Summary
Clinical signs and patient verbal responses have traditionally been used to assess patient comfort and the depth of sedation. Recently, level-of-consciousness monitors have been used to guide sedation. The SNAP IIc is a two-lead electroencephalogram device which displays a SNAPc Index - a value derived based on both high and low frequency electroencephalogram signals. Much of the current clinical research on monitoring during sedation involves the bispectral index monitor. We compared simultaneous readings recorded by the SNAP II and bispectral index during sedation in 51 consecutive patients undergoing surgery. The anaesthesia team was blinded to the SNAP II and bispectal index values. Concurrent SNAP II and bispectral index readings displayed similarly-shaped trajectories during sedation, but further studies are needed to establish the routine clinical utility of both these monitors.
Traditionally, patients’ clinical signs and verbal responses have been used to assess patient comfort and depth of sedation. When patients are non-responsive, objective physiological signs are easy to observe, but may be non-specific. When autonomic activity is altered due to medications or intrinsic disease, the usefulness of heart rate and blood pressure monitoring for sedation or anaesthesia may be reduced. When patients are sedated, tactile and verbal stimulation are often used to elicit a response from the patient to help assess psychological and physical comfort. This repetitive activity may become intrusive to patients, distracting to surgeons and tedious for anaesthesia providers during a procedure.
Several clinical semi-quantitative tools have been developed to assess the response to sedative medications. These include the Observer Assessment of Alertness/Sedation (OAAS) Scale, the Ramsay Scale, and the Richmond Agitation Scale. Clinical sedation tools have been used in a critical care setting with reasonable results [1]. However, as an adjunct, commercially available electronic monitors have been developed to assess patients’ level of consciousness (LOC) [2,3]. Examples of monitors include: standard electroencephalogram, Mid-latency Auditory Evoked Potentials (MLAEP), Bispectral Index (BIS©; Aspect Medical Systems, Norwood, MA, USA), SNAP II© (Stryker, Kalamazoo, MI, U.S.A.), Entropy© (GE Healthcare, Wauwatosa, WI USA), Narcotrend© (Monitortechnik GMBH, Bad Bramstedt, Germany) and Sedline© (Hospira, Lake Forest, IL, USA). There have been many studies comparing clinical observational tools with these devices [4–6]. The utility of these monitors in clinical practice is debated, with widely ranging claims of usefulness [5–12]. Many articles address the use of LOC monitors during general anaesthesia. However, there is also a growing interest in the utility of LOC monitors during light to moderate sedation [5–9,11–18], including sedation in intensive care [10, 19–20]. The BIS monitor has been most widely studied [15–17] and consequently most new LOC devices are often compared to the BIS. [15–18,22].
The SNAP II is a relatively newer, small, single lead electroencephalogram (EEG) device which displays a SNAP Index©-a derived value based on an algorithm utilizing spectral analysis of both ultra high (80–420Hz) and low frequency (0–18Hz) EEG signals [23]. Most other LOC monitors utilise primarily traditional lower frequency EEG signals [3, 23–26], although they may add higher frequency electomyogram (EMG) analysis for additional information. For example, the BIS index is derived from a two lead processed EEG signal via a proprietary algorithm that computes a numerical value by using fast Fourier transformation, power spectrum and bi-spectrum analysis. The BIS also displays an indication of EMG signal (70–110Hz) activity [3,24,27]. For patients undergoing sevoflurane-nitrous oxide anaesthesia, a correlation between the SNAP II index and the Bispectral (BIS) index has been shown by Ruiz-Gimono et al. [28. However the two monitors have typically been compared during general anesthesia in the operating theatre. The purpose of this study was to compare the BIS and SNAP II during sedation in the operating theatre.
Methods
This study protocol was approved by the University of Wisconsin Hospital and Clinics Ethics Committee and written informed consent was obtained. ASA class I, II and III patients undergoing outpatient surgery with sedation and local anaesthesia or sedation and regional anaesthesia were eligible. Exclusion criteria were: age < 18 y, ASA IV or V, general anaesthesia, prisoners, or women who were pregnant. Additionally, we excluded patients with a significant history of neurological deficit related to stroke, seizure or brain injury, or those on medications that depress neurological function. A convenience sample was taken, which consisted of 51 patients undergoing a day case surgical procedure with sedation in the outpatient center at the University of Wisconsin Hospital and Clinics. Surgical procedures included a wide range of procedures in upper and lower peripheral limb surgery. Medications used for sedation were unrestricted and included midazolam, fentanyl, ketamine, hydromorphone, and propofol. Local anesthetics were used, either as a field block, intravenous block, or as regional block
After cleaning the patient’s forehead with alcohol, two 3-gel electrode strips were placed on the forehead in accordance with the manufacturer’s guidelines. To standardise, the BIS electrodes were placed on the dominant hand side and the SNAP II opposite and slightly above that. The electrodes were then attached to their respective monitors: a SNAP II module (SNAP II version 1.0.9, Algorithm ver. 1.79 Everest Biomedical Instruments Company), and BIS XP module (Aspect System Revision 3.21, BIS Engine Revision 1.18). Automatic electrode impendence checking was performed by both devices and, to mimic real world situations, the automatic impedance checking was not disabled and was allowed to occur as set by the software for each device. Following this, a baseline reading from each machine was taken before giving any sedative/hypnotic or narcotic. The patient was then disconnected from the LOC monitors and transported to the operating room where the monitors where reattached following patient positioning. Monitoring was continuous and an independent observer recorded vital signs every 5 min, recording types and amounts medications given by the anaesthesia providers. The anaesthesia team was blinded to the values of both BIS and SNAP II. Monitoring was stopped and electrodes were removed when the patient appeared awake and alert at the conclusion of the anesthetic care. Data were recorded into Microsoft Office Excel 2003 ® (Microsoft Corporation One Microsoft Way, Redmond, WA 98052-7329 USA) spreadsheets.
Data were collected to represent minute-by-minute measurements of the SNAP II and BIS values during the entire surgical procedure. SNAP II and BIS measurements ranged from 0% to 100% in 1%-unit increments. The duration of surgery differed for the participants, and anaesthetic choice also differed.
Since the main purpose of our study was simply to ascertain if SNAP II and BIS produce shape-equivalent readings, we did not submit our data to complex statistical analysis. Shape-equivalence is defined simply as similarly-shaped SNAP II and BIS trajectories, or, in other words, the two monitoring systems track well together over time. This is best represented graphically.
Results
Due to equipment malfunction, interference or lack of contiguous data, the data from seven patients were deemed to be inadequate, hence all subsequent analyses are based on data obtained from the remaining 44 patients. Figure 1 shows the SNAP II and BIS trajectories of four participants. BIS values seem always lower than same-minute SNAP II, by an average of ~20 points. However, the overall shapes of the two trajectories appear to agree very closely.
Figure 1.
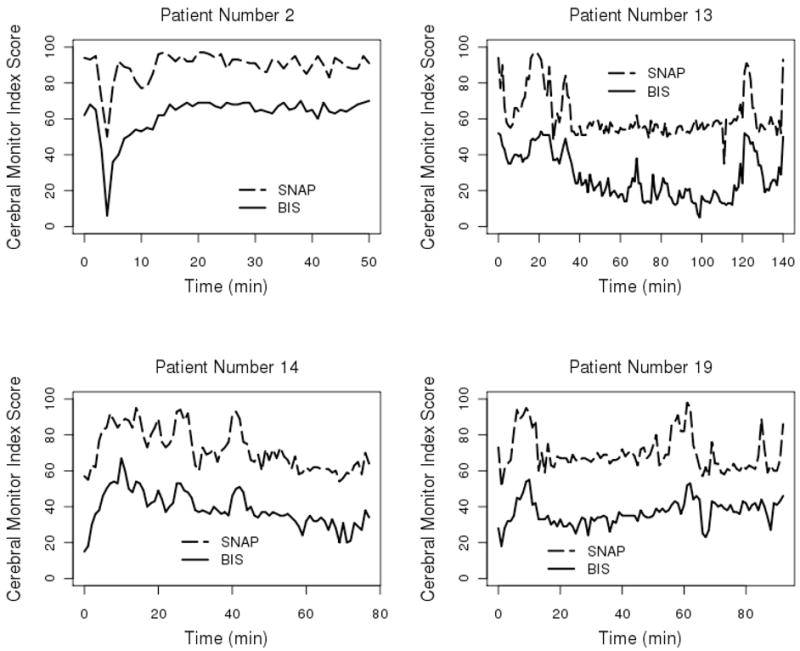
Four selected patients’ data. SNAP II© and BIS trajectories are shown in the lower part of each set. The vertical axis shows the SNAPc and BIS Index scores while the horizontal axis represents the surgical procedure duration in minutes from initiation.
Figure 2 shows individual minute-by-minute values for SNAP II and BIS for all study participants. For simplicity, participants have been grouped into three categories: short- (0–25 min), medium- (25–40 min) and extended- (>40 min) duration surgery. These differences oscillate, in random fashion, around a constant value, albeit slightly different on a patient-to-patient basis, with no discernable pattern.
Figure 2.
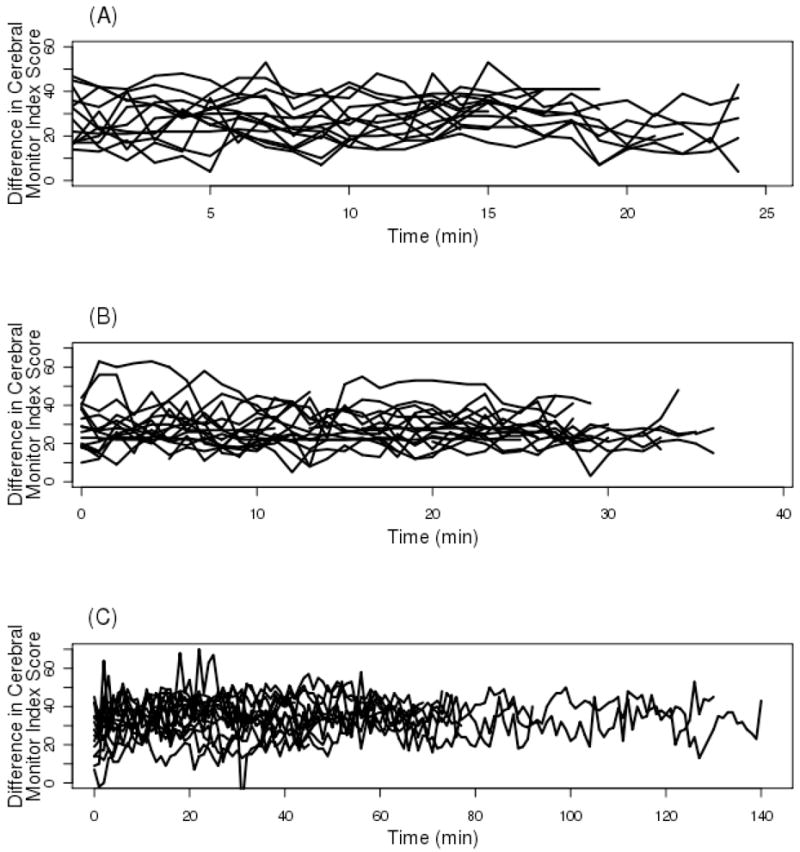
Individual minute-by-minute differences between SNAP II© and BIS cerebral monitor index scores for all study participants, grouped as follows: Panel A, short- (0–25 min); Panel B, medium- (25–40 min) and Panel C, extended- (≥ 40 min or more) duration surgery.
Discussion
While our study is a simple indication that the two monitors track sedation in a similar way, there are still questions that remain about the overall utility of LOC devices. Measures of improvement in quality of care could include case turn-over time, patient discharge time, patient satisfaction, amount of medication use, or incidence adverse cardio respiratory events when using LOC monitors.
A recent study questions the usefulness of processed EEG monitors for detecting awareness during general anaesthesia [29,30] but these monitors have also been used for tracking depth of sedation. Sedation can often be challenging due to patient variability in responses to medications. LOC monitors could be especially useful in patients who are difficult to assess clinically such as patients who cannot verbalise, or where it may be difficult to interpret clinical signs of discomfort, or where frequent interactions between anaesthetist and patient are disruptive to the procedure.
Our findings suggest that although SNAP II and BIS are not producing identical measurements, they are capturing essentially the same (albeit perhaps ‘shifted’ in time) cerebral activity pattern. The existence of this shift is not surprising and the most logical explanation for it resides in the differences between the mathematical algorithms upon which SNAP II and BIS measurement are based. Unfortunately, these algorithms are proprietary and not publicly available, hence it would be impossible to pinpoint the exact source leading to differential SNAP II and BIS measurements. A distinct difference between SNAP II and BIS values was found during general anesthesia by Ruiz-Gimeno et al. [28]. In the present study, we too have found that SNAP II Index values are greater than their BIS counterparts, even under moderate sedation.
Our data do not support the use of one monitor over the other. A future study might compare continuous monitor readings and standardised clinical observations, or with standardised anaesthesia and surgery. It has been reported that the ability of the BIS to determine the anaesthxetic depth or likelihood of movement to surgical stimulation may be affected by the use of non-volatile agent anesthetics, such as ketamine, or opioids [31]. The current study had multiple sedation agents and did not focus on comparing single medications, but on presenting an overall assessment. Further useful studies could allow the anaesthesia providers to view the device data to guide anesthetic management.
Acknowledgments
Acknowledgements and Conflicts of Interests
Adin-Cristian Andrei’s research is supported in part by following NIH grants: P30 CA014520-33 and UL1 RR025011. The authors have also received a grant from Everest Biomedical Instruments Co Inc., Fenton, MO, USA, providing support for the institutional research nurse and the SNAP II© module and electrodes. The company was not involved in the design of the study, the collection of the data, the analysis of the data or the writing of the manuscript and it has not reviewed the manuscript.
References
- 1.De Jonghe B, Cook D, et al. Using and understanding sedation scoring systems: a systematic review. Intensive Care Medicine. 2000;26:275–85. doi: 10.1007/s001340051150. [DOI] [PubMed] [Google Scholar]
- 2.Anderson RE, Jakobsson JG. Cerebral state monitor, a new small handheld EEG monitor for determining depth of anesthesia: a clinical comparison with the Bispectral index during day-surgery. European Journal of Anaesthesiology. 2006;23:208–12. doi: 10.1017/S0265021505002206. [DOI] [PubMed] [Google Scholar]
- 3.Jameson LC, Sloan TB. Using EEG to monitor anesthesia drug effects during surgery. Journal of Clinical Monitoring and Computing. 2006;20:445–72. doi: 10.1007/s10877-006-9044-x. [DOI] [PubMed] [Google Scholar]
- 4.Turkmen A, Altan A, Turgut N, Vatansever S, Gokkaya S. The correlation between the Richmond agitation-sedation scale and Bispectral index during dexmedetomidine sedation. European Journal of Anaesthesiology. 2006;23:300–4. doi: 10.1017/S0265021506000081. [DOI] [PubMed] [Google Scholar]
- 5.Chisholm CJ, Zurica J, Mironov D, Sciacca RR, Ornstein E, Heyer EJ. Comparison of electrophysiological monitors with clinical assessment of level of sedation. Mayo Clinic Proceedings. 2006;81:46–52. doi: 10.4065/81.1.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dahaba AA, Lischnig U, Kronthaler R, et al. Bispectral-index-guided versus clinically guided remifentanil/propofol analgesia/sedation for interventional radiological procedures: an observer-blinded randomized study. Anesthesia and Analgesia. 2006;103:378–84. doi: 10.1213/01.ane.0000223681.77120.ca. [DOI] [PubMed] [Google Scholar]
- 7.Bower AL, Ripepi A, Dilger J, Boparai N, Brody FJ, Ponsky JL. Bispectral index monitoring of sedation during endoscopy. Gastrointestinal Endoscopy. 2000;52:192–6. doi: 10.1067/mge.2000.107284. [DOI] [PubMed] [Google Scholar]
- 8.Bailey PL, Zuccaro G., Jr Sedation for endoscopic procedures: not as simple as it seems. American Journal of Gastroenterology. 2006;101:2008–10. doi: 10.1111/j.1572-0241.2006.00807.x. [DOI] [PubMed] [Google Scholar]
- 9.Drake LM, Chen SC, Rex DK. Efficacy of bispectral monitoring as an adjunct to nurse-administered propofol sedation for colonoscopy: a randomized controlled trial. American Journal of Gastroenterology. 2006;101:2003–7. doi: 10.1111/j.1572-0241.2006.00806.x. [DOI] [PubMed] [Google Scholar]
- 10.LeBlanc JM, Dasta JF, Kane-Gill SL. Role of the bispectral index in sedation monitoring in the ICU. Annals of Pharmacotherapy. 2006;40:490–500. doi: 10.1345/aph.1E491. [DOI] [PubMed] [Google Scholar]
- 11.Park KS, Hur EJ, Han KW, Kil HY, Han TH. Bispectral index does not correlate with observer assessment of alertness and sedation scores during 0.5% bupivicaine epidural anesthesia with nitrous oxide sedation. Anesthesia and Analgesia. 2006;103:385–9. doi: 10.1213/01.ane.0000226090.13170.25. [DOI] [PubMed] [Google Scholar]
- 12.Stonell CA, Leslie K, Absalom AR. Effect-site targeted patient-controlled sedation with propofol: comparison with anesthetist administration for colonoscopy. Anaesthesia. 2006;61:240–7. doi: 10.1111/j.1365-2044.2005.04509.x. [DOI] [PubMed] [Google Scholar]
- 13.von Delius S, Schmid RM, Huber W. Bispectral index monitoring of sedation during endoscopy: buried alive? American Journal of Gastroenterology. 2007;102:686–7. doi: 10.1111/j.1572-0241.2007.01040_1.x. [DOI] [PubMed] [Google Scholar]
- 14.Stonell CA, Leslie K, Absalom AR. Effect-site targeted patient-controlled sedation with propofol: comparison with anesthetist administration for colonoscopy. Anaesthesia. 2006;61:240–7. doi: 10.1111/j.1365-2044.2005.04509.x. [DOI] [PubMed] [Google Scholar]
- 15.Hadzidiakos D, Petersen S, Baars J, Herold K, Rehberg B. Comparison of a new composite index based on midlatency auditory evoked potentials and electroencephalographic parameters with Bispectral index (BI) during moderate propofol sedation. European Journal of Anaesthesiology. 2006;23:931–6. doi: 10.1017/S0265021506001050. [DOI] [PubMed] [Google Scholar]
- 16.Ellerkmann RK, Soehle M, Alves TM, et al. Spectral entropy and Bispectral index as measures of the electroencephalographic effects of propofol. Anesthesia and Analgesia. 2006;102:1456–62. doi: 10.1213/01.ane.0000204289.47792.56. [DOI] [PubMed] [Google Scholar]
- 17.Zhong T, Guo QL, Pang YD, Peng LF, Li CL. Comparative evaluation of the cerebral state index and the Bispectral index during target-controlled infusion of propofol. British Journal of Anaesthesiology. 2005;95:798–802. doi: 10.1093/bja/aei253. [DOI] [PubMed] [Google Scholar]
- 18.Zou L, Quan X, Lin SF, Tian SY, Wang LP, Ye TH. Comparison of cerebral state index and Bispectral index accuracies in sedation monitoring during target control infusion of midazolam. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2008;30:330–3. [PubMed] [Google Scholar]
- 19.Tukenmez B, Memis D, Pamukcu Z. The addition of haloperidol, propofol, or midazolam to sufentanil for intravenous sedation in the intensive care unit using Bispectral index. Journal of Opioid Management. 2008;4:34–40. doi: 10.5055/jom.2008.0006. [DOI] [PubMed] [Google Scholar]
- 20.Lu CH, Ou-Yang HY, Man KM, et al. Relative reliability of the auditory evoked potential and Bispectral Index for monitoring sedation level in surgical intensive care patients. Anaesthesia Intensive Care. 2008;36:553–9. doi: 10.1177/0310057X0803600409. [DOI] [PubMed] [Google Scholar]
- 21.Lamas A, Lopez-Herce J, Sancho L, et al. Assessing sedation in critically ill children by Bispectral index, auditory-evoked potentials and clinical scales. Intensive Care Medicine. 2008;34:2092–9. doi: 10.1007/s00134-008-1198-1. [DOI] [PubMed] [Google Scholar]
- 22.Soto R, Nguyen TC, Smith RA. A comparison of Bispectral index and entropy, or how to misinterpret both. Anesthesia and Analgesia. 2005;100:1059–61. doi: 10.1213/01.ANE.0000146435.58751.17. [DOI] [PubMed] [Google Scholar]
- 23.Willmann K, Springman S, Rusy D, Daily E. A preliminary evaluation of a new derived EEG index monitor in anesthetized patients. Journal of Clinical Monitoring and Computing. 2002;17:345–50. doi: 10.1023/a:1024266026091. [DOI] [PubMed] [Google Scholar]
- 24.Rampil IJ. A primer for EEG signal processing in anesthesia. Anesthesiology. 1998;89:980–1002. doi: 10.1097/00000542-199810000-00023. [DOI] [PubMed] [Google Scholar]
- 25.Wong CA, Fragen RJ, Fitzgerald PC, McCarthy RJ. The association between propofol-induced loss of consciousness and the SNAP index. Anesthesia and Analgesia. 2005;100:141–8. doi: 10.1213/01.ANE.0000138057.61904.FD. [DOI] [PubMed] [Google Scholar]
- 26.Sing HC, Kautz MA, Thorne DR, et al. High-frequency EEG as measure of cognitive function capacity: a preliminary report. Aviation, Space and Environmental Medicine. 2005;76:114–35. [PubMed] [Google Scholar]
- 27.Bruhn J, Myles PS, Sneyd R, Struys MM. Depth of anesthesia monitoring: what’s available, what’s validated and what’s next? British Journal of Anaesthesiology. 2006;97:85–94. doi: 10.1093/bja/ael120. [DOI] [PubMed] [Google Scholar]
- 28.Ruiz-Gimeno P, Soro M, Perez-Solaz A, et al. Comparison of the EEG-based SNAP index and the Bispectral (BIS) index during sevoflurane-nitrous oxide anaesthesia. Journal of Clinical Monitoring and Computing. 2005;19:383–9. doi: 10.1007/s10877-005-5871-4. [DOI] [PubMed] [Google Scholar]
- 29.Orser BA. Depth-of-anesthesia monitor and the frequency of intraoperative awareness. New England Journal of Medicine. 2008;358:1189–91. doi: 10.1056/NEJMe0800998. [DOI] [PubMed] [Google Scholar]
- 30.Avidan MS, Zhang L, Burnside BA, et al. Anesthesia awareness and the Bispectral index. New England Journal of Medicine. 2008;358:1097–108. doi: 10.1056/NEJMoa0707361. [DOI] [PubMed] [Google Scholar]
- 31.Bowdle TA. Depth of anesthesia monitoring. Anesthesiology Clinics. 2006;24:793–822. doi: 10.1016/j.atc.2006.08.006. [DOI] [PubMed] [Google Scholar]


