Abstract
The morphogen Sonic Hedgehog (Shh) promotes neovascularization in adults by inducing pro-angiogenic cytokine expression in fibroblasts; however, the direct effects of Shh on endothelial cell (EC) function during angiogenesis are unknown. Our findings indicate that Shh promotes capillary morphogenesis (tube length on Matrigel™ increased to 271±50% of the length in untreated cells, p=0.00003), induces EC migration (modified Boyden chamber assay, 191±35% of migration in untreated cells, p=0.00009), and increases EC expression of matrix metalloproteinase 9 (MMP-9) and osteopontin (OPN) mRNA (real-time RT-PCR), which are essential for Shh-induced angiogenesis both in vitro and in vivo. Shh activity in ECs is mediated by Rho, rather than through the “classic” Shh signaling pathway, which involves the Gli transcription factors. The Rho dependence of Shh-induced EC angiogenic activity was documented both in vitro, with dominant-negative RhoA and Rho kinase (ROCK) constructs, and in vivo, with the ROCK inhibitor Y27632 in the mouse corneal angiogenesis model. Finally, experiments performed in MMP-9- and OPN-knockout mice confirmed the roles of the ROCK downstream targets MMP-9 and OPN in Shh-induced angiogenesis. Collectively, our results identify a “non-classical” pathway by which Shh directly modulates EC phenotype and angiogenic activity.
Keywords: angiogenesis, Sonic Hedgehog, endothelial cells, Gli transcription factors, ischemia
Introduction
Hedgehog (Hh) proteins, which include Sonic hedgehog (Shh), Indian hedgehog, and Desert hedgehog, act as morphogens in multiple tissues during embryonic development [1–8]. The role of Shh in vasculogenesis has been documented in a variety of contexts. Hypervascularization of the neuroectoderm has been observed after transgenic overexpression of Shh in the dorsal neural tube of mouse embryos [9], Shh-deficient zebrafish exhibit disorganized endothelial precursors and are unable to form the dorsal aorta or axial vein [10], and the developing lungs of Shh-deficient mice lack proper vascularization [5].
Our previous studies have shown that the embryonic Hh pathway is reactivated in adult animal models of ischemic injury, including hind limb-ischemia [11] and myocardial infarction [12], and that administration of Shh as a recombinant protein or via gene therapy enhances neovascularization of ischemic tissue by promoting both angiogenesis [11] and the recruitment of endothelial progenitor cells (EPCs) [12–13]. Shh induces fibroblasts and cardiomyocytes to overexpress several pro-angiogenic growth factors, including vascular endothelial growth factor (VEGF) and angiopoietin-1 (Ang1) [11–14], which suggests that the effect of Shh on vascular cells is indirect. Recently, we have shown that Shh influences EPCs directly to promote EPC adhesion, migration, and proliferation [13].
In the classically described Hh-signaling cascade, interactions between Shh and the receptor Patched-1 (Ptch1) repress Ptch-1–mediated inhibition of Smoothened (Smo), which subsequently leads to activation of the Gli transcription factors, then activated Gli induces expression of Ptch1, Gli1, and other downstream target genes [15]. However, the nature and extent of Shh activity in mature endothelial cells (ECs) remain controversial. Corneal neovessel ECs and human umbilical vein endothelial cells (HUVECs) do not overexpress Ptch1 in response to Shh stimulation [11]; nevertheless, Shh promotes capillary morphogenesis in both HUVECs and murine brain capillary ECs [16]. Shh activates the phosphoinositide kinase 3 (PI3K)/Akt pathway in ECs [16], and other signal transduction pathways, such as the cyclic AMP/protein kinase A axis [17], can modulate the activity of the Gli transcription factors. Shh also induces expression of chicken ovalbumin upstream promoter transcription factor II (COUP TFII) in mouse embryonic carcinoma cells [18] and extracellular signal-regulated kinases 1/2 (ERK 1/2) and protein kinase Cδ (PKCδ) in fibroblasts [19]; Shh activates the Rho/Rho kinase (ROCK) pathway in neuronal cells [20].
The potential role of a nonclassical signaling pathway in Shh-mediated angiogenesis has not been fully explored. Accordingly, we performed a series of in vitro and in vivo experiments to characterize the potential direct angiogenic effects of Shh. Our results reveal that Shh influences angiogenesis directly through the Rho/ROCK signaling pathway and by inducing expression of downstream target genes such as matrix metalloproteinase 9 (MMP-9) and osteopontin (OPN). Thus, our investigation has identified a novel, nonclassical mechanism of Shh signaling that modulates neovascularization.
Materials and Methods
All studies were approved by the Northwestern University Animal Care and Use Committee.
Cell lines
Experiments were performed with bovine aortic endothelial cells (BAECs) (ATCC, Manassas, VA, USA), HUVECs and human coronary-artery endothelial cells (HCAECs) (Cambrex Corporation, East Rutherford, NJ, USA), NIH 3T3 embryonic fibroblasts (ATCC) and HeLa cells. Culture conditions are provided in the Supplemental Methods.
Tube formation assay
25×103 serum-starved BAECs were seeded in the presence or absence of Shh protein in each well of a 48-well plate coated with 120 μL of growth-factor–reduced Matrigel™ (BD Biosciences, San Jose, CA, USA). Tube formation was examined by phase-contrast microscopy 6 hours later.
Migration assay
Cell migration was evaluated by using a modified Boyden’s chamber (Neuro Probe, Inc., Bethesda, MD, USA) as detailed in the Supplemental Methods. Each condition was assayed in triplicate, and each experiment was performed at least three times.
In vitro proliferation assay
Cells were incubated with 200 μg/mL bromodeoxyuridine (BrdU) for 24 hours, fixed with 100% methanol for 10 minutes at 4°C, then stained with immunofluorescent sheep anti-BrdU antibodies (Abcam Inc., Cambridge, MA, USA); nuclei were stained with DAPI. Each experiment was performed at least three times, and each condition in each experiment was assayed in triplicate.
Quantitative real-time reverse transcriptase–polymerase chain reaction (RT-PCR)
RNA was isolated from 3×105 cells or homogenized tissue with RNA STAT-60 (TEL-TEST Electronics Labs Inc, Austin, TX, USA) according to the manufacturer’s instructions. Total RNA was reverse transcribed with a Taqman cDNA Synthesis Kit (Applied Biosystems, Foster City, CA, USA) and amplification was performed with a Taqman 7500 (Applied Biosystems). Primer and probe sequences are listed in the Supplemental Table. The relative expression of each mRNA was calculated by the comparative threshold cycle (CT) method and normalized to 18S expression.
Plasmid and siRNA transfection
Transfection was performed as detailed in the Supplemental Methods; assays were performed 48 hours after transfection. The Gli-BS luciferase and mutGli luciferase plasmids were kindly provided by Dr H. Sasaki [21]. The ROCK RB/PH (pEF-BOS-myc-Rho-kinase RB/PH)- and RhoAN19 (pEF-BOS-HA-RhoA-N19)-expressing plasmids were kindly provided by Dr K. Kaibuchi [22]. The Ezrin dominant-negative plasmid (pCB-6-Ezrin NTer) was kindly provided by Dr M. Arpin [23]. The sequences of the OPN- and MMP-9–inhibiting siRNAs and the nonsilencing, GFP siRNA (Dharmacon, Lafayette, CO, USA) are provided in the Supplemental Methods.
Gene reporter assay
BAECs and NIH3T3 fibroblasts were transfected in 190 mm2 wells by using TransFast™ transfection reagent (Promega Corporation) according to the manufacturer’s instructions. Cells were co-transfected with 0.08 μg Gli-BS luciferase plasmid [21] or 0.08 μg mutGli luciferase plasmid [21] and 0.08 μg RSV-βgal plasmid. HUVECs were transfected by using JetPEI™-HUVEC (Polyplus Transfection) according to the manufacturer’s instructions. Briefly, 2.5×105 cells were seeded in 950 mm2 wells; 1 day later, the cells were co-transfected with 0.65 μg Gli-BS luciferase or mutGli luciferase, and 0.65 μg pCMVnLacZ. Luciferase was assayed with a Luciferase assay system (Promega Corporation) and β-galactosidase activity was assayed as previously described [24]. For each sample, luciferase activity was normalized to β-galactosidase activity to compensate for differences in transfection efficiency. Each condition was assayed in triplicate, and each experiment was performed al least three times.
Rho pull down assay, Western blot
Rho activity was assayed by using a Rho activation kit (Upstate, Lake Placid, NY, USA) according to the manufacturer’s protocol. Briefly, Rho-GTP was precipitated with Rotekin RBD agarose beads, then the amount of RhoA-GTP was evaluated by performing SDS PAGE with an anti-RhoA antibody (Cell signaling Technology, Inc., Danvers, MA, USA).
Phosphorylation of Akt, mitogen-activated protein kinase (MAPK) ERK 1/2 and p38 was evaluated by performing SDS PAGE with anti-Akt1, anti-phospho-Akt (Santa Cruz Biotechnology, Santa Cruz, CA, USA), anti-p42-p44, anti-phospho-p42-p44, anti-p38 and anti-phospho-p38 (Cell signaling) antibodies. VEGFA and fibroblast growth factor 2 (FGF2) proteins were detected with anti-VEGF (A20) and anti-FGF2 (147) antibodies (Santa Cruz Biotechnology) respectively.
Immunostaining
Cells were fixed with 100% methanol for 10 minutes at 4°C. Immunostaining was performed with goat anti-Actin antibody 1/50 (Santa Cruz Biotechnology) or with rabbit anti-Ezrin antibody 1/150 (Upstate). Normal goat IgG (Santa-Cruz Biotechnology) or rabbit IgG (Santa-Cruz Biotechnology) was used as controls, and secondary antibodies were obtained from Jackson ImmunoResearch Laboratories (West Grove, PA, USA). Nuclei were stained with DAPI 1/5000.
Corneas were fixed and dissected in methanol, then embedded in paraffin. Sections were stained with rat anti-CD31 antibodies (BD Biosciences), goat anti-MMP-9 antibodies (Santa Cruz Biotechnology), and goat anti-OPN antibodies (R&D Systems Inc., Minneapolis, MN, USA); primary antibodies were visualized with Alexa-Fluor–conjugated secondary antibodies (Invitrogen). Nuclei were stained with DAPI (1/5000).
Mouse corneal angiogenesis assay
C57BL/6J, C3HeB/FeJ, and FVB wild-type mice, MMP-9-null mice, and Gli3-haploinsufficient (Gli3XtJ) mice were obtained from The Jackson Laboratory, Bar Harbor, ME, USA. OPN-null mice were kindly provided by Dr. S. R Rittling [25], and Gli1-null (Gli1LacZ/LacZ) mice were kindly provided by Dr. A. Dlugosz [26]. Pellets were prepared and implanted in the corneas of 6- to 8-week-old mice as previously described [27]. Eight days after pellet implantation, mice were injected with 50 μL fluorescien-BS1-Lectin I (Vector Laboratories Inc.) and sacrificed 15 minutes later, then eyes were harvested and fixed with 1% paraformaldehyde, and corneas were excised and prepared for fluorescent microscopy as detailed in the Supplemental Methods.
Statistics
Results are reported as mean±SD. Comparisons within groups were evaluated via analysis of variance and comparisons between individual groups were evaluated with the Student’s t test.
Results
Shh promotes EC migration and capillary morphogenesis
The direct effects of Shh on EC angiogenic activity were investigated in vitro with two cultured EC lines: BAECs and HUVECs. In the modified Boyden chamber assay, BAEC migration increased at progressively higher Shh concentrations, reaching a maximum at 100 ng/mL Shh (191±35% of migration in the absence of Shh, p=9×10−5) (Figure 1A); maximum HUVEC migration (280±82%, p=1×10−25) was observed at 10 ng/mL Shh (Figure 1B). Pretreatment with cyclopamine, which interrupts Shh signaling by inhibiting Smo, abolished Shh-induced migration in both cell lines (Supplemental Figure 1), thereby demonstrating that the enhanced migration was an Shh-specific effect. Capillary morphogenesis assays in Matrigel™ indicated that Shh concentrations as low as 10 ng/mL significantly promote the formation of tube-like structures (Figure 1C) by BAECs; total tube length increased to 271±50% (p=3×10−5) of the length attained in the absence of Shh. Shh treatment did not significantly alter the proliferation of BAECs (Figure 1D) or HUVECs (Figure 1E) when assessed via BrdU uptake.
Figure 1. Shh significantly induces EC migration and tube formation.
50×103 (A) serum-starved BAECs or (B) cultured HUVECs were seeded in the upper chamber of a modified Boyden chamber; the lower chamber contained 0–1000 ng/mL Shh. Migration was evaluated 6 hours later. (C) BAECs were suspended and cultured on Matrigel™ with or without 10 ng/mL Shh. Six hours later, tube formation was assessed under a phase-contrast microscope. Tube formation was quantified as the total length of tubes per high-power field. Results are presented as a percentage of measurements obtained in the absence of Shh. (D) Serum-starved BAECs and (E) HUVECs were treated with 0–1000 ng/mL Shh for 24 hours in the presence of 200 μM BrdU, then cells were stained for BrdU uptake with anti-BrdU antibodies (green); nuclei were stained with DAPI (blue). Proliferation was quantified as the number of BrdU+ cells. Three independent experiments, n=4 per condition for each experiment. ***p≤0.001 versus 0 ng/mL Shh, **p≤0.01 versus 0 ng/mL Shh; NS indicates not significant (p>0.05).
Shh does not alter the expression of VEGF or Ang1 in ECs but increases MMP-9 and OPN expression
Previously, we have shown that Shh increases the expression of several angiogenic factors, including VEGF and Ang1, in fibroblasts. Here, we determined whether Shh treatment increased VEGF and Ang1 expression in ECs by performing real-time RT-PCR analyses for mRNA expression and Western blot analyses for protein expression. Neither VEGF nor Ang1 expression differed significantly in HUVECs treated with 0–1000 ng/mL Shh for 24 or 48 hours (Figures 2A–D). However, exposure to 100 ng/mL Shh enhanced the expression of MMP-9 and OPN to 488±68% (p=1.52×10−4) and 230±35% (p=2.04×10−5), respectively, of the levels measured in untreated cells (Figures 2E–F).
Figure 2. OPN and MMP-9 activity is essential for in vitro Shh-induced EC migration.
(A–B) HUVECs were cultured with 0–1000 ng/mL Shh for 24 hours, then (A) VEGFA and (B) Ang1 mRNA expression was analyzed by real time RT-PCR and normalized to 18S rRNA expression. (C–D) HUVECs were cultured with 0–1000 ng/mL Shh for 24 hours or 48 hours, then (C) VEGFA and (D) Ang1 protein expression was analyzed by Western blot; α-tubulin expression was evaluated to control for loading. (E–F) HUVECs were cultured with 0 or 100 ng/mL Shh for 6 hours, then (E) MMP-9 and (F) OPN mRNA expression was analyzed by real-time RT-PCR and normalized to 18S rRNA expression. (G) HUVECs were transfected with GFP siRNA, MMP-9 siRNA, or OPN siRNA and cultured for 48 hours, then 50×103 cells were seeded in the upper chamber of a Boyden chamber; the lower chamber contained 0 or 100 ng/mL Shh. Migration was evaluated 6 hours later. Migration is presented as a percentage of the measurement obtained for GFP-siRNA–transfected cells in the absence of Shh. Panels A, B, E–G: 3 independent experiments, n=3 per condition for each experiment. Panels C–D: 3 independent experiments, n=1 per condition for each experiment. ***p≤0.001, **p≤0.01; NS indicates not significant (p>0.05).
Shh-induced EC migration is dependent on MMP-9 and OPN
The role of MMP-9 and OPN in Shh-induced EC migration was evaluated by transfecting HUVECs with MMP-9 siRNA, OPN siRNA, or a control GFP siRNA; migration toward 100 ng/mL Shh was assessed 48 hours later. When either MMP-9 or OPN mRNA expression was inhibited (Figure 2G), Shh no longer induced HUVEC migration, indicating that MMP-9 and OPN are essential for Shh-induced EC migration.
Shh does not activate Gli expression or Gli-dependent transcription in ECs
To determine whether Shh increased Gli expression in ECs, Gli1, Gli2, and Gli3 mRNA expression was measured via real-time RT-PCR in HUVECs, HCAECs, and NIH3T3 fibroblasts; Gli mRNA expression was also measured in HeLa cells to serve as a positive control for the human cell lines. HUVECs and HCAECs expressed substantial amounts of Gli3 mRNA, but very little Gli1 or Gli2 mRNA, and Gli expression did not change significantly after Shh treatment (Supplemental Figures 2A–C); however, Gli1 and Gli2 expression were enhanced 200- and 2.5-fold, respectively, in NIH 3T3 fibroblasts treated with Shh.
The effect of Shh treatment on Gli-dependent transcription was evaluated in HUVECs, BAECs, and NIH3T3 fibroblasts transfected with a Gli-luciferase reporter plasmid; in transfected NIH3T3 fibroblasts, Shh treatment typically increases luciferase activity 7- to 14-fold [19]. Shh treatment yielded no change in luciferase activity in HUVECs or BAECs (Figures 3A–B), but substantially increased luciferase activity in NIH3T3 fibroblasts (Figure 3C). The lack of Shh-induced Gli transcriptional activity is also supported by the absence of enhanced Ptch1 mRNA expression in Shh-treated HUVECs (Figure 3D).
Figure 3. Shh does not activate Gli-dependent transcription in ECs.

(A) HUVECs, (B) BAECs, and (C) NIH3T3 fibroblasts were transiently co-transfected with GliBS luciferase or mutGli luciferase and RSV-β-galactosidase plasmids, cultured for 24 hours, then treated with 0–1000 ng/mL Shh for 6 or 24 hours. Cell lysates were assayed for luciferase and β-galactosidase activity, and luciferase activity was normalized to β-galactosidase activity to compensate for differences in transfection efficiency. (D) HUVECs were cultured with 0–1000 ng/mL Shh for 24 hours, then Ptch1 mRNA expression was analyzed by real time RT-PCR and normalized to 18S rRNA expression. Three independent experiments, n=3 per condition for each experiment. ***p≤0.001; NS indicates not significant (p>0.05).
Shh-induced EC migration, tube formation, and gene expression are mediated by the Rho/ROCK pathway
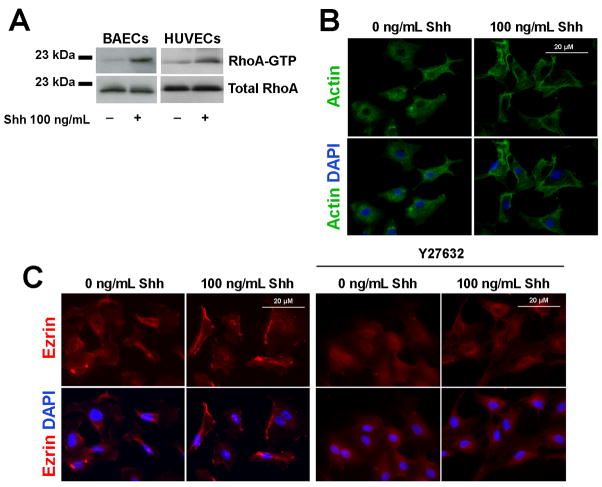
The phosphorylation states of several kinases were assayed by Western blot in ECs treated with 0–1000 ng/mL Shh for 30 minutes. Shh increased Akt phosphorylation slightly (Supplemental Figure 3A) in BAECs, which is consistent with the findings of Kanda et al. [16], but had no effect on the phosphorylation of p38 and ERK 1/2 MAPK or on COUP TFII mRNA expression in HUVECs (Supplemental Figure 3B). However, just 5 minutes of Shh treatment increased phosphorylation of the G-protein Rho in BAECs and HUVECs (Figure 4A).
Figure 4. Shh activates RhoA in ECs.
(A) Serum-starved BAECs and HUVECs were treated with 0 or 100 ng/mL Shh for 5 minutes, then the level of RhoA-GTP was evaluated with the Rho pull down assay. (B) Serum-starved BAECs were treated with 0 or 100 ng/mL Shh for 10 minutes, then the actin cytoskeleton was stained with an anti-actin antibody (green), and nuclei were stained with DAPI (blue). (C) HUVECs cultured in 1% FBS-containing medium were treated with 0 or 100 ng/mL Shh in the presence or absence of 20 nM Y27632 for 10 minutes, then stained for Ezrin (red); nuclei were stained with DAPI (blue). Panel A: 3 independent experiments, n=1 per condition for each experiment; Panels B–C: 3 independent experiments, n=1 per condition for each experiment.
Because the Rho proteins are involved in cytoskeletal rearrangement and phosphorylate the Ezrin/Radixin/Moesin proteins [28], ECs were treated with or without Shh for 10 minutes, then the actin cytoskeleton and Ezrin localization were examined by immunostaining. Shh treatment was associated with evidence of actin cytoskeletal rearrangement and with Ezrin relocation to the membrane ruffles (Figures 4B–C); furthermore, Shh-induced Ezrin relocalization was abolished by the presence of the ROCK inhibitor Y27632. Taken together, these results indicate that Shh rapidly activated the Rho/ROCK pathway in ECs, which subsequently led to cytoskeletal changes that are associated with EC motility.
The role of RhoA and ROCK during Shh-induced EC activity was investigated by treating ECs with Y27632 and by transfecting BAECs with plasmids that expressed dominant-negative mutations for RhoA (RhoA N19), ROCK (ROCK RB/PH) [22], or Ezrin (Ez NTer). Shh-induced EC migration was inhibited by both Y27632 (Figures 5A–B) and the expression of RhoAN19 or ROCK RB/PH (Figure 5C), and RhoA N19 expression inhibited Shh-induced tube formation (Figure 5D). Shh-induced EC migration was also inhibited in cells transfected with the Ez NTer plasmid (Figure 5E), indicating that Ezrin, a downstream target of ROCK [28], is also involved in Shh-induced EC migration. The Shh-induced expression of both OPN and MMP-9 (Figures 5F–G) was inhibited in the presence of Y27632, suggesting that the Rho/ROCK pathway is essential for Shh-induced gene expression in ECs.
Figure 5. Shh-induced EC migration, tube formation, and gene expression are dependent on ROCK.
(A) HUVECs and (B) BAECs were pretreated with 20 ng/mL Y27632, or control vehicle for 30 minutes then seeded in the upper chamber of a modified Boyden chamber; the lower chamber contained the indicated concentration of Shh. Migration was evaluated 6 hours later and expressed as the fold-change from measurements obtained in the absence of Shh. (C–D) BAECs were transfected with plasmids containing an empty vector (pcDNA3) or plasmids expressing RhoA N19 or ROCK RB/PH, then starved for 24 hours in serum-free medium. (C) 50×103 transfected BAECs were seeded in the upper chamber of a modified Boyden chamber; the lower chamber contained 0 or 100 ng/mL Shh. Migration was evaluated 6 hours later. (D) Transfected BAECs were suspended and cultured on Matrigel™ with or without 10 ng/mL Shh; tube formation was assessed 6 hours later, quantified as the total length of tubes per high-power field. (E) BAECs were transfected with plasmids containing an empty vector (pCB6) or dominant-negative Ezrin (Ez Nter), starved for 24 hours in serum-free medium, then 50×103 cells were seeded in the upper chamber of a modified Boyden chamber; the lower chamber contained 0 or 100 ng/mL Shh. Migration was evaluated 6 hours later. The results in panels C–E are presented as a percentage of the measurements obtained for pcDNA3- or pCB6-transfected cells in the absence of Shh. (F–G) HUVECs were treated with 0 or 100 ng/mL Shh in the presence of 20 ng/mL Y27632, or control vehicle for 6 hours. (F) OPN and (G) MMP-9 mRNA expression were analyzed by real time RT-PCR and normalized to 18S rRNA expression. Three independent experiments, n=3 per condition for each experiment. ***p≤0.001, **p≤0.01, *p≤0.05.
Shh-induced corneal angiogenesis is dependent on the activity of Smo and the Rho/ROCK pathway and on MMP-9 and OPN expression, but not on Gli expression
The potential involvement of Smo, the Rho/ROCK pathway, MMP-9, OPN, and the Gli transcription factors in Shh-induced angiogenesis was assessed in vivo with the mouse corneal angiogenesis model; the pro-angiogenic effect of Shh in this model has been demonstrated previously [11].
The involvement of Smo and the Rho/ROCK pathway were investigated in wild-type (WT) mice. Smo was evaluated by implanting pellets containing phosphate-buffered saline (PBS) or cyclopamine alone and in combination with Shh in the corneas of WT mice, then monitoring angiogenesis via in vivo fluorescein-BS-1 lectin perfusion. When paired with PBS, Shh dramatically increased angiogenesis, but this enhancement was abolished by the presence of cyclopamine (Figure 6). To assess the contribution of the Rho/ROCK pathway, pellets containing PBS, Shh, Y27632, or Shh and Y27632 were implanted. Pellets containing Shh alone substantially increased angiogenesis, but no enhancement was observed after transplantation of pellets containing both Shh and Y27632. Collectively, these observations indicate that both Smo and the RhoA/ROCK pathway contribute to Shh-induced angiogenesis.
Figure 6. Shh-induced corneal angiogenesis is dependent on the activity of Smo and the Rho/ROCK pathway and on MMP-9 and OPN expression, but not on Gli expression.
Pellets containing the indicated combinations of PBS, Shh, the Smo inhibitor cyclopamine (CYC), and the ROCK inhibitor Y27632 were implanted in the corneas of WT mice, MMP-9-knockout mice (MMP-9−/−) and their WT littermates, OPN-knockout mice (OPN−/−) and their WT littermates, Gli1-knockout (Gli1−/−) C57BL/6 mice, WT C57BL/6 mice, Gli3-haploinsufficient (Gli3+/−) C3HeB/FeJ mice, and WT C3HeB/FeJ mice; angiogenesis was evaluated 8 days later via in vivo fluorecein-BS-1 lectin injection. n≥4 mice (8 corneas) per group
We confirmed that Shh increased MMP-9 and OPN expression in the corneas of WT mice by implanting Shh- or PBS-containing pellets and evaluating mRNA expression 7 days later. Corneal levels of both MMP-9 and OPN mRNA were higher after implantation with the Shh pellet than after PBS-pellet implantation (Supplemental Figures 4A–B). Corneal sections double-stained for MMP-9 or OPN and the endothelial-cell marker CD31 (Supplemental Figure 4C) indicated that OPN, but not MMP-9, was expressed by ECs. To determine whether MMP-9 and OPN contributed to Shh-induced angiogenesis, Shh- or PBS-containing pellets were implanted in the corneas of MMP-9- or OPN-null mice and their WT littermates. Shh induced angiogenesis in the corneas of WT mice, but this enhancement was impaired in the corneas of MMP-9-null and OPN-null mice (Figure 6). These results demonstrate that both MMP-9 and OPN are essential for in vivo Shh-induced angiogenesis, and that the contribution of OPN evolves, at least in part, from ECs, whereas MMP-9 functions through different cell types.
Because Gli1 is the strongest transactivator among the Gli transcription factors [29], and Gli3 is expressed in ECs (Supplemental Figure 2C), Gli activation during Shh-induced angiogenesis was tested in Gli1-null mice and in Gli3-haploinsufficient mice. Shh-induced angiogenesis was similar in WT and Gli1-null mice and in WT and Gli3-haploinsufficient mice (Figure 6), which suggests that the Gli transcription factors may not be involved in Shh-induced angiogenesis in adult mice.
Discussion
Previous investigations indicate that Shh is upregulated in ischemic tissue and enhances revascularization [11–12]. Shh is known to activate several transduction pathways, and most involve the modulation of Gli activity. For example, Shh-induced activation of the Rho/ROCK pathway promoted Gli3 nuclear translocation [20] in neuronal cells, and the PKCδ, MAPK ERK 1/2, and PI3K/Akt pathways have been implicated in Shh-induced, Gli-dependent transcription in NIH 3T3 fibroblasts [19, 30]. Shh induces expression of several pro-angiogenic factors, such as VEGF and Ang1, in fibroblasts and cardiomyocytes [11–12].
The results reported here indicate that Shh-induced EC migration and capillary morphogenesis does not occur through the classical Shh mechanism: Gli transcriptional activation. Instead, Shh activated the Rho/ROCK pathway, which is consistent with the findings of a recent report [31]. Our results also demonstrate that Shh-induced ROCK activation increases the expression of OPN and MMP-9, and that these two factors are essential for Shh-induced EC migration. OPN is an RGD-containing extracellular matrix protein that induces angiogenesis, both in vivo and in vitro, by promoting EC survival and migration [32], and matrix metalloproteinases such as MMP-9 are believed to promote cell migration [33] by degrading type IV collagen in the basement membranes of vessels and the extracellular matrix [34].
ROCK regulates cell polarity and migration by inducing cellular contractions, protrusions, and focal adhesions. Like Shh, VEGF activates ROCK in ECs, which leads to the phosphorylation of focal adhesion kinase and subsequent EC migration [35], and VEGF-induced EC migration and tube formation are impeded by the ROCK inhibitor Y27632 [36]. ROCK also regulates the actin-binding proteins Ezrin, Radixin, and Moesin. RhoA colocalizes with Ezrin in the lamellipodia and ruffles of ECs [37], and phosphorylation of Moesin by ROCK has been implicated in estradiol-induced EC migration [38].
The PI3K/Akt pathway is activated by Shh in ECs, but is not involved in Shh-induced Gli1 nuclear translocation [16]. ROCK inhibits the PI3K/Akt pathway and the expression of endothelial nitrous oxide synthase [39], which could lead to detrimental effects in the vascular system; however, little is known about the role of ROCK during angiogenesis in vivo. Y27632 has been shown to attenuate hypoxia-induced angiogenesis in the lung [40], and fasudil, another ROCK inhibitor, inhibited VEGF-induced angiogenesis in an in vivo Matrigel™ assay [41] and in the mouse corneal angiogenesis model [42]. Conditional activation of ROCK has been shown to promote tumor angiogenesis [43].
Gli1 is not essential for initial Shh signaling and ectopic activation of the Hh pathway during development [26], and Bijlsma et al. have recently presented evidence indicating that Shh-induced fibroblast migration and cytoskeletal rearrangement may not require Gli activation [44]. Here, significant (14-fold) activation of Gli-dependent transcription was not apparent in NIH3T3 fibroblasts until 16 hours after Shh stimulation, whereas the Rho/ROCK-mediated modifications of Ezrin localization and cytoskeletal actin in ECs were observed 10 minutes after Shh treatment, and increases in MMP-9 and OPN mRNA expression as well as EC migration were detected within 6 hours. Furthermore, Shh did not increase Gli expression in ECs, and Shh-induced angiogenesis was intact in Gli1-null and Gli3-haploinsufficient mice. Nevertheless, we cannot exclude the potential involvement of Gli transcription factors in Shh-induced angiogenesis, because compensatory mechanisms could alleviate the loss or downregulation of any single Gli transcription factor.
In conclusion, our current investigation identifies a novel mechanism for Shh-induced neovascularization: the direct enhancement of EC migration and tube formation via ROCK-mediated increases in OPN and MMP-9 expression. Several of these effects (e.g., increased migration, MMP-9 expression, and OPN expression) were induced by a Shh concentration of 10 ng/mL, which is likely similar to physiological Shh concentrations. It is reasonable to consider whether these Shh-induced, ROCK-mediated effects support the function of other nearby cells, such as EPCs, smooth muscle cells [45–46], and muscle-derived progenitor cells [47], and whether Shh activation of the PI3K/Akt pathway, reported in ECs [16], EPCs [48], and fibroblasts [30] in vitro, may also induce favorable effects. These data cumulatively indicate that Shh has an important and complex role in neovascularization, and the diverse nature of Shh activity could provide many promising approaches to therapeutic revascularization.
Supplementary Material
Acknowledgments
This work was supported by NIH Grants HL53354, HL57516, HL80137 and HL95874 (DWL). The authors report no conflicts of interest.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Bitgood MJ, Shen L, McMahon AP. Sertoli cell signaling by Desert hedgehog regulates the male germline. Curr Biol. 1996;6:298–304. doi: 10.1016/s0960-9822(02)00480-3. [DOI] [PubMed] [Google Scholar]
- 2.Chiang C, Litingtung Y, Lee E, Young KE, Corden JL, Westphal H, et al. Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature. 1996;383:407–13. doi: 10.1038/383407a0. [DOI] [PubMed] [Google Scholar]
- 3.Johnson RL, Tabin CJ. Molecular models for vertebrate limb development. Cell. 1997;90:979–90. doi: 10.1016/s0092-8674(00)80364-5. [DOI] [PubMed] [Google Scholar]
- 4.Parmantier E, Lynn B, Lawson D, Turmaine M, Namini SS, Chakrabarti L, et al. Schwann cell-derived Desert hedgehog controls the development of peripheral nerve sheaths. Neuron. 1999;23:713–24. doi: 10.1016/s0896-6273(01)80030-1. [DOI] [PubMed] [Google Scholar]
- 5.Pepicelli CV, Lewis PM, McMahon AP. Sonic hedgehog regulates branching morphogenesis in the mammalian lung. Curr Biol. 1998;8:1083–6. doi: 10.1016/s0960-9822(98)70446-4. [DOI] [PubMed] [Google Scholar]
- 6.Ramalho-Santos M, Melton DA, McMahon AP. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development. 2000;127:2763–72. doi: 10.1242/dev.127.12.2763. [DOI] [PubMed] [Google Scholar]
- 7.St-Jacques B, Dassule HR, Karavanova I, Botchkarev VA, Li J, Danielian PS, et al. Sonic hedgehog signaling is essential for hair development. Curr Biol. 1998;8:1058–68. doi: 10.1016/s0960-9822(98)70443-9. [DOI] [PubMed] [Google Scholar]
- 8.St-Jacques B, Hammerschmidt M, McMahon AP. Indian hedgehog signaling regulates proliferation and differentiation of chondrocytes and is essential for bone formation. Genes Dev. 1999;13:2072–86. doi: 10.1101/gad.13.16.2072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rowitch DH, S-Jacques B, Lee SM, Flax JD, Snyder EY, McMahon AP. Sonic hedgehog regulates proliferation and inhibits differentiation of CNS precursor cells. J Neurosci. 1999;19:8954–65. doi: 10.1523/JNEUROSCI.19-20-08954.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brown LA, Rodaway AR, Schilling TF, Jowett T, Ingham PW, Patient RK, et al. Insights in to early vasculogenesis revealed by expression of the ETS-domain transcription factor Fli-1 in type and mutant zebrafish embryos. Mech Dev. 2000;90:237–52. doi: 10.1016/s0925-4773(99)00256-7. [DOI] [PubMed] [Google Scholar]
- 11.Pola R, Ling LE, Silver M, Corbley MJ, Kearney M, Blake Pepinsky R, et al. The morphogen Sonic hedgehog is an indirect angiogenic agent upregulating two families of angiogenic growth factors. Nat Med. 2001;7:706–11. doi: 10.1038/89083. [DOI] [PubMed] [Google Scholar]
- 12.Kusano KF, Pola R, Murayama T, Curry C, Kawamoto A, Iwakura A, et al. Sonic hedgehog myocardial gene therapy: tissue repair through transient reconstitution of embryonic signaling. Nat Med. 2005;11:1197–204. doi: 10.1038/nm1313. [DOI] [PubMed] [Google Scholar]
- 13.Asai J, Takenaka H, Kusano KF, Ii M, Luedemann C, Curry C, et al. Topical sonic hedgehog gene therapy accelerates wound healing in diabetes by enhancing endothelial progenitor cell-mediated microvascular remodeling. Circulation. 2006;113:2413–24. doi: 10.1161/CIRCULATIONAHA.105.603167. [DOI] [PubMed] [Google Scholar]
- 14.Pola R, Ling LE, Aprahamian TR, Barban E, Bosch-Marce M, Curry C, et al. Postnatal recapitulation of embryonic hedgehog pathway in response to skeletal muscle ischemia. Circulation. 2003;108:479–85. doi: 10.1161/01.CIR.0000080338.60981.FA. [DOI] [PubMed] [Google Scholar]
- 15.Villavicencio EH, Walterhouse DO, Iannaccone PM. The sonic hedgehog-patched-gli pathway in human development and disease. Am J Hum Genet. 2000;67:1047–54. doi: 10.1016/s0002-9297(07)62934-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kanda S, Mochizuki Y, Suematsu T, Miyata Y, Nomata K, Kanetake H. Sonic hedgehog induces capillary morphogenesis by endothelial cells through phosphoinositide 3-kinase. J Biol Chem. 2003;278:8244–9. doi: 10.1074/jbc.M210635200. [DOI] [PubMed] [Google Scholar]
- 17.Sheng T, Chi S, Zhang X, Xie J. Regulation of Gli1 localization by the cAMP/protein kinase A signaling axis through a site near the nuclear localization signal. J Biol Chem. 2006;281:9–12. doi: 10.1074/jbc.C500300200. [DOI] [PubMed] [Google Scholar]
- 18.Krishnan V, Pereira FA, Qiu Y, Chen CH, Beachy PA, Tsai SY, et al. Mediation of Sonic hedgehog-induced expression of COUP-TFII by a protein phosphatase. Science. 1997;278(5345):1947–50. doi: 10.1126/science.278.5345.1947. [DOI] [PubMed] [Google Scholar]
- 19.Riobo NA, Haines GM, Emerson CP., Jr Protein kinase C-delta and mitogen-activated protein/extracellular signal-regulated kinase-1 control GLI activation in hedgehog signaling. Cancer Res. 2006;66:839–45. doi: 10.1158/0008-5472.CAN-05-2539. [DOI] [PubMed] [Google Scholar]
- 20.Kasai K, Takahashi M, Osumi N, Sinnarajah S, Takeo T, Ikeda H, et al. The G12 family of heterotrimeric G proteins and Rho GTPase mediate Sonic hedgehog signalling. Genes Cells. 2004;9:49–58. doi: 10.1111/j.1356-9597.2004.00701.x. [DOI] [PubMed] [Google Scholar]
- 21.Sasaki H, Hui C, Nakafuku M, Kondoh H. A binding site for Gli proteins is essential for HNF-3beta floor plate enhancer activity in transgenics and can respond to Shh in vitro. Development. 1997;124:1313–22. doi: 10.1242/dev.124.7.1313. [DOI] [PubMed] [Google Scholar]
- 22.Amano M, Chihara K, Kimura K, Fukata Y, Nakamura N, Matsuura Y, et al. Formation of actin stress fibers and focal adhesions enhanced by Rho-kinase. Science. 1997;275:1308–11. doi: 10.1126/science.275.5304.1308. [DOI] [PubMed] [Google Scholar]
- 23.Algrain M, Turunen O, Vaheri A, Louvard D, Arpin M. Ezrin contains cytoskeleton and membrane binding domains accounting for its proposed role as a membrane-cytoskeletal linker. J Cell Biol. 1993;120:129–39. doi: 10.1083/jcb.120.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Renault MA, Jalvy S, Belloc I, Pasquet S, Sena S, Olive M, et al. AP-1 is involved in UTP-induced osteopontin expression in arterial smooth muscle cells. Circ Res. 2003;93:674–81. doi: 10.1161/01.RES.0000094747.05021.62. [DOI] [PubMed] [Google Scholar]
- 25.Rittling SR, Matsumoto HN, McKee MD, Nanci A, An XR, Novick KE, et al. Mice lacking osteopontin show normal development and bone structure but display altered osteoclast formation in vitro. J Bone Miner Res. 1998;13:1101–11. doi: 10.1359/jbmr.1998.13.7.1101. [DOI] [PubMed] [Google Scholar]
- 26.Bai CB, Auerbach W, Lee JS, Stephen D, Joyner AL. Gli2, but not Gli1, is required for initial Shh signaling and ectopic activation of the Shh pathway. Development. 2002;129:4753–61. doi: 10.1242/dev.129.20.4753. [DOI] [PubMed] [Google Scholar]
- 27.Kenyon BM, Voest EE, Chen CC, Flynn E, Folkman J, D’Amato RJ. A model of angiogenesis in the mouse cornea. Invest Ophthalmol Vis Sci. 1996;37:1652–32. [PubMed] [Google Scholar]
- 28.Kishore R, Qin G, Luedemann C, Bord E, Hanley A, Silver M, et al. The cytoskeletal protein ezrin regulates EC proliferation and angiogenesis via TNF-alpha-induced transcriptional repression of cyclin A. J Clin Invest. 2005;115:1785–96. doi: 10.1172/JCI22849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sasaki H, Nishizaki Y, Hui C, Nakafuku M, Kondoh H. Regulation of Gli2 and Gli3 activities by an amino-terminal repression domain: implication of Gli2 and Gli3 as primary mediators of Shh signaling. Development. 1999;126:3915–24. doi: 10.1242/dev.126.17.3915. [DOI] [PubMed] [Google Scholar]
- 30.Riobo NA, Lu K, Ai X, Haines GM, Emerson CP., Jr Phosphoinositide 3-kinase and Akt are essential for Sonic Hedgehog signaling. Proc Natl Acad Sci U S A. 2006;103:4505–10. doi: 10.1073/pnas.0504337103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chinchilla P, Xiao L, Kazanietz MG, Riobo NA. Hedgehog proteins activate pro-angiogenic responses in endothelial cells through non-canonical signaling pathways. Cell Cycle. 2010:9. doi: 10.4161/cc.9.3.10591. [DOI] [PubMed] [Google Scholar]
- 32.Takagi H, Suzuma K, Otani A, Oh H, Koyama S, Ohashi H, et al. Role of vitronectin receptor-type integrins and osteopontin in ischemia-induced retinal neovascularization. Jpn J Ophthalmol. 2002;46:270–8. doi: 10.1016/s0021-5155(02)00482-3. [DOI] [PubMed] [Google Scholar]
- 33.Van Den Steen PE, Wuyts A, Husson SJ, Proost P, Van Damme J, Opdenakker G. Gelatinase B/MMP-9 and neutrophil collagenase/MMP-8 process the chemokines human GCP-2/CXCL6, ENA-78/CXCL5 and mouse GCP-2/LIX and modulate their physiological activities. Eur J Biochem. 2003;270:3739–49. doi: 10.1046/j.1432-1033.2003.03760.x. [DOI] [PubMed] [Google Scholar]
- 34.Creemers EE, Cleutjens JP, Smits JF, Daemen MJ. Matrix metalloproteinase inhibition after myocardial infarction: a new approach to prevent heart failure? Circ Res. 2001;89:201–10. doi: 10.1161/hh1501.094396. [DOI] [PubMed] [Google Scholar]
- 35.Le Boeuf F, Houle F, Sussman M, Huot J. Phosphorylation of focal adhesion kinase (FAK) on Ser732 is induced by rho-dependent kinase and is essential for proline-rich tyrosine kinase-2-mediated phosphorylation of FAK on Tyr407 in response to vascular endothelial growth factor. Mol Biol Cell. 2006;17:3508–20. doi: 10.1091/mbc.E05-12-1158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.van Nieuw Amerongen GP, Koolwijk P, Versteilen A, van Hinsbergh VW. Involvement of RhoA/Rho kinase signaling in VEGF-induced endothelial cell migration and angiogenesis in vitro. Arterioscler Thromb Vasc Biol. 2003;23:211–7. doi: 10.1161/01.atv.0000054198.68894.88. [DOI] [PubMed] [Google Scholar]
- 37.Menager C, Vassy J, Doliger C, Legrand Y, Karniguian A. Subcellular localization of RhoA and ezrin at membrane ruffles of human endothelial cells: differential role of collagen and fibronectin. Exp Cell Res. 1999;249:221–30. doi: 10.1006/excr.1999.4481. [DOI] [PubMed] [Google Scholar]
- 38.Simoncini T, Scorticati C, Mannella P, Fadiel A, Giretti MS, Fu XD, et al. Estrogen receptor alpha interacts with Galpha13 to drive actin remodeling and endothelial cell migration via the RhoA/Rho kinase/moesin pathway. Mol Endocrinol. 2006;20:1756–71. doi: 10.1210/me.2005-0259. [DOI] [PubMed] [Google Scholar]
- 39.Noma K, Oyama N, Liao JK. Physiological role of ROCKs in the cardiovascular system. Am J Physiol Cell Physiol. 2006;290:C661–8. doi: 10.1152/ajpcell.00459.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hyvelin JM, Howell K, Nichol A, Costello CM, Preston RJ, McLoughlin P. Inhibition of Rho-kinase attenuates hypoxia-induced angiogenesis in the pulmonary circulation. Circ Res. 2005;97:185–91. doi: 10.1161/01.RES.0000174287.17953.83. [DOI] [PubMed] [Google Scholar]
- 41.Yin L, Morishige K, Takahashi T, Hashimoto K, Ogata S, Tsutsumi S, et al. Fasudil inhibits vascular endothelial growth factor-induced angiogenesis in vitro and in vivo. Mol Cancer Ther. 2007;6:1517–25. doi: 10.1158/1535-7163.MCT-06-0689. [DOI] [PubMed] [Google Scholar]
- 42.Hata Y, Miura M, Nakao S, Kawahara S, Kita T, Ishibashi T. Antiangiogenic properties of fasudil, a potent Rho-Kinase inhibitor. Jpn J Ophthalmol. 2008;52:16–23. doi: 10.1007/s10384-007-0487-5. [DOI] [PubMed] [Google Scholar]
- 43.Croft DR, Sahai E, Mavria G, Li S, Tsai J, Lee WM, et al. Conditional ROCK activation in vivo induces tumor cell dissemination and angiogenesis. Cancer Res. 2004;64:8994–9001. doi: 10.1158/0008-5472.CAN-04-2052. [DOI] [PubMed] [Google Scholar]
- 44.Bijlsma MF, Borensztajn KS, Roelink H, Peppelenbosch MP, Spek CA. Sonic hedgehog induces transcription-independent cytoskeletal rearrangement and migration regulated by arachidonate metabolites. Cell Signal. 2007;19:2596–604. doi: 10.1016/j.cellsig.2007.08.011. [DOI] [PubMed] [Google Scholar]
- 45.Sung HJ, Johnson CE, Lessner SM, Magid R, Drury DN, Galis ZS. Matrix metalloproteinase 9 facilitates collagen remodeling and angiogenesis for vascular constructs. Tissue Eng. 2005;11:267–76. doi: 10.1089/ten.2005.11.267. [DOI] [PubMed] [Google Scholar]
- 46.Jalvy S, Renault MA, Leen LL, Belloc I, Bonnet J, Gadeau AP, et al. Autocrine expression of osteopontin contributes to PDGF-mediated arterial smooth muscle cell migration. Cardiovasc Res. 2007;75:738–47. doi: 10.1016/j.cardiores.2007.05.019. [DOI] [PubMed] [Google Scholar]
- 47.Ogata T, Ueyama T, Nomura T, Asada S, Tagawa M, Nakamura T, et al. Osteopontin is a myosphere-derived secretory molecule that promotes angiogenic progenitor cell proliferation through the phosphoinositide 3-kinase/Akt pathway. Biochem Biophys Res Commun. 2007;359:341–7. doi: 10.1016/j.bbrc.2007.05.104. [DOI] [PubMed] [Google Scholar]
- 48.Fu JR, Liu WL, Zhou JF, Sun HY, Xu HZ, Luo L, et al. Sonic hedgehog protein promotes bone marrow-derived endothelial progenitor cell proliferation, migration and VEGF production via PI 3-kinase/Akt signaling pathways. Acta Pharmacol Sin. 2006;27:685–93. doi: 10.1111/j.1745-7254.2006.00335.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.