Abstract
Barrett’s esophagus (BE) increases the risk for development of esophageal adenocarcinoma. Because of the rapid rise in incidence of esophageal adenocarcinoma, screening for BE with subsequent surveillance when found has been proposed as a method of early detection. Sedated endoscopy, however, is too expensive for wide spread screening. As a result, other techniques including unsedated transnasal esophagoscopy and capsule esophagoscopy have been proposed to expand screening programs.
Barrett’s esophagus (BE) is a condition in which the normal squamous epithelium of the esophagus is replaced with metaplastic intestinal-type epithelium. This epithelium can progress sequentially from metaplasia to low-grade dysplasia to high-grade dysplasia and finally to invasive adenocarcinoma. BE is associated with a 0.5% annual incidence of high grade dysplasia or esophageal adenocarcinoma (1). Esophageal adenocarcinoma is a deadly illness for which the prognosis depends upon early detection. Given the stage-like progression of BE from dysplasia to adenocarcinoma, identification and endoscopic surveillance of patients with BE could reduce mortality from esophageal adenocarcinoma. This strategy is supported by data showing that esophageal adenocarcinoma detected during surveillance programs is found at an earlier stage than that detected outside of surveillance programs (2–4).
Screening the entire population for BE is unreasonable and costly, so risk factors for Barrett’s esophagus have been identified, including age over 50, white race, male gender, obesity, smoking, and gastroesophageal reflux disease (GERD) (2–11). Professional society guidelines recommend consideration on an individual basis of screening endoscopy in older patients (particularly white males over age 50) with long-standing symptoms of GERD. Further, these guidelines recommend consideration of endoscopic surveillance of patients diagnosed with BE (12–14). To date, no prospective, randomized-controlled trials have evaluated the benefits of surveillance.
More controversial, and currently not recommended outside the research setting, is the concept of screening asymptomatic patients for BE. BE is known to be present in patients without GERD (15) and up to 57% of patients with esophageal adenocarcinoma have never reported symptoms of typical GERD (16–18). As a result, a significant portion of patients at risk for esophageal adenocarcinoma will not be screened under current guidelines.
Because of the large population that could benefit from screening, in lieu of controlled trials, cost analyses have been performed. Modeling studies comparing screening and surveillance of patients over age 50 (predominantly white males) with standard upper endoscopy to no screening have shown incremental cost effectiveness ratios between $10,440–$86,833 per quality-adjusted life year (19–21) or $4,530–$12,140 per life year gained (22, 23) depending upon different modeling assumptions and surveillance intervals. Given the standard incremental cost effectiveness ratio that society is willing to pay of $50,000 per QALY, this would theoretically make screening appear cost-effective. However, these studies have a number of limitations including: a] underestimating costs, b] failure to consider the impact of screening on quality of life, c] overestimating the accuracy of endoscopy and biopsies, and d] overly optimistic assumptions of patient compliance with screening (24). As a result, research efforts have evaluated unsedated examinations and capsule esophagoscopy as ways to reduce cost of screening. Additionally, efforts are underway to identify the patient population that would benefit most from screening examinations. Currently screening for BE, while widely practiced, is controversial and should be considered on a case-by-case basis. The purpose of this article is to review the techniques available for screening.
SCREENING METHODS
STANDARD SEDATED UPPER ENDOSCOPY
Evaluation for BE on upper endoscopy requires knowledge of the anatomy of the gastroesophageal junction (Figure 1). Examination of the esophagus begins with gastric decompression (a stomach full of air will impede full opening of the esophagus). Once the stomach is deflated, the endoscope is withdrawn slowly, seeking the diaphragmatic attachment. This can be identified as the point at which the esophagus (or cardia in the case of a hiatal hernia) expands and collapses during breathing or when the patient is asked to sniff (“sniff” test). The next landmark to identify is the top of the gastric folds (the transition between the gastric rugae and the smooth esophagus). The final landmark is the squamo-columnar junction (or z-line) where the gastric mucosa abruptly transitions to squamous mucosa. Normally, the top of the gastric folds coincides with the attachment of the diaphragm and the squamocolumnar junction. In a hiatal hernia, the top of the gastric folds is found proximal to the diaphragmatic attachment but coincides with the z-line. BE is suspected endoscopically when the squamocolumnar junction is proximal to the top of the gastric folds (Figure 2). When the distance between the top of the gastric folds and the z-line is less than 3 cm, short segment BE is diagnosed, whereas a distance greater than 3 cm is consistent with long-segment BE.
Figure 1.
Anatomy of hiatal hernia and Barrett’s esophagus.
Figure 2.
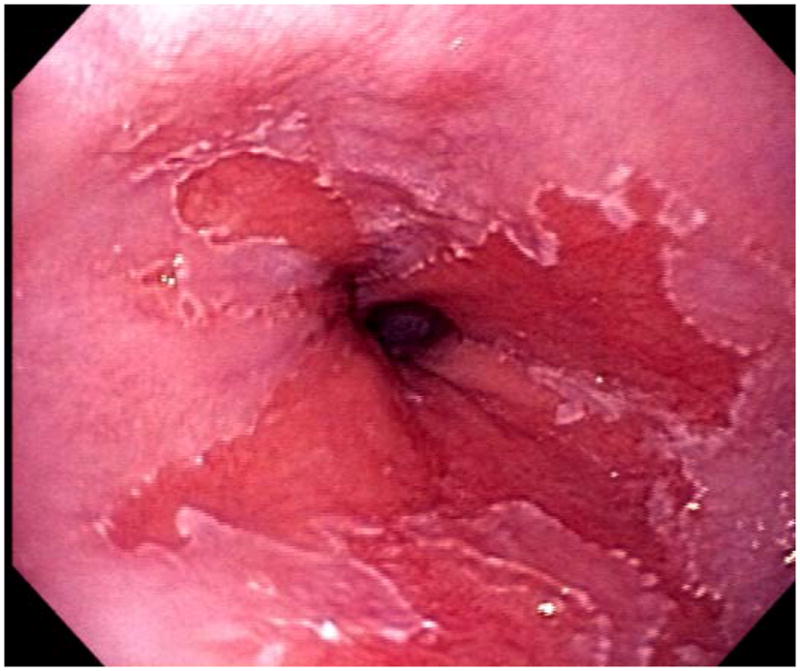

Endoscopic pictures of Barrett’s esophagus. A. Non-dysplastic Barrett’s esophagus. B. Barrett’s esophagus with a nodule of high grade dysplasia.
In an attempt to uniformly classify BE, the Prague C & M criteria were developed (25). The extent of circumferential BE (denoted C) as well as the maximal extent (denoted M) is noted. For example, a segment of BE that is circumferential from 35–40 cm with tongues of BE that extend up to 33 cm would be labeled as C5M7 for 5 cm of circumferential BE and 7 cm maximum extent.
Some controversy exists as to the best way to identify the gastroesophageal junction. While the Prague C & M criteria use the upper extent of the gastric folds to identify the end of the esophagus, other groups use the disappearance of the distal esophageal palisade vessels as the landmark for the gastroesophageal junction (Figure 3). The palisade vessels are endoscopically visible as they course through the mucosa of the distal esophagus. However, they disappear into the submucosa at the gastroesophageal junction and can no longer be visualized endoscopically in the cardia. These vessels are predominantly used in Japan, where there is an increased prevalence of gastric atrophy, making the upper extent of the gastric folds more difficult to identify. A comparative study showed improved agreement between endoscopists in the localization of the gastroesophageal junction through use of the upper extent of the gastric folds as opposed to the palisade vessels (26). Clinically, both landmarks can aid the endoscopist in localization of the gastroesophageal junction.
Figure 3.
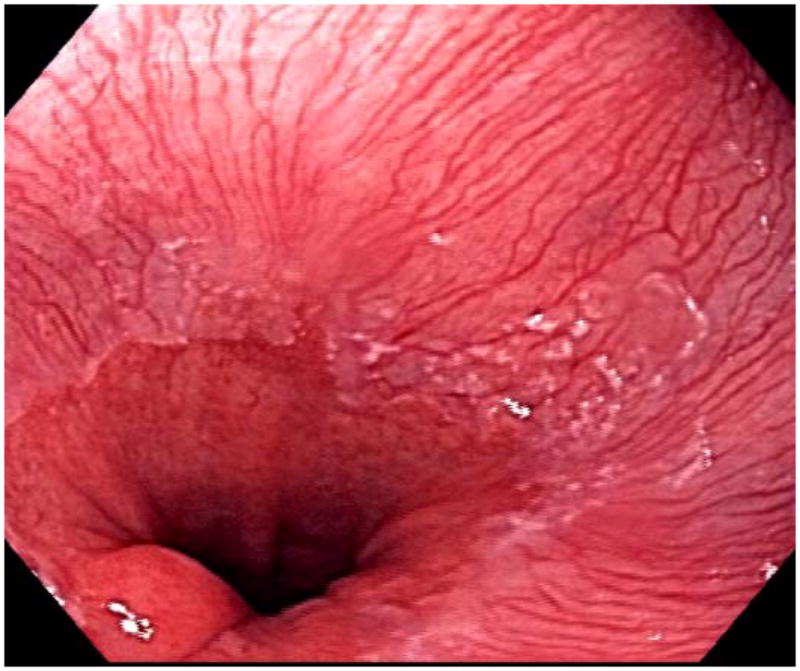
Pallisade vessels of the distal esophagus.
To confirm endoscopic suspicion of BE, biopsies should be taken of the salmon-colored mucosa above the gastric folds. It is critical that biopsies are taken above the gastric folds as intestinal metaplasia of the esophagus (BE) warrants surveillance whereas intestinal metaplasia of the cardia does not. In other words, diagnosis of BE requires both endoscopic and pathologic diagnosis. Identification of goblet cells in columnar epithelium of esophageal biopsies confirms the diagnosis of BE (Figure 4). Biopsies of the esophagus, especially in the region just proximal to the lower esophageal sphincter, are somewhat more challenging than other parts of the gastrointestinal tract because of the frequent motor activity and the tangential angle from which biopsies are obtained. One helpful maneuver termed the “turn and suck” technique is to pull an open biopsy forceps back against the endoscope and turn the scope into the mucosa. This can be followed by pressing the suction button to draw the mucosa into the forceps prior to closing the forceps and taking the biopsy. Prior to obtaining surveillance biopsies, the segment of BE should be examined closely for suspicious nodules or irregular mucosa that could represent areas of dysplasia or early malignancy. Focused biopsies should be obtained first from mucosal irregularities. Next, 4-quadrant biopsies should be taken at a minimal interval of every 2 cm of BE and placed in separate jars labeled by distance from incisors. Biopsies should be sent to pathology with a clear description of endoscopic findings. This allows for aiding localization of dysplasia (if discovered) on repeat endoscopy.
Figure 4.

Pathology of Barrett’s esophagus. A. Non-dysplastic Barrett’s esophagus. B. Barrett’s esophagus with high-grade dysplasia.
UNSEDATED ESOPHAGOSCOPY
Because of the large population at risk for BE, techniques to decrease cost of a screening program have been evaluated. One such technique uses a small-caliber (3–5 mm in diameter) upper endoscope passed transorally or transnasally into the esophagus solely with topical anesthesia in an unsedated patient. This technique has been shown to be tolerable, safe, and accurate in evaluating the esophagus (27–33). Our current procedure is to have the patient sit in a chair for the procedure after a four-hour fast. (The supplementary video is available online at www.techgiendoscopy.com.) We use an atomizer to instill 7 mL of aerosolized 4% lidocaine and 0.05% oxymetazoline hydrochloride into the patent nares over five minutes. Next, we administer a 3-second instillation of aerosolized 14% benzocaine into the oropharynx. We then have the patient put their chin to their chest and pass a 4.9 mm diameter flexible endoscope with a 2 mm working channel transnasally through the nasal turbinates and into the oropharynx, hypopharynx, esophagus, and stomach. We have found that the 5 mm caliber scope that we use remains tolerable and allows for biopsies to be taken at the time of unsedated exam, obviating the need for sedated EGD if non-dysplastic BE is discovered.
Unsedated small caliber endoscopic screening for BE has been shown to be more cost-effective than standard endoscopy (22). However, in the current environment where many patients expect sedation prior to endoscopy, availability of this technique has not been shown to increase the referral pattern for screening (34). At present, in the United States, unsedated transnasal esophagoscopy is used predominantly for selected patients or in the research setting.
CAPSULE ESOPHAGOSCOPY
Although not studied, capsule esophagoscopy may be a more patient-friendly technique for unsedated BE screening. The esophageal capsule has two optical domes that allows for capture of 14 images per second (compared to two images per second with the small bowel capsule) (35). The operating time is limited to 20 minutes. This technique has used varying ingestion protocols to gain optimal views of the esophagus. The original protocol called for an 8-hour fast prior to ingestion of 100 mL of water (to wash saliva away) followed immediately by laying supine. The patient remained supine for 2 minutes, then was passively raised to 30 degrees for 2 minutes, then 60 degrees for 2 minutes, and finally allowed to sit upright. The patients were instructed not to speak or swallow for 4 minutes after the capsule entered the esophagus. A new protocol in which the capsule is swallowed with the patient lying in the right lateral supine position and takes sips of water every 30 seconds during the 3 minutes following ingestion provides better visualization of the z-line (36, 37). A third technique (38, 39) involves attaching a tethered sleeve onto the capsule allowing the operator to control the capsule during the examination (string capsule endoscopy). In this technique, the patient swallows the capsule in the seated position and the capsule travels to 50 cm from the incisors. Next, the operator pulls the capsule back through the esophagus to the upper esophageal sphincter where it is swallowed again allowing for three passes through the esophagus. The capsule can be retrieved and disinfected for re-use, allowing for four capsules to be used to evaluate 100 patients in one study (38). The act of pulling the string back and forth can be uncomfortable for patients and might obviate the perceived attractiveness of capsule endoscopy over unsedated endoscopy.
This technique has shown a variable sensitivity of 60%–96%, a specificity of 84%–100%, a positive predictive value of 22–100%, and a negative predictive value of 61%–98% when compared with standard EGD (37, 39, 40–42). The low sensitivity can be attributed to failure to see the entire gastroesophageal junction in certain patients. Results in known short segment BE were even less encouraging with 44% of cases being identified by one observer and only 16% by another. The procedure is also more expensive and has not been shown to be cost effective in its current form compared with standard endoscopy in two modeling studies (22, 24). Additionally, its reading time of 8–15 minutes (40) is similar to that of EGD, so capsule esophagoscopy is unlikely to decrease physician time in BE screening (43). Finally, while patients prefer capsule esophagoscopy to EGD, this preference is likely to reverse once a patient discovers EGD is more accurate and may be necessary anyway after a non-diagnostic capsule study (43). Thus, in its present form, capsule esophagoscopy is unlikely to replace standard EGD in BE screening. However, as further innovation improves the capsule’s accuracy and decreases cost, it is conceivable that capsule esophagoscopy could play a major role in BE screening.
CONCLUSION
BE is a pre-cancerous condition for which known risk factors exist and for which surveillance programs have been shown to diagnose cancer at earlier stages. Current recommendations allow for consideration of EGD in patients at risk (particularly white males over 50 with chronic GERD) on a case by case basis. However, because screening this population alone has been shown to miss esophageal cancer and Barrett’s esophagus, less expensive alternatives to standard EGD are being evaluated as a way to broaden to population screened. At present, we agree that screening should be considered on a case-by-case basis in older patients with chronic GERD. Patients at risk should be offered less expensive alternatives to standard sedated endoscopy where this is available. Patients with severe comorbidities who are poor surgical candidates should be excluded from screening programs. Research efforts should continue to identify those patients who would benefit most from screening and to improve technology allowing for cheaper yet effective screening techniques.
Supplementary Material
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Sharma P, Falk GW, Weston AP, et al. Dysplasia and cancer in a large multicenter cohort of patients with Barrett’s esophagus. Clin Gastroenterol Hepatol. 2006;4:566–572. doi: 10.1016/j.cgh.2006.03.001. [DOI] [PubMed] [Google Scholar]
- 2.Streitz JM, Andrews CW, Ellis FH. Endoscopic surveillance of Barrett’s esophagus. J Thorac Cardiovasc Surg. 1993;105:383–388. [PubMed] [Google Scholar]
- 3.Peters JH, Clark GWB, Ireland AP, et al. Outcome of adenocarcinoma arising in Barrett’s esophagus in endoscopically surveyed and nonsurveyed patients. J Thorac Cardiovasc Surg. 1994;108:811–822. [PubMed] [Google Scholar]
- 4.vanSandick J, vanLanschot J, Kuiken B, et al. Impact of endoscopic biopsy of surveillance of Barrett’s oesophagus on pathological stage and clinical outcome of Barrett’s carcinoma. Gut. 1998;43:216–22. doi: 10.1136/gut.43.2.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Spechler SJ, Goyal RK. Barrett’s esophagus. N Engl J Med. 1986;315:362–371. doi: 10.1056/NEJM198608073150605. [DOI] [PubMed] [Google Scholar]
- 6.Williamson WA, Ellis FH, Gibb SP, et al. Barrett’s esophagus: Prevalence and incidence of adenocarcinoma. Arch Intern Med. 1991;151:2212–6. doi: 10.1001/archinte.151.11.2212. [DOI] [PubMed] [Google Scholar]
- 7.Kabat GC, Ng SK, Wynder EL. Tobacco, alcohol intake, and diet in relation to adenocarcinoma of the esophagus and gastric cardia. Cancer Causes Control. 1993;4:123–32. doi: 10.1007/BF00053153. [DOI] [PubMed] [Google Scholar]
- 8.Brown LM, Swanson CA, Gridley G, et al. Adenocarcinoma of the esophagus: Role of obesity and diet. J Natl Cancer Inst. 1995;87:104–9. doi: 10.1093/jnci/87.2.104. [DOI] [PubMed] [Google Scholar]
- 9.Vaughan TL, Davis S, Kristal A, et al. Obesity, alcohol, and tobacco as risk factors for cancers of the esophagus and gastric cardia: Adenocarcinoma versus squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 1995;4:85–92. [PubMed] [Google Scholar]
- 10.Gammon MD, Schoenberg JB, Ahsan H, et al. Tobacco, alcohol, and socioeconomic status and adenocarcinomas of the esophagus and gastric cardia. J Natl Cancer Inst. 1997;89:1277–84. doi: 10.1093/jnci/89.17.1277. [DOI] [PubMed] [Google Scholar]
- 11.Chow WH, Blot WJ, Vaughan TL, et al. Body mass index and risk of adenocarcinomas of the esophagus and gastric cardia. J Natl Cancer Inst. 1998;90:150–5. doi: 10.1093/jnci/90.2.150. [DOI] [PubMed] [Google Scholar]
- 12.Wang KK, Wongkeesong M, Buttar NS, et al. American Gastroenterological Association medical position statement: Role of the gastroenterologist in the management of esophageal carcinoma. Gastroenterology. 2005;128:1468–70. doi: 10.1053/j.gastro.2005.03.076. [DOI] [PubMed] [Google Scholar]
- 13.Hirota WK, Zuckerman MJ, Adler DG, et al. ASGE guideline. The role of endoscopy in the surveillance of premalignant conditions of the upper GI tract. Gastrointest Endosc. 2006;63:570–80. doi: 10.1016/j.gie.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 14.Wang KK, Sampliner RE. Updated guidelines 2008 for the diagnosis, surveillance, and therapy of Barrett’s esophagus. Am J Gastroenterol. 1998;103:788–97. doi: 10.1111/j.1572-0241.2008.01835.x. [DOI] [PubMed] [Google Scholar]
- 15.Lagergren J, Bergstrom R, Lindgren A, et al. The role of tobacco, snuff and alcohol use in the aetiology of cancer of the oesophagus and gastric cardia. Int J Cancer. 2000;85:340–6. [PubMed] [Google Scholar]
- 16.Chak A, Faulx A, Eng C, et al. Gastroesophageal reflux symptoms in patients with adenocarcinoma of the esophagus or cardia. Cancer. 2006;107:2160–6. doi: 10.1002/cncr.22245. [DOI] [PubMed] [Google Scholar]
- 17.Lagergren J, Bergstrom R, Lindgren A, et al. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N Engl J Med. 1999;340:825–831. doi: 10.1056/NEJM199903183401101. [DOI] [PubMed] [Google Scholar]
- 18.Reavis KM, Morris CD, Gopal DV, et al. Laryngopharyngeal reflux symptoms better predict the presence of esophageal adenocarcinoma than typical gastroesophageal reflux symptoms. Ann Surg. 2004;239:849–856. doi: 10.1097/01.sla.0000128303.05898.ee. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Inadomi JM, Sampliner R, Lagergren J, et al. Screening and surveillance for Barrett esophagus in high-risk groups: a cost-utility analysis. Ann Intern Med. 2003;138:176–186. doi: 10.7326/0003-4819-138-3-200302040-00009. [DOI] [PubMed] [Google Scholar]
- 20.Nietert PJ, Silverstein MD, Mokhashi MS, et al. Cost-effectiveness of screening a population with chronic gastroesophageal reflux. Gastrointest Endosc. 2003;57:311–318. doi: 10.1067/mge.2003.101. [DOI] [PubMed] [Google Scholar]
- 21.Rubenstein JH, Inadomi JM, Brill JV, et al. Cost utility of screening for Barrett’s esophagus with esophageal capsule endoscopy versus conventional upper endoscopy. Clin Gastroenterol Hepatol. 2007;5:312–8. doi: 10.1016/j.cgh.2006.12.008. [DOI] [PubMed] [Google Scholar]
- 22.Gerson LB, Groeneveld PW, Triadafilopoulos G. Cost-effectiveness model of endoscopic screening and surveillance in patients with gastroesophageal reflux disease. Clin Gastroenterol Hepatol. 2004;2:868–879. doi: 10.1016/s1542-3565(04)00394-5. [DOI] [PubMed] [Google Scholar]
- 23.Gerson L, Lin OS. Cost-benefit analysis of capsule endoscopy compared with standard upper endoscopy for the detection of Barrett’s esophagus. Clin Gastroenterol Hepatol. 2007;5:319–325. doi: 10.1016/j.cgh.2006.12.022. [DOI] [PubMed] [Google Scholar]
- 24.Barbiere J, Lyratzopoulos G. Cost-effectiveness of endoscopic screening followed by surveillance for Barrett’s Esophagus. A review. Gastroenterology. doi: 10.1053/j.gastro.2009.10.011. (in press) [DOI] [PubMed] [Google Scholar]
- 25.Sharma P, Dent J, Armstrong D, et al. The development and validation of an endoscopic grading system for Barrett’s esophagus: The Prague C & M criteria. Gastroenterology. 2006;131:1329–9. doi: 10.1053/j.gastro.2006.08.032. [DOI] [PubMed] [Google Scholar]
- 26.Amano Y, Ishimura N, Furuta K, et al. Which landmark results in a more consistent diagnosis of Barrett’s esophagus, the gastric folds or palisade vessels? Gastrointest Endosc. 2006;64(2):206–211. doi: 10.1016/j.gie.2006.04.029. [DOI] [PubMed] [Google Scholar]
- 27.Wilkins T, Gillies RA. Office-based unsedated ultrathin esophagoscopy in a primary care setting. Ann Fam Med. 2005;3:126–30. doi: 10.1370/afm.262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Madhotra R, Mokhashi M, Willner I, et al. Prospective evaluation of a 3.1-mm battery-powered esophagoscope in screening for esophageal varices in cirrhotic patient. Am J Gastroenterol. 2003;98:807–12. doi: 10.1111/j.1572-0241.2003.07374.x. [DOI] [PubMed] [Google Scholar]
- 29.Thota PN, Zuccaro G, Jr, Vargo JJ, 2nd, et al. A randomized prospective trial comparing unsedated esophagoscopy via transnasal and transoral routes using a 4-mm video endoscope with conventional endoscopy with sedation. Endoscopy. 2005;37:559–65. doi: 10.1055/s-2005-861476. [DOI] [PubMed] [Google Scholar]
- 30.Faulx AL, Catanzaro A, Zyzanski S, et al. Patient tolerance and acceptance of unsedated ultrathin esophagoscopy. Gastrointest Endosc. 2002;55:620–3. doi: 10.1067/mge.2002.123274. [DOI] [PubMed] [Google Scholar]
- 31.Wildi SM, Wallace MB, Glenn TF, et al. Accuracy of esophagoscopy performed by a non-physician endoscopist with a 4-mm diameter battery-powered endoscope. Gastrointest Endosc. 2003;57:305–10. doi: 10.1067/mge.2003.111. [DOI] [PubMed] [Google Scholar]
- 32.Saeian K, Staff DM, Vasilopoulos S, et al. Unsedated transnasal endoscopy accurately detects Barrett’s metaplasia and dysplasia. Gastrointest Endosc. 2002;56:472–8. doi: 10.1067/mge.2002.128131. [DOI] [PubMed] [Google Scholar]
- 33.Jobe BA, Hunter JG, Chang EY, et al. Office-based unsedated small-caliber endoscopy is equivalent to conventional sedated endoscopy in screening or surveillance for Barrett’s esophagus: A randomized and blinded comparison. Am J Gastroenterol. 2006;107:2160–6. doi: 10.1111/j.1572-0241.2006.00890.x. [DOI] [PubMed] [Google Scholar]
- 34.Atkinson M, Das A, Faulx A, et al. Ultrathin esophagoscopy in screening for Barrett’s esophagus at a Veterans Administration hospital: Easy access does not lead to referrals. Am J Gastroenterol. 2008;103:92–97. doi: 10.1111/j.1572-0241.2007.01501.x. [DOI] [PubMed] [Google Scholar]
- 35.Delvaux M, Gay G. Capsule endoscopy: Technique and indications. Best Practice & Research Clinical Gastroenterology. 2008;22:813–837. doi: 10.1016/j.bpg.2008.06.003. [DOI] [PubMed] [Google Scholar]
- 36.Gralnek IM, Rabinovitz R, Afik D, et al. A simplified ingestion procedure for esophageal capsule endoscopy: initial evaluation in healthy volunteers. Endoscopy. 2006;38:913–918. doi: 10.1055/s-2006-944718. [DOI] [PubMed] [Google Scholar]
- 37.De Jonge PJF, Van Eijck BC, Geldof H, et al. Capsule endoscopy for the detection of oesophageal mucosal disorders: a comparison of two different ingestion protocols. Scand Journal of Gastroenterol. 2008;43:870–877. doi: 10.1080/00365520801908878. [DOI] [PubMed] [Google Scholar]
- 38.Ramirez FC, Shaukat MS, Young MA, et al. Feasibility and safety of string, wireless capsule endoscopy in the diagnosis of Barrett’s esophagus. Gastrointest Endosc. 2005;61:741–6. doi: 10.1016/s0016-5107(05)00322-6. [DOI] [PubMed] [Google Scholar]
- 39.Ramirez FC, Akins R, Shaukat M. Screening of Barrett’s esophagus with string-capsule endoscopy: a prospective blinded study of 100 consecutive patients using histology as the criterion standard. Gastrointest Endosc. 2008;68:25–31. doi: 10.1016/j.gie.2007.10.040. [DOI] [PubMed] [Google Scholar]
- 40.Lin OS, Schembre DB, Mergener K, et al. Blinded comparison of esophageal capsule endoscopy versus conventional endoscopy for a diagnosis of Barrett’s esophagus in patients with chronic gastroesophageal reflux. Gastrointest Endosc. 2007;65:577–83. doi: 10.1016/j.gie.2006.06.035. [DOI] [PubMed] [Google Scholar]
- 41.Delvaux M, Papanikolaou IS, Fassler I, et al. Esophageal capsule endoscopy in patients with suspected esophageal disease: double blinded comparison with esophagogastroduodenoscopy and assessment of interobserver variability. Endoscopy. 2008;40:16–22. doi: 10.1055/s-2007-966935. [DOI] [PubMed] [Google Scholar]
- 42.Galmiche JP, Sacher-Huvelin S, Coron E, et al. Screening for esophagitis and Barrett’s esophagus with wireless esophageal capsule endoscopy: A multicenter prospective trial in patients with reflux symptoms. Am J Gastroenterol. 2008;103:538–545. doi: 10.1111/j.1572-0241.2007.01731.x. [DOI] [PubMed] [Google Scholar]
- 43.Hur C. Esophageal capsule endoscopy for Barrett’s esophagus screening: a hard pill to swallow? Clin Gastroenterol Hepatol. 2007;5:307–309. doi: 10.1016/j.cgh.2006.11.010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.




