Abstract
Goals of work
This paper presents an observational study of the longitudinal effects of cancer treatment on quality of life (QoL) in patients treated for head and neck squamous cell carcinoma (HNSCC), and evaluated the contribution of patients' baseline illness cognitions to the prediction of QoL 2 years after diagnosis.
Patients and methods
One hundred seventy-seven patients eligible for primary treatment for HNSCC completed the Illness Perception Questionnaire-Revised at baseline and the European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire-30 at baseline, at 1-year and 2-year follow-ups.
Main results
Compared to baseline, patients reported better emotional functioning at both follow-ups (p < 0.001), worse social functioning at 12 months (p < 0.05), and better global health status at 24 months (p < 0.05). Patients' own implicit common sense beliefs about their illness added small but significant amounts of variance to the prediction of QoL after 2 years. Less belief in own behavior causing the illness predicted better functioning and better global health. Strong illness identity beliefs predicted worse functioning and worse global health. Negative perceptions about the duration of the illness (chronic timeline beliefs) and more negative perceived consequences also predicted worse QoL.
Conclusions
Our results on the negative perceptions about the duration of the illness, perceived consequences, and high symptom awareness predicting worse QoL illustrate the detrimental effects of uncertainty and negative expectations about the future course of the illness. The identification of these cognitive factors provides possible targets for counseling strategies to assist patients in long-term adjustment to HNSCC.
Keywords: Head and neck cancer, Oral oncology, Illness cognitions, Quality of life, Fear of recurrence (FoR) management
Introduction
Over more than 40 years, a variety of individual and group-based psychosocial interventions has been developed specifically for people with cancer. Although the findings of several meta-analyses and research reviews have suggested the overall usefulness of psychosocial interventions for enhancing coping and improving quality of life (QoL) in adult cancer patients [1–4], other researchers have concluded their reviews with a more pessimistic assessment that the evidence on efficacy is inconclusive at best [5, 6].
One of the suggestions to shed light on these mixed conclusions regarding effectiveness of psychosocial interventions is to study potential moderators of intervention efficacy, such as cancer type, interventionist, and type of intervention [7, 8]. With regard to type of intervention, results from meta-analyses studying the efficacy of various components of interventions show that those with a greater number of components related to social cognitive theory (self-efficacy beliefs, information processing, and expectations regarding outcome and self-regulation, i.e., the process of planning, monitoring, and changing one's behaviors and cognitions) predicted better short- and/or long-term QoL outcomes for cancer patients than do interventions lacking those components [3, 9].
Another suggested approach is to evaluate whether psychological process-level variables (e.g., self-esteem, perceived control) contribute to the outcome through mediating and/or moderating effects [10]. With respect to cognitive factors, studies identifying potential mechanisms by which interventions in cancer care may exert their effects on QoL are few. Only one intervention study tested whether a cognitive behavioral intervention modified cognitive factors (perceived stress management skills), and whether improvement in QoL was mediated by greater perceived skills [11]. Other studies suggested cognitive factors such as: fear of recurrence [12, 13], belief in curability [14], recovery expectations [15–17], cognitive appraisal of cancer threat [18], and patients' perceptions of their illness and treatment [19–21].
Although there has been minimal research into effective psychosocial intervention programs specifically for head and neck cancer patients, there is evidence to indicate that psychosocial interventions focusing on monitoring and altering cancer-relevant cognitions, emotions, and behaviors also have a positive effect in enhancing QoL in these patients [22–26].
Our study aims to identify specific cognitions that are (un)helpful in longer term adjustment to head and neck squamous cell carcinoma (HNSCC) as a useful step toward designing interventions for HNSCC patients. The self-regulation model (SRM) of Leventhal et al. [27] was adopted as a general conceptual framework for identifying the subjective cognitions (ideas, views, and beliefs) and emotional responses generated by the illness that contribute to adjustment after cancer treatment. According to the SRM, patients will form commonsense representations when presented with information that constitutes a threat to health, integrating internal and external stimulus information with their own preexisting illness theory. The SRM identifies two simultaneous processes—i.e., cognitive and affective—that make independent and potentially interactive contributions to health and illness behavior [28]. The model posits that the cognitive representations are conceptualized by patients along five core dimensions: beliefs about the illness' identity (beliefs about the disease label and the symptoms associated with the illness), possible cause (beliefs about how one gets the disease), timeline of the illness (beliefs about the likely duration of illness and its expected course), controllability (beliefs about which strategies can cure/control the illness), and consequences (the perceived severity and beliefs about the physical, social, economic, and emotional effects of the illness). Preliminary support for the use of this model for explaining changes in health behavior and QoL in cancer patients comes from studies in patients with breast cancer [29–32] and patients with head and neck cancer [33].
Our specific objectives were to extend our prior research in this area, in which we demonstrated that illness cognitions were significantly related to pretreatment QoL [21], by analyzing longitudinal data, determining relationships between baseline illness cognitions, and QoL outcome 2 years after diagnosis.
Patients and methods
Study population
This is a prospective observational study, in which the study population consisted of all incident patients with head and neck squamous cell carcinoma who were diagnosed at the Department of Otolaryngology and Head and Neck Surgery at the Leiden University Medical Center, The Netherlands, between March 2002 and August 2005. Patients were followed-up until death or 2 years after diagnosis.
Patients were included if they were eligible for primary treatment for squamous cell carcinomas in the pharynx, larynx, oral cavity, nose, sinuses, esophagus, or salivary glands. Patients who were mentally retarded, demented, or unable to fill in the questionnaires for other reasons were excluded.
One hundred seventy-seven patients meeting study criteria were enrolled. The characteristics of the study sample are depicted in Table 1.
Table 1.
Demographic, treatment, and tumor-related characteristics of the study population
| At baseline (n = 177) | Dead (n = 53) | Missing (n = 29) | Final sample (n = 95) | |
|---|---|---|---|---|
| Age (year) | ||||
| Mean (SD) | 59.5 (11.3) | 62.1 (11.8) | 54.1 (10.1) | 59.6 (10.8) |
| Range | 28–84 | 40–84 | 28–70 | 36–84 |
| Gender (n) | ||||
| Male | 131 | 41 | 18 | 72 |
| Female | 46 | 12 | 11 | 23 |
| Treatment (n) | ||||
| None | 4 | 2 | – | 2 |
| Irradiation only | 72 | 23 | 14 | 35 |
| Surgery and irradiation | 42 | 11 | 10 | 20 |
| Surgery only | 39 | 8 | 4 | 27 |
| Chemotherapy only or combination with chemotherapy | 20 | 9 | 1 | 11 |
| Tumor (sub)sites (n) | ||||
| Pharynx | ||||
| Nasopharynx | 10 | 4 | 1 | 5 |
| Oropharynx | 46 | 15 | 8 | 23 |
| Hypopharynx | 18 | 9 | 2 | 7 |
| Larynx | 42 | 10 | 7 | 25 |
| Oral cavity | 38 | 8 | 9 | 21 |
| Other | 23 | 7 | 2 | 14 |
| Tumor stage (n) | ||||
| T0 | 4 | 1 | – | 3 |
| T1 | 32 | 3 | 2 | 27 |
| T2 | 51 | 13 | 15 | 23 |
| T3 | 37 | 12 | 5 | 20 |
| T4 | 44 | 18 | 6 | 20 |
| Tx | 9 | 6 | 1 | 2 |
| Nodal stage (n) | ||||
| N0 | 90 | 20 | 15 | 55 |
| N1 | 21 | 3 | 6 | 12 |
| N2 | 49 | 21 | 5 | 23 |
| N3 | 8 | 4 | 1 | 3 |
| Nx | 9 | 5 | 2 | 2 |
| Distant metastasis (n) | ||||
| M0 | 160 | 42 | 28 | 90 |
| M1 | 6 | 5 | – | 1 |
| Mx | 11 | 6 | 1 | 4 |
| AJCC/UICC tumor stage (n) | ||||
| I | 27 | 2 | 1 | 24 |
| II | 29 | 8 | 9 | 12 |
| III | 34 | 6 | 8 | 20 |
| IV | 80 | 33 | 10 | 37 |
| Unknown | 7 | 4 | 1 | 2 |
| Prior malignancy (n) | ||||
| No | 162 | 45 | 28 | 89 |
| Yes | 15 | 8 | 1 | 6 |
Procedure
Patients were invited to participate by a research nurse on the day of diagnostic testing. All patients were informed on the purpose of the study. The study was exempted from institutional review board approval because the questionnaires were administered as part of standard clinical care to assist treatment planning. For baseline assessment consenting, patients were handed a questionnaire packet, and they completed the questionnaires while waiting for the next diagnostic procedure, with the research nurse available to respond to questions. For follow-up measurements, patients received a questionnaire packet by mail. Upon completion, the packet was returned to the research nurse by mail, or handed in by the patient at their follow-up visit.
Measurement
The Illness Perception Questionnaire-Revised (IPQ-R) [34] was used to measure patients' cognitive and emotional representations of their illness. The first part of the questionnaire measures the illness identity dimension with a list of 14 commonly occurring symptoms: pain, sore throat, nausea, breathlessness, weight loss, fatigue, stiff joints, sore eyes, wheezinass, headaches, upset stomach, sleep difficulties, dizziness, and loss of strength. Patients are asked to rate whether or not they have experienced each symptom since their illness, and if they believe the symptom to be specifically related to their illness (yes or no). The summed yes-rated items on the second question form the illness identity scale, with higher scores indicating a stronger belief that the experienced symptoms are part of the patient's illness.
The second part of the IPQ-R consists of 38 statements using five-point Likert scale (range “strongly agree” to “strongly disagree”) and provides separate scores for the consequences, timeline (divided into the subscales: cyclical timeline and acute/chronic timeline), control (divided into the subscales: personal control and treatment control), illness coherence, and emotional representations scales. High scores indicate stronger beliefs in: serious consequences of the disease, a chronic long-term disease, illness and/or symptoms as cyclical in nature, the patients' own ability to control symptoms, and the effectiveness of treatment in controlling the illness. Higher scores on the illness coherence scale indicate a higher degree to which patients feel they have a coherent model of the illness, and higher scores on the emotional representations scale indicate a stronger emotional response to illness.
The third part, questions about causal attributions, uses the same five-point scale and consists of 18 items. Only the causal attributions that were endorsed by more than 20% of this patient group (“chance”, “smoking”, “own behavior”, “alcohol”, and “stress”) were included in further analyses. Since “smoking”, “alcohol” and “own behavior” were highly correlated (Pearson correlation > 0.50, p < 0.000), these items were summed together to form the “own behavior” subscale. Higher scores indicate stronger beliefs in own behavior, chance, or stress causing the illness. Means (± standard deviation (SD)) and reliability coefficients (Cronbach's α) for the IPQ-R scales are shown in Table 2. Reliability coefficients ranged from 0.70 to 0.92, indicating satisfactory internal consistency.
Table 2.
Mean scores (SD) and internal reliability scores (Cronbach's α) of the Illness Perception Questionnaire-Revised (IPQ-R) scales
| Scales IPQ-R | Mean (SD) | Cronbach's α |
|---|---|---|
| Illness identity | 2.61 (2.62) | 0.79 |
| Consequences | 19.11 (4.40) | 0.76 |
| Timeline | ||
| Cyclical | 9.68 (2.96) | 0.70 |
| Acute/chronic | 17.12 (4.45) | 0.86 |
| Control | ||
| Personal | 18.33 (3.51) | 0.74 |
| Treatment | 17.53 (3.15) | 0.78 |
| Illness coherence | 15.78 (4.05) | 0.75 |
| Emotional representations | 18.93 (6.43) | 0.92 |
| Causal attributions | ||
| Behavioral (alcohol, smoking, my own behavior) | 8.28 (3.20) | 0.80 |
| Chance | 3.28 (1.22) | – |
| Stress | 2.35 (1.19) | – |
The European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire 30 (EORTC QLQ-30; version 3) [35] was used to measure quality of life. It consists of 30 items, yielding nine summary scales: physical functioning, role functioning, emotional functioning, social functioning, cognitive functioning, global health status, fatigue, pain, and nausea, and six single items. With the exception of the global health status items, patients respond on four-point Likert scale (difficulties during the last week, range “not at all” to “very much”). Global quality of life is rated on a seven-point visual analog scale (health and quality of life during the last week, range “very poor” to “excellent”). Higher scores for functional scales and global health status represent a high level of functioning/quality of life, whereas high scores for symptoms represent high levels of impairment. The scales and single items of the QLQ-C30 were scored and transformed to scales of 0 to100 according to the EORTC Quality of Life Study Group scoring guidelines.
Statistical analysis
All data were analyzed using the Statistical Package for the Social Sciences (SPSS 14.0 for Windows). The significance of differences between patients who completed measurements and those who died or dropped out was tested using chi-square statistics and Student's t test. Descriptive means, standard deviations, and Cronbach's alpha for the IPQ-R subscales were calculated at baseline. A one-way repeated measures analysis of variance was conducted to explore changes in QLQ-C30 functioning and global health status scales at diagnosis, at 12 months and 24 months follow-ups. Multiple regressions were used to examine if baseline illness perceptions explained variance on QLQ-C30 functioning and global health status scales 2 years after diagnosis. For each QoL scale, multiple regression equations were computed. Age and American Joint Committee on Cancer (AJCC)/International Union Against Cancer (UICC) tumor stages were force-entered on the first step, and baseline measurement of the outcome variable was force-entered on the second step to correct for the possible influence of these factors on QoL at 24 months. On the third step, a selection of the IPQ-R subscales were entered stepwise into the regressions to examine which illness perceptions explain variance independently from and on top of age, tumor stage, and baseline QoL. To limit the number of variables entered into the regressions and avoid multicollinearity, only the IPQ-R cognitive and emotional representations scales that correlated significantly with QoL at 2 years were entered.
Results
Patients
Patients were followed prospectively over 2 years. From the initial patient sample, 53 patients died before the end of the study (30%), 29 patients withdrew (16%), and 95 patients completed measurements at 24 months. Patients who died within study period and patients who withdrew did not differ from participants who completed on baseline QoL or illness cognitions, except for chronic timeline beliefs—patients who died had higher chronic belief at baseline (F(2,149) = 6.9, p < 0.001). With respect to demographic, treatment, and tumor-related characteristics (see Table 1), patients who withdrew differed from patients who completed on AJCC/UICC tumor stage (χ2 = 10.3, df = 3; p < 0.05). Patients who withdrew were significantly less frequently diagnosed in stage I (4% vs 26%) and more frequently in stage II (32% vs. 13%). Patients who died also differed from patients who completed on AJCC/UICC tumor stage (χ2 = 9.8, df = 3; p < 0.05), with the majority of patients who died being diagnosed in stage IV (62.5%).
Quality of life over time
Scores on the QLQ-C30 functioning and global health status scales were compared across time for patients who completed measurements at 24 months (see Fig. 1). Significant effects for time were found for emotional functioning (Wilks' lambda = 0.73; F(2,65) = 11.84, p < 0.001), social functioning (Wilks' lambda = 0.90; F(2,65) = 3.52, p < 0.05), and global health status (Wilks' lambda = 0.90; F(2,62) = 3.44, p < 0.05), with significantly better emotional functioning at 12 and 24 months as compared to baseline (p < 0.001), social functioning being worse at 12 months (p < 0.05), but not significantly different from baseline at 24 months (p = 0.06), and better global health status scores at 24 months as compared to baseline (p < 0.05).
Fig. 1.
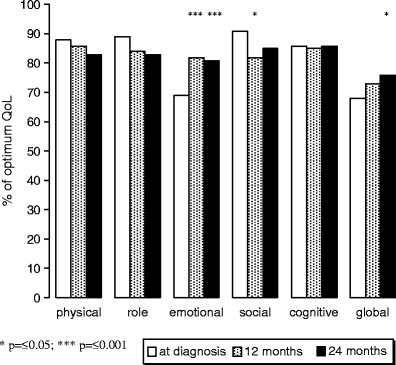
Differences between QLQ-C30 functioning scales and global health scale scores at diagnosis and at follow-up (12 months and 24 months); * p = ≤0.05; *** p = ≤0.001
Predicting quality of life
As evident in Table 3, tests of the significance of the variables in the equations revealed that age and AJCC/UICC tumor stages (step 1) did not significantly contribute to the variance in any of the QLQ-C30 scales at 24 months postdiagnosis. With the exception of role functioning, baseline QLQ-C30 functioning (step 2) explained most of the variance in functioning (47%, 42%, 26%, and 64% in physical, emotional, social, and cognitive functioning, respectively) and in global health (31%). Better baseline functioning predicted better functioning at 24 months.
Table 3.
Regression analyses: percentages of variance on the QLQ-C30 functioning scales and global health scale explained by age, illness stage, functioning at baseline, and illness perceptions
| Step and variablea | Beta | Summarized R2 (%) | R2 change (%) | F of change (p value) |
|---|---|---|---|---|
| Physical functioning | ||||
| 1. Age | 3% | 3% | NS | |
| Illness stage | ||||
| 2. Physical functioning T1 | 0.60 | 50% | 47% | *** |
| 3. Consequences | −0.19 | 54% | 4% | * |
| Behavioral attributions | −0.18 | 56% | 3% | * |
| Role functioning | ||||
| 1. Age | 1% | 1% | NS | |
| Illness stage | ||||
| 2. Role functioning T1 | 9% | 8% | NS | |
| 3. Illness identity | −0.44 | 24% | 15% | *** |
| Behavioral attributions | −0.26 | 32% | 9% | ** |
| Consequences | −0.22 | 36% | 4% | * |
| Emotional functioning | ||||
| 1. Age | 1% | 1% | NS | |
| Illness stage | ||||
| 2. Emotional functioning T1 | 0.65 | 42% | 42% | *** |
| Social functioning | ||||
| 1. Age | 2% | 2% | NS | |
| Illness stage | ||||
| 2. Social functioning T1 | 0.29 | 27% | 26% | *** |
| 3. Illness identity | −0.50 | 50% | 23% | *** |
| Behavioral attributions | −0.22 | 55% | 5% | * |
| Cognitive functioning | ||||
| 1. Age | 1% | 1% | NS | |
| Illness stage | ||||
| 2. Cognitive functioning T1 | 0.78 | 64% | 64% | *** |
| 3. Timeline chronic | −0.15 | 67% | 2% | * |
| Global health | ||||
| 1. Age | 4% | 4% | NS | |
| Illness stage | ||||
| 2. Global health T1 | 0.23 | 35% | 31% | *** |
| 3. Illness identity | −0.39 | 48% | 14% | *** |
| Behavioral attributions | −0.25 | 55% | 7% | ** |
| Consequences | −0.18 | 58% | 3% | * |
a Illness perceptions dimensions entered for the prediction of the QLQ-C30 functioning scales included: physical functioning: identity, consequences, and behavioral attributions; role functioning: identity, consequences, and behavioral attributions; emotional functioning: identity, consequences, emotional representations, and stress attributions; cognitive functioning: identity, timeline chronic, timeline cyclical, consequences, emotional representations, and stress attributions; social functioning: identity, timeline cyclical, consequences, illness coherence, emotional representations, behavioral attributions, stress attributions; global health: identity, timeline chronic, consequences, emotional representations, behavioral attributions, and stress attributions
NS nonsignificant
*p < 0.05; **p < 0.01, ***p < 0.001
When we examine the additional variance explained by baseline illness cognitions, results show that consequences beliefs (4%) and behavioral attributions (3%) significantly and uniquely added to the variance explained in QLQ-C30 physical functioning. Lower perceived consequences scores and less belief in own behavior causing the illness were associated with better physical functioning. Together with identity perceptions, behavioral attributions and perceived consequences beliefs also contributed significantly to the variance in QLQ-C30 role functioning (15%, 9%, and 4%, respectively) and global health status (14%, 7%, and 3%, respectively) with fewer perceived symptoms, less belief in own behavior causing the illness, and lower consequences scores being associated with better role functioning and higher global health status ratings. Behavioral attributions also contributed significantly to the variance in QLQ-C30 social functioning (5%), with less belief in own behavior causing the illness being associated with better social functioning. With regard to cognitive functioning, chronic timeline perceptions added a small but significant 2% to the prediction. Belief that the illness will be permanent was associated with worse cognitive functioning.
Discussion
This paper presents an observational study of the longitudinal effects of cancer treatment on QoL in HNSCC patients, exploring the contribution of baseline illness cognitions to the prediction of QoL 2 years after diagnosis. The general findings in the present study suggest that, although pretreatment QoL is by far the strongest predictor of QoL at 2-year follow-up, illness cognitions add small but significant amounts of variance to the prediction, with behavioral attributions, perceived consequences, and illness identity beliefs as the most consistent predictors.
Longitudinal results from prospective studies in head and neck cancer patients show a general trend that QoL deteriorates during and directly after treatment and restores to pretreatment levels after 12 months, despite functional disabilities [36]. Studies with longer follow-up periods report no clinical significant improvements in QoL after 1 year, with physical functioning and role functioning deteriorating over time and emotional functioning and global health improving [37–39]. Findings in our sample on quality of life over time closely parallel these results.
Less belief in own behavior causing the illness was predictive of better physical, role, and social functioning and better global health in our sample. These results strengthen our previous cross-sectional findings [21] and confirm the negative effects of self-blame in cancer found by others, who reported that stronger self-blame attributions were related to more psychological distress and poorer QoL in patients with breast cancer [40–43]. Also, self-blame was found to be associated with potentially health-damaging behavior following treatment in patients with head and neck cancer [44]. Christensen et al. found that patients who attributed the cause of their cancer to their past substance use exhibited a higher likelihood of continued smoking, but only if they also reported a low degree of perceived control over their future cancer-related health. Other studies found that blaming one's past behaviors for cancer does not (automatically) enhance perceptions of control over future recovery/recurrence [40, 42, 43] and conclude that healthcare professionals should offer support to discourage the use of self-blame.
Strong baseline illness identity beliefs predicted worse social and role functioning and worse global health after 2 years. High symptom awareness predicting distress was also found in a study by Millar et al. [29]. It is suggested that persistent vigilance to somatic signs is associated with fear of recurrence (FoR) and that even common, noncancer-like symptoms (such as tiredness and headaches) will become part of patients' illness models, thus playing a major role in evoking FoR [13, 45]. FoR can arise already before treatment [46, 47], is one of the most frequently reported concerns for many cancer patients [48], and is an important stressor in the lives of both patients and significant others [49–51]. Our results on the negative perceptions about the duration of the illness (chronic timeline beliefs) and more negative perceived consequences at baseline predicting worse QoL also illustrate the detrimental effects of uncertainty and negative expectations about the future course of the illness. The results are consistent with previous research in showing that the way patients conceptualize their cancer appears to be more influential in determining levels of posttreatment distress than objective indicators of the likelihood of recurrence (e.g., stage of disease) or performance status (Karnofsky status) [15, 30]. Studies examining QoL in long-term cancer survivors have reported that uncertainty in illness extends the impact of cancer well beyond the treatment phase and has a direct negative influence on patients' QoL [12, 45, 50, 52–54]. According to the self-regulation model, patients' illness cognitions help determine their illness-related behaviors and attempts to control distress. We speculate that those who have more negative expectations about the future course of the illness may be less motivated to engage in adaptive strategies to remain well and psychologically healthy, both during and after treatment. The stronger associations between negative illness cognitions and social and role functioning in our study might indicate that these patients are less successful in organizing social support.
One of the limitations of the current study is that the sample is inherently biased toward patients who survived. Patients who remained in the study were recruited into the study with significantly earlier stage tumors, and our data are, therefore, biased toward patients with relatively less physical consequences of treatment. Also, our longitudinal design does not take into account the concerns raised by Llewellyn et al. [33] that baseline factors such as illness cognitions are likely to change over the course of the illness. Within the SRM, it is posited that illness cognitions, health-related responses, and evaluation of health outcome are recursive stages in illness trajectories, thus suggesting that studying the process of change in illness cognitions might be more appropriate than studying pretreatment beliefs as predictors of longitudinal outcomes. In our study, illness cognitions were measured in the initial stages of treatment (even before patients received their definitive diagnosis), and since, it is likely that over the course of a (very burdensome) treatment patients' beliefs will fluctuate, this might explain why baseline illness cognitions only added small amounts of variance to the prediction of follow-up QoL. From our results, however, it can also be tentatively concluded that by changing pretreatment cognitive factors, long-term functional outcome after treatment for HNSCC can be improved.
Understanding the experience of illness from the perspective of the patient provides clinically relevant information to guide intervention and treatment. In particular, when assisting patients to adjust to HNSCC, from our results, it seems that supportive care need to target patients' cognitions about own behavior causing the illness, illness identity beliefs, chronic timeline beliefs, and perceived consequences. Intervention trials addressing uncertainty and FoR underline the importance of influencing negative cognitions in reducing uncertainty and strengthening QoL outcome after cancer [13, 55, 56]. One intervention was especially designed to assist patients with uncertainty during watchful waiting in prostate cancer. This study documents benefits of cognitive reframing and refocusing attention to satisfying and rewarding areas of life [55].The very recently developed intervention by Humphris and Ozakinci [13] seems promising and concentrates on symptom identification and recurrence fear management, discussion of illness beliefs, and moderation of excessive checking behavior in patients with head and neck cancer. Results from intervention studies with a focus on self-regulation in other illnesses show that changing illness perceptions is possible and can result in improved functional outcome [57–59].
Acknowledgments
Conflict of interest statement
None of the authors has a conflict of interest to declare in relation to this work.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Gysels M, Higginson IJ, Rajasekaran M, Davies E, Harding R (2004) Improving supportive and palliative care for adults with cancer. National Institute for Clinical Excellence. http://www.nice.org.uk/nicemedia/pdf/csgspresearchevidence.pdf. Date accessed: 30 July 2008
- 2.Meyer TJ, Mark MM. Effects of psychosocial interventions with adult cancer patients: a meta-analysis of randomized experiments. Health Psychol. 1995;14:101–108. doi: 10.1037/0278-6133.14.2.101. [DOI] [PubMed] [Google Scholar]
- 3.Osborn RL, Demoncada AC, Feuerstein M. Psychosocial interventions for depression, anxiety, and quality of life in cancer survivors: meta-analyses. Int J Psychiatry Med. 2006;36:13–34. doi: 10.2190/EUFN-RV1K-Y3TR-FK0L. [DOI] [PubMed] [Google Scholar]
- 4.Rehse B, Pukrop R. Effects of psychosocial interventions on quality of life in adult cancer patients: meta-analysis of 37 published controlled outcome studies. Patient Educ Counsel. 2003;50:179–186. doi: 10.1016/s0738-3991(02)00149-0. [DOI] [PubMed] [Google Scholar]
- 5.Lepore SJ, Coyne JC. Psychological interventions for distress in cancer patients: a review of reviews. Ann Behav Med. 2006;32:85–92. doi: 10.1207/s15324796abm3202_2. [DOI] [PubMed] [Google Scholar]
- 6.Newell SA, Sanson-Fisher WR, Salvolainen NJ. Systematic review of psychological therapies for cancer patients: overview and recommendations for future research. J Natl Cancer Inst. 2002;94:558–584. doi: 10.1093/jnci/94.8.558. [DOI] [PubMed] [Google Scholar]
- 7.Stanton AL. Psychosocial concerns and interventions for cancer survivors. J Clin Oncol. 2006;24:5132–5137. doi: 10.1200/JCO.2006.06.8775. [DOI] [PubMed] [Google Scholar]
- 8.Zimmermann T, Heinrichs N, Baucom DH. “Does one size fit all?” Moderators in psychosocial interventions for breast cancer patients: a meta-analysis. Ann Behav Med. 2007;34:225–239. doi: 10.1007/BF02874548. [DOI] [PubMed] [Google Scholar]
- 9.Graves KD. Social cognitive theory and cancer patients' quality of life: a meta-analysis of psychosocial intervention components. Health Psychol. 2003;22:210–219. doi: 10.1037/0278-6133.22.2.210. [DOI] [PubMed] [Google Scholar]
- 10.Owen JE, Klapow JC, Hicken B, Tucker DC. Psychosocial interventions for cancer: review and analysis using a three-tiered outcomes model. Psychooncology. 2001;10:218–230. doi: 10.1002/pon.509.abs. [DOI] [PubMed] [Google Scholar]
- 11.Penedo FJ, Dahn JR, Molton I, Gonzalez JS, Kinsinger D, Roos BA, Carver CS, Schneiderman N, Antoni MH. Cognitive-behavioral stress management improves stress-management skills and quality of life in men recovering from treatment of prostate carcinoma. Cancer. 2004;100:192–200. doi: 10.1002/cncr.11894. [DOI] [PubMed] [Google Scholar]
- 12.Hart SL, Latini DM, Cowan JE, Carroll PR, Investigators CaPSURE. Fear of recurrence, treatment satisfaction, and quality of life after radical prostatectomy for prostate cancer. Support Care Cancer. 2008;16:161–169. doi: 10.1007/s00520-007-0296-x. [DOI] [PubMed] [Google Scholar]
- 13.Humphris G, Ozakinci G. The AFTER intervention: a structured psychological approach to reduce fears of recurrence in patients with head and neck cancer. Br J Health Psychol. 2008;13:223–230. doi: 10.1348/135910708X283751. [DOI] [PubMed] [Google Scholar]
- 14.Beadle GF, Yates PM, Najman JM, Clavarino A, Thomson D, Williams G, Kenny L, Roberts S, Mason B, Schlect D. Beliefs and practices of patients with advanced cancer: implications for communication. Br J Cancer. 2004;91:254–257. doi: 10.1038/sj.bjc.6601950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Koller M, Lorenz W, Wagner K, Keil A, Trott D, Engenhart-Cabillic R, Nies C. Expectations and quality of life of cancer patients undergoing radiotherapy. J R Soc Med. 2000;93:621–628. doi: 10.1177/014107680009301205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wagner MK, Armstrong D, Laughlin JE. Cognitive determinants of quality of life after onset of cancer. Psychol Rep. 1995;77:147–154. doi: 10.2466/pr0.1995.77.1.147. [DOI] [PubMed] [Google Scholar]
- 17.Winterling J, Glimelius B, Nordin K. The importance of expectations on the recovery period after cancer treatment. Psychooncology. 2008;17:190–198. doi: 10.1002/pon.1216. [DOI] [PubMed] [Google Scholar]
- 18.Steginga SK, Lynch BM, Hawkes A, Dunn J, Aitken J. Antecedents of domain-specific quality of life after colorectal cancer. Psychooncology. 2008 doi: 10.1002/pon.1388. [DOI] [PubMed] [Google Scholar]
- 19.Llewellyn CD, McGurk M, Weinman J. Head and neck cancer: to what extent can psychological factors explain differences between health-related quality of life and individual quality of life? Br J Oral Maxillofac Surg. 2006;44:351–357. doi: 10.1016/j.bjoms.2005.06.033. [DOI] [PubMed] [Google Scholar]
- 20.Llewellyn CD, McGurk M, Weinman J. The relationship between the Patient Generated Index (PGI) and measures of HR-QoL following diagnosis with head and neck cancer: are illness and treatment perceptions determinants of judgment-based outcomes? Br J Health Psychol. 2007;12:421–437. doi: 10.1348/135910706X118422. [DOI] [PubMed] [Google Scholar]
- 21.Scharloo M, Baatenburg de Jong RJ, Langeveld TP, van Velzen-Verkaik E, Doorn-op den Akker MM, Kaptein AA. Quality of life and illness perceptions in patients with recently diagnosed head and neck cancer. Head Neck. 2005;27:857–863. doi: 10.1002/hed.20251. [DOI] [PubMed] [Google Scholar]
- 22.Allison PJ, Edgar L, Nicolau B, Archer J, Black M, Hier M. Results of a feasibility study for a psycho-educational intervention in head and neck cancer. Psychooncology. 2004;13:482–485. doi: 10.1002/pon.816. [DOI] [PubMed] [Google Scholar]
- 23.Hammerlid E, Persson LO, Sullivan M, Westin T. Quality-of-life effects of psychosocial intervention in patients with head and neck cancer. Otolaryngol Head Neck Surg. 1999;120:507–516. doi: 10.1053/hn.1999.v120.a90352. [DOI] [PubMed] [Google Scholar]
- 24.Semple CJ, Dunwoody L, Sullivan K, Kernohan WG. Patients with head and neck cancer prefer individualized cognitive behavioural therapy. Eur J Cancer Care (Engl) 2006;15:220–227. doi: 10.1111/j.1365-2354.2005.00643.x. [DOI] [PubMed] [Google Scholar]
- 25.Semple CJ, Dunwoody L, Kernohan WG, McCaughan E. Development and evaluation of a problem-focused psychosocial intervention for patients with head and neck cancer. Support Care Cancer. 2008 doi: 10.1007/s00520-008-0480-7. [DOI] [PubMed] [Google Scholar]
- 26.Vilela LD, Nicolau B, Mahmud S, Edgar L, Hier M, Black M, Franco EL, Allison PJ. Comparison of psychosocial outcomes in head and neck cancer patients receiving a coping strategies intervention and control subjects receiving no intervention. J Otolaryngol. 2006;35:88–96. doi: 10.2310/7070.2005.5002. [DOI] [PubMed] [Google Scholar]
- 27.Leventhal H, Leventhal EA, Contrada RJ. Self-regulation, health, and behavior: a perceptual-cognitive approach. Psychol Health. 1998;13:717–733. doi: 10.1080/08870449808407425. [DOI] [Google Scholar]
- 28.Hagger MS, Orbell S. A meta-analytic review of the commonsense model of illness representations. Psychol Health. 2003;18:141–184. doi: 10.1080/088704403100081321. [DOI] [Google Scholar]
- 29.Millar K, Purushotham AD, McLatchie E, George WD, Murray GD. A 1-year prospective study of individual variation in distress, and illness perceptions, after treatment for breast cancer. J Psychosom Res. 2005;58:335–342. doi: 10.1016/j.jpsychores.2004.10.005. [DOI] [PubMed] [Google Scholar]
- 30.Rabin C, Leventhal H, Goodin S. Conceptualization of disease timeline predicts posttreatment distress in breast cancer patients. Health Psychol. 2004;23:407–412. doi: 10.1037/0278-6133.23.4.407. [DOI] [PubMed] [Google Scholar]
- 31.Rabin C, Pinto B. Cancer-related beliefs and health behavior change among breast cancer survivors and their first-degree relatives. Psychooncology. 2006;15:701–712. doi: 10.1002/pon.961. [DOI] [PubMed] [Google Scholar]
- 32.Stewart DE, Cheung AM, Duff S, Wong F, McQuestion M, Cheng T, Purdy L, Bunston T. Attributions of cause and recurrence in long-term breast cancer survivors. Psychooncology. 2001;10:179–183. doi: 10.1002/pon.499.abs. [DOI] [PubMed] [Google Scholar]
- 33.Llewellyn CD, McGurk M, Weinman J. Illness and treatment beliefs in head and neck cancer: is Leventhal's commonsense model a useful framework for determining changes in outcomes over time? J Psychosom Res. 2007;63:17–26. doi: 10.1016/j.jpsychores.2007.01.013. [DOI] [PubMed] [Google Scholar]
- 34.Moss-Morris R, Weinman J, Petrie KJ, Horne R, Cameron LD, Buick D. The revised Illness Perception Questionnaire (IPQ-R) Psychol Health. 2002;17:1–16. doi: 10.1080/08870440290001494. [DOI] [Google Scholar]
- 35.Sherman AC, Simonton S, Adams DC, Vural E, Owens B, Hanna E. Assessing quality of life in patients with head and neck cancer: cross-validation of the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Head and Neck module (QLQ-H&N35) Arch Otolaryngol Head Neck Surg. 2000;126:459–467. doi: 10.1001/archotol.126.4.459. [DOI] [PubMed] [Google Scholar]
- 36.Ledeboer QC, Velden LA, Boer MF, Feenstra L, Pruyn JF. Physical and psychosocial correlates of head and neck cancer: an update of the literature and challenges for the future (1996–2003) Clin Otolaryngol. 2005;30:303–319. doi: 10.1111/j.1365-2273.2005.01035.x. [DOI] [PubMed] [Google Scholar]
- 37.Abendstein H, Nordgren M, Boysen M, Jannert M, Silander E, Ahlner-Elmqvist M, Hammerlid E, Bjordal K. Quality of life and head and neck cancer: a 5-year prospective study. Laryngoscope. 2005;115:2183–2192. doi: 10.1097/01.MLG.0000181507.69620.14. [DOI] [PubMed] [Google Scholar]
- 38.Nordgren M, Hammerlid E, Bjordal K, Ahlner-Elmqvist M, Boysen M, Jannert M. Quality of life in oral carcinoma: a 5-year prospective study. Head Neck. 2008;30:461–470. doi: 10.1002/hed.20735. [DOI] [PubMed] [Google Scholar]
- 39.Nordgren M, Jannert M, Boysen M, Ahlner-Elmqvist M, Silander E, Bjordal K, Hammerlid E. Health-related quality of life in patients with pharyngeal carcinoma: a 5-year follow-up. Head Neck. 2006;28:339–349. doi: 10.1002/hed.20334. [DOI] [PubMed] [Google Scholar]
- 40.Bennett KK, Compas BE, Beckjord E, Glinder JG. Self-blame and distress among women with newly diagnosed breast cancer. J Behav Med. 2005;28:313–323. doi: 10.1007/s10865-005-9000-0. [DOI] [PubMed] [Google Scholar]
- 41.Friedman LC, Romero C, Elledge R, Chang J, Kalidas M, Dulay MF, Lynch GR, Osborne CK. Attribution of blame, self-forgiving attitude, and psychological adjustment in women with breast cancer. J Behav Med. 2007;30:351–357. doi: 10.1007/s10865-007-9108-5. [DOI] [PubMed] [Google Scholar]
- 42.Houldin AD, Jacobsen B, Lowery BJ. Self-blame and adjustment to breast cancer. Oncol Nurs Forum. 1996;23:75–79. [PubMed] [Google Scholar]
- 43.Malcarne VL, Compas BE, Epping-Jordan JE, Howell DC. Cognitive factors in adjustment to cancer: attributions of self-blame and perceptions of control. J Behav Med. 1995;18:401–417. doi: 10.1007/BF01904771. [DOI] [PubMed] [Google Scholar]
- 44.Christensen AJ, Moran PJ, Ehlers SL, Raichle K, Karnell L, Funk G. Smoking and drinking behavior in patients with head and neck cancer: effects of behavioral self-blame and perceived control. J Behav Med. 1999;22:407–418. doi: 10.1023/A:1018669222706. [DOI] [PubMed] [Google Scholar]
- 45.Easterling DV, Leventhal H. Contribution of concrete cognition to emotion: neutral symptoms as elicitors of worry about cancer. J Appl Psychol. 1989;74:787–796. doi: 10.1037/0021-9010.74.5.787. [DOI] [PubMed] [Google Scholar]
- 46.Llewellyn CD, Weinman J, McGurk M, Humphris G. Can we predict which head and neck cancer survivors develop fears of recurrence? J Psychosom Res. 2008;65:525–532. doi: 10.1016/j.jpsychores.2008.03.014. [DOI] [PubMed] [Google Scholar]
- 47.Mehta SS, Lubeck DP, Pasta DJ, Litwin MS. Fear of cancer recurrence in patients undergoing definitive treatment for prostate cancer: results from CaPSURE. J Urol. 2003;170:1931–1933. doi: 10.1097/01.ju.0000091993.73842.9b. [DOI] [PubMed] [Google Scholar]
- 48.Meyerowitz BE, Kurita K, D'Orazio LM. The psychological and emotional fallout of cancer and its treatment. Cancer J. 2008;14:410–413. doi: 10.1097/PPO.0b013e31818d8757. [DOI] [PubMed] [Google Scholar]
- 49.Hodges LJ, Humphris GM. Fear of recurrence and psychological distress in head and neck cancer patients and their carers. Psychooncology. 2008 doi: 10.1002/pon.1346. [DOI] [PubMed] [Google Scholar]
- 50.Lasry JC, Margolese RG. Fear of recurrence, breast-conserving surgery, and the trade-off hypothesis. Cancer. 1992;69:2111–2115. doi: 10.1002/1097-0142(19920415)69:8<2111::AID-CNCR2820690817>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- 51.Mellon S, Kershaw TS, Northouse LL, Freeman-Gibb L. A family-based model to predict fear of recurrence for cancer survivors and their caregivers. Psychooncology. 2007;16:214–223. doi: 10.1002/pon.1074. [DOI] [PubMed] [Google Scholar]
- 52.Simard S, Savard J. Fear of Cancer Recurrence Inventory: development and initial validation of a multidimensional measure of fear of cancer recurrence. Support Care Cancer. 2008 doi: 10.1007/s00520-008-0444-y. [DOI] [PubMed] [Google Scholar]
- 53.Wonghongkul T, Dechaprom N, Phumivichuvate L, Losawatkul S. Uncertainty appraisal coping and quality of life in breast cancer survivors. Cancer Nurs. 2006;29:250–257. doi: 10.1097/00002820-200605000-00014. [DOI] [PubMed] [Google Scholar]
- 54.Zhao L, Portier K, Stein K, Baker F, Smith T. Exploratory factor analysis of the cancer problems in living scale: a report from the American Cancer Society's Studies of Cancer Survivors. J Pain Symptom Manage. 2009;37:676–686. doi: 10.1016/j.jpainsymman.2008.04.011. [DOI] [PubMed] [Google Scholar]
- 55.Bailey DE, Mishel MH, Belyea M, Stewart JL, Mohler J. Uncertainty intervention for watchful waiting in prostate cancer. Cancer Nurs. 2004;27:339–346. doi: 10.1097/00002820-200409000-00001. [DOI] [PubMed] [Google Scholar]
- 56.Gil KM, Mishel MH, Belyea M, Germino B, Porter LS, Clayton M. Benefits of the uncertainty management intervention for African American and White older breast cancer survivors: 20-month outcomes. Int J Behav Med. 2006;13:286–294. doi: 10.1207/s15327558ijbm1304_3. [DOI] [PubMed] [Google Scholar]
- 57.Fortune DG, Richards HL, Griffiths CE, Main CJ. Targeting cognitive-behaviour therapy to patients' implicit model of psoriasis: results from a patient preference controlled trial. Br J Clin Psychol. 2004;43:65–82. doi: 10.1348/014466504772812977. [DOI] [PubMed] [Google Scholar]
- 58.Petrie KJ, Cameron LD, Ellis CJ, Buick D, Weinman J. Changing illness perceptions after myocardial infarction: an early intervention randomized controlled trial. Psychosom Med. 2002;64:580–586. doi: 10.1097/00006842-200207000-00007. [DOI] [PubMed] [Google Scholar]
- 59.Skinner TC, Carey ME, Cradock S, Daly H, Davies MJ, Doherty Y, Heller S, Khunti K, Oliver L, DESMOND Collaborative Diabetes Education and Self-Management for Ongoing and Newly Diagnosed (DESMOND): process modelling of pilot study. Patient Educ Couns. 2006;64:369–377. doi: 10.1016/j.pec.2006.04.007. [DOI] [PubMed] [Google Scholar]


