Abstract
The design and synthesis of a multivalent gold nanoparticle therapeutic is presented. SDC-1721, a fragment of the potent HIV inhibitor TAK-779, was synthesized and conjugated to 2.0 nm diameter gold nanoparticles. Free SDC-1721 had no inhibitory effect on HIV infection; however, the (SDC-1721)-gold nanoparticle conjugates displayed activity comparable to that of TAK-779. This result suggests that multivalent presentation of small molecules on gold nanoparticle surfaces can convert inactive drugs into potent therapeutics.
It has been suggested that biological systems exploit multivalency in the synthesis of high-affinity ligands because they allow an organism to take advantage of an existing set of monovalent ligands without the need for evolving completely new molecules for every required function.1 Multivalent displays are ubiquitous in biology and can confer dramatically enhanced affinity. For example, binding between multiple hemagglutinin (HA) ligands of an influenza virus and sialic acid (SA) surface receptors of an erythrocyte during viral infection is estimated to occur with an affinity of 1013 M−1, while the association constant for a single SA–HA interaction is 103 M−1.1
In the development of therapeutics, a multivalent binding strategy could offer many advantages. Synthetic systems that present multiple low-affinity biomolecule binding ligands may prove to be more synthetically accessible and could allow one to tune binding affinity over several orders of magnitude. Moreover, nanoparticles that are comparable in size to proteins and present multiple protein-binding ligands may be effective at disrupting protein/protein interactions that drive disease pathogenesis. Multivalent linear polymers, dendrimers, proteins, and liposomes have successfully targeted pathogenic biomolecule targets.1,2 Of particular note are SA-coated liposomes and dendrimers that bind to HA on the surface of an influenza virus with affinity enhancements of 102–106.1
Particle-based displays of multiple ligands have the additional advantage of creating a high local concentration of binding molecules. Consequently, binding equilibrium between a surface-bound ligand and receptor favors formation of more ligand-receptor pairs. For instance, DNA hybridization is thermodynamically favored by 1 order of magnitude if one of the single-stranded DNA sequences is conjugated to a 10 nm diameter gold nanoparticle surface.3,4
The advantages of multivalent therapeutics are illustrated here with the demonstration that gold nanoparticles transform a weakly binding and biologically inactive small molecule into a multivalent conjugate that effectively inhibits HIV-1 fusion to human T cells. We employed 2.0 nm diameter, mercaptobenzoic acid modified gold particles as a nanoscale platform to construct our multivalent therapeutic. These particles have a proposed empirical formula of [Au144(SC6H4COOH)52] and are related to the [Au102(SC6H4-COOH)44] monolayer-protected gold nanoparticles that were recently characterized by X-ray crystallography.5 Importantly, this class of gold particles is similar to proteins and dendrimers in that they are atomically precise and monodisperse nanoscale molecules.
The 2.0 nm diameter mercaptobenzoic acid coated gold nano-particles were conjugated to SDC-1721, a derivative of TAK-779, a known CCR5 antagonist (Figure 1). CCR5 serves as the principal entry co-receptor for most commonly transmitted strains of HIV-1 and those that predominate in the early years of infection.6 A significant amount of structure–activity relationship (SAR) data has been generated with regard to TAK-779, most notably that the quaternary ammonium salt is essential for high-affinity binding and effective inhibition of HIV fusion. TAK-779 homologues that lack this quaternary ammonium salt are devoid of activity.7 Unfortunately, the quaternary ammonium salt imbues TAK-779 with poor pharmacological properties, such as significant irritation at the injection site, which led to an exhaustive search for alternate small molecule CCR5 antagonists.7 As an alternative approach and proof-of-concept, we synthesized SDC-1721, a TAK-779 homologue that lacks the quaternary ammonium salt moiety, to show that it is possible to conjugate a low-affinity, biologically inactive small molecule to a gold nanoparticle to create biologically active multivalent gold nanoparticle therapeutics. The ability to transform small molecules that are not therapeutically viable into potential therapeutics by simply conjugating them to a gold nanoparticle scaffold could greatly accelerate the discovery of effective new drug formulations.
Figure 1.
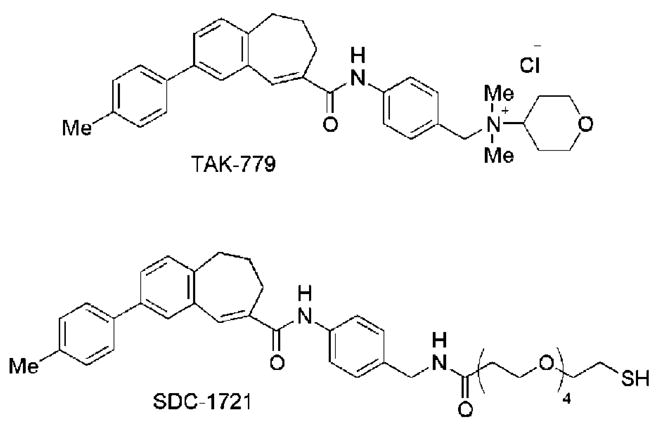
TAK-779 and SDC-1721.
SDC-1721 was synthesized (Supporting Information) using a modification of the synthetic approach to TAK-7798 and coupled to the 2.0 nm diameter gold nanoparticles via a simple ligand exchange reaction. The average number of SDC-1721 molecules per particle was determined to be 12, and the particle size was verified by transmission electron microscopy. Examination of the micrographs revealed no substantial differences in core size or dispersion when compared to the 2.0 nm diameter gold nanoparticles prior to ligand exchange.
To evaluate the antiviral activity of the nanoparticle conjugate, phytohemagglutinin (PHA)-stimulated peripheral blood mono-nuclear cells (PBMCs) were infected with the CCR5-tropic HIV-1 clone JR-CSF in the presence or absence of the test compounds. On day 7 postinfection, supernatants were collected and HIV-1 capsid p24 antigen was measured by ELISA. As shown in Figure 2a, TAK-779 inhibited HIV-1 replication with an IC50 of 10 nM. The IC50 for TAK-779 against four different CCR5-tropic viral isolates ranged from 1.6 to 3.7 nM.9 However, JR-CSF was not one of the viruses tested. With a similar virus, JR-FL, TAK-779 revealed an IC50 of 20 nM.10 Sensitivity to CCR5 entry inhibitors is impacted by receptor expression levels and HIV-1 envelope/receptor affinity, mediated by both cellular and viral determinants.11 Thus, JR-CSF appears to be inherently less sensitive to TAK-779.
Figure 2.
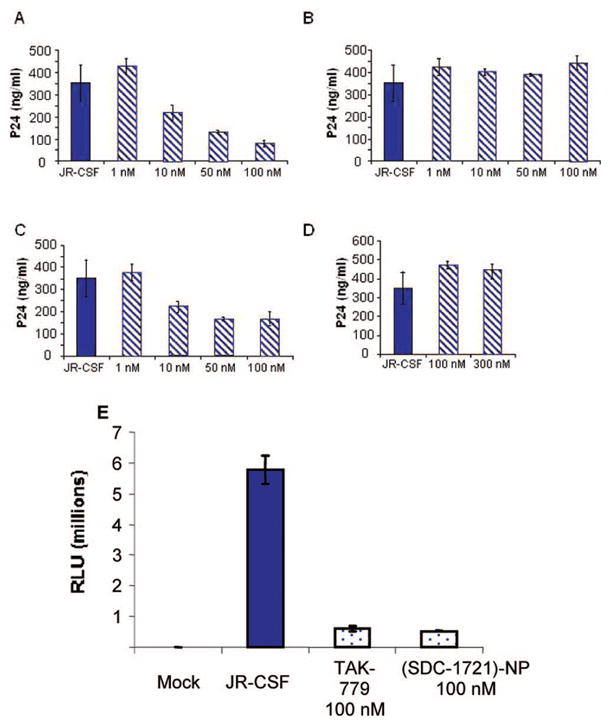
(A–D) Multiple-cycle inhibition assays. (A) TAK-779 HIV-1 inhibition; (B) SDC-1721 HIV-1 inhibition; (C) (SDC-1721)–NP HIV-1 inhibition; (D) GSH–NP HIV-1 inhibition; (E) single-cycle HIV-1 infectivity inhibition with TAK-779 and (SDC-1721)–NP.
We next tested whether, as had been reported, the quaternary ammonium group was essential to TAK-779 activity.7 SDC-1721 did not inhibit viral replication (Figure 2b). Conjugation of SDC-1721 ((SDC-1721)–NP) to gold nanoparticles at an average ratio of 12:1 restored activity with an IC50 of 10 nM (Figure 2c). To ensure that the inhibitory effect was not due to the gold particle, a glutathione-modified gold nanoparticle (GSH–NP) control was tested. The GSH–NP is unable to inhibit viral replication (Figure 2d). In addition, we verified that multivalent display of SDC-1721 on the nanoparticle surface was required for activity by assaying (SDC-1721)–NP conjugates with an average ratio of 0.93/1. This nanoparticle preparation was unable to inhibit viral replication up to 300 μM (Supporting Information). There was no difference in cell viability between samples at these concentrations.
We further verified that inhibition of viral replication was specific for viral entry. This was assayed by performing single-cycle infection experiments using the TZM-bl luciferase reporter gene assay system.12 This cell line is engineered to express high levels of CCR5 and CD4 and contains a luciferase reporter gene under control of the HIV-1 promoter, which is inducible in trans by the viral protein Tat.13 TZM-bl cells were infected with JR-CSF in the presence or absence of the test compounds. Forty-eight hour postinfection, luciferase activity was measured as relative luminescence units (RLU). SDC-1721-modified gold nanoparticles inhibited viral entry as well as TAK-779 (Figure 2e). At 48 h, viral production is insignificant, thus establishing inhibition of viral replication at the stage of viral entry.
In conclusion, we have documented the first application of small-molecule coated gold nanoparticles as effective inhibitors of HIV fusion. The results presented here demonstrate that therapeutically inactive monovalent small organic molecules may be converted into highly active drugs by simply conjugating them to gold nanoparticles.
Supplementary Material
Acknowledgments
This research was funded by NCSU (C.M.), GlaxoSmithKline (T.E.B.), The University of Colorado (D.F.), the NIH (AI045297 to D.M.M., EY017568 to D.F.), and the UNC Chapel Hill CFAR (AI50410) to M.C.B. TZM-bl was obtained through the NIH AIDS Research and Reference Reagent Program, Division of AIDS, NIAID, NIH, from Dr. John C. Kappes, Dr. Xiaoyun Wu, and Tranzyme Inc.
Footnotes
Supporting Information Available: Experimental procedures and characterization for all new compounds; TEM and cell assay procedures. This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Mammen M, Choi SK, Whitesides GM. Angew Chem, Int Ed. 1998;37:2755–2794. doi: 10.1002/(SICI)1521-3773(19981102)37:20<2754::AID-ANIE2754>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- 2.Caruthers SD, Wickline SA, Lanza GM. Curr Opin Biotechnol. 2007;18:26–30. doi: 10.1016/j.copbio.2007.01.006. [DOI] [PubMed] [Google Scholar]
- 3.Nicewarner Pena SR, Raina S, Goodrich GP, Fedoroff NV, Keating CD. J Am Chem Soc. 2002;124:7314–7323. doi: 10.1021/ja0177915. [DOI] [PubMed] [Google Scholar]
- 4.Lytton-Jean AK, Mirkin CA. J Am Chem Soc. 2005;127:12754–12755. doi: 10.1021/ja052255o. [DOI] [PubMed] [Google Scholar]
- 5.Jadzinsky PD, Calero G, Ackerson CJ, Bushnell DA, Kornberg RD. Science. 2007;318:430–433. doi: 10.1126/science.1148624. [DOI] [PubMed] [Google Scholar]
- 6.Moore JP, Kitchen SG, Pugach P, Zack JA. AIDS Res Hum Retroviruses. 2004;20:111–126. doi: 10.1089/088922204322749567. [DOI] [PubMed] [Google Scholar]
- 7.Barbaro G, Scozzafava A, Mastrolorenzo A, Supuran CT. Curr Pharm Des. 2005;11:1805–1843. doi: 10.2174/1381612053764869. [DOI] [PubMed] [Google Scholar]
- 8.Ikemoto T, et al. Org Process Res Dev. 2000;4:520–525. [Google Scholar]
- 9.Baba M, et al. Proc Natl Acad Sci USA. 1999;96:5698–5703. doi: 10.1073/pnas.96.10.5698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Safarian D, Carnec X, Tsamis F, Kajumo F, Dragic T. Virology. 2006;352:477–484. doi: 10.1016/j.virol.2006.05.016. [DOI] [PubMed] [Google Scholar]
- 11.Reeves JD, Gallo SA, Ahmad N, Miamidian JL, Harvey PE, Sharron M, Pöhlmann S, Sfakianos JN, Derdeyn CA, Blumenthal R, Hunter E, Doms RW. Proc Natl Acad Sci USA. 2002;99:16249–16254. doi: 10.1073/pnas.252469399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wei X, Decker JM, Wang S, Hui H, Kappes JC, Xiaoyun Wu X, Salazar-Gonzalez JF, Salazar MG, Kilby JM, Saag MS, Komarova NL, Nowak MA, Hahn BH, Kwong PD, Shaw GM. Nature. 2003;422:307–312. doi: 10.1038/nature01470. [DOI] [PubMed] [Google Scholar]
- 13.Wei X, Decker JM, Liu H, Zhang Z, Arani RB, Kilby JM, Saag MS, Wu X, Shaw GM, Kappe JC. Antimicrob Agents Chemother. 2002;46:1896–1905. doi: 10.1128/AAC.46.6.1896-1905.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


