Abstract
Objective
Development of an in vivo imaging method to assess lymphatic draining function in the K/B×N mouse model of inflammatory arthritis.
Methods
Indocyanine green (ICG), a near-infrared (NIR) fluorescent dye, was injected intradermally into the footpad of wild-type mice, the limb was illuminated with an 806 nm NIR laser, and the movement of ICG from the injection site to the draining popliteal lymph node (PLN) was recorded with a CCD camera. ICG-NIR images were analyzed to obtain 5 measures of lymphatic function across time. K/B×N arthritic mice and control non-arthritic littermates were imaged at one-month of age when acute joint inflammation commenced, and repeated at 3 months when joint inflammation became chronic. Lymphangiogenesis in PLNs was assessed by immunochemistry.
Results
ICG and its transport within lymphatic vessels were readily visualized and quantitative measures derived. During the acute phase of arthritis, the lymphatic vessels were dilated with increased ICG signal intensity and lymphatic pulses, and PLNs became fluorescent quickly. During the chronic phase, new lymphatic vessels were present near the foot. However, ICG appearance in lymphatic vessels was delayed. The size and area of PLN lymphatic sinuses progressively increased in the K/B×N mice.
Conclusion
ICG-NIR lymphatic imaging is a valuable method to assess the lymphatic draining function in mice with inflammatory arthritis. ICG-NIR imaging of K/B×N mice identified two distinct lymphatic phenotypes during the acute and chronic phase of inflammation. This technique can be used to assess new therapies for lymphatic disorders.
Keywords: Near infrared, lymphatic drainage, lymphangiogenesis, inflammation, lymph nodes, in vivo imagining
INTRODUCTION
Inflammatory erosive arthropathies, such as rheumatoid arthritis (RA), are characterized by pannus formation and joint tissue damage. While it is known that inflammation in RA induces angiogenesis and tissue edema due to the accumulation of fluid leaking from newly formed blood vessels into interstitial space (1), and this interstitial fluid contains high levels of cytokines and chemokines that may contribute to joint tissue destruction (2), little is known about the compensatory lymphatic mechanisms responsible for the removal of accumulated fluid from joint interstitial space that attenuate local edema, inflammation, and bone and cartilage erosion. As this is largely due to a lack of physiological outcome measures to assess lymphatics and lymph flow during arthritis, novel approaches to quantify this central biomarker of disease pathogenesis are warranted.
The lymphatic system drains interstitial fluid to local draining lymph nodes through lymphatic vessels (3). The lymph generated in RA joints, as well as lymphatic flow, are increased during disease progression and contain significantly elevated levels of pro-inflammatory cytokines and chemokines (4). ELISA of synovial fluid from RA patients demonstrated elevated levels of vascular endothelial growth factor C (VEGF-C), the primary lymphatic growth factor, which correlated significantly with tumor necrosis factor (TNF) and interleukin-1 (IL-1) levels of in RA (5). Immunostaining of synovial tissues from patients with RA and osteoarthritis using antibodies that recognize lymphatic endothelial cells or lymphatic growth factors demonstrated increased lymphatic vessel formation (6–8). We recently reported that the blockade of lymphangiogenesis at the beginning of arthritis development increased the severity of joint tissue injury (9). Thus the expression levels of lymphatic growth factors, lymphatic vessel formation and lymph flow are all increased in RA joints, suggesting that increased lymphangiogenesis is a compensatory mechanism to ameliorate inflammatory arthritis. However, the role of the lymphatic system in the pathogenesis of inflammatory arthritis has not been elucidated readily because inadequate imaging modalities to assess lymphatic function in murine models.
Several imaging methods have been used in rodents to measure lymph flow, including whole body X-ray, MRI, optical imaging, and intravital microscopy (10–15). Although these technologies can visualize the path of a contrast agent within lymphatic vessels, they are expensive, require special instruments and highly trained personnel thus limiting routine use in laboratory animals. Indocyanine green (ICG) is a dye safe for intravenous bolus used for multiple purposes (16). Once injected it stays in blood and is excreted in bile without being reabsorbed from the gut. Although green, ICG has an absorption and emission spectrum within the near infrared (NIR) range, enabling deep tissue penetration and fluorescence visualization. Sharma et al. utilized ICG-NIR fluorescence to examine lymphatics in swine (17). They completed a phase I clinical trail using small doses of ICG to assess lymph traffic from tumor to sentinel lymph nodes in breast cancer (18). However, this method has not been used in laboratory mouse models of arthritis.
In the current study, we used ICG-NIR lymphatic imaging to examine progressive changes in lymphatic function in the legs of K/B×N mice (19), a murine model of inflammatory arthritis. During the acute phase of arthritis, lymphatic flow increases, resulting in dilated lymphatic vessels and severe ankle joint swelling. During the chronic phase, new lymphatic vessels develop in the foot with decreased lymph flow. These ICG-NIR findings demonstrated impaired lymphatic function in inflamed joints, suggesting that the improvement of local lymph flow may represent a new target for RA therapy in patients with severe disease. Additionally, we find that ICG-NIR imaging is an effective method for assessing lymphatic function in vivo.
MATERIALS AND METHODS
Animals
The KRN/NOD transgenic mice used in this work were obtained by crossing KRN transgenic males (B6 genetic background; provided by Dr. C. Benoist, Harvest University) with femaleNon-obese diabetic (NOD) mice (NOD/LtJ, Jackson Laboratory). (KRN/NOD)F1 offspring mice were bledon day 21 after birth, and those expressing the αVβ6 TCR transgene were identified by flow cytometry. These mice were named as K/B×N mice. Controls were F1 offspringmice not expressing the transgene. All K/B×N mice develop severe ankle joint inflammation around 1-month of age. For flow cytometry, whole blood was incubatedon ice with PE-monoclonal anti-mouse CD4 T cell (0.5 mg) and FITC-monoclonal anti-mouse αVβ6-TCRβ-chain (0.5 mg) antibodies (BD PharMingen). The animals that carry CD4+αVβ6+ cells were identified as KRN transgene positive.
For ICG-NIR imaging, animals were anesthetized with 2% and maintained with 1% isoflurane (Bulter animal health supply. United Kingdom) in oxygen. Animals received ICG-NIR imaging at 1 and 3 months of age. The Institutional Animal Care and Use Committee approved all animal studies. For measurement of the severity of edema, the ankle thickness (mm) was measured at one month and 3 month of age using a caliper.
ICG-NIR lymphatic imaging
A Spy1000 system (Novadaq Technologies Inc Mississauga, Ontario, Canada) was used. This system consists of an 806nm laser providing maximum illumination intensity approximately 30 mW/cm2 at a fixed focal length. This instrument was modified to provide continuous operation and adjustment of laser intensity, and was equipped with a second PAL camera. Filters permitted the measurement of light above 815nm. The video outputs of the camera were attached to the network using an Axis 241SA video server (Axis, Sweden, Lund) that was monitored using SecuritySpy (Ben Bird, www.bensoftware.com) that converted motion JPEG image streams to QuickTime movies (Apple Computer Inc). Individual JPEG image sequences were then exported and read for further analysis with ImageJ.
ICG (Acorn or Pulsion) was dissolved in the distilled water at 0.1 μg/μl and 10 μl (1 μg) of ICG solution was intra-dermally injected into the footpad using a 30G needle. Before ICG injection, fur was removed from legs with hair removal lotion. Animals were placed in recumbent position and legs were restrained with tape on an isothermal gel pad during imaging sessions. ICG fluorescence was recorded for 1–2 hours immediately and again 24 hours for 5 min after the ICG injection. The QuickTime movies were examined and the time for ICG appearance in popliteal lymph node (PLNs) after the footpad injection was recorded. Sequential images from the movie file were exported and the intensity of ICG fluorescence of PLNs and footpads were determined using ImageJ. A fixed circular region of interest (ROI) was drawn over the vessels or PLNs and nearby background tissue. The signal intensity was defined as Signal intensity – background intensity. The following outcome measures were derived from the ICG-NIR images: 1) T-initial (T-in), which is the time that it takes for the ICG to be detected in vessels; 2) S-max, which is the maximum ICG signal intensity observed in the PLN during the first day imaging session; 3) T-max, which is the time it takes for a PLN to achieve maximal ICG signal intensity; 4) % Clearance, which is an assessment of ICG wash out through the lymphatics and is quantified as the percent difference of ICG signal intensity between the two ICG-NIR images from the ROI of the PLNs or footpad at a) S-max (first day) and b) 24 hours post ICG injection; and 5) pulse, which is the ICG pulses that pass the ROI within 400 seconds.
Immunostaining and histomorphometric analysis
PLNs were frozen in liquid nitrogen and embedded in OCT matrix. Frozen sections (10 μm) were stained with a mixture of FITC-rabbit anti-Lymphatic Vessel Endothelial Receptor 1 (LYVE-1) antibody followed by Alexa Fluor 488 goat anti-rabbit IgG and PE-anti-CD31 antibody as we described previously (8). Four pictures were taken from each section (×10) from different fields. Lymphatic vessels were quantified by a point-counting method, as we described previously (20). For each mouse, 2 sections were cut at 250 μm apart and the area and size of LYVE-1+ lymphatic vessels were measured for entire PLN and expressed as percentage of LYVE+ vessels per PLN, as we previously described (8). The data were presented as the mean ± SD from 4–6 PLNs.
Statistics
All results are given as mean ± SD. Comparisons between two groups were analyzed using two-tailed unpaired Student’s t-test. One-way ANOVA and Dunnett’s Post Hoc multiple comparisons were used for comparisons among three or more groups. P values < 0.05 were considered statistically significant.
RESULTS
Establishment of an ICG-NIR lymphatic imaging protocol to quantify lymph drainage in the mouse leg
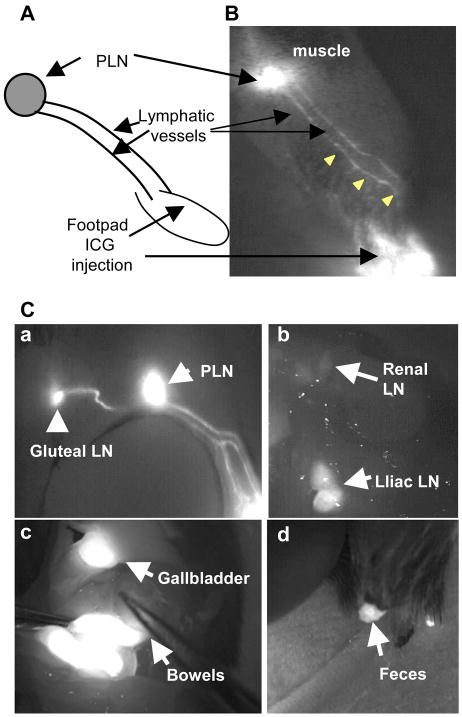
To establish a quantitative ICG-NIR lymphatic imaging protocol for the mouse leg, we first evaluated the clearance behavior of ICG throughout the animal. ICG was intradermally injected into the footpad, and the path of ICG clearance through two parallel lymphatic vessels in the lower limb to the PLN was clearly visible by NIR imaging (Figure 1A&B). Whole body ICG-NIR imaging demonstrated that the fluorescent dye migrates from the PLN to the gluteal and iliac lymph nodes (Figure 1C), from there it is efficiently cleared through the digestive system such that no detectable ICG remains in WT mice 48hrs after injection. These findings are consistent with previous lymphatic tracer studies (21), and confirm that measurement of ICG flow between the foot and the PLN reflects lymphatic function in the lower limbs of mice.
Figure 1. Lymphatic flow in the mouse leg.
Adult C57/B6 wild type mice were used. (A) A photo taken 5 min after intradermal injection of blue ink into the footpad shows the path of ink flow from the injection site to the PLN through 2 lymphatic vessels (green arrows). (B) A schematic diagram and (C) an ICG-NIR image illustrate the movement of ICG along the lymphatic vessels from the footpad to the PLN. (D) ICG-NIR images show the ICG clearance from lymphatics (a) to internal organs (b,c), and excreted in feces (d). In this experiment, a double dose of ICG was intradermally injected into the footpad (a), and massage was applied upon the injection site to enhance lymphatic flow. The abdomen was opened and exposed to the NIR laser for observation of the deep lymph nodes and internal organs (b,c).
Assessment of lymphatic function from ICG-NIR images
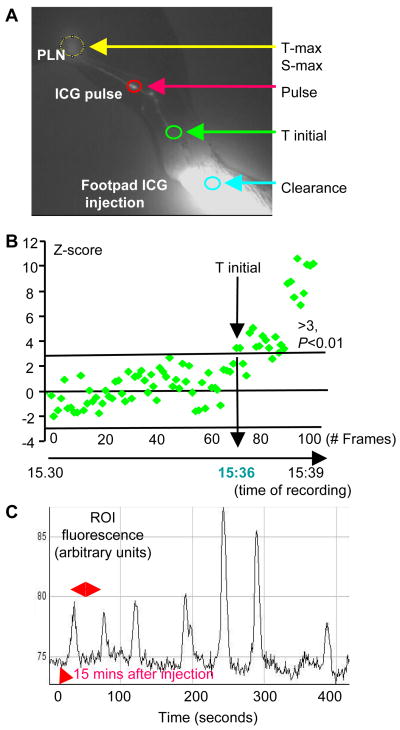
To quantify lymphatic function, a regions of interest (ROI) were retrospectively identified on the ICG-NIR images from the 1–2hr real time video to quantify five independent metrics (Figure 2A): 1) the time it takes for ICG to reach lymphatic vessels was named T initial (T-in); 2) the peak signal intensity achieved in the PLN (S-max); 3) the time it takes to achieve S-max (T-max); 4) the lymphatic pulse (amplitude in signal intensity and time interval between pulses); and 5) the rate of ICG clearance from the injection site in the foot (% ICG signal at 24hr). Figure 2B is a z-score data set from a real time ICG-NIR video analyzed over a ten minutes period during which T-in was achieved. Z-score was calculated as the follows: (current observation - mean)/standard deviation. The z-scores were plotted against the frame number (time) where the z-score that was greater than 3 (p<0.01) was considered as T-in time. Figure 2C illustrates the mean fluorescence intensity profile of the ROI for lymphatic pulse over time, which shows that the lymphatic vessels in the leg of a WT mouse under deep anesthesia beat every 55 seconds. This is in dramatic contrast to the mouse’s vascular pulse, which is driven by a heart rate of ~550–600 beats per minute. These data also draw attention to a common misconception that lymph migration is achieved via adjacent muscle tissues contraction, when in fact ICG migration in our system is entirely dependent on lymphatic vessel contraction that contains its own pulse.
Figure 2. Quantitative assessment of ICG-NIR images to evaluate lymphatic draining function in the mouse leg.
Adult C57/B6 wild type mice were used (n=4), and data from a representative animal are shown. (A) An ICG-NIR image indicates selected regions of interest (ROI) for quantitative analyses. (B) The primary data from a real time NIR imaging video demonstrates the time it takes for ICG to travel from the footpad to the lymphatic vessel (green circle in A) as T initial. (C) The histogram shows a typical ICG pulses from a wild type mouse under anesthesia. For quantification, a ROI for pulse was identified (red circle in A). The number of lymphatic pulses that pass the ROI over 400 seconds was calculated from the NIR real time video to derive the number of lymphatic pulses per min and the pulse interval (double headed arrow).
Distinct lymphatic draining phenotypes during the acute and chronic phase of inflammatory arthritis
As a prelude to our studies examining the effects of altered lymphatic flow on the severity of joint inflammation, we first excluded the possibility that ICG footpad injection itself causes local inflammation. Injection of ICG into the footpad of wild-type mice did not result in histologic changes at the injection sites on H&E-stained sections (data not shown). We then defined the behavior of ankle synovitis in K/B×N mice at the acute and chronic phases of arthritis by comparing paw thickness of same animals at 1 and 3 months of age, and found that foot swelling decrease with age (the ankle thickness: 1 month vs 3 month = 3.3+0.2 vs. 4.5+0.5 mm, p<0.05). Collectively, these findings demonstrate that our ICG-NIR experimental approach is innocuous, and that 1-month-old K/B×N mice display acute ankle synovitis, which is abated during the chronic phase of arthritis as seen in 3-month-old K/B×N mice.
To examine the changes of lymphatic function during the natural history of inflammatory arthritis, we performed ICG-NIR imaging on the legs of K/B×N arthritic mice and control non-arthritic littermates prospectively. K/B×N mice develop severe inflammatory arthritis around 1 month of age. Joint redness and swelling occurs within 2–3 days during the acute phase. Afterwards, inflammation persists into the chronic phase (>3 months old) where the redness and swelling decline but the tissue damage remains (22, 23).
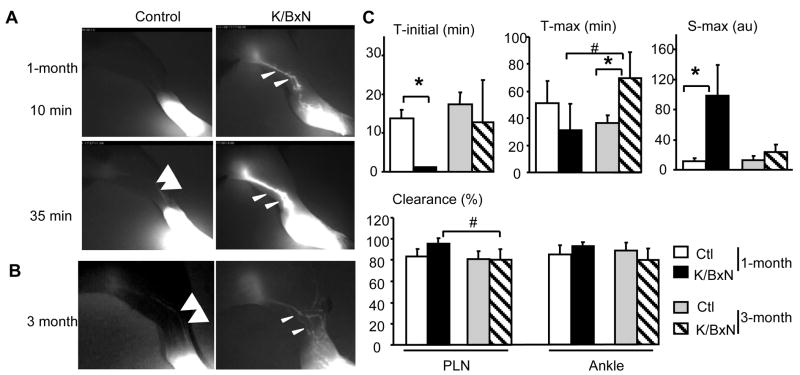
The ICG-NIR images show that control mice had one or two major lymphatic vessels linking the foot area to the PLN. These vessels were thin with weak ICG fluorescence (arrows in Figure 3A). Accordingly, S-max was low and T-max was about 30–50 min. There were no remarkable differences in lymphatic parameters between 1 and 3 month old control mice (Figure 3A). These observations suggest a consistent level of lymph movement from the foot area to the PLN in the normal mouse leg under anesthesia. In contrast, K/B×N arthritic mice had a dramatically different lymphatic phenotype. During the acute phase (1-month-old), extensively dilated and irregular lymphatic vessels (arrow-head in Figure 3A) were observed, and a very bright ICG signal was present in vessels and PLNs. During the chronic phase, the ICG signal intensity and the size of the lymphatic vessels were returned to the control levels. However, multiple newly formed lymphatic vessels near the foot area were visualized (arrow-heads in Figure 3B). These observations were confirmed by quantitative analysis (Figure 3C). During the acute phase of arthritis in K/B×N mice, T-in is significantly decreased 13-fold, and S-max is significantly increased 10-fold. In contrast, during the chronic inflammation phase in 3-month-old K/B×N mice, T-in, S-max and ICG clearance were all similar to that of their non-transgenic littermates. However, the lymphatics in the legs of K/B×N mice with chronic arthritis were still abnormal as evidences by a 2-fold increase in T-max, which was also significantly greater than that seen in 1-month-old K/B×N mice with acute arthritis. Taken together, these data predict that the lymphatic pulse is significantly increased during the acute phase, to achieve accelerated ICG migration. However, during chronic arthritis, this accelerated pulse decreases, and lymphangiogenesis predominates to increase lymphatic vessels numbers and volume.
Figure 3. Lymphatic flow is significantly increased during acute arthritis but not during chronic disease.
K/B×N mice and non-arthritic control littermates were subjected to ICG-NIR imaging at 1 and 3 months of age. (A) At 1-month, the ICG-NIR images that were taken at 10 and 35 min after ICG injection at a low camera threshold intensity show very bright and dilated lymphatic vessels (arrow-heads) in a K/B×N mouse with acute arthritis compared to control mice (arrows). (B) At 3-month, the ICG-NIR images that were taken at the maximum camera threshold intensity after ICG injection show numerous lymphatic vessels (arrow-heads) in the K/B×N leg with chronic arthritis compared to the two major lymphatic vessels in the control leg (arrows). (C) The lymphatic draining function was measured from ICG-NIR images as Ti, T-max, S-max and clearance. Values are the mean + SD of 4–8 mice. *p<0.05 vs control mice; # p<0.05 vs 1-month-old K/B×N mice.
To evaluate the effects of acute and chronic ankle arthritis on the lymphatic pulse in the leg, we performed ICG-NIR analyses as described in Figure 2C. We found that during the acute phase of arthritis, K/B×N mice had a significantly increased pulse rate, reaching to about 5 pulses per minute (Figure 4). In contrast, the lymphatic pulses in the chronic phase returned to the control level ~ 1 pulse per minute. Thus, these data support a model of increased lymphatic flow in the acute inflammation, followed by increased lymphangiogenesis with decreased lymphatic flow during the progression of inflammatory arthritis.
Figure 4. Lymphatic pulses are significantly increased during acute arthritis but not during chronic disease.
(A) Representative histograms of lymphatic pulses in a 1-month-old K/B×N mouse (upper) compared to its control littermate (lower). Histograms from 3-month-old K/B×N mice with chronic arthritis were similar to control (data not shown). ICG-NIR images from which the histogram was derived show the pulsatile nature of ICG movement through the lymphatic vessels (arrows) in real time as illustrated by the change in signal intensity from the dilated phase to the contracted phase of the pulse. (B) Quantitative analyses of lymphatic pulses and the time interval among pulses. Values are the mean + SD of 4–8 mice. * p<0.05 vs control mice.
Draining lymph node lymphangiogenesis is positively associated with the capacity of lymphatic drainage and the severity of arthritis
As our lymphatic flow data clearly predicts that there must be significant changes in the number of lymphatic vessels and overall lymphatic volume of the lower limb during arthritis progression in K/B×N, we examined the lymphatic vasculature in PLNs by immunostaining with anti-LYVE-1 antibody to identify lymphatic sinuses, as we reported previously (9, 24). The K/B×N PLNs had significantly increased volume and weight at 1 month, which increased to 10 time that of control mice by 3 months (Figure 5A). Compared to control PLN, PLNs from 1-month old K/B×N mice had increased numbers of LYVE-1+ lymphatic sinuses with narrow lumens (Figure 5Ba, c, e). PLNs from 3 month-old K/B×N mice had even more LYVE-1+ lymphatic sinuses, the majority of them having extensive and dilated lumens (Figure 5Bb, d, f, arrows). The immunostaining observations were confirmed by histomorphometric analysis (Figure 5C). We also performed IHC for CD31+ blood vessels, and found that angiogenesis was also increased in K/B×N PLNs compared to control LNs, but to a lesser degree than lymphangiogenesis (Figure 5B and data not show).
Figure 5. Lymphangiogenesis in PLNs of K/B×N mice is continually increase from the acute through the chronic phases of arthritis.
(A) Weight of PLNs from 1 and 3 month-old control and K/B×N mice (n=4–8). (B) Representative micrographs of PLN sections from 1-month-old (a–c) and 3-month-old (d–f) WT (a,d) and K/B×N (b,c,e,f) mice. Sections were stained with rabbit anti-LYVE-1 antibody followed by Alexa Fluor 488 goat anti-rabbit IgG to identify lymphatic vessels (green), and PE-anti-CD31 antibody for blood vessels (red). Pictures were taken at ×5 (a,b,d,e) and ×10 (c,f), showing markedly dilated lymphatic sinuses in 3-month-old K/B×N mice (yellow arrows). (C) Histomorphormetric analysis of LYVE-1-stained PLN sections. The percentage of LYVE-1+ lymphatic vessels per PLN (left) and the area within the lymphatic vessels are shown. Values are the mean of + SD of 4 PLNs. *p<0.05 vs control mice; # p<0.05 vs 1-month-old K/B×N mice.
DISCUSSION
Although there has been significant advances in our understanding of the etiology and pathogenesis of RA, several enigmatic aspects of this disease remain to be elucidated. One of them is the manner in which the disease waxes and wanes over long periods of time, and the sudden onset of exacerbated joint inflammation and pain commonly referred to as arthritic flare. As repeated efforts to identify changes in autoimmunity continually fail to identify significant findings, it appears more likely that these varying disease states are caused by environmental and epigenetic influences on a chronic condition. One mechanism by which these influences could dramatically affect RA is by altering the efferent lymphatic flow from the joint, which functions to perpetually removes immune cells and catabolic factors that would otherwise accumulate causing swelling, connective tissue destruction and pain. Unfortunately, the absence of a minimally invasive outcome measure to quantify lymphatic flow has prevented this field from moving forward such that the direct role of lymphatic drainage on RA pathogenesis remains largely unknown. Thus, in order to overcome this obstacle and better understand the relationship between lymphatic flow and inflammatory arthritis progression, we established an ICG-NIR lymphatic imaging protocol in mouse legs to visualize and quantify changes during the natural history of inflammatory arthritis in K/B×N mice.
In our longitudinal studies, we observed two distinct lymphatic phenotypes during the acute vs. chronic phases of arthritis in K/B×N mice. At the onset of severe ankle inflammation, lymphatic flow is dramatically increased as determined by a 5-fold increase in the lymphatic pulse and several quantitative measures of ICG migration in lymphatic vessels. We also observed that the lymphatic vessels appeared to be extremely dilated and very bright, indicating that a large amount of lymph was entering afferent lymphatic vessels within a short time as a consequence of acute inflammation where fluid is rapidly accumulating in the interstitial space due to blood vascular leakage (25). This is consistent with the clinical observation of increased lymph flow in lymphatic vessels draining inflamed joints in RA patients (4). However, this increase in lymphatic flow is not sustained, and the joint disease progresses to a chronic phase, which is characterized by numerous new lymphatic vessels that drain the inflamed tissue with a decreased lymphatic pulse and slower lymphatic flow. This de novo lymphangiogenesis is supported by studies demonstrating increased lymphatic vasculature in the synovium of RA patients and animals by immunohistochemistry (6–8). In other models of inflammation such as bacteria-induced lung inflammation, newly formed lymphatic vessels are also observed (26, 27). These observations raises the question as to why there is slower lymphatic flow during the chronic phase, when the large number of newly formed lymphatic vessels should be able to increase lymphatic flow. The simplest explanations are that either the vessels are immature and leaky or that they are nonfunctional. Interestingly, it has been reported that transgenic over-expression or administration of VEGF-A or VEGF-C induces lymphatic vessel formation, but these newly formed vessels leak (28, 29), However, we did not observe ICG fluorescence around the lymphatic vessels, suggesting that the slower lymph flow is unlikely due to vessel leakage. As such, our attention has turned towards understanding the mechanisms responsible for the lymphatic pulse, with the goal of identifying factors that could be pharmacologically manipulated to increase lymphatic flow, and decrease joint inflammation.
Our studies suggest that there is a direct relationship between dysregulated lymph flow and inflammation. This theory is now supported by 2 lines of experimental evidence in mouse models of inflammatory arthritis. First, loss-of-function studies have shown that inhibition of lymphangiogenesis with anti-VEGFR3 neutralizing antibodies decreases lymphatic flow from the foot and increases the severity of ankle joint inflammation (9). Secondly, we have recently completed a series of gain-of-function studies in which lymphangiogenesis was induced via intra-articular injection of recombinant adeno-associated virus that over expresses VEGF-C, which significantly increased lymph flow, and reduces inflammation in arthritic joints (unpublished data). These data support the hypothesis of lymphatic involvement in RA. The lymphatic machinery is composed of lymphatic endothelium, muscle (30) and valve (31). Currently, it is not known if chronic joint inflammation in RA patients is associated with a defect in any of these components. Similarly, very little is known about the cellular and molecular mechanisms controlling the function of lymphatic endothelium, muscle and valve under normal and arthritic condition. By using ICG-NIR imaging of lymphatic flow in animal models and clinical studies, it may be possible to elucidate the mechanisms responsible for arthritic flares and develop therapeutic interventions to prevent and treat RA progression.
Acknowledgments
The authors would like to thank Drs. C. Benoist and D. Mathis (Harvard University, USA) for providingmale KRN transgenic mice, Dr. B. Boyce for helpful discussion, and Ms. Yanyun Li for technical assistance with the histology. This work was supported by research grants from the National Institutes of Health PHS awards (AR48697, AR53586 and UL1 RR24160 to LX, AR54041 and AR56702 to ES, DK075036 to RW). Part of Dr. Zhou, Quan’s salary was supported by a grant from the National Science Fund for Distinguished Young Scholars of China (30625043 to YJW) and National Basic Research Program of China (973 Program, 2010CB530400 to YJW). Dr. D. Golijanin provided advice, encouragement and access to the instrument obtained under a research agreement between the University of Rochester and Novadaq Technologies Inc. Drs. Wood and Golijanin have licensed intellectual property to Novadaq Technologies.
Footnotes
AUTHOR CONTRIBUTIONS
Dr. Lianping Xing has full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study design. Drs. Zhou, Wood, Schwarz, and Xing.
Acquisition of data. Drs. Zhou, Wood, and Xing.
Analysis and interpretation of data. Drs. Zhou, Wood, Wang, Schwarz, Xing.
Manuscript preparation. Drs. Zhou, Wood, Wang, Schwarz, and Xing.
Statistical analysis. Dr. Zhou.
References
- 1.Koch AE. Review: angiogenesis: implications for rheumatoid arthritis. Arthritis Rheum. 1998;41:951–962. doi: 10.1002/1529-0131(199806)41:6<951::AID-ART2>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 2.Firestein GS. Evolving concepts of rheumatoid arthritis. Nature. 2003;423:356–361. doi: 10.1038/nature01661. [DOI] [PubMed] [Google Scholar]
- 3.Gerli R, Solito R, Weber E, Agliano M. Specific adhesion molecules bind anchoring filaments and endothelial cells in human skin initial lymphatics. Lymphology. 2000;33:148–157. [PubMed] [Google Scholar]
- 4.Olszewski WL, Pazdur J, Kubasiewicz E, Zaleska M, Cooke CJ, Miller NE. Lymph draining from foot joints in rheumatoid arthritis provides insight into local cytokine and chemokine production and transport to lymph nodes. Arthritis Rheum. 2001;44:541–549. doi: 10.1002/1529-0131(200103)44:3<541::AID-ANR102>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 5.Cha HS, Bae EK, Koh JH, Chai JY, Jeon CH, Ahn KS, Kim J, Koh EM. Tumor necrosis factor-alpha induces vascular endothelial growth factor-C expression in rheumatoid synoviocytes. J Rheumatol. 2007;34:16–19. [PubMed] [Google Scholar]
- 6.Xu H, Edwards J, Banerji S, Prevo R, Jackson DG, Athanasou NA. Distribution of lymphatic vessels in normal and arthritic human synovial tissues. Ann Rheum Dis. 2003;62:1227–1229. doi: 10.1136/ard.2003.005876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Paavonen K, Mandelin J, Partanen T, Jussila L, Li TF, Ristimaki A, Alitalo K, Konttinen YT. Vascular endothelial growth factors C and D and their VEGFR-2 and 3 receptors in blood and lymphatic vessels in healthy and arthritic synovium. J Rheumatol. 2002;29:39–45. [PubMed] [Google Scholar]
- 8.Zhang Q, Lu Y, Proulx ST, Guo R, Yao Z, Schwarz EM, Boyce BF, Xing L. Increased lymphangiogenesis in joints of mice with inflammatory arthritis. Arthritis Res Ther. 2007;9:R118. doi: 10.1186/ar2326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Guo R, Zhou Q, Proulx ST, Wood R, Ji RC, Ritchlin CT, Pytowski B, Zhu Z, Wang YJ, Schwarz EM, et al. Inhibition of lymphangiogenesis and lymphatic drainage via vascular endothelial growth factor receptor 3 blockade increases the severity of inflammation in a mouse model of chronic inflammatory arthritis. Arthritis Rheum. 2009;60:2666–2676. doi: 10.1002/art.24764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Engeset A, Tjotta E. Lymphatic pathways from the tail in rats and mice. Cancer Res. 1960;20:613–617. [Google Scholar]
- 11.Leu AJ, Berk DA, Yuan F, Jain RK. Flow velocity in the superficial lymphatic network of the mouse tail. Am J Physiol. 1994;267:H1507–1513. doi: 10.1152/ajpheart.1994.267.4.H1507. [DOI] [PubMed] [Google Scholar]
- 12.Kim S, Lim YT, Soltesz EG, De Grand AM, Lee J, Nakayama A, Parker JA, Mihaljevic T, Laurence RG, Dor DM, et al. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat Biotechnol. 2004;22:93–97. doi: 10.1038/nbt920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kobayashi H, Kawamoto S, Brechbiel MW, Bernardo M, Sato N, Waldmann TA, Tagaya Y, Choyke PL. Detection of lymph node involvement in hematologic malignancies using micromagnetic resonance lymphangiography with a gadolinum-labeled dendrimer nanoparticle. Neoplasia. 2005;7:984–991. doi: 10.1593/neo.05454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kobayashi H, Kawamoto S, Star RA, Waldmann TA, Tagaya Y, Brechbiel MW. Micro-magnetic resonance lymphangiography in mice using a novel dendrimer-based magnetic resonance imaging contrast agent. Cancer Res. 2003;63:271–276. [PubMed] [Google Scholar]
- 15.Kobayashi H, Kawamoto S, Sakai Y, Choyke PL, Star RA, Brechbiel MW, Sato N, Tagaya Y, Morris JC, Waldmann TA. Lymphatic drainage imaging of breast cancer in mice by micro-magnetic resonance lymphangiography using a nano-size paramagnetic contrast agent. J Natl Cancer Inst. 2004;96:703–708. doi: 10.1093/jnci/djh124. [DOI] [PubMed] [Google Scholar]
- 16.Matsui M. ICG fluorescence fundus angiography; the past, the present and the future. Nippon Ganka Gakkai Zasshi. 1993;97:553–554. [PubMed] [Google Scholar]
- 17.Sharma R, Wang W, Rasmussen JC, Joshi A, Houston JP, Adams KE, Cameron A, Ke S, Kwon S, Mawad ME, et al. Quantitative imaging of lymph function. Am J Physiol Heart Circ Physiol. 2007;292:H3109–3118. doi: 10.1152/ajpheart.01223.2006. [DOI] [PubMed] [Google Scholar]
- 18.Sevick-Muraca EM, Sharma R, Rasmussen JC, Marshall MV, Wendt JA, Pham HQ, Bonefas E, Houston JP, Sampath L, Adams KE, et al. Imaging of lymph flow in breast cancer patients after microdose administration of a near-infrared fluorophore: feasibility study. Radiology. 2008;246:734–741. doi: 10.1148/radiol.2463070962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kouskoff V, Korganow AS, Duchatelle V, Degott C, Benoist C, Mathis D. Organ-specific disease provoked by systemic autoimmunity. Cell. 1996;87:811–822. doi: 10.1016/s0092-8674(00)81989-3. [DOI] [PubMed] [Google Scholar]
- 20.Boyce BF. Bone biopsy and histomorphometry in metabolic bone disease. In: Stevenson JC, editor. New techniques in metabolic bone disease. London: Butterworths; 1990. pp. 110–131. [Google Scholar]
- 21.Tilney NL. Patterns of lymphatic drainage in the adult laboratory rat. J Anat. 1971;109:369–383. [PMC free article] [PubMed] [Google Scholar]
- 22.Kyburz D, Corr M. The KRN mouse model of inflammatory arthritis. Springer Semin Immunopathol. 2003;25:79–90. doi: 10.1007/s00281-003-0131-5. [DOI] [PubMed] [Google Scholar]
- 23.Korganow AS, Ji H, Mangialaio S, Duchatelle V, Pelanda R, Martin T, Degott C, Kikutani H, Rajewsky K, Pasquali JL, et al. From systemic T cell self-reactivity to organ-specific autoimmune disease via immunoglobulins. Immunity. 1999;10:451–461. doi: 10.1016/s1074-7613(00)80045-x. [DOI] [PubMed] [Google Scholar]
- 24.Zhang Q, Lu Y, Proulx S, Guo R, Yao Z, Schwarz EM, Boyce BF, Xing L. Increased lymphangiogenesis in joints of mice with inflammatory arthritis. Arthritis Res Ther. 2007;9:R118. doi: 10.1186/ar2326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Maruotti N, Cantatore FP, Crivellato E, Vacca A, Ribatti D. Angiogenesis in rheumatoid arthritis. Histol Histopathol. 2006;21:557–566. doi: 10.14670/HH-21.557. [DOI] [PubMed] [Google Scholar]
- 26.Baluk P, Tammela T, Ator E, Lyubynska N, Achen MG, Hicklin DJ, Jeltsch M, Petrova TV, Pytowski B, Stacker SA, et al. Pathogenesis of persistent lymphatic vessel hyperplasia in chronic airway inflammation. J Clin Invest. 2005;115:247–257. doi: 10.1172/JCI22037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kataru RP, Jung K, Jang C, Yang H, Schwendener RA, Baik JE, Han SH, Alitalo K, Koh GY. Critical role of CD11b+ macrophages and VEGF in inflammatory lymphangiogenesis, antigen clearance, and inflammation resolution. Blood. 2009 doi: 10.1182/blood-2008-09-176776. [DOI] [PubMed] [Google Scholar]
- 28.Goldman J, Le TX, Skobe M, Swartz MA. Overexpression of VEGF-C causes transient lymphatic hyperplasia but not increased lymphangiogenesis in regenerating skin. Circ Res. 2005;96:1193–1199. doi: 10.1161/01.RES.0000168918.27576.78. [DOI] [PubMed] [Google Scholar]
- 29.Kunstfeld R, Hirakawa S, Hong YK, Schacht V, Lange-Asschenfeldt B, Velasco P, Lin C, Fiebiger E, Wei X, Wu Y, et al. Induction of cutaneous delayed-type hypersensitivity reactions in VEGF-A transgenic mice results in chronic skin inflammation associated with persistent lymphatic hyperplasia. Blood. 2004;104:1048–1057. doi: 10.1182/blood-2003-08-2964. [DOI] [PubMed] [Google Scholar]
- 30.Tammela T, Saaristo A, Holopainen T, Lyytikka J, Kotronen A, Pitkonen M, Abo-Ramadan U, Yla-Herttuala S, Petrova TV, Alitalo K. Therapeutic differentiation and maturation of lymphatic vessels after lymph node dissection and transplantation. Nat Med. 2007;13:1458–1466. doi: 10.1038/nm1689. [DOI] [PubMed] [Google Scholar]
- 31.Lynch PM, Delano FA, Schmid-Schonbein GW. The primary valves in the initial lymphatics during inflammation. Lymphat Res Biol. 2007;5:3–10. doi: 10.1089/lrb.2007.5102. [DOI] [PubMed] [Google Scholar]