Abstract
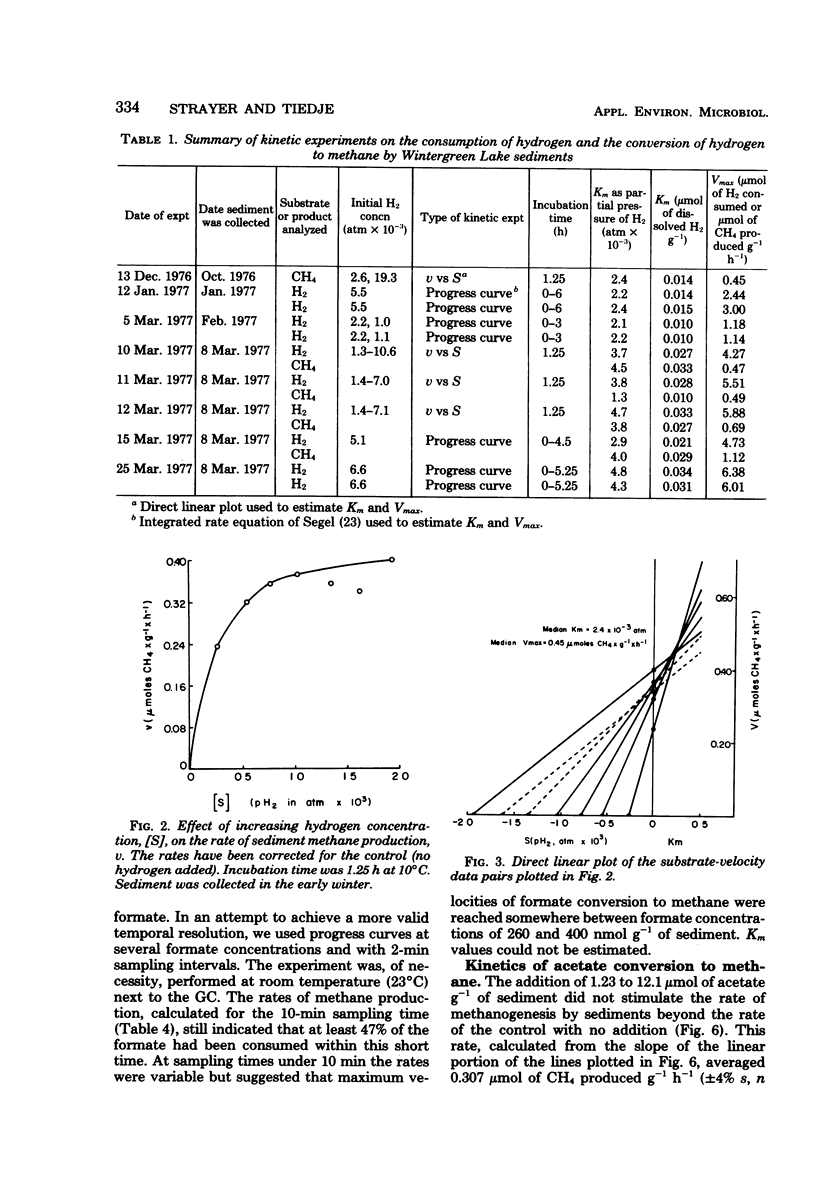
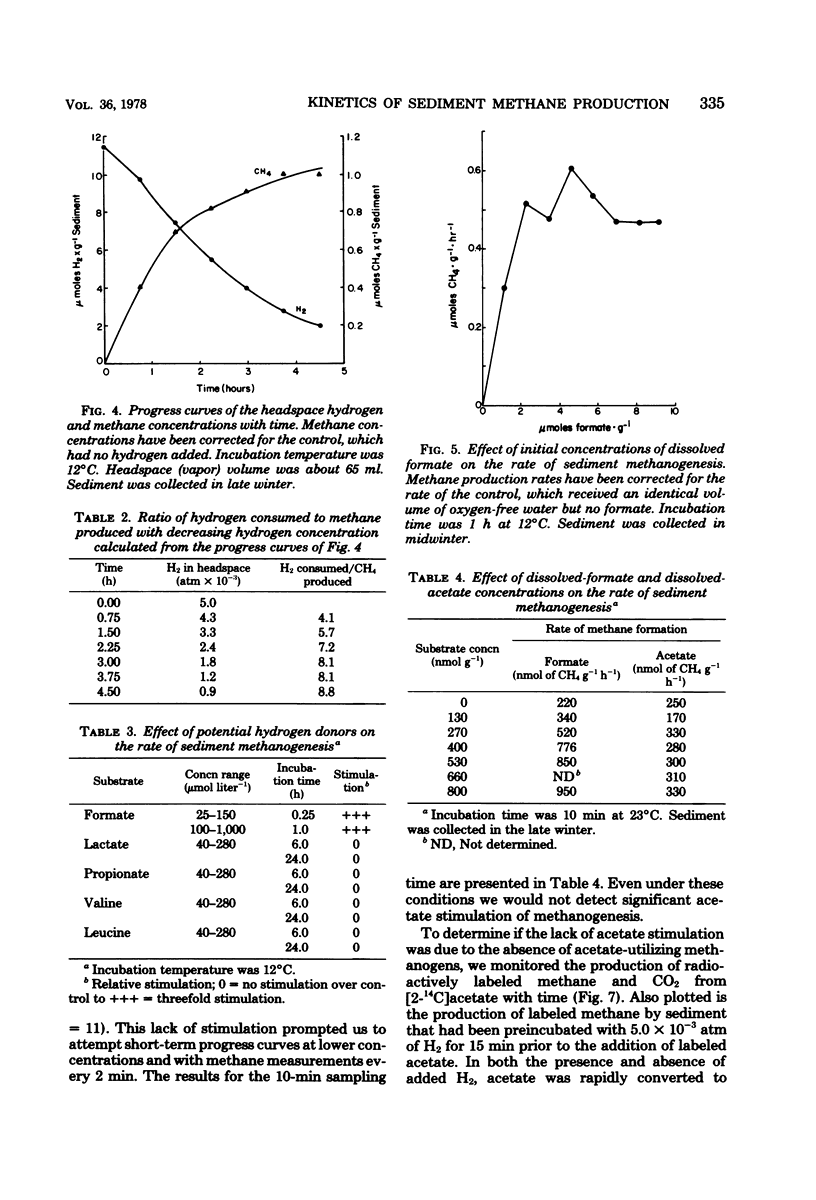
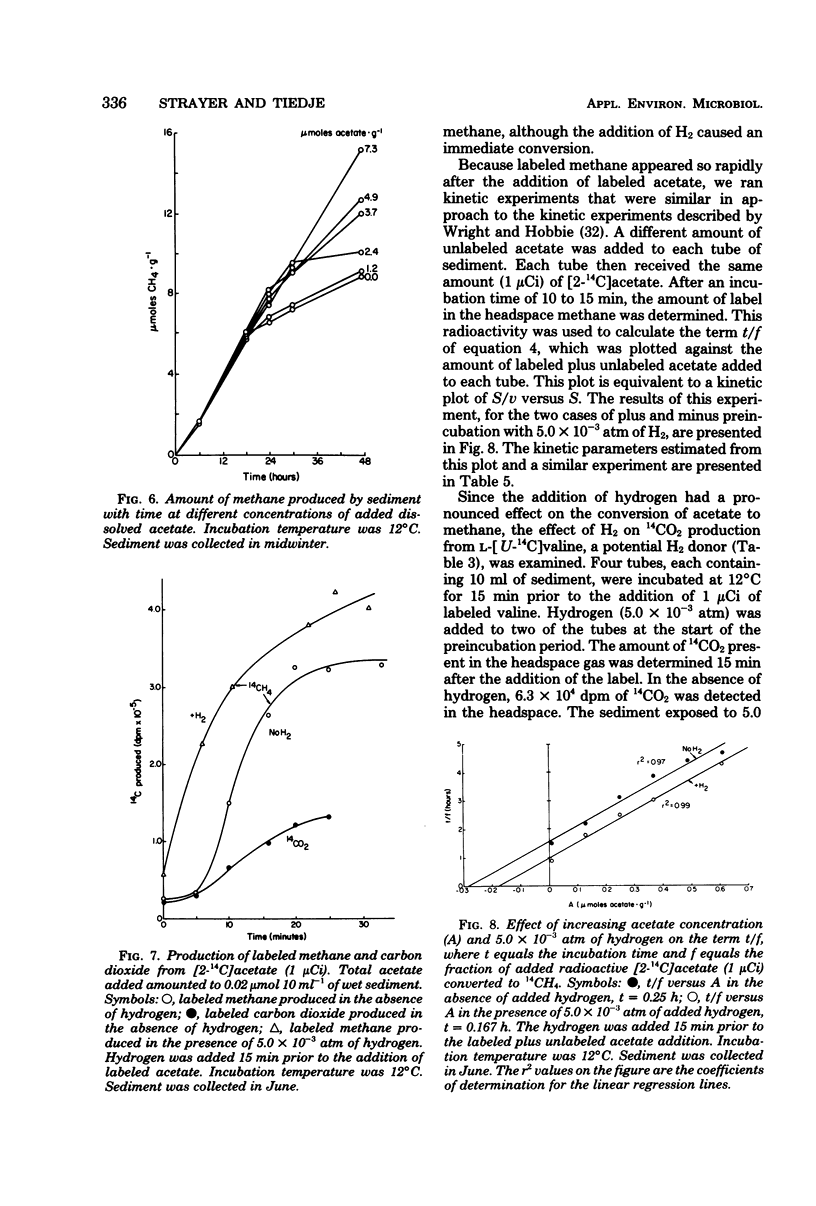
The kinetic parameters Km, Vmax, Tt (turnover time), and v (natural velocity) were determined for H2 and acetate conversion to methane by Wintergreen Lake sediment, using short-term (a few hours) methods and incubation temperatures of 10 to 14°C. Estimates of the Michaelis-Menten constant, Km, for both the consumption of hydrogen and the conversion of hydrogen to methane by sediment microflora averaged about 0.024 μmol g−1 of dry sediment. The maximal velocity, Vmax, averaged 4.8 μmol of H2 g−1 h−1 for hydrogen consumption and 0.64 μmol of CH4 g−1 h−1 for the conversion of hydrogen to methane during the winter. Estimated natural rates of hydrogen consumption and hydrogen conversion to methane could be calculated from the Michaelis-Menten equation and estimates of Km, Vmax, and the in situ dissolved-hydrogen concentration. These results indicate that methane may not be the only fate of hydrogen in the sediment. Among several potential hydrogen donors tested, only formate stimulated the rate of sediment methanogenesis. Formate conversion to methane was so rapid that an accurate estimate of kinetic parameters was not possible. Kinetic experiments using [2-14C]acetate and sediments collected in the summer indicated that acetate was being converted to methane at or near the maximal rate. A minimum natural rate of acetate conversion to methane was estimated to be about 110 nmol of CH4 g−1 h−1, which was 66% of the Vmax (163 nmol of CH4 g−1 h−1). A 15-min preincubation of sediment with 5.0 × 10−3 atm of hydrogen had a pronounced effect on the kinetic parameters for the conversion of acetate to methane. The acetate pool size, expressed as the term Km + Sn (Sn is in situ substrate concentration), decreased by 37% and Tt decreased by 43%. The Vmax remained relatively constant. A preincubation with hydrogen also caused a 37% decrease in the amount of labeled carbon dioxide produced from the metabolism of [U-14C]valine by sediment heterotrophs.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bryant M. P., Campbell L. L., Reddy C. A., Crabill M. R. Growth of desulfovibrio in lactate or ethanol media low in sulfate in association with H2-utilizing methanogenic bacteria. Appl Environ Microbiol. 1977 May;33(5):1162–1169. doi: 10.1128/aem.33.5.1162-1169.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cappenberg T. E. Interrelations between sulfate-reducing and methane-producing bacteria in bottom deposits of a fresh-water lake. I. Field observations. Antonie Van Leeuwenhoek. 1974;40(2):285–295. doi: 10.1007/BF00394387. [DOI] [PubMed] [Google Scholar]
- Cappenberg T. E. Interrelations between sulfate-reducing and methane-producing bacteria in bottom deposits of a fresh-water lake. II. Inhibition experiments. Antonie Van Leeuwenhoek. 1974;40(2):297–306. doi: 10.1007/BF00394388. [DOI] [PubMed] [Google Scholar]
- Cappenberg T. E., Prins R. A. Interrelations between sulfate-reducing and methane-producing bacteria in bottom deposits of a fresh-water lake. 3. Experiments with 14C-labeled substrates. Antonie Van Leeuwenhoek. 1974;40(3):457–469. doi: 10.1007/BF00399358. [DOI] [PubMed] [Google Scholar]
- Chung K. T. Inhibitory effects of H2 on growth of Clostridium cellobioparum. Appl Environ Microbiol. 1976 Mar;31(3):342–348. doi: 10.1128/aem.31.3.342-348.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornish-Bowden A., Eisenthal R. Statistical considerations in the estimation of enzyme kinetic parameters by the direct linear plot andother methods. Biochem J. 1974 Jun;139(3):721–730. doi: 10.1042/bj1390721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenthal R., Cornish-Bowden A. The direct linear plot. A new graphical procedure for estimating enzyme kinetic parameters. Biochem J. 1974 Jun;139(3):715–720. doi: 10.1042/bj1390715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flett R. J., Hamilton R. D., Campbell N. E. Aquatic acetylene-reduction techniques: solutions to several problems. Can J Microbiol. 1976 Jan;22(1):43–51. doi: 10.1139/m76-006. [DOI] [PubMed] [Google Scholar]
- Hungate R. E., Smith W., Bauchop T., Yu I., Rabinowitz J. C. Formate as an intermediate in the bovine rumen fermentation. J Bacteriol. 1970 May;102(2):389–397. doi: 10.1128/jb.102.2.389-397.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iannotti E. L., Kafkewitz D., Wolin M. J., Bryant M. P. Glucose fermentation products in Ruminococcus albus grown in continuous culture with Vibrio succinogenes: changes caused by interspecies transfer of H 2 . J Bacteriol. 1973 Jun;114(3):1231–1240. doi: 10.1128/jb.114.3.1231-1240.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molongoski J. J., Klug M. J. Characterization of anaerobic heterotrophic bacteria isolated from freshwater lake sediments. Appl Environ Microbiol. 1976 Jan;31(1):83–90. doi: 10.1128/aem.31.1.83-90.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohwaki K., Hungate R. E. Hydrogen utilization by clostridia in sewage sludge. Appl Environ Microbiol. 1977 Jun;33(6):1270–1274. doi: 10.1128/aem.33.6.1270-1274.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfennig N., Biebl H. Desulfuromonas acetoxidans gen. nov. and sp. nov., a new anaerobic, sulfur-reducing, acetate-oxidizing bacterium. Arch Microbiol. 1976 Oct 11;110(1):3–12. doi: 10.1007/BF00416962. [DOI] [PubMed] [Google Scholar]
- Reddy C. A., Bryant M. P., Wolin M. J. Characteristics of S organism isolated from Methanobacillus omelianskii. J Bacteriol. 1972 Feb;109(2):539–545. doi: 10.1128/jb.109.2.539-545.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheifinger C. C., Linehan B., Wolin M. J. H2 production by Selenomonas ruminantium in the absence and presence of methanogenic bacteria. Appl Microbiol. 1975 Apr;29(4):480–483. doi: 10.1128/am.29.4.480-483.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith P. H., Mah R. A. Kinetics of acetate metabolism during sludge digestion. Appl Microbiol. 1966 May;14(3):368–371. doi: 10.1128/am.14.3.368-371.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strayer R. F., Tiedje J. M. Application of the fluorescent-antibody technique to the study of a methanogenic bacterium in lake sediments. Appl Environ Microbiol. 1978 Jan;35(1):192–198. doi: 10.1128/aem.35.1.192-198.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weimer P. J., Zeikus J. G. Fermentation of cellulose and cellobiose by Clostridium thermocellum in the absence of Methanobacterium thermoautotrophicum. Appl Environ Microbiol. 1977 Feb;33(2):289–297. doi: 10.1128/aem.33.2.289-297.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Widdel F., Pfennig N. A new anaerobic, sporing, acetate-oxidizing, sulfate-reducing bacterium, Desulfotomaculum (emend.) acetoxidans. Arch Microbiol. 1977 Feb 4;112(1):119–122. doi: 10.1007/BF00446665. [DOI] [PubMed] [Google Scholar]
- Winfrey M. R., Nelson D. R., Klevickis S. C., Zeikus J. G. Association of hydrogen metabolism with methanogenesis in Lake Mendota sediments. Appl Environ Microbiol. 1977 Feb;33(2):312–318. doi: 10.1128/aem.33.2.312-318.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winfrey M. R., Zeikus J. G. Effect of sulfate on carbon and electron flow during microbial methanogenesis in freshwater sediments. Appl Environ Microbiol. 1977 Feb;33(2):275–281. doi: 10.1128/aem.33.2.275-281.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolfe R. S. Microbial formation of methane. Adv Microb Physiol. 1971;6:107–146. doi: 10.1016/s0065-2911(08)60068-5. [DOI] [PubMed] [Google Scholar]
- Wolin M. J. Metabolic interactions among intestinal microorganisms. Am J Clin Nutr. 1974 Nov;27(11):1320–1328. doi: 10.1093/ajcn/27.11.1320. [DOI] [PubMed] [Google Scholar]




