Abstract
Introduction
We wanted to assess the importance of the levels of anti-citrullinated peptide antibody (anti-CCP) and immunoglobulin M (IgM) rheumatoid factor (RF) in predicting development of persistent arthritis from undifferentiated arthritis (UA), and to investigate whether there is an added predictive value for persistent arthritis in testing for both anti-CCP and IgM RF.
Methods
Patients with UA (exclusion of definite non-rheumatoid arthritis (RA) diagnoses) included in the Norwegian very early arthritis clinic were assessed for development of persistent arthritic disease. The effect of antibody level on the likelihood of persistent arthritis was investigated, and the sensitivity and specificity for persistent arthritis for anti-CCP and IgM RF, separately and combined, was determined.
Results
A total of 376 UA patients were included (median arthritis duration 32 days). 59 (15.7%) patients were IgM RF positive, and 62 (16.5%) anti-CCP positive. One hundred, seventy-four (46.3%) had persistent disease after one year. Overlap of anti-CCP and IgM RF positivity was 58%. Sensitivity/specificity for persistent arthritis was 28/95% for IgM RF alone, 30/95% for anti-CCP alone, and 37/92% for positivity of both anti-CCP and IgM RF. The likelihood for persistent disease increased with increasing levels of both anti-CCP and IgM RF.
Conclusions
The likelihood of developing persistent arthritis in UA patients increases with the level of anti-CCP and IgM RF. Testing both anti-CCP and IgM RF has added predictive value in UA patients. This study suggests that antibody level should be taken into account when making risk assessments in patients with UA.
Introduction
Rheumatoid factor (RF) has traditionally been regarded as the main serologic marker in inflammatory arthritis [1,2]. In recent years anti-citrullinated protein antibodies (ACPA), most commonly measured by assays for antibodies against cyclic citrullinated peptide (anti-CCP), have also been identified as important predictors both for diagnosis and prognosis in rheumatoid arthritis (RA) [3,4]. RF has similar sensitivity as anti-CCP in RA diagnosis but lower specificity [3,5-8], and RF and anti-CCP are both independent predictors of erosive progression [9].
The paradigm of a window of opportunity in the treatment of inflammatory arthritis has raised awareness of seeing patients at the earliest possible stage of disease. The 1987 American Rheumatism Association (ARA) classification criteria for RA [1] do not perform well in early disease [10] and early arthritis is often undifferentiated and may develop into RA [11,12]. A few studies have identified anti-CCP or RF as predictors of persistent arthritis (as opposed to remission of disease) [13-17].
Several questions remain unanswered regarding the predictive role of anti-CCP and RF in patients with early undifferentiated arthritis. What is the optimal cut-off level for defining a positive antibody status? Is a high positive level of anti-CCP or RF more predictive of an unfavourable outcome than a low positive level? What is the added value (if any) of testing for both markers? Regarding the optimal cut-off of anti-CCP level, a recent study on pre-RA sera [18] suggested that lowering thresholds below that of the manufacturer's recommended cut-off level gave more sensitive prediction of future RA development. Increased levels of ACPA are associated with worse radiographic progression and higher disease activity in RA [9,19,20], whereas no such relationship was found in a recent study of prognosis in early arthritis patients [21]. No studies have assessed the predictive value of the levels of anti-CCP and RF in patients with early undifferentiated arthritis.
The objectives of this study were 1) to investigate whether there is an added predictive value for persistent arthritis in testing for both anti-CCP and IgM RF and 2) to assess the predictive performance for persistent arthritis of the level of anti-CCP and RF in patients with arthritis duration <16 weeks.
Materials and methods
Early arthritis clinic
The Norwegian Very Early Arthritis Clinic (NOR-VEAC) study was started in 2004 as a multicenter observational study in the South-Eastern part of Norway. The five participating hospitals serve a region with approximately 1.7 million inhabitants. The cohort includes patients (age 18 to 75) presenting with at least one clinically swollen joint of ≤ 16 weeks duration, and patients are followed longitudinally for two years. One year outcome was used in the present study.
Joint swelling due to trauma, osteoarthritis, crystal arthropathies, and septic arthritis are exclusion diagnoses; if any of these diagnoses are made during follow-up, patients are excluded from further follow-up. The details of the data collection have been described elsewhere [22], and are summarised in Additional file 1. Imaging procedures were not a part of the data collection for the patients included in this analysis. The study was approved by the regional Ethics Board and the Data Inspectorate, and patients gave an informed consent.
Laboratory markers
Erythrocyte sedimentation rate (ESR) and C-reactive protein levels were determined locally at the participating centres. Serum was frozen at baseline and stored at -70°C and used to analyse anti-citrullinated protein antibodies (ACPA) (anti-CCP2, INOVA Diagnostics, Inc.® San Diego, CA, USA) and IgM rheumatoid factor (RF) (in-house enzyme-linked immunosorbent assay) [9]. Analyses of the serologic markers were performed in one batch. Anti-CCP levels were reported in units from 2 to 250. Any level greater than 250 was reported as >250 and analysed as 251, and levels less than 2 were reported as <2 and analysed as 1. Similarly, IgM RF levels were reported in units from 2 to 300. Any level greater than 300 was reported as >300 and analysed as 301, and levels less than 2 were reported as <2 and analysed as 1. The cut-off levels recommended by the central laboratory for positivity of the serologic markers are: Anti-CCP2 ≥ 25 units/ml, IgM RF ≥ 25 units/ml [9].
Patient selection and outcome determination
Patients included before 1 June 2007, who had baseline information about IgM RF and anti-CCP, were eligible for the current analysis. Patients who were judged by the treating rheumatologist to have a definite diagnosis other than RA were excluded from the present analysis, leaving only patients with very early undifferentiated arthritis in the study. Persistent arthritis (the outcome) was defined as presence of joint swelling at one year. For patients lost to follow-up before one year, the last registered follow-up visit was used in a last observation carried forward (LOCF) manner with regard to joint swelling, provided that at least one follow-up visit was recorded. Patients prescribed with disease-modifying anti-rheumatic drug (DMARD) therapy within one year of presentation were also included in the persistent arthritis group. To determine whether it was correct to include these DMARD-starting patients in the persistent arthritis group, baseline characteristics were compared between patients with persistent synovitis (with or without concomitant DMARD prescription) and patients in which DMARDs were prescribed without coexisting persistent arthritis.
Statistical analysis
Means and standard deviations were calculated for continuous variables following a Gaussian distribution, otherwise median values and inter quartile ranges (25th to 75th percentiles) were calculated. Independent samples T-tests/Mann-Whitney-U tests were used for group comparisons where appropriate. Frequencies were calculated for categorical variables and compared using Chi-square tests.
The relationship between anti-CCP as a continuous/dichotomised variable and the outcome (persistent arthritis) was investigated through univariate and subsequently multivariate logistic regression analyses. The multivariate analyses were adjusted for factors previously found to affect the outcome (age, sex, small joint arthritis, functional status according to Health Assessment Questionnaire (HAQ) score, 28-tender joint count, C-reactive protein, morning stiffness >1 hour) [13,16,23]. IgM RF and anti-CCP were assessed as continuous variables with receiver operating characteristic (ROC) curve analysis. ROC curve analysis was also used to investigate whether alternative cut-offs had more optimal combined sensitivity/specificity than the recommended cut-off of 25 units/ml. Likelihood ratios and their 95% confidence intervals were calculated for these alternative cut-offs.
To assess the effect of increasing levels of anti-CCP and IgM RF on the likelihood of developing persistent disease, anti-CCP and IgM RF levels were divided into ordinal categories (based on the recommended cut-off as well as with tertiles/median of antibody positive patients for anti-CCP and IgM RF, respectively). This resulted in two new, categorized variables (anti-CCP: ≤ 25, >25 to 100, >100 to 250, >250 units/ml; IgM RF: ≤ 25, >25 to 75, >75 units/ml), and these variables were entered into separate univariate logistic regression models. Likelihood ratios for each of the categories compared to the reference category (≤ 25 units/ml) were calculated. The prediction models were also tested in each subgroup of patients with persistent disease (persistent synovitis or DMARD prescription alone) as a sensitivity analysis.
Results
Baseline information about anti-CCP and IgM RF was available in 483/572 (84%) patients enrolled in the study. Patients with and without available sera were similar regarding baseline characteristics and outcome (data not shown). Eighty-nine patients were excluded from the analysis because they were diagnosed with a specific rheumatological non-RA condition at the initial assessment (Löfgren's syndrome/sarcoidosis-associated arthritis 39, psoriatic arthritis according to the Moll & Wright criteria for psoriatic arthritis (PsA) [24] 38, gout 10, ulcerative colitis-arthritis 1, polymyalgia rheumatica 1). Further, 18 patients were excluded from analysis because they had no follow-up data recorded, leaving 376 patients with very early undifferentiated inflammatory arthritis for the current analysis.
Baseline characteristics of the 376 patients are presented in Table 1. The baseline data were complete except for some missing data for morning stiffness. Median (interquartile range, IQR) anti-CCP level in the 62 (16.5%) patients who were anti-CCP2 positive (cut-off 25 units/ml) was 205 (76 to 251) units/ml. Fifty-nine (15.7%) patients were IgM RF positive (>25 units/ml), with median (IQR) IgM RF level 69 (44 to 125) units/ml). Twenty-one patients were solely anti-CCP positive and 18 solely RF positive, while 41 patients were positive for both serologic markers. One hundred seventy-four (46.3%) patients had persistent disease after one year (persistent joint swelling 65, DMARD prescribed 52, both 57). In 32 patients the outcome was determined by the LOCF strategy due to missing data (persistent arthritis 9, remission 23). The DMARDs prescribed were distributed as follows: methotrexate 72 (66.1%) patients, sulphasalazine 21 (19.3%), hydroxychloroquine 4 (3.7%), biologics (+ methotrexate) 5 (1.3%), combinations of traditional DMARDs 5 (1.3%), other 2 (0.5%) patients. Patients with and without persistent disease at one year differed significantly with respect to several baseline characteristics (Table 1).
Table 1.
Baseline characteristics of the patients with comparison between patients with/without persistent arthritis at one year
| All patients (n = 376) |
Persistent disease (N = 174) |
Self-limiting disease (N = 202) |
P* | ||
|---|---|---|---|---|---|
| Persistent synovitis (with or without DMARD) (N = 122) |
DMARD prescribed (without persistent synovitis)$ (N = 52) |
||||
| Female gendera | 218 (58.0) | 78 (63.9) | 32 (61.5) | 108 (53.5) | 0.06 |
| Age, yearsb | 46.3 (14.8) | 49.0 (14.9) | 45.6 (17.1) | 44.9 (14.0) | 0.04 |
| Arthritis duration, daysc | 32 (10 to 64) | 50 (21 to 69) | 59 (27 to 76) | 21 (7 to 49) | <0.001 |
| SJC (0 to 68)c | 2 (1 to 5) | 4 (1 to 9) | 4 (2 to 8) | 1 (1 to 3) | <0.001 |
| TJC (0 to 28)c | 1 (1 to 3) | 2 (1 to 6) | 2 (1 to 5) | 1(0 to 2) | <0.001 |
| ESR, mm/hc | 24 (11 to 46) | 27 (10 to 50) | 32 (17 to 51) | 19 (10 to 38) | 0.005 |
| CRP, mg/lc | 15.0 (5.0 to 36.4) | 15 (5 to 35) | 18 (9 to 45) | 14.0 (3.5 to 36.2) | 0.16 |
| IgM RF level, units/mlc | 4 (1 to 10) | 6 (2 to 40) | 6 (2 to 30) | 3 (1 to 7) | <0.001 |
| IgM RF positivea | 59 (15.7) | 36 (29.5) | 13 (25.0) | 10 (5.0) | <0.001 |
| Anti-CCP2 level, units/mlc | 3 (2 to 6) | 4 (2 to 83) | 4 (2 to 25) | 2 (2 to 4) | <0.001 |
| Anti-CCP2 positivea | 62 (16.5) | 39 (32.0) | 13 (25.0) | 10 (5.0) | <0.001 |
| Assessor's global VAS, mmb | 35.8 (20.7) | 39.5 (21.8) | 44.7 (23.0) | 31.3 (18.2) | <0.001 |
| Patient's global VAS, mmb | 53.8 (24.0) | 57.4 (21.9) | 59.5 (26.1) | 50.2 (24.1) | 0.001 |
| Small joint arthritisa | 173 (46.0) | 73 (59.8) | 33 (63.5) | 67 (33.2) | <0.001 |
| Morning stiffness>1 houra | 195 (51.9)§ | 70 (57.9) | 31 (60.8) | 101 (58.0) | 0.04 |
| DAS28b | 4.05 (1.32) | 4.47 (1.35) | 4.61 (1.36) | 3.65 (1.15) | <0.001 |
| HAQ (0-3)b | 0.89 (0.67) | 0.98 (0.64) | 1.07 (0.80) | 0.78 (0.63) | 0.001 |
| Monoarthritisa | 154 (41.0) | 37 (30.3) | 12 (23.1) | 105 (52.0) | <0.001 |
| -of the kneea | 80 (51.9) | 20 (54.1) | 6 (50.0) | 54 (51.4) | 0.85 |
| Fulfilment of 1987 ACR criteriaa† | 70 (18.6) | 37 (30.3) | 16 (30.8) | 17 (8.4) | <0.001 |
ACR: American College of Rheumatology; anti-CCP2: 2nd generation anti-citrullinated peptide antibody; CRP: C-reactive protein; DAS: Disease Activity Score; ESR: erythrocyte sedimentation rate; HAQ: Health Assessment Questionnaire; RF: rheumatoid factor; SJC: swollen joint count; TJC: tender joint count; VAS: visual analogue scale; small joint arthritis, joint swelling in PIP-, MCP- or MTP joint(s).
$No statistical differences between the persistent arthritis group and DMARD group for any of the baseline variables were found.
*Comparing persistent disease group with self-limiting disease group.aCount (%).bMean (standard deviation).cMedian (inter-quartile range).§N = 369.†Modified from the 1987 ARA criteria according to available data (excluding criteria on erosive disease and duration of symptoms).
Associations between persistent disease and the serologic markers
Logistic regression analyses showed significant univariate associations with persistent disease for anti-CCP and IgM RF which were maintained in the multivariate analysis (Table 2). The odds ratios for persistence were almost doubled when both serologic markers were present.
Table 2.
Logistic regression analysis exploring the relationship between anti-CCP/IgM RF and persistent arthritic disease
| Univariate | Multivariate* | |||||
|---|---|---|---|---|---|---|
| estimate | OR (95% CI) |
P | estimate | OR (95% CI) |
P | |
| Anti-CCP2 (units/ml) |
0.013 | 1.013 (1.008 to 1.018) |
<0.001 | 0.008 | 1.008 (1.003 to 1.013) |
0.002 |
| Anti-CCP2+ (>25 units/ml) |
2.102 | 8.184 (4.008 to 16.709) |
<0.001 | 1.272 | 3.567 (1.572 to 8.094) |
0.002 |
| IgM RF (units/ml) | 0.022 | 1.022 (1.012 to 1.033) |
<0.001 | 0.009 | 1.009 (1.001 to 1.017) |
0.026 |
| IgM RF+ (>25 units/ml) | 2.018 | 7.526 (3.677 to 15.407) |
<0.001 | 0.999 | 2.716 (1.189 to 6.202) |
0.018 |
| IgM RF+ and anti-CCP+ | 2.593 | 13.369 (4.658 to 38.369) |
<0.001 | 2.073 | 7.948 (2.659 to 23.752) |
<0.001 |
*Adjusted for age, sex, small joint arthritis (swelling of MTP-, PIP- and/or MCP joints), Health Assessment Questionnaire, 28-tender joint count, C-reactive protein (mg/l), morning stiffness >1 hour, as well as IgM rheumatoid factor (for the analysis with anti-CCP) or anti-CCP (for the analysis with IgM RF), respectively.
The distribution of anti-CCP- and RF positive patients with and without persistent disease is shown in Table 3. Only 58% (37/64) of antibody positive patients with persistent arthritis were positive for both RF and anti-CCP. Testing for only anti-CCP would identify 52 of the 174 patients (sensitivity 30%) with persistent arthritis. Specificity was 95.0%. Testing for only IgM RF would identify slightly fewer patients, 49/174 (sensitivity 28%), with equal specificity as for anti-CCP alone (95.0%). Testing for both markers would identify 64/174 patients (sensitivity 37%). The number of false-positives for anti-CCP and RF combined was 16 patients, yielding a specificity of 92.1% for the and/or combination. A serial measuring strategy starting with measuring IgM RF would not identify 21/80 (26.3%) autoantibody positive patients, and 15/174 (8.6%) of patients with persistent arthritis would be missed.
Table 3.
Distribution of anti-CCP- and IgM RF positive patients with/without persistent arthritis
| Persistent arthritis | Self-limiting disease | Total | |
|---|---|---|---|
| Anti-CCP positive only | 15 | 6 | 21 |
| Anti-CCP and IgM RF positive | 37 | 4 | 41 |
| IgM RF positive only | 12 | 6 | 18 |
| No antibody positivity | 110 | 186 | 296 |
| Total | 174 | 202 | 376 |
ROC curve analysis and alternative cut offs
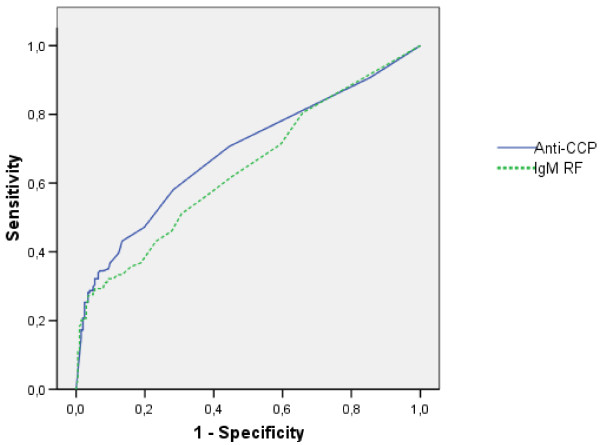
The ROC curve analysis showed moderate discriminatory capacity of anti-CCP (Figure 1), with an area under the curve (AUC) of 0.69. The combination of slightly increased accuracy and positive likelihood ratios remaining above 4 suggested some gain in informative value of lowering the threshold down to about 50% of the recommended cut-off for anti-CCP (Table 4). The ROC curve analysis for IgM RF showed slightly less discriminatory capacity than anti-CCP, with an AUC of 0.64 (Figure 1). Lowering the threshold for defining a positive status for RF resulted in a rather large loss of specificity and positive likelihood ratio (LR) with little gain in sensitivity or negative LR (Table 4).
Figure 1.
Receiver operating characteristic curve analysis. Receiver operating characteristic (ROC) curve analysis investigating the discriminatory capacity for persistent arthritis at one year of baseline antibody levels. Area under the curve (AUC) (95% confidence interval): Anti-CCP: 0.687 (0.633 to 0.742), IgM RF: 0.641 (0.585 to 0.697).
Table 4.
Sensitivity, specificity, and likelihood ratios for persistent arthritis for different minimum cut-off levels for anti-CCP2 and IgM RF
| Cut-off anti-CCP2 (units/ml) |
Sensitivity | Specificity | Accuracy | LR+ (95% CI) |
LR- (95% CI) |
|---|---|---|---|---|---|
| 2 | 0.71 | 0.55 | 0.63 | 1.59 (1.32 to 1.91) | 0.53 (0.41 to 0.69) |
| 3 | 0.58 | 0.72 | 0.65 | 2.06 (1.60 to 2.65) | 0.58 (0.48 to 0.71) |
| 4 | 0.47 | 0.80 | 0.65 | 2.38 (1.73 to 3.27) | 0.66 (0.56 to 0.77) |
| 5 | 0.43 | 0.87 | 0.67 | 3.23 (2.18 to 4.77) | 0.66 (0.57 to 0.77) |
| 10 | 0.35 | 0.93 | 0.66 | 4.98 (2.88 to 8.58) | 0.71 (0.69 to 1.79) |
| 15 | 0.32 | 0.95 | 0.66 | 5.91 (3.20 to 10.92) | 0.72 (0.64 to 0.80) |
| 20 | 0.31 | 0.95 | 0.65 | 5.59 (3.02 to 10.37) | 0.74 (0.66 to 0.82) |
| 25 | 0.30 | 0.95 | 0.64 | 6.04 (3.17 to 11.51) | 0.74 (0.67 to 0.82) |
| Cut-off IgM RF (units/ml) |
Sensitivity | Specificity | Accuracy | LR+ | LR- |
| 2 | 0.71 | 0.41 | 0.55 | 1.20 (1.04 to 1.39) | 0.71 (0.53 to 0.94) |
| 3 | 0.62 | 0.54 | 0.58 | 1.34 (1.13 to 1.65) | 0.70 (0.55 to 0.88) |
| 5 | 0.51 | 0.69 | 0.61 | 1.67 (1.30 to 2.15) | 0.71 (0.59 to 0.84) |
| 7 | 0.43 | 0.77 | 0.61 | 1.85 (1.37 to 2.51) | 0.74 (0.64 to 0.86) |
| 10 | 0.33 | 0.87 | 0.62 | 2.49 (1.66 to 3.76) | 0.77 (0.68 to 0.87) |
| 15 | 0.31 | 0.92 | 0.64 | 3.69 (2.22 to 6.12) | 0.75 (0.68 to 0.84) |
| 20 | 0.29 | 0.95 | 0.64 | 5.28 (2.84 to 9.81) | 0.75 (0.68 to 0.83) |
| 25 | 0.28 | 0.95 | 0.64 | 5.69 (2.97 to 10.89) | 0.76 (0.69 to 0.83) |
CI: confidence interval; LR+: positive likelihood ratio; LR-: negative likelihood ratio.
Effect of increasing antibody levels
When patients were divided into categories based on anti-CCP level (as described in the Methods section) the numbers of patients in each category were as follows: ≤ 25 units/ml (n = 314), >25 to 100 units/ml (n = 19), >100 to 250 units/ml (n = 14), >250 units/ml (n = 29). The likelihood for having persistent arthritic disease increased with increasing levels of anti-CCP (Table 5), suggesting that the level of ACPA can be informative in prediction of persistent arthritis.
Table 5.
Univariate logistic regression for persistent arthritis with anti-CCP/IgM RF as categorical (grouped) variables according to level
| Anti-CCP (units/ml) | OR (95% C.I.) | P | LR+ | LR- | Persistent arthritis patients/patients in category n/N |
|---|---|---|---|---|---|
| ≤ 25 | 1.0 | ref | ref | 122/314 (38.9%) | |
| >25 to 100 | 4.4 (1.6 to 12.5) | 0.005 | 4.1 (1.5 to 11.0) | 0.9 (0.9 to 1.0) | 14/19 (73.7%) |
| >100 to 250 | 9.4 (2.1 to 42.9) | 0.004 | 8.7 (2.0 to 38.2) | 0.9 (0.8 to 1.0) | 12/14 (85.7%) |
| >250 | 13.6 (4.0 to 46.0) | <0.001 | 11.4 (3.5 to 37.0) | 0.8 (0.8 to 0.9) | 26/29 (89.7%) |
| IgM RF (units/ml) | OR (95% C.I.) | P | LR+ | LR- | |
| ≤ 25 | 1.0 | ref | ref | 125/317 (39.4%) | |
| >25 to 75 | 4.6 (2.0 to 10.6) | <0.001 | 4.0 (1.9 to 8.7) | 0.9 (0.8 to 0.9) | 24/32 (75.0%) |
| >75 | 19.2 (4.5 to 82.5) | <0.001 | 16.2 (3.9 to 67.2) | 0.8 (0.8 to 0.9) | 25/27 (92.6%) |
CI: confidence interval; LR+: positive likelihood ratio; LR-: negative likelihood ratio; OR, odds ratio
Similarly, the numbers of patients in each of the categories for IgM RF level were: ≤ 25 units/ml (n = 317), >25 to 75 units/ml (n = 32), >75 units/ml (n = 27). As for anti-CCP, increasing levels of IgM RF were associated with higher positive likelihood of developing persistent arthritis. The negative likelihood ratios for all cut-offs were rather high but consistent with increasing levels. Sensitivity analyses with DMARD start alone or persistent synovitis alone as outcome did not change these results.
Discussion
This study is the first to show that the likelihood of persistent arthritis increases with the levels of IgM RF and anti-CCP. We also demonstrate an added predictive value in testing for both biomarkers in patients with very early undifferentiated arthritis. Several studies have assessed anti-CCP and/or RF as predictors of persistent arthritic disease in patients with undifferentiated arthritis [7,13,15-17] but none of these investigated the influence of antibody level on diagnostic outcome. The association between levels of these serologic markers and outcome also supports the new American College of Rheumatology (ACR)/European League Against Rheumatism (EULAR) classification criteria for RA which provide different points in the classification system according to level of anti-CCP or RF. (Aletaha D, et al. The 2010 American College of Rheumatology/European League Against Rheumatism Classification Criteria for Rheumatoid Arthritis. Arthritis Rheum/Ann Rheum Dis 2010, submitted.)
When working with prediction models, the challenge of circularity arises when the predictive factor is a part of diagnostic or classification criteria. Presence of the factor of interest can bias the physician to label or diagnose a patient (for example, rheumatoid factor or anti-CCP and RA) [13,25]. This challenge can be overcome by using alternative definitions of the outcome, like persistent joint swelling or erosive disease, which are examples of more objective outcomes in early arthritis patients. Their determination relies solely on the presence of physical features, and therefore is less likely to be biased. Moreover, 17 patients who fulfilled the 1987 ARA criteria for RA at baseline turned out to have self-limiting disease after one year (Table 1), which is in line with previous early arthritis studies demonstrating the low specificity of these criteria [10,26-28].
As evidence of the superior specificity and similar sensitivity of anti-CCP as compared to RF as a predictor of an adverse outcome in inflammatory arthritis has increased [3,5-7], the question of whether one should stop testing for RF has been raised [29]. The overlap between RF and anti-CCP positivity may be greater in established RA (93% in a study of 784 RA patients, mean disease duration 18 years) [30], than in early arthritis (50 to 80% in different studies) [5,7,31]. In the present study only 58% (37/64) of antibody positive patients with persistent arthritis were positive for both RF and anti-CCP. Testing for both markers increased sensitivity for persistent arthritis from about 30% to 37%. Our results support that each of these antibodies should be evaluated when making risk assessments in early arthritis, especially if the one first tested is negative.
We also investigated the benefits and drawbacks of lowering the threshold for defining a positive antibody status. Although lowering the threshold resulted in a rather steep loss of specificity for IgM RF, we found some, but limited, added predictive value of lowering the threshold for anti-CCP positivity to 10 units/ml. This is in line with the findings in a recent study [18]. We believe that the clinician should be aware of the potential value also of low levels of anti-CCP when evaluating early arthritis patients, but we cannot recommend a change in the current threshold for positivity based on our findings.
Is there a dose-response relationship between antibody level and likelihood of developing persistent arthritis? Kudo-Tanaka and colleagues reported a higher mean level of anti-CCP2 (Axis-Shield Diagnostics Ltd., Dundee, Scotland, cut-off 4.6 U/ml for positive status) in patients developing RA (167 U/ml) than in patients developing other arthropathies or persistent UA (55 U/ml) in a study of 146 anti-CCP positive UA patients [32]. A Dutch study of patients with arthralgia demonstrated higher median levels of ACPA and IgM RF in patients developing arthritis than in patients who did not [33]. Our findings support that patients with very high levels of anti-CCP or rheumatoid factor are more prone to enter a serious disease course than individuals with low positive levels. This study is the first to demonstrate the effect of levels of anti-CCP and IgM RF on the likelihood of persistent disease, although the numbers of patients with a positive antibody status is moderate and the confidence intervals for the point estimates are wide. A dose-response relationship could support the theory of a pathophysiologic role for anti-CCP antibodies in the development of RA and persistent arthritic diseases, but further studies are needed to shed more light on this complex issue. Importantly, our study supports that presence of high antibody levels give more points than low levels in the new ACR/EULAR classification criteria for RA.
We have shown in this and a previous report [23] that anti-CCP is the most important predictor of an adverse outcome in early arthritis. However, as most patients who developed persistent disease were antibody negative, it is important to bear in mind that early referral and assessment is needed in all patients with significant inflammatory arthritis.
Conclusions
This study of persistent arthritis in patients with arthritis of less than 16 weeks' duration demonstrates that high positive levels of IgM RF and anti-CCP, as compared to low positive levels increase the likelihood of developing chronic arthritic disease, and also suggests an added value of testing for both antibodies in early disease. We conclude that antibody level should be taken into account when making risk assessments in patients with early undifferentiated arthritis.
Abbreviations
ACPA: anti-citrullinated protein antibodies; ACR: American College of Rheumatology; anti-CCP: anti-citrullinated peptide antibody; anti-CCP2: 2nd generation anti-citrullinated peptide antibody; ARA: American Rheumatism Association; AUC: area under the curve; CRP: C-reactive protein; DAS: Disease Activity Score; DMARD: disease-modifying anti-rheumatic drug; ESR: erythrocyte sedimentation rate; HAQ: Health Assessment Questionnaire; IgM: immunoglobuline M; IQR: inter-quartile range; LOCF: last observation carried forward; LR: likelihood ratio; NOR-VEAC: Norwegian Very Early Arthritis Clinic; PsA: psoriatic arthritis; RA: rheumatoid arthritis; RF: rheumatoid factor; ROC: receiver operating characteristic; SJC: swollen joint count; TJC: tender joint count; UA: undifferentiated arthritis; VAS: visual analogue scale
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
MDM performed the statistical analyses and drafted the manuscript, as well as participated in the study design. DvdH contributed to the analysis and interpretation of data and helped draft the manuscript. TU helped to draft the manuscript and participated in the data collection. AJH, HN and KH participated in the study design and data collection. GS participated in the data collection. TKK was the main designer of the study and helped draft the manuscript. All authors read and approved the final manuscript.
Supplementary Material
Summary of data collection NOR-VEAC. MS Word file containing a summary of data collection NOR-VEAC.
Contributor Information
Maria D Mjaavatten, Email: maria_mjaavatten@hotmail.com.
Désirée van der Heijde, Email: d.vanderheijde@kpnplanet.nl.
Till Uhlig, Email: till.uhlig@diakonsyk.no.
Anne J Haugen, Email: Anne.Julsrud.Haugen@so-hf.no.
Halvor Nygaard, Email: halvor.nygaard@revmatismesykehuset.no.
Göran Sidenvall, Email: goran.sidenvall@sykehuset-innlandet.no.
Knut Helgetveit, Email: knut.helgetveit@mhh.no.
Tore K Kvien, Email: t.k.kvien@medisin.uio.no.
Acknowledgements
The authors thank the patients for participating in this study and the local rheumatology staff for data collection. We would also like to thank Per Ivar Gaarder and Gro Jaaberg Talgø at the Department of Immunology and Transfusion Medicine, Ullevål University Hospital for performing the analyses of IgM RF and anti-CCP, and Inova Inc. for providing the kits for analysing anti-CCP. We are grateful to Inge C. Olsen for helpful discussions regarding the statistical analyses. Dr. Mjaavatten's work was supported by a grant from the South-Eastern Norway Regional Health Authority. The NOR-VEAC study was funded by the Norwegian Foundation for Health and Rehabilitation and the South-Eastern Norway Regional Health Authority.
References
- Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, Healey LA, Kaplan SR, Liang MH, Luthra HS. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31:315–324. doi: 10.1002/art.1780310302. [DOI] [PubMed] [Google Scholar]
- Scott DL. Prognostic factors in early rheumatoid arthritis. Rheumatology (Oxford) 2000;39:24–29. doi: 10.1093/oxfordjournals.rheumatology.a031490. [DOI] [PubMed] [Google Scholar]
- Avouac J, Gossec L, Dougados M. Diagnostic and predictive value of anti-cyclic citrullinated protein antibodies in rheumatoid arthritis: a systematic literature review. Ann Rheum Dis. 2006;65:845–851. doi: 10.1136/ard.2006.051391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helm-van Mil AH van der, le Cessie S, van Dongen H, Breedveld FC, Toes RE, Huizinga TW. A prediction rule for disease outcome in patients with recent-onset undifferentiated arthritis: how to guide individual treatment decisions. Arthritis Rheum. 2007;56:433–440. doi: 10.1002/art.22380. [DOI] [PubMed] [Google Scholar]
- Jansen AL, Horst-Bruinsma IE van der, van Schaardenburg D, Stadt RJ van de, de Koning MH, Dijkmans BA. Rheumatoid factor and antibodies to cyclic citrullinated peptide differentiate rheumatoid arthritis from undifferentiated polyarthritis in patients with early arthritis. J Rheumatol. 2002;29:2074–2076. [PubMed] [Google Scholar]
- Schellekens GA, Visser H, de Jong BA, Hoogen FH van den, Hazes JM, Breedveld FC, van Venrooij WJ. The diagnostic properties of rheumatoid arthritis antibodies recognizing a cyclic citrullinated peptide. Arthritis Rheum. 2000;43:155–163. doi: 10.1002/1529-0131(200001)43:1<155::AID-ANR20>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- van Gaalen FA, Linn-Rasker SP, van Venrooij WJ, de Jong BA, Breedveld FC, Verweij CL, Toes RE, Huizinga TW. Autoantibodies to cyclic citrullinated peptides predict progression to rheumatoid arthritis in patients with undifferentiated arthritis: a prospective cohort study. Arthritis Rheum. 2004;50:709–715. doi: 10.1002/art.20044. [DOI] [PubMed] [Google Scholar]
- Nishimura K, Sugiyama D, Kogata Y, Tsuji G, Nakazawa T, Kawano S, Saigo K, Morinobu A, Koshiba M, Kuntz KM, Kamae I, Kumagai S. Meta-analysis: diagnostic accuracy of anti-cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis. Ann Intern Med. 2007;146:797–808. doi: 10.7326/0003-4819-146-11-200706050-00008. [DOI] [PubMed] [Google Scholar]
- Syversen SW, Gaarder PI, Goll GL, Ødegård S, Haavardsholm EA, Mowinckel P, Heijde DM van der, Landewe R, Kvien TK. High anti-cyclic citrullinated peptide levels and an algorithm of four variables predict radiographic progression in patients with rheumatoid arthritis: results from a 10-year longitudinal study. Ann Rheum Dis. 2008;67:212–217. doi: 10.1136/ard.2006.068247. [DOI] [PubMed] [Google Scholar]
- Banal F, Dougados M, Combescure C, Gossec L. Sensitivity and specificity of the American College of Rheumatology 1987 criteria for the diagnosis of rheumatoid arthritis according to disease duration: a systematic literature review and meta-analysis. Ann Rheum Dis. 2009;68:1184–1191. doi: 10.1136/ard.2008.093187. [DOI] [PubMed] [Google Scholar]
- van Dongen H, van Aken J, Lard LR, Visser K, Ronday HK, Hulsmans HM, Speyer I, Westedt ML, Peeters AJ, Allaart CF, Toes RE, Breedveld FC, Huizinga TW. Efficacy of methotrexate treatment in patients with probable rheumatoid arthritis: a double-blind, randomized, placebo-controlled trial. Arthritis Rheum. 2007;56:1424–1432. doi: 10.1002/art.22525. [DOI] [PubMed] [Google Scholar]
- Wiles N, Symmons DP, Harrison B, Barrett E, Barrett JH, Scott DG, Silman AJ. Estimating the incidence of rheumatoid arthritis: trying to hit a moving target? Arthritis Rheum. 1999;42:1339–1346. doi: 10.1002/1529-0131(199907)42:7<1339::AID-ANR6>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- Visser H, le Cessie S, Vos K, Breedveld FC, Hazes JM. How to diagnose rheumatoid arthritis early: a prediction model for persistent (erosive) arthritis. Arthritis Rheum. 2002;46:357–365. doi: 10.1002/art.10117. [DOI] [PubMed] [Google Scholar]
- El Miedany Y, Youssef S, Mehanna AN, El Gaafary M. Development of a scoring system for assessment of outcome of early undifferentiated inflammatory synovitis. Joint Bone Spine. 2008;75:155–162. doi: 10.1016/j.jbspin.2007.04.021. [DOI] [PubMed] [Google Scholar]
- Green M, Marzo-Ortega H, McGonagle D, Wakefield R, Proudman S, Conaghan P, Gooi J, Emery P. Persistence of mild, early inflammatory arthritis: the importance of disease duration, rheumatoid factor, and the shared epitope. Arthritis Rheum. 1999;42:2184–2188. doi: 10.1002/1529-0131(199910)42:10<2184::AID-ANR20>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- Harrison B, Symmons D. Early inflammatory polyarthritis: results from the Norfolk Arthritis Register with a review of the literature. II. Outcome at three years. Rheumatology (Oxford) 2000;39:939–949. doi: 10.1093/rheumatology/39.9.939. [DOI] [PubMed] [Google Scholar]
- Tunn EJ, Bacon PA. Differentiating persistent from self-limiting symmetrical synovitis in an early arthritis clinic. Br J Rheumatol. 1993;32:97–103. doi: 10.1093/rheumatology/32.2.97. [DOI] [PubMed] [Google Scholar]
- Chibnik LB, Mandl LA, Costenbader KH, Schur PH, Karlson EW. Comparison of threshold cutpoints and continuous measures of anti-cyclic citrullinated peptide antibodies in predicting future rheumatoid arthritis. J Rheumatol. 2009;36:706–711. doi: 10.3899/jrheum.080895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rönnelid J, Wick MC, Lampa J, Lindblad S, Nordmark B, Klareskog L, van Vollenhoven RF. Longitudinal analysis of citrullinated protein/peptide antibodies (anti-CP) during 5 year follow up in early rheumatoid arthritis: anti-CP status predicts worse disease activity and greater radiological progression. Ann Rheum Dis. 2005;64:1744–1749. doi: 10.1136/ard.2004.033571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Syversen SW, Goll GL, Heijde DM van der, Landewe R, Lie BA, Ødegård S, Uhlig T, Gaarder PI, Kvien TK. Prediction of radiographic progression in rheumatoid arthritis and the role of antibodies against mutated citrullinated vimentin: results from a 10-year prospective study. Ann Rheum Dis. 2010;69:345–351. doi: 10.1136/ard.2009.113092. [DOI] [PubMed] [Google Scholar]
- Ursum J, Bos WH, van Dillen N, Dijkmans BA, van Schaardenburg D. Levels of anti-citrullinated protein antibodies and IgM rheumatoid factor are not associated with outcome in early arthritis patients: a cohort study. Arthritis Res Ther. 2010;12:R8. doi: 10.1186/ar2907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mjaavatten MD, Haugen AJ, Helgetveit K, Nygaard H, Sidenvall G, Uhlig T, Kvien TK. Pattern of joint involvement and other disease characteristics in 634 patients with arthritis of less than 16 weeks' duration. J Rheumatol. 2009;36:1401–1406. doi: 10.3899/jrheum.081217. [DOI] [PubMed] [Google Scholar]
- Mjaavatten MD, Uhlig T, Haugen AJ, Nygaard H, Sidenvall G, Helgetveit K, Kvien TK. Positive anti-citrullinated protein antibody status and small joint arthritis are consistent predictors of chronic disease in patients with very early arthritis: results from the NOR-VEAC cohort. Arthritis Res Ther. 2009;11:R146. doi: 10.1186/ar2820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moll JM, Wright V. Psoriatic arthritis. Semin Arthritis Rheum. 1973;3:55–78. doi: 10.1016/0049-0172(73)90035-8. [DOI] [PubMed] [Google Scholar]
- Aletaha D, Huizinga TW. The use of data from early arthritis clinics for clinical research. Best Pract Res Clin Rheumatol. 2009;23:117–123. doi: 10.1016/j.berh.2008.11.008. [DOI] [PubMed] [Google Scholar]
- Harrison BJ, Symmons DP, Barrett EM, Silman AJ. The performance of the 1987 ARA classification criteria for rheumatoid arthritis in a population based cohort of patients with early inflammatory polyarthritis. American Rheumatism Association. J Rheumatol. 1998;25:2324–2330. [PubMed] [Google Scholar]
- Kaarela K, Kauppi MJ, Lehtinen KE. The value of the ACR 1987 criteria in very early rheumatoid arthritis. Scand J Rheumatol. 1995;24:279–281. doi: 10.3109/03009749509095163. [DOI] [PubMed] [Google Scholar]
- Saraux A, Berthelot JM, Chales G, Le Henaff C, Thorel JB, Hoang S, Valls I, Devauchelle V, Martin A, Baron D, Pennec Y, Botton E, Mary JY, Le GP, Youinou P. Ability of the American College of Rheumatology 1987 criteria to predict rheumatoid arthritis in patients with early arthritis and classification of these patients two years later. Arthritis Rheum. 2001;44:2485–2491. doi: 10.1002/1529-0131(200111)44:11<2485::AID-ART428>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- Symmons DP. Classification criteria for rheumatoid arthritis--time to abandon rheumatoid factor? Rheumatology (Oxford) 2007;46:725–726. doi: 10.1093/rheumatology/kel418. [DOI] [PubMed] [Google Scholar]
- Mewar D, Coote A, Moore DJ, Marinou I, Keyworth J, Dickson MC, Montgomery DS, Binks MH, Wilson AG. Independent associations of anti-cyclic citrullinated peptide antibodies and rheumatoid factor with radiographic severity of rheumatoid arthritis. Arthritis Res Ther. 2006;8:R128. doi: 10.1186/ar2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klareskog L, Stolt P, Lundberg K, Kallberg H, Bengtsson C, Grunewald J, Rönnelid J, Harris HE, Ulfgren AK, Rantapää-Dahlqvist S, Eklund A, Padyukov L, Alfredsson L. A new model for an etiology of rheumatoid arthritis: smoking may trigger HLA-DR (shared epitope)-restricted immune reactions to autoantigens modified by citrullination. Arthritis Rheum. 2006;54:38–46. doi: 10.1002/art.21575. [DOI] [PubMed] [Google Scholar]
- Kudo-Tanaka E, Ohshima S, Ishii M, Mima T, Matsushita M, Azuma N, Harada Y, Katada Y, Ikeue H, Umeshita-Sasai M, Miyatake K, Saeki Y. Autoantibodies to cyclic citrullinated peptide 2 (CCP2) are superior to other potential diagnostic biomarkers for predicting rheumatoid arthritis in early undifferentiated arthritis. Clin Rheumatol. 2007;26:1627–1633. doi: 10.1007/s10067-007-0558-5. [DOI] [PubMed] [Google Scholar]
- Bos WH, Wolbink GJ, Boers M, Tijhuis GJ, de Vries N, Horst-Bruinsma I van der, Tak PP, Stadt R van de, Laken CJ van der, Dijkmans BA, van SD. Arthritis development in arthralgia patients is strongly associated with anti-citrullinated protein antibody status: a prospective cohort study. Ann Rheum Dis. 2009;69:490–494. doi: 10.1136/ard.2008.105759. Published Online First: 9 April 2009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Summary of data collection NOR-VEAC. MS Word file containing a summary of data collection NOR-VEAC.