Abstract
Rationale: permissive hypercapnia, a stretch-limiting ventilation strategy, often results in high PaCO2. This strategy is associated with reduced morbidity and mortality in premature infants and its benefits have been attributed to diminished barotrauma. However, little is known about the independent effect of high CO2 levels during the lung development. Methods: mice were exposed to 8% CO2 or room air for 2 wk either from postnatal day 2 through 17 or as adults (∼2 mo of age). Lungs were excised and processed for protein, RNA, histology, and total lung volumes. Results: histologic analysis demonstrated that alveolar walls of CO2-exposed mouse pups were thinner than those of controls and had twice the total lung volume. Molecular analysis revealed that several matrix proteins in the lung were downregulated in mouse pups exposed to hypercapnia. Interstitial collagen type I α1, type III α1, elastin and fibronectin protein, and mRNA levels were less than half of controls while collagen IV α5 was unaffected. This decrease in interstitial collagen could thus account for the thinning of the interstitial matrix and the altered lung biomechanics. Matrix metalloproteinase (MMP)-8, a collagenase that has specificity for collagen types I and III, increased in hypercapnic mouse pups, suggesting increased collagen degradation. Moreover, tissue inhibitor of MMP (TIMP)-1, a potent inhibitor of MMP-8, was significantly decreased. However, unlike pups, adult mice exposed to hypercapnia demonstrated only a mild increase in total lung volumes and did not exhibit similar molecular or histologic changes. Conclusions: although permissive hypercapnia may prevent lung injury from barotrauma, our study revealed that exposure to hypercapnia may be an important factor in lung remodeling and function, especially in early life.
Keywords: matrix metalloproteinase, TIMP, collagen, fibronectin, permissive hypercapnia
hypercapnia is a manifestation of many conditions, including pulmonary diseases such as asthma, chronic obstructive pulmonary disease, adult respiratory distress syndrome, and cystic fibrosis. While primary pulmonary disease is a common cause of hypercapnia, other disorders involving the central nervous system (brain stem dysfunction) and neuromuscular disease (proximal muscle disease, amyotrophic lateral sclerosis, and diaphragm dysfunction) can also lead to similar acute or chronic elevations in carbon dioxide (CO2).
Clinical reports have demonstrated that critically ill patients ventilated with a stretch-minimizing ventilation strategy have some improved clinical outcomes (1). While chronic hypercapnia was not the goal in these trials, this ventilation method resulted in hypercapnia and is commonly referred to as “permissive hypercapnia” (4). Yet it is still unclear as to which aspects of permissive hypercapnia are responsible for the beneficial effect. Some theories have proposed that the beneficial effect of this strategy is due to reduced barotrauma. At present, high CO2 exposure is now being studied to determine whether it contributes to the improved clinical outcome (15, 17). Although there are now data that suggest that CO2 may protect the lung against inflammatory and hyperoxic injury after prolonged ventilation (17), the studies on the effect of chronic hypercapnia per se are limited.
There are even fewer investigations related to the effect of hypercapnia on immature animals or infants. Premature infants are born before adequate lung maturation and therefore often require prolonged mechanical ventilation for survival. This group is commonly exposed not only to high oxygen concentrations but also to chronic hypercapnia. As in Adult Respiratory Distress Syndrome (ARDS), studies in the newborn have shown that there may be a decrease in morbidity if a low tidal volume ventilator strategy is used (21). While stretch-limiting strategies may help improve clinical outcomes, it is not known whether the reduced morbidity is attributable to reduced stretch-injury or increased CO2. Hence, there are several unanswered questions regarding the effect of chronic hypercapnia in the perinatal period during which lung development continues.
Lung development is a dynamic process that involves the restructuring of lung tissue from an almost solid tissue to one that is air filled with a tremendous surface area and millions of alveoli. Throughout all the stages of normal lung development, extensive remodeling and changes in extracellular matrix composition occurs (5, 20). Components of the extracellular matrix such as collagen and fibronectin have distinct developmental expression profiles that help direct lung maturation. Premature infants are often born prior to the period of alveolarization and frequently at even earlier stages than the saccular phase, which begins at 28 wk gestation when the lung is comprised of terminal sacs and very primitive alveoli. Thus, due to vast changes in extracellular matrix composition during lung development, we hypothesized that exposure to chronic hypercapnia during this critical period alters lung structure and function.
To test this hypothesis, we exposed spontaneously breathing mouse pups, to chronic hypercapnia from postnatal day 2 through 17 (representing the late saccular and early alveolar stages of lung development) and focused on the changes that were visible in structure and consequently function. In addition, to highlight the importance of hypercapnia during lung development, we compared the effect of chronic hypercapnia on both pups and adult mice. Previous studies on the effect of hypoxia during lung development have already demonstrated that immature animals respond to hypoxia more profoundly with arrested alveolar development, whereas mature animals exposed to similar conditions did not exhibit the same degree of lung remodeling (29). In this study, our goal was to understand the singular effect of hypercapnia (independent of hypoxia) on lung structure and function specifically in pups to help explain how it may impact clinical outcomes in infants.
METHODS
Overview.
Mice of two ages were studied to analyze the effect of chronic hypercapnia on postnatal lung development. We exposed newborn pups (from postnatal day 2 through 17) and adult mice (∼2 mo of age) to either 2 wk of 8% CO2 (experimental) or room air (control group). Separate litters and/or adult mice were used for each condition and analyzed for lung volume measurements, histology, and molecular analysis. All experiments had ≥6 animals/group for each methodology performed unless stated otherwise.
CO2 exposure.
A computer-controlled system (OxyCycler, Reming Bioinstruments, Redfield, NY) was used to introduce and maintain constant levels of 8% CO2 and oxygen levels of 21%, as described previously (19). CD-1 mice were used in all experiments. This strain was chosen because it has been extensively evaluated in our laboratory. All litters were culled to eight pups each and weight-matched with controls. At postnatal day 2, litters and their dams were placed in Plexiglas chambers with regular 12:12-h light/dark cycles. Control litters were housed in identical chambers and exposed to room air. Adult male mice (∼2 mo of age) were placed in either 8% CO2/21% O2 or room air for 2 wk. All studies were conducted in pathogen-free conditions. All experimental protocols were approved by the University of California, San Diego (UCSD) Institutional Animal Care and Use Committee, which is accredited by the American Association of Laboratory Animal Care; animals were treated according to guidelines approved by UCSD.
Blood chemistries.
Animals were anesthetized and venous blood was obtained on the day of death. Total carbon dioxide levels were measured and calculated to reduce the impact of acute pH and Pco2 levels. Blood gases, bicarbonate, total carbon dioxide, and chloride levels were processed by the Animal Care Program Diagnostic Laboratory at UCSD.
Respiratory rate.
Respiratory rates, oxygen saturation, and pulse rates were recorded on unrestrained unanesthetized mice either in room air or in the hypercapnia chamber using the MouseOx system (Starr Life Sciences, Oakmont, PA) as per manufacturer's instruction. Data were collected for at least 5 min and only used when no error code was given; respiratory rates were then calculated for an average as well as a mode.
Measurement of lung capacity.
Animals were euthanized and lungs were inflated through a tracheostomy (24-gauge Angiocath, BD Medical, Sandy, UT) secured with a silk ligature. The lungs were inflated, with the thorax intact, to 30 cmH2O for 1 min with a calibrated glass syringe (1 ml, Hamilton, Reno, NV). Pressure was measured with a calibrated manometer (model HHP-201, Omega Engineering, Meriam Instruments, Cleveland, OH). Lungs were then deflated to 5 cmH2O for 1 min and again to 30 cmH2O for 1 min. This cycle was repeated 3 times. Volumes at 30 cmH2O for the last three inflations were averaged. Internal compliance of the syringe/manometer/tubing system was corrected for in each experiment. Eight animals were used per group in the younger animals and four per group were used in the adults with no air leaks in any animal.
Processing of lung tissue.
For the histological analysis, animals were euthanized using a lethal dose of inhaled isoflourane, and lung tissue was processed as described previously (19). Briefly, lungs were inflated through tracheotomies to 25 cm pressure with 4% paraformaldehyde (pH 7.4). Lungs and hearts were excised en bloc, submersed in 4% paraformaldehyde overnight, and processed for paraffin embedding and sectioned into 5-μm-thick sections by the core histology facility at UCSD. Lung tissues were then stained with PCNA (DAKO, Carpinteria, CA) or ProLong Gold antifade reagent with DAPI (Invitrogen Molecular Probes, Carlsbad, CA) as per manufacturer's instructions. For Western blot analysis, animals were euthanized as described above, and lungs were excised, frozen in liquid nitrogen, and stored at −80°C until analysis.
Image analysis.
Four random lung trichrome sections from four separate animals per study group were taken at ×10 magnification using AxioVision (Carl Zeiss, Thornwood, NY). Images were then processed in Photoshop CS4 (Adobe Systems, San Jose, CA) and masked for any empty areas, blood vessels, and large airways. Metamorph (Molecular Devices, Sunnyvale, CA) image analysis software was used to threshold images for air spaces (white areas). Air spaces were calculated as a percentage of white area/total image area minus masked areas.
Protein analysis.
We studied the biochemistry of the lung structure and matrix components in frozen lung tissue, processed as described previously (19). Briefly, frozen tissues were homogenized, sheared, and centrifuged, and supernatants were taken as whole tissue lysates. Protein concentration was measured using the bicinchoninic acid kit, according to the manufacturer's instructions (Sigma-Aldrich, St. Louis, MO). Equal amounts of protein (50 μg) were separated on 4–12% precast NuPAGE bis-Tris SDS-PAGE gels (Invitrogen, Carlsbad, CA) and transferred to polyvinylidene difluoride membranes (Immobilin-P; Millipore, Bedford, MA). Western blots were performed with antibodies against HSC-70 (Stressgen/Assay Designs, Ann Arbor, MI), 1:10,000; Collagen types I α1, III α1, IV α5, VI α1, Elastin, MMP-13 and MMP-8 1:200 (Santa Cruz Biotechnology, Santa Cruz, CA); MMP-14 1:500 (R&D Systems, Minneapolis, MN); Fibronectin 1:200 (B&D Bioscience, San Jose, CA). Specific bands were visualized after incubation with the respective secondary antibodies using enhanced chemiluminescense (GE Healthcare/Amersham Biosciences, Buckinghamshire, UK). Densitometry of Western blots from each experimental group were obtained (n ≥ 6), and absolute values were normalized to HSC-70. Results were reported in arbitrary units, comparing each value with that obtained from each respective HSC-70 measurement on each blot. TIMP-1 ELISA assay was performed on total lung homogenates using R&D TIMP-1 ELISA (R&D Systems) assay as per manufacturer's instructions.
Soluble collagen assay.
Collagen level was measured using a Sircol Soluble Collagen Assay (Biocolor Ltd, Belfast, UK), an assay comparable to the hydroxyproline method of collagen analysis, as per manufacturer's instructions. In brief, lungs were removed after 2 wk of chronic hypercapnia or room air at postnatal day 17 and homogenized with 0.5 M acetic acid solution. Soluble collagen levels were determined by using Sirius Red, an anionic dye with sulphonic acid side chain groups that reacts with the side chain groups of the basic amino acids present in collagen. Samples were shaken for 30 min then centrifuged for 5 min at 16,000 rpm. Unbound dye solution was drained and an alkali reagent was added to release the bound dye. One hundred microliters of each sample and standard was then transferred to a 96-well plate, and the optical density was measured at 540nm wavelength using a microplate reader (BioTek Microplate Reader, BioTek Instruments, Winooski, VT). Lung collagen content was calculated using a standard curve and adjusted to the sample's wet lung weight.
Real-time PCR.
For RNA analysis, animals were euthanized as previously described and lung tissue removed using sterile techniques. Lungs were immediately frozen in liquid nitrogen and stored in −80°C until samples were ready for RNA extraction. Total RNA was extracted using a RNA midi prep (Qiagen, Valencia, CA) as per manufacturer's instructions. RNA was tested for quality and measured for concentration using a spectrophotometer (Beckman Coulter, Fullerton, CA). One microgram of total RNA was used to synthesize the cDNA library with random hexamers and Superscript RT III First Strand (Invitrogen) as per manufacturer's directions. Real-time PCR was then used to quantify mRNA levels of collagen types I α1 and type III α1 and fibronectin. Specific primers were designed for collagen type I α1, fibronectin, GAPDH (using Primer-Blast, a web-based primer design program) and collagen type III α1 (16). Experiments were performed under the following conditions: 95°C for 10 min followed by 40 cycles of 95°C/15 s, 60°C/60 s, then 95°C/15 s, 60°C/15 s, and 95°C/15 s, using SYBR-green (Applied Biosystems, Foster City, CA) on a AB 7600 RT-PCR machine (Applied Biosystems).
Statistical analysis.
Student's t-test was used for comparison of effects of hypercapnia on protein (Western blot), RNA, ELISA, histology, blood chemistries, image analysis, and lung function; differences between sample means were considered significant if the P values <0.05.
RESULTS
Blood chemistries.
Blood gases were obtained on the day of death after 2 wk of hypercapnia. After 2 wk of hypercapnia, pups developed elevated Pco2 levels (control 36 vs. experimental 63 mmHg, P < 0.01) but did not significantly differ in pH (control pH 7.34 vs. experimental pH 7.28, P = 0.05). Since animals were anesthetized before blood sampling, Pco2 levels may not accurately represent the level of hypercarbia, thus additional blood chemistries that reflect chronic hypercapnia were obtained. Total carbon dioxide levels (measured, not calculated) and bicarbonate were obtained. Both bicarbonate (control 20 vs. experimental 29.5 mM/l, P < 0.0001) and total carbon dioxide (control 21 vs. experimental 31 mmHg, P < 0.0001) levels were elevated in the experimental group. In addition, pups exposed to 2 wk of constant 8% CO2 also had decreased serum chloride levels (control 109.5 vs. experimental 97.8 meq/l, P < 0.001, n = 6 each group for all blood chemistries).
Body weight and lung-to-body weight ratios.
Body weights (controls 9.3 vs. experimental 9.5 gm, n ≥ 20 per group, P = 0.22) and the wet-to-dry lung ratios (controls 1.23 vs. experimental 1.25, n = 6 per group, P = 0.06) of mouse pups were not altered by 2 wk of chronic hypercapnia. In addition no differences in morbidity or mortality were noted between groups. Litters were weight-matched and culled to 8 pups/litter to minimize interlitter differences.
Lung structure.
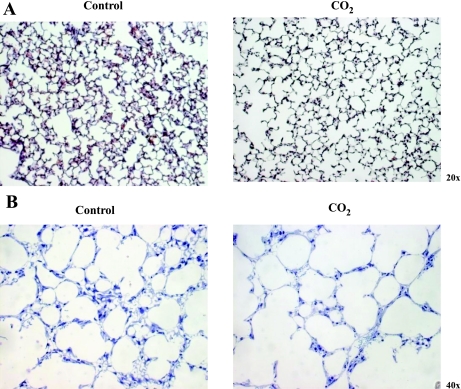
Pups exposed to hypercapnia had significantly thinner alveolar walls but similar body weights between control and hypercapnic groups (Fig. 1). Alveolar air spaces were 5% greater in pups exposed to hypercapnia on lung sections at ×10 magnification (70.1 vs. 74.9% air space/image, P = 0.05, n = 4 images/animal, 4 animals/group). Thinning of alveolar walls was not associated with fewer alveoli as determined by mean linear intercept measurements (MLI) (19).
Fig. 1.
A: 2-wk exposure to 8% CO2 results in decreased alveolar interstitial thickness without changing mean linear intercept (×20 magnification). B: ×40 magnification of 1-μm-thick sections demonstrate that interstitial spaces between alveoli are thinner in the CO2 group.
Lung volumes and respiratory rate.
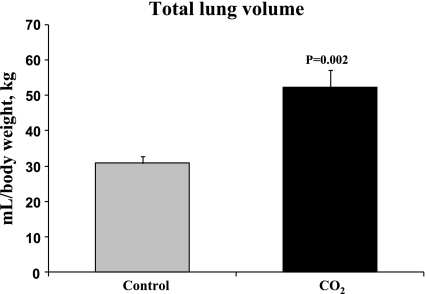
To determine if the alveolar wall thinning had functional significance, total lung volume was determined by inflating the lungs in situ at 30 cmH2O pressure. At this pressure, the experimental pups had lung volumes that were approximately twice those of controls (controls 30.9 vs. 52.4 ml/kg body wt, n ≥ 8 each group, P < 0.002; Fig. 2), suggesting that static compliance in the exposed group was much larger than that of controls. Respiratory rates on unrestrained, unanesthetized mice breathing room air (control) or 8% CO2/21% O2 balanced with nitrogen were not significantly different between groups [average: control 212 vs. experimental 241 respirations/min (rpm), P = 0.12 or mode: control 215 vs. 218 rpm, P = 0.62]. In addition, exposure to hypercapnia did not alter oxygen saturation (control 99 vs. experimental 98% saturation, P > 0.05) or pulse rate (control 650 vs. experimental 690 beats/min, P > 0.05, n = 13 control n = 11 experimental for all respiratory measurements).
Fig. 2.
Static lung volumes at 30 cmH20 pressure (representing total lung volume) demonstrate an increase in lung volume in mice exposed to chronic hypercapnia, n ≥ 8 for each group.
Cellular proliferation and cell death.
Cellular proliferation was unaffected by hypercapnia as demonstrated by PCNA staining. There were on average 360 PCNA-positive cells/20 images for the control group, and 346 PCNA-positive cells for the experimental pup group, P = 0.51, n = 4 per group. In addition, Caspase 3 protein levels were similar between the two groups, suggesting the rate of cell death was not affected (data not shown). To determine if a decrease in cell number could account for thinning of the interstitial space, alveolar areas (areas with no bronchi or blood vessel) were analyzed for cell number, as determined by DAPI-staining cells, per high power view. There were on average 353 DAPI-positive cells in the control group and 375 DAPI-positive cells/40 images in the experimental group, P = 0.21, n = 4 animals/group and 5 random images/animal.
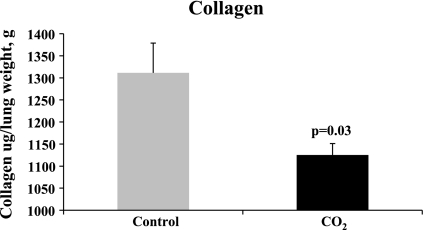
Collagen levels are decreased.
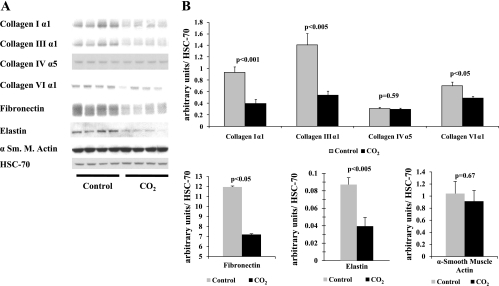
Total collagen levels were assessed using the Sircol Collagen Assay, a well-established method for collagen quantification. Using this method, overall soluble collagen level was significantly decreased in the experimental group. As demonstrated in Fig. 3, soluble collagen was reduced by 14% in the experimental group (1,311 vs. 1,125 μg collagen/g lung, P = 0.03, n = 8 each group). To determine which collagen types were affected, we tested several collagen subtypes by Western blot analysis. Figure 4 demonstrates that both collagen type I α1 and III α1 decreased by more than half in the experimental pup group (0.93 vs. 0.39 arbitrary units/Hsc-70 for collagen I α1, P < 0.001 and 1.41 vs. 0.53 arbitrary units/ Hsc-70 for collagen III, n ≥ 6 each group, P < 0.005). Collagen type VI α1 was also decreased by hypercapnia (control 0.70 vs. experimental 0.48 arbitrary units/Hsc-70, n ≥ 6 each group, P < 0.05) whereas collagen type IV α5 (which resides predominantly in the basement membrane) did not change with hypercapnia (control 0.30 vs. experimental 0.29 arbitrary units/Hsc-70, n ≥ 8 for each group, P = 0.59). In addition we found that mRNA levels of both collagen types I α1 and III α1 were also significantly reduced by fivefold (collagen type I α1: control 0.212 vs. experimental 0.04 arbitrary units/GAPDH, n = 8 per group, P < 0.005; collagen type III α1 control 0.051 vs. experimental 0.009 arbitrary units/GAPDH, n = 8/group, P < 0.001).
Fig. 3.
Collagen was significantly reduced in the CO2 group compared controls using a Sircol Collagen Assay. Collagen was reduced by 14% in the experimental group (1,311 vs. 1,125 μg collagen/g lung, P = 0.03, n = 8 each group).
Fig. 4.
A: representative Western blots collagen types I α1, III α1, IV α5, and VI α1, fibronectin, elastin, α smooth muscle actin, and Hsc-70. B: densitometry of Western blots demonstrates a reduction of collagen types I α1, III α1, and VI α1, whereas collagen type IV α5 was unaltered. Fibronectin and elastin levels were also significantly reduced but CO2 did not affect alpha smooth muscle actin protein levels, n ≥ 6 each group.
Fibronectin and elastin levels were decreased.
Fibronectin protein levels were also examined since it is a component of the matrix and a molecule that connects collagen fibrils (26). Figure 4 demonstrates that fibronectin levels were reduced to nearly half in the experimental group compared with nonexposed pups (control 11.96 vs. experimental 7.20 arbitrary units/Hsc-70, n ≥ 6 each group, P < 0.004). Similarly, fibronectin mRNA levels were reduced by fivefold in experimental animals (control 0.005 vs. experimental 0.001 arbitrary units/GAPDH, n = 8 per group, P < 0.05). In addition, elastin (precursor tropoelastin and mature elastin) protein levels were reduced in the experimental groups (control 0.087 vs. experimental 0.039 arbitrary units/Hsc-70, n ≥ 6 each group, P < 0.005).
Alpha smooth muscle actin was not significantly altered by hypercapnia.
Despite the significant changes in several matrix proteins in pups, Western blot analysis of whole lung tissues demonstrated that smooth muscle actin protein level was not affected (Fig. 4).
Hypercapnia increased collagenase activity.
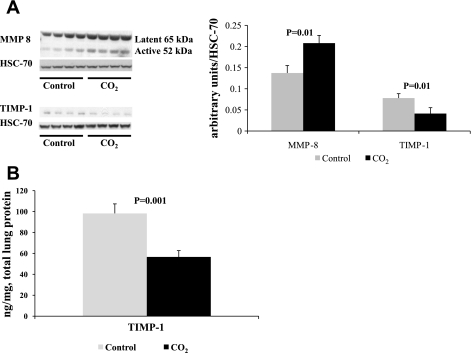
Western blot analysis of whole lung tissues demonstrated that MMP-8 levels were significantly increased by exposure to hypercapnia in pups (Fig. 5). While both latent and active forms were detected, the active form increased 1.5 times from control (control 0.14 vs. experimental 0.21 arbitrary units/Hsc-70, n ≥ 6 for each group, P = 0.01). This increase in MMP-8 levels may account for the decrease in collagen levels since MMP-8 is a major collagenase and is able to degrade collagen types I and III. MMP-14 was also examined due to its ability to degrade collagen but its levels were not significantly different between the control and experimental groups (0.21 vs. 0.16 arbitrary units/Hsc-70, n ≥ 6 for each group, P > 0.05). In addition, MMP-13 another potent collagenase was not significantly altered (0.17 vs. 0.14 arbitrary units/Hsc-70, n ≥ 6 for each group, P > 0.05). However, TIMP-1 protein, an inhibitor of MMP-8, was decreased in pups exposed to hypercapnia (Fig. 5A, 0.077 vs. 0.041 arbitrary units/Hsc-70, respectively, n ≥ 6 for each group, P = 0.01). ELISA assays also revealed that TIMP-1 decreased to nearly half of control levels (98 vs. 56 ng TIMP-1/mg total lung protein, n = 5 per group, P = 0.001, Fig. 5B).
Fig. 5.
A: representative Western blots illustrate that active MMP-8 levels increase with hypercapnia, whereas TIMP-1 protein levels are significantly decreased. Densitometry of Western blots demonstrate significance at P = 0.01 for both proteins, n ≥ 6 for each group. B: TIMP-1 ELISA assay also confirms the reduction of function in the CO2 group, P = 0.001, n = 5.
Impact of age.
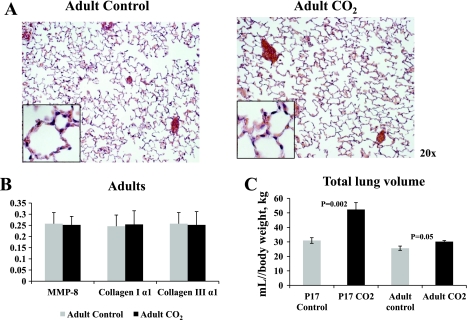
Adult mice were exposed to the same level of chronic constant hypercapnia but demonstrated only a subtle response to hypercapnia. As in pups, there were no differences in body weights between adult experimental and control groups (36.5 vs. 37.4 g, n ≥8 for each group, P = 0.22). At the end of the experimental period, respiratory rates between controls and adults were not significantly different [average: control 223 respirations per min (rpm) vs. experimental 206 rpm, P = 0.30 or mode: control 222 rpm vs. 200 rpm, P = 0.31]. In addition, exposure to hypercapnia did not alter oxygen saturation (control 98% vs. experimental 97% saturation, P = 0.5) or pulse rate (control 645 vs. experimental 597 beats/min, P = 0.43, n = 9 control n = 9 experimental for all respiratory measurements). Adult mice exposed to hypercapnia developed an increase in total blood carbon dioxide (control 27 vs. adult experimental 33 mM/l, P < 0.01) and decrease in serum chloride (control 115 meq/l vs. adult experimental 112 meq/l, n ≥ 4 for each group, P = 0.02). While hypercapnia induced a thinning of interstitial spaces in pups, adults exposed to similar conditions did not develop alveolar wall thinning (Fig. 6A). There was no significant difference in air space area between control and experimental groups on image analysis (78% air space/image at ×10 magnification for control and experimental groups, P = 0.9). In addition, matrix proteins as wells as MMP-8 were comparable between the groups (Fig. 6B). Collagen type I α1 (0.25 vs. 0.25 arbitrary units/Hsc-70, n ≥ 4 for each group, P > 0.05) and collagen type III α1 levels (0.26 vs. 0.25 arbitrary units/Hsc-70, n = 4, P > 0.05) were only minimally changed. Additionally, MMP-8 protein levels were not significantly altered (control 0.26 vs. experimental 0.25 arbitrary units/Hsc-70, n = 4, P > 0.05). Interestingly however, adult hypercapnic mice still demonstrated a modest increase in total lung volumes (25.6 vs. 30.2 ml/body wt, kg, n = 4, P = 0.05, Fig. 6C) and compliance (control 0.85 vs. experimental 1 ml·cmH2O−1·kg−1, n = 4, P = 0.05).
Fig. 6.
A: adult mice exposed to a 2-week exposure to 8% CO2 did not demonstrate a significant change in lung morphology (×20 magnification with a ×40 insert). B: densitometry of Western blots did not demonstrate a significant difference between adult control and experimental groups for collagen types I α1. III α1, or MMP-8 protein levels, n ≥ 5 for each group, P > 0.05. C: static lung volumes at 30 cmH2O pressure (representing total lung volume) demonstrate that adult mice exposed to hypercapnia only demonstrated a 15% increase in lung volume (P = 0.05), whereas pups exposed to the same conditions increased 40% (P < 0.002) from age matched controls, n ≥ 8 for pup groups and n = 4 for adult groups.
DISCUSSION
Our study demonstrated that exposure to hypercapnia significantly increased total lung volumes in mouse pups. To probe whether hypercapnia induced increased total lung volumes through stretch mechanisms, two stages of development, pups and adults were studied. According to literature, the mechanical response (increased tidal volume and respiratory rate) to hypercapnia should be greater in adults than in pups assuming that CO2 is sensed to the same extent by pups and adults (2, 23, 27). However, the opposite resulted. We found that pups exposed to hypercapnia developed a greater increase in total lung volumes than adults. Hence, the mechanical response to hypercapnia cannot ultimately be responsible for the changes seen in pups. Other investigators have also demonstrated that only immature animals exposed to hypercapnia develop increased lung compliance even weeks after the exposure (24). In these studies, adults did not develop changes in lung compliance, alveolar air space volume, respiratory rate or septal tissue volumes when exposed to the same conditions (18, 24). Although the mechanical response to hypercapnia may contribute to the modest increase in lung volumes seen in adults, we propose that the mechanical response alone cannot account for the changes seen in pups exposed to chronic hypercapnia.
The phenotype seen in the pup is clearly different from the adult exposed to hypercapnia. We propose two possibilities to account for the increase in total lung volume seen in the immature animal. First, it is possible that exposure to hypercapnia during lung development induced more lung growth. It was recently reported that mouse pups exposed to chronic hypercapnia developed more alveoli as evidenced by an increase in alpha smooth muscle actin localizing to tips of alveolar buds by 1 wk of hypercapnia (9). The study by Das et al. (9) suggests that exposure to hypercapnia induces alveoli formation. While our data did not demonstrate a change in cellular proliferation or apoptosis after 2 wk, it is possible that there was an increase in proliferation in the first week of hypercapnia/earlier in development that stabilized by 2 wk of hypercapnia. Despite this finding, it is still unclear whether hypercapnia accelerates lung growth or permanently alters it. Alveolarization, grossly determined by counting alveoli (MLI), was not significantly different between groups. Our data demonstrate that alveolar air spaces were only slightly enlarged in pups exposed to hypercapnia while static lung volumes were significantly increased at 30 cmH2O pressure. This difference between histologic analysis and physiological changes seen may be due to irregular postmortem shrinkage in paraffin embedded lungs processed for histology (13). Thus another explanation for the increased total lung volumes in pups is due to changes in molecular lung structure resulting in increased lung compliance.
Lung compliance can be altered through changes in matrix composition. Given that the matrix is made up of mostly collagen [collagens 60–70% (22), 20–30% elastins (22), 1% glycosaminoglycans, and 0.5% fibronectins (10)], we focused our investigation on collagen. Exposure to chronic hypercapnia resulted in a decrease in total soluble collagen and elastin protein. Interstitial collagen types I and III, the major constituents in lung parenchyma (3), were most affected. It is worthwhile noting that while both collagen protein and RNA were decreased, they were not decreased to the same degree, suggesting that RNA and protein turnover at different rates. Alternatively, collagen may be controlled at both transcriptional and translational levels.
Matrix metalloproteinases (MMPs) are proteases that can directly degrade all the known components of the extracellular matrix, including collagens and fibronectin (11, 30). MMPs are abundantly expressed during development and disease states where dramatic changes in lung structure are taking place (7, 8, 11, 25). While the mechanisms that act trigger MMP activation are still largely unknown, mechanical stimuli as well as hormones, growth factors, and cytokines have all been shown to activate MMPs in vitro (6, 12). MMPs are important modulators of lung remodeling because they have specific affinities for matrix components. This ability to alter specific matrix components allows MMPs to orchestrate precise structural changes that are required during processes such as morphogenesis and wound repair. We hypothesized that an increase in MMP-8, a potent collagenase with high substrate affinities to both collagen types I/III (28), could account for the specific decrease in collagen types I and III. We found that MMP-8 increased at the same time that its targets decreased. In addition, other MMPs with collagenase activity but less affinity to collagens types I and III were not significantly altered. Furthermore, tissue inhibitor of metalloproteinases-1 (TIMP-1), a potent MMP-8 inhibitor, was also decreased thus further shifting the balance toward proteolysis and possibly increasing lung compliance. When MMP-8 levels did not increase in adults exposed to hypercapnia, collagen types I and III were unaffected. Since MMP-8 is only minimally expressed in the healthy adult, a pathologic state where MMP levels are already high may be required to achieve the same proteolytic state as the pups.
We found that chronic exposure to hypercapnia can lead to increased lung volumes with decreased collagen and elastin levels, yet we do not know whether this is beneficial to the lung or to respiration. As to the benefit of chronic hypercapnia in the adult, we speculate that our studies on normal mice exposed to moderate hypercapnia may not accurately predict what may occur in a disease state. In addition, while chronic hypercapnia may provide some benefit in the lung, it may adversely affect other organs such as the brain (14). Furthermore, our data might underestimate the impact of hypercapnia on premature infants since they are often hypercapnic as early as 24 wk of gestation (canalicular stage of lung development), whereas our mice were exposed from postnatal day 2 through 17, which encompasses the saccular and alveolar stages of lung development. Clearly further studies are needed to determine the impact of hypercapnia in the development or prevention of lung injury; however, our study provides insight into a consequence of prolonged hypercapnia during lung development.
GRANTS
This work was supported by National Institutes of Health Grant HD-P01-32573-14 to G. G. Haddad.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the authors.
ACKNOWLEDGMENTS
We thank Peter Wagner and Grace Ryu for editorial review and Shirley Reynolds and Christopher Lingad for help with animal care.
REFERENCES
- 1. Amato MB, Barbas CS, Medeiros DM, Magaldi RB, Schettino GP, Lorenzi-Filho G, Kairalla RA, Deheinzelin D, Munoz C, Oliveira R, Takagaki TY, Carvalho CR. Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med 338: 347– 354, 1998 [DOI] [PubMed] [Google Scholar]
- 2. Bamford OS, Schuen JN, Carroll JL. Effect of nicotine exposure on postnatal ventilatory responses to hypoxia and hypercapnia. Respir Physiol 106: 1– 11, 1996 [DOI] [PubMed] [Google Scholar]
- 3. Bradley KH, McConnell SD, Crystal RG. Lung collagen composition and synthesis. Characterization and changes with age. J Biol Chem 249: 2674– 2683, 1974 [PubMed] [Google Scholar]
- 4. Burchardi H. New strategies in mechanical ventilation for acute lung injury. Eur Respir J 9: 1063– 1072, 1996 [DOI] [PubMed] [Google Scholar]
- 5. Burri PH. Postnatal growth and maturation of the lung. Chest 67: 2S– 3S, 1975 [DOI] [PubMed] [Google Scholar]
- 6. Chakraborti S, Mandal M, Das S, Mandal A, Chakraborti T. Regulation of matrix metalloproteinases: an overview. Mol Cell Biochem 253: 269– 285, 2003 [DOI] [PubMed] [Google Scholar]
- 7. D'Armiento J, Dalal SS, Okada Y, Berg RA, Chada K. Collagenase expression in the lungs of transgenic mice causes pulmonary emphysema. Cell 71: 955– 961, 1992 [DOI] [PubMed] [Google Scholar]
- 8. Dalal S, Imai K, Mercer B, Okada Y, Chada K, D'Armiento JM. A role for collagenase (matrix metalloproteinase-1) in pulmonary emphysema. Chest 117: 227S– 228S, 2000 [DOI] [PubMed] [Google Scholar]
- 9. Das S, Du Z, Bassly S, Singer L, Vicencio AG. Effects of chronic hypercapnia in the neonatal mouse lung and brain. Pediatr Pulmonol 44: 176– 182, 2009. [DOI] [PubMed] [Google Scholar]
- 10. Field J. Handbook of Physiology: a critical, comprehensive presentation of physiological knowledge and concepts. Bethesda, MD: Am Physiol Soc, 1959, p. 1– 10 [Google Scholar]
- 11. Greenlee KJ, Werb Z, Kheradmand F. Matrix metalloproteinases in lung: multiple, multifarious, and multifaceted. Physiol Rev 87: 69– 98, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Haseneen NA, Vaday GG, Zucker S, Foda HD. Mechanical stretch induces MMP-2 release and activation in lung endothelium: role of EMMPRIN. Am J Physiol Lung Cell Mol Physiol 284: L541– L547, 2003 [DOI] [PubMed] [Google Scholar]
- 13. Hsia CC, Hyde DM, Ochs M, Weibel ER. ATS/ERS Joint Task Force on Quantitative Assessment of Lung Structure An Official Research Policy Statement of the American Thoracic Society/European Respiratory Society: Standards for Quantitative Assessment of Lung Structure. Am J Respir Crit Care Med 181: 394– 418, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Kaiser JR, Gauss CH, Pont MM, Williams DK. Hypercapnia during the first 3 days of life is associated with severe intraventricular hemorrhage in very low birth weight infants. J Perinatol 26: 279– 285, 2006 [DOI] [PubMed] [Google Scholar]
- 15. Kantores C, McNamara PJ, Teixeira L, Engelberts D, Murthy P, Kavanagh BP, Jankov RP. Therapeutic hypercapnia prevents chronic hypoxia-induced pulmonary hypertension in the newborn rat. Am J Physiol Lung Cell Mol Physiol 291: L912– L922, 2006 [DOI] [PubMed] [Google Scholar]
- 16. Krag S, Nyengaard JR, Wogensen L. Combined effects of moderately elevated blood glucose and locally produced TGF-[beta]1 on glomerular morphology and renal collagen production. Nephrol Dial Transplant 22: 2485– 2496, 2007 [DOI] [PubMed] [Google Scholar]
- 17. Laffey JG, Tanaka M, Engelberts D, Luo X, Yuan S, Tanswell AK, Post M, Lindsay T, Kavanagh BP. Therapeutic hypercapnia reduces pulmonary and systemic injury following in vivo lung reperfusion. Am J Respir Crit Care Med 162: 2287– 2294, 2000 [DOI] [PubMed] [Google Scholar]
- 18. Lechner AJ, Blake CI, Banchero N. Pulmonary development in growing guinea pigs exposed to chronic hypercapnia. Respiration 52: 108– 114, 1987 [DOI] [PubMed] [Google Scholar]
- 19. Li G, Zhou D, Vicencio AG, Ryu J, Xue J, Kanaan A, Gavrialov O, Haddad GG. Effect of carbon dioxide on neonatal mouse lung: a genomic approach. J Appl Physiol 101: 1556– 1564, 2006 [DOI] [PubMed] [Google Scholar]
- 20. McGowan SE. Extracellular matrix and the regulation of lung development and repair. FASEB J 6: 2895– 2904, 1992 [PubMed] [Google Scholar]
- 21. Miller JD, Carlo WA. Safety and effectiveness of permissive hypercapnia in the preterm infant. Curr Opin Pediatr 19: 142– 144, 2007 [DOI] [PubMed] [Google Scholar]
- 22. Pierce JA, Ebert RV. Fibrous Network of the Lung and its Change with Age. Thorax 20: 469– 476, 1965 [Google Scholar]
- 23. Putnam RW, Conrad SC, Gdovin MJ, Erlichman JS, Leiter JC. Neonatal maturation of the hypercapnic ventilatory response and central neural CO2 chemosensitivity. Respir Physiol Neurobiol 149: 165– 179, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Rezzonico R, Gleed RD, Mortola JP. Respiratory mechanics in adult rats hypercapnic in the neonatal period. J Appl Physiol 68: 2274– 2279, 1990 [DOI] [PubMed] [Google Scholar]
- 25. Ryu J, Vicencio AG, Yeager ME, Kashgarian M, Haddad GG, Eickelberg O. Differential expression of matrix metalloproteinases and their inhibitors in human and mouse lung development. Thromb Haemost 94: 175– 183, 2005 [DOI] [PubMed] [Google Scholar]
- 26. Sottile J, Hocking DC. Fibronectin polymerization regulates the composition and stability of extracellular matrix fibrils and cell-matrix adhesions. Mol Biol Cell 13: 3546– 3559, 2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Stunden CE, Filosa JA, Garcia AJ, Dean JB, Putnam RW. Development of in vivo ventilatory and single chemosensitive neuron responses to hypercapnia in rats. Respir Physiol 127: 135– 155, 2001 [DOI] [PubMed] [Google Scholar]
- 28. Van Lint P, Libert C. Matrix metalloproteinase-8: cleavage can be decisive. Cytokine Growth Factor Rev 17: 217– 223, 2006 [DOI] [PubMed] [Google Scholar]
- 29. Vicencio AG, Eickelberg O, Stankewich MC, Kashgarian M, Haddad GG. Regulation of TGF-beta ligand and receptor expression in neonatal rat lungs exposed to chronic hypoxia. J Appl Physiol 93: 1123– 1130, 2002. [DOI] [PubMed] [Google Scholar]
- 30. Woessner JF, Nagase H. Nagase Matrix Metalloproteinases and TIMPs. Oxford University Press, 2000 [Google Scholar]