Abstract
Background
Risk estimation of gastrointestinal stromal tumours (GIST) is based on tumour size and mitotic rate according to the National Institutes of Health consensus classification. The indication for adjuvant treatment of patients with high risk GIST after R0 resection with small molecule inhibitors is still a controversial issue, since these patients represent a highly heterogeneous population. Therefore, additional prognostic indicators are needed. Here, we evaluated the prognostic value of cyclin H expression in GIST.
Methods
In order to identify prognostic factors of GIST we evaluated a single centre cohort of ninety-five GIST patients. First, GISTs were classified with regard to tumour size, mitotic rate and localisation according to the NIH consensus and to three additional suggested risk classifications. Second, Cyclin H expression was analysed.
Results
Of ninety-five patients with GIST (53 female/42 male; median age: 66.78a; range 17-94a) risk classification revealed: 42% high risk, 20% intermediate risk, 23% low risk and 15% very low risk GIST. In patients with high risk GIST, the expression of cyclin H was highly predictive for reduced disease-specific survival (p = 0.038). A combination of cyclin H expression level and high risk classification yielded the strongest prognostic indicator for disease-specific and disease-free survival (p ≤ 0.001). Moreover, in patients with tumour recurrence and/or metastases, cyclin H positivity was significantly associated with reduced disease-specific survival (p = 0.016) regardless of risk-classification.
Conclusion
Our data suggest that, in addition to high risk classification, cyclin H expression might be an indicator for "very-high risk" GIST.
Background
Gastrointestinal stromal tumours (GIST) display a wide range of clinical and pathological features and represent the largest group of mesenchymal tumours of the gastrointestinal tract. They are mostly characterised by a gain-of function mutation of the c-kit gene encoding a receptor tyrosine kinase [1-3]. The original NIH (National Institutes of Health) classification of GIST into four subgroups (very low, low, intermediate or high risk) is based on tumour size and mitotic rate [1] and supplemented by the addition of further parameters [4-7]; (Additional file 1: Table S1). Tumour stage, as well as tumour size, and mitotic rate are relevant markers for clinical outcome of GIST. The combination of large tumour size and/or high mitotic rate is used to identify high risk GIST which is associated with an unfavourable prognosis [1,2]. The identification of genes and gene products correlating with prognosis might have therapeutic implications for further differentiation of high risk GIST in the adjuvant setting. While surgery remains the only curative treatment for GIST, small molecule inhibitors like Imatinib targeting KIT and PDGFR are used as the standard first-line treatment in advanced disease [3,8]. At present, the use of Imatinib in adjuvant treatment is controversial. The suggestion by De Matteo and co-workers generally to use Imanitib in adjuvant therapy of high risk GIST [9,10], has led to pressing demands to introduce additional parameters that help to stratify patients within the high risk GIST group for additional adjuvant therapy concepts [9,10].
Genes involved in cell-cycle regulation, such as cyclins and cyclin-dependent kinases (CDKs), are among such potential markers [11,12]. The impact of deregulation in regard to members of the cyclin-CDK-system on tumourigenesis/tumour progression of GIST has only been evaluated in a few studies [13-15]. There is evidence that a low expression of p27KIP1 (CDKN1B, a cyclin-dependent-kinase-inhibitor) is associated with reduced progression-free survival [16-18]. Furthermore, expression of cyclin A, cyclin B, cyclin D1 and cyclin E seems to be associated with high risk grading but not with clinical outcome [17,19,20]. At present there are no data available regarding the role of cyclin H expression in the progression of GIST. Cyclin H plays a key role in cell cycle regulation by modulating the activity of CDK7 which phosphorylates CDK1, 2, 4 and 6 [21].
In this study, we investigated the expression pattern of cyclin H in a single-centre population of 95 GIST and evaluated its prognostic value, since our gene expression analysis in normal and tumour tissue of a high-risk GIST patient revealed a 10 fold upregulation of cyclin H in tumour tissue.
Methods
Human tissue
Medical records as well as paraffin-embedded and frozen specimens of 95 gastrointestinal stromal tumours were included in the study. The clinocopathological features are outlined in Table 1. Informed consent was obtained from all patients. The study was performed with the permission of the independent local ethics committee of the University of Ulm (No. 90/2006 MuZGi).
Table 1.
Clinicopathologic Features
| Initial symptoms (multiple mentions possible) | n | % of 90 | |
|---|---|---|---|
| No clinical symptoms | 25 | 28% | |
| Pain | 33 | 37% | |
| GI bleeding | 25 | 28% | |
| Anemia | 6 | 7% | |
| Localization of primary tumour | n | % of 95 | |
| Stomach | 58 | 61% | |
| Small bowel | 29 | 31% | |
| Jejunum | 9 | 9% | |
| Ileum | 17 | 18% | |
| Duodenum | 3 | 3% | |
| Colon | 1 | 1% | |
| Esophagus | 2 | 2% | |
| Others (EGIST, etc.) | 5 | 5% | |
| Second neoplasias | n | % of 95 | |
| Total | 30 | 32% | |
| n | % of 30 | ||
| Colorectal cancer | 5 | 17% | |
| Prostate cancer | 4 | 13% | |
| Breast cancer | 3 | 10% | |
| Gastric cancer | 3 | 10% | |
| Myometrial or cervical cancer | 2 | 7% | |
| Renal or urothelian cancer | 2 | 7% | |
| Pancreatic cancer | 1 | 3% | |
| Others | 10 | 33% | |
| Histomorphology | n | % of 92 | |
| Spindle cell GIST | 80 | 87% | |
| Epithelioid/Mixed pattern | 12 | 13% | |
| Mixed pattern | 11 | 12% | |
| Epithelioid | 1 | 1% | |
| Risk of Malignancy (Fletcher et al.) | n (sto/smbo/ot) | n | % of 93 |
| High risk | 21/13/5 | 39 | 42% |
| Intermediate risk | 13/5/1 | 19 | 20% |
| Low risk | 14/7/0 | 21 | 23% |
| Very low risk | 8/4/2 | 14 | 15% |
sto = stomach, smbo = small bowel, ot = others
Quantification and detection of Cyclin H mRNA
In a pilot study, total RNA was isolated from frozen normal jejunal tissue and from a clinically very aggressive tumour relapse which occurred 1.91 years after the initial diagnosis of a jejunal high risk GIST in a 53-year old female patient (clinical data: age at primary tumour diagnosis: 51 years, features of the primary tumour: size: 9.5 cm, MR: 47 per 50 HPFs; Fletcher classification: high risk of malignancy, no Imatinib treatment prior to resection) using the RNAeasy Kit (Qiagen, Germany). Total RNA (2 μg) was reversely transcribed into complementary DNA using the RT2 First Strand Kit (SuperArray Bioscience Corp., USA). Gene profiling was done as described by the manufacturer using the RT2 profiler PCR array for the human p53 signalling pathway (84 genes, SuperArray Bioscience Corp., USA). For CCNH (gene encoding cyclin H), housekeeping gene HPRT1 (hypoxanthine Phosphoribosyltransferase 1) was used for normalisation. The reactions were carried out in a 7500 Fast Real-Time PCR System (Applied Biosystems, USA). The results were analysed with the 7500 Fast System SDS Software 1.4 (Applied Biosystems, USA).
Histological evaluation
Original haematoxylin and eosin-stained tumour sections were used to evaluate cell type features and to determine the mitotic rate in 50 high power fields (HPF, magnification: 40×). GISTs were classified according to Fletcher et al. [1] - (see Table 1). Additional risk estimation was performed for all tumours according to the classifications suggested by Miettinen et al. [7], Hornick et al. [4] and Joensuu [6].
Immunohistochemical analysis
For immunohistochemical analysis the following antibodies were used: anti cyclin H (ab54903, monoclonal, dilution 1:100, abcam, GB), anti KIT (CD117, C-KIT, polyclonal, dilution 1:200, Dako, Glostrup Denmark), anti smooth muscle actin (dilution 1:400, clone 1A4, Dako), anti CD34 (dilution 1:100, clone QBEND10, Dako), anti desmin (D33, monoclonal, 1:10, Linaris, Germany), anti vimentin (Vim3B4, 1:300, Dako), anti NSE (BBC/NC/VI-H14; polyclonal, 1:500, Dako), anti S100 (polyclonal, 1:1000, Dako), anti Ki-67 (clone Mib-1; dilution 1:200, Dako).
In brief, deparaffinised and re-hydrated tissue sections (3 μm) were pretreated in a microwave in CitraPlus solution (Biogenex, USA; 2 minutes on 450 watt then on 80 watt for 20 minutes). After blocking the endogenous peroxidase activity (peroxidase blocking agent, Dako, Denmark), the sections were incubated with the monoclonal mouse anti cyclin H antibody followed by incubation with anti-mouse immunoglobulins conjugated with peroxidase-labeled dextran polymers (N-Histofine, Nichirei Corporation, Japan). Staining was detected with 3, 3'- diaminobenzidine (liquid DAB +, Dako, Denmark) as chromogen and counterstained with hematoxylin before being cover slipped. Cyclin H positivity was defined as positive staining of ≥10% of the tumour cell nuclei according to the modified classifications of Bondi et al. and Kayaselcuk et al. [22,23]. Assessment was done by estimating the rate of positive cells in 10 consecutive fields of view (magnification: 20×) and calculation of the arithmetic mean value of two independent reviewers (TFEB, KK). Results of routine immunhistochemical diagnostics such as expression of CD34, smooth muscle actin, desmin, vimentin and NSE were included in statistical analysis.
Statistical analysis
For investigation of the obtained data, an exploratory data analysis was performed using SPSS 16 (SPSS Inc., USA) and Excel 2007 (Microsoft Corporation, USA). All criteria were rated equally important, without adjustment of p-values for multiple testing. Tumour size, mitotic rate and age were considered to be continuous variables, while others, such as positivity for immunohistochemical markers, initial symptoms and sex, are treated as categorical variables.
To analyse hypotheses regarding the independence of variables, a contingency table was created and either a chi-square test (X2), or (if one or more cells contain less than 5 respondents) Fisher's exact test was performed. Testing was always done two-sided. For statistical analysis of timeline-dependent parameters such as disease-specific survival, a Kaplan-Meier estimation was created and significance was tested using log-rank test. For the calculation of disease-specific-survival (DSS), non GIST-related death-events were censored. P-values < 0.05 were considered statistically significant (α = 0.05). No correction for multiple testing was done.
Results
A single-centre population of ninety five patients with a mean age of 64.3 years (median 66.9 ± 13.5) ranging from 17 to 94 years and a male/female ratio of 42/53 (44% men, 56% women) underwent surgical resection for a GIST via laparotomy (except one: only biopsy, see below). Clinical manifestations, pathological findings, treatment options, and clinical outcome were evaluated, and statistical analyses were carried out with regard to a potential predictive value of cyclin H.
Major clinical symptoms, tumour location, histomorphology, immunohistology and risk classification are summarised in Table 1 and 2. Tumour size varied between 0.4 and 30.0 centimetres (mean 7.4 ± 5.6) and the mitotic rate per 50 HPF ranged from 0 to 116 (mean 10.8 ± 21). No major differences were obtained by using alternative risk estimations (risk of progressive disease, risk of tumour progression, risk category, according to Miettinen et al. [7], Hornick et al. [4] and Joensuu [6]. All four classification scales did significantly differentiate between high risk and non-high risk GIST (p < 0.001); Table 3.
Table 2.
Immunohistochemical Results in GIST
| Immunohistochemistry | Positive, n (%) | Negative, n (%) | Total, n | ||
|---|---|---|---|---|---|
| c-kit (CD117) | 92 | (98%) | 2 | (2%) | 94 |
| cyclin H | 23 | (24%) | 72 | (76%) | 95 |
| CD34 | 63 | (85%) | 11 | (15%) | 74 |
| smooth muscle actin | 11 | (17%) | 53 | (83%) | 64 |
| desmin | 4 | (9%) | 43 | (91%) | 47 |
| vimentin | 32 | (100%) | 0 | (0%) | 32 |
| NSE | 6 | (55%) | 5 | (45%) | 11 |
| S100 | 0 | (0%) | 55 | (100%) | 55 |
Two c-kit negative GIST turned to be PDGFRα positive, confirmed as GIST by mutational analysis
Table 3.
P values regarding different parameters in GIST
| Independent Variables | TRD | DSS | DFS | Met/Rec | Count |
|---|---|---|---|---|---|
| Tests: | χ2/Fisher Exact | Log-Rank | Log-Rank | χ2/Fisher Exact | (n) |
| Sex | 0.180 | 0.226 | 0.647 | 0.626 | 95 |
| Localization (stomach vs. small bowel) | 0.671 | 0.618 | 0.433 | 0.611 | 87 |
| Tumour size (≥ 5 cm vs. <5 cm) | 0.005 | 0.008 | <0.001 | <0.001 | 92 |
| Tumour size (≥ 10 cm vs. <10 cm) | <0.001 | <0.001 | <0.001 | <0.001 | 92 |
| Mitotic rate (≥ 5/50 HPF vs. <5/50 HPF) | 0.001 | 0.001 | <0.001 | <0.001 | 90 |
| Mitotic rate (≥ 10/50 HPF vs. <10/50 HPF) | <0.001 | <0.001 | <0.001 | <0.001 | 90 |
| Histomorphology (spindle vs. not) | 0.103 | 0.099 | 0.217 | 0.187 | 92 |
| Fletcher (high vs. non-high) | <0.001 | <0.001 | <0.001 | <0.001 | 93 |
| Joensuu (high vs. non-high) | <0.001 | <0.001 | <0.001 | <0.001 | 89 |
| Miettinen (high vs. non-high) | 0.001 | <0.001 | <0.001 | <0.001 | 86 |
| Hornick (high vs. non-high) | <0.001 | <0.001 | <0.001 | <0.001 | 90 |
| Primary tumour state (unifocal vs. not) | <0.001 | <0.001 | <0.001 | -- | 93 |
| CD34 (pos vs. neg) | 0.680 | 0.511 | 0.744 | 1.000 | 74 |
| Aktin (pos vs. neg) | 1.000 | 0.818 | 0.844 | 1.000 | 64 |
| Desmin (pos vs. neg) | 1.000 | 0.930 | 0.602 | 1.000 | 47 |
| Cyclin H (pos vs. neg) | 0.369 | 0.189 | 0.692 | 0.806 | 95 |
| Fletcher (high vs. non-high) | <0.001 | <0.001 | <0.001 | <0.001 | 93 |
| Cyclin H (pos vs. neg, only high-risk) | 0.435 | 0.038 | 0.552 | 1.000 | 39 |
| Cyclin H (pos high risk vs rest) | 0.004 | <0.001 | 0.001 | 0.011 | 95 |
| Joensuu (high vs. non-high) | <0.001 | <0.001 | <0.001 | <0.001 | 89 |
| Cyclin H (pos vs. neg, only high-risk) | 0.281 | 0.009 | 0.330 | 0.730 | 40 |
| Cyclin H (pos. high-risk vs. rest) | 0.004 | <0.001 | <0.001 | 0.007 | 89 |
| Miettinen (high vs. non-high) | 0.001 | <0.001 | <0.001 | <0.001 | 86 |
| Cyclin H (pos vs. neg, only high-risk) | 0.681 | 0.050 | 0.967 | 0.696 | 32 |
| Cyclin H (pos. high-risk vs rest) | 0.035 | <0.001 | 0.012 | 0.053 | 86 |
| Hornick (high vs. non-high) | <0.001 | <0.001 | <0.001 | <0.001 | 90 |
| Cyclin H (pos vs. neg, only high-risk) | 0.306 | 0.020 | 0.502 | 1.000 | 36 |
| Cyclin H (pos. high-risk vs. rest) | 0.003 | <0.001 | 0.001 | 0.007 | 90 |
Dependent on the primary tumour site and the extent of tumour growth, final resection state was R0 in 83 (88%), R2 in 7 (8%) and R1 in 4 (4%) of 94 patients; one 94-year-old patient with a huge gastric GIST was not resected and only biopsies were performed due to a coexisting metastatic obstructive colonic cancer. 16 out of 27 patients who had tumour recurrence and/or metastases were treated with imatinib (200-800 mg/d) in addition to surgery. Of these 15 patients, 8 (50%) achieved stable disease, 4 (25%) attained a partial remission and 4 (25%) had tumour progression. Imatinib did not lead to a complete remission in any of our patients.
Outcome
Median follow-up time of the surviving patients was 5.37 years (64.39 months) (mean 6 years, range 0.4 to 20.2 years). At the time of diagnosis, 15% (n = 14) of all GIST patients showed metastatic disease. 16% (n = 15) of the patients died due to GIST-related causes (overall survival 69% (n = 66)) and the rate of patients with metastases or tumour recurrence increased to 27% (n = 26) during the time of observation. Disease-specific 1-, 3-, and 5-year survival probability (DSS) was 96%, 87%, and 84%. Respectively, disease-free survival (DFS) was 80%, 76% and 72%. The median disease-free interval after primary diagnosis of patients with primary unifocal disease and later developed metastases or tumour recurrence was 2.1 years (25 months), mean 2.6 years (31.5 months), range 0.5 to 6.1 years (5.6 to 73 months). Interestingly, 32% (n = 30) of the patients exhibited additional malignant neoplasms. Concurrent benign neoplasias were found in 17% (n = 16). Results of the survival analysis are summarised in Tables 3, 4, 5 and in Figure 1, 2, 3, 4, 5.
Table 4.
Results of the Survival Analysis in GIST
| Non-Disease-specific | 1-year | 3-year | 5-year | |||
|---|---|---|---|---|---|---|
| Overall survival | 91% | 79% | 71% | |||
| 1-year DSS (%) | 3-year DSS (%) | 5-year DSS (%) | 1-year DFS (%) | 3-year DFS (%) | 5-year DFS (%) | |
| Whole cohort | ||||||
| Survival | 96 | 87 | 84 | 80 | 76 | 72 |
| Risk of malignancy according to Fletcher et al. 2002 [1] | ||||||
| Very low | 100 | 100 | 100 | 100 | 100 | 100 |
| Low | 100 | 95 | 95 | 95 | 95 | 95 |
| Intermediate | 100 | 100 | 100 | 100 | 100 | 100 |
| High | 89 | 71 | 65 | 53 | 44 | 38 |
| Non-high | 100 | 98 | 98 | 98 | 98 | 98 |
| Cyclin H | ||||||
| Positive | 90 | 80 | 72 | 78 | 67 | 67 |
| Negative | 97 | 89 | 87 | 80 | 79 | 73 |
| Other criteria | ||||||
| Prim-local | 100 | 97 | 94 | 95 | 90 | 85 |
| Prim-metastasis | 72 | 33 | 33 | -- | -- | -- |
| Met: any time | 84 | 56 | 46 | 27 | 19 | 8 |
| R0 resection | 97 | 94 | 90 | 88 | 86 | 85 |
| R1/2 resection | 83 | 40 | 40 | 33 | 25 | 25 |
DSS = disease specific survival; DSF = disease free survival
Table 5.
Survival and p-values with focus on subgroups of GIST regarding Cyclin H expression
| High-Risk | DSS | DFS | ||||
|---|---|---|---|---|---|---|
| Cyclin H |
positive (n = 11) |
negative (n = 28) |
all (n = 39) |
positive (n = 11) |
negative (n = 28) |
all (n = 39) |
| 1-year probability (%) | 75 | 93 | 89 | 51 | 53 | 52 |
| 3-year probability (%) | 50 | 77 | 71 | 26 | 49 | 44 |
| 5-year probability (%) | 33 | 73 | 65 | 26 | 41 | 38 |
| Log-Rank Test (p) | p = 0.038 | p = 0.522 | ||||
| Tumour Recurrence or Metastases | DSS | DFS | ||||
| Cyclin H |
positive (n = 7) |
negative (n = 20) |
all (n = 27) |
positive (n = 7) |
negative (n = 20) |
all (n = 27) |
| 1-year probability (%) | 67 | 90 | 84.9 | 27 | 30 | 30 |
| 3-year probability (%) | 33 | 64 | 57.0 | 0 | 25 | 19 |
| 5-year probability (%) | 0 | 58 | 47.5 | 0 | 10 | 7 |
| Log-Rank Test (p) | p = 0.016 | p = 0.362 | ||||
(results of immunostaining)
Figure 1.
Disease specific survival - high versus non-high. Disease specific survival (DSS) of the different risk of malignancy groups according to Fletcher; p < 0.001 (high versus non-high).
Figure 2.
Disease free survival - high versus non-high. Disease free survival (DFS) of the different risk of malignancy groups according to Fletcher; p < 0.001 (high versus non-high).
Figure 3.
DSS according to Cyclin H expression in high risk GIST. DSS in comparison of Cyclin H staining in high risk GIST; positive versus negative (p = 0.038).
Figure 4.
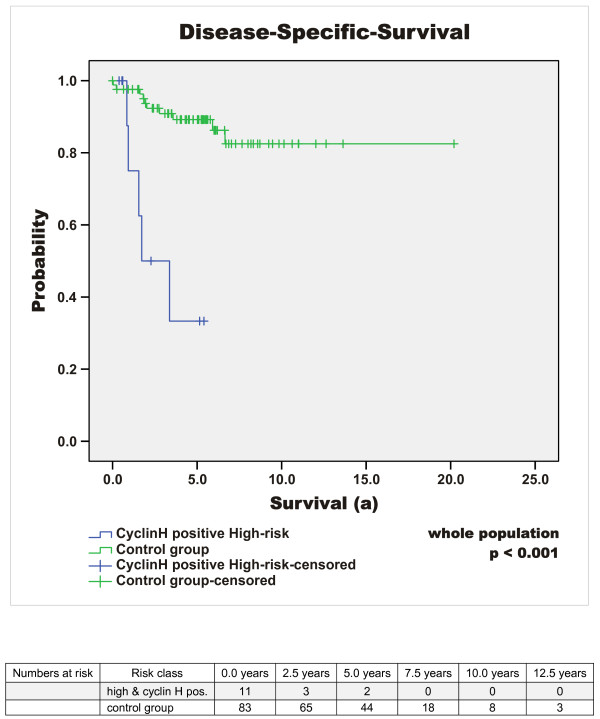
Cyclin H expression and high risk classification as predictor (DSS). Comparison of DSS of all cyclin H positive high risk GIST versus all cyclin H negative high risk GIST as well as all other GIST classified as intermediate, low and very low summarised as control group (p < 0.001).
Figure 5.
Cyclin H expression and high risk classification as predictor (DFS). Comparison of DFS of all cyclin H positive high risk GIST versus all cyclin H negative high risk GIST as well as all other GIST classified as intermediate, low and very low summarised as control group (p < 0.001).
Cyclin H expression and clinical outcome (see also Table 3, 4, 5)
Quantification of cyclin H expression by Real time PCR in normal intestinal tissue and in a relapse of a jejunal high risk GIST indicated that cyclin H transcription is increased by 10 fold in the tumour tissue. Based on this high transcription level of cyclin H in one GIST, we analysed cyclin H expression at a protein level in the tumour tissue of the same patient. A high nuclear staining of cyclin H was detected. Consequently, immunohistochemical analysis of cyclin H expression in the tumour tissue of 95 GIST patients was undertaken and revealed nuclear positivity of cyclin H in 24% (n = 23) (cut-off value of ≥10% reactive cells) (Figure 6). In the majority of these tumours the intensity was moderate positive (19/23) and only 5 tumours showed a very strong cyclin H staining. Of these five tumours 3 are classified as low risk, 1 as very low risk and 1 as high risk GIST. Further interpretation of strongly positive or moderate positive cannot be assessed because of the limited number of cases. Analysis of the relationship between nuclear cyclin H positivity and risk of malignancy according to Fletcher and co-workers [1] revealed that high risk GIST tumours are 3 times more frequently cyclin H positive than very-low risk GIST (p = 0.176). The disease-specific survival (DSS) of cyclin H positive GIST-patients after 1, 3 and 5 years is approximately 10% below the cyclin H negative cohort (log-rank test, p = 0.189) and within the cyclin H positive GIST patients the disease specific mortality rate is 22% compared to 14% in patients with cyclin H negative GIST (p = 0.369); Table 3. Of the 23 tumours stained positive for Cyclin H the distribution with regard to the different risk categories was as follows: 11 of 39 high risk GIST (28%), 4 of 19 intermediate risk (21%) and 8 of 35 low or very low risk GIST (23%). With focus on the high risk group, cyclin H positivity is significantly related to reduction of disease-specific survival (log-rank: p = 0.038; see Tables 3 &5; Figure 3). In patients who were affected by tumour recurrence or metastases, cyclin H positivity indicated a significantly lower disease-specific survival (e.g. 33.3% vs. 64.2% after 3 years, p = 0.016 log-rank test, see Table 5). The combination of cyclin H positivity and high risk GIST showed the strongest predictive p-value for poor disease-free as well as disease-specific survival (p = 0.001 and p < 0.001, log-rank test; cyclin H positive high risk GIST in comparison with the rest of the whole population; see Table 3 and Figure 5 &6).
Figure 6.
Expression of cyclin H in gastrointestinal stromal tumours. Serial sections of gastrointestinal stromal tumours (GISTs) were immunostained using the anti cyclin H specific monoclonal antibody ab54903. A: negative staining; B: GIST showing a fraction of positively stained cell nuclei; C: positivity of almost all cells with strong nuclear and faint cytoplasmic staining.
No association was found between cyclin H status and localisation of the primary tumour (p = 0.471), sex (p = 0.935), and routine immunohistochemical markers such as CD34 (p = 1.000), smooth muscle actin (p = 0.479), desmin (p = 0.564) and Ki-67 (p = 0.227); Table 3. In multivariate analyses, no statistically relevant related factors could be detected.
Discussion
The aim of the present study was to evaluate the prognostic value of cyclin H expression in GIST. To this end, the expression of cyclin H was analysed at the mRNA (single case pilot analysis) and consecutively at a protein level of the whole cohort. The cyclin H immunostaining pattern of tumours of 95 patients with GIST was characterised and correlated to clinicopathologic features and clinical outcome.
Due to the absence of reliable genetic or immunohistochemical predictors, tumour size and mitotic rate are still the determining NIH criteria for risk estimation in GIST [1]. Although the classification distinguishes the tumours using a four-point scale (very low, low, intermediate and high risk of malignancy), this scale is only useful to differentiate between high-risk and non-high-risk GIST. High risk classification according to Fletcher et al. [1] is a reliable predictor for the development of metastases or tumour relapse since 85-97% of the affected patients originate from the high risk group. No major differences were found by using alternative risk classification scales according to Miettinen et al., Hornick et al. and Joensuu [4,6,7]. In contrast the high risk group itself remains inhomogeneous based on the period of long-term disease-specific and disease-free survival of GIST patients even without adjuvant treatment (DSS 65%, DFS 38% after 5 years in our study cohort; Table 5; Figure 1 &2). These findings are in line with previous published data and comments highlighting differences up to 100% within this group [9]. In the light of adverse drug effects, the development of secondary resistance to commercial available and broadely used tyrosine kinase inhibitor (TKI) in molecular target based therapy concepts and high annual treatment costs (40,000-80,000 €/year) [3,9,24] it seems mandatory to detect prognostic factors helping to use adjuvant therapy more selectively by identifying patients with a "very-high" risk within the high risk group particularly after an R0 resection.
Quantification of cyclin H mRNA revealed a 10.2 fold increased transcription of cyclin H in a high risk jejunal GIST compared to normal tissue, suggesting an important role of cyclin H and the cyclin-CDK-system in GIST pathogenesis. By immunohistochemical analysis of cyclin H expression in 95 tumour specimens of a single centre population we found a high expression of cyclin H (≥ 10% reactive cells) in 24% of the tumours, which correlated well with the risk of malignancy (p = 0.176). Among all Cyclin H positive patients enrolled in our study there is a tendency for a poor prognosis, although there was no statistical significance (p = 0.692 for DFS and p = 0.189 for DSS). In patients with high risk GIST the expression of cyclin H was highly predictive for the reduction of DSS (p = 0.038). Accordingly, cyclin H expression differentiated high risk and "very-high risk" GIST with regard to disease-specific mortality and might be a valuable tool for further treatment decisions. Moreover, with regard to the whole population, the combination of cyclin H positivity and high risk classification according to Fletcher was strongly predictive of a poor DFS as well as DSS (p = 0.001 and p < 0.001) [1]. Additionally, in patients with tumour recurrence and/or metastases, cyclin H positivity was significantly associated with reduced disease-specific survival (p = 0.016) regardless of risk-classification.
Although the predictive value of Cyclin H is limited with regard to the whole population, our results suggest for the first time a predictive value of cyclin H expression in high risk GIST patients, underlining the importance of the cyclin-CDK system for GIST pathogenesis. Therewith, our data strengthen previous reports on the predictive value of cyclin A, cyclin B, cyclin D1, cyclin E, cdc2, p27 and p21 [16-20,25-27]. However, immunohistochemical positivity of cyclin A, cyclin B, cyclin D1, cyclin E and cdc2 [17,19,20] or the loss of cyclin kinase inhibitors p27 and p21 [16-18,25-27] has only been shown to be associated with high risk grading in GIST, while investigations in regard to clinical outcome are missing. In contrast, our data show a significant correlation of Cyclin H expression with reduced DSS in high risk GIST and in patients with metastases or tumour recurrence.
The efforts to identify parameters that clearly correlate with the clinical outcome of GIST are not restricted to the cyclin-CDK-system. Similarly, the prognostic value of various other factors, such as p53, p16, p21, pRb, E2F1, p27KIP1, Mdm2, Bcl-2 and Bax is not yet entirely clarified. Although changes in their expression have been evaluated in regard to risk ranking, in most cases their correlation with the clinical outcome (DSF, DSS, PFS) has not been validated in detail. Furthermore, the conclusions drawn are still in some cases contradictory [3,17,18,28,29]. Moreover, data relating to the prognostic value of the mutational status are still under debate [30-34]. Altogether, this suggests that one factor does not satisfy the multi-factorial and multidimensional complexity of tumourigenesis and tumour progression of GIST. The identification of factors with a potential prognostic value, as here described for cyclin H, is an important prerequisite for multi-factorial analyses. Simultaneous analysis of cyclin H and a previously investigated factor, p16 [28], within an intersection sub-cohort revealed that the combined positivity of both parameters indicates poor outcome of GIST, irrespective of the mitotic rate or tumour size (p = 0.039) (Additional file 2: Table S2 & S3 and Figure S1). After one year, 50% of the patients with cyclin H- and p16-positive high risk GIST died and none of these patients was tumour free in comparison to 98% survivors and 83% tumour-free patients in the control group. These findings indicate the necessity of multi-factorial follow-up studies (in larger series) for the future.
Conclusions
In conclusion, conventional risk estimations including tumour size, mitotic rate and tumour location are useful to differentiate high risk and non-high risk GIST. The combination of positivity for cyclin H and high risk classification predicts highly significant poor prognosis in GIST. Also in patients with recurrence or metastases, the expression of cyclin H is the only relevant clinical predictor. Therefore, protein expression of cyclin H may allow subclassification of "very-high risk" (cyclin H-positive high risk GIST) from high risk GIST. Whether cyclin H alone or in combination with any other factor will be an indicator for the necessity of adjuvant treatment of R0-resected high risk GIST with a tyrosine kinase inhibitor remains to be further investigated.
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
Study concepts: JD, HS, UK, KK; Study design: JD, HS, MS, UK, KK; Data aquisition: JD, HS, MS, TFEB, UK, KK; Quality control of data and algorithms: JD, HS, MS, TFEB, UK, KK; Data analysis and interpretation: JD, HS, MS, AB, TFEB, DHB, UK, KK; Statistical analysis: JD, MS, AB, UK, KK; Manuscript preparation: JD, HS, UK, KK; Manuscript editing: JD, HS, AB, TFEB, DHB, UK, KK; Manuscript review: JD, HS, MS, AB, TFEB, DHB, UK, KK
Pre-publication history
The pre-publication history for this paper can be accessed here:
Supplementary Material
Table S1. Table 1: Suggested risk classifications
Table S2 & S3 and Figure S1. Table S2: Combined Cyclin H and p16 positivity - Results of the Survival Analysis Table S3: P values for Combined Cyclin H and p16 positivity Figure S1: Disease specific survival in high-risk GIST with combined positivity for cyclin H and p16 (p < 0.001).
Contributor Information
Julian Dorn, Email: julian.dorn@gmx.de.
Hanno Spatz, Email: johann.Spatz@klinikum-augsburg.de.
Michael Schmieder, Email: michael-Schmieder@freenet.de.
Thomas FE Barth, Email: thomas.barth@uniklinik-ulm.de.
Annette Blatz, Email: annette.blatz@uniklinik-ulm.de.
Doris Henne-Bruns, Email: doris.henne-bruns@uniklinik-ulm.de.
Uwe Knippschild, Email: uwe.knippschild@uniklinik-ulm.de.
Klaus Kramer, Email: gzu.kramer@gmx.de.
Acknowledgments
The authors thank Dr. Bettina Danner (Institute of Biometrics) for excellent cooperation in statistical analysis, Dr. Cathy Dew (Worcester, Great Britain) for editorial assistance and Heidrun Hirner, Arnhild Grothey, Elena Yasovskaya and Helena Krist for technical assistance.
References
- Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, Miettinen M, O'Leary TJ, Remotti H, Rubin BP. Diagnosis of gastrointestinal stromal tumors: A consensus approach. Hum Pathol. 2002;33:459–465. doi: 10.1053/hupa.2002.123545. [DOI] [PubMed] [Google Scholar]
- Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006;130:1466–1478. doi: 10.5858/2006-130-1466-GSTROM. [DOI] [PubMed] [Google Scholar]
- Steigen SE, Eide TJ. Gastrointestinal stromal tumors (GISTs): a review. APMIS. 2009;117:73–86. doi: 10.1111/j.1600-0463.2008.00020.x. [DOI] [PubMed] [Google Scholar]
- Hornick JL, Fletcher CD. The role of KIT in the management of patients with gastrointestinal stromal tumors. Hum Pathol. 2007;38:679–687. doi: 10.1016/j.humpath.2007.03.001. [DOI] [PubMed] [Google Scholar]
- Hou YY, Lu SH, Zhou Y, Xu JF, Ji Y, Hou J, Qi WD, Shi Y, Tan YS, Zhu XZ. Predictive values of clinical and pathological parameters for malignancy of gastrointestinal stromal tumors. Histol Histopathol. 2009;24:737–747. doi: 10.14670/HH-24.737. [DOI] [PubMed] [Google Scholar]
- Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol. 2008;39:1411–1419. doi: 10.1016/j.humpath.2008.06.025. [DOI] [PubMed] [Google Scholar]
- Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach: a clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29:52–68. doi: 10.1097/01.pas.0000146010.92933.de. [DOI] [PubMed] [Google Scholar]
- Wente MN, Buchler MW, Weitz J. [Gastrointestinal stromal tumors (GIST). Surgical therapy] Chirurg. 2008;79:638–643. doi: 10.1007/s00104-008-1527-5. [DOI] [PubMed] [Google Scholar]
- Hohenberger P. Adjuvant imatinib in GIST: a self-fulfilling prophecy, or more? Lancet. 2009;373:1058–1060. doi: 10.1016/S0140-6736(09)60562-6. [DOI] [PubMed] [Google Scholar]
- DeMatteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, Blackstein ME, Blanke CD, von Mehren M, Brennan MF. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373:1097–1104. doi: 10.1016/S0140-6736(09)60500-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson M, Persson JL. Cancer therapy: targeting cell cycle regulators. Anticancer Agents Med Chem. 2008;8:723–731. doi: 10.2174/187152008785914833. [DOI] [PubMed] [Google Scholar]
- Malumbres M. Cyclins and related kinases in cancer cells. J BUON. 2007;12(Suppl 1):S45–52. [PubMed] [Google Scholar]
- Ali S. Role of c-kit/SCF in cause and treatment of gastrointestinal stromal tumors (GIST) Gene. 2007;401:38–45. doi: 10.1016/j.gene.2007.06.017. [DOI] [PubMed] [Google Scholar]
- Bauer S, Duensing A, Demetri GD, Fletcher JA. KIT oncogenic signaling mechanisms in imatinib-resistant gastrointestinal stromal tumor: PI3-kinase/AKT is a crucial survival pathway. Oncogene. 2007;26:7560–7568. doi: 10.1038/sj.onc.1210558. [DOI] [PubMed] [Google Scholar]
- Tornillo L, Terracciano LM. An update on molecular genetics of gastrointestinal stromal tumours. J Clin Pathol. 2006;59:557–563. doi: 10.1136/jcp.2005.031112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirin H, Kravtsov V, Shahmurov M, Shabat VS, Krinshpon I, Alin A, Avinoach I, Avni Y. The cyclin-dependent kinase inhibitor, p27, has no correlation with the malignant potential of GIST. Digestion. 2007;75:4–9. doi: 10.1159/000101457. [DOI] [PubMed] [Google Scholar]
- Nemoto Y, Mikami T, Hana K, Kikuchi S, Kobayashi N, Watanabe M, Okayasu I. Correlation of enhanced cell turnover with prognosis of gastrointestinal stromal tumors of the stomach: relevance of cellularity and p27kip1. Pathol Int. 2006;56:724–731. doi: 10.1111/j.1440-1827.2006.02038.x. [DOI] [PubMed] [Google Scholar]
- Sabah M, Cummins R, Leader M, Kay E. Altered expression of cell cycle regulatory proteins in gastrointestinal stromal tumors: markers with potential prognostic implications. Hum Pathol. 2006;37:648–655. doi: 10.1016/j.humpath.2006.01.023. [DOI] [PubMed] [Google Scholar]
- Nakamura N, Yamamoto H, Yao T, Oda Y, Nishiyama K, Imamura M, Yamada T, Nawata H, Tsuneyoshi M. Prognostic significance of expressions of cell-cycle regulatory proteins in gastrointestinal stromal tumor and the relevance of the risk grade. Hum Pathol. 2005;36:828–837. doi: 10.1016/j.humpath.2005.03.012. [DOI] [PubMed] [Google Scholar]
- Koon N, Schneider-Stock R, Sarlomo-Rikala M, Lasota J, Smolkin M, Petroni G, Zaika A, Boltze C, Meyer F, Andersson L. Molecular targets for tumour progression in gastrointestinal stromal tumours. Gut. 2004;53:235–240. doi: 10.1136/gut.2003.021238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lolli G, Johnson LN. CAK-Cyclin-dependent Activating Kinase: a key kinase in cell cycle control and a target for drugs? Cell Cycle. 2005;4:572–577. [PubMed] [Google Scholar]
- Bondi J, Husdal A, Bukholm G, Nesland JM, Bakka A, Bukholm IR. Expression and gene amplification of primary (A, B1, D1, D3, and E) and secondary (C and H) cyclins in colon adenocarcinomas and correlation with patient outcome. J Clin Pathol. 2005;58:509–514. doi: 10.1136/jcp.2004.020347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kayaselcuk F, Erkanli S, Bolat F, Seydaoglu G, Kuscu E, Demirhan B. Expression of cyclin H in normal and cancerous endometrium, its correlation with other cyclins, and association with clinicopathologic parameters. Int J Gynecol Cancer. 2006;16:402–408. doi: 10.1111/j.1525-1438.2006.00407.x. [DOI] [PubMed] [Google Scholar]
- Dirnhofer S, Leyvraz S. Current standards and progress in understanding and treatment of GIST. Swiss Med Wkly. 2009;139:90–102. doi: 10.4414/smw.2009.12166. [DOI] [PubMed] [Google Scholar]
- Di Vizio D, Demichelis F, Simonetti S, Pettinato G, Terracciano L, Tornillo L, Freeman MR, Insabato L. Skp2 expression is associated with high risk and elevated Ki67 expression in gastrointestinal stromal tumours. BMC Cancer. 2008;8:134. doi: 10.1186/1471-2407-8-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu FY, Qi JP, Xu FL, Wu AP. Clinicopathological and immunohistochemical analysis of gastrointestinal stromal tumor. World J Gastroenterol. 2006;12:4161–4165. doi: 10.3748/wjg.v12.i26.4161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J, Du X, Lazar AJ, Pollock R, Hunt K, Chen K, Hao X, Trent J, Zhang W. Genetic aberrations of gastrointestinal stromal tumors. Cancer. 2008;113:1532–1543. doi: 10.1002/cncr.23778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmieder M, Wolf S, Danner B, Stoehr S, Juchems MS, Wuerl P, Henne-Bruns D, Knippschild U, Hasel C, Kramer K. p16 expression differentiates high-risk gastrointestinal stromal tumor and predicts poor outcome. Neoplasia. 2008;10:1154–1162. doi: 10.1593/neo.08646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider-Stock R, Boltze C, Lasota J, Peters B, Corless CL, Ruemmele P, Terracciano L, Pross M, Insabato L, Di Vizio D. Loss of p16 protein defines high-risk patients with gastrointestinal stromal tumors: a tissue microarray study. Clin Cancer Res. 2005;11:638–645. [PubMed] [Google Scholar]
- Agaimy A, Haller F, Gunawan B, Wunsch PH, Fuzesi L. Distinct biphasic histomorphological pattern in gastrointestinal stromal tumours (GISTs) with common primary mutations but divergent molecular cytogenetic progression. Histopathology. 2009;54:295–302. doi: 10.1111/j.1365-2559.2008.03214.x. [DOI] [PubMed] [Google Scholar]
- Bachet JB, Hostein I, Le Cesne A, Brahimi S, Beauchet A, Tabone-Eglinger S, Subra F, Bui B, Duffaud F, Terrier P. Prognosis and predictive value of KIT exon 11 deletion in GISTs. Br J Cancer. 2009;101:7–11. doi: 10.1038/sj.bjc.6605117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoeben A, Schoffski P, Debiec-Rychter M. Clinical implications of mutational analysis in gastrointestinal stromal tumours. Br J Cancer. 2008;98:684–688. doi: 10.1038/sj.bjc.6604217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubin BP, Heinrich MC, Corless CL. Gastrointestinal stromal tumour. Lancet. 2007;369:1731–1741. doi: 10.1016/S0140-6736(07)60780-6. [DOI] [PubMed] [Google Scholar]
- Schildhaus HU, Merkelbach-Bruse S, Buttner R, Wardelmann E. Pathology and molecular biology of gastrointestinal stromal tumors (GIST) Radiologe. 2009;49(12):1104–8. doi: 10.1007/s00117-009-1850-y. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1. Table 1: Suggested risk classifications
Table S2 & S3 and Figure S1. Table S2: Combined Cyclin H and p16 positivity - Results of the Survival Analysis Table S3: P values for Combined Cyclin H and p16 positivity Figure S1: Disease specific survival in high-risk GIST with combined positivity for cyclin H and p16 (p < 0.001).