In addition to CNS tumors, more than 50% of patients with neurofibromatosis type 2 (NF2) develop posterior subcapsular cataracts (PSCs). This study demonstrated that conditional deletion of Nf2 from the lens causes defects in fiber cell proliferation, terminal differentiation, and polarity. The lens provides an assay system to identify pathways critical for fiber cell differentiation and could shed light on how PSCs form in NF2 patients.
Abstract
Purpose.
Neurofibromatosis type 2 (NF2) is an autosomal-dominant CNS tumor syndrome that affects 1:25,000 children and young adults. More than 50% of NF2 patients also develop posterior subcapsular cataracts (PSCs). The authors deleted Nf2 from the lens to determine its role in fiber cell differentiation.
Methods.
Nf2 was conditionally deleted from murine lenses using the LeCre transgene. Standard histology and immunohistochemical and immunofluorescent methods were used to analyze lens morphology and markers of cell cycle progression, differentiation, and cell junctions in wild-type and knockout lenses from embryonic day 10.5 through postnatal day 3.
Results.
Fiber cells lacking Nf2 did not fully exit the cell cycle and continued to express epithelial cell markers, such as FoxE3 and E-cadherin, despite expressing the fiber cell marker Prox1. Many fiber cells lost their elongated morphology. Markers of apical-basal polarity, such as ZO-1, were mislocalized along the lateral and basal membranes of fiber cells. The lens vesicle failed to separate from the surface ectoderm, and prospective lens and corneal epithelial cells formed a multilayered mass of cells at the surface of the eye. Herniation of this membrane caused the fiber mass to erupt through the cornea.
Conclusions.
Nf2 is required for complete fiber cell terminal differentiation, maintenance of cell polarity, and separation of lens vesicle from corneal epithelium. Defects identified in fiber cell differentiation may explain the formation of PSCs in patients with NF2. The lens provides an assay system to identify pathways critical for fiber cell differentiation and to test therapies for the tumors that occur in patients with NF2.
Neurofibromatosis type 2 (NF2) is an autosomal dominant disease that affects 1 in 25,000 children and young adults and is characterized by the predisposition to develop multiple types of nervous system tumors.1 Tumor development in NF2 patients follows the classic “two-hit” model for tumor suppressor genes. Children typically inherit one mutant Nf2 allele. Random inactivation of the remaining wild-type (WT) allele in one cell of a susceptible tissue is sufficient to induce tumorigenesis.2 Characteristic NF2 tumors include schwannomas, meningiomas, ependymomas, and astrocytomas.3 In addition to tumors, more than 50% of NF2 patients develop posterior subcapsular cataracts (PSCs), adding debilitated vision to the complications of the disease.
The product of the Nf2 gene, merlin, is a member of the band 4.1 superfamily of proteins, which often link the actin cytoskeleton to plasma membrane proteins. Merlin is required for contact-dependent growth arrest in cultured primary keratinocytes.4 The interaction and colocalization of merlin with the components of adherens junctions, such as N-cadherin and β-catenin, suggested that merlin may control junctional dynamics, though this has not been explicitly demonstrated. In vivo studies, in which floxed Nf2 was deleted with Nestin-Cre, suggested that merlin is required for the assembly, but not the persistence, of adherens junctions.5 Merlin may control cell growth by suppressing the downstream pathways that are activated by growth factor receptors. Under different conditions, merlin has been shown to alter signaling by the epidermal growth factor receptor6 Ras,7 phosphoinositol-3-kinase,8 and MAP kinase.9
PSCs are one of the three main types of age-related cataracts. Risk factors for PSC formation include diabetes, exposure to immunosuppressive steroids, and therapeutic radiation treatment.10 Although the cellular and molecular mechanisms of PSC formation are not well understood, it is thought that abnormal proliferation of epithelial cells or failure of the proper differentiation of fiber cells may be involved.11,12
Two previous studies reported that loss of Nf2 caused defects in lens development.5,13 However, these defects were not studied further. To better understand PSC formation in general and the PSCs that occur in NF2 patients, we examined the cellular and molecular consequences of conditional deletion of Nf2 from the developing lens.
Nf2 was required for the cessation of cell proliferation that normally accompanies the terminal differentiation of fiber cells, the full expression of markers of fiber cell differentiation, maintenance of cell apical-basal polarity, and successful separation of the lens vesicle from the surface ectoderm. Any of these defects in fiber cell terminal differentiation could contribute to the formation of PSCs in patients with NF2.
Materials and Methods
Generation of Nf2CKO and Genotyping
All animals were treated in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research and with the approval of the Animal Studies Committee of the Washington University School of Medicine. Mice homozygous for a floxed Nf2 allele (Nf2fx/fx), one of which also expressed a Cre-transgene (LeCre),14 were mated to generate offspring, approximately 50% of which were Cre-positive (conditional knockout, Nf2CKO). Cre-negative offspring were considered to be wild type (Nf2WT). Cre-positive animals were always mated to Cre-negative animals, ensuring that Cre-positive offspring inherited only one copy of the Cre transgene (Nf2fx/fx; LeCre−/− X Nf2fx/fx; LeCre±). Cre-mediated deletion of the floxed allele removes exon 2, resulting in the production of a truncated, nonfunctional protein.13 Timed matings were set up, and noon of the day of identification of a vaginal plug was considered embryonic day (E) 0.5. For genotyping, mouse genomic DNA was extracted from toe tissue, and standard PCR was performed as described previously.15
Histology
Embryos or postnatal day (P) 3 mouse heads were fixed in 10% neutral-buffered formalin overnight at 4°C, dehydrated through a series of ethanol concentrations, embedded in paraffin, and sectioned at 4 μm. For morphologic studies, sections were stained with hematoxylin and eosin (Surgipath, Richmond, IL).
Immunohistochemistry
Embryos or P3 mouse heads were fixed in 10% neutral-buffered formalin overnight at 4°C, embedded in paraffin, and sectioned at 4 μm. For antibody staining, sections were deparaffinized and rehydrated. Endogenous peroxidase activity was inactivated with 3% H2O2 in methanol for 30 minutes at room temperature (RT) for those samples that would be treated for horseradish peroxidase (HRP). Epitope retrieval was performed in 0.01 M citrate buffer (pH 6.0). Slides were incubated in blocking solution containing 20% inactivated normal donkey serum for 30 minutes at RT, followed by incubation in primary antibodies overnight at 4°C. Slides were incubated for 1 hour at RT with biotinylated secondary antibodies and were treated with the ABC-peroxidase reagent (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA), followed by treatment with diaminobenzidine and H2O2. The slides were washed with PBS and counterstained with hematoxylin. Slides were visualized on an upright fluorescence microscope (BX60; Olympus, Tokyo, Japan), and images were collected with a digital camera (Spot; Diagnostic Instruments, Sterling Heights, MI). Primary antibodies used for immunohistochemical staining were rabbit anti–human p57KIP2 (8298; Santa Cruz Biotechnology, Santa Cruz, CA) at a 1:1000 dilution and mouse anti–human Ki67 (556003; BD PharMingen, San Diego, CA) at a 1:400 dilution.
BrdU and TUNEL Staining
Pregnant female mice were injected with 50 mg/kg of a mixture of 10 mM BrdU (Roche, Indianapolis, IN) and 1 mM 5-fluoro-5′-deoxyuridine (Sigma, St. Louis, MO) and were killed after 1 hour. A monoclonal anti–BrdU antibody (1:250) (Dako, Carpinteria, CA) was used with an ABC kit (Vectastain Elite Mouse IgG ABC kit; Vector Laboratories). Sections were counterstained with hematoxylin. Terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine triphosphate nick end-labeling (TUNEL) was performed with a fluorescein direct in situ apoptosis detection kit (ApopTag; Chemicon, Temecula, CA). The deparaffinized slides were treated with 3% H2O2 in methanol for 30 minutes, followed by proteinase K treatment (20 μg/mL) for 15 minutes. Slides were incubated with TdT enzyme in equilibration buffer for 1 hour at 37°C. The reaction was terminated with wash buffer, provided by the manufacturer, for 10 minutes at room temperature. Anti–digoxigenin-peroxidase conjugate was added for 30 minutes at room temperature, followed by diaminobenzidine and H2O2 treatment. Slides were counterstained with hematoxylin.
Immunofluorescence
Embryos were fixed as described. After rinsing in PBS, heads were dissected in half, embedded in 4% agarose, and allowed to set overnight at 4°C. Thick sections (120 μm) were cut using a tissue slicer (Electron Microscopy Sciences, Hatfield, PA). Sections containing the lens were blocked in 5% normal goat serum, 0.5% Triton X-100 for permeabilization, and 0.03% sodium azide for 1 hour at RT and were incubated with primary antibodies overnight at 4°C. After rinsing, sections were incubated with fluorescence-labeled secondary antibodies for 1 hour at RT and counterstained with DRAQ-5 (Biostatus Limited, Shepshed, Leicestershire, UK), a vital, fluorescent DNA dye. Sections were mounted in medium (Vectashield; Vector Laboratories) mixed with PBS at a 1:1 ratio, mounted on glass coverslips, and visualized using a confocal microscope (LSM 510; Carl Zeiss, Inc., Thornwood, NY). Primary antibodies used for immunofluorescence in this study were rabbit anti–phospho-histone H3 (3475B; Cell Signaling, Danvers, MA) at 1:1000, rabbit anti–FoxE3 (a gift from Peter Carlsson) at 1:1000, mouse anti–Prox1 (MAB5652; Chemicon InternationalA) at 1:1000, rabbit anti–β-catenin (9587; Cell Signaling) at 1:1000, anti–E-cadherin (13–1900; Invitrogen, Carlsbad, CA) at 1:1000, and anti–ZO-1 (61–7300; Zymed Laboratories, San Francisco, CA) at 1:500. AlexaFluor-labeled phalloidin (Invitrogen) was used to stain for F-actin at 1:1000. AlexaFluor-conjugated anti–mouse and anti–rabbit secondary antibodies (Invitrogen) were used at 1:1000.
Results
To study the role of Nf2 in lens development, we used Cre recombinase to delete floxed alleles of Nf2. Transgenic Cre (LeCre) is first expressed on E9 in the lens placode, the surface ectoderm that will go on to form the lens.14
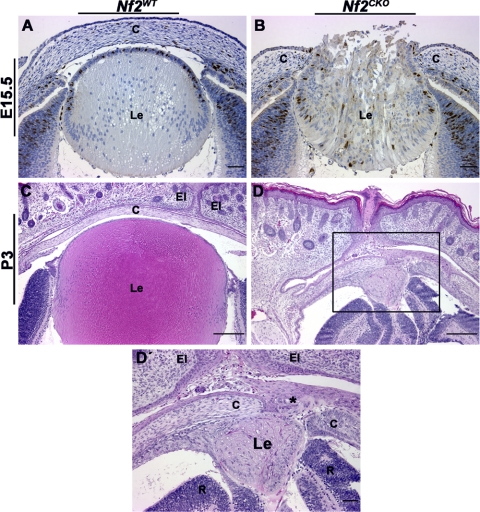
As described previously,5 the failure of lens vesicle separation and the resultant changes in cell organization caused many Nf2CKO lenses to herniate, leading to expulsion of lens fiber cells through a perforation in the cornea (Fig. 1B). By P3, Nf2WT pups had mature lenses and corneas (Fig. 1C). When the lens herniated, Nf2CKO pups were born with abnormally small lenses with serious structural defects (Figs. 1D, 1D′). Presumably because of the lack of normal-size lenses, these eyes were small and retinas were folded.
Figure 1.
Nf2CKO lenses typically herniated through the area of lens-corneal fusion. (A) By E15.5, Nf2WT lenses show clear separation between the developing lens and the cornea. (B) Nf2CKO lenses usually developed lens herniation; fiber cells were expelled through the perforated cornea. (C) At P3, Nf2WT lenses were normal. (D, D′) Nf2CKO pups typically are born with abnormally small, disrupted lenses. Higher magnification shows the lens herniation phenotype. Expelled lens material is trapped between the cornea and the closed eyelids (asterisk). Sections in (A) and (B) were labeled with antibody to BrdU. Scale bars: 50 μm (A, B), 200 μm (C, D), 50 μm (D′, area highlighted by the black box in D). c, cornea; Le, lens; El, eyelid; R, retina.
Nf2 Required for Cell Cycle Withdrawal
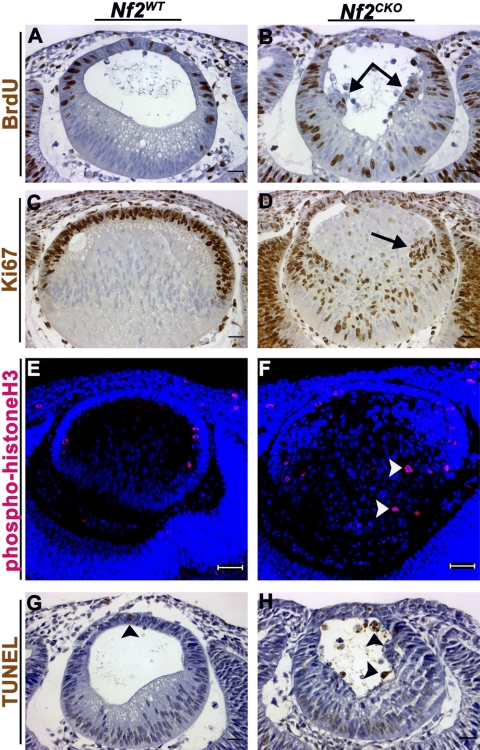
Lens fiber cells normally withdraw from the cell cycle as they initiate terminal differentiation. Nf2WT E11.5 embryos from females that were pulse-labeled with BrdU showed strong labeling in the anterior epithelial cells but not in elongating primary fiber cells (Fig. 2A). However, Nf2CKO lenses had abundant BrdU staining in the nuclei of primary fiber cells (Fig. 2B). BrdU-labeled fiber cells were detected in Nf2CKO lenses throughout subsequent development (not shown). We also stained lenses for Ki67, which labels cells that are actively cycling. Ki67 staining was restricted to the lens epithelium in Nf2WT lenses (Fig. 2C). However, numerous Ki67-positive cells were present in the posterior fiber cell region of Nf2CKO lenses (Fig. 2D). Nf2CKO lenses contained clusters of cells near the lens equator, many of which were BrdU-positive and displayed prominent nuclear accumulation of Ki67 (Figs. 2B, 2D, arrows). Nf2CKO lenses also had phospho-histone H3-positive mitotic figures in the epithelium and in the posterior fiber region, indicating that DNA synthesis was followed by mitosis in at least some fiber cells (Fig. 2F). Mitotic cells were restricted to the epithelial layer of Nf2WT lenses (Fig. 2E).
Figure 2.
Nf2CKO lens fiber cells failed to withdraw from the cell cycle. (A) At E11.5, BrdU labeling was restricted to epithelial cells in Nf2WT lenses. (B) In Nf2CKO lenses. BrdU labeling was present in fiber cells throughout the posterior fiber region. (C) Staining for Ki67 was seen only in anterior lens epithelia in Nf2WT lenses at E13.5. (D) Ki67-positive cells were seen throughout the posterior region in Nf2CKO lenses. (E) At E11.5, phospho-histone H3 staining was seen only at the apical ends of epithelial cells in Nf2WT lenses. (F) Phospho-histone H3-positive mitotic figures were present in the fiber mass of Nf2CKO lenses (arrowheads). (G) At E11.5, Nf2WT lenses displayed normal levels of apoptosis that accompanied normal development (arrowhead). (H) Nf2CKO lenses showed increased TUNEL labeling, indicating increased apoptotic cell death, compared with Nf2WT lenses (arrowheads). Scale bars: 25 μm (A–D, G, H), 50 μm (E, F).
Aberrant proliferation of cells in the process of terminal differentiation is often accompanied by increased apoptotic cell death. TUNEL-positive cells were rarely seen in Nf2WT lenses (Fig. 2G), whereas Nf2CKO lenses showed a marked increase in TUNEL-positive cells in the lens vesicle (Fig. 2H) and, at later stages, in fiber cells (not shown). Increased apoptotic death in Nf2CKO lenses was confirmed using immunofluorescence labeling for cleaved (activated) caspase-3 (data not shown).
Incomplete Terminal Differentiation of Nf2CKO Fiber Cells
In normal lens development, cells at the lens equator withdraw from the cell cycle, move posteriorly, and then elongate to form fiber cells. Withdrawal of differentiating lens cells from the cell cycle requires the cyclin-dependent kinase inhibitor p57KIP2.16 Fiber cell differentiation is also accompanied by the loss of epithelial cell-specific markers, such as the transcription factor FoxE3,17 and increased expression of lens fiber cell-specific markers, such as the transcription factors Prox118 and c-Maf.19,20
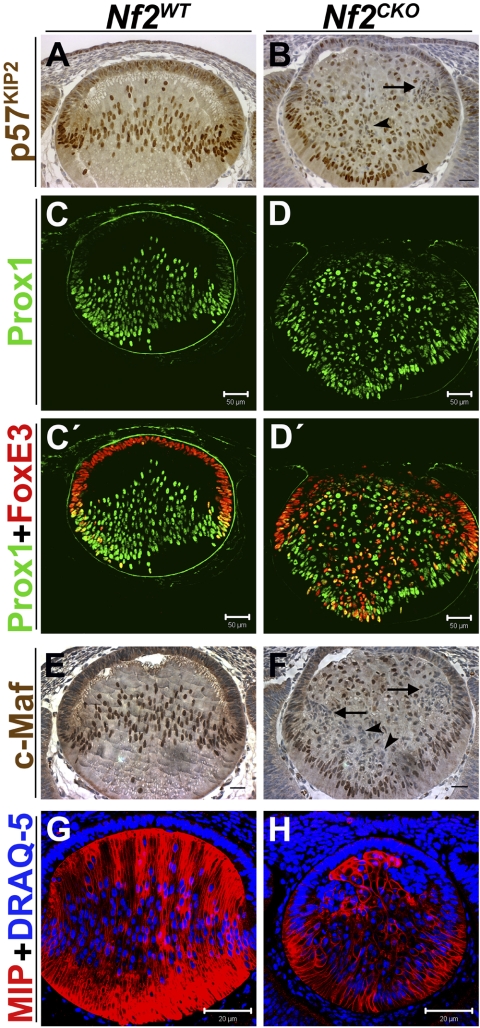
As expected, in Nf2WT lenses, fiber cells showed strong nuclear localization of p57KIP2, beginning near the equatorial region of the lens and extending throughout the posterior fiber cells (Fig. 3A). In Nf2CKO lenses, p57KIP2 was present in the nuclei of many fiber cells. However, even when overstained, some cells in the fiber region displayed weak p57KIP2 staining, or lacked detectable p57KIP2 (Fig. 3B, arrowheads). Lack of p57KIP2 expression was especially noticeable in the clusters of cells that consistently accumulated near the equator of Nf2CKO lenses (Fig. 3B, arrow).
Figure 3.
Most Nf2CKO fiber cells express fiber-specific differentiation markers, but many coexpress epithelium-specific proteins. (A) At E13.5, high levels of p57KIP2 were present in nuclei at the lens equator and throughout the posterior fiber region of Nf2WT lenses. (B) In Nf2CKO lenses, many cells had weak expression or lacked detectable p57KIP2 (arrow and arrowheads). (C) In E12.5 Nf2WT lenses, antibodies to Prox1 labeled the nuclei of cells near the equator and throughout the posterior fiber region. (D) As in WT lenses, Prox1 staining was first detected in nuclei at the equator of Nf2CKO lenses. Careful examination revealed that all cells in the fiber region of the knockout lenses expressed Prox1, though levels were low in some cells. (C′) FoxE3 staining was present in the nuclei of all Nf2WT epithelial cells. Notably, several nuclei near the lens equator were stained with both antibodies. (D′) Antibodies to FoxE3 stained many cells in the posterior fiber compartment of these lenses. Therefore, all fiber cells in Nf2CKO lenses that expressed FoxE3 were also Prox1-positive. (E) In Nf2WT lenses, c-Maf is strongly expressed in the nuclei of all elongating fiber cells. (F) In Nf2CKO lenses, c-Maf is also localized to the nuclei of many fiber cells. However, there are many fiber cells that display either weak labeling or that lack positive labeling for c-Maf (arrowheads). This is especially evident in the clusters of cells near the equatorial region (arrow). Even when the images were contrast enhanced to give the impression of overstaining, the fiber cells in Nf2CKO lenses appeared unstained (arrow and arrowheads). (G, H) At E12.5, antibody to MIP stained fiber cells in Nf2WT and Nf2CKO lenses. However, not all Nf2CKO cells in the fiber compartment were stained. Cells in the epithelioid clusters near the equator had little or no detectable MIP staining. Scale bars: 25 μm (A, B, E, F), 50 μm (C, C′, D, D′), 20 μm (G, H).
In Nf2WT lenses, staining for Prox1, a marker of fiber cell differentiation, was absent in epithelial cells, appeared at the equator, and was strong in all fiber cell nuclei (Figs. 3C, 3C′). In Nf2CKO lenses, all cells in the fiber zone stained for Prox1, though staining intensity varied more than it did in Nf2WT lenses (Figs. 3D, 3D′). In Nf2WT lenses, staining for FoxE3 was strong in the nuclei of lens epithelial cells but was lost near the equator, where epithelial cells differentiate into fiber cells (Fig. 3C′). In Nf2CKO lenses, peripheral lens epithelial cells stained for FoxE3. However, expression was retained in many cells in the posterior fiber region. Many of the cell clusters that contained Ki67- and BrdU-positive cells also had many FoxE3-positive cells (Fig. 3D′), suggesting that the cells in these clusters retain the characteristics of lens epithelial cells. Conversely, in Nf2CKO lenses, many of the nuclei of cells near the central region of the epithelium, where the lens failed to separate from the prospective corneal epithelium, had low or undetectable levels of FoxE3. Occasional double-labeled cells were also seen at the boundary between epithelial and fiber cells in Nf2WT lenses (Fig. 3C′). However, a number of cells throughout the fiber compartment of Nf2CKO lenses were double labeled for Prox1 and FoxE3 (Fig. 3D′).
The expression in Nf2CKO lenses of another fiber-specific transcription factor, c-Maf, differed from that observed for Prox1. As expected, c-Maf expression was strong and localized to the nuclei of all Nf2WT fiber cells, beginning at the bow region (Fig. 3E). In Nf2CKO lenses, c-Maf was correctly expressed in many fiber cells. However, a number of fiber cells either showed weak staining or lacked c-Maf staining altogether (Fig. 3F, arrow and arrowheads). Even when the images were contrast enhanced to create the appearance of overstaining, these Nf2CKO fiber cells showed no detectable c-Maf staining.
MIP, also known as aquaporin 0, is exclusively expressed in lens fiber cells and constitutes approximately 60% of total membrane protein in the fibers, making it a good marker for mature, terminally differentiated fiber cells.21,22 Nf2WT lenses showed strong staining for MIP that was restricted to the fiber cell layer (Fig. 3G). Consistent with data observed for FoxE3 and Prox1, MIP was detected in most cells in the fiber region of Nf2CKO lenses, though staining was generally low or undetectable in the cell clusters near the lens equator (Fig. 3H).
Defects in Cell Adhesion and Loss of Apical-Basal Polarity
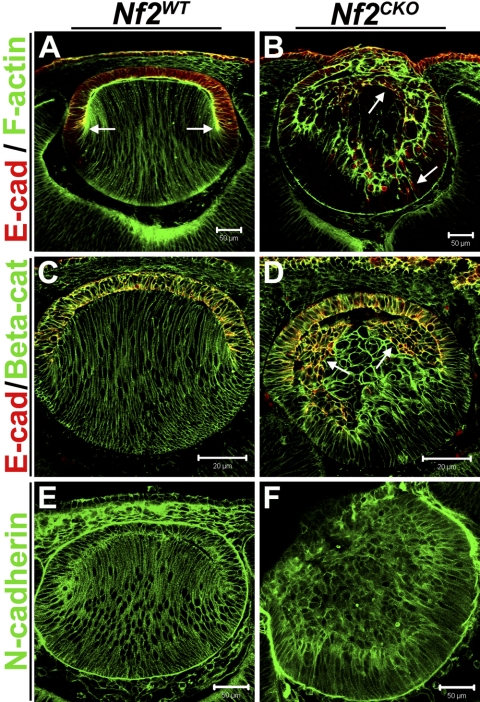
At E12.5, filamentous actin localized at the apical ends of lens epithelial and fiber cells and along the lateral membranes of elongated fiber cells in Nf2WT lenses (Fig. 4A). Although there were no discernible differences in the amount of filamentous actin between Nf2WT and Nf2CKO lenses, F-actin was also more concentrated at the apical ends of fibers cells in the knockout lenses (Fig. 4B).
Figure 4.
Nf2CKO lens fiber cells retained expression of the epithelial cell marker E-cadherin, and some lost their elongated morphology. (A) Staining WT lenses with fluorescent-conjugated phalloidin to label F-actin showed prominent actin staining throughout the lens. Higher levels of F-actin were especially visible at the apical ends of both epithelial and fiber cells. E-cadherin expression labeled cell–cell junctions in the epithelial layer but was absent from differentiated fiber cells (arrow). F-actin colocalized with E-cadherin at the apical ends of epithelial cells. (B) F-actin was also more prominent at the apical ends of elongating fibers cells in Nf2CKO lenses. F-actin was typically seen along the lateral membranes and at the basal ends of Nf2CKO fiber cells. The image shown here is an extreme case of apical F-actin in Nf2CKO lenses, but it also shows double labeling of F-actin and E-cadherin. In Nf2CKO lenses, E-cadherin expression was retained in many fiber cells. Although not completely colocalized, there were areas in the fiber region of CKO lenses in which F-actin colocalized with E-cadherin (arrows). (C) In Nf2WT lenses, β-catenin labeled cellular junctions throughout the lens and specifically colocalized with E-cadherin in the epithelium. (D) In Nf2CKO lenses, E-cadherin that was retained in the posterior fiber zone was completely colocalized with β-catenin and in the epithelium, as would be expected (arrows). (E, F) There were no notable differences in the amount and organization of F-actin, β-catenin, or N-cadherin between Nf2WT and Nf2CKO lenses. Scale bars: 50 μm (A, B, E, F); 20 μm (C, D).
In Nf2WT lenses, E-cadherin was strictly localized to the lens epithelium, where it demarcated cell-cell junctions (Figs. 4A, 4C). Like FoxE3, E-cadherin staining was lost at the lens equator as fiber cells terminally differentiated (Fig 4A, arrows). In contrast, Nf2CKO lenses showed aberrant localization of E-cadherin (Figs. 4B, 4D). Although E-cadherin labeling was present in the epithelium, it was also prominent in cells throughout the posterior fiber region of Nf2CKO lenses (Figs 4B, 4D, arrows). Many of these cells also displayed a rounded morphology rather than their characteristic elongated shape. In some areas in Nf2CKO lenses, F-actin was colocalized with the E-cadherin that was retained in the posterior fiber zone (Fig. 4B). The subcellular localization of β-catenin- and N-cadherin was similar in Nf2WT and Nf2CKO lenses, though the abnormal cell morphology of cells in the Nf2CKO lenses was associated with altered distribution of these proteins (Figs. 3C–F). To test whether loss of merlin function altered adherens junction formation, we performed double labeling for β-catenin and E-cadherin. In Nf2WT lenses, β-catenin labeled cell junctions throughout the entire lens and colocalized with E-cadherin in the epithelium (Fig. 4C). Even though E-cadherin expression was retained in the fiber zone of Nf2CKO lenses, it still colocalized with β-catenin, suggesting that merlin is not required for adherens junction formation in the lens (Fig. 4D).
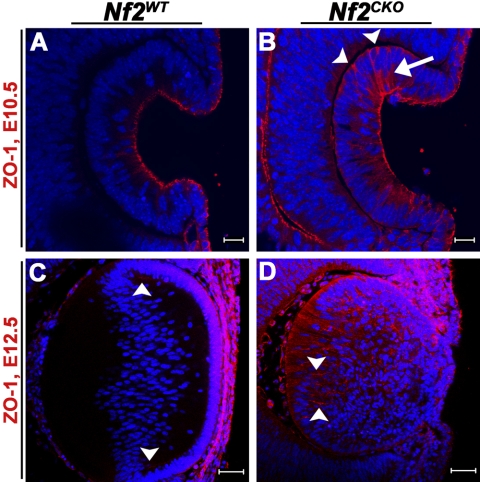
The tight junction protein ZO-1 has previously been shown to localize to the apical membranes of epithelial cells within the developing lens.23,24 In Nf2WT lenses at E10.5, ZO-1 staining was restricted to the apical ends of cells in the invaginating lens pit (Fig. 5A). Nf2CKO lenses also showed intense apical staining for ZO-1. However, in the knockout lenses, ZO-1 expression also extended along the lateral membranes of the cells (Fig. 5B, arrow) and, in some instances, was seen at their basal ends (Fig. 5B, arrowheads). At E12.5, ZO-1 still localized to the apical ends of epithelial and fiber cells of WT lenses, most prominently near the lens equator (Fig. 5C, arrowheads). However, in Nf2CKO lenses, ZO-1 staining was seen throughout the posterior fiber cell region, most intensely along the lateral membranes of the fiber cells (Fig. 5D, arrowheads).
Figure 5.
Nf2CKO lens cells lost apical-basal polarity. (A) Staining for ZO-1 was restricted to the apical ends of WT cells in the invaginating lens vesicles. (B) In Nf2CKO lens vesicle cells, ZO-1 was also present along the lateral (arrow) and basal (arrowheads) ends. (C) At E12.5, ZO-1 was expressed at the apical ends of epithelial and fiber cells of Nf2WT lenses, especially near the lens equator (arrowheads). (D) In Nf2CKO lenses, ZO-1 was expressed throughout the fiber region, especially along fiber cell lateral membranes (arrowheads). Scale bars: 20 μm (A, B); 50 μm (C, D).
Failure of Separation of the Lens Vesicle from the Surface Ectoderm
The lens placode invaginates and separates from the surface ectoderm to form the lens vesicle.25 Formation of the vesicle and its separation from the ectoderm is associated with the fusion and subsequent separation of cell layers, increased cell death, changes in cell-cell associations, maintenance of cell polarity, and the destruction and resynthesis of the basal laminae of the prospective lens and corneal epithelia (Fig. 6).26–29 In Nf2CKO lenses, the margins of the lens pit fused, but the lens vesicle failed to complete the process of separating from the surface ectoderm (Figs. 2B, 2H). Corneal and lens cells in the zone of fusion lost their epithelial morphology and appeared multipolar and multilayered, with extensive extracellular space. In addition, Figure 3D shows that the basal lamina of the lens vesicle, which normally breaks down during separation of the vesicle from the ectoderm (Fig. 6), remained continuous with the basal lamina of the surface epithelial cells in the Nf2CKO lenses. Thus, several aspects of cell behavior that normally accompany vesicle separation failed to occur in Nf2CKO lenses.
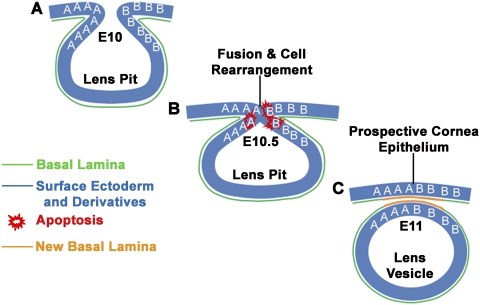
Figure 6.
Diagram of lens vesicle formation. (A) At E10, the lens placode invaginates, forming the lens pit. The letters A and B represent cells located on either side of the lens pit. (B) At E10.5, the sides of the lens pit fuse. (C) By E11, the lens vesicle separates from the surface ectoderm that will become the corneal epithelium. This process involves the fusion of epithelia, the exchange of cell neighbors (some A cells now lie next to B cells) with concomitant destruction and reformation of cell–cell junctions, the destruction of part of the original basal lamina, and the production of a new basal lamina where the epithelia fuse and separate.
Discussion
Examination of developing lenses after conditional deletion of the Nf2 gene revealed four major phenotypes: loss of cell apical-basal polarity, failure of the lens vesicle to separate from the surface ectoderm, failure to properly exit the cell cycle during fiber cell differentiation, and incomplete terminal differentiation of fiber cells.
Conditional deletion of the floxed Nf2 construct used in this study causes in-frame deletion of exon 2, resulting in the expression of a nonfunctional protein. For this reason, it was not possible to use antibody staining to determine at what stage merlin was lost from the lens because antibodies specific to exon 2 were not available. The earliest phenotype detected, aberrant distribution of the tight junction protein ZO-1, was seen at E10.5, followed soon by failure of the lens vesicle to separate from the ectoderm. Because we could not determine when merlin was lost from the lens, it remains possible that defects occurred between E9 and E10.5 that were not detected in this study.
Nf2 Function: Role in Early Fiber Cell Terminal Differentiation
Its unique spatial organization makes the lens a useful model in which to study the mechanisms that control the choice between continued cell proliferation and terminal differentiation.16,30–33 Although it is known that Nf2 is required to suppress excess cell growth in tumors, the mechanisms by which it regulates cell proliferation or quiescence are not understood.4,34–36 The lens may provide a useful in vivo model to shed light on how merlin normally inhibits cells from entering the cell cycle.
Targeted gene deletion in mice revealed that the cyclin-dependent kinase inhibitor Cdkn1c (p57KIP2) and the cell cycle regulator Rb are required for lens fiber cells to withdraw from the cell cycle.16,33 Deletion of the transcription factor Prox1 or receptors for the FGF or BMP families of growth factors decreased p57KIP2 levels and increased cell proliferation in the lens fiber cell compartment.15,18,37 Deletion of c-Maf caused effects similar to those of the loss of Prox1, but p57KIP2 expression was not altered.19,20 Given that the deletion of Nf2 resulted in increased cell proliferation and decreased expression of c-Maf and p57KIP2 in the fiber cell compartment, it may be that merlin is required to link the activation of FGF or BMP receptors to the expression of these factors. In this case, the deletion of Nf2 would interrupt the normal differentiation program, permitting cell proliferation and the continued expression of epithelial cell markers, such as FoxE3 and E-cadherin. However, our data showed that, in the absence of Nf2, most of the cells in the fiber cell compartment elongated and expressed the fiber cell-specific protein MIP. Therefore, though merlin appears to be required for the suppression of lens epithelial characteristics during fiber cell formation, it is not required to activate all aspects of the fiber cell differentiation program.
Although the loss of merlin affects fiber cell terminal differentiation, it is unclear why the loss of merlin leads to the loss of c-Maf and p57KIP2 in some cells without also preventing the expression of Prox1. Further studies are needed to determine where merlin fits in the signaling pathways that control fiber cell differentiation. Based on Ki67 staining, we can conclude that most cells in the posterior Nf2CKO lens are actively cycling. It seems reasonable to suggest that the inhibition of fiber terminal differentiation in the absence of merlin is a secondary effect resulting from a failure of Nf2CKO fiber cells to exit the cell cycle.
Nf2 Required for Lens Cell Adhesion and Polarity
Previous studies suggested that merlin functions in cell-cell adhesion by stabilizing newly formed adherens junctions.5 During the process of lens vesicle invagination, clusters of cells separated from the epithelial layer. These cells became trapped in the lens vesicle and persisted beneath the epithelium near the lens equator. In spite of their loss of association with the lens basement membrane and their rounded appearance, these cells retained several of the molecular characteristics of lens epithelial cells, such as FoxE3 and E-cadherin expression. Together with the redistribution of ZO-1 in Nf2CKO lenses, these results suggest defects in the maintenance of cell adhesion and apical-basal polarity.
Localization of the epithelial-specific cadherin E-cadherin was greatly altered in Nf2CKO lenses. Many fiber cells retained expression of E-cadherin, and many of these cells appeared to lose adhesion or normal polarity. It is unclear whether the loss of fiber cell polarity in Nf2CKO lenses simply reflected the failure to stop cell proliferation and silence the expression of epithelial-specific genes or whether ectopic E-cadherin expression caused defects in polarity by interfering with the function of other adhesion molecules in fiber cells.
Failure of Separation of Lens Vesicle from the Surface Ectoderm: Functional Evidence of Defects in Epithelial Cell Adhesion and Polarity
In humans, an eye defect resulting from the failure of separation of the lens and prospective corneal epithelial layers is called Peters anomaly (OMIM 604229, http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi?id=604229), a syndrome that may occur sporadically or in association with mutations in the transcription factors PAX6, PITX2, or FOXC1 or the enzyme CYP1B1.38 Peters anomaly also occurs in several mouse knockout models used to study lens development.38–41 In Peters anomaly, the lens epithelium remains continuous with the surface ectoderm, the future corneal epithelium. The persistence of this “corneal-lenticular stalk” prevents the migration and differentiation of the mesenchyme cells that would normally form the corneal stroma and endothelium, leading to severe defects in the central cornea.
A significant difference between the phenotype of Nf2CKO eyes and the typical appearance of Peters anomaly was the absence of an obvious corneal-lenticular stalk. In lenses lacking Nf2, cells that would normally form the corneal and lens epithelia lose their epithelial characteristics and form a membrane composed of several layers of loosely organized cells rather than a continuous epithelium connecting the corneal and lens epithelia. These cells appear to lose their apical-basal polarity and their cell-cell and cell-substrate adhesions. As in typical Peters anomaly, the basal lamina of Nf2CKO lenses, the future lens capsule, is continuous with the basal lamina of the surface ectoderm. During normal lens vesicle formation, the basal lamina must be disrupted and then reformed for the lens and future corneal epithelia to separate (Fig. 6). Evidence of more severe defects in cell organization in Nf2CKO eyes was the herniation that typically occurred in later gestation. Although expulsion of the lens contents through the cornea can occur in Peters anomaly, it is a rare complication of this syndrome.42 It was previously suggested that failure of cells lacking Nf2 to properly complete the invagination of the neural tube and lens resulted from the inability to reform cadherin-containing adherens junctional complexes.5 Our observations are consistent with defects in the reformation of cell junctions, but they also suggest more severe deficiencies in cell polarity, cell-substrate adhesion, and substrate remodeling.
Successful separation of the lens vesicle from the surface ectoderm depends on the function of FoxE3.39,43 FoxE3 expression decreased in the anterior epithelial cells of Nf2CKO lenses, especially in the area of fusion between the lens and corneal epithelial cells (Fig. 3D′). This suggests that reduced Foxe3 expression could have contributed to the failure of separation of the lens and cornea. However, the separation defect in Nf2CKO lenses appeared to be more severe than in Foxe3 null lenses, suggesting that additional aspects of merlin function contributed to the severe “fusion” phenotype of Nf2CKO lenses. The apparent dependence of Foxe3 expression on merlin function may warrant further analysis.
Significance of Studying Nf2 in Lens Development
The aberrant fiber cell differentiation or loss of polarity seen in Nf2CKO lenses could help explain the posterior subcapsular cataracts seen in NF2 patients. Because the NF2 phenotype requires the inactivation of both NF2 alleles, loss of merlin function would be expected in only a few cells in the lenses of NF2 patients. If these cells had increased rates of proliferation and aberrant differentiation, they could contribute to PSC formation. The early expression of the Cre transgene used in the present study complicated the analysis of this aspect of the phenotype because the lens separation phenotype often led to expulsion of the fiber cells from the eye. Use of a Cre transgene expressed later in development or induced in the adult eye could provide a more realistic model of the PSCs that develop in patients with NF2 and a genetic model in which to study the cell biology of PSC formation in general. Future studies of merlin function in the lens could identify pathways that might be targeted to provide therapies for the tumors that develop in patients with NF2.
Acknowledgments
The authors thank Ruth Ashery-Padan for providing the LeCre mice, Peter Carlsson for the FoxE3 antibody, Belinda McMahan and Jean Jones for their assistance with histology and immunohistochemistry, and Chenghua Wu for genotyping.
Footnotes
Supported by National Institutes of Health Grant EY04853; Department of Ophthalmology and Visual Sciences Core Grant EY002687; an unrestricted grant from Research to Prevent Blindness, Inc.; National Heart, Lung, and Blood Institute Grant 2 T32 HL007873; and a Ruth L. Kirschstein National Research Service Award from the Developmental Cardiology and Pulmonary Training Program at Washington University (LAW).
Disclosure: L.A. Wiley, None; L.K. Dattilo, None; K.B. Kang, None; M. Giovannini, None; D.C. Beebe, None
References
- 1.Saito H, Yoshida T, Yamazaki H, Suzuki N. Conditional N-rasG12V expression promotes manifestations of neurofibromatosis in a mouse model. Oncogene 2007;26:4714–4719 [DOI] [PubMed] [Google Scholar]
- 2.Gautreau A, Manent J, Fievet B, Louvard D, Giovannini M, Arpin M. Mutant products of the NF2 tumor suppressor gene are degraded by the ubiquitin-proteasome pathway. J Biol Chem 2002;277:31279–31282 [DOI] [PubMed] [Google Scholar]
- 3.McClatchey AI. Neurofibromatosis. Annu Rev Pathol 2007;2:191–216 [DOI] [PubMed] [Google Scholar]
- 4.Lallemand D, Curto M, Saotome I, Giovannini M, McClatchey AI. NF2 deficiency promotes tumorigenesis and metastasis by destabilizing adherens junctions. Genes Dev 2003;17:1090–1100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McLaughlin ME, Kruger GM, Slocum KL, et al. The Nf2 tumor suppressor regulates cell-cell adhesion during tissue fusion. Proc Natl Acad Sci U S A 2007;104:3261–3266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Curto M, Cole BK, Lallemand D, Liu CH, McClatchey AI. Contact-dependent inhibition of EGFR signaling by Nf2/Merlin. J Cell Biol 2007;177:893–903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tikoo A, Varga M, Ramesh V, Gusella J, Maruta H. An anti-Ras function of neurofibromatosis type 2 gene product (NF2/Merlin). J Biol Chem 1994;269:23387–23390 [PubMed] [Google Scholar]
- 8.Rong R, Tang X, Gutmann DH, Ye K. Neurofibromatosis 2 (NF2) tumor suppressor merlin inhibits phosphatidylinositol 3-kinase through binding to PIKE-L. Proc Natl Acad Sci U S A 2004;101:18200–18205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chadee DN, Xu D, Hung G, et al. Mixed-lineage kinase 3 regulates B-Raf through maintenance of the B-Raf/Raf-1 complex and inhibition by the NF2 tumor suppressor protein. Proc Natl Acad Sci U S A 2006;103:4463–4468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Robman L, Taylor H. External factors in the development of cataract. Eye 2005;19:1074–1082 [DOI] [PubMed] [Google Scholar]
- 11.Streeten BW, Eshaghian J. Human posterior subcapsular cataract: a gross and flat preparation study. Arch Ophthalmol 1978;96:1653–1658 [DOI] [PubMed] [Google Scholar]
- 12.Eshaghian J, Streeten BW. Human posterior subcapsular cataract: an ultrastructural study of the posteriorly migrating cells. Arch Ophthalmol 1980;98:134–143 [DOI] [PubMed] [Google Scholar]
- 13.Giovannini M, Robanus-Maandag E, van der Valk M, et al. Conditional biallelic Nf2 mutation in the mouse promotes manifestations of human neurofibromatosis type 2. Genes Dev 2000;14:1617–1630 [PMC free article] [PubMed] [Google Scholar]
- 14.Ashery-Padan R, Marquardt T, Zhou X, Gruss P. Pax6 activity in the lens primordium is required for lens formation and for correct placement of a single retina in the eye. Genes Dev 2000;14:2701–2711 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Rajagopal R, Dattilo LK, Kaartinen V, et al. Functions of the type 1 BMP receptor Acvr1 (Alk2) in lens development: cell proliferation, terminal differentiation, and survival. Invest Ophthalmol Vis Sci 2008;49:4953–4960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang P, Wong C, DePinho RA, Harper JW, Elledge SJ. Cooperation between the Cdk inhibitors p27(KIP1) and p57(KIP2) in the control of tissue growth and development. Genes Dev 1998;12:3162–3167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Landgren H, Blixt A, Carlsson P. Persistent FoxE3 expression blocks cytoskeletal remodeling and organelle degradation during lens fiber differentiation. Invest Ophthalmol Vis Sci 2008;49:4269–4277 [DOI] [PubMed] [Google Scholar]
- 18.Wigle JT, Chowdhury K, Gruss P, Oliver G. Prox1 function is crucial for mouse lens-fibre elongation. Nat Genet 1999;21:318–322 [DOI] [PubMed] [Google Scholar]
- 19.Ring BZ, Cordes SP, Overbeek PA, Barsh GS. Regulation of mouse lens fiber cell development and differentiation by the Maf gene. Development 2000;127:307–317 [DOI] [PubMed] [Google Scholar]
- 20.Yoshida K, Kim JI, Imaki J, et al. Proliferation in the posterior region of the lens of c-maf−/− mice. Curr Eye Res 2001;23:116–119 [DOI] [PubMed] [Google Scholar]
- 21.King LS, Agre P. Pathophysiology of the aquaporin water channels. Annu Rev Physiol 1996;58:619–648 [DOI] [PubMed] [Google Scholar]
- 22.Broekhuyse RM, Kuhlmann ED, Stols AL. Lens membranes II: isolation and characterization of the main intrinsic polypeptide (MIP) of bovine lens fiber membranes. Exp Eye Res 1976;23:365–371 [DOI] [PubMed] [Google Scholar]
- 23.Nguyen MM, Rivera C, Griep AE. Localization of PDZ domain containing proteins Discs Large-1 and Scribble in the mouse eye. Mol Vis 2005;11:1183–1199 [PubMed] [Google Scholar]
- 24.Nielsen PA, Baruch A, Shestopalov VI, et al. Lens connexins alpha3Cx46 and alpha8Cx50 interact with zonula occludens protein-1 (ZO-1). Mol Biol Cell 2003;14:2470–2481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cvekl A, Tamm ER. Anterior eye development and ocular mesenchyme: new insights from mouse models and human diseases. Bioessays 2004;26:374–386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schook P. A review of data on cell actions and cell interaction during the morphogenesis of the embryonic eye. Acta Morphol Neerl Scand 1978;16:267–286 [PubMed] [Google Scholar]
- 27.Webster EH, Jr, Silver AF, Gonsalves NI. The extracellular matrix between the optic vesicle and presumptive lens during lens morphogenesis in an anophthalmic strain of mice. Dev Biol 1984;103:142–150 [DOI] [PubMed] [Google Scholar]
- 28.Mizuno S, Ikeda K, Majima A, Hirabayashi Y, Yamada K. [Histochemical studies on the separation of the lens vesicle]. Nippon Ganka Gakkai Zasshi 1997;101:46–51 [PubMed] [Google Scholar]
- 29.Mohamed YH, Amemiya T. Apoptosis and lens vesicle development. Eur J Ophthalmol 2003;13:1–10 [DOI] [PubMed] [Google Scholar]
- 30.Bassnett S. On the mechanism of organelle degradation in the vertebrate lens. Exp Eye Res 2009;88:133–139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu Y, Zacksenhaus E. E2F1 mediates ectopic proliferation and stage-specific p53-dependent apoptosis but not aberrant differentiation in the ocular lens of Rb-deficient fetuses. Oncogene 2000;19:6065–6073 [DOI] [PubMed] [Google Scholar]
- 32.Pan H, Griep AE. Temporally distinct patterns of p53-dependent and p53-independent apoptosis during mouse lens development. Genes Dev 1995;9:2157–2169 [DOI] [PubMed] [Google Scholar]
- 33.Morgenbesser SD, Williams BO, Jacks T, DePinho RA. p53-dependent apoptosis produced by Rb-deficiency in the developing mouse lens. Nature 1994;371:72–74 [DOI] [PubMed] [Google Scholar]
- 34.Gutmann DH, Haipek CA, Burke SP, Sun CX, Scoles DR, Pulst SM. The NF2 interactor, hepatocyte growth factor-regulated tyrosine kinase substrate (HRS), associates with merlin in the “open” conformation and suppresses cell growth and motility. Hum Mol Genet 2001;10:825–834 [DOI] [PubMed] [Google Scholar]
- 35.Houshmandi SS, Emnett RJ, Giovannini M, Gutmann DH. The neurofibromatosis 2 protein, merlin, regulates glial cell growth in an ErbB2- and Src-dependent manner. Mol Cell Biol 2009;29:1472–1486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Striedinger K, VandenBerg SR, Baia GS, McDermott MW, Gutmann DH, Lal A. The neurofibromatosis 2 tumor suppressor gene product, merlin, regulates human meningioma cell growth by signaling through YAP. Neoplasia 2008;10:1204–1212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang P, Liegeois NJ, Wong C, et al. Altered cell differentiation and proliferation in mice lacking p57KIP2 indicates a role in Beckwith-Wiedemann syndrome. Nature 1997;387:151–158 [DOI] [PubMed] [Google Scholar]
- 38.Prosser J, van Heyningen V. PAX6 mutations reviewed. Hum Mutat 1998;11:93–108 [DOI] [PubMed] [Google Scholar]
- 39.Medina-Martinez O, Brownell I, Amaya-Manzanares F, Hu Q, Behringer RR, Jamrich M. Severe defects in proliferation and differentiation of lens cells in Foxe3 null mice. Mol Cell Biol 2005;25:8854–8863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pontoriero GF, Smith AN, Miller LA, Radice GL, West-Mays JA, Lang RA. Co-operative roles for E-cadherin and N-cadherin during lens vesicle separation and lens epithelial cell survival. Dev Biol 2009;326:403–417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pontoriero GF, Deschamps P, Ashery-Padan R, et al. Cell autonomous roles for AP-2α in lens vesicle separation and maintenance of the lens epithelial cell phenotype. Dev Dyn 2008;237:602–617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Heckenlively J, Kielar R. Congenital perforated cornea in Peter's anomaly. Am J Ophthalmol 1979;88:63–65 [DOI] [PubMed] [Google Scholar]
- 43.Blixt A, Landgren H, Johansson BR, Carlsson P. Foxe3 is required for morphogenesis and differentiation of the anterior segment of the eye and is sensitive to Pax6 gene dosage. Dev Biol 2007;302:218–229 [DOI] [PubMed] [Google Scholar]