two-tailed Fisher’s exact test. With regard to the ambulatory status (Frankel A, B or C) in those patients with neurology, 9/12 (75%) from the noncontiguous group were nonambulatory at presentation versus 38/49 (77.5%) in the single focus group. This was not statistically significant (P = 0.7) using the two-tailed Fisher’s exact test.
One of the noncontiguous groups died (an elderly diabetic with paraplegia and significant co morbidities, who became septicaemic in ICU post-surgery.). Of the remaining 11 patients with a neurological deficit, all improved. Eight patients were nonambulatory at presentation, seven improved to be ambulant (Frankel D or E) on completion of treatment. The one nonambulant outcome was an elderly, demented, chronically noncompliant patient who improved from a Frankel B to a C.
The combination of microbiology, skin testing and histology confirmed the diagnosis in 15/16 patients. The results of the diagnostic tests are depicted in Fig. 1. One patient, who was on prolonged treatment prior to surgery, had no supportive histology or microbiology findings. The diagnosis of TB was made due to the history, imaging, macroscopic findings and good response to treatment.
Fig. 1.
Diagnostic testing indicating that histology gave the highest yield with 11 positive, 3 suggestive (granuloma 0 and only 2 negative). When combined with other modalities, 13 were confirmed positive and 2 suggestive
In our series, histology was more sensitive than microbiology. Microbiology produced 8 positive cultures out of the 15 performed. Six of the cultures were sensitive to first-line TB drugs, whereas the cultures of two patients showed multi-drug resistant (MDR) TB. Both of these cases were incompletely treated prior to presentation. The initial diagnosis of MDR TB was made on clinical grounds and a poor response to standard treatment. Second-line drugs were introduced with a rapid favourable response and confirmation obtained when sensitivities became available after approximately 6 weeks. There were no cases of extremely drug-resistant (XDR) TB cultured in either group.
Besides poor nutritional status, three patients had immunosuppressive factors, possibly contributing to their condition. One was HIV-positive, one diabetic and one pregnant. Routine HIV testing was only done in the latter part of the series. A total of 12 patients were tested for HIV, 11 were negative and one positive and already on retroviral medication (8.3%). Of the 48 single focus patients tested, 10 were found to be positive (20.8%). There was no significant difference between the groups (P value of 0.43) using the two-tailed Fisher’s exact test.
On plain films, 15/16 patients had at least one typical spinal TB focus. The HIV positive patient had no lesion on plain radiology. Of the 15 with X-ray changes, almost all had clear evidence of noncontiguous lesions on the plain films. These varied from typical to atypical, such as missing pedicles, scalloping of the bodies, disc space loss and centrosomic osseous lesions.
One patient presenting to us with paraplegia was worked up, scanned and referred to neurosurgery colleagues as a metastatic malignancy on the basis of the MRI scan, with the biopsies only coming back as tuberculosis. An MRI scan is by no means diagnostic [13].
With regard to the distribution of lesions, 11 patients had two noncontiguous foci involved, two patients had three foci and two had four discrete lesions (Figs. 3, 4, 5). The distribution of involved vertebrae showed a predilection for the lower thoracic and lumbar vertebrae, in keeping with distribution as described by Hodgson [19]. All 16 patients had a thoracic focus, and 14 had a thoracic and a lumbar focus. The distribution of affected levels is depicted in Fig. 2. The red indicates the single focus levels involved, whilst the blue represents the multifocus cases.
Fig. 3.

A 16-year-old girl who presented complaining only of persistent vague neck pain after a fall. Apart from a clinical neck deformity, she was systemically asymptomatic, completely neurologically intact and HIV negative. On MRI, atlanto-axial, subaxial, thoracic and lumbar TB were identified. She was managed by means of anterior cervical surgery and medical management
Fig. 4.
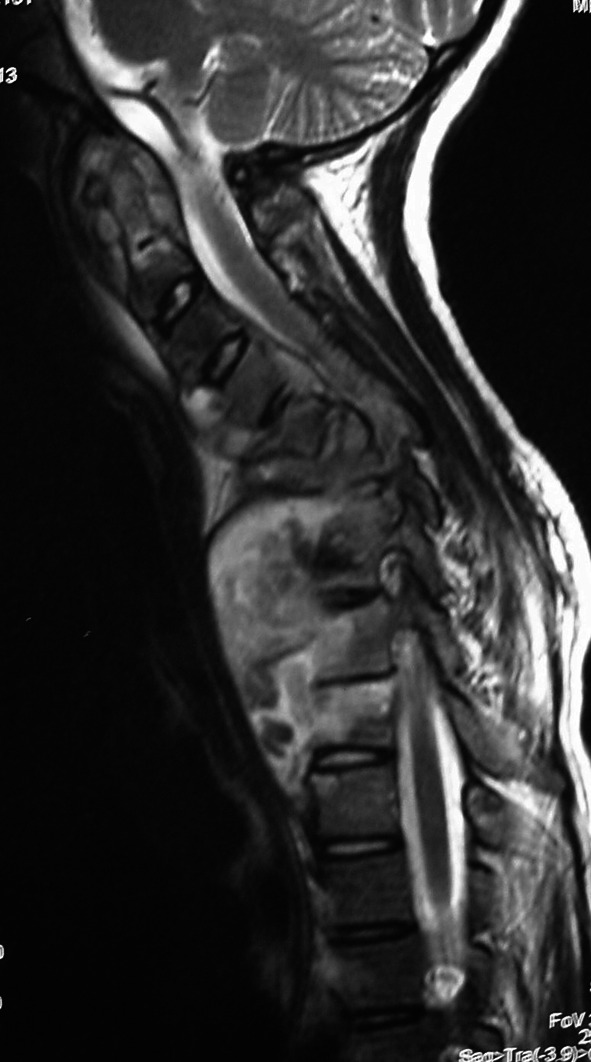
A 16-year-old girl who presented complaining only of persistent vague neck pain after a fall. Apart from a clinical neck deformity, she was systemically asymptomatic, completely neurologically intact and HIV negative. On MRI, atlanto-axial, subaxial, thoracic and lumbar TB were identified. She was managed by means of anterior cervical surgery and medical management. Cevical MRI of patient in Fig. 3 demonstrating atlanto-axial and sub-axial involvement
Fig. 5.
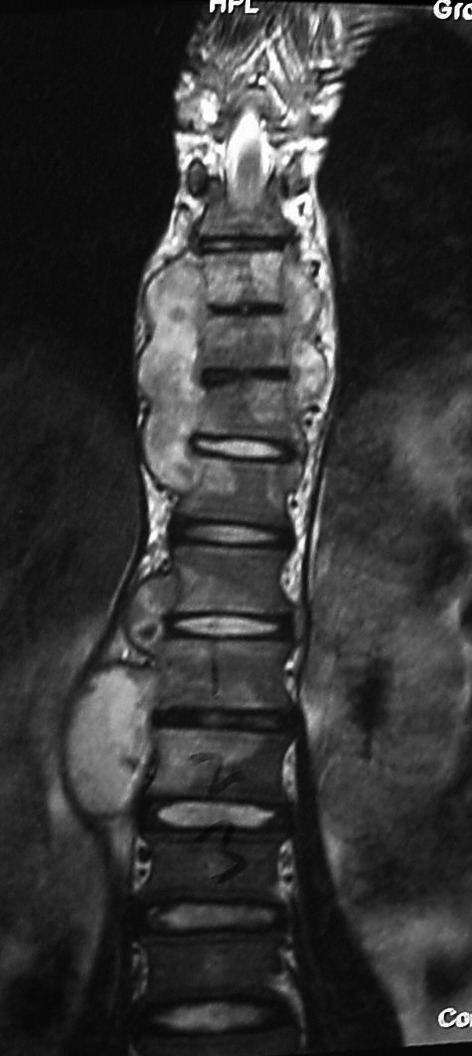
A 16-year-old girl who presented complaining only of persistent vague neck pain after a fall. Apart from a clinical neck deformity, she was systemically asymptomatic, completely neurologically intact and HIV negative. On MRI, atlanto-axial, subaxial, thoracic and lumbar TB were identified. She was managed by means of anterior cervical surgery and medical management. MRI of case in Fig. 3 depicting thoracic and lumbar involvement
Fig. 2.
Distribution of involved vertebral bodies. Noncontiguous in blue and single focus in red
Discussion
Although TB spondylitis frequently involves multiple adjacent spinal vertebrae, noncontiguous, remote involvement is reported as rare. A literature review produced mostly episodic single case reports [2, 6, 7, 9, 11, 13, 21, 22, 24]. A case report of three foci spinal TB in “British Journal Neurosurgery 2001” was described as the first such report in English language literature [7, 21]. Turgut reported 1 out of 694 cases, Rezai et al. 1/20, Nussbaum 1/29, and a nuclear medicine study from Saudi Arabia 3/63 [14].
One paper stands out in reporting a 25% incidence of multicentricity in spinal TB. This study by Pandit et al. [17] was a bone scan study of 40 confirmed cases of TB spine. There was, however, a 25% (10/40) incidence of skeletal multicentricity reported rather than spinal multicentricity. Of the 40 patients, 6 had vertebral skip lesions giving an incidence for noncontiguous spinal TB involvement of 15%. No comment was made as to whether these multiple lesions were confirmed on plain radiographs or MRI scans. Pandit et al’s finding of a 15% incidence is much higher than reported in the other papers, but correlates well with our findings of a 16.3% incidence.
The treatment regimes utilised in these studies varied between purely medical and a combination of chemotherapy and surgery, all reported with favourable results.
The classic appearance of spinal TB on plain films is well described [8, 12]. The lesion is typically seen to affect two or more adjacent vertebral bodies with loss of the intervening disc space, often accompanied by a paravertebral soft tissue shadow. Atypical radiographic features are described by Pande et al.: centrosomic (single isolated body), which may progress to vertebrae plana and be confused with malignancy; “ivory vertebrae”; isolated neural arch involvement; circumferential or pan-vertebral involvement, which causes significant instability; and multiple vertebral disease, which may be in continuity or noncontiguous [16, 18].
In a study of 103 cases by Pertuiset et al. [18], pathology consistent with spinal TB was evident on plain films in 94% of patients. The literature reports the sensitivity of plain radiography to be between 91 and 99%. In our series, 15/16 patients had at least one typical spinal TB focus. Of the 15 with X-ray changes, almost all had clear evidence of noncontiguous lesions on the plain films. These varied from typical to atypical.
The HIV positive patient had no lesion on plain radiology; however, on MRI and at surgery he had granulomas in the canal and a dorsal soft tissue abscess without any bony destruction. This is a recognised atypical form of spinal TB known as “extradural tuberculomas” or “spinal tumour syndrome” [3, 16]. There was a second intraosseous lesion in L2 on MRI and at follow-up there were bony changes evident on the plain radiographs.
Although there is a recognised association between HIV and TB, this was not evident in our series with only one positive case. The CD4 lymphocytes play an integral part in the development and maintenance of the cellular response, which develops against TB. It is this cellular immune response, which is responsible for the development of granulomas and cell-mediated immunity, which prevents reactivation of latent bacilli, and new infections. It is this very cellular response, which is reduced by HIV infection. This may explain the lack of bony destruction evident in our HIV-positive case. Bono [5] commented that “MRI findings in patients with HIV and TB can be deceptively benign”.
The reasons for the high incidence of noncontiguous disease is unclear. It may well be due to the high prevalence of tuberculosis in the community with delayed presentation of a few months before the diagnosis is made. This series may have a selection bias as all patients were operated on. However, in Western Cape, spinal tuberculosis is not managed in smaller hospitals due to limited access to MRI and specialist staff. They are generally referred to one of the two tertiary care centres, of which we are one. A minimum of a biopsy or abscess debridement is performed, which would have allowed the data to be included in this study. The most likely reason is the increased accuracy of diagnosis with the use of MRI and the more recently established protocol of including a full spine sagittal sequence. One suspects that many lesions are overlooked on X-ray once one obvious lesion is identified.
There are a number of proposed mechanisms of spinal TB involvement: the valveless venous system; arterial seeding; lymphatic spread; direct invasion from adjacent viscera/nodes and via spread along soft tissue planes [8, 16, 20, 21].
With noncontiguous TB, it is probable that the venous system is involved in the pathological process. Once the first TB focus is established in the spine by arterial/venous/lymphatic inoculation, there will be shedding of bacilli into the valveless venous plexus, which allows retrograde flow with changes in abdominal pressure. This would allow the bacilli to travel to new vertebrae without being filtered through the lungs or lymphatic system and thus establish “skip lesions”, much like that done by sarcomas in the venous sinusoids of long bones. This would explain how a patient may present with the involvement of multiple, isolated spinal levels and no other obvious pulmonary/visceral/bony involvement. The alternative would have to be multiple haematogenous arterial seedings, preferentially targeting different spinal areas, which is less likely.
Tuberculosis has a propensity to spread along soft tissue planes, particularly anteriorly under the anterior longitudinal ligament (ALL) involving multiple vertebrae, in time giving the so-called “aneurysmal syndrome” (scalloping of the anterior vertebral margins), as described by Ghormley and Bradley [4]. It may also affect some vertebrae more obviously than others on X-ray. This sub-ALL collection will not be evident on bone scan due to stripping of the soft tissue and vasculature [17]. Thus, plain radiographs and bone scan may give a false impression of noncontiguous TB. The MRI scans of our series were reviewed and no soft tissue abscess communications could be visualised, confirming the cases as true noncontiguous lesions.
Conclusion
Noncontiguous spinal TB is far more common (16.3%) than that reported in literature. There was a trend towards a higher incidence of neurology in the noncontiguous group compared to the single focus. This trend may not necessarily reflect more severe disease, but instead may be a product of heightened probability due to the increased levels involved and/or the double-crush neural phenomenon.
Noncontiguous spinal TB is not an obvious manifestation of HIV, MDR TB or chronicity in our series. It is not a “fulminant” type of TB as suggested by Emel et al. [7], as some of our most severe cases on imaging studies were neurologically intact with minimal systemic symptoms and good outcomes with standard care.
Noncontiguous spinal TB is usually evident on plain radiography, but it requires a higher index of suspicion. MRI scanning is extremely useful for assisting in the diagnosis and surgical planning, but is by no means diagnostic. Confusion, particularly with metastatic malignancy, is possible and a tissue diagnoses is still essential [1, 8, 9, 16].
We recommend that the entire spine should be x-rayed in TB spine patients, and preferably a whole spine sagittal MRI performed. Once identified, the lesions should be managed based on their merit.
References
- 1.Ahmadi J, Bajaj A, Destian S, Segall HD, Chi-Shing Z. Spinal tuberculosis: atypical observations at MR imaging. Radiology. 1993;189(2):489–493. doi: 10.1148/radiology.189.2.8210378. [DOI] [PubMed] [Google Scholar]
- 2.Amogne W, Abubaker A. Multifocal vertebral tuberculosis with involvement of the ribs: a case report. Ethiop Med J. 2002;40(4):397–405. [PubMed] [Google Scholar]
- 3.Babhulkar SS, Tayade WB, Babhulkar SK. Atypical spinal tuberculosis. J Bone Joint Surg Br. 1984;66-B(2):239–242. doi: 10.1302/0301-620X.66B2.6707060. [DOI] [PubMed] [Google Scholar]
- 4.Bailey HL, Gabriel M, Hodgson AR, Shin JS. Tuberculosis of the spine in children. J Bone Joint Surg Am. 1972;54:1633–1657. [PubMed] [Google Scholar]
- 5.Bono CM. Spectrum of spine infections in patients with HIV: a case report and review of the literature. Clin Orthop Relat Res. 2006;444:83–91. doi: 10.1097/01.blo.0000203450.28899.ef. [DOI] [PubMed] [Google Scholar]
- 6.Dickinson FL, Finlay DB, Belton IP. Multifocal skeletal tuberculosis: bone scan appearances. Nucl Med Commun. 1996;17(11):957–962. doi: 10.1097/00006231-199611000-00006. [DOI] [PubMed] [Google Scholar]
- 7.Emel E, et al. Noncontiguous multifocal spinal tuberculosis involving cervical, thoracic, lumbar and sacral segments: a case report. Eur Spine J. 2006;15:1019–1024. doi: 10.1007/s00586-005-0989-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Griffith JF et al. (2002) Imaging of musculoskeletal tuberculosis: a new look at an old disease. Clin Orthop Relat Res (398): 32–39. doi:10.1097/00003086-200205000-00006 [DOI] [PubMed]
- 9.Hasegawa K, Murata H, Naitoh K, Nagano A (2002) Spinal tuberculosis: report of an atypical presentation. Clin Orthop Relat Res (403):100–103. doi:10.1097/00003086-200210000-00016 [PubMed]
- 10.Hodgson AR, Stock FE. Anterior spinal fusion: a preliminary communication on the radical treatment of potts disease and Potts paraplegia. Br J Surg. 1956;XLIV:266. doi: 10.1002/bjs.18004418508. [DOI] [PubMed] [Google Scholar]
- 11.Islam N, Ahmedani MY. Cervical spine tuberculosis. J Coll Physicians Surg Pak. 2004;14(8):499–500. [PubMed] [Google Scholar]
- 12.Jevtic V. Vertebral infection. Eur Radiol. 2004;14(suppl 3):E43–E52. doi: 10.1007/s00330-003-2046-x. [DOI] [PubMed] [Google Scholar]
- 13.Sj Kim, Seok JW, Kim IJ, Kim YK, Kim DS. Multifocal Potts disease incidentally detected on Tc-99 m MDP bone and Ga-67 citrate scintigraphy in a patient with diabetes. Clin Nucl Med. 2003;28(4):286–289. doi: 10.1097/00003072-200304000-00003. [DOI] [PubMed] [Google Scholar]
- 14.Lindahl S, Nyman R, Brismar J, Hugsson C, lundstedt C. Imaging of tuberculosis: spinal manifestations in 63 patients. Acta Radiol. 1996;37(4):506–511. doi: 10.3109/02841859609175433. [DOI] [PubMed] [Google Scholar]
- 15.Naim-ur-Rahman Atypical forms of spinal tuberculosis. J Bone Joint Surg Br. 1980;62(2):162–165. doi: 10.1302/0301-620X.62B2.7364827. [DOI] [PubMed] [Google Scholar]
- 16.Pande KC, Babhulkar SS (2002) Atypical spinal tuberculosis. Clin Orthop Relat Res (398):67–74. doi:10.1097/00003086-200205000-00010 [DOI] [PubMed]
- 17.Pandit HG, Sonsale PD, Shikare SS, Bhojraj SY. Bone scintgraphy in tuberculous spondylodiscitis. Eur Spine J. 1999;8:205–209. doi: 10.1007/s005860050159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pertuiset E, et al. Spinal tuberculosis in adults: a study of 103 cases in a developed country, 1980–1994. Medicine (Baltimore) 1999;78(5):309–320. doi: 10.1097/00005792-199909000-00003. [DOI] [PubMed] [Google Scholar]
- 19.Rothman Rh, FA Simeone. The spine. Philadelphia: WB Saunders; 1975. [Google Scholar]
- 20.Rothman RH, Simeone FA. The spine. 5. Philadelphia: WB Saunders; 2006. [Google Scholar]
- 21.Turgut M. Multifocal extensive spinal tuberculosis (Potts disease) involving cervical, thoracic and lumbar vertebrae. Br J Neurosurg. 2001;15(2):142–146. doi: 10.1080/02688690120036856. [DOI] [PubMed] [Google Scholar]
- 22.Uno M, et al. A case report of atypical tuberculous spondylitis. Kansenshogaku Zasshi. 1994;68(8):982–985. doi: 10.11150/kansenshogakuzasshi1970.68.982. [DOI] [PubMed] [Google Scholar]
- 23.World Health Organisation website: http:/www.who.int
- 24.Yalniz E, Pekindil G, Aktas S. Atypical tuberculosis of the spine. Yonsei Med J. 2000;41(5):657–661. doi: 10.3349/ymj.2000.41.5.657. [DOI] [PubMed] [Google Scholar]




