Abstract
Successful virotherapy requires efficient virus spread within tumors. We tested whether the expression of hyaluronidase, an enzyme which dissociates the extracellular matrix (ECM), could enhance the intratumoral distribution of an oncolytic adenovirus and improve its therapeutic activity. As a proof of concept, we demonstrated that intratumoral coadministration of hyaluronidase in mice-bearing tumor xenografts improves the antitumor activity of an oncolytic adenovirus. Next, we constructed a replication-competent adenovirus expressing a soluble form of the human sperm hyaluronidase (PH20) under the control of the major late promoter (MLP) (AdwtRGD-PH20). Intratumoral treatment of human melanoma xenografts with AdwtRGD-PH20 resulted in degradation of hyaluronan (HA), enhanced viral distribution, and induced tumor regression in all treated tumors. Finally, the PH20 cDNA was inserted in an oncolytic adenovirus that selectively kills pRb pathway-defective tumor cells. The antitumoral activity of the novel oncolytic adenovirus expressing PH20 (ICOVIR17) was compared to that of the parental virus ICOVIR15. ICOVIR17 showed more antitumor efficacy following intratumoral and systemic administration in mice with prestablished tumors, along with an improved spread of the virus within the tumor. Importantly, a single intravenous dose of ICOVIR17 induced tumor regression in 60% of treated tumors. These results indicate that ICOVIR17 is a promising candidate for clinical testing.
Introduction
Oncolytic adenoviruses are promising agents for the treatment of cancer. These viruses are designed to selectively target, replicate in and kill cancer cells, while sparing normal cells. The release of virus progeny from lysed tumor cells results in an exponential increase of the virus inoculum, which can spread laterally throughout the tumor mass and potentially induce tumor regression. Early clinical trials with oncolytic adenoviruses established the safety of this therapy,1 even after i.v. administration.2,3 However, only limited responses were noted, and treatment with first generation of oncolytic adenoviruses did not significantly alter the course of the disease.
One of the main limitations that contributes to these suboptimal results is the inability of oncolytic adenoviruses to effectively spread in the solid tumors. Oncolytic adenoviruses, along with most novel targeted therapeutics, face tremendous transport barriers in the tumor interstitium, due in part to their relatively large size (90 nm), much larger than chemotherapeutic drugs. Also, solid tumor presents unique characteristics that hinder the transport of macromolecules.4 Among them, the presence of high amounts of extracellular matrix (ECM) and high interstitial fluid pressure in tumor interstitium are the main source of physical resistance to drug transport.
One attractive method for improving viral spread through the tumor mass is to degrade ECM with proteolytic enzymes. In this regard, pretreatment of tumors with trypsin or collagenase has been shown to enhance distribution of viral vectors after intratumoral administration.5,6 Also, replication-competent adenoviruses expressing relaxin, a peptide hormone that modulates collagen levels, have been constructed, and results showed that ECM degradation can enhance viral spread and prolong the survival of tumor-bearing mice.7,8
Hyaluronan (HA) is an important structural element of the ECM. HA is a high molecular weight linear glycosaminoglycan consisting of repeating disaccharide units. Concentrations of HA are elevated in several cancers, and often HA levels correlate with invasive and metastatic behavior.9 Degradation of HA with hyaluronidase has been shown to enhance the action of various chemotherapeutic agents in patients.10,11 Hyaluronidase is assumed to facilitate penetration and to decrease interstitial fluid pressure,12 permitting anticancer agents to reach malignant cells. Moreover, it has been proposed that hyaluronidase may itself have intrinsic anticancer activity.13
Interestingly, a recent report indicated that intratumoral coadministration of hyaluronidase and replication-competent adenoviruses can enhance viral distribution and improve virus potency in vivo.14 In the present study, we confirmed these results by administering a single dose of hyaluronidase and an oncolytic adenovirus in a melanoma model that expresses high levels of HA. Although successful results were obtained, this strategy is limited to intratumoral administration. As many patients have metastasic disease, intravenous administration would improve the utility of an oncolytic virotherapeutic approach for cancer. Therefore, we sought to determine whether expression of hyaluronidase by an oncolytic adenovirus, instead of coadministration, could enhance viral spread throughout the tumor after systemic administration.
As an initial step, we constructed a replication-competent adenovirus expressing PH20 hyaluronidase under the control of the major late promoter (MLP). PH20 is a glycosylphosphatidylinositol (GPI)-anchored hyaluronidase found in mammalian sperm.15 Human PH20 hyaluronidase was chosen for its ability to degrade HA at neutral pH (pH 5–8), in contrast to other human hyaluronidases (as HYAL1 and HYAL2) that are active only at acid pH (pH 3–4). In order to obtain a soluble hyaluronidase, we removed the GPI membrane attachment motif of PH20. Expression of such a soluble PH20 enhanced virus spread and improved therapeutic outcome when compared to the parental virus, resulting in tumor regression in all treated tumors. We next constructed an oncolytic adenovirus expressing PH20 hyaluronidase (ICOVIR17). The insertion of eight E2F-binding sites and one Sp1-binding site in the E1a endogenous promoter renders ICOVIR17 selective for replication in pRb pathway-defective tumor cells. HA degradation enhanced the intratumoral spread of ICOVIR17 and greatly improved its antitumor efficacy without increasing its toxicity.
Results
HA expression in tumor cell lines
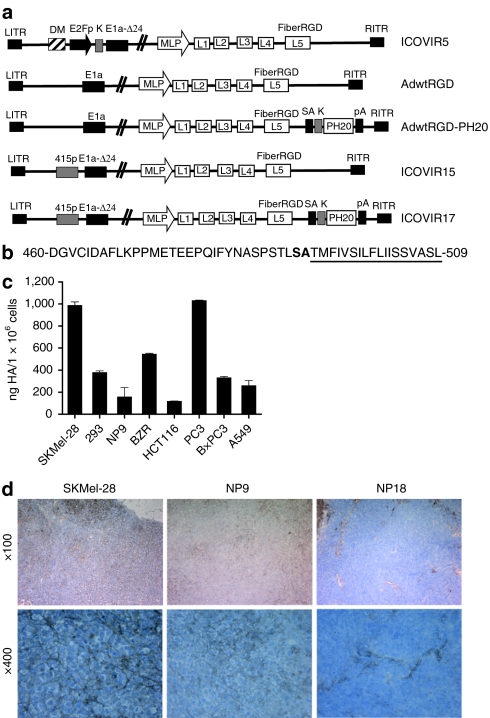
In order to choose a suitable model for in vivo experiments, we first measured HA expression levels in supernatants of various tumor cell lines using an ELISA-like assay. As shown in Figure 1c, SkMel-28 melanoma cells and PC3 prostate carcinoma cells produced the highest amounts of HA. Next, we analyzed HA production in human xenograft tumors (Figure 1d) by histochemical staining. Quantification of the percentage of stained areas indicated that both SKMel-28 and NP9 tumors expressed high amounts of HA (18.4 ± 6.6% and 14.2 ± 9.8%, respectively) around tumor cells. By contrast, NP18 tumors expressed sixfold less HA than SKMel-28 cells (2.7 ± 0.1%; P = 0.028), and this HA was mainly expressed within the tumor stroma. SkMel-28 melanoma cells, which produced high amounts of HA both in vitro and in vivo, were selected for in vivo experiments.
Figure 1.
Adenoviruses and cells used in this study. (a) Schematic representation of viruses used in this study. All viruses contain the RGD peptide in the HI loop of the fiber knob protein, and all oncolytic adenoviruses contain a deletion of 24 base pairs within the E1a region (E1a-Δ24). ICOVIR5 is an oncolytic adenovirus in which the endogenous E1A promoter has been replaced by the human E2F-1 promoter insulated with the DM-1. AdwtRGD and AdwtRGD-PH20 are RGD-modified wild-type adenoviruses. AdwtRGD-PH20 also contains a PH20 cassette inserted downstream of the fiber gene, consisting of a splice acceptor (SA) and a kozak sequence (k) in front of the PH20 cDNA and a polyadenylation sequence (pA). ICOVIR15 and ICOVIR17 are novel oncolytic adenoviruses that contain an E1a promoter modified by the insertion of eight E2F-binding sites and one Sp1-binding site at the nucleotide site 415 of the Ad genome. ICOVIR17 also contains the PH20 cassette after the fiber gene. DM, myotonic dystrophy locus insulator; MLP, major late promoter. (b) Amino acid sequence of the C-terminal human PH20 protein. The hydrophobic region responsible for signal attachment of a GPI anchor is underlined, and the cleavage site is represented in boldface type. The viruses used in this study express a soluble hyaluronidase lacking the cleavage site and the GPI membrane attachment motif. (c) Measurement of HA levels in the supernatants of various tumor cell lines by an ELISA-like assay. Mean ± SD is plotted. (d) Evaluation of HA levels within SKMel-28, NP9 and NP18 tumors by histochemical analysis using the biotinylated hyaluronan-binding protein (HABP-b). HA, hyaluronan.
Intratumoral administration of hyaluronidase enhances the efficacy of an oncolytic adenovirus in vivo
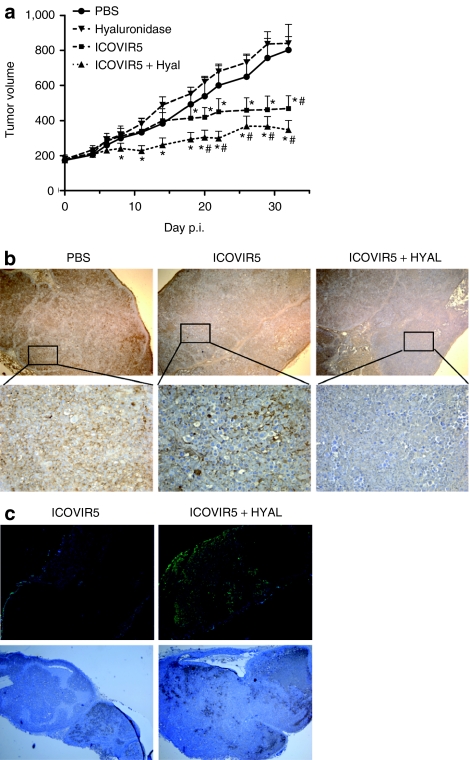
To investigate whether the degradation of the HA present in the ECM of solid tumors can enhance virus spread, we used an oncolytic adenovirus, ICOVIR5,16 in which replication is restricted to cancer cells with a disrupted Rb pathway (Figure 1a). Established s.c. SkMel-28 melanoma tumor xenografts (average size of 170 m3) were treated with a single intratumoral injection of phosphate-buffered saline (PBS), testicular bovine hyaluronidase (100 U/tumor), ICOVIR5 (1 × 109 viral particles (vp)/tumor), or a combination of hyaluronidase and ICOVIR5 (Figure 2a). A significant amount of tumor growth inhibition was detectable in tumors treated with the combination of virus and hyaluronidase compared with tumors treated with PBS (P ≤ 0.01) or hyaluronidase alone (P ≤ 0.01). Importantly, hyaluronidase administration increased the antitumor efficacy of ICOVIR5 compared with the virus administered in monotherapy. Although differences in tumor volume between these groups do not reach statistical significance, they do when percentage of tumor growth is analyzed (P ≤ 0.05 at days 11 and 14 after administration).
Figure 2.
Coadministration of testicular bovine hyaluronidase enhances spread and antitumor activity of ICOVIR5. (a) SkMel-28 s.c. tumor xenografts were treated intratumorally with PBS, testicular bovine hyaluronidase (PH20), ICOVIR5, or a combination of PH20 hyaluronidase and ICOVIR5. Tumor volume + SEM is plotted (n = 10). #Significant (P ≤ 0.01) compared with tumors treated with PBS; *Significant (P ≤ 0.01) compared with tumors treated with PH20 hyaluronidase. (b,c) Analysis of extracellular matrix and adenovirus distribution in melanoma tumors treated with ICOVIR5 or ICOVIR5, and hyaluronidase obtained 14 days after administration. (b) Deparaffinized tumor sections were stained for HA using the HABP-b and counterstained with hematoxylin. Upper panel: original magnification ×40. Lower panel: original magnification ×200. (c) Upper panel: adenovirus immunodetection was performed in frozen sections with an antiadenovirus antibody and counterstained with 4′,6′-diaminidino-2-phenylindole (DAPI); original magnification ×40. Lower panel: adenovirus immunodetection was performed in deparaffinized tumor sections with an antiadenovirus antibody and counterstained with hematoxylin; original magnification ×40. PBS, phosphate-buffered saline.
To assess whether the enhanced efficacy observed in animals treated with the combination was related to a decrease in HA levels in the tumor mass, as well as an improved distribution profile of ICOVIR5, immunohistochemical analysis was done on tumors obtained at 14 days after virus treatment (Figure 2b,c). SkMel-28 tumors treated with ICOVIR5 and hyaluronidase showed lower levels of expression of HA and more diffuse adenoviral capsid protein expression (a marker of virus replication) compared with tumors treated with ICOVIR5 alone. Control tumors injected with PBS presented high content of HA but no adenovirus positive cells (data not shown).
Expression of PH20 hyaluronidase enhances the efficacy of a replication-competent adenovirus in vivo
Next, we constructed AdwtRGD-PH20 (Figure 1a), a replication-competent adenovirus that expresses a soluble form of the human testicular hyaluronidase (PH20) lacking the GPI membrane attachment motif (Figure 1b). To restrict hyaluronidase expression to the later phases of the viral life cycle, PH20 cDNA was inserted in the AdwtRGD genome (Figure 1a) downstream of the fiber gene, under the control of the MLP. To confirm that AdwtRGD-PH20 expressed a functional soluble hyaluronidase, A549 cells were infected with AdwtRGD and AdwtRGD-PH20. After 48 hours, supernatants were collected and analyzed for hyaluronidase activity. Although no hyaluronidase activity was detected in supernatants of A549 cells infected with AdwtRGD, supernatants of AdwtRGD-PH20 infected cells were able to digest high molecular weight HA to oligosaccharides of different sizes (data not shown). In vitro characterization of AdwtRGD-PH20 in a panel of tumor cell lines showed similar levels of viral replication and cell cytotoxicity compared with the control virus, AdwtRGD (data not shown).
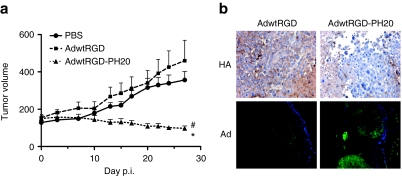
To analyze whether expression of the human PH20 hyaluronidase by a replication-competent adenovirus enhances the antitumor effect in vivo, mice-bearing SkMel-28 tumors were administered with a single dose of AdwtRGD or AdwtRGD-PH20 (1 × 109 vp/tumor). In this experiment, tumors treated with AdwtRGD showed a pattern of tumor growth similar to that of PBS-treated tumors (Figure 3a). In contrast, treatment with AdwtRGD-PH20 showed a significant reduction in tumor growth compared with PBS (P ≤ 0.005) and AdwtRGD (P ≤ 0.025). At day 27 after administration, 100% of tumors treated with AdwtRGD-PH20 showed partial regressions (10–70% of tumor reduction), whereas no tumor regressions were observed in mice treated with PBS or AdwtRGD. The enhanced antitumor activity displayed by the hyaluronidase expressing adenovirus, AdwtRGD-PH20, correlated with a decrease in tumor HA levels and wider areas of virus replication compared with tumors treated with AdwtRGD, as shown by immunohistochemical analysis (Figure 3b).
Figure 3.
Expression of human testicular hyaluronidase enhances spread and antitumor activity of a replication-competent adenovirus. (a) SkMel-28 s.c. tumor xenografts were treated intratumorally with PBS, AdwtRGD, or AdwtRGD-PH20. Tumor volume + SEM is plotted (n = 10). #Significant (P ≤ 0.005) compared with tumors treated with PBS from day 10 to day 27; *Significant (P ≤ 0.025) compared with tumors treated with AdwtRGD from day 7 to day 27. (b) Immunohistochemistry of melanoma tumors treated with AdwtRGD or AdwtRGD-PH20. Upper panel: tumor sections from tumors obtained 14 days after administration were stained for HA using the HABP-b; original magnification ×400. Lower panel: adenovirus immunodetection was performed in frozen sections from tumors obtained at day 27 after administration with an antiadenovirus antibody and counterstained with DAPI; original magnification ×100. HA, hyaluronan; PBS, phosphate-buffered saline.
Generation and characterization of ICOVIR17, an oncolytic adenovirus expressing PH20 hyaluronidase
On the basis of the observations described above, we constructed an oncolytic adenovirus expressing PH20 hyaluronidase. Insertion of the PH20 cDNA into ICOVIR5, in the absence of compensatory deletions, would result in a net increase in virion DNA of 2.5 kilobases (kb), exceeding the accepted packaging size limit of 1.8 kb. To address this problem, we used a new oncolytic adenovirus, ICOVIR15, which displays similar selectivity and potency to that of ICOVIR5 (J.J. Rojas, S. Guedan, A. Gros, M. Cascallo, and R. Alemany, unpublished results) but with a shorter genome. In ICOVIR15, selectivity was achieved by the insertion of eight E2F-binding sites and one Sp1-binding site in the E1a endogenous promoter (Figure 1a). Insertion of the PH20 expression cassette from AdwtRGD-PH20 in ICOVIR15, downstream of the fiber gene, resulted in the vector ICOVIR17 (Figure 1a). The genome of ICOVIR17 is ~1.7 kb larger than Adwt genome.
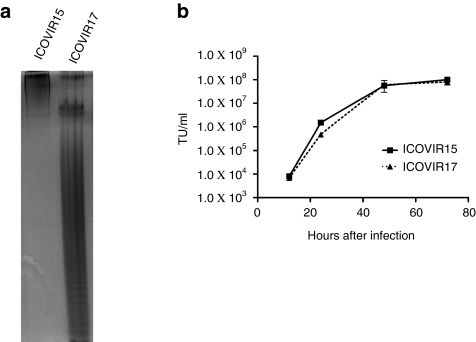
Analysis of hyaluronidase activity in the supernatants of tumor cells infected with ICOVIR15 and ICOVIR17 indicated that ICOVIR17 expresses a soluble hyaluronidase that is capable of digesting high molecular weight HA. As expected, no hyaluronidase activity was detected in supernatants of tumor cells infected with ICOVIR15 (Figure 4a).
Figure 4.
In vitro characterization of ICOVIR17. (a) PAGE-stained patterns of HA samples digested with supernatants of A549 cells obtained 48 hours after infection with ICOVIR15 and ICOVIR17. Electrophoresis shows that ICOVIR17 expresses a soluble protein that mediates the depolymerization of hyaluronan. (b) SkMel-28 cells were infected with ICOVIR15 or ICOVIR17 at high multiplicity of infection. At the indicated time points, cell extracts were harvested and titrated by α-hexon staining. Mean ± SD is plotted. TU, transduction units.
To analyze the impact of incorporating the PH20 cDNA on viral replication, we compared the replication kinetics of ICOVIR15 and ICOVIR17 in SkMel-28 cells. As shown in Figure 4b, both viruses reached approximately the same viral titer at all time points, indicating similar viral replication. Next, we examined the ability of ICOVIR17 to kill tumor cells when compared with ICOVIR15. IC50 values obtained in SkMel-28 and PC3 were 0.5 and 0.065 for ICOVIR15 and 0.33 and 0.099 for ICOVIR17, respectively. These results indicate that in vitro, where virus spread is not impaired, expression of PH20 hyaluronidase does not increase cytotoxicity of a conditionally replicative adenovirus.
ICOVIR17 displays potent antitumor activity after intratumoral administration in vivo
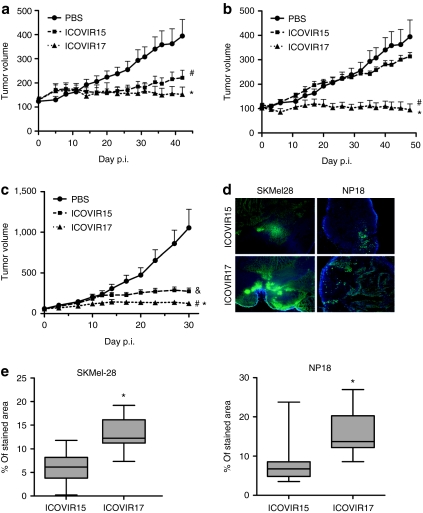
We next analyzed the antitumor activity of ICOVIR17 in established melanoma tumors after intratumoral administration. Mice-bearing SkMel-28 tumors (average size of 100 mm3) were treated with a single intratumoral injection of PBS, ICOVIR15, or ICOVIR17 (1 × 109 vp/tumor). Treatment with both viruses resulted in considerable inhibition of tumor growth when compared with untreated tumors. These effects were observed throughout the duration of the study (Figure 5a). At day 42 after administration, ICOVIR15 and ICOVIR17 induced a reduction of tumor growth of 60% (P ≤ 0.02) and 95% (P ≤ 0.002), respectively, compared to the control group. At this time point, animals were killed, and the weight of the tumors was measured. PBS- and ICOVIR15-treated tumors reached an average weight of 0.552 ± 0.4 and 0.342 ± 0.18 g, respectively. Tumors treated with the PH20-expressing adenovirus, ICOVIR17, were significantly smaller (0.199 ± 0.11 g) when compared with tumors treated with PBS (P = 0.022) or with the control virus, ICOVIR15 (P = 0.05), indicating that PH20 expression can significantly enhance the antitumor activity of an oncolytic adenovirus after intratumoral administration.
Figure 5.
Expression of human PH20 enhances the antitumor activity of an oncolytic adenovirus. (a) SkMel-28 s.c. tumor xenografts were treated intratumorally with PBS, ICOVIR15, or ICOVIR17. Tumor volume + SEM is plotted (n = 10). #Significant (P ≤ 0.05) compared with PBS-treated tumors from day 29 to day 42; *Significant (P ≤ 0.05) compared with PBS-treated tumors from day 23 to day 42. (b) SkMel-28 s.c. tumor xenografts were treated i.v. with a single dose of PBS, ICOVIR15, or ICOVIR17. Tumor volume + SEM is plotted (n = 7–10). #Significant (P ≤ 0.03) compared with tumors treated with PBS from day 20 to day 48; *Significant (P ≤ 0.05) compared with tumors treated with ICOVIR15 from day 14 to day 48. (c) NP18 s.c. tumor xenografts were treated i.v. with a single dose of PBS, ICOVIR15, or ICOVIR17. Tumor volume + SEM is plotted (n = 10). #Significant (P ≤ 0.02) compared with PBS-treated tumors from day 14 to day 30; &Significant (P ≤ 0.05) compared with PBS-treated tumors from day 14 to day 30; *Significant (P ≤ 0.02) compared with ICOVIR15-treated tumors from day 12 to day 30. (d) SkMel-28 and NP18 tumors treated with ICOVIR15 or ICOVIR17 were obtained at the end point of each experiment (b,c) and were frozen in OCT. Adenovirus immunodetection was performed in frozen sections with an antiadenovirus antibody and counterstained with DAPI. Representative sections of ICOVIR15- and ICOVIR17-treated tumors are shown. Original magnification ×40. (e) To quantify stained areas (which are indicative of virus replication), five random fields in viable tissue zones for each tumor (n = 7–10) were captured at original magnification ×100. Quantification of percentage of stained areas was done using ImageJ software. *Significant (P ≤ 0.01) compared with ICOVIR15-treated tumors. PBS, phosphate-buffered saline.
ICOVIR17 displays potent antitumor activity after systemic administration in vivo
The efficacy of ICOVIR17 via intratumoral administration led us to evaluate the ability of ICOVIR17 to control tumor growth when administered i.v. Mice-bearing SkMel-28 melanoma tumor xenografts (average size of 100 mm3) were treated with a single dose of PBS, ICOVIR15, or ICOVIR17 at 5 × 1010 vp/mouse administered via the tail vein. In this tumor model, which expresses high levels of HA, tumors treated with ICOVIR15 showed no therapeutic response, and all tumors continued to grow in a similar pattern to those treated with PBS (Figure 5b). In contrast, a single intravenous injection of ICOVIR17 had an immediate effect on tumor growth that lasted throughout the study. Within 1 week of virus administration, the tumors treated with ICOVIR17 showed a significantly reduction on tumor growth compared with PBS (P = 0.0012) and ICOVIR15 (P = 0.0057) treated mice. At the end of the study (day 49 after treatment), the PH20-expressing adenovirus, ICOVIR17, displayed a significantly greater antitumor effect than ICOVIR15 (P = 0.000008), with 60% of ICOVIR17-treated tumors showing regression between 20 and 40% of their pretreatment size.
To confirm that the antitumor activity shown by the PH20-expressing adenoviruses was not restricted to a single cell line, we performed an additional study in human pancreatic tumor xenografts. Mice carrying NP18 tumors (average size of 60 mm3) were treated with a single i.v. dose of PBS, ICOVIR15, or ICOVIR17 at 5 × 1010 vp/mouse (Figure 5c). By day 30 after treatment, animals were killed due to the large size of PBS-treated tumors. In these tumors, which express lower levels of HA compared with SkMel-28 tumors (Figure 1d), ICOVIR15 induced a significant tumor growth inhibition when compared with PBS-treated mice. Importantly, ICOVIR17 showed a drastically greater reduction in tumor progression that was significant compared with PBS and ICOVIR15 groups from early after the start of the treatment through the end of the study.
Finally, immunofluorescence detection of virus capsid proteins was performed in SkMel-28 and NP18 tumors harvested at the end of the study, and percentage of stained areas was quantified (Figure 5d,e). All tumors treated with ICOVIR15 and ICOVIR17 showed areas of virus replication that were not present in untreated tumors. ICOVIR15-treated tumors showed compact islands of infected cells that represented 6–8% of the total viable tissue. Importantly, tumors treated with the PH20-expressing adenovirus, ICOVIR17, showed a more diffuse pattern of adenovirus replication (about 13% of viable tissue was positive for virus capsid protein expression), which was significant compared to ICOVIR15-treated tumors in both tumor models.
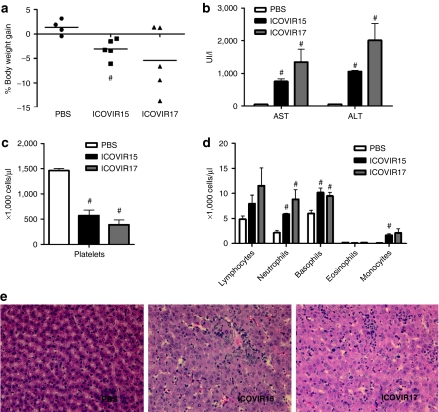
ICOVIR17 displays similar toxicity to ICOVIR15 after systemic administration in hamsters
Our in vivo studies showed that hyaluronidase expression enhanced the intratumoral spread of ICOVIR17 and greatly improved its antitumor efficacy. An important question was therefore whether this also resulted in increased toxicity. Because mice are poorly permissive for human adenoviruses, we performed this safety study in Syrian hamsters, which are immunocompetent and permissive for Ad5 replication. Hamsters were injected with PBS or with a previously determined maximum tolerable dose of 4 × 1011 vp of ICOVIR15 or ICOVIR17 via the cephalic vein. Weight loss, liver enzymes, and hematological profile were determined at day 5 after viral injection. Both ICOVIR15 and ICOVIR17 viruses slightly reduced body weight and caused a modest increase in AST and ALT levels (Figure 6a,b). Also, both viruses induced a modest but significant decrease in platelets and a significant increase in neutrophils and basophils compared with the PBS-treated hamsters (Figure 6c,d). Importantly, no significant differences in body weight loss, serum levels of liver enzymes, or hematological profile were observed between mice treated with ICOVIR15 or ICOVIR17, indicating that PH20 expression did not significantly increase toxicity caused by an oncolytic adenovirus.
Figure 6.
Toxicity profile after systemic administration of ICOVIR17 in hamsters. Syrian hamsters were injected intravenously with vehicle or with 4 × 1011 viral particles of ICOVIR15 or ICOVIR17. After 5 days of virus administration, animals were weighted and killed, and blood and liver samples were collected. (a) Percentage weight gain of individual animals. The average values for (b) serum transaminases, (c) platelet, and (d) leukocyte concentration in hamster peripheral blood at day 5 after virus administration. Mean values + SE of 4–5 hamsters/group are plotted. #Significant (P ≤ 0.05) compared with PBS-treated animals. (e) Livers of hamsters treated with PBS, ICOVIR15, or ICOVIR17 were excised at day 5 after virus administration. Paraffin sections of the livers were prepared, and each section was stained with hematoxylin and eosin. Original magnification ×400. ALT, alanine transaminase; AST, aspartate aminotransferase; PBS, phosphate-buffered saline.
In addition, histological analysis was performed on liver samples obtained at day 5 after systemic administration. Microscopic examination of liver sections from animals treated with ICOVIR15 and ICOVIR17 showed some areas of infiltration with marginal Councilman bodies in the more superficial areas of the liver. No apparent differences were noted between liver sections treated with ICOVIR15 or ICOVIR17.
Discussion
A major obstacle to the successful application of oncolytic adenovirus in cancer therapy is their inability to effectively spread in the solid tumors. In the present study, we attempted to enhance the intratumoral distribution of an oncolytic adenovirus by arming it with a soluble form of the human sperm hyaluronidase, PH20. By using this strategy, we have demonstrated that PH20 expression by an oncolytic adenovirus degrades the ECM, improves viral spread through the tumor mass and enhances the overall antitumor efficacy without increasing its toxicity. We have also demonstrated, for the first time, that an oncolytic adenovirus armed with hyaluronidase displays great antitumor efficacy after systemic administration.
As a proof of principle, we demonstrated that a single dose of hyaluronidase in combination with an oncolytic adenovirus is enough to improve adenovirus distribution within the tumor mass and enhance its antitumor activity. This data confirm previous results suggesting that ECM degradation enhances viral spread and is therefore greatly advantageous for the virotherapy for cancer.7,8,14 However, this approach presents some limitations. The first limitation relies on the fact that the effect of hyaluronidase on the ECM modulation is transient and reversible within 48 hours. Thus, the enhancement of the antitumoral activity observed with the combination therapy is probably due to an improvement in the spread of viruses produced early after the start of the treatment. Therefore, one might expect that maintained modulation of the ECM would further enhance adenoviral spread through the tumor. One possibility, which has already been used by Ganesh et al. is to inject hyaluronidase one every other day. By using this method, Ganesh et al. demonstrated that combination treatment of an oncolytic adenovirus and human recombinant PH20 significantly increases the antitumor activity and survival of tumor-bearing animals in two different models. However, although these strategies gave encouraging results, the utility of hyaluronidase co-injection is limited to intratumoral administration into localized tumors.
Our goal in this study was to develop an oncolytic adenovirus with improved spread capabilities for the treatment of disseminated disease. We speculated that hyaluronidase expression by replication-competent adenoviruses should allow maintained ECM degradation around areas of viral replication, enhancing therefore virus spread within the tumor. To this end, two viruses were generated expressing a soluble version of PH20 hyaluronidase under the control of the MLP: AdwtRGD-PH20, a replication-competent adenovirus; and ICOVIR17, a replication-selective adenovirus. In vitro characterization of the new viruses showed that expression of a soluble hyaluronidase lacking the GPI attachment motive was feasible, and did not impair virus replication. In vivo, both PH20-expressing viruses displayed greater antitumor efficacy compared with their nonarmed counterparts in the tumor models tested. Importantly, PH20 expression increased oncolytic activity of ICOVIR17 after i.v. administration in two different tumor models compared to its counterpart, ICOVIR15. In all the experiments, this enhanced oncolytic activity was achieved after a single administration of the virus, and the tumor burden at the time of treatment (2 or 3 weeks after tumor cell implantation) was extensive. It is important to note that although 60% of tumors showed partial regressions and all tumors showed active ICOVIR17 replication areas, complete tumor responses were rarely achieved. One possible explanation to the low rate of complete tumor regressions is that tumor stroma of murine origin precludes a complete intratumoral spread, as mouse cells are not permissive for human adenovirus replication. However, fibrotic tissue may not be such an important issue for viral spread in the clinical setting as preliminary results indicate that ICOVIR15 efficiently replicates in carcinoma-associated fibroblast of human origin (data not shown).
Other studies have sought to improve adenovirus spread by degrading matrix components. For example, two different groups constructed oncolytic adenoviruses armed with relaxin, a peptide hormone that downregulates expression of collagen and upregulates expression of matrix metalloproteinases. Consistent with our results, ECM degradation by relaxin-expressing oncolytic viruses increased virus spread and improved antitumor efficacy7,8 supporting the idea that ECM components inhibit viral spread following administration of adenoviruses. One important issue that may be considered is whether expression of these proteolytic enzymes can increase the toxicity of the oncolytic virotherapy. In this regard, Kim et al. demonstrated that injections of subcutaneous tumors with a relaxin-expressing virus yielded no increase in metastases, although their in vitro results showed that relaxin expression could increase cell invasion, which raises potential safety concerns.8 In our work, we sought to prevent uncontrolled expression of hyaluronidase in normal cells by expressing PH20 late in the adenoviral infection cycle, under the control of the MLP. Late PH20 expression by an oncolytic adenovirus should be restricted to tumor cells, thus maintaining the safety of the original virus in normal cells. In the toxicology studies performed in hamsters, we have not detected significant differences in body weight loss, serum levels of liver enzymes, or hematological profile following i.v. injection of ICOVIR17 compared to ICOVIR15, indicating that PH20 expression does not seem to affect the safety profile of a conventional oncolytic adenovirus. Importantly, the safety and tolerability of hyaluronidase has been analyzed in clinical trials, showing that hyaluronidase seemingly has no toxicity toward normal tissues or other adverse effects.10,17,18 In particular, hyaluronidase administration in preclinical studies and clinical trials has not been related to an increase of metastasis.13,14,19,20,21 In this regard, Bookbinder et al. demonstrated that the size of channels created in the interstitial matrix after depletion of HA by PH20 hyaluronidase allowed the diffusion of particles up to 200 nm in diameter.22 Therefore, adenovirus particles, but not tumor cells, would be allowed to penetrate through these channels.
Hyaluronidase has shown promise as an adjuvant in local and systemic cancer therapy. The mechanism by which hyaluronidase enhances therapeutic effect of chemotherapeutics is not well known, but degradation of the ECM is assumed to improve the penetration of the drug into the tumor tissue. However, it has also been suggested that hyaluronidase may itself have intrinsic anticancer activity. In this regard, Shuster et al. reported that hyaluronidase administration in severe combined immunodeficient mice-bearing human breast tumors caused rapid reduction in tumor size, and they suggest that hyaluronidase treatment of tumor cells appears to have induced an irreversible change in cell cycle kinetics.13 Recently, Lokeshwar et al. stably transfected prostate cancer cell lines to generate moderate, high, and low hyaluronidase producers.23 They demonstrated that when hyaluronidase is expressed at high doses, it can act as a tumor suppressor by inducing apoptosis. In this report, we show that either hyaluronidase coadministration or hyaluronidase expression by an oncolytic adenovirus enhances virus distribution within the tumor, resulting in a widespread viral replication and improved therapeutic outcome. In contrast, when PH20-expressing viruses were analyzed in vitro, where viral spread is not impaired, all viruses showed similar cytotoxicity in all conditions tested. Therefore, our results suggest that the main mechanism by which hyaluronidase increases oncolytic potency in vivo is probably related to the ECM degradation and interstitial fluid pressure reduction. However, we cannot rule out the possibility that other mechanisms, such as apoptosis induction in cancer cells, could be contributing to the enhanced potency of PH20-expressing adenoviruses.
Taken together, our results provide conclusive evidence that ECM disruption by hyaluronidase greatly enhances the distribution of oncolytic adenovirus within the tumor mass, which results in a more potent antitumor effect in all tumor models tested. Also, we show that ICOVIR17, an oncolytic adenovirus expressing PH20, has strong antitumor activity after both intratumoral and systemic administration, resulting in tumor regression. Taken into account that oncolytic adenoviruses have shown promising results in combination with chemotherapy,24 and that hyaluronidase administration improves antitumor effects of various chemotherapeutics, we believe that combination of ICOVIR17 with chemotherapy may present synergistic effects. We believe that ICOVIR17 is highly promising for clinical development for the treatment of disseminated cancers, and it might also be combined with other therapies, such as chemotherapy.
Materials and Methods
Cell lines. HEK293, A549 lung adenocarcinoma cells, SkMel-28 melanoma cells, PC3 prostate carcinoma cells, BxPC3 pancreatic tumor cells, and HTC116 colon cancer cells were obtained from the American Type Culture Collection (ATCC, Manassas, VA). BZR cells were derived from Beas-2B normal lung cells and were also obtained from the ATCC. NP9 and NP18 pancreatic tumor cells were established in our laboratory.25 All cell lines were routinely tested by mycoplasma presence and authenticated by morphology and growth curve analysis. All cell lines were grown in Dulbecco's modified Eagle's medium supplemented with 5% fetal bovine serum.
HA determination. To determine the HA content in the supernatants of various cell lines, cell cultures were grown to 60% confluence in Dulbecco's modified Eagle's medium containing 5% fetal bovine serum. Medium was removed and cells were incubated in serum-free medium. After 24 hours, medium was collected and digested overnight with 0.1 mg/ml of pronase. HA content in samples was measured using an ELISA assay as previously described.26
Recombinant adenoviruses. A schematic diagram outlining the structure of the viruses used in this study is shown in Figure 1a. AdwtRGD and ICOVIR5 have been described previously.16 ICOVIR15 is a conditionally replicative adenovirus in which expression of the adenovirus E1A-Δ24 gene is regulated by a modified endogenous E1A promoter, which contains eight E2F-1 binding sites and one Sp1-binding site. To generate PH20-expressing adenoviruses, a shuttle plasmid, pNKFiberPH20, was constructed as described below. PH20 exons II, III, and IV were amplified by PCR using A549 cellular DNA as the template DNA. The reverse primer used to amplify exon IV was designed such that the nucleotide sequence encoding for the GPI membrane attachment motif was excluded. The PCR products corresponding to exons II, III, and IV were mixed and reamplified with specific primers engineered to amplify the complete PH20 cDNA. The forward primer contained the acceptor splicing sequence of the virus protein IIIa and the kozak sequence, and the reverse primer contained a polyadenylation signal for PH20. Both primers contained MfeI restriction sites. The PCR product, containing the PH20 cassette, was digested with MfeI and ligated to the pNKFiberRGD plasmid27 to generate pNKFiberPH20. The NotI-KpnI fragment of pNKFiberPH20 and the partially SwaI-digested pVK50cau a plasmid derived from pVK50 (ref. 28), containing the yeast replication elements and a selectable marker to allow the homologous recombination in yeast) were recombined by homologous recombination, performed in yeast. This recombination event resulted in generation of the pAdwtRGD-PH20. pICOVIR17 was obtained using a similar strategy; pICOVIR15 was partially digested with SpeI and recombined with the SpeI-PacI fragment of pAdwtRGD-PH20. AdwtRGD-PH20 and ICOVIR17 were generated in HEK293 and plaque purified in A549 cells. All viruses were amplified in A549 cells and purified on CsCl gradients according to standard techniques.
Production assays. A549 and SkMel-28 cells were infected with 25 transduction units (TU)/cell of each virus to allow for 80–100% infection. After 4 hours, infection medium was removed; cells were washed twice with PBS and incubated with fresh medium. At the indicated time points, cells and medium (CE) were harvested and subjected to three rounds of freeze–thaw lysis. Viral titers of CE were determined in triplicate according to an antihexon staining–based method in HEK293 cells.29
Assay for hyaluronidase activity. A549 cells were infected with 25 TU/cell of each virus to allow for 80–100% infection. After 24 hours, infection medium was removed, and cells were incubated with serum-free medium. After 24 hours, medium was collected and concentrated by filtration using Amicon Ultra-4 columns (Millipore, Billerica, MA). The hyaluronidase activity of concentrated supernatants was analyzed as previously described.30 Briefly, supernatant samples containing soluble hyaluronidase were mixed with a HA (Sigma, St Louis, MO) solution in phosphate buffer (pH 6.0) containing 0.1 mol/l NaCl and 0.05% bovine serum albumin, and samples were incubated overnight at 37 °C. HA fragment sizes after digestion with samples were analyzed by polyacrylamide gel electrophoresis and visualized using a combined Alcian blue and silver staining protocol.
In vitro cytotoxicity assay. Cytotoxicity assay was performed by seeding 10,000 SkMel-28 and PC3 cells in 96-well plates in Dulbecco's modified Eagle's medium with 5% fetal bovine serum. Cells were infected with serial dilutions starting with 180 TU/cell. At day 5 after infection (PC3) or day 8 after infection (SkMel-28), plates were washed with PBS and stained for total protein content (bicinchoninic acid assay; Pierce Biotechnology, Rockford, IL), and absorbance was quantified. The TU per cell required to produce 50% growth inhibition (IC50 value) was estimated from dose–response curves by standard nonlinear regression (GraFit; Erithacus Software, Surrey, UK), using an adapted Hill equation.
In vivo efficacy studies. Animal studies were performed at the IDIBELL facility (AAALAC unit 1,155) and approved by the IDIBELL's Ethical Committee for Animal Experimentation. For in vivo coadministration studies, subcutaneous SkMel-28 melanoma tumors were established by injection of 5 × 106 cells into the flanks of 6-week-old male Balb/C nu/nu mice. Once tumors reached 170 mm3, mice were randomized (n = 6), and tumors were treated with a single intratumoral injection of 25 µl of PBS, bovine hyaluronidase (100 U/tumor), ICOVIR5 (1 × 109 vp/tumor), or a combination of ICOVIR5 (1 × 109 vp/tumor) and PH20 hyaluronidase (100 U/tumor).
To evaluate the intratumoral efficacy of viruses expressing the PH20 hyaluronidase, mice-bearing established SkMel-28 tumors (100–150 mm3) received a single intratumoral injection of PBS or 1 × 109 vp of each virus. To evaluate systemic efficacy, subcutaneous SkMel-28 melanoma or NP18 pancreatic tumors were established by injection of 5 × 106 cells into the flanks of 6-week-old male Balb/C nu/nu mice. Once tumors reached the desired mean tumor volume (100 mm3 for SkMel-28 tumors and 60 mm3 for NP18 tumors), mice were randomly distributed into treatment groups (n = 10 tumors per group) and treated with a single intravenous injection of PBS or 5 × 1010 vp of ICOVIR15 or ICOVIR17 given via tail vein.
In all animal experiments, tumor progression and morbidity status were monitored three times weekly. Tumor volume and the percentage of tumor growth were calculated as previously described.16 The two-tailed Student's t-test was used to compare the differences in tumor volume and tumor progression in mice in each cohort.
At the desired time points, animals from each group were euthanized, and tumors were collected and cut into halves. One half was frozen in O.C.T. and the other half was fixed in 4% formaldehyde for 24 hours and embedded in paraffin.
In vivo toxicity studies. Female 5-week-old immunocompetent Syrian (golden) hamsters (Mesocricetus auratus) were used. A single dose of 4 × 1011 vp of ICOVIR15 and ICOVIR17 was administered intravenously into the cephalic vein on day 0 in a volume of 300 µl of PBS. The control group was injected with an equal volume of PBS. Body weights were measured at days 0, 3, and 5 after administration. At these times, animals were examined for clinical signs of toxicity. On day 5, Hamsters were killed, and whole blood and serum samples were collected through cardiac puncture. Clinical biochemistry of transaminase levels and hematological determinations were performed by the Clinical Biochemistry and Hematological Services of the Veterinary Faculty at the Autonomous University of Barcelona. Hamster livers were collected, and portions were fixed in 4% formaldehyde for 24 hours or frozen in O.C.T.
Immunohistochemistry. Adenovirus late protein immunofluorescence was performed on O.C.T.-embedded tumor sections, as previously described.16,29 Images of sections were obtained on an Olympus BX60 fluorescence microscope (Olympus, Tokyo, Japan). To quantify adenovirus positive replication areas, five random fields in viable tissue zones for each tumor were captured at ×100 magnification. Quantification of percentage of stained areas was done using ImageJ software. For immunohistochemical analysis, paraffin-embedded sections of SKMel-28 tumors were treated with an antiadenovirus antibody (Ab6982; Abcam, Cambridge, UK). Immunohistochemical staining was performed with EnVision (Dako, Hamburg, Germany), according to the manufacturer's instructions.
Histochemical staining of HA. Paraffin-embedded blocks were cut into 4 µm thick sections. Sections were deparaffinized, and endogenous peroxidase activity was blocked by incubation for 30 minutes in 0.3% H2O2 in methanol. After rehydration, sections were blocked for 30 minutes with 10% normal goat serum diluted in PBS. For HA staining, the slides were incubated with 5 µg/ml of a biotinylated hyaluronic acid binding protein (HABP-b; Seikagaku, Tokyo, Japan) overnight at 4 °C. The specificity of HA staining was tested by pretreating some samples with 20 U/ml of bovine testes hyaluronidase (Sigma) at 37 °C for 1 hour, prior to the addition of the HABP-b. After incubation with HABP-b, the slides were washed in PBS and treated with avidin–biotin–peroxidase kit (ABC KIT PK-4000; Vectastain, Burlingame, CA). After washings, sections were developed with DAB (Dako Laboratories, Glostrup, Denmark), and counterstained with hematoxylin. To quantify HA positive areas, five random fields in viable tissue zones for each tumor (n = 4) were captured at ×400 magnification. Quantification of percentage of stained areas was done using ImageJ software.
Acknowledgments
We thank Blanca Luena for her technical assistance in animal experiments; Anna Bassols for valuable advice (Universitat Autonoma de Barcelona, Barcelona, Spain); Laia Miquel-Serra and Daniel Hernandez from the Universitat Autonoma de Barcelona, Barcelona, Spain and Felip Vilardell from Hospital Arnau de Vilanova, Lleida, Spain for their technical assistance; and Lynda Coughlan (University of Glasgow, Glasgow, UK) for extensive revision of this manuscript. S.G. and J.J.R. were supported by a predoctoral fellowship (FI) granted by the Generalitat de Catalunya. This work was supported by a grant from the Spanish Ministry of Education and Science, BIO2005-08682-C03-02/01 and received partial support from the Generalitat de Catalunya SGR0500008 and 200556R00066, and the Theradpox contract LSHB-CT-2005-018700 from the European Commission. No conflict of interest from any of the authors exists.
REFERENCES
- Kirn D. Clinical research results with dl1520 (Onyx-015), a replication-selective adenovirus for the treatment of cancer: what have we learned. Gene Ther. 2001;8:89–98. doi: 10.1038/sj.gt.3301377. [DOI] [PubMed] [Google Scholar]
- Nemunaitis J, Senzer N, Sarmiento S, Zhang YA, Arzaga R, Sands B, et al. A phase I trial of intravenous infusion of ONYX-015 and Enbrel in solid tumor patients. Cancer Gene Ther. 2007;14:885–893. doi: 10.1038/sj.cgt.7701080. [DOI] [PubMed] [Google Scholar]
- Reid T, Warren R., and, Kirn D. Intravascular adenoviral agents in cancer patients: lessons from clinical trials. Cancer Gene Ther. 2002;9:979–986. doi: 10.1038/sj.cgt.7700539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang SH, Wientjes MG, Lu D., and, Au JL. Drug delivery and transport to solid tumors. Pharm Res. 2003;20:1337–1350. doi: 10.1023/a:1025785505977. [DOI] [PubMed] [Google Scholar]
- Kuriyama N, Kuriyama H, Julin CM, Lamborn KR., and, Israel MA. Protease pretreatment increases the efficacy of adenovirus-mediated gene therapy for the treatment of an experimental glioblastoma model. Cancer Res. 2001;61:1805–1809. [PubMed] [Google Scholar]
- Kuriyama N, Kuriyama H, Julin CM, Lamborn K., and, Israel MA. Pretreatment with protease is a useful experimental strategy for enhancing adenovirus-mediated cancer gene therapy. Hum Gene Ther. 2000;11:2219–2230. doi: 10.1089/104303400750035744. [DOI] [PubMed] [Google Scholar]
- Ganesh S, Gonzalez Edick M, Idamakanti N, Abramova M, Vanroey M, Robinson M, et al. Relaxin-expressing, fiber chimeric oncolytic adenovirus prolongs survival of tumor-bearing mice. Cancer Res. 2007;67:4399–4407. doi: 10.1158/0008-5472.CAN-06-4260. [DOI] [PubMed] [Google Scholar]
- Kim JH, Lee YS, Kim H, Huang JH, Yoon AR., and, Yun CO. Relaxin expression from tumor-targeting adenoviruses and its intratumoral spread, apoptosis induction, and efficacy. J Natl Cancer Inst. 2006;98:1482–1493. doi: 10.1093/jnci/djj397. [DOI] [PubMed] [Google Scholar]
- Toole BP. Hyaluronan: from extracellular glue to pericellular cue. Nat Rev Cancer. 2004;4:528–539. doi: 10.1038/nrc1391. [DOI] [PubMed] [Google Scholar]
- Baumgartner G, Gomar-Höss C, Sakr L, Ulsperger E., and, Wogritsch C. The impact of extracellular matrix on the chemoresistance of solid tumors–experimental and clinical results of hyaluronidase as additive to cytostatic chemotherapy. Cancer Lett. 1998;131:85–99. [PubMed] [Google Scholar]
- Desoize B., and, Jardillier J. Multicellular resistance: a paradigm for clinical resistance. Crit Rev Oncol Hematol. 2000;36:193–207. doi: 10.1016/s1040-8428(00)00086-x. [DOI] [PubMed] [Google Scholar]
- Brekken C, Bruland ØS., and, de Lange Davies C. Interstitial fluid pressure in human osteosarcoma xenografts: significance of implantation site and the response to intratumoral injection of hyaluronidase. Anticancer Res. 2000;20 5B:3503–3512. [PubMed] [Google Scholar]
- Shuster S, Frost GI, Csoka AB, Formby B., and, Stern R. Hyaluronidase reduces human breast cancer xenografts in SCID mice. Int J Cancer. 2002;102:192–197. doi: 10.1002/ijc.10668. [DOI] [PubMed] [Google Scholar]
- Ganesh S, Gonzalez-Edick M, Gibbons D, Van Roey M., and, Jooss K. Intratumoral coadministration of hyaluronidase enzyme and oncolytic adenoviruses enhances virus potency in metastatic tumor models. Clin Cancer Res. 2008;14:3933–3941. doi: 10.1158/1078-0432.CCR-07-4732. [DOI] [PubMed] [Google Scholar]
- Cherr GN, Yudin AI., and, Overstreet JW. The dual functions of GPI-anchored PH-20: hyaluronidase and intracellular signaling. Matrix Biol. 2001;20:515–525. doi: 10.1016/s0945-053x(01)00171-8. [DOI] [PubMed] [Google Scholar]
- Cascallo M, Alonso MM, Rojas JJ, Perez-Gimenez A, Fueyo J., and, Alemany R. Systemic toxicity-efficacy profile of ICOVIR-5, a potent and selective oncolytic adenovirus based on the pRB pathway. Mol Ther. 2007;15:1607–1615. doi: 10.1038/sj.mt.6300239. [DOI] [PubMed] [Google Scholar]
- Bruera E, Neumann CM, Pituskin E, Calder K., and, Hanson J. A randomized controlled trial of local injections of hyaluronidase versus placebo in cancer patients receiving subcutaneous hydration. Ann Oncol. 1999;10:1255–1258. doi: 10.1023/a:1008331727535. [DOI] [PubMed] [Google Scholar]
- Menon P, Smith KJ, Crittenden J., and, Skelton H. Adjuvant therapy with hyaluronidase prior to excision of dermatofibrosarcoma protuberans. Dermatol Surg. 1999;25:205–209. doi: 10.1046/j.1524-4725.1999.08148.x. [DOI] [PubMed] [Google Scholar]
- Spruss T, Bernhardt G, Schönenberger H., and, Schiess W. Hyaluronidase significantly enhances the efficacy of regional vinblastine chemotherapy of malignant melanoma. J Cancer Res Clin Oncol. 1995;121:193–202. doi: 10.1007/BF01366962. [DOI] [PubMed] [Google Scholar]
- Smith KJ, Skelton HG, Turiansky G., and, Wagner KF. Hyaluronidase enhances the therapeutic effect of vinblastine in intralesional treatment of Kaposi's sarcoma. Military Medical Consortium for the Advancement of Retroviral Research (MMCARR) J Am Acad Dermatol. 1997;36 2 Pt 1:239–242. doi: 10.1016/s0190-9622(97)70288-3. [DOI] [PubMed] [Google Scholar]
- Klocker J, Sabitzer H, Raunik W, Wieser S., and, Schumer J. Combined application of cisplatin, vindesine, hyaluronidase and radiation for treatment of advanced squamous cell carcinoma of the head and neck. Am J Clin Oncol. 1995;18:425–428. doi: 10.1097/00000421-199510000-00013. [DOI] [PubMed] [Google Scholar]
- Bookbinder LH, Hofer A, Haller MF, Zepeda ML, Keller GA, Lim JE, et al. A recombinant human enzyme for enhanced interstitial transport of therapeutics. J Control Release. 2006;114:230–241. doi: 10.1016/j.jconrel.2006.05.027. [DOI] [PubMed] [Google Scholar]
- Lokeshwar VB, Cerwinka WH, Isoyama T., and, Lokeshwar BL. HYAL1 hyaluronidase in prostate cancer: a tumor promoter and suppressor. Cancer Res. 2005;65:7782–7789. doi: 10.1158/0008-5472.CAN-05-1022. [DOI] [PubMed] [Google Scholar]
- Chu RL, Post DE, Khuri FR., and, Van Meir EG. Use of replicating oncolytic adenoviruses in combination therapy for cancer. Clin Cancer Res. 2004;10:5299–5312. doi: 10.1158/1078-0432.CCR-0349-03. [DOI] [PubMed] [Google Scholar]
- Villanueva A, García C, Paules AB, Vicente M, Megías M, Reyes G, et al. Disruption of the antiproliferative TGF-beta signaling pathways in human pancreatic cancer cells. Oncogene. 1998;17:1969–1978. doi: 10.1038/sj.onc.1202118. [DOI] [PubMed] [Google Scholar]
- Serra M, Rabanal RM, Miquel L, Domenzain C., and, Bassols A. Differential expression of CD44 in canine melanocytic tumours. J Comp Pathol. 2004;130:171–180. doi: 10.1016/j.jcpa.2003.10.006. [DOI] [PubMed] [Google Scholar]
- Cascante A, Abate-Daga D, Garcia-Rodríguez L, González JR, Alemany R., and, Fillat C. GCV modulates the antitumoural efficacy of a replicative adenovirus expressing the Tat8-TK as a late gene in a pancreatic tumour model. Gene Ther. 2007;14:1471–1480. doi: 10.1038/sj.gt.3303008. [DOI] [PubMed] [Google Scholar]
- Dmitriev I, Krasnykh V, Miller CR, Wang M, Kashentseva E, Mikheeva G, et al. An adenovirus vector with genetically modified fibers demonstrates expanded tropism via utilization of a coxsackievirus and adenovirus receptor-independent cell entry mechanism. J Virol. 1998;72:9706–9713. doi: 10.1128/jvi.72.12.9706-9713.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cascallo M, Gros A, Bayo N, Serrano T, Capella G., and, Alemany R. Deletion of VAI and VAII RNA genes in the design of oncolytic adenoviruses. Hum Gene Ther. 2006;17:929–940. doi: 10.1089/hum.2006.17.929. [DOI] [PubMed] [Google Scholar]
- Ikegami-Kawai M., and, Takahashi T. Microanalysis of hyaluronan oligosaccharides by polyacrylamide gel electrophoresis and its application to assay of hyaluronidase activity. Anal Biochem. 2002;311:157–165. doi: 10.1016/s0003-2697(02)00425-6. [DOI] [PubMed] [Google Scholar]