Abstract
Diabetes leads to elevated levels of glucose in blood which, in turn, can lead to the non-enzymatic glycation of serum proteins such as human serum albumin (HSA). It has been suggested that this increase in glycation can alter the ability of HSA to bind to drugs and other small solutes. This study used high-performance affinity chromatography (HPAC) to see if there is any significant change related to glycation in the binding of HSA to warfarin and L-tryptophan, which are often used as probe compounds for Sudlow sites I and II of HSA in drug binding studies with this protein. It was found through frontal analysis studies that both of these compounds gave a good fit to a single-site binding model with glycated HSA under the conditions used in this study. There were was no significant change in the association equilibrium constants or specific activities for warfarin with HSA at pH 7.4 and 37°C under glycation conditions that were representative of those expected in pre-diabetes or diabetes, but a 4.7–5.8 fold increase binding affinity for L-tryptophan with glycated HSA was observed. These results indicate that warfarin and L-tryptophan can be successively used as site-selective probes for glycated HSA; however, changes in the affinity of L-tryptophan may need to be considered in such an application. These results should be valuable in future competition studies using these compounds as probes to examine the interactions of other drugs and solutes with Sudlow sites I and II and to determine how changes in HSA glycation can affect the serum protein-binding of various pharmaceutical agents during diabetes.
Keywords: Human serum albumin, Glycation, Drug-protein binding, Warfarin, L-Tryptophan
1. Introduction
Diabetes is a growing problem in the United States. In 2007, over 23 million people within the U.S. (7.8% of the population) were reported to have this disease, with over 1 million people being diagnosed every year [1]. The most common form of this disease is type 2 (non-insulin dependent) diabetes, in which the body suffers from a shortage of insulin. Diabetes is characterized by an elevated level of glucose in the blood. This glucose can form covalent adducts with proteins in blood through a non-enzymatic process known as glycation [2–4]. For example, glycated hemoglobin is now commonly used by physicians to monitor the long-term control of diabetes by a patient [4]. However, many other blood proteins also become glycated including human serum albumin (HSA). Because HSA has a shorter half-life than hemoglobin in blood, monitoring the extent of HSA glycation has been considered as a way to look at short-term diabetes management [4].
HSA is the most prominent protein in human plasma. This protein is synthesized in the liver and contains 585 amino acids with a total molar mass of 66,438 Da [5–7]. Two-thirds of HSA is made up of α-helix structures while 10% of the protein contains β-turns. This protein has three homologous domains (I, II, and III) that each contain two subunits (A and B). Approximately 6–13% of this protein is glycated in normal individuals [2–5], but this level can increase to over 20–30% in individuals with diabetes [3–5]. Glycation occurs by a condensation reaction of glucose with a lysine residue or the N-terminus of HSA to first form a Schiff base [3,6–8]. Rearrangements of this product create a ketoamine (i.e., an Amadori product) [3,6,8]. Further rearrangements of this adduct can create advanced glycation end-products (AGEs). Increased levels of these adducts and products can lead to severe health complications in diabetic individuals [3].
HSA is a major transport protein in blood for carrying various endogenous and exogenous compounds throughout the body. It greatly influences drug distribution and can play a major role in affecting drug absorption, distribution, metabolism, and excretion [2,9–11]. Binding to HSA also allows hydrophobic drugs to be more soluble in blood and increases the overall lifetime of a drug before it is metabolized [2,11]. It has previously been determined that HSA has two major binding sites for drugs (i.e., Sudlow sites I and II, located in subunits IIA and IIIB of HSA), as well as additional minor binding sites [12–14]. Sudlow site I binds anticoagulant drugs such as warfarin and anti-inflammatory drugs such as azapropazone, phenylbutazone, and salicylate. Sudlow site II binds drugs such as ibuprofen, fenoprofen, ketoprofen, and benzodiazepines, along with the essential amino acid L-tryptophan [9].
The purpose of this study was to see if the binding of HSA to warfarin and L-tryptophan (see structures in Figure 1) is altered as the level of HSA glycation is increased, as occurs in diabetes. Warfarin and L-tryptophan were of interest to this work because they are often used as site-selective probes for Sudlow sites I and II, respectively, in examining the binding of other drugs to HSA at these sites [15–17]. Glycation has noted in previous work to occur at locations that are near both Sudlow sites I and II [2,3,8,16,17]. It has been suggested in earlier studies based on fluorescence spectroscopy or equilibrium dialysis that the binding of HSA to some drugs and other solutes [5,18,19], including both warfarin [4,5] and L-tryptophan [18], can be affected by modifications resulting from glycation. However, past studies examining the binding of warfarin with glycated HSA at 20–25°C have resulted in apparently conflicting results, in which some authors have reported an increase in binding strength while others reported a decrease [4,5]. Studies with L-tryptophan and glycated HSA at 4°C found cases in which there was decreased and increased binding as the levels of glycation were increased [18]. It is apparent from these results that there is still a need for a better understanding of how glycation affects the binding of both warfarin and L-tryptophan with glycated HSA. Because these previous studies were conducted at 4–25°C and using non-physiological levels of HSA or glucose [4,5,19], there is also a need for further binding studies under the temperature and concentration conditions that are more typical of those expected in blood during diabetes.
Figure 1.
Structures of warfarin and L-tryptophan.
The goal of this current study was to obtain binding information on these probe compounds by using high-performance affinity chromatography (HPAC). HPAC is a chromatographic technique that can make use of an immobilized protein (e.g., HSA) to provide a precise, reproducible, and convenient means of examining drug-protein binding, while also being capable of automation [20]. This approach has been utilized successfully in the past to obtain drug-protein binding data on interactions of both warfarin and L-tryptophan with normal HSA [21–24]. This current report employed HPAC to also examine the interactions of these solutes with in vitro glycated HSA. This was accomplished by preparing HPAC columns that contained HSA with various known levels of glycation. The binding parameters that were found with warfarin and L-tryptophan on these columns were then compared to those noted with normal HSA. Knowledge of how these interactions are affected by glycation should be valuable in future work that uses warfarin or L-tryptophan as probes to examine the binding of other drugs of solutes with glycated HSA. This information, in turn, could be important in the future in obtaining a better picture of how such compounds bind to serum proteins such as HSA during diabetes and in how these interactions affect properties such as the metabolism, excretion and distribution of these drugs in this disease state.
2. Materials and Methods
2.1 Materials
Racemic warfarin (98% pure), L-tryptophan (98%), monobasic and dibasic potassium phosphate salts, D-(+)-glucose (99.5%), sodium azide (>95%), acetohexamide, sodium chloride, sodium nitrate, sodium phosphate salts, HSA (essentially fatty acid free albumin from human serum, ≥ 96%), and in vitro glycated HSA (Lot 058K6087, referred to here as “gHSA1”) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Reagents used in the bicinchoninic acid (BCA) protein assay were from Pierce (Rockford, IL, USA). Nucleosil Si-300 silica (300 Å pore size, 7 micron particle diameter) was obtained from Macherey-Nagel (Düren, Germany). The enzymatic assay kits for fructosamine were from Diazyme Laboratories (San Diego, CA, USA). Sterilized 17 × 100 mm culture tubes were purchased from Fisher Scientific (Pittsburg, PA, USA). Slide-A-Lyzer 7K dialysis cassettes (7 kDa MW cutoff; 0.5–3, 3–12, and 12–30 ml sample volume) were acquired from Thermo Scientific (Rockford, IL, USA). Econo-Pac 10 DG disposable chromatography desalting columns were purchased from Bio-Rad Laboratories (Hercules, CA, USA). Solutions were made using water from a Nanopure system (Barnstead, Dubuque, IA, USA) and filtered with a 0.20 μm GNWP nylon membrane from Millipore (Billerica, MA, USA).
2.2 Apparatus
The chromatographic system consisted of a Jasco DG-2080-53 three-solvent degasser (Tokyo, Japan), two Jasco PU-2080 isocratic HPLC pumps, a Rheodyne Advantage PF six-port valve (Cotati, CA, USA), a Jasco AS-2055 autosampler, a Jasco CO-2060 column oven (i.e., to maintain a column temperature of 37 °C), and a Jasco UV-2075 UV/Vis detector. This chromatographic system hardware was controlled through EZChrom Elite software v3.2.1 (Agilent, CA, USA) and a Jasco LC Net component. The chromatographic data were analyzed using in-house programs written using Labview 5.1 software (National Instruments, Austin, TX, USA) or using Peakfit 4.12 (Jandel Scientific Software, San Rafael, CA, USA). Linear regression was performed using Excel 2003 (Microsoft Corporation, Redmond, WA, USA) and non-linear regression was performed using DataFit (Oakdale Engineering, PA, USA).
2.3 Methods
2.3.1. Preparation of Glycated HSA
Studies were performed using three different glycated HSA samples. The first of which was purchased from a commercial source (Sigma-Aldrich), and the other two glycated HSA samples were made in vitro using a modified version of previously published methods [23,24]. To perform in vitro glycation of the HSA samples, all glassware and spatulas that would come in contact with the glycated HSA solution were previously sterilized by autoclaving to prevent bacterial growth during the preparation of the glycated HSA. One liter of pH 7.4, 0.2 M potassium phosphate buffer was prepared and also sterilized by autoclaving to minimize bacterial growth during this process. Sodium azide (i.e., a strong antibacterial agent) was added to this buffer once it had cooled to make a 1 mM solution of this agent. A 15 mM or 30 mM glucose solution (used to make gHSA2 and gHSA3, respectively, as described later in this report), was prepared using the sterilized sodium azide solution, with 20 ml of this solution then being utilized to dissolve 840 mg of HSA to provide a working solution containing 42 g/L, or 0.63 mM, HSA. This concentration of HSA was within the typical range (35–50 g/L, or 0.53–0.75 mM) seen in humans under normal physiological conditions [2,21,22].
This HSA solution was placed into a series of sterile culture tubes as 3–4 ml fractions, covered with the culture tube cap, further sealed with parafilm, and incubated in a water bath at 37 °C for four weeks (i.e., a time interval equal to approximately two half-lives for HSA in the circulation) [23]. After this incubation period, each glycated HSA solution was passed through a size-exclusion desalting column to separate the protein from any excess glucose and sodium azide in the solution. The collected protein fraction was dialyzed against water to remove any remaining glucose and buffer salts. The water used during dialysis was 200–500 times the volume of the sample. The sample was placed into a sterile dialysis cassette and allowed to dialyze for 2 h at room temperature, with gentle stirring using a magnetic stir bar, followed by a second dialysis cycle against water for another 2 h under the same conditions. The sample was then dialyzed against water a third time, without stirring, at 4 °C for approximately 14–18 h. The final samples were stored at −80 °C. Lyophilization was performed until samples were completely dry. Samples were again stored at −80 °C until use.
2.3.2. Fructosamine Assay
A commercial fructosamine assay from Diazyme was modified for use in determining the overall level of glycation of each batch of HSA that was used in this study. A 0.85% (w/v) saline solution was first prepared by dissolving 0.85 g sodium chloride in 100 ml of water. A pH 7.4, 0.025 M sodium phosphate buffer was also prepared. Although normal HSA concentrations in serum are around ~40 g/L [2], an HSA sample solution of ~20 g/L was prepared to stay within the linear range of the assay (i.e., 30–1354 μmol/L or 2–90 g/L). Each sample of glycated HSA for this assay was prepared by weighing out roughly 10 mg of this protein and adding 500 μL of pH 7.4, 0.025 M sodium phosphate buffer, followed by the solution then being placed on a vortex mixer for a short period of time. A temperature-controlled UV/vis spectrometer was used to perform the assay with the temperature held at 37 °C for the entire duration. A 300 μL portion of Reagent 1 from the kit was added to an empty cuvette. A 30 μL portion of the sample was then added, the resulting solution was mixed quickly, and placed into the spectrometer, with absorbance measurements being taking after 5 min at 600 and 700 nm. A 75 μL portion of Reagent 2 from the kit was then added to the cuvette and mixed quickly with the other contents, with this mixture then being allowed to react for 5 min before measurements were again taken at 600 and 700 nm. Each sample was run in duplicate or triplicate. A 0.85% (w/v) saline solution was used as a blank to subtract out the background while calibration standards and a control were measured during this assay; this procedure was repeated several times using these solutions in place of a 30 μL sample. The difference in the absorbance readings at 600 nm and 700 nm was calculated at each time, and the difference in the resulting values for the first and second 5 min readings was then also found. A calibration curve was made per the manufacturer’s instructions using a calibration standard included with the kit. This gave a plot that was used to find the μmol of fructosamine (i.e., a value equivalent to μmol hexose) in each protein sample.
2.3.3. HPAC Column and Solution Preparation
Nucleosil Si-300 silica was used to prepare diol-bonded silica according to a previously published procedure [25]. Glycated HSA was immobilized to the resulting support by using the Schiff base method, as previously discussed for normal HSA; and a control support was made in the same manner but with no glycated HSA being added during the immobilization step [17]. It should be noted here that although both the Schiff base immobilization and glycation involve free amine residues, previous studies have determined that these process mainly involve different residues on HSA, with the Schiff base method predominantly using the N-terminus or a few lysines that are not present at Sudlow sites I or II [26,27]. As a result, the prior glycation of HSA should not have affected the ability of this protein to be immobilized by the Schiff base method or the relative activities noted at Sudlow sites I and II for this immobilized protein, as was confirmed later in this study (see Section 3). A BCA assay was performed in triplicate to determining the content of each support, using soluble glycated HSA as the standard and the control support as the blank. The protein content of gHSA 1, 2, and 3 samples were found by this method to be 29 (± 4), 47 (± 8), and 40 (± 3) mg HSA/g silica, respectively (see Table 1). Each glycated HSA silica sample was downward slurry-packed into separate 2.0 cm x 2.1 mm I.D. columns at 3500 psi (24.1 MPa) using pH 7.4, 0.067 M potassium phosphate buffer as the packing solution. A column with the same dimensions but packed with the control support was also prepared. All columns were stored at 4 °C in pH 7.4, 0.067 M potassium phosphate buffer and were used over the course of one year and for fewer than 500 sample applications or injections per column. HSA columns have been shown in previous studies to retain good stability for drug-protein binding studies under such conditions [17].
Table 1.
Properties of glycated HSA supportsa
| Type of support | |||
|---|---|---|---|
| gHSA1 | gHSA2 | gHSA3 | |
| Protein content (mg HSA/g silica) | 29 (± 4) | 47 (± 8) | 40 (± 3) |
| Glycation level (mol hexose/mol HSA) | 1.31 (± 0.05) | 2.34 (± 0.13) | 3.35 (± 0.14) |
The values shown in parentheses represent a range of ± 1 S.D.
The warfarin, L-tryptophan and all other sample solutions for the chromatographic studies were prepared in pH 7.4, 0.067 M potassium phosphate buffer. The same pH 7.4, 0.067 M potassium phosphate buffer was used as the application and elution in the chromatographic studies. All solutions were filtered through a 0.2 μm nylon filter and degassed under vacuum for at least 15 min prior to use. Solutions of L-tryptophan have been found to decrease in stability over 2–9 days (depending on storage conditions) [16], so for this work the L-tryptophan solutions were made fresh daily prior to use. Racemic warfarin has been shown to have a slow structural conversion over time from its minor cyclic hemiketal to its major cyclic hemiketal [28]. When stored at 5 °C, a 5% conversion occurs in 45–52 hours whereas 95% conversion occurs between 109–127 days. Warfarin solutions were stored at 4 °C and used within 10 days for these experiments. All chromatographic experiments described in this report were performed at 37.0 (±0.1) °C. The elution of R-warfarin and L-tryptophan was monitored at 308 nm and 280 nm, respectively, while sodium nitrate (i.e., a non-retained solute) was monitored at 205 nm.
2.2.4. Chromatographic Studies
The competition studies described later in this report were conducted by using zonal elution on the columns containing normal HSA or glycated HSA. These studies were conducted using varying concentrations of acetohexamide (0–20 μM) in the mobile phase while 20 μL of R-warfarin or L-tryptophan were injected on to the column at a concentration of 5 μM. These studies were performed for the HSA or gHSA columns and corresponding control columns. Sodium nitrate was used to determine the void time of the columns and the system by injecting 20 μL of 20 μM sodium nitrate onto each column and injecting it onto the system with a zero-volume union. A pH 7.4, 0.067 M potassium phosphate buffer was used to make all solutions. PeakFit v.4.12 and an exponentially-modified Gaussian curve fit were used to determine the central moments of the eluting peaks.
All frontal analysis experiments were carried out at 0.5 ml/min. This flow rate has been shown in similar studies to obtain reproducible binding capacities with the given analytes and on columns containing normal HSA [29,30]. The concentrations for the applied analyte solutions ranged from 1 and 10 μM. A six-port injection valve was used to switch between the mobile phases (i.e., sample application and elution/column regeneration). The column was first equilibrated using pH 7.4, 0.067 M potassium phosphate buffer. When performing the frontal analysis experiments, the six-port valve was programmed to switch from this buffer to the given analyte solution after one minute from time zero to ensure an initial steady baseline for data analysis. Once the analyte had fully saturated the column and a breakthrough curve had been obtained, the six-port valve was switched back to the original pH 7.4 phosphate buffer to wash the retained analyte from the column and re-equilibrate the column before the next run. All experiments were performed at each analyte concentration on both the glycated HSA columns and the control column. The resulting breakthrough curves were analyzed using Labview 5.1 or Peakfit v.4.12 and were corrected for non-specific binding by the analytes to the support by subtracting control column data from data for the glycated HSA columns.
3. Results and Discussion
3.1 Preparation and Initial Studies of Glycated HSA
One issued noted with previous studies looking at warfarin or L-tryptophan interactions with glycated HSA is that the conditions employed in such work differed significantly in one or more ways from those expected within the body. For instance, these earlier reports [4,5,19] examined binding at 4°C or 20–25°C rather than at a physiological temperature of 37°C, as used in this current report. In addition, many of these earlier reports used a much smaller concentration of HSA than is present in the circulation (e.g., 0.075 or 0.3 mM HSA in Refs. 4 and 5 versus a typical serum level of 0.6–0.63 mM, as used in this current study and Ref. 18). These same studies also tended to use a much larger concentration and excess of glucose versus HSA than would be expected in diabetes (e.g., a 165- to 3330-fold mol excess of glucose versus HSA in Refs. 4 and 5 vs. up to the 48-fold excess in this study and Ref. 18).
Three samples of in vitro glycated HSA were used in this study. The first preparation (referred to here as gHSA1, and obtained from a commercial source) had a relatively low level of glycation of 1.31 (± 0.05) mol hexose/mol HSA and was used to represent minimally glycated HSA, as might be found during pre-diabetes or early state diabetes. The second sample, gHSA2, was made using glucose conditions typical of those seen in diabetes and had a glycation level of 2.34 (± 0.13) mol hexose/mol HSA [31]. The third sample, referred to here as gHSA3, contained 3.35 (± 0.14) mol hexose/mol HSA and was used to represent a case of uncontrolled, advanced diabetes. Protein assays were also conducted on each of these preparations after they had been immobilized by the same method to HPLC grade silica, giving final protein contents in the range of 29 to 47 mg protein/g silica, or roughly 440 to 710 nmol glycated HSA/g silica. These latter results were in the same range as seen for non-glycated HSA when using the same immobilization method and type of support material [17,28].
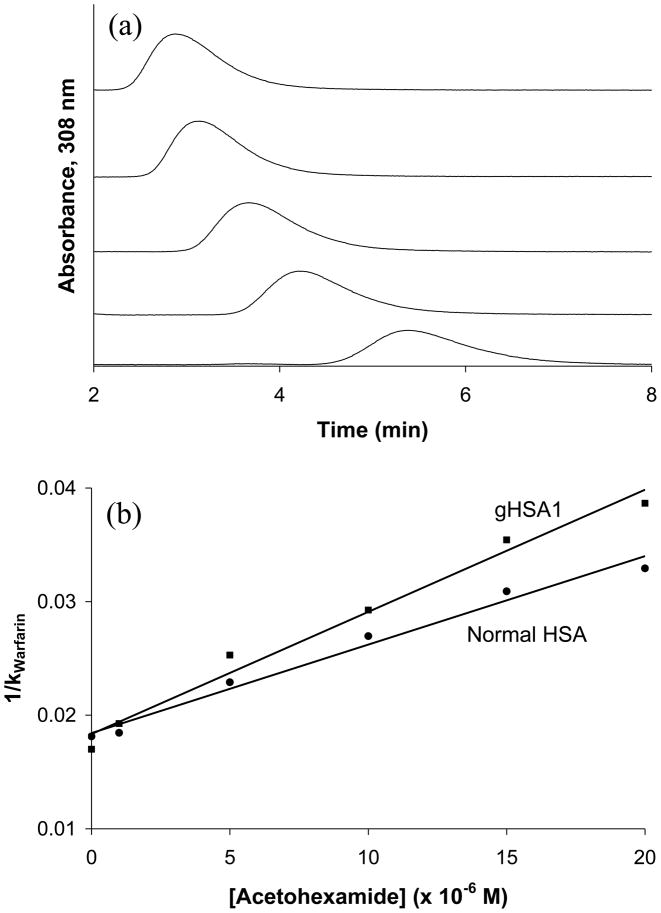
Preliminary competition studies were first conducted with these supports using the method of zonal elution. In these studies, similar retention factors (when corrected for differences in column protein content) were noted for R-warfarin and L-tryptophan in the absence of any competing agents. An example of such a study is shown in Figure 2 for columns containing HSA or glycated HSA. In this type of study, the retention of these site-selective probe compounds was examined while varying the concentration of a mobile phase additive and possible competing agent (i.e., acetohexamide, in this case). In this type of experiment, Eqn. (1) describes the predicted change in retention of the injected analyte (A) if it has competition at a single type of site on an immobilized ligand with the competing agent (I).
Figure 2.
(a) Competition studies for the injection of R-warfarin onto glycated HSA 1 column with acetohexamide solutions in the mobile phase. Acetohexamide concentrations from left to right: 20, 15, 10, 5, and 1 μM. The R-warfarin samples had a concentration of 5 μM and the injection volume was 20 μL. (b) Plots of 1/k vs. [Acetohexamide] while injecting R-warfarin on to a normal HSA column (●) and glycated HSA column 1 (■). The best-fit lines were as follows: normal HSA, y = 779 (± 51) x + 0.0184 (± 0.0006) (r = 0.991); glycated HSA, y = 1075 (± 72) x + 0.0183 (± 0.0008) (r = 0.991).
| (1) |
In this equation, the retention factor (k) is determined by using the retention time of the analyte (tR) and the void time of the system (tM), where k = (tR – tM)/tM. The terms KaA and KaI are the association equilibrium constants for the analyte and the competing agent, respectively, at their site of competition on the immobilized ligand and VM is the void volume. If direct competition is present between A and I at a single type of binding site, a plot of 1/k versus [I] should be linear, as occurs for the plots in Figure 2. In addition the value of k at the intercept will be directly related to the equilibrium constant and relative activity of the immobilized ligand for the injected analyte (i.e., at [I] = 0, 1/k = VM/{KaA mL} or k = KaA mL/VM).
The similarities of the retention factors noted when [I] = 0 in plots such as Figure 2 for the normal HSA and glycated HSA columns suggested that warfarin and L-tryptophan (data not shown) had relatively consistent binding to Sudlow sites I and II, respectively. This appeared to be the case even though the competition of these probes with drugs like acetohexamide did create a change in the response of these plots. Additional studies based on frontal analysis were next undertaken (see following sections) to further examine the interactions of warfarin and L-tryptophan with these columns prior to more in depth studies using these compounds as probes in competition studies with additional drugs on glycated HSA columns.
3.2 Binding of Warfarin to Glycated HSA
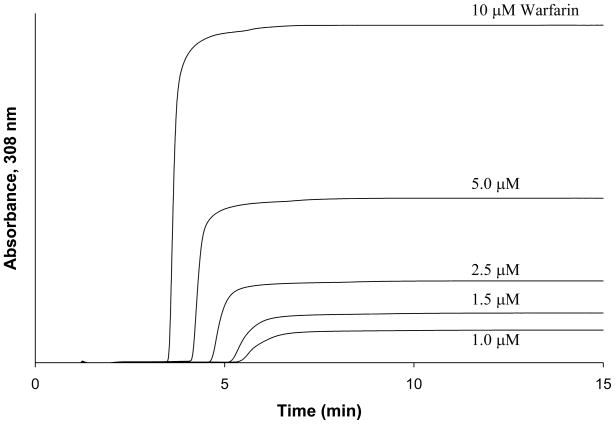
Frontal analysis studies were performed to obtain a more detailed analysis of the binding parameters for glycated HSA with warfarin. This method is performed by continuously applying a solution with a known amount of analyte to a column with an immobilized ligand. As the analyte binds to the ligand, the column becomes saturated and a characteristic breakthrough curve is formed (Figure 3). A breakthrough time can be calculated when the area under the first half of the curve equals the area above the second half of the curve [20]. The middle point represents the breakthrough time. When fast association/dissociation kinetics are present, the breakthrough time can be related to the concentration of the analyte [A], the association equilibrium constant for the analyte-ligand system (Ka), and the moles of active binding sites on the column for the analyte (mL). The following equations show this relationship for a system where there is a single type of binding site on the ligand for the analyte (single-site model) [20],
Figure 3.
Breakthrough curves for warfarin at applied concentrations 10, 5, 2.5, 1.5, and 1 μM (from left to right) on glycated HSA column 1. Conditions for these studies are given in the text.
| (2) |
| (3) |
in which mLapp is the apparent moles of analyte that are required to reach the mean position of the breakthrough curve at any given concentration of applied analyte and [A] is the molar concentration of the applied analyte. Eqn. (3) would be expected to produce a linear relationship for a 1:1 binding model when a plot is made of 1/mLapp versus 1/[A], while a non-linear fit and a plot of mLapp versus [A] would be used when applying Eqn. (2) to the same data. Either type of fit make it possible to determine Ka and mL simultaneously for the interaction, which can be used to provide information on how a change in the ligand might affect its affinity or relative activity for the applied analyte [32]. In this current study, this approach was used to monitor any changes that occurred in binding as the degree of glycation was varied for HSA.
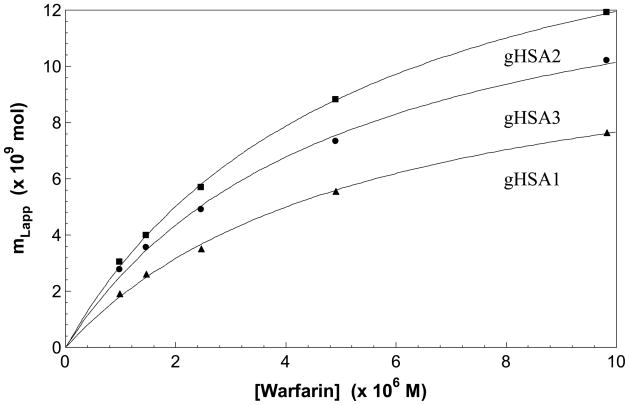
Some typical frontal analysis results that were obtained for warfarin on the glycated HSA columns are shown in Figures 3 and 4. In each case, warfarin produced breakthrough curves that shifted to shorter breakthrough times as the concentration of the applied analyte was increased (see Figure 3). The data obtained from these breakthrough curves were then analyzed according to Eqn. (2). Some typical results are shown in Figure 4. It was found for each of the glycated HSA columns that there was good agreement with the linear behavior predicted by Eqn. (3) at the concentration range which was examined in this study. The correlation coefficients ranged from 0.997 to 0.998 (n = 5), with only random variations generally being noted in the data points about the best-fit line (Note: a small amount of curvature may have been present in the results for the gHSA3 column, but using a higher-order fit such as a two-site model did not give any further improvement in the quality of the overall fit to this set of results). Similar agreement between the data and a one-site binding model was obtained when fitting a plot of mLapp versus [A] according to Eqn. (2), as demonstrated in Figure 5. These results agreed with those of previous examining the binding of warfarin with glycated HSA at 20–25°C, which also noted a good fit to a single site binding model [4,5].
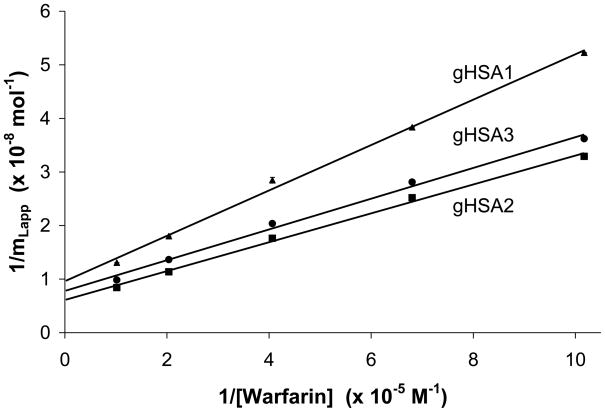
Figure 4.
Double reciprocal plots prepared according to Eqn. (3) for warfarin binding to columns prepared with samples of gHSA1 (▲), gHSA2 (■) or gHSA3 column (●). The solid line shows the best-fit line for each data set. The error bars represent a range of ± 1 S.D. The best-fit lines were as follows: gHSA1, y = 424 (± 15) x + [9.62 (± 0.87)] × 107 (r = 0.998); gHSA2, y = 270 (± 10) x + [6.10 (± 0.56)] × 107 (r = 0.998); gHSA3, y = 287 (± 13) x + [7.80 (± 0.77)] × 107 (r = 0.997).
Figure 5.
Plots of mLapp versus [Warfarin] prepared according to Eqn. (2) for the gHSA1 column (▲), gHSA2 column (■) and gHSA3 column (●). That data used in these plots were the same as used in Figure 4. The best-fit lines were as follows: gHSA1, y = [{1.81 (± 0.15)} × 105 × {1.19 (± 0.05) × 10−8 x]/[1 + {1.81 (± 0.15)} × 105 x] (r = 0.999); gHSA2, y = [{1.89 (± 0.09)} × 105 × {1.83 (± 0.05) × 10−8 x]/[1 + {1.89 (± 0.09)} × 105 x] (r = 0.999); gHSA3, y = [{2.01 (± 0.20)} × 105 × {1.52 (± 0.05) × 10−8 x]/[1 + {2.01 (± 0.20)} × 105 x] (r = 0.998).
Table 2 summarizes the association equilibrium constants that were obtained when the plotting the data according to Eqn. (3) and using a single-site model. These values ranged from 2.3 × 105 M−1 to 2.7 × 105 M−1 and had relative precisions of ± 9–10%. These results did not show any significant variations at the 95% confidence level between the various glycated HSA columns that were examined in this study, with all Ka values agreeing within 17–20% and within ± 2 S.D. of any other Ka value for warfarin in this same data set. This range of values also agreed within an average association equilibrium constant of 2.4 (± 0.4) × 105 M−1 that was determined based on previous results reported for the separate R- and S-enantiomers of warfarin on a similar column that contained normal HSA and using the same method of data analysis [29]. These results indicated glycation did not have any observable effect on the average Ka that was measured for R/S-warfarin on these columns as the level of glycation of HSA was increased up to the level present in the gHSA column (i.e., 3.35 mol hexose/mol HSA, as shown in Table 1).
Table 2.
Binding parameters for R-warfarin and L-tryptophan on various HSA columns
| Type of HSA column | ||||
|---|---|---|---|---|
| Normal HSA | gHSA1 | gHSA2 | gHSA3 | |
| Racemic Warfarin | ||||
| Ka (× 105 M−1) | 2.4 (± 0.4)b | 2.3 (± 0.2) | 2.3 (± 0.2) | 2.7 (± 0.3) |
| mL (× 10−8 mol) | ------- | 1.04 (± 0.09) | 1.64 (± 0.15) | 1.28 (± 0.13) |
| Specific Activity (mol/mol HSA) | ------- | 0.76 (± 0.12) | 0.74 (± 14) | 0.68 (± 0.09) |
| L-Tryptophan | ||||
| Ka (× 104 M−1) | 1.1 (± 0.3)c | 5.2 (± 0.9) | 6.4 (± 1.7) | 5.7 (± 0.5) |
| mL (× 10−9 mol) | ------- | 5.0 (± 0.9) | 6.7 (± 1.8) | 5.8 (± 0.6) |
| Specific Activity (mol/mol HSA) | ------- | 0.36 (± 0.08) | 0.30 (± 0.05) | 0.31 (± 0.04) |
It was also possible from the results in Figure 4 to determine the binding capacity of each affinity column for warfarin. This information was combined with the known protein content to obtain the specific activity for warfarin on each column and to compare these results to those expected for normal HSA. These results are also summarized in Table 2. When normalized for the protein content, it was found that comparable specific activities were present on each of the glycated HSA columns for warfarin. The relative precision of these specific activities was ± 12–19% and all of these specific activities were within 11% of all other values in this same data set. These values also ranged from 98% to 104% of the activity expected for columns containing normal HSA (e.g., as determined by using Eqn. (1) to analyze the results in Figure 2) and showed no significant variations at the 95% confidence level. It was concluded from the results in Table 2 that there was no significant change in either the association equilibrium constants or the relative activity of Sudlow site I for warfarin in going from normal HSA to glycated HSA at the levels of glycation that were examined in this study.
The lack of any apparent change in warfarin binding to glycated HSA at 37°C under the conditions used in this study falls between the conflicting conclusions reached in previous studies at 20–25°C, noting either up to a 1.8-fold increase in the strength of this binding or a 6.9-fold decrease [4,5]. However, a much larger excess of glucose than employed in this report was generally used to make the glycated HSA in these previous studies (i.e., up to a 165- to 3,300-fold mol excess of glucose compared to the 28- to 48-fold mol excess in this report). It is suggested when comparing the results of all these studies that glycation conditions that more closely mimic those occurring in blood will lead to fewer changes in warfarin binding than seen using much harsher glycation conditions. The conclusion is supported by the past use of circular dichroism to look for any major structural changes in HSA after glycation. It was found when using a 33-fold mol excess of glucose during glycation (i.e., a level similar to that used in this current report), no changes in secondary structure were seen for glycated HSA versus normal HSA. However, the use of a 670-fold mol excess of glucose during glycation did produce noticeable changes in the secondary structure of HSA, which were believed to be linked in the change in warfarin-HSA binding strength seen under such conditions [4,5].
3.3 Binding of L-Tryptophan to Glycated HSA
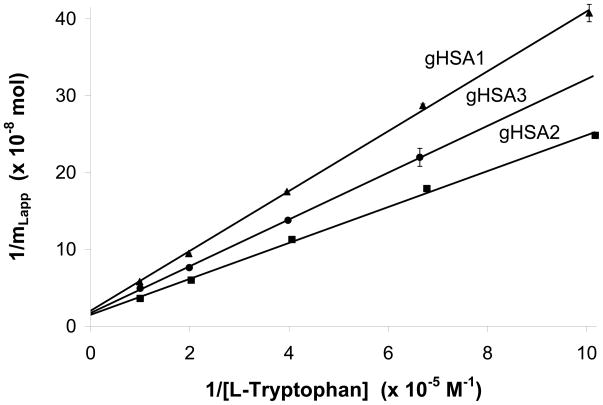
Similar frontal analysis studies to those in the previous section were conducted with L-tryptophan to examine the binding of this compound to Sudlow site II as glycation levels on HSA were increased. All of the tested columns gave results that gave a good fit to Eqn. (2) under the conditions used in this study (see Figure 6). The correlation coefficients for all of these plots was 0.999 (n = 5) and only random variations were noted in the data points about the best-fit line. These results indicated that the binding of L-tryptophan with the various samples of glycated HSA could be described effectively by a single-site when examined at 37°C. The same single site model has been shown in past studies to also give a good fit for the binding of L-tryptophan with glycated HSA at 4°C [18].
Figure 6.
Double reciprocal plots of L-tryptophan binding to affinity columns containing glycated HSA. These results are for the gHSA1 column (▲), gHSA2 column (■), and gHSA3 column (●). The solid line shows the best-fit line for each data set. The error bars represent a range of ± 1 S.D. The best-fit lines were as follows: gHSA1, y = [3.89 (± 0.06)] × 103 x + [2.02 (± 0.36)] × 108 (r = 0.999); gHSA2, y = [2.34 (± 0.07)] × 103 x + [1.49 (± 0.39)] × 108 (r = 0.999); gHSA3, y = [3.04 (± 0.04)] × 103 x + [1.72 (± 0.16)] × 108 (r = 0.999).
Table 2 summarizes association equilibrium constants and relative activities that were determined from the frontal analysis experiments for L-tryptophan with the various glycated HSA columns. The Ka values for this solute ranged from 5.2 × 104 M−1 to 6.4 × 104 M−1 with relative precisions for these values of ± 9 to 26% (Figure 6). These Ka values for the glycated HSA columns all overlapped within a range of 2 S.D. and were not significantly different at the 95% confidence level. However, these values were all 4.7–5.8 times higher than an association equilibrium constant of 1.1 × 104 M−1 that has been reported at 37°C for L-tryptophan on similar columns containing normal HSA [30,33,34]. These results indicated that even the levels seen in minimally-glycated HSA can affect the strength of L-tryptophan interactions with this protein. Possible changes in the binding strength of L-tryptophan with HSA versus glycated HSA have also been noted at 4°C under comparable glycation conditions, although at this latter temperature up to a 0.6-fold decrease in affinity may have been observed [18].
The measured binding capacities and the protein contents were used to also examine the specific activity of L-tryptophan on each glycated HSA column. When the binding capacities had been normalized for the amount of protein on the column, the specific activities for L-tryptophan ranged between 30.5 and 36.5% on each column, with precisions of ± 2–22%. These values differed by a maximum of only 16% and all fell within 1 S.D. of the values of the other columns, showing no significant differences at the 95% confidence level. These specific activities were also within the range that has been noted previously for L-tryptophan on columns containing non-glycated HSA that were prepared by the same immobilization method and on comparable silica supports [30]. It was concluded from these data that the levels of glycation used in this report did not create any appreciable changes in the specific activity of HSA for L-tryptophan.
Conclusion
Uncontrolled diabetes increases blood sugar levels which in turn facilitate a boost in the level of glycation that blood proteins undergo. This increase in glycation can potentially alter the binding of drugs to the protein thereby changing the transport of the drug around the body. These studies examined the binding at 37°C of warfarin and L-tryptophan for the major drug binding sites on HSA (i.e., Sudlow sites I and II, respectively). For warfarin, a good fit to a single-site model was noted for all samples of glycated HSA that were examined. No significant changes in the association equilibrium constants or relatively binding activities were seen for warfarin when comparing references values for normal HSA with the results obtained for glycated HSA that was representative of conditions expected during pre-diabetes or diabetes. The binding of L-tryptophan with glycated HSA also showed good agreement with a single-site binding model. The association equilibrium constants for L-tryptophan with even minimally-glycated HSA appeared to increase versus reference values for normal HSA when working with even minimally-glycated HSA, however, the specific activity of the glycated HSA for L-tryptophan was comparable to what would be expected for normal HSA.
These findings indicate the warfarin and L-tryptophan can be used as probes for Sudlow sites I and II in work with in vitro glycated HSA, although the changes in binding strength for L-tryptophan should be considered in such an application. Similar studies are now in progress with in vivo glycated HSA; changes due to glycation in drug binding with other serum proteins is also of interest for future studies. The results of this current study should be valuable in future competition studies using warfarin and L-tryptophan as probes to examine the interactions of other drugs and solutes with Sudlow sites I and II on HSA. Such information, in turn, should make it possible to better describe and predict how changes in HSA glycation can affect the serum protein-binding and effective activity of various pharmaceutical agents during diabetes.
Acknowledgments
This research was supported by National Institutes of Health under grant R01 DK069629 and was conducted in facilities that were renovated under NIH grant RR015468-01.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.National diabetes fact sheet: General information and national estimates on diabetes in the United States, 2007. U.S. Centers for Disease Control; Atlanta, GA: 2008. [Google Scholar]
- 2.Colmenarejo G. In silico prediction of drug-binding strengths to human serum albumin. Med Res Rev. 2003;23:275–301. doi: 10.1002/med.10039. [DOI] [PubMed] [Google Scholar]
- 3.Mendez DL, Jensen RA, McElroy LA, Pena JM, Esquerra RM. The effect of non-enzymatic glycation on the unfolding of human serum albumin. Arch Biochem Biophys. 2005;444:92–99. doi: 10.1016/j.abb.2005.10.019. [DOI] [PubMed] [Google Scholar]
- 4.Nakajou K, Watanabe H, Kragh-Hansen U, Maruyama T, Otagiri M. The effect of glycation on the structure, function and biological fate of human serum albumin as revealed by recombinant mutants. Biochim Biophys Acta. 2003;1623:88–97. doi: 10.1016/j.bbagen.2003.08.001. [DOI] [PubMed] [Google Scholar]
- 5.Fitzpatrick G, Duggan PF. The effect of non-enzymatic glycation on ligand binding to human serum albumin. Biochem Soc Trans. 1987;15:267–268. [Google Scholar]
- 6.Garlick RL, Mazer JS. The principal site of nonenzymatic glycosylation of human serum albumin in vivo. J Biol Chem. 1983;258:6142–6146. [PubMed] [Google Scholar]
- 7.Marashi S-A, Safarian S, Moosavi-Movahedi AA. Why major nonenzymatic glycation sites of human serum albumin are preferred to other residues? Med Hypotheses. 2005;64:881. doi: 10.1016/j.mehy.2004.11.007. [DOI] [PubMed] [Google Scholar]
- 8.Lapolla A, Fedele D, Seraglia R, Traldi P. The role of mass spectrometry in the study of non-enzymatic protein glycation in diabetes: An update. Mass Spectrom Rev. 2006;25:775–797. doi: 10.1002/mas.20090. [DOI] [PubMed] [Google Scholar]
- 9.Ascoli GA, Domenici E, Bertucci C. Drug binding to human serum albumin: Abridged review of results obtained with high-performance liquid chromatography and circular dichroism. Chirality. 2006;18:667–679. doi: 10.1002/chir.20301. [DOI] [PubMed] [Google Scholar]
- 10.Dockal M, Carter DC, Ruker F. The three recombinant domains of human serum albumin. J Biol Chem. 1999;274:29303–29310. doi: 10.1074/jbc.274.41.29303. [DOI] [PubMed] [Google Scholar]
- 11.Herve F, Urien S, Albengres E, Duche JC, Tillement JP. Drug binding in plasma: A summary of recent trends in the study of drug and hormone binding. Clin Pharmacokinet. 1994;26:44–58. doi: 10.2165/00003088-199426010-00004. [DOI] [PubMed] [Google Scholar]
- 12.Sudlow G, Birkett DJ, Wade DN. Characterization of two specific drug binding sites on human serum albumin. Mol Pharmacol. 1975;11:824–832. [PubMed] [Google Scholar]
- 13.Sudlow G, Birkett DJ, Wade DN. Further characterization of specific drug binding sites on human serum albumin. Mol Pharmacol. 1976;12:1052–1061. [PubMed] [Google Scholar]
- 14.Sengupta A, Hage DS. Characterization of minor site probes for human serum albumin by high performance affinity chromatography. Anal Chem. 1999;71:3824–3827. doi: 10.1021/ac9903499. [DOI] [PubMed] [Google Scholar]
- 15.Bertucci C, Andrisano V, Gotti R, Cavrini V. Use of an immobilized human serum albumin HPLC column as a probe of drug–protein interactions: The reversible binding of valproate. J Chromatogr B. 2002;768:147–155. doi: 10.1016/s0378-4347(01)00494-7. [DOI] [PubMed] [Google Scholar]
- 16.Conrad ML, Moser AC, Hage DS. Evaluation of indole-based probes for high-throughput screening of drug binding to human serum albumin: Analysis by high-performance affinity chromatography. J Sep Sci. 2009;32:1145–1155. doi: 10.1002/jssc.200800567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Joseph KS, Moser AC, Basiaga S, Schiel JE, Hage DS. Evaluation of alternatives to warfarin as probes for Sudlow site I of human serum albumin: Characterization by high-performance affinity chromatography. J Chromatogr A. 2009;1216:3492–3500. doi: 10.1016/j.chroma.2008.09.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Barzegar A, Moosavi-Movahedi AA, Sattarahmady N, Hosseinpour-Faizi MA, Aminbakhsh M, Ahmad F, Saboury AA, Ganjali MR, Norouzi P. Spectroscopic studies of the effects of glycation of human serum albumin on L-trp binding. Protein Peptide Lett. 2007;14:13–18. doi: 10.2174/092986607779117191. [DOI] [PubMed] [Google Scholar]
- 19.Koyama H, Sugioka N, Uno A, Mori S, Nakajima K. Effects of glycosylation of hypoglycaemic drug binding to serum albumin. Biopharm Drug Dispos. 1997;18:791–801. doi: 10.1002/(sici)1099-081x(199712)18:9<791::aid-bdd66>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 20.Hage DS. High-performance affinity chromatography: A powerful tool for studying serum protein binding. J Chromatogr B. 2002;768:3–30. doi: 10.1016/s0378-4347(01)00482-0. [DOI] [PubMed] [Google Scholar]
- 21.Ascenzi P, Bocedi A, Notari S, Fanali G, Fesce R, Fasano M. Allosteric modulation of drug binding to human serum albumin. Mini-Rev Med Chem. 2006;6:483–489. doi: 10.2174/138955706776361448. [DOI] [PubMed] [Google Scholar]
- 22.Fasano M, Curry S, Terreno E, Galliano M, Fanali G, Narciso P, Notari S, Ascenzi P. The extraordinary ligand binding properties of human serum albumin. IUBMB Life. 2005;57:787–796. doi: 10.1080/15216540500404093. [DOI] [PubMed] [Google Scholar]
- 23.Lapolla A, Fedele D, Reitano R, Arico NC, Seraglia R, Traldi P, Marotta E, Tonani R. Enzymatic digestion and mass spectrometry in the study of advanced glycation end products/peptides. J Am Soc Mass Spectrom. 2004;15:496–509. doi: 10.1016/j.jasms.2003.11.014. [DOI] [PubMed] [Google Scholar]
- 24.Ney KA, Colley KJ, Pizzo SV. The standardization of the thiobarbituric acid assay for nonenzymatic glucosylation of human serum albumin. Anal Biochem. 1981;118:294–300. doi: 10.1016/0003-2697(81)90585-6. [DOI] [PubMed] [Google Scholar]
- 25.Ruhn PF, Garver S, Hage DS. Development of dihydrazide-activated silica supports for high-performance affinity chromatography. J Chromatogr A. 1994;669:9–19. doi: 10.1016/0021-9673(94)80332-3. [DOI] [PubMed] [Google Scholar]
- 26.Wa C, Cerny RL, Clarke WA, Hage DS. Characterization of glycation adducts on human serum albumin by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Clin Chim Acta. 2007;385:48–60. doi: 10.1016/j.cca.2007.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wa C, Cerny RL, Hage DS. Identification and quantitative studies of protein immobilization sites by stable isotope labeling and mass spectrometry. Anal Chem. 2006;78:7967–7977. doi: 10.1021/ac0609935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Moser AC, Kingsbury C, Hage DS. Stability of warfarin solutions for drug-protein binding measurements: Spectroscopic and chromatographic studies. J Pharm Biomed Anal. 2006;41:1101–1109. doi: 10.1016/j.jpba.2006.02.012. [DOI] [PubMed] [Google Scholar]
- 29.Loun B, Hage DS. Chiral separation mechanisms in protein-based HPLC columns. 1. Thermodynamic studies of (R)- and (S)-warfarin binding to immobilized human serum albumin. Anal Chem. 1994;66:3814–3822. doi: 10.1021/ac00093a043. [DOI] [PubMed] [Google Scholar]
- 30.Yang J, Hage DS. Characterization of the binding and chiral separation of D- and L-tryptophan on a high-performance immobilized human serum albumin column. J Chromatogr. 1993;645:241–250. doi: 10.1016/0021-9673(93)83383-4. [DOI] [PubMed] [Google Scholar]
- 31.Powers AC. In: Harrison’s principles of internal medicine. Kasper DL, Fauci AS, Longo DL, Braunwald E, Hauser SL, Jameson JL, editors. McGraw-Hill Companies, Inc; 2005. [Google Scholar]
- 32.Chattopadhyay A, Tian T, Kortum L, Hage DS. Development of tryptophan-modified human serum albumin columns for site-specific studies of drug-protein interactions by high-performance affinity chromatography. J Chromatogr B. 1998;715:183–190. doi: 10.1016/s0378-4347(98)00140-6. [DOI] [PubMed] [Google Scholar]
- 33.Talbert AM, Tranter GE, Holmes E, Francis PL. Determination of drug-plasma protein binding kinetics and equilibria by chromatographic profiling: Exemplification of the method using L-tryptophan and albumin. Anal Chem. 2002;74:446–452. doi: 10.1021/ac010643c. [DOI] [PubMed] [Google Scholar]
- 34.Lagercrantz C, Larsson T. Comparative studies of the binding of some ligands to human serum albumin non-covalently attached to immobilized Cibacron Blue, or covalently immobilized on Sepharose, by column affinity chromatography. Biochem J. 1983;213:387–390. doi: 10.1042/bj2130387. [DOI] [PMC free article] [PubMed] [Google Scholar]