Abstract
Analogs of vitamin D attenuate renal injury in several models of kidney disease, but the mechanism underlying this renoprotective effect is unknown. To address the role of the vitamin D receptor (VDR) in renal fibrogenesis, we subjected VDR-null mice to unilateral ureteral obstruction for 7 days. Compared with wild-type mice, VDR-null mice developed more severe renal damage in the obstructed kidney, with marked tubular atrophy and interstitial fibrosis. Significant induction of extracellular matrix proteins (fibronectin and collagen I), profibrogenic and proinflammatory factors (TGF-β, connective tissue growth factor, and monocyte chemoattractant protein 1), and epithelial-to-mesenchymal transition accompanied this histologic damage. Because VDR ablation activates the renin-angiotensin system and leads to accumulation of angiotensin II (AngII) in the kidney, we assessed whether elevated AngII in the VDR-null kidney promotes injury. Treatment with the angiotensin type 1 antagonist losartan eliminated the difference in obstruction-induced interstitial fibrosis between wild-type and VDR-null mice, suggesting that AngII contributes to the enhanced renal fibrosis observed in obstructed VDR-null kidneys. Taken together, these results suggest that the VDR attenuates obstructive renal injury at least in part by suppressing the renin-angiotensin system.
Interstitial fibrosis is a hallmark of chronic renal failure and strongly correlates with deterioration of renal function, regardless of the underlying disease. Unilateral ureteral obstruction (UUO) is a well-established model of tubulointerstitial fibrosis of the kidney. In this model, damage of the obstructed kidney is accompanied by tubular atrophy and progressive interstitial fibrosis, as a result of excessive production and deposition of extracellular matrix (ECM) in the interstitium.1 The ECM proteins (e.g., fibronectin [FN], collagens) are secreted from myofibroblasts derived from resident interstitial fibroblasts or from transformed epithelial cells, a process termed epithelial-to-mesenchymal transition (EMT).2 During this transition process, the epithelial cells lose epithelial markers and gain mesenchymal markers. With the loss of epithelial cell properties, myofibroblasts proliferate, migrate, and produce and deposit a large amount of ECM. In addition, infiltration of immune cells, particularly macrophages, which secret numerous profibrotic factors, is important for the progression of tubulointerstitial fibrosis.3
Angiotensin II (AngII) and TGF-β are two major fibrogenic factors that mediate fibrogenesis in the kidney.4 The intrarenal renin-angiotensin system (RAS) is activated in obstructive nephropathy, leading to increased production of AngII within the kidney.3 AngII promotes renal fibrosis directly via the angiotensin type 1 receptor (AT1R) or activation of the TGF-β pathway.5 TGF-β is a potent profibrotic factor that directly stimulates the expression of many ECM proteins in renal cells6 and promotes EMT in the kidney.2 AngII and TGF-β also activate other fibrogenic factors, such as connective tissue growth factor (CTGF), which also plays an important role in renal fibrosis and EMT.7,8
The vitamin D receptor (VDR) mediates the activity of 1,25-dihydroxyvitamin D3 [1,25(OH)2D3], the active hormonal form of vitamin D.9 VDR is highly expressed in the kidney and has been shown to play a renoprotective role by targeting the RAS.10 1,25(OH)2D3 negatively regulates renin expression,11 and VDR deletion leads to hyperreninemia and activation of the RAS.12 Mice carrying VDR-null mutation develop substantially worse diabetic renal injury than their wild-type diabetic counterparts,13 and vitamin D analogs can prevent or attenuate renal injury in a number of experimental models of kidney diseases.14–17 Liu and colleagues reported that a vitamin D analog was able to ameliorate tubulointerstitial fibrosis by blocking EMT in the UUO model.18 In this study, by using this model, we demonstrate that VDR attenuates renal fibrosis in vivo by suppressing the RAS.
Results
VDR(−/−) Mice Develop More Severe Renal Fibrosis in the UUO Model
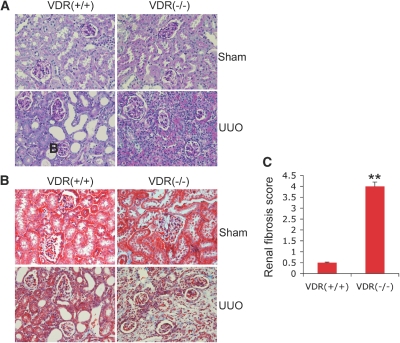
To determine the role of VDR in renal fibrogenesis, we subjected wild-type [VDR(+/+)] and VDR knockout [VDR(−/−)] mice to UUO and analyzed the mice on day 7 after surgery. Histologic examination by periodic acid-Schiff (PAS; Figure 1A) and Masson's trichrome (Figure 1B) staining showed normal renal cortex in the sham-operated kidney from either VDR(+/+) or VDR(−/−) mice. The obstructed VDR(+/+) kidney exhibited prominent dilation with sloughed cells or cellular debris in the tubular lumina, but no obvious interstitial fibrosis or tubular atrophy was detectable. In contrast, the obstructed VDR(−/−) kidney showed marked reduction in kidney mass with severe thinning of the renal cortex; the glomeruli were clustered close together, indicating substantial tubular loss. There was severe tubular atrophy and interstitial fibrosis in the obstructed VDR(−/−) kidney, as shown by the strong collagen deposition (blue staining in Figure 1B) in the interstitium. Semiquantitative scoring (on a scale of 0 to 4) confirmed that all VDR(−/−) mice examined developed severe interstitial fibrosis, whereas VDR(+/+) mice showed only mild renal fibrosis within 7 days of UUO (Figure 1C).
Figure 1.
VDR-null mice develop more severe renal fibrosis in UUO. VDR(+/+) and VDR(−/−) mice are subjected to sham-operation or UUO surgery and killed after 7 days. (A) PAS-stained sections from sham-operated and obstructed kidneys of VDR(+/+) and VDR(−/−) mice. (B) Masson's trichrome–stained sections from VDR(+/+) and VDR(−/−) mice. (C) Semiquantitative score (on a scale of 0 to 4) of renal fibrosis in VDR(+/+) and VDR(−/−) mice. **P < 0.01 versus VDR(+/+) (n = 4).
We then examined the expression of ECM protein FN and collagen I, surrogate markers of renal fibrosis, in the kidney. Quantitative reverse transcriptase–PCR (RT-PCR) showed a marked increase in FN and collagen I in the obstructed kidney compared with the sham controls; however, the increase in VDR(−/−) mice was more robust than in VDR(+/+) mice (Figure 2A). Immunostaining with anti-FN antibody showed strong staining in the fibrotic areas that was more intense in the obstructed VDR(−/−) kidney than in the VDR(+/+) counterpart (Figure 2B). Western blot analyses of total kidney lysates confirmed the more robust induction of FN protein in the obstructed VDR(−/−) kidney (Figure 2, C and D).
Figure 2.
More robust induction of ECM proteins in VDR-null mice. (A) Real-time RT-PCR quantification of FN and collagen I expression in sham-operated and obstructed kidneys from VDR(+/+) and VDR(−/−) mice. (B) Immunostaining of kidney sections with anti-FN antibody. Note the strong staining in the obstructed VDR(−/−) kidney. (C) Western blot analysis of FN levels in total lysates from sham-operated and obstructed kidneys. (D) Densitometric quantification of FN proteins. *P < 0.05 versus sham of the same genotype; #P < 0.05 versus VDR(+/+)-UUO (n = 4 to 5). Each lane represents one mouse.
Induction of Fibrogenic and Inflammatory Factors
RT-PCR showed that TGF-β and CTGF were induced in the obstructed kidney in both VDR(+/+) and VDR(−/−) mice, but the induction was more robust in VDR(−/−) mice than in the VDR(+/+) counterpart (Figure 3). The expression of monocyte chemoattractant protein 1 (MCP-1), a chemokine promoting macrophage infiltration, was also highly induced in the obstructed kidney; however, the induction was also more dramatic in VDR(−/−) mice than in VDR(+/+) mice (Figure 3). This is consistent with a previous observation that MCP-1 is downregulated by vitamin D hormone19; therefore, VDR deficiency leads to more robust induction of profibrotic and proinflammatory factors in UUO.
Figure 3.
More robust induction of pro-fibrotic and pro-inflammatory factors in VDR-null mice. (A) RT-PCR examination of TGF-β, CTGF, and MCP-1 in sham-operated and obstructed kidneys from VDR(+/+) and VDR(−/−) mice. (B) Densitometric quantification of the RT-PCR data. *P < 0.05, **P < 0.01 versus sham of the same genotype; #P < 0.05, ##P < 0.01 versus VDR(+/+)-UUO. Each lane represents one mouse.
VDR(−/−) Mice Show More Robust EMT
EMT is a key feature of UUO. E-cadherin, an epithelial marker, was moderately reduced at mRNA and protein levels in the obstructed VDR(+/+) kidney, consistent with the relatively well-preserved tubular structure; in contrast, the obstructed VDR(−/−) kidney showed very dramatic reduction of E-cadherin (Figure 4A), with mRNA and protein levels reduced by >80% (Figure 4B) relative to the sham VDR(+/+) control. The loss of epithelial markers accompanied the appearance of fibroblast markers in the obstructed kidney. α-Smooth muscle actin (α-SMA), a marker of myofibroblasts,20 was barely detectable in sham-operated kidneys (Figure 4, C and D), and immunostaining showed positive staining only in the smooth muscle layer of the blood vessels in sham-operated kidneys (Figure 4G). UUO led to marked upregulation of α-SMA in the obstructed kidneys, as shown by Western blot (Figure 4, C and D) and immunostaining (Figure 4G). The induction was much more robust in the obstructed VDR(−/−) kidney than in the VDR(+/+) counterpart (Figure 4, C, D, and G). The induction of α-SMA was mainly localized in the highly fibrotic area (Figure 4G). Consistently, Snail, the transcription factor known to play a key role in EMT,21 was also highly induced in the obstructed kidney, and, as expected, Snail induction was much higher in the obstructed VDR(−/−) kidney than in the VDR(+/+) counterpart (Figure 4, E and F).
Figure 4.
VDR ablation promotes EMT. (A) Western blot analysis of E-cadherin (E-cad) in sham-operated and obstructed kidneys from VDR(+/+) and VDR(−/−) mice. (B) Real time RT-PCR and densitometric quantifications of E-cadherin mRNA and protein levels in kidneys from VDR(+/+) and VDR(−/−) mice. (C and D) Western blot (C) and densitometric quantification (D) of α-SMA protein. (E and F) Western blot (E) and densitometric quantification (F) of Snail protein in these kidneys. (G) Immunostaining of kidney sections with anti-α-SMA antibody. *P < 0.05, **P < 0.01, ***P < 0.001 versus sham of the same genotype; #P < 0.05, ##P < 0.01 versus VDR(+/+)-UUO.
Activation of the RAS in UUO
To understand the mechanism underlying the enhanced renal fibrosis in VDR(−/−) mice, we focused on the RAS, which was previously shown to be regulated by 1,25(OH)2D3.12 Immunostaining showed marked accumulation of AngI/II in the interstitium of the sham VDR(−/−) kidney (Figure 5A), which thus predisposes the VDR(−/−) kidney to fibrosis under ureteral ligation. AngI/II remained high in the fibrotic region of VDR(−/−) mice 7 days after UUO (Figure 5A). Semiquantitative analysis of the immunostained sections confirmed that the sham VDR(−/−) kidneys showed more intense AngI/II staining than the VDR(+/+) counterpart, and the obstructed VDR(−/−) kidney had the highest score (Figure 5B). Consistently, Northern blot showed that the baseline renin level was elevated in the sham VDR(−/−) kidney, as expected, and renin expression was induced in the obstructed kidney from both VDR(+/+) and VDR(−/−) mice, with VDR(−/−) mice showing much more robust induction (by >35-fold on the basis of the quantitative data; Figure 5, C and D). Further examinations by real-time RT-PCR revealed that renin, (pro)renin receptor, angiotensinogen (AGT), and AT1R all were induced in the obstructed kidney in both VDR(+/+) and VDR(−/−) mice, with VDR(−/−) mice showing more robust induction in renin, (pro)renin receptor, AGT, and AT1R (Figure 5D). In fact, as reported previously,12,22 the baseline levels of renin and AGT were already elevated in VDR(−/−) mice (Figure 5D). These data support the notion that the local renal RAS is activated in UUO, and in the absence of VDR, the RAS is even more activated.
Figure 5.
More robust activation of the RAS in VDR-null mice under UUO. (A) Immunostaining of sham-operated and obstructed kidney sections with AngI/II-specific antibody. (B) Semiquantitative score of the AngI/II immunostaining of the kidney sections. (C) Northern blot analysis of renin expression in these kidneys. (D) Real-time RT-PCR analysis of the components of the RAS in the sham-operated and obstructed kidneys from VDR(+/+) and VDR(−/−) mice. Renin R, (pro)renin receptor; ACE, angiotensin-converting enzyme. *P < 0.05, **P < 0.01, ***P < 0.001 versus sham of the same genotype; #P < 0.05, ##P < 0.01, ###P < 0.001 versus corresponding VDR(+/+) (sham or UUO).
Fibrogenic Effect of AngII on Primary Tubular Cells
To confirm the fibrogenic activity of AngII in kidney cells, we treated primary tubular cells isolated from VDR(+/+) and VDR(−/−) mice with AngII, using TGF-β as a positive control. As shown in Figure 6, like TGF-β, AngII markedly suppressed E-cadherin expression while stimulating α-SMA and Snail in these cells (Figure 6, A and B). Interestingly, the effect of AngII on VDR(−/−) cells seemed to be more robust than on VDR(+/+) cells (Figure 6C), suggesting that VDR(−/−) tubular cells might be more susceptible to AngII EMT stimulation. Given that these cells were cultured in the absence of 1,25(OH)2D3, it is plausible that VDR might negatively regulate EMT in a ligand-independent manner.
Figure 6.
TGF-β and Ang II induce EMT in primary tubular cells derived from VDR(+/+) and VDR(−/−) mice. (A and B) Primary tubular cells isolated from VDR(+/+) and VDR(−/−) mice are exposed to 10 ng/ml TGF-β (A) or 100 nM AngII (B) for 24 hours, and protein levels of E-cadherin, α-SMA, and Snail are determined by Western blotting. (C) Densitometric quantification of E-cadherin, α-SMA, and Snail proteins. *P < 0.05 versus control of the same genotype; #P < 0.05 versus VDR(+/+) treated.
Blockade of the RAS Blunts the Enhanced Renal Fibrosis in VDR(−/−) Mice
The data prompted us to hypothesize that activation of the local RAS and hence AngII overproduction in the kidney are responsible for the more robust renal fibrosis seen in the VDR(−/−) kidney. To test this hypothesis, we treated VDR(+/+) and VDR(−/−) mice under UUO with the AT1R blocker losartan for 7 days. Histologic examination with PAS (Figure 7A) and Masson's trichrome (Figure 7B) staining showed mild tubular dilation but little fibrosis in the obstructed kidneys; importantly, the morphologic abnormalities became indistinguishable between VDR(+/+) and VDR(−/−) mice (Figure 7, A and B). As expected, there was very mild induction of FN and collagen I (Figure 7C), as well as TGF-β and CTGF (Figure 7D), in the obstructed kidneys; however, there was no difference in the magnitude of induction for these proteins between VDR(+/+) and VDR(−/−) kidneys (Figure 7, C and D). The mild induction of FN and collagen I led to no detectable fibrosis in the kidney, because their induction usually precedes the formation of detectable fibrosis. Our renal pathologist's assessment of the fibrosis score was 0 and 0 (on a scale of 0 to 4) for the obstructed VDR(+/+) and VDR(−/−) kidneys, respectively. Compared with the score of 0.5 and 4.0 obtained in untreated obstructed VDR(+/+) and VDR(−/−) kidneys (Figure 1C), these results demonstrated a clear renoprotective effect of losartan treatment in UUO.
Figure 7.
Losartan blocks renal fibrosis in VDR(+/+) and VDR(−/−) mice. Sham-operated or obstructed VDR(+/+) and VDR(−/−) mice are treated with losartan at 30 mg/kg per d for 7 days. (A and B) Kidney sections from the losartan-treated VDR(+/+) and VDR(−/−) mice are stained with PAS (A) or Masson's trichrome (B). (C) RT-PCR quantification of FN and collagen I expression in these kidneys. (D) RT-PCR quantification of MCP-1, TGF-β, and CTGF expression in these kidneys. Shown also are the PCR ethidium bromide staining gels of these cytokines. *P < 0.05, ***P < 0.001 versus sham of the same genotype (n = 3).
In losartan-treated mice, there was marked reduction of E-cadherin and marked induction of α-SMA and Snail in the obstructed kidneys compared with the sham controls; however, the magnitude of changes of these proteins was indistinguishable between VDR(+/+) and VDR(−/−) mice (Figure 8). These data indicate that blockade of the RAS in the kidney blunts the enhanced EMT and renal fibrosis seen in VDR(−/−) mice.
Figure 8.
Losartan treatment eliminates the difference in EMT between VDR(+/+) and VDR(−/−) mice. (A) Western blot analyses of E-cadherin, α-SMA, and Snail in the sham-operated and obstructed kidneys of losartan-treated VDR(+/+) and VDR(−/−) mice. (B) Densitometric quantification of these proteins. *P < 0.05, **P < 0.01, ***P < 0.001 versus sham of the same genotype.
Discussion
Renal fibrosis is a common downstream event leading to renal failure; thus, understanding the development of renal fibrosis has important implications for therapeutic intervention of kidney disease. Previous studies demonstrated that paricalcitol, an activated vitamin D analog, was able to attenuate interstitial fibrosis, EMT, and renal inflammation in the UUO model.18,23 In those studies, paricalcitol was shown to suppress TGF-β expression as well as block directly TGF-β–induced EMT and ECM proteins in tubular cell cultures and to inhibit RANTES expression via antagonizing NF-κB activity. In this study, we assessed the effect of VDR deficiency on the development of renal fibrosis using the UUO model. Our data suggest that VDR attenuates renal fibrosis by blocking the RAS. Together, these studies suggest that vitamin D–VDR signaling suppresses renal fibrosis through multiple mechanisms.
In this study, we demonstrated that VDR(−/−) mice developed severe tubular atrophy and tubulointerstitial fibrosis with robust EMT and ECM deposition after 7 days of UUO; in contrast, VDR(+/+) mice showed only moderate, histologically identifiable renal injury and minimal fibrosis. At the molecular level, the expression of ECM proteins (FN and collagens), fibrogenic and inflammatory factors (TGF-β, CTGF, and MCP-1), and EMT markers (E-cadherin, α-SMA, and Snail) was increased more in VDR(−/−) mice than in VDR(+/+) mice in the obstructed kidney. EMT is a major pathway that leads to fibrosis in the kidney. The induction of α-SMA, the marker of myofibroblasts, and the reduction of E-cadherin, the marker of tubular epithelial cells, were much more dramatic in VDR(−/−) mice compared with VDR(+/+) mice. Most striking is the observation that E-cadherin was almost undetectable in the VDR(−/−) kidney after 7 days of UUO, indicating an extensive loss of tubular epithelial cells, which is consistent with the massive tubular atrophy and fibrosis seen in the obstructed VDR(−/−) kidney. Also consistent is Snail, a transcription factor that is involved in EMT and negatively regulates E-cadherin.21 Snail was induced more in VDR(−/−) mice than in VDR(+/+) mice after UUO. Together, these data suggest that VDR(−/−) mice developed more severe renal injury mostly because of more robust EMT.
The main mechanism underlying the severe renal fibrosis in VDR(−/−) mice appears to be the action of the local RAS in the kidney. VDR mediates the action of 1,25(OH)2D3 to suppress renin and AGT,11,22 and VDR inactivation leads to activation of the RAS and overproduction of AngII.12 AngII is a potent inducer of EMT and renal fibrosis4 and stimulates TGF-β production and renal inflammation in UUO.24,25 We confirmed with in vitro tubular cell cultures that AngII has potent fibrogenic activity by directly inducing EMT in VDR(+/+) and VDR(−/−) cells. Thus, the high baseline intrarenal AngII level likely predisposes the VDR(−/−) kidney to fibrosis. Indeed, we showed that treatment with losartan markedly reduced interstitial fibrosis in both VDR(+/+) and VDR(−/−) mice. The fibrosis scores as well as the magnitude of induction for ECM proteins (FN and collagen I) and profibrotic factors (TGF-β and CTGF) were dramatically reduced in VDR(+/+) and VDR(−/−) mice after losartan treatment (compare the data in Figure 7 with Figures 1, 2, and 3). Interestingly, whereas losartan completely blocked fibrosis in UUO, it did not seem to block completely the EMT response (see Figure 8). This disconnect between EMT and fibrosis suggests that AngII might act downstream of EMT and upstream of fibrosis in this model. Obviously, more studies are required to clarify this issue.
Importantly, we demonstrated that blockade of AngII activity with losartan not only reduced the development of renal fibrosis but also eliminated the difference in the severity of the renal phenotypes between VDR(−/−) and VDR(+/+) mice in UUO. The difference in the induction of ECM proteins, profibrotic cytokines, and EMT markers between VDR(+/+) and VDR(−/−) mice also disappeared after losartan treatment, suggesting that these genes are directly or indirectly affected by the high AngII level in VDR(−/−) mice. Therefore, VDR seems to attenuate renal fibrosis in large part by suppressing the RAS.
VDR is a pleiotropic ligand-activated transcription factor. In addition to the RAS, VDR may directly or indirectly regulate other targets involved in renal fibrosis. In VDR or vitamin D deficiency, these targets might lead to a network of cascades to accelerate renal fibrosis. For example, MCP-1 is known to be directly downregulated by 1,25(OH)2D3 in kidney cells.19 Recently, the Wnt/β-catenin pathway was shown to play a key role in renal fibrosis.26 We found that β-catenin was induced in the obstructed kidney but the induction was not different between VDR(+/+) and VDR(−/−) mice (data not shown). This is not unexpected, because VDR inhibits β-catenin signaling by physically interacting with β-catenin, not by transcriptional regulation.27 In the absence of VDR, β-catenin may be more activated in the kidney. VDR may also directly suppress Snail18 and stimulate E-cadherin expression27 in cell cultures as a way to prevent EMT. Together, the data obtained in this study further enforce the notion established in recent years that VDR plays a crucial renoprotective role in pathophysiologic conditions.
Concise Methods
Animal Studies
Three-month-old C57BL/6J wild-type [VDR(+/+)] and VDR-null [VDR(−/−)] mice were used in the study. These mice were subjected to UUO operation. Briefly, UUO was performed under ketamine/xylazine anesthesia in which a midline incision was made and the left ureter was exposed and tied off at two points. Sham operation was performed similarly but without ureter ligation. All mice were killed on day 7 after the surgical operation by cardiac exsanguination. Kidneys were collected for analyses. For losartan treatment, mice were fed water containing 0.1 mg/ml (30 mg/kg per d) losartan starting 3 days before UUO surgery until day 7 after surgery. The animal study protocols were approved by the Institutional Animal Care and Use Committee at the University of Chicago.
Histology and Immunohistochemistry
Freshly dissected kidneys were fixed overnight with 4% formaldehyde in PBS (pH 7.2), processed, and embedded in paraffin. Kidney sections were cut at 3 mm, and the sections were stained with PAS or Masson's trichrome by standard procedure. The kidneys were scored by a renal pathologist (A.C.) for interstitial fibrosis and tubular atrophy on a scale from 0 to 4.28 For immunostaining, paraffin sections were first boiled in 10 mM Na citrate solution (pH 6.0) for 10 minutes to retrieve the antigens before antibody staining. The sections were stained with primary antibodies (against α-SMA [Chemicon], FN [Sigma] or AngI/II [Santa Cruz Biotechnology]), followed by incubation with horseradish peroxidase–conjugated secondary antibodies. Antigens were visualized with a peroxidase substrate diaminobenzidine kit (Vector Laboratories, Burlingame, CA) under a microscope. The intensity of AngI/II immunostaining was evaluated using a semiquantitative scale of 0 to 3 (none, mild, moderate, and severe) by a single observer (A.C.) without previous knowledge of the treatment groups. Staining with secondary antibodies only served as negative controls, and no stained signals were detected in the negative control slides.
Primary Tubular Cell Cultures
Primary tubular cells were isolated from VDR(+/+) and VDR(−/−) mice according to our previously described methods.29 The tubular cells were cultured to confluence when they were treated with TGF-β (4 ng/ml) or AngII (100 nM) for 2 days. Then cell lysates were prepared for Western blot analyses.
Western Blot
Whole kidneys or tubular cells were homogenized or lysed in Laemmli buffer (Boston Bioproducts, Worcester, MA), followed by 5 minutes of boiling and centrifugation to obtain the lysates. Protein concentrations were determined using a BioRad Protein Assay kit (BioRad, Hercules, CA). Proteins were separated by SDS-PAGE and transferred onto Immobilon membranes. Western blotting was carried out as described previously,30 using antibodies against α-SMA, E-cadherin (Santa Cruz Biotechnology), or Snail (Santa Cruz Biotechnology).
RT-PCR and Northern Blot
Total cellular RNAs were isolated using TRIzol reagents (Invitrogen, Carlsbad, CA). First-strand cDNAs were synthesized from 2 μg of total RNAs in a 20-μl reaction using MML-V reverse transcriptase (Invitrogen) and hexanucleotide random primers. The first-strand cDNAs served as the template for the PCR performed using a BioRad DNA Engine (BioRad). Real time RT-PCR was performed in Applied Biosystems 7900 Real Time PCR System using a SYBR green PCR reagent kit (Applied Biosystems, Foster City, CA) as described previously.13 Glyceraldehyde-3-phosphate dehydrogenase or β2 microglobulin served as the internal control. The PCR primers used in this study were as described previously.13,17 Northern blot analysis of renin expression was performed as described.12
Statistical Analysis
Data were presented as means ± SEM. Statistical comparisons were made using t test, with P < 0.05 being considered statistically significant.
Disclosures
None.
Acknowledgments
This work was supported in part by National Institutes of Health grant HL085793.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
REFERENCES
- 1. Iwano M, Neilson EG: Mechanisms of tubulointerstitial fibrosis. Curr Opin Nephrol Hypertens 13: 279–284, 2004. [DOI] [PubMed] [Google Scholar]
- 2. Kalluri R, Neilson EG: Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest 112: 1776–1784, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Chevalier RL: Obstructive nephropathy: Towards biomarker discovery and gene therapy. Nat Clin Pract Nephrol 2: 157–168, 2006. [DOI] [PubMed] [Google Scholar]
- 4. Wolf G: Renal injury due to renin-angiotensin-aldosterone system activation of the transforming growth factor-beta pathway. Kidney Int 70: 1914–1919, 2006. [DOI] [PubMed] [Google Scholar]
- 5. Wolf G, Mueller E, Stahl RA, Ziyadeh FN: Angiotensin II-induced hypertrophy of cultured murine proximal tubular cells is mediated by endogenous transforming growth factor-beta. J Clin Invest 92: 1366–1372, 1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Border WA, Noble NA: Cytokines in kidney disease: The role of transforming growth factor-beta. Am J Kidney Dis 22: 105–113, 1993. [DOI] [PubMed] [Google Scholar]
- 7. Burns WC, Twigg SM, Forbes JM, Pete J, Tikellis C, Thallas-Bonke V, Thomas MC, Cooper ME, Kantharidis P: Connective tissue growth factor plays an important role in advanced glycation end product-induced tubular epithelial-to-mesenchymal transition: Implications for diabetic renal disease. J Am Soc Nephrol 17: 2484–2494, 2006. [DOI] [PubMed] [Google Scholar]
- 8. Gupta S, Clarkson MR, Duggan J, Brady HR: Connective tissue growth factor: Potential role in glomerulosclerosis and tubulointerstitial fibrosis. Kidney Int 58: 1389–1399, 2000. [DOI] [PubMed] [Google Scholar]
- 9. Haussler MR, Whitfield GK, Haussler CA, Hsieh JC, Thompson PD, Selznick SH, Dominguez CE, Jurutka PW: The nuclear vitamin D receptor: Biological and molecular regulatory properties revealed. J Bone Miner Res 13: 325–349, 1998. [DOI] [PubMed] [Google Scholar]
- 10. Li YC: Renoprotective effects of vitamin D analogs. Kidney Int May 27, 2009. [epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 11. Yuan W, Pan W, Kong J, Zheng W, Szeto FL, Wong KE, Cohen R, Klopot A, Zhang Z, Li YC: 1,25-Dihydroxyvitamin D3 suppresses renin gene transcription by blocking the activity of the cyclic AMP response element in the renin gene promoter. J Biol Chem 282: 29821–29830, 2007. [DOI] [PubMed] [Google Scholar]
- 12. Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP: 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest 110: 229–238, 2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zhang Z, Sun L, Wang Y, Ning G, Minto AW, Kong J, Quigg RJ, Li YC: Renoprotective role of the vitamin D receptor in diabetic nephropathy. Kidney Int 73: 163–171, 2008. [DOI] [PubMed] [Google Scholar]
- 14. Freundlich M, Quiroz Y, Zhang Z, Zhang Y, Bravo Y, Weisinger JR, Li YC, Rodriguez-Iturbe B: Suppression of renin-angiotensin gene expression in the kidney by paricalcitol. Kidney Int 74: 1394–1402, 2008. [DOI] [PubMed] [Google Scholar]
- 15. Makibayashi K, Tatematsu M, Hirata M, Fukushima N, Kusano K, Ohashi S, Abe H, Kuze K, Fukatsu A, Kita T, Doi T: A vitamin D analog ameliorates glomerular injury on rat glomerulonephritis. Am J Pathol 158: 1733–1741, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Mizobuchi M, Morrissey J, Finch JL, Martin DR, Liapis H, Akizawa T, Slatopolsky E: Combination therapy with an angiotensin-converting enzyme inhibitor and a vitamin D analog suppresses the progression of renal insufficiency in uremic rats. J Am Soc Nephrol 18: 1796–1806, 2007. [DOI] [PubMed] [Google Scholar]
- 17. Zhang Z, Zhang Y, Ning G, Deb DK, Kong J, Li YC: Combination therapy with AT1 blocker and vitamin D analog markedly ameliorates diabetic nephropathy: Blockade of compensatory renin increase. Proc Natl Acad Sci U S A 105: 15896–15901, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Tan X, Li Y, Liu Y: Paricalcitol attenuates renal interstitial fibrosis in obstructive nephropathy. J Am Soc Nephrol 17: 3382–3393, 2006. [DOI] [PubMed] [Google Scholar]
- 19. Zhang Z, Yuan W, Sun L, Szeto FL, Wong KE, Li X, Kong J, Li YC: 1,25-Dihydroxyvitamin D(3) targeting of NF-kappaB suppresses high glucose-induced MCP-1 expression in mesangial cells. Kidney Int 72: 193–201, 2007. [DOI] [PubMed] [Google Scholar]
- 20. Liu Y: Epithelial to mesenchymal transition in renal fibrogenesis: Pathologic significance, molecular mechanism, and therapeutic intervention. J Am Soc Nephrol 15: 1–12, 2004. [DOI] [PubMed] [Google Scholar]
- 21. Cano A, Perez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG, Portillo F, Nieto MA: The transcription factor snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol 2: 76–83, 2000. [DOI] [PubMed] [Google Scholar]
- 22. Deb DK, Chen Y, Zhang Z, Zhang Y, Szeto FL, Wong KE, Kong J, Li YC: 1,25-Dihydroxyvitamin D3 suppresses high glucose-induced angiotensinogen expression in kidney cells by blocking the NF-kappaB pathway. Am J Physiol Renal Physiol 296: F1212–F1218, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Tan X, Wen X, Liu Y: Paricalcitol inhibits renal inflammation by promoting vitamin D receptor-mediated sequestration of NF-kappaB signaling. J Am Soc Nephrol 19: 1741–1752, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Esteban V, Lorenzo O, Ruperez M, Suzuki Y, Mezzano S, Blanco J, Kretzler M, Sugaya T, Egido J, Ruiz-Ortega M: Angiotensin II, via AT1 and AT2 receptors and NF-kappaB pathway, regulates the inflammatory response in unilateral ureteral obstruction. J Am Soc Nephrol 15: 1514–1529, 2004. [DOI] [PubMed] [Google Scholar]
- 25. Pimentel JL, Jr, Sundell CL, Wang S, Kopp JB, Montero A, Martinez-Maldonado M: Role of angiotensin II in the expression and regulation of transforming growth factor-beta in obstructive nephropathy. Kidney Int 48: 1233–1246, 1995. [DOI] [PubMed] [Google Scholar]
- 26. He W, Dai C, Li Y, Zeng G, Monga SP, Liu Y: Wnt/beta-catenin signaling promotes renal interstitial fibrosis. J Am Soc Nephrol 20: 765–776, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Palmer HG, Gonzalez-Sancho JM, Espada J, Berciano MT, Puig I, Baulida J, Quintanilla M, Cano A, de Herreros AG, Lafarga M, Munoz A: Vitamin D(3) promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and the inhibition of beta-catenin signaling. J Cell Biol 154: 369–387, 2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Bao L, Wang Y, Chang A, Minto AW, Zhou J, Kang H, Haas M, Quigg RJ: Unrestricted C3 activation occurs in Crry-deficient kidneys and rapidly leads to chronic renal failure. J Am Soc Nephrol 18: 811–822, 2007. [DOI] [PubMed] [Google Scholar]
- 29. Cao LP, Bolt MJ, Wei M, Sitrin MD, Li YC: Regulation of calbindin-d9k expression by 1,25-dihydroxyvitamin d(3) and parathyroid hormone in mouse primary renal tubular cells. Arch Biochem Biophys 400: 118–124, 2002. [DOI] [PubMed] [Google Scholar]
- 30. Li YC, Bolt MJ, Cao L-P, Sitrin MD: Effects of vitamin D receptor inactivation on the expression of calbindins and calcium metabolism. Am J Physiol Endocrinol Metab 281: E558–E564, 2001. [DOI] [PubMed] [Google Scholar]