Abstract
Objective
T and B lymphocytes are associated with atherosclerosis and aneurysms. Angiotensin II (AngII) infusion into hypercholesterolemic mice results in augmentation of atherosclerosis and abdominal aortic aneurysm (AAA) formation. In this study, we determined whether total lymphocyte deficiency reduced AngII-induced vascular diseases.
Methods and Results
ApoE deficient (apoE −/−) mice were cross-bred with recombination activating gene-1 (Rag-1) deficient mice that lack mature T and B lymphocytes. Heterozygous littermates (Rag-1 +/−) have normal lymphocytic function and served as controls. Male and female apoE −/− mice that were either Rag-1 +/− or −/− were fed a normal laboratory diet and infused with either saline or AngII (1,000 ng/kg/min) subcutaneously via osmotic mini pump for 28 days. Total lymphocyte deficiency had no significant effect on body weight and systolic blood pressure prior to and during AngII infusion. However, it was associated with decreased serum cholesterol concentrations. AngII infusion increased atherosclerotic lesion area in Rag-1 +/− mice compared to saline (P = 0.017 in males and P = 0.004 in females). This effect was significantly blunted in Rag-1 −/− male (P = 0.044), but not female mice. AngII-infusion promoted increased width of the abdominal aorta, with a greater effect in males. Despite the reduction in atherosclerosis in males, Rag-1 deficiency had no significant effect on AngII-induced aortic dilation in either gender.
Conclusion
T and B lymphocyte deficiency attenuates AngII-induced atherosclerosis in males but not AAA formation in apoE −/− mice.
Keywords: angiotensin II, lymphocytes, atherosclerosis, aneurysm
INTRODUCTION
Chronic subcutaneous infusion of angiotensin II (AngII) into hypercholesterolemic mice results in augmented atherosclerotic lesion formation [1,2]. Similar to atherosclerotic tissue formed in hypercholesterolemic mice, AngII-induced lesions are characterized by infiltration of macrophages and T lymphocytes [1,3]. Monocyte deficiency virtually ablates development of atherosclerotic lesions that form in hypercholesterolemic mice in the absence and presence of AngII infusion.[4,5,6]. Total lymphocyte deficiency more modestly reduces atherosclerosis, although this effect has gender specificity and does not occur under conditions of severe hypercholesterolemia [7,8]. It has not been defined whether total lymphocyte deficiency has an effect on the development of AngII-augmented atherosclerosis.
Infusion of AngII into mice also leads to the formation of abdominal aortic aneurysms (AAAs) [1,9]. Development of AngII-induced AAAs is characterized by pronounced luminal expansion that is associated with complex temporal changes in cells and extracellular matrix components [10]. Macrophages infiltrate the aortic media of the aneurysm-prone region early in the disease process, and are prominent throughout the disease progression. T lymphocytes are also prominent in aneurysmal tissue following luminal dilation. Unlike atherosclerosis, B lymphocytes are also present in AngII-induced aneurysmal disease, as occurs in the human disease [3,10,11]. Although leukocytic infiltrates are prominent in AngII-induced aneurysmal disease, there have been no studies to define the function of specific leukocytic cell types on aortic dilation.
In the present study, we have examined the effects of total lymphocyte deficiency on the development of AngII-induced atherosclerosis and AAAs in apoE −/− mice. This was achieved by developing apoE −/− mice that lacked the recombination activating gene-1 (Rag-1) gene. Rag-1 deficiency leads to an inability to perform VDJ recombination which prevents the formation of mature T and B lymphocytes. Although total lymphocyte deficiency reduced the size of AngII-augmented atherosclerotic lesions in males, it had no detectable effect on development of AAAs.
MATERIAL AND METHODS
Mice and Diet
Rag-1 −/− (stock # 002216) and apoE −/− mice (stock # 002052) on C57BL/6 background (N10) were purchased from the Jackson Laboratory (Bar Harbor). Rag-1 −/− females were bred to apoE−/− males with subsequent breeding to develop littermates of Rag-1 +/− and −/− x apoE −/− mice. Rag-1 +/− mice are immunocompetent. All mice were maintained in a barrier facility and fed a normal laboratory diet. No mice developed any immunological or infectious disorders. All study procedures were approved by the University of Kentucky Institutional Animal Care and Use Committee.
Mouse Genotyping and Phenotyping
Mouse genotypes were determined by PCR. DNA was isolated from tail snip samples of mice using DNeasy tissue kits (Qiagen). Rag-1 genotyping used the following primers: 5'-ACTCAATTCTGACTCAACG, 5'-AACAGATGTCACAGGACG, and 5'-AGAAAGTATCCATCATGGC. The PCR cycle was: 35 cycles − 95°C for 1 min, 49.5°C for 1 min, 72°C for 2 min, and 1 elongation cycle of 72°C for 7 min. The wild type and disrupted alleles generated amplicons of 1.1 kb and 425 bp, respectively. Spleen sections were analyzed by immunostaining for mature T and B lymphocytes to verify phenotype as described previously [10]. ApoE genotypes were defined by PCR as described previously.[12]
AngII Infusion and Blood Pressure Measurement
Saline or AngII (1,000 ng/kg/min; Bachem) was infused into 8–10 week old male and female mice subcutaneously via Alzet osmotic minipumps (Model 2004, Durect Corp) for 28 days as described previously [1]. Systolic blood pressure (SBP) was measured on conscious mice for 5 consecutive days prior to and the last week during AngII infusion using a Visitech tail cuff system, as described previously [13].
Serum Total Cholesterol Measurement
Serum total cholesterol concentrations were evaluated in individual serum samples with a commercially available enzymatic-based kit (Wako Chemical) [8].
Quantification of Atherosclerosis and Aneurysm
Areas of atherosclerotic lesions covering the aortic arch and thorax were measured by en face methods as described previously [14]. AAAs were quantified as described previously [13]. Briefly, aortas were pinned and suprarenal aortic diameters were measured using Image-Pro software. AAA incidence was defined as a 50% increase in external aortic diameter.
Statistics
Continuous data are summarized as mean ± SEM. Two-way ANOVA with follow-up pairwise comparisons (t tests) were used to examine body weight and serum total cholesterol concentration. Because the usual assumptions for two-way ANOVA were questionable with the atherosclerosis and abdominal aortic diameter data, we analyzed these data using the nonparametric Kruskal Wallis procedure with follow-up pairwise comparisons (Z tests). A repeated measures ANOVA was performed to evaluate blood pressure data. Fisher's exact test was used to determine differences between groups in the incidence of AAA. Analyses were performed using SigmaStat 3.5 or SAS 9.2 software. The specific test used is indicated in the Table and Figure legends. Results with P < 0.05 were deemed statistically significant.
EXPERIMENTAL RESULTS
Rag-1 Deficiency on Characteristics of ApoE −/− Mice Prior to and During AngII infusions
Total lymphocyte deficiency had no significant effect on body weights in both male and female mice (Table 1). AngII-infused Rag-1 −/− mice had lower serum total cholesterol concentration compared to Rag-1 +/− mice in both males and females (P = 0.004 and P < 0.001, respectively).
Table 1.
Effects of AngII infusion into apoE −/− mice that were either Rag-1 +/− or −/−.
| Infusion | Rag-1 genotype | Male | Female | ||||
|---|---|---|---|---|---|---|---|
| n | Body weight (g) | Cholesterol (mg/dl) | n | Body weight (g) | Cholesterol (mg/dl) | ||
| Saline | +/− | 6 | 31.7 ± 1.2 | 291 ± 17 | 5 | 23.6 ± 1.0 | 194 ± 16 |
| −/− | 5 | 30.8 ± 0.5 | 265 ± 19 | 4 | 22.6 ± 0.4 | 152 ± 14 | |
| AngII | +/− | 16 | 29.3 ± 0.5 | 334 ± 20 | 18 | 23.2 ± 0.4 | 291 ± 19** |
| −/− | 15 | 29.4 ± 0.8 | 256 ± 19* | 19 | 22.8 ± 0.4 | 219 ± 12*** | |
Body weight and serum cholesterol concentrations were measured at the end of the study. Values are represented as mean ± SEM. Statistical analyses were performed by two way ANOVA.
Denotes P = 0.004 for comparison of versus Rag-1 +/− treated with AngII.
Denotes P = 0.003 for comparison of versus saline in Rag-1 +/−.
Denotes P < 0.001 for comparison of versus Rag-1 +/− infused with AngII.
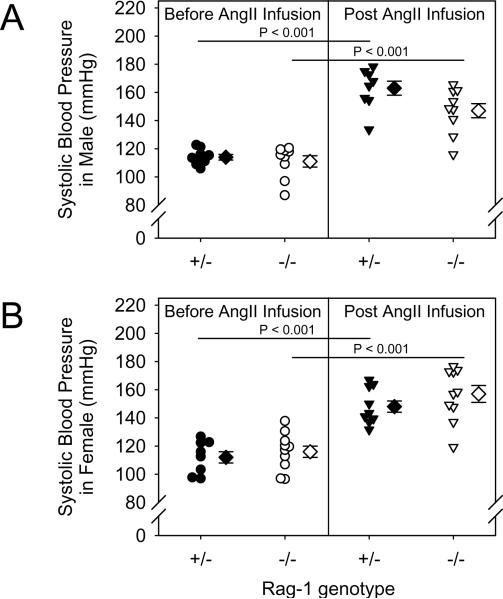
Rag-1 deficiency had no significant effect on SBP at baseline in both male and female mice compared to controls. Chronic AngII infusion (1,000 ng/kg/min) increased systolic blood pressure in Rag-1 +/− mice similar to Rag-1 −/− mice in both males and females (Fig. 1A and B, P < 0.001 for both genotypes and genders). There were no significant differences in the increase of SBP between Rag-1 +/− and −/− mice during AngII infusion in both males and females.
Figure 1. Total lymphocyte deficiency had no significant effect on basal or AngII-induced systolic blood pressure.
Systolic blood pressure was determined from the mean of measurements for 5 consecutive days prior to and the last week during AngII infusion in male (A) and female groups (B). Histobars represent means and bars are SEMs. Statistical analyses were performed by ANOVA with repeated measures.
Rag-1 Deficiency Attenuated AngII-induced Atherosclerotic Lesion Formation in Male Mice
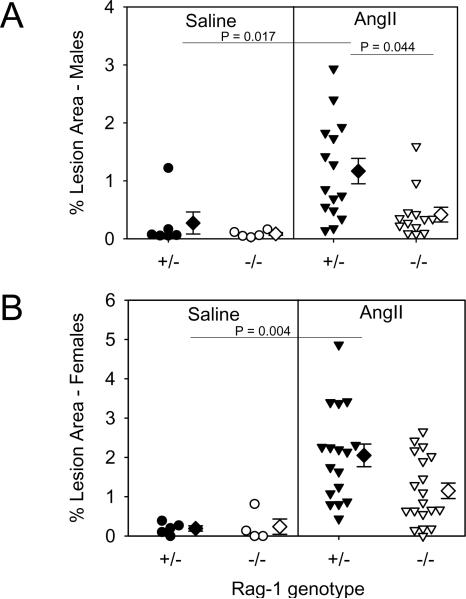
Atherosclerotic lesion size was quantified on the intimal surface of arches and thoracic regions of aortas. Abdominal segments were excluded because of the occurrence of AAAs by AngII infusion. Lesions were either absent or extremely small in mice infused with saline for 28 days. AngII increased aortic atherosclerotic lesions in both male and female Rag-1 +/− mice (P = 0.017 and P = 0.004, respectively), which was significantly reduced in males (P = 0.044), but not in female Rag-1 −/− mice (Fig. 2A and B). Also, AngII infusion failed to significantly increase the size of atherosclerotic lesions in male Rag-1 −/− mice, but did in female mice (P = 0.021).
Figure 2. Total lymphocyte deficiency reduced AngII-induced atherosclerosis in male mice.
The percent area of grossly discernable atherosclerotic lesions was measured in the arch and thoracic regions of aortas recovered from apoE −/− mice that are either Rag-1 +/− or −/− in male (A) and female groups (B). The area for each individual mouse is represented by circle or inverted triangle; diamonds represent means and bars represent SEMs. Statistical analyses were performed by Kruskal Wallis with follow up Z test.
Rag-1 Deficiency Had no Significant Effect on AngII-induced AAAs
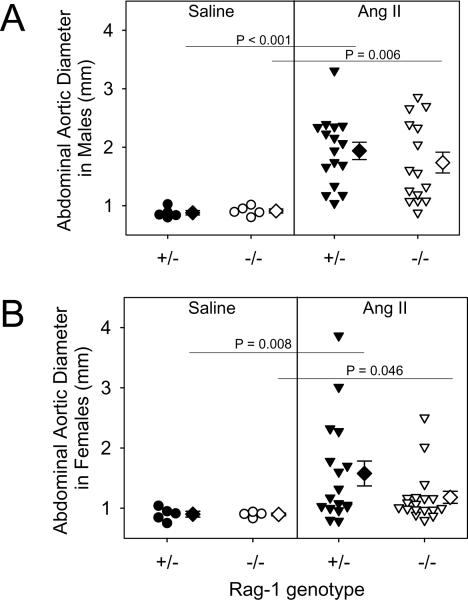
AAAs were not detected in saline-infused mice of both genotypes and genders. During AngII infusion, 4 Rag-1 +/− and 3 Rag-1 −/− male, and 1 Rag-1+/− and 1 Rag-1 −/− female mice died due to rupture of the aneurysm-prone aortic region. AngII increased the maximal external diameters of suprarenal aortas in Rag-1 +/− male mice (0.88 ± 0.04 versus 1.94 ± 0.15 mm, saline and AngII-infused, respectively; P < 0.001), while female mice had a more modest increase (0.90 ± 0.05 versus 1.58 ± 0.20, saline and AngII-infused, respectively; P = 0.008). For both genders, Rag-1 deficiency had no significant effect on maximal external diameters of the suprarenal region compared to heterozygous mice (Fig. 3A and B). AAA incidence in male mice was 84% and 69% in Rag-1 +/− and −/−, respectively (not statistically significant). Furthermore, AAA incidence in females was not significantly different between Rag-1 +/− and −/−.
Figure 3. Total lymphocyte deficiency had no significant effect on the size and incidence of AngII-induced AAAs.
Size was measured as maximal diameter of suprarenal aortas in male (A) and female mice (B). The diameter for each individual mouse is represented by circle or inverted triangle; diamonds represent means, and bars are SEMs. Statistical analyses were performed by Kruskal Wallis with follow up Z test.
DISCUSSION
Many groups have consistently demonstrated that chronic subcutaneous Angll infusion into hypercholesterolemic mice augments atherosclerosis and leads to AAA development [15]. The effects of AngII on atherosclerosis have no gender specificity, while males have a greater susceptibility to developing AAAs [16,17]. The arterial tissue in both vascular diseases is characterized by accumulation of lymphocytes. Consistent with studies in modestly hypercholesterolemic mice, in the present study, Angll augmented atherosclerosis was attenuated in males by total lymphocyte deficiency [7]. However, total lymphocyte deficiency had no detectable effect on the development of Angll-induced AAAs.
Angll has well known vasoconstrictor properties and a sufficient rate of infusion increases blood pressure. It has been consistently demonstrated that infusion rates of 1,000 ng/kg/min or above lead to persistent increases in systolic blood pressure in conscious hypercholesterolemic mice [1,9,18,19]. However, this increased blood pressure, per se, is not directly responsible for the profound development of Angll-induced vascular pathology [20]. This has been demonstrated most directly by infusing hypercholesterolemic mice with norepinephrine to promote similar increases in systolic blood pressure to Angll-infused mice [2]. In contrast to Angll infusion, norepinephrine infusion had only a minor effect in augmenting atherosclerosis and did not lead to AAA formation [2,21]. In the present study, we observed no significant effect of Rag-1 deficiency on systolic blood pressure, either prior to AngII infusion, or during the 28 day interval of infusion. In contrast, a recent report has demonstrated Rag-1 deficiency had no significant effect on basal systolic blood pressure, but attenuated Angll-induced increases [22]. Strain differences do not account for the differences since most of the Angll infusions into hypercholesterolemic mice have been performed using the C57BL/6 strain; as were the studies of Guzik et al [22]. A difference in the experimental designs is the use of normo- versus hypercholesterolemic apoE −/− mice. Increased plasma cholesterol concentrations have well characterized effects on vascular contractility and relaxation [23,24]. Further studies will be needed to determine whether plasma cholesterol concentrations and characteristics of lipoproteins may explain the divergent results.
Several studies have determined the effects of total lymphocyte deficiency, either in Rag-1 or Rag-2 deficiency, on atherosclerosis development in hypercholesterolemic mice. These studies have demonstrated that total lymphocyte deficiency reduced hypercholesterolemia-induced atherosclerosis in both apoE −/ and LDL receptor −/− mice, with a more profound effect in males [7,25,26,27]. However, this protective effect is either transient or lost in apoE −/− or LDL receptor −/− mice when they are rendered severely hypercholesterolemic by feeding saturated fat-enriched diets [7,8,27]. In the present study, apoE −/− mice were fed a normal laboratory diet and infused with AngII. Similar to the previous studies in modest hypercholesterolemic states, total lymphocyte deficiency only significantly reduced the size of atherosclerotic lesions in males [7]. A modest reduction in plasma cholesterol concentrations may contribute to the reduced atherosclerosis [26]. Current studies indicate that mechanisms of hypercholesterolemia induced atherosclerosis are the same as those of AngII augmented disease. Indeed, previous studies have demonstrated that inhibition of the renin angiotensin system by pharmacological or genetic approaches, profoundly reduces atherosclerosis in hypercholesterolemic mice [28,29,30].
Many studies have demonstrated that AngII infusion into mice leads to AAA development, which is more prevalent in males as confirmed in the current study [1,9,16,17,19]. Both T and B lymphocytes have been detected in aneurysmal tissue of AngII-infused mice [10]. Despite the presence of these cells in aneurysmal tissue of wild type mice, their total absence had no significant effect on the incidence or maximal diameter of AAAs in either gender. Interferon gamma is a major cytokine released by T lymphocytes and its deficiency augments AngII-induced AAAs [31]. However, interferon gamma is released from several cell types other than T lymphocytes, including natural killer cells, macrophages, and smooth muscle cells. Therefore, the combination of these studies is consistent with this previously described effect of interferon gamma on AngII-induced AAAs not being attributable to release of this cytokine from lymphocytes. While total lymphocyte deficiency had no detectable effect on the development of AngII-induced AAAs during a 28 day interval, this does not preclude a possible role for these cell types in subsequent progression. Also, our study was not powered to detect small effect sizes, so our negative results should not be interpreted as definitively ruling out minor effects of total lymphocyte deficiency. It should be noted that Rag-1 deficiency also influences other non lymphocytic leukocyte populations, such as macrophages and NK cells [32,33]. Therefore, while Rag-1 deficiency has the direct effect of inhibiting development of mature lymphocytes, there may also be indirect effects on other types of leukocytes.
In conclusion, these studies demonstrate that total lymphocyte deficiency due to Rag-1 deficiency had no detectable effect on development of AngII-induced AAAs in apoE −/− mice. Furthermore, this study provides another illustration of the disparate mechanisms in the formation of AngII-induced atherosclerosis compared to AAAs [17,31,34].
ACKNOWLEDGMENTS
This work was supported by a grant from the NHLBI (P01 HL80100). Haruhito A. Uchida was partially supported by a fellowship grant from the Sumitomo Life Social Welfare Services Foundation, Japan. We acknowledge the skilled technical assistance of Deborah A. Howatt and Jessica J. Moorleghen.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Daugherty A, Manning MW, Cassis LA. Angiotensin II promotes atherosclerotic lesions and aneurysms in apolipoprotein E-deficient mice. J Clin Invest. 2000;105:1605–1612. doi: 10.1172/JCI7818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Weiss D, Kools JJ, Taylor WR. Angiotensin II-induced hypertension accelerates the development of atherosclerosis in ApoE-deficient mice. Circulation. 2001;103:448–454. doi: 10.1161/01.cir.103.3.448. [DOI] [PubMed] [Google Scholar]
- 3.Roselaar SE, Kakkanathu PX, Daugherty A. Lymphocyte populations in atherosclerotic lesions of apoE −/− and LDL receptor −/− mice. Decreasing density with disease progression. Arterioscler Thromb Vasc Biol. 1996;16:1013–1018. doi: 10.1161/01.atv.16.8.1013. [DOI] [PubMed] [Google Scholar]
- 4.Smith JD, Trogan E, Ginsberg M, Grigaux C, Tian J, Miyata M. Decreased atherosclerosis in mice deficient in both macrophage colony-stimulating factor (op) and apolipoprotein E. Proc Natl Acad Sci USA. 1995;92:8264–8268. doi: 10.1073/pnas.92.18.8264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rajavashisth T, Qiao JH, Tripathi S, Tripathi J, Mishra N, Hua M, Wang XP, Loussararian A, Clinton S, Libby P, Lusis A. Heterozygous osteopetrotic (op) mutation reduces atherosclerosis in LDL receptor-deficient mice. J Clin Invest. 1998;101:2702–2710. doi: 10.1172/JCI119891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Babamusta F, Rateri DL, Moorleghen JJ, Howatt DA, Li XA, Daugherty A. Angiotensin II infusion induces site-specific intra-laminar hemorrhage in macrophage colony-stimulating factor-deficient mice. Atherosclerosis. 2005;186:282–290. doi: 10.1016/j.atherosclerosis.2005.08.006. [DOI] [PubMed] [Google Scholar]
- 7.Dansky HM, Charlton SA, Harper MM, Smith JD. T and B lymphocytes play a minor role in atherosclerotic plaque formation in the apolipoprotein E-deficient mouse. Proc Natl Acad Sci USA. 1997;94:4642–4646. doi: 10.1073/pnas.94.9.4642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Daugherty A, Pure E, Delfel-Butteiger D, Chen S, Leferovich J, Roselaar SE, Rader DJ. The effects of total lymphocyte deficiency on the extent of atherosclerosis in apolipoprotein E−/− mice. J Clin Invest. 1997;100:1575–1580. doi: 10.1172/JCI119681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang YX, Martin McNulty B, Freay AD, Sukovich DA, Halks Miller M, Li WW, Vergona R, Sullivan ME, Morser J, Dole WP, Deng GG. Angiotensin II increases urokinase-type plasminogen activator expression and induces aneurysm in the abdominal aorta of apolipoprotein E-deficient mice. Am J Pathol. 2001;159:1455–1464. doi: 10.1016/S0002-9440(10)62532-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Saraff K, Babamusta F, Cassis LA, Daugherty A. Aortic dissection precedes formation of aneurysms and atherosclerosis in angiotensin II-infused, apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2003;23:1621–1626. doi: 10.1161/01.ATV.0000085631.76095.64. [DOI] [PubMed] [Google Scholar]
- 11.Ocana E, Bohorquez JC, Perez-Requena J, Brieva JA, Rodriguez C. Characterisation of T and B lymphocytes infiltrating abdominal aortic aneurysms. Atherosclerosis. 2003;170:39–48. doi: 10.1016/s0021-9150(03)00282-x. [DOI] [PubMed] [Google Scholar]
- 12.Zhang SH, Reddick RL, Piedrahita JA, Maeda N. Spontaneous hypercholesterolemia and arterial lesions in mice lacking apolipoprotein E. Science. 1992;258:468–471. doi: 10.1126/science.1411543. [DOI] [PubMed] [Google Scholar]
- 13.Cassis LA, Rateri DL, Lu H, Daugherty A. Bone marrow transplantation reveals that recipient AT1a receptors are required to initiate angiotensin II-induced atherosclerosis and aneurysms. Arterioscler Thromb Vasc Biol. 2007;27:380–386. doi: 10.1161/01.ATV.0000254680.71485.92. [DOI] [PubMed] [Google Scholar]
- 14.Daugherty A, Whitman SC. Quantification of atherosclerosis in mice. Methods Mol Biol. 2003;209:293–309. doi: 10.1385/1-59259-340-2:293. [DOI] [PubMed] [Google Scholar]
- 15.Lu H, Rateri DL, Cassis LA, Daugherty A. The role of the renin-angiotensin system in aortic aneurysmal diseases. Curr Hypertens Rep. 2008;10:99–106. doi: 10.1007/s11906-008-0020-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Henriques TA, Huang J, D'Souza SS, Daugherty A, Cassis LA. Orchiectomy, but not ovariectomy, regulates angiotensin II-induced vascular diseases in apolipoprotein E deficient mice. Endocrinology. 2004;145:3866–3872. doi: 10.1210/en.2003-1615. [DOI] [PubMed] [Google Scholar]
- 17.Henriques T, Zhang X, Yiannikouris FB, Daugherty A, Cassis LA. Androgen increases AT1a receptor expression in abdominal aortas to promote angiotensin II-induced AAAs in apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2008;28:1251–1256. doi: 10.1161/ATVBAHA.107.160382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bruemmer D, Collins AR, Noh G, Wang W, Territo M, Arias-Magallona S, Fishbein MC, Kintscher U, Fleck E, Law RE, Hsueh WA. Angiotensin II accelerated atherosclerosis and aneurysm formation is attenuated in osteopontin deficient mice. J Clin Invest. 2003;112:1318–1331. doi: 10.1172/JCI18141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thomas M, Gavrila D, McCormick ML, Miller FJ, Jr, Daugherty A, Cassis LA, Dellsperger KC, Weintraub NL. Deletion of p47phox attenuates angiotensin II-induced abdominal aortic aneurysm formation in apolipoprotein E-deficient mice. Circulation. 2006;114:404–413. doi: 10.1161/CIRCULATIONAHA.105.607168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lu H, Cassis LA, Daugherty A. Atherosclerosis and arterial blood pressure in mice. Curr Drug Targets. 2007;8:1181–1189. doi: 10.2174/138945007782403829. [DOI] [PubMed] [Google Scholar]
- 21.Cassis LA, Gupte M, Thayer S, Zhang X, Charnigo R, Howatt DA, Rateri DL, Daugherty A. Angiotensin II infusion promotes abdominal aortic aneurysms independent of increased blood pressure in hypercholesterolemic mice. Am J Physiol Heart Circ Physiol. 2009;296:H1660–1665. doi: 10.1152/ajpheart.00028.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guzik TJ, Hoch NE, Brown KA, McCann LA, Rahman A, Dikalov S, Goronzy J, Weyand C, Harrison DG. Role of the T cell in the genesis of angiotensin II induced hypertension and vascular dysfunction. J Exp Med. 2007;204:2449–2460. doi: 10.1084/jem.20070657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nickenig G, Jung O, Strehlow K, Zolk O, Linz W, Scholkens BA, Bohm M. Hypercholesterolemia is associated with enhanced angiotensin AT1-receptor expression. Am J Physiol. 1997;272:H2701–7. doi: 10.1152/ajpheart.1997.272.6.H2701. [DOI] [PubMed] [Google Scholar]
- 24.Bonthu S, Heistad DD, Chappell DA, Lamping KG, Faraci FM. Atherosclerosis, vascular remodeling, and impairment of endothelium- dependent relaxation in genetically altered hyperlipidemic mice. Arterioscler Thromb Vasc Biol. 1997;17:2333–40. doi: 10.1161/01.atv.17.11.2333. [DOI] [PubMed] [Google Scholar]
- 25.Elhage R, Clamens S, Reardon Alulis C, Getz GS, Fievet C, Maret A, Arnal JF, Bayard F. Loss of atheroprotective effect of estradiol in immunodeficient mice. Endocrinology. 2000;141:462–465. doi: 10.1210/endo.141.1.7377. [DOI] [PubMed] [Google Scholar]
- 26.Reardon CA, Blachowicz L, White T, Cabana V, Wang YG, Lukens J, Bluestone J, Getz GS. Effect of immune deficiency on lipoproteins and atherosclerosis in male apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2001;21:1011–1016. doi: 10.1161/01.atv.21.6.1011. [DOI] [PubMed] [Google Scholar]
- 27.Song L, Leung C, Schindler C. Lymphocytes are important in early atherosclerosis. J Clin Invest. 2001;108:251–259. doi: 10.1172/JCI11380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wassmann S, Czech T, Van Eickels M, Fleming I, Bohm M, Nickenig G. Inhibition of diet-induced atherosclerosis and endothelial dysfunction in apolipoprotein E/Angiotensin II type 1A receptor double-knockout mice. Circulation. 2004;110:3062–3067. doi: 10.1161/01.CIR.0000137970.47771.AF. [DOI] [PubMed] [Google Scholar]
- 29.Daugherty A, Rateri DL, Lu H, Inagami T, Cassis LA. Hypercholesterolemia stimulates angiotensin peptide synthesis and contributes to atherosclerosis through the AT1A receptor. Circulation. 2004;110:3849–3857. doi: 10.1161/01.CIR.0000150540.54220.C4. [DOI] [PubMed] [Google Scholar]
- 30.Lu H, Rateri DL, Feldman DL, RJ, Fukamizu A, Ishida J, Oesterling EG, Cassis LA, Daugherty A. Renin inhibition reduces hypercholesterolemia-induced atherosclerosis in mice. J Clin Invest. 2008;118:984–993. doi: 10.1172/JCI32970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.King VL, Lin AY, Kristo F, Anderson TJ, Ahluwalia N, Hardy GJ, Owens AP, 3rd, Howatt DA, Shen D, Tager AM, Luster AD, Daugherty A, Gerszten RE. Interferon-gamma and the interferon-inducible chemokine CXCL10 protect against aneurysm formation and rupture. Circulation. 2009;119:426–435. doi: 10.1161/CIRCULATIONAHA.108.785949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sendobry SM, Cornicelli JA, Welch K, Grusby MJ, Daugherty A. Absence of T lymphocyte-derived cytokines fails to diminish macrophage 12/15-lipoxygenase expression in vivo. J Immunol. 1998;161:1477–82. [PubMed] [Google Scholar]
- 33.Grundy MA, Sentman CL. Immunodeficient mice have elevated numbers of NK cells in non-lymphoid tissues. Exp Cell Res. 2006;312:3920–3926. doi: 10.1016/j.yexcr.2006.08.019. [DOI] [PubMed] [Google Scholar]
- 34.Manning MW, Cassis LA, Daugherty A. Differential effects of doxycycline, a broad-spectrum matrix metalloproteinase inhibitor, on angiotensin II-induced atherosclerosis and abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol. 2003;23:483–488. doi: 10.1161/01.ATV.0000058404.92759.32. [DOI] [PubMed] [Google Scholar]