Abstract
Hydrophobic or amphiphilic tetrapyrrole sensitizers are taken up by cells and are usually located in cellular lipid membranes. Singlet oxygen is photogenerated by the sensitizer and it diffuses in the membrane and causes oxidative damage to membrane components. This damage can occur to membrane lipids and to membrane-localized proteins. Depolarization of the Nernst electric potential on cells’ membranes has been observed in cellular photosensitization, but it was not established whether lipid oxidation is a relevant factor leading to abolishing the resting potential of cells’ membranes and to their death. In this work we studied the effect of liposomes’ lipid composition on the kinetics of hematoporphyrin-photosensitized dissipation of K+-diffusion electric potential that was generated across the membranes. We employed an electrochromic voltage-sensitive spectroscopic probe that possesses a high fluorescence signal response to the potential. We found a correlation between the structure and unsaturation of lipids and the leakage of the membrane, following photosensitization. As the extent of non-conjugated unsaturation of the lipids is increased from 1 to 6 double bonds, the kinetics of depolarization become faster. We also found that the kinetics of depolarization is affected by the percentage of the unsaturated lipids in the liposome: as the fraction of the unsaturated lipids increases the leakage trough the membrane is enhanced. When liposomes are composed of a lipid mixture similar to that of natural membranes and photosensitization is being carried out under usual photodynamic therapy (PDT) conditions, photodamage to the lipids is not likely to cause enhanced permeability of ions through the membrane, which would have been a mechanism that leads to cell death.
Keywords: photodynamic therapy (PDT), di-4-ANBDQPQ, voltage sensitive dye, Nernst electric potential
INTRODUCTION
The application of porphyrins and their analogs as photosensitizers for selective photodamage of inflicted tissue in photodynamic therapy (PDT) is based on two important properties: their preferential uptake by solid tumors and their efficient production of electronically excited singlet oxygen (1O2), via energy transfer from the sensitizer’s long-lived excited triplet state to molecular oxygen. These attributes, together with the porphyrins’ characteristic fluorescence, were also used diagnostically to identify and mark malignant tissue. Various tumors (including skin, lung, breast and bladder) have been treated by applying, intravenously, various porphyrin derivatives and illuminating with low-intensity light. Malignant cells undergo necrosis or apoptosis with minimal damage to surrounding tissue. PDT has become a clinical protocol for treating malignancies. Recent reviews on photosensitization properties and results of some pre-clinical tests are given (1–9).
There is evidence indicating that the locus of PDT action by amphiphilic sensitizers is the membrane (10–12). Thus, the binding of hematoporphyrin to cellular membranes was found to be a prerequisite for their action (13). It was also found that most of the damage caused by porphyrins in tumor cells - including increased permeability, membrane rupture and cell lysis - occurred in the plasma and organelle membranes (14–17). These observations suggest that generation of 1O2 by amphiphilic sensitizers occurs within the bilayer, which is probably also the site of photodamage, because of singlet oxygen’s fast diffusion out of the membrane and its short lifetime in aqueous media.
As explained above, much of the cellular damage that is inflicted by 1O2 is in the membrane’s phase. It was demonstrated, with amphiphilic sensitizers, that PDT’s lethal action and the injury which is caused, leads to damage of transport mechanisms, depolarization of electric potential and leakage across the cytoplasmic and intracellular membranes (17–27). It is obvious that the depolarization of the cell’s electric potential and free passage of ions and molecules through the cytoplasmic membrane are mutually connected. However, these studies did not indicate whether the change in the electrical field was the result, or the cause, of the lethal action of sensitization, and did not correlate kinetics with likely mechanisms. Still, it is taken sometimes as axiomatic that photosensitized damage to unsaturated lipids in the membrane is involved in the death of the cell. In a preliminary study we showed that depolarization of vesicles was much slower than cell killing under identical photosensitization conditions (28), which may hint that lipid peroxidation is less important than other possible biological targets. A detailed study of the effect of lipid components on the leakage of the electric potential was, however, never carried out.
In this manuscript we report our studies on the correlation between lipid composition of liposomes and the kinetics of depolarization of Nernst, cross-membrane, potential that was set by diffusion of K+ ions. Studying the depolarization of the membrane potential allows the usage of a very subtle parameter of membrane intactness. Our results show that liposomes composed of saturated phospholipids, or even of natural lecithin, remain intact under usual conditions of photosensitization, which usually leads to ionic leakage and depolarization of the cross membrane potential. Such damage is observed only when poly-unsaturated lipids are present at high concentrations. This study bears on the general issue of the correlation between modifications to constituting lipids and the maintenance of membrane integrity.
MATERIALS AND METHODS
Chemicals and sample preparation
The following saturated and unsaturated lipids were all purchased from Avanti Polar Lipids (Alabaster, AL): 1,2-dimyristoyl-sn-glycero-3-phosphocholine (14:0, where 14 is the number of carbons in the lipid’s fatty acid chain and 0 is the number of unconjugated double bonds in the chain), 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (16:0), 1,2-dimyristoleoyl-sn-glycero-3-phosphocholine (14:1 c Δ 9cis, where the double bond is placed in the ninth carbon at the cis position in the fatty acid chain), 1,2-dipalmitoleoyl-sn-glycero-3-phosphocholine (16:1 c Δ 9cis), 1,2-dioleoyl-sn-glycero-3-phospho-choline (18:1 c Δ 9cis), 1,2-dieicosenoyl-sn-glycero-3-phosphocholine (20:1 c Δ 9cis), 1,2-dilinoleoyl-sn-glycero-3-phosphocholine (18:2 c Δ 9,12), 1,2-dilinolenoyl-sn-glycero-3-phospho-choline (18:3 c Δ 9,12,15), 1,2-diarachidonoyl-sn-glycero-3-phosphocholine (20:4 c Δ 5,8,11,14), 1,2-didocosahexaenoyl-sn-glycero-3-phosphocholine (22:6 c Δ 4,7,10,13,16,19). We also used natural egg-yolk lecithin, purchased from Sigma (St. Louis, MO), in which the composition of fatty acids is as follows: 33% palmitic (C16:0), 31% oleic (C18:1), 13% stearic (C18:0), and 15% linoleic (C18:2). The remaining 8% is a mixture of several other fatty acids.
The voltage sensitive dye, 4-(1-[2-(di-n-butylamino)-6-naphthyl]-4-butadienyl)-1-(3-propyl trimethylammonium) quinolinium dibromide (di-4-ANBDQPQ), was synthesized at the University of Connecticut Health Center. We used a 5.5 mM stock solution in ethanol. Valinomycin (Sigma) was also kept as a stock solution in ethanol, at 0.1 mM concentration. We employed hematoporphyrin IX (HP) (Sigma) as the sensitizer, from a stock solution of 5 mM in DMF. The singlet oxygen quencher 9,10-dimethylanthracene (DMA) was also purchased from Sigma.
Spectroscopic measurements
Absorption spectra were recorded on a Shimadzu (Kyoto, Japan) UV-2501PC UV–vis. spectrophotometer. Fluorescence excitation and emission spectra and fluorescence time-drive measurements were performed on a Perkin-Elmer LS-50B digital fluorimeter. For all fluorescence measurements, the samples’ optical density per cm was kept below 0.05 at the wavelength of fluorescence excitation, in order to maintain linear dependence of the fluorescence intensity on concentration.
Preparation of liposomes
A chloroform and ethanol solution of the lipids was evaporated under a stream of nitrogen forming a layer of lipids at the bottom of a vial. The dry lipids were re-dissolved in chloroform, which was then re-evaporated under nitrogen to complete dryness. This process was repeated with diethyl ether. A solution of 0.075 M K2SO4 was added to obtain a lipid concentration of 2.5–10 mg/ml. This sample was vortexed for 1 minute and was then sonicated for 20 minutes at 4°C, using a probe sonicator (MSE Soniprep 150, Crawley, UK) which formed an almost clear solution. This suspension was used as the liposomes’ stock solution.
Since liposomes composed strictly from lipids of unsaturated fatty acids did not sustain a stable electric potential, as evidenced by our voltage-sensitive dye, the preparation of those liposomes was made from a mixture of 70% of L-α-lecithin and 30% of the desired unsaturated lipid.
Preparation of solutions for illumination
Hematoporphyrin IX (HP) was added from its DMF stock solution to the sample of liposomes’ suspension, to a final HP concentration of 6.66μM. The partitioning of HP into the liposomal membrane by diffusion was very rapid, within less than 1 minute. This was established by following the time course of the fluorescence intensity at 621 nm, emitted by membrane-bound HP.
The reaction solution, containing liposomes’ suspension in water and membrane-bound HP, was illuminated by an Ar+ laser (Stabilite 2017, Spectra-Physics, Mountain View, CA) beam at 501.7 nm, which is within one of the excitation bands of the sensitizer. The sample was stirred magnetically throughout the illumination, to obtain uniform exposure of the whole content of the sample. The laser power (around 10 mW) was measured at the sample’s surface with a power meter (model PD2-A, Ophir, Jerusalem, Israel), before and after the measurements, to ensure that the power remained constant during the experiment.
Measurement of membrane electric potential
Every several minutes, during the process of illumination, a small aliquot was taken out of the sample and was diluted 20–100 fold into an aqueous solution of 0.075 M Na2SO4, the same concentration as the K2SO4 inside the liposomes. We chose SO42− as the anion to eliminate the possibility of its leakage through the membrane. The voltage sensitive dye, di-4-ANBDQPQ, was added at a final concentration of 8 μM and its fluorescence intensity was monitored in a time-drive mode for up to 100 minutes. The fluorescence was excited at 650 nm and measured at 800 nm. Two or three minutes after starting the monitoring, the K+-ionophore, valinomycin, was added to a final concentration of 25 nM. This initiates the diffusion of K+ ions from inside the liposomes outside, down their concentration gradient, to generate a cross-membrane Nernst potential. The generation of the electric potential results, at the abovementioned wavelengths, in a rise of the dye’s fluorescence intensity. We found that intact, undamaged, liposomes could sustain an electrical potential difference for a long time. In such case the raised level of fluorescence intensity, which was obtained after the addition of valinomycin, exhibited a very slow down-drift with a time constant of more than 20 minutes. Thus, only processes that were significantly faster than this were considered as depolarization.
According to the Nernst equation a 20-fold dilution of liposomes that contain, inside and outside, K2SO4 into a solution of the same concentration of Na2SO4 generates a potential difference of 76 mV, negative inside.
| (Eq. 1) |
The cross-membrane Nernst potential can be measured electrically, but also by several spectroscopic techniques, employing potentiometric molecular probes, as was done in this study.
Calculation of the reaction rates
The characteristic rate of liposomes’ depolarization, kdepol, was calculated by fitting the time dependence of the dye’s fluorescence intensity, F, to an exponential decay, in which the intensity returns to a non-zero level at very long time, F∞:
| (Eq. 2) |
The rate constant of depolarization is expected to be enhanced as the liposomes, with the photosensitizer, are illuminated for longer periods of time, prior to testing the sustainability of the potential. We thus correlated, for each lipid composition, the dependence of kdepol on the length of photosensitization, and fitted a linear equation to these data. The slope of this line represents the dependence of the rate of depolarization on the length of illumination, e.g. reaction rate.
Estimates of the mean kdepol and its error
The values for kdepol were determined by statistically averaging three to five independent measurements. The weighted average, μ, of several measurements, each yielding a result xi and possessing a measurement error σi, was calculated using the formula (29):
| (eq. 3) |
RESULTS
Spectroscopic properties of the electrochromic probe di-4-ANBDQPQ
In this study we employed the voltage sensitive dye di-4-ANBDQPQ. This dye was demonstrated in previous studies with cells (30,31) to have an electrochromic response mechanism (32) and to respond instantaneously to the change of membrane electric potential, due to the direct interaction of the molecular electronic charge distribution with the electric field. More importantly, it was also reported to have an exceptionally high fluorescence response, of up to 20% change in fluorescence intensity per 100mV potential (33). In addition, this dye is chemically and photochemically highly stable. For these reasons we chose to use this voltage sensitive dye in the current study and also because the excitation wavelengths of the dye do not overlap the absorption bands of the sensitizer HP, which we used. We thus first characterized the potentiometric properties of this indicator.
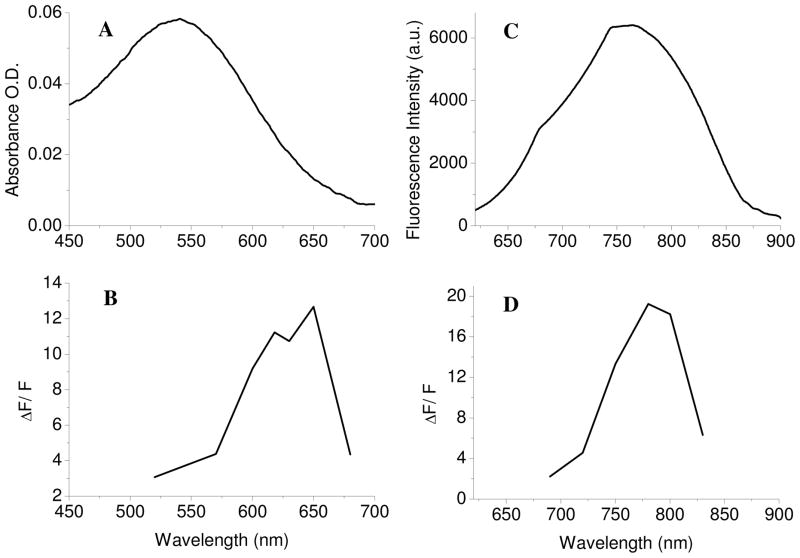
Di-4-ANBDQPQ has structureless absorption and emission spectra, peaking, when bound to liposomes, at 537 and 756 nm, respectively (Fig. 1A, C). The large Stokes shift arises probably from the charge redistribution upon excitation and, as a result, solvation of the vertically excited state that is different from the solvation of the relaxed excited state. We measured the binding constant of the dye to lecithin liposomes, Kb, by a spectroscopic titration method (34) and found it to be 67.88 ± 4.11 ml · mg−1. The intuitive meaning of this number is that at a lipid concentration of Kb−1, namely 0.0147 mg/ml, half of the dye’s molecules are partitioned into the liposomes’ membrane. Since the dye’s fluorescence level in aqueous solution is negligible, the fluorescent signal at much higher lipid concentration than this value, as were used in this study, is contributed only by membrane-residing dye molecules.
Fig. 1.
Spectral properties of the dye di-4-ANBDQPQ. A, C: Absorbance and emission of the dye when bound to liposomes. B: The relative change in its fluorescence intensity, measured at 770 nm, as a function of excitation wavelengths. D: The relative change in its fluorescence intensity excited at 650 nm, as a function of emission wavelengths. A K+-diffusion potential of 59 mV was generated in Figs. 1B and 1D.
In order to assess the response of the dye to cross-membrane diffusion potential, we searched for the optimal combination of excitation and emission wavelengths of the dye that give the strongest change of fluorescence as a result of an induced electric potential across the membrane. We assessed the response of the dye to membrane potential, by measuring the magnitude of the relative change in its fluorescence intensity as a function of excitation or emission wavelengths. The results, which are shown in Figure 1B, D indicate that the optimal wavelengths were 650 nm and 800 nm, respectively. These results with a biphasic response also indicate to a characteristic behavior of electrochromic mechanism.
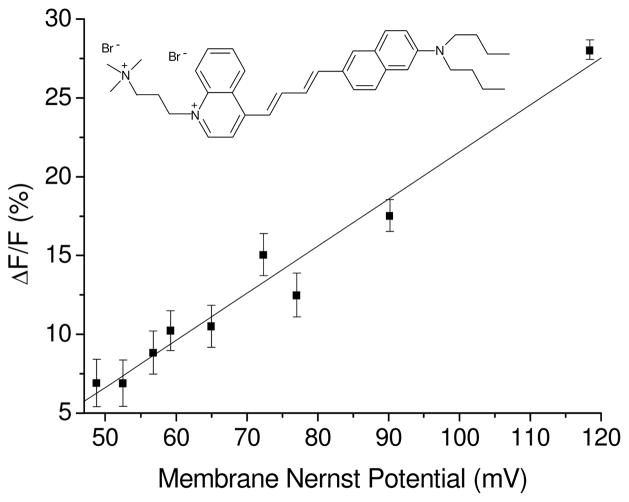
We also measured the response of the dye to the magnitude of the potential, by measuring the size of the relative change in fluorescence intensity as a function of the size of the Nernst diffusion potential on liposomes. We prepared Na2SO4 solutions into which we added different volumes from the liposomes’ suspensions in K2SO4. Hence the concentration gradient of K+ ions between inside and outside the liposomes, in each sample, was different and was used to calculate the membrane potential difference by the Nernst equation. We then calculated the relative change of the fluorescence, ΔF/F, which was measured at the above F optimal pair of wavelengths. The results are plotted in Figure 2. As can be seen, the response is linear, as could be expected from a dye with a potentiometric mechanism. At the specific wavelengths that were used the fluorescence changes by 21.6% at 100 mV potential difference.
Fig. 2.
Relative change of fluorescence intensity of di-4-ANBDQPQ (λexc.= 650 nm; λem.= 800 nm) vs. magnitude of K+ Nernst diffusion potential on liposomes.
Photosensitization of lecithin liposomes
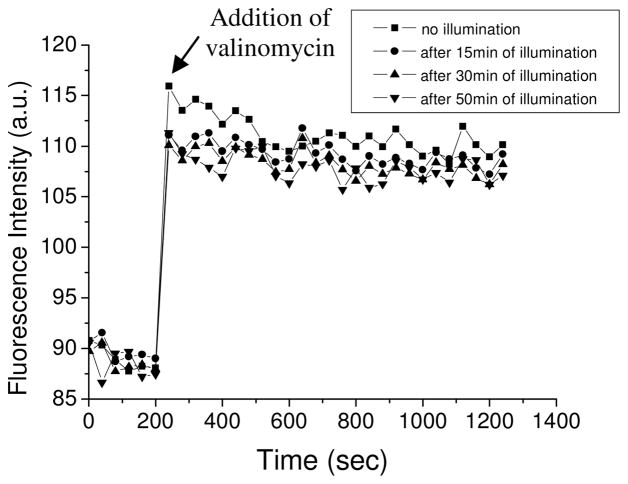
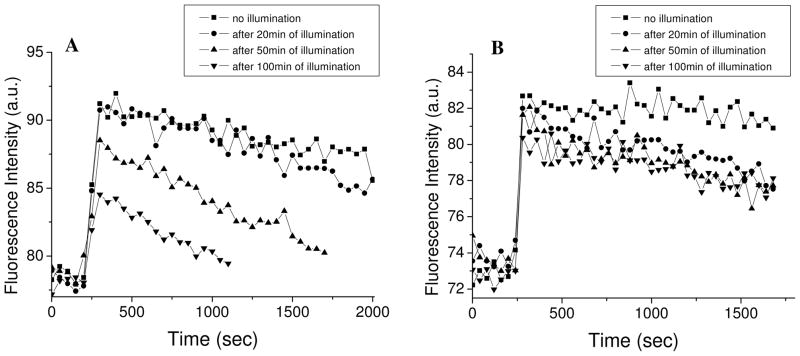
We first investigated the effect of photosensitization by HP on the intactness of liposomes composed of natural egg phospholipids, and their ability to sustain an electric membrane potential. Liposomes composed of natural L-α-lecithin contain a mixture of saturated and unsaturated lipids that mimics the natural lipid content of many cells’ membranes. Figure 3 shows a time-dependent trace of the fluorescence intensity of di-4-ANBDQPQ, before and after the generation of K+ diffusion potential, following different periods of photosensitization by HP. The immediate change in fluorescence intensity that was observed after the addition of valinomycin, which reflects the immediate generation of an electric field across the membrane, was independent of the time the sample had been pre-illuminated. Thus, this indicates that the gradient of K+ ions generates the same electric potential in each of the samples. More importantly, following its generation, the electric potential is stable even after long periods of photosensitization. This indicates that the liposomes do not leak K+ ions across the membrane, which would have dissipated the electric field. This is despite of possible oxidation, peroxidation and breakup at the double bonds in the unsaturated lipids in lecithin. In other words, the degree of unsaturated phospholipids that is present in lecithin is not enough to allow ionic leakage through the membrane following photosensitization.
Fig. 3.
Fluorescence intensity of di-4-ANBDQPQ upon generation of a K+-diffusion potential of 118 mV, on liposomes composed of lecithin.
In light of these results, we shall present in the following sections the photodamaging effect that was observed in liposomes containing a variety of phospholipids that are diverse in the number of non-conjugated double bonds and the length of fatty-acid chains. All these experiments were carried out under identical concentrations of the sensitizer and illumination conditions.
The effect of fatty acids unsaturation
We wanted to find out to what extent fatty acid unsaturation affects photosensitized induction of depolarization of cross-membrane potential. We examined different phosphatidylcholine lipids, with a varying number of double bonds (from one double bond to six non-conjugated double bonds). For each case we measured the kinetics of dissipation of membrane potential after different durations of photosensitization. We then calculated the reaction rate, namely the illumination time-dependent dissipation rate, for each lipid.
It is important to indicate that in liposomes composed strictly of saturated lipids, namely 1,2-dimyristoyl-sn-glycero-3-phosphocholine (DMPC, 14:0) or 1,2-dipalmitoyl-sn-glycero-3-phosphocholine (DPPC, 16:0), there was no depolarization of the potential by photosensitization, even after more than an hour of illumination of the liposomes which contained HP. Also, liposomes containing 30% of 1,2-dioleoyl-sn-glycero-3-phosphocholine (18:1) did not exhibit any remarkable depolarization of the membrane’s electric potential, even after 100 minuets of illumination.
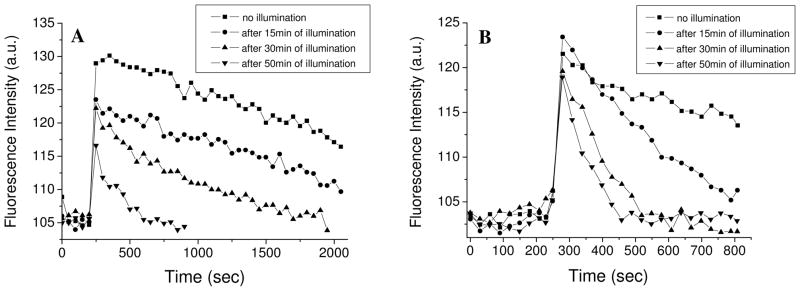
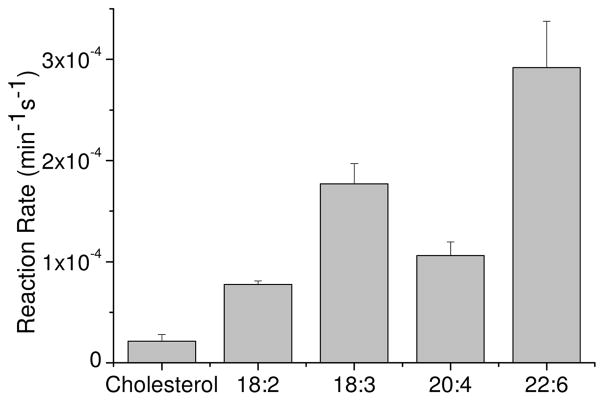
We show in figure 4, as representative examples, the fluorescence intensity traces of di-4-ANBDQPQ, after generation of the diffusion potential, following photosensitization by HP, in mixed liposomes. After 15 minutes of illumination of liposomes composed of 70% lecithin and 30% of 18:2 or 22:6 phospholipids, the time constants of depolarization were 769 seconds and 322 seconds, respectively. After 30 minutes the time constants decreased to 474 seconds and 102 seconds, respectively, and after 50 minutes illumination the values dropped to 258 and 72 seconds. Correlating the depolarization rates (1/(time constant)) with the illumination time yielded the illumination time-dependent dissipation rates, which are shown in Table 1 and in figure 5. As can be seen, liposomes containing 30% of the lipid 1,2-dilinoleoyl-sn-glycero-3-phosphocholine (18:2) possess the most moderate depolarization due to photosensitization. The rate is enhanced 4-fold when the liposomes contain 30% of 1,2-didocosahexaenoyl-sn-glycero-3-phosphocholine (22:6). Thus, the liposomes containing the 22:6 fatty acids exhibit the fastest developing leakage of ions through the membrane after illumination. We might have expected that the dissipation reaction rate of liposomes containing 1,2-diarachidonoyl-sn-glycero-3-phosphocholine (20:4) will be larger than the reaction rate of liposomes containing 1,2-dilinolenoyl-sn-glycero-3-phosphocholine (18:3) but the latter displayed a stronger ionic leakage. We think that a possible reason for these results is the length of the fatty acid chains. The lipid 1,2-diarachidonoyl-sn-glycero-3-phosphocholine (20:4), has longer chains, which makes the packaging of the liposome more stable than the packaging of those containing 1,2-dilinolenoyl-sn-glycero-3-phosphocholine (18:3), which has only 18 carbons in the acid chain.
Fig. 4.
Fluorescence intensity of di-4-ANBDQPQ upon generation of a K+-diffusion potential of 76.9 mV, on liposomes composed of 70% lecithin and 30% phosphatidylcholine with 18:2 acids (A) and with 22:6 acids (B).
Table 1.
Reaction rate of depolarization of liposomes, containing 70% lecithin and 30% of the given lipid.
| Type of phospholipid | Unsaturation of fatty acids | Chemical structure | Reaction rate (min−1s−1) |
|---|---|---|---|
| Cholesterol | -------- | 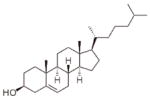 |
(2.14 ± 0.65)×10−5 |
| 1,2-dilinoleoyl-sn-glycero-3-phosphocholine | 18:2 c Δ9,12 |
(7.74 ± 0.35)×10−5 | |
| 1,2-dilinolenoyl-sn-glycero-3-phosphocholine | 18:3 c Δ9,12, 15 |
(1.77 ± 0.20)×10−4 | |
| 1,2-diarachidonoyl-sn-glycero-3-phosphocholine | 20:4 c Δ5,8,11,14 |
(1.06 ± 0.13)×10−4 | |
| 1,2-didocosahexaenoyl-sn-glycero-3-phosphocholine | 22:6c Δ4,7,10,13,16,19 |
(2.92 ± 0.45)×10−4 |
Fig 5.
The illumination time-dependent depolarization rate constants, for liposomes composed of 70% lecithin and 30% of the given lipids or cholesterol.
We examined the formation of malondialdehyde, which is one of the peroxidation products of non-conjugated fatty acids, from the lipid 1,2-didocosahexaenoyl-sn-glycero-3-phosphocholine (22:6). We employed the thiobarbituric acid assay (35) and found that as the illumination time increased the amount of the malondialdehyde that was created increase as well. The reaction of singlet oxygen with a set of two non-conjugated double bonds releases malondialdehyde and shortens the fatty acid chain by three carbon atoms (36). This may constitute a local perturbation in the bilayer’s structure and may lead to increased permeability of the membrane. Additional reactions of the lipids with 1O2, which may lead to breakage of the double bonds, are of course also possible.
The singlet oxygen quencher 9,10-dimethylanthracene (DMA) was added to the liposomes’ suspension to a final concentration of 100μM. We found that after 15 minutes of illumination the DMA fluorescence decreased by more than 90%. DMA is known for its very efficient chemical reaction with singlet oxygen (37), we thus conclude that the damage to the liposomes is caused by singlet oxygen.
We also measured the reaction rate of liposomes composed of 70:30 w:w lecithin and cholesterol. We found that the leakage of the membrane’s electric potential was the slowest, compared to the other unsaturated phosphatidylcholines which were used. Schnitzer et al. (38) have indicated that peroxidation of liposomes containing cholesterol is much slower then peroxidation of liposomes containing palmitoyl-linoleoyl-phosphatidylcholine (PLPC) (16:0–18:2), and our results are compatible with their observation.
The effect of the length of fatty acid chains
In order to check whether the length of the fatty acids affects the efficiency of photosensitized induction of depolarization, we examined lipids with only one double bond but with different lengths of the fatty acid chains, from 14 carbons to 20 carbons in a single chain. Liposomes composed of 30% lipids with long fatty acid chains of 16–20 carbons sustain the membrane’s electric potential like non-sensitized liposomes. In contrast, a slow leakage of the membrane’s potential was seen in liposomes containing 30% of 1,2-dimyristoleoyl-sn-glycero-3-phosphocholine (14:1) after about 100 minutes of illumination. We show in Figure 6 the change of membrane potential as a function of illumination time in liposomes containing lipids with 14:1 acids and 20:1 acids.
Fig. 6.
Fluorescence intensity of di-4-ANBDQPQ upon generation of a K+-diffusion potential of 76.9 mV on liposomes composed of 70% lecithin and 30% phosphatidylcholine with 14:1 (A) and 20:1 fatty acids (B).
The effect of the fraction of unsaturated lipid in the liposome composition
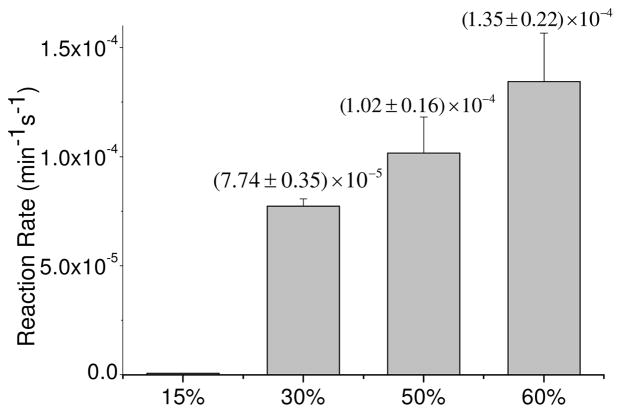
We prepared liposomes from lecithin and 1,2-dilinoleoyl-sn-glycero-3-phosphocholine (18:2) at varying proportions. This lipid gave the most stable liposomes among the poly-unsaturated lipids, as was shown previously. The experiments were executed in the following weight percentages of the 18:2 lipid: 15%, 30%, 50% and 60%, with lecithin as the complementary lipid. We found that at higher 18:2 ratios, the liposomes were not stable enough to sustain a steady membrane potential. The depolarization time was inversely dependent on the fraction of the 18:2 PC. For example after 30 minutes of illumination, the depolarization time constants were 2974 sec, 714 sec, 443 sec and 211 sec, for the above respective ratios. Figure 7 shows the depolarization reaction rate at each composition. There is a clear trend of increase of the rate of depolarization by photosensitization, as the content of this unsaturated lipid increases. In liposomes containing 15% of the unsaturated 18:2 lipid we observed only a marginal rate of depolarization by photosensitization, even after 100 minutes of illumination. In contrast, liposomes containing 60% of this lipid displayed fast ionic leakage through the membrane after 35 minutes of illumination, having a depolarization time constant of only 170 sec.
Fig. 7.
Ionic leakage rate constants as a function of the weight percentage of 18:2 lipid in the liposomes.
It should be remembered, that all the above-described experiments were executed with liposomes containing a mixture of lecithin and of the given additive lipid (the percentage of each component is as reported at each of the experiments). However, as was shown in the previous experiments, liposomes made of lecithin alone were not damaged by the photosensitization under the current conditions. We can therefore state, undoubtedly, that whatever damage that was caused to the membrane of the liposomes resulted from the oxidation of the added lipid and not from lecithin.
DISCUSSION
Photodynamic therapy inflicts an irreversible damage to the cell’s membrane, following light absorption by the photosensitizer. From the early days of photodynamic research, the involvement of 1O2 was postulated to be important in biological photosensitization, through oxidation of fatty acids (39–41). Furthermore, different types of cells were shown to have their membrane potential depolarized (17–25,42,43) and liposome models were demonstrated to undergo lysis (41,44,45).
Two questions remained, however, unanswered. The first is whether the electric depolarization is an initial step in the non-apoptotic process of cellular damage, caused by damage to the membrane as a barrier, or is it the result of the cell’s death, which might have been caused by other mechanisms. Namely, it is not clear whether membrane depolarization is the cause or the result of sensitized cell death. An additional question is whether the critical damage in the membrane is caused to lipids or to other membrane-bound entities, mainly proteins. In summary, is the depolarization of the membrane critical and is it attributable to damage to the lipids that build the membrane bilayer or rather to damage to membrane proteins or other components that are located in the membrane?
In this study we took the first step and measured the extent of depolarization of liposomes, which had different compositions, as a result of photosensitization with hematoporphyrin. By this approach, we probed a delicate attribute of a membrane, namely its intactness and action as a barrier to the diffusion of ions under a concentration gradient. Such leakage can compromise the membrane even before massive mechanical destruction of the membrane occurs. First, we examined liposomes composed of a lipid mixture similar to that of natural membranes. We did not find any significant change in the ability of the liposomes to sustain a K+- diffusion potential as a result of photosensitization by HP, over an hour of illumination.
The intactness of the liposomes was not compromised at conditions similar to those employed in PDT. Lysis of liposomes that was observed in the group of Grossweiner (45–47) was achieved with light intensities that were higher than in normal PDT. Thus, liposomal membrane diffusion potential is not abolished as a consequence of photosensitization, under conditions that lead to massive death of animal and bacterial cells. This is similar to our previous observation (28). This may indicate that the change in the electric membrane potential that occurs due to photodynamic sensitization, at least in the case of model liposomes under usual conditions, is not arising from simple poration of the bilayer. The same negative effect was observed when we employed liposomes that were composed of fully saturated lipids, and the electric potential of the membrane was not affected.
However, when the degree of unsaturation of the lipids that build the bilayer membrane was increased, depolarization could be achieved by photosensitization. We found that as the number of non-conjugated double bonds in the fatty-acid chains of the lipid increased, from 1 to 6, the light dose-dependent reaction rate for depolarization increased strongly. Thus the ability of the liposomes to sustain the electric potential decreased, outside the exception of lipids 20:4 and 18:3. It should be stressed again, that when liposomes composed of saturated lipid or of natural lecithin were examined, the electric potential of the membrane was sustainable and no ionic leakage due to photosensitization was detected. The initial step in lipid oxidation by singlet oxygen is usually the formation of peroxide at the double bond of the fatty acid, which in turn leads to reorganization of the non-conjugated double bonds and to the formation of conjugated double bond (35,48). The ionic leakage through the membrane can be attributed to the formation of shortened fatty acid chains, formation of alcoholic or aldehyde groups at the breakage points of double bonds and a general rearrangement and packaging of the hydrophobic core of the membrane. These changes form void volumes as well as polar environments within the bilayer and the insertion of oxygen atoms may cause repulsion between fatty acids chains, increasing the distance between the oxidized and the nonoxidized residues. Disintegration products such as short-chain carbonyl produce phospholipids that tend to collapse the bilayer structure (49,50).
The length of the fatty acid chains could be an important parameter which can affect the leakage of K+ ions across the membrane. Fatty acid chains with only 14 carbon atoms and one double bound are not long enough to preserve the stability of the membrane for long and are probably the reason for the leakage of the membrane that we observed. Additionally, when the fatty acid possesses more then one double bond the length of the chain is significant in preserving the stability of the liposomes. The fraction of the added unsaturated lipid in the liposome composition significantly affects the oxidation of the membrane. The larger the content of unsaturated lipids in the membrane, the more susceptible is the membrane to oxidation, and as a result to depolarization.
CONCLUSIONS
The diffusion of ions through the membrane may require just a subtle change in the memrane’s structure. This is indeed very different from complete lysis, as was observed in some cases in past studies. However, the diffusion of K+ ions, which was studied here, can be an immediate cause of the depolarization of the cross-membrane potential, leading to cell death. This study shows that under conditions similar to those used in biological photosensitization, such process may indeed be relevant to depolarization of membrane potential on liposomes only if they contain high concentrations of poly-unsaturated fatty acids, such as 18:3, 20:4 and 22:6.
Acknowledgments
This research was supported by a grant of the US-Israel Binational Science Foundation, by the Michael David Falk Chair in Laser Phototherapy and by the Katz Family Grant Incentive Program (to BE) and by USPHS NIH grant EB001963 (to LML).
References
- 1.Dougherty TJ, Kaufman JE, Goldfarb A, Weishaupt KR, Boyle D, Mittleman A. Photoradiation therapy for the treatment of malignant tumors. Cancer Res. 1978;38:2628–2635. [PubMed] [Google Scholar]
- 2.Dougherty TJ. Photosensitizers: therapy and detection of malignant tumors. Photochem Photobiol. 1987;45:879–889. doi: 10.1111/j.1751-1097.1987.tb07898.x. [DOI] [PubMed] [Google Scholar]
- 3.Henderson BW, Dougherty TJ. How does photodynamic therapy work? Photochem Photobiol. 1992;55:145–157. doi: 10.1111/j.1751-1097.1992.tb04222.x. [DOI] [PubMed] [Google Scholar]
- 4.Jori G, Brown SB. Photosensitized inactivation of microorganisms. Photochem Photobiol Sci. 2004;3:403–405. doi: 10.1039/b311904c. [DOI] [PubMed] [Google Scholar]
- 5.Wainwright M. Photoinactivation of viruses. Photochem Photobiol Sci. 2004;3:406–411. doi: 10.1039/b311903n. [DOI] [PubMed] [Google Scholar]
- 6.Juzeniene A, Peng Q, Moan J. Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci. 2004;6:1234–1245. doi: 10.1039/b705461k. [DOI] [PubMed] [Google Scholar]
- 7.Verma S, Watt GM, Mal Z, Hasan T. Strategies for enhanced photodynamic therapy effects. Photochem Photobiol. 2007;83:996–1005. doi: 10.1111/j.1751-1097.2007.00166.x. [DOI] [PubMed] [Google Scholar]
- 8.Plaetzer K, Krammer B, Berlanda J, Berr F, Kiesslich T. Photophysics and photochemistry of photodynamic therapy: fundamental aspects. Lasers Med Sci. 2009;24:259–268. doi: 10.1007/s10103-008-0539-1. [DOI] [PubMed] [Google Scholar]
- 9.Robertson CA, Evans DH, Abrahamse H. Photodynamic therapy (PDT): A short review on cellular mechanisms and cancer research applications for PDT. J Photochem Photobiol B-Biol. 2009;96:1–8. doi: 10.1016/j.jphotobiol.2009.04.001. [DOI] [PubMed] [Google Scholar]
- 10.Moan J, Berg K, Kvam E, Western A, Malik Z, Ruck A, Schneckenburger H. In: Photosensitizing Compounds: Their Chemistry, Biology and Clinical Use. Bock G, Harnett S, editors. John Wiley and Sons; New York: 1989. pp. 95–111. [Google Scholar]
- 11.Berg K, Moan J. Lysosomes and microtubules as targets for photochemotherapy of cancer. Photochem Photobiol. 1997;65:403–409. doi: 10.1111/j.1751-1097.1997.tb08578.x. [DOI] [PubMed] [Google Scholar]
- 12.Mojzisova H, Bonneau S, Brault D. Structural and physico-chemical determinants of the interactions of macrocyclic photosensitizers with cells. Europ Biophys J. 2007;36:943–953. doi: 10.1007/s00249-007-0204-9. [DOI] [PubMed] [Google Scholar]
- 13.Ehrenberg B, Malik Z, Nitzan Y. Fluorescent spectral changes of hematoporphyrin derivative upon binding to lipid vesicles, s. aureus and e. coli cells. Photochem Photobiol. 1985;41:429–435. doi: 10.1111/j.1751-1097.1985.tb03508.x. [DOI] [PubMed] [Google Scholar]
- 14.Kessel D. Hematoporphyrin and HPD: photophysics, photochemistry and photo-therapy. Photochem Photobiol. 1984;39:851–859. doi: 10.1111/j.1751-1097.1984.tb08871.x. [DOI] [PubMed] [Google Scholar]
- 15.Malik Z, Djaldetti M. Destruction of erythroleukemia myelocytic leukemia and Burkitt lymphoma cells by photoactivated protoporphyrin. Int J Cancer. 1980;26:495–500. doi: 10.1002/ijc.2910260415. [DOI] [PubMed] [Google Scholar]
- 16.Boegheim JPJ, Lagerberg JWM, Dubbelman TMAR, Tijssen K, Tanke HJ, Van der Meulen J, Van Steveninck J. Photodynamic effects of HPD on the uptake of rhodamine 123 by mitochondria of intact murine L929 fibroblasts and Chinese hamster ovary K1 cells. Photochem Photobiol. 1988;48:613–620. doi: 10.1111/j.1751-1097.1988.tb02871.x. [DOI] [PubMed] [Google Scholar]
- 17.Paardekooper M, Van den Broek PJA, De Bruijne AW, Elferink JGR, Dubbelman TMAR, Van Steveninck J. Photodynamic treatment of yeast cells with the dye toluidine blue: all-or-none loss of plasma membrane barrier properties. Biochim Biophys Acta. 1992;1108:86–90. doi: 10.1016/0005-2736(92)90117-5. [DOI] [PubMed] [Google Scholar]
- 18.Bellnier DA, Dougherty TJ. Membrane lysis in Chinese hamster ovary cells treated with hematoporphyrin derivative plus light. Photochem Photobiol. 1982;36:43–47. doi: 10.1111/j.1751-1097.1982.tb04338.x. [DOI] [PubMed] [Google Scholar]
- 19.Henderson BW, Donovan JM. Release of prostaglandin E2 from cells by photodynamic treatment in vitro. Cancer Res. 1989;49:6896–6900. [PubMed] [Google Scholar]
- 20.Specht KG, Rodgers MAJ. Depolarization of mouse myeloma cell membranes during photodynamic action. Photochem Photobiol. 1990;51:319–324. doi: 10.1111/j.1751-1097.1990.tb01717.x. [DOI] [PubMed] [Google Scholar]
- 21.Specht KG, Rodgers MAJ. Plasma membrane depolarization and calcium influx during cell injury by photodynamic action. Biochim Biophys Acta. 1991;1070:60–68. doi: 10.1016/0005-2736(91)90146-y. [DOI] [PubMed] [Google Scholar]
- 22.Krammer-Reubel B. Transmembrane Potential measurements of normal and transformed human fibroblasts following photodynamic laser therapy. Bioelectrochem Bioenerg. 1992;27:19–22. [Google Scholar]
- 23.Kochevar IE, Bouvier J, Lynch M, Lin CW. Influence of dye and protein location on photosensitization of the plasma membrane. Biochim Biophys Acta-Biomembranes. 1994;1196:172–180. doi: 10.1016/0005-2736(94)00236-3. [DOI] [PubMed] [Google Scholar]
- 24.Kunz L, Stark G. Photofrin II sensitized modifications of ion transport across the plasma membrane of an epithelial cell line: I. Electrical measurements at the whole-cell level. J Memb Biol. 1998;166:179–185. doi: 10.1007/s002329900459. [DOI] [PubMed] [Google Scholar]
- 25.Chaloupka R, Obsil T, Plasek J, Sureau F. The effect of hypericin and hypocrellin-A on lipid membranes and membrane potential of 3T3 fibroblasts. Biochim Biophys Acta-Biomembranes. 1999;1418:39–47. doi: 10.1016/s0005-2736(99)00016-4. [DOI] [PubMed] [Google Scholar]
- 26.Lagerberg JWM, Uberriegler KP, Krammer B, Vansteveninck J, Dubbelman TMAR. Plasma membrane properties involved in the photodynamic efficacy of merocyanine 540 and tetrasulfonated aluminum phthalocyanine. Photochem Photobiol. 2000;71:341–346. doi: 10.1562/0031-8655(2000)071<0341:pmpiit>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 27.Kessel D, Luguya R, Vicente MGH. Localization and photodynamic efficacy of two cationic porphyrins varying in charge distribution. Photochem Photobiol. 2003;78:431–435. doi: 10.1562/0031-8655(2003)078<0431:lapeot>2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 28.Ehrenberg B, Gross E, Nitzan Y, Malik Z. Electric depolarization of photosensitized cells: lipid vs. protein alterations. Biochim Biophys Acta. 1993;1151:257–264. doi: 10.1016/0005-2736(93)90110-l. [DOI] [PubMed] [Google Scholar]
- 29.Bevington PR. Data Reduction and Error Analysis for the Physical Sciences. Chapter 5 McGraw-Hill; New York, USA: 1969. [Google Scholar]
- 30.Wuskell JP, Boudreau D, Wei M, Jin L, Engl R, Chebolu R, Bullen A, Hoffacker KD, Kerimo J, Cohen LB, Zochowski MR, Loew LM. Synthesis, spectra, delivery and potentiometric responses of new styryl dyes with extended spectral ranges. J Neurosc Methods. 2006;151:200–215. doi: 10.1016/j.jneumeth.2005.07.013. [DOI] [PubMed] [Google Scholar]
- 31.Teisseyre TZ, Millard AC, Yan P, Wuskell JP, Wei MD, Lewis A, Loew LM. Nonlinear optical potentiometric dyes optimized for imaging with 1064-nm light. J Biomed Opt. 2007;12(4):044001. doi: 10.1117/1.2772276. [DOI] [PubMed] [Google Scholar]
- 32.Loew LM. Spectroscopic Membrane probes. Vol. 2. CRC Press; Boca Raton, FL: 1988. How to choose a potentiometric membrane probe; pp. 139–151. [Google Scholar]
- 33.Matiukas A, Mitrea BG, Qin M, Pertsov AM, Shvedko AG, Warren MD, Zaitsev AV, Wuskell JP, Wei M, Watras J, Loew LM. Near-infrared voltage-sensitive fluorescent dyes optimized for optical mapping in blood-perfused myocardium. Heart Rhythm. 2007;4:1442–1451. doi: 10.1016/j.hrthm.2007.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ehrenberg B. Assessment of the partitioning of probes to membranes by spectroscopic titration. J Photochem Photobiol B. 1992;14:383–386. doi: 10.1016/1011-1344(92)85117-d. [DOI] [PubMed] [Google Scholar]
- 35.Buege JA, Aust SD. Microsomal lipid-peroxidation. Methods in Enzymology. 1978;52:302–310. doi: 10.1016/s0076-6879(78)52032-6. [DOI] [PubMed] [Google Scholar]
- 36.Niehaus WG, Jr, Samyelsson B. Formation of malonaldehyde from phospholipid arachidonate during microsomal lipid peroxidation. Eur J Biochem. 1968;6:126–130. doi: 10.1111/j.1432-1033.1968.tb00428.x. [DOI] [PubMed] [Google Scholar]
- 37.Castro-Olivares R, Gunther G, Zanocco AL, Lemp E. Linear free energy relationship analysis of solvent effect on singlet oxygen reactions with mono and disubstituted anthracene derivatives. J Photochem Photobiol A-Chem. 2009;207:160–166. [Google Scholar]
- 38.Schnitzer E, Pinchuk I, Bor A, Leikin-Frenkel A, Lichtenberg D. Oxidation of liposomal cholesterol and its effect on phospholipid peroxidation phospholipid peroxidation. Chem Phys Lipids. 2007;146:43–53. doi: 10.1016/j.chemphyslip.2006.12.003. [DOI] [PubMed] [Google Scholar]
- 39.Foote CS. Mechanisms of Photosensitized Oxidation. Science. 1968;162:963–970. doi: 10.1126/science.162.3857.963. [DOI] [PubMed] [Google Scholar]
- 40.Rawls HR, Van Santen PJ. A possible role for singlet oxygen in the initiation of fatty acid autoxidation. J Amer Oil Chem Soc. 1970;47:121–125. [Google Scholar]
- 41.Anderson SM, Krinsky NI. Protective action of carotenoid pigments against photodynamic damage to liposomes. Photochem Photobiol. 1973;18:403–408. doi: 10.1111/j.1751-1097.1973.tb06440.x. [DOI] [PubMed] [Google Scholar]
- 42.Schothorst AA, van Steveninck J, Went LN, Suurmond D. Metabolic aspects of the photodynamic effect of protoporphyrin in protoporphyria and in normal red blood cells. Clin Chim Acta. 1971;33:207–213. doi: 10.1016/0009-8981(71)90269-5. [DOI] [PubMed] [Google Scholar]
- 43.Goldstein BD, Harber LC. Erythropoietic Protoporphyria: Lipid Peroxidation and Red Cell Membrane Damage Associated with Photohemolysis. J Clin Invest. 1972;51:892–901. doi: 10.1172/JCI106884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Leibowitz MC, Johnson MC. Relation of lipid peroxidation to loss of cations trapped in liposomes. J Lipid Res. 1971;12:662–670. [PubMed] [Google Scholar]
- 45.Muller-Runkel R, Blais J, Grossweiner LI. Photodynamic damage to egg lecithin liposomes. Photochem Photobiol. 1981;33:683–687. [Google Scholar]
- 46.Grossweiner LI, Patel AS, Grossweiner JB. Type I and type II mechanisms in the photosensitized lysis of phosphatidylcholine liposomes by hematoporphyrin. Photochem Photobiol. 1982;36:159–167. doi: 10.1111/j.1751-1097.1982.tb04358.x. [DOI] [PubMed] [Google Scholar]
- 47.Grossweiner LI, Goyal GC. Photosensitized lysis of liposomes by hematoporphyrin derivative. Photochem Photobiol. 1983;37:529–532. doi: 10.1111/j.1751-1097.1983.tb04513.x. [DOI] [PubMed] [Google Scholar]
- 48.Catal’a A. An overview of lipid peroxidation with emphasis in outer segments of photoreceptors and the chemiluminescence assay. The Intl J Biochem Cell Biol. 2006;38:1482–1495. doi: 10.1016/j.biocel.2006.02.010. [DOI] [PubMed] [Google Scholar]
- 49.Van Ginkel G, Sevanian A. Lipid peroxidation-induced membrane structural alteration. Methods in Enzymology. 1994;233:273–288. doi: 10.1016/s0076-6879(94)33031-x. [DOI] [PubMed] [Google Scholar]
- 50.Van den Berg JJM, Op den Kamp JAF, Lubin BH, Kuypers FA. Conformational change in oxidized phospholipid and their preferential hydrolysis by phospholipase A2: a monolayer study. Biochemistry. 1993;32:4962–4967. doi: 10.1021/bi00069a035. [DOI] [PubMed] [Google Scholar]