Abstract
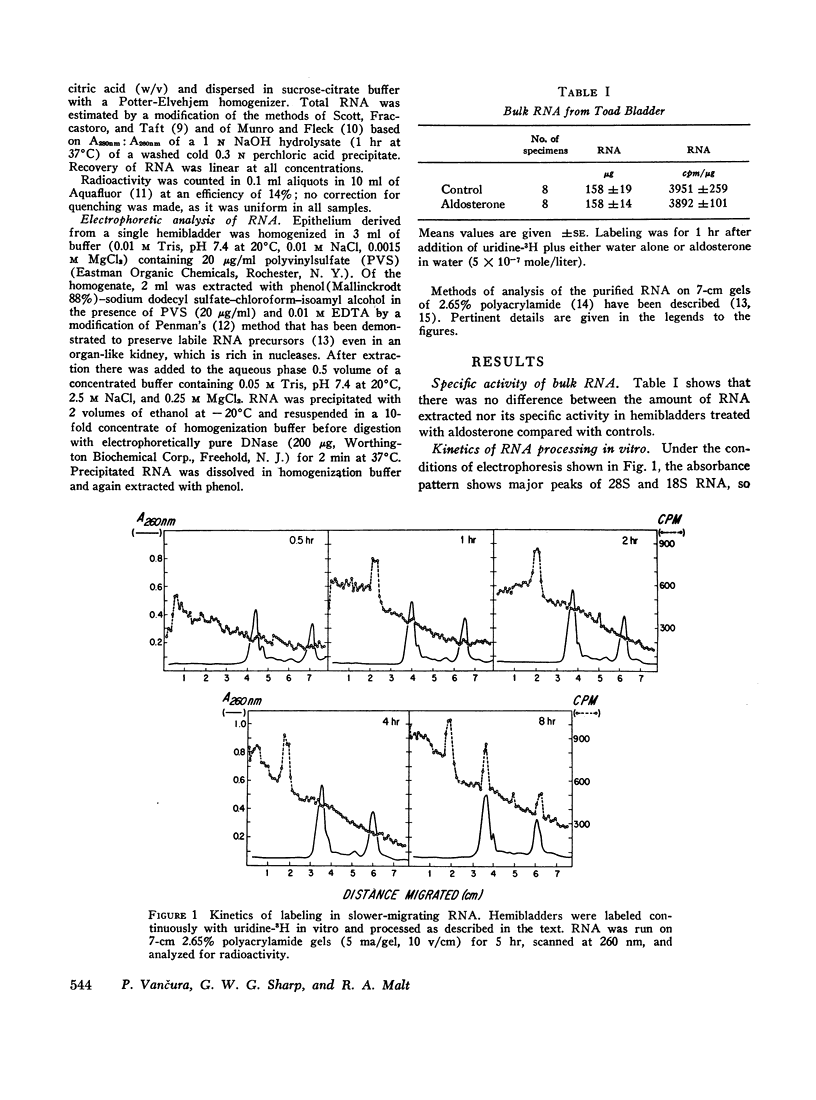
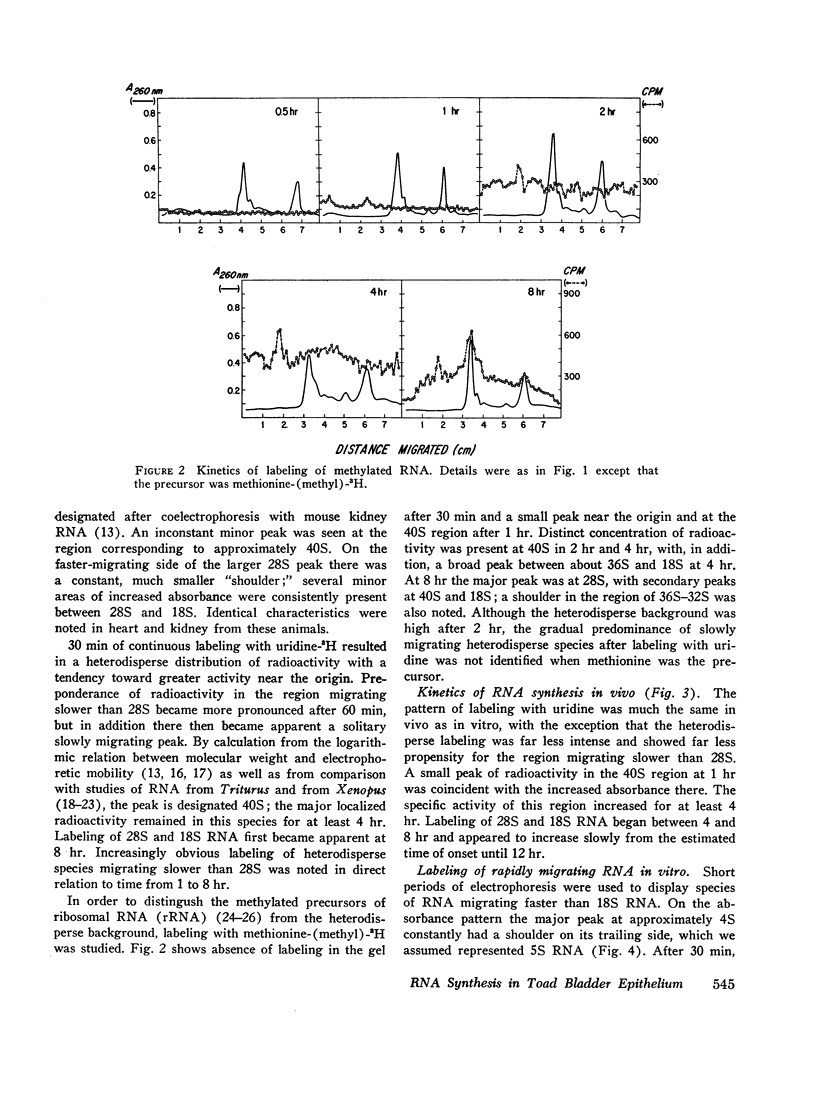
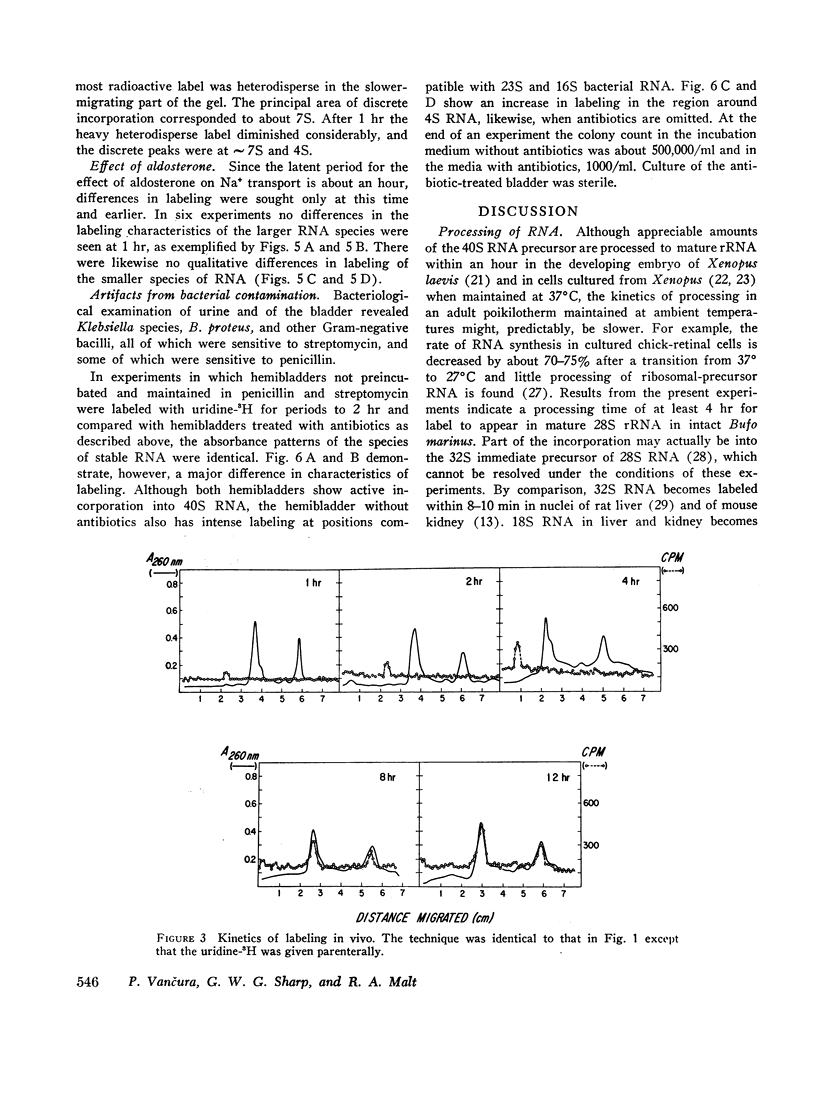
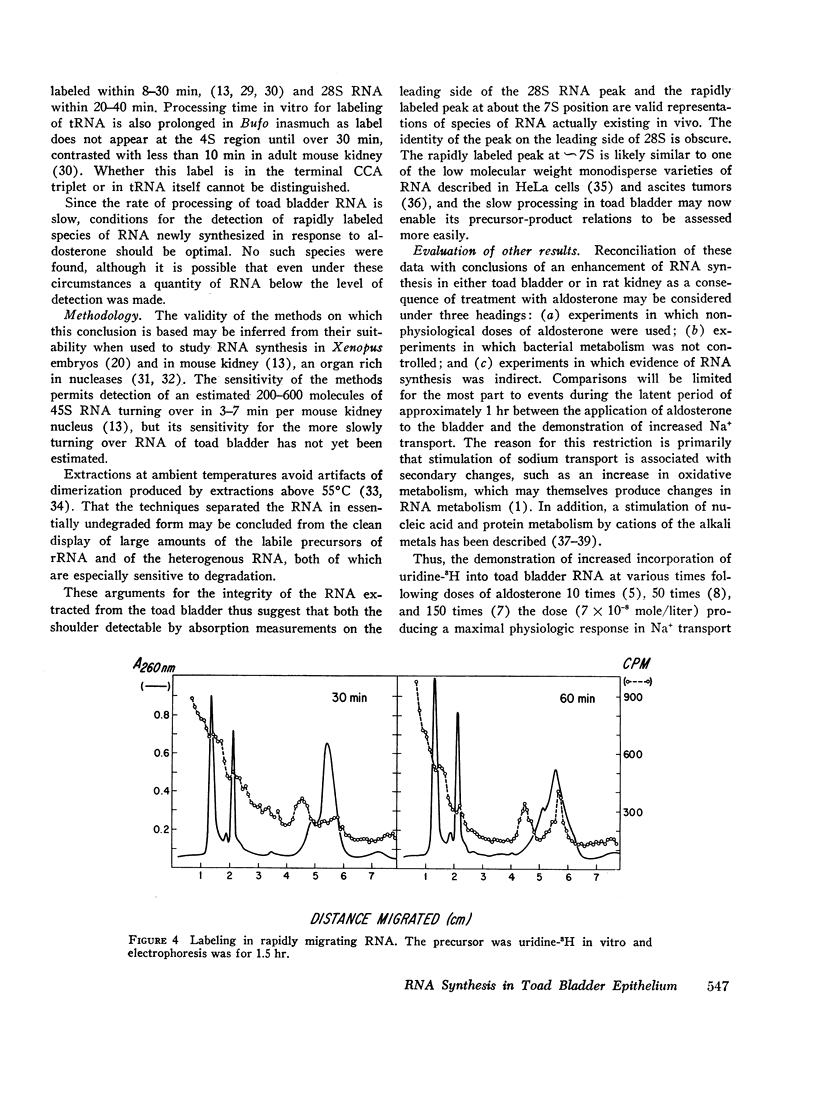
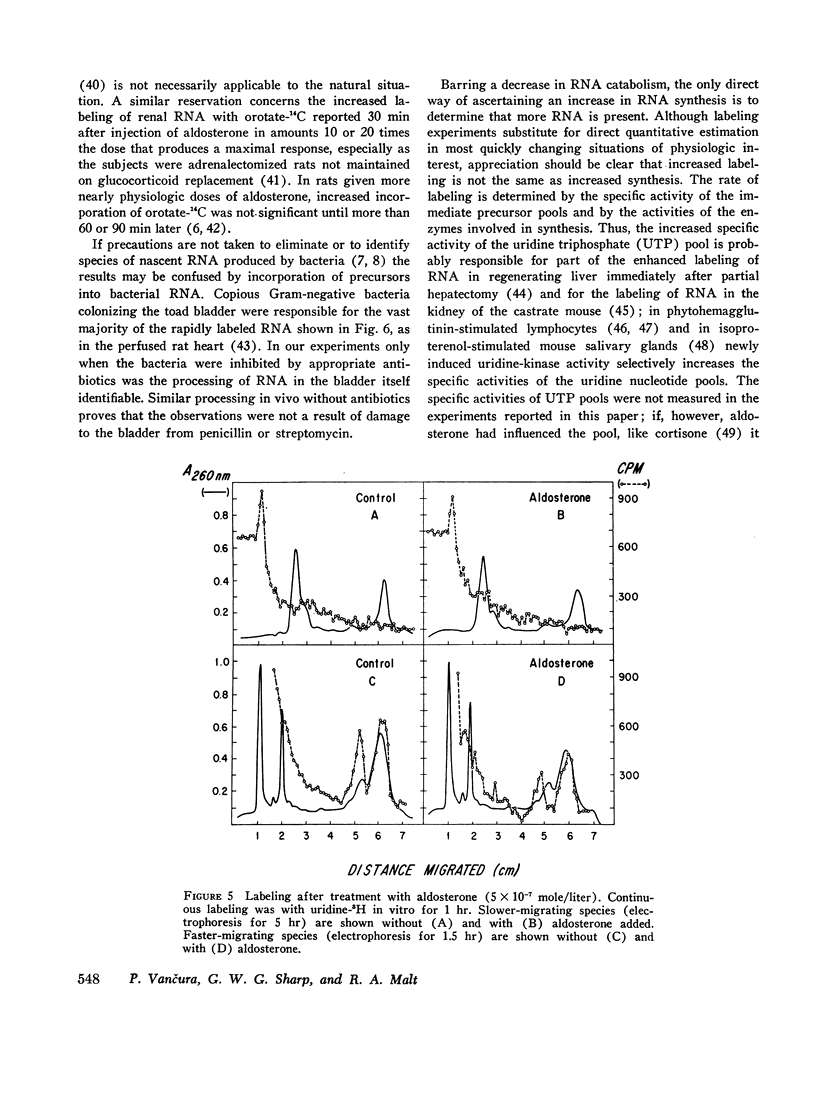
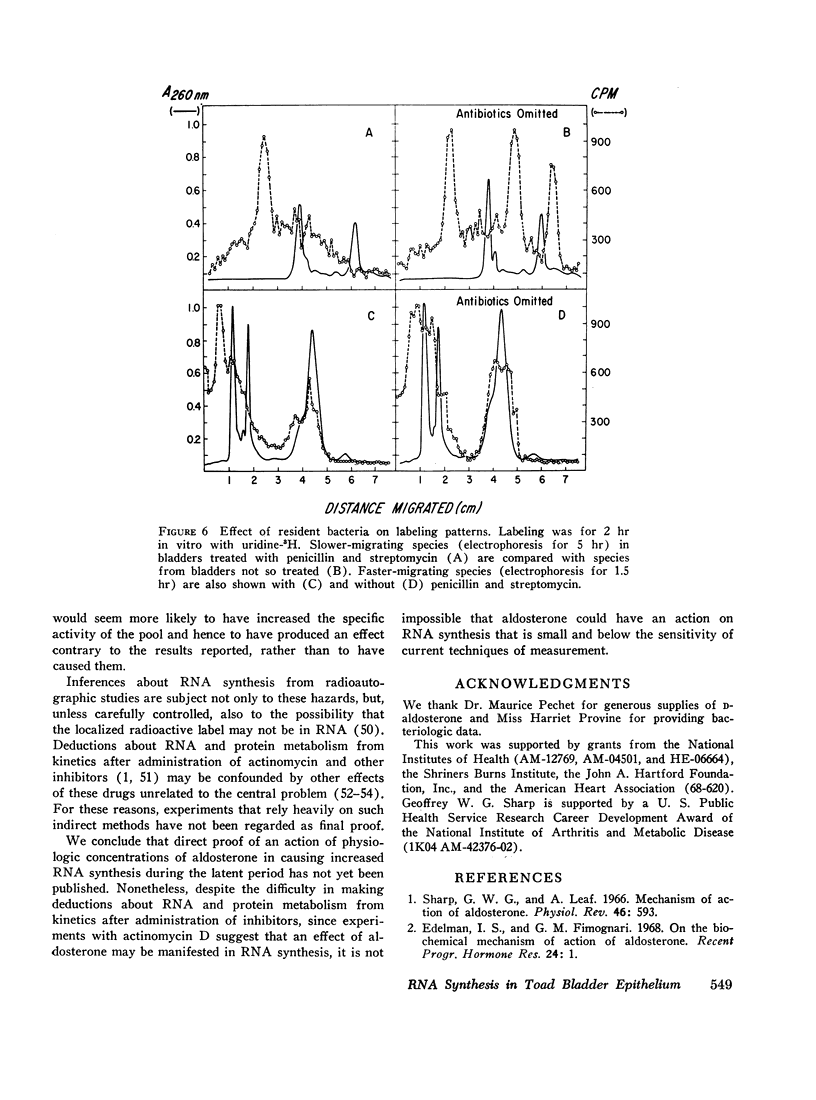
Processing of RNA in the toad bladder was analyzed by polyacrylamide-gel electrophoresis to determine whether aldosterone causes any changes in the 1 hr before it potentiates transport of sodium ion. No change was found in the quantity or in the specific activity of bulk RNA labeled with uridine-5-3H. In vivo and in vitro with either uridine-5-3H or with methionine-(methyl)-3H as precursors, processing of RNA was extremely slow. Heterodisperse RNA was obvious after 30 min of continuous labeling, but labeling of the 40S precursor of ribosomal RNA was not apparent for 60 min. Labeling of mature 28S and 18S RNA first became apparent after 8 hr. ∼7S RNA was the principal fastmigrating species labeled at 30 min, and 4S RNA was not heavily labeled until 1 hr. Aldosterone (5 × 10-7 mole/liter) produced no changes. If care were not taken to inhibit metabolism of native bacteria colonizing the bladder, bacterial RNA of high specific activity predominated. We conclude that RNA metabolism in the toad bladder is extraordinarily slow, that a major acceleration of de novo synthesis in response to physiologic doses of aldosterone was not demonstrable, and that some reports to the contrary may have been influenced by artifacts from bacterial RNA metabolism. Earlier evidence for obligatory alterations in RNA metabolism during the latent period is not strong.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ab G., Malt R. A. Metabolism of ribosomal precursor ribonucleic acid in kidney. J Cell Biol. 1970 Aug;46(2):362–369. doi: 10.1083/jcb.46.2.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnstiel M., Speirs J., Purdom I., Jones K., Loening U. E. Properties and composition of the isolated ribosomal DNA satellite of Xenopus laevis. Nature. 1968 Aug 3;219(5153):454–463. doi: 10.1038/219454a0. [DOI] [PubMed] [Google Scholar]
- Bishop D. H., Claybrook J. R., Spiegelman S. Electrophoretic separation of viral nucleic acids on polyacrylamide gels. J Mol Biol. 1967 Jun 28;26(3):373–387. doi: 10.1016/0022-2836(67)90310-5. [DOI] [PubMed] [Google Scholar]
- Bucher N. L., Swaffield M. N. Rate of incorporation of [6-14C]orotic acid into uridine 5'-triphosphate and cytidine 5'-triphosphate and nuclear ribonucleic acid in regenerating rat liver. Biochim Biophys Acta. 1965 Dec 9;108(4):551–567. doi: 10.1016/0005-2787(65)90052-3. [DOI] [PubMed] [Google Scholar]
- Castles T. R., Williamson H. E. Mediation of aldosterone induced anti-natriuresis via RNA-synthesis de novo. Proc Soc Exp Biol Med. 1967 Mar;124(3):717–719. doi: 10.3181/00379727-124-31834. [DOI] [PubMed] [Google Scholar]
- Chaudhuri S., Lieberman I. Time required by the normal and regenerating rat liver cell to make a ribosome. J Mol Biol. 1968 Apr 14;33(1):323–326. doi: 10.1016/0022-2836(68)90299-4. [DOI] [PubMed] [Google Scholar]
- EDELMAN I. S., BOGOROCH R., PORTER G. A. ON THE MECHANISM OF ACTION OF ALDOSTERONE ON SODIUM TRANSPORT: THE ROLE OF PROTEIN SYNTHESIS. Proc Natl Acad Sci U S A. 1963 Dec;50:1169–1177. doi: 10.1073/pnas.50.6.1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edelman I. S., Fimognari G. M. On the biochemical mechanism of action of aldosterone. Recent Prog Horm Res. 1968;24:1–44. doi: 10.1016/b978-1-4831-9827-9.50007-1. [DOI] [PubMed] [Google Scholar]
- FEIGELSON P., FEIGELSON M. Studies on the mechanism of regulation by cortisone of the metabolism of liver purine and ribonucleic acid. J Biol Chem. 1963 Mar;238:1073–1077. [PubMed] [Google Scholar]
- Fanburg B. L., Posner B. I. Labeling of RNA in the perfused heart: the problem of bacterial contamination. Biochim Biophys Acta. 1969 Jun 17;182(2):577–579. doi: 10.1016/0005-2787(69)90214-7. [DOI] [PubMed] [Google Scholar]
- Fanestil D. D. Mechanism of action of aldosterone. Annu Rev Med. 1969;20:223–232. doi: 10.1146/annurev.me.20.020169.001255. [DOI] [PubMed] [Google Scholar]
- Fimognari G. M., Fanestil D. D., Edelman I. S. Induction of RNA and protein synthesis in the action of aldosterone in the rat. Am J Physiol. 1967 Oct;213(4):954–962. doi: 10.1152/ajplegacy.1967.213.4.954. [DOI] [PubMed] [Google Scholar]
- Forte L., Landon E. J. Aldosterone-induced RNA synthesis in the adrenalectomized rat kidney. Biochim Biophys Acta. 1968 Apr 22;157(2):303–309. doi: 10.1016/0005-2787(68)90084-1. [DOI] [PubMed] [Google Scholar]
- Gall J. G. Nuclear RNA of the salamander oocyte. Natl Cancer Inst Monogr. 1966 Dec;23:475–488. [PubMed] [Google Scholar]
- Goldman D., Schultz S. G., Epstein W. Repressive control of potassium transport in Escherichia coli. Biochim Biophys Acta. 1966 Dec 28;130(2):546–548. doi: 10.1016/0304-4165(66)90259-5. [DOI] [PubMed] [Google Scholar]
- Hausen P., Stein H. On the synthesis of RNA in lymphocytes stimulated by phytohemagglutinin. 1. Induction of uridine-kinase and the conversion of uridine to UTP. Eur J Biochem. 1968 Apr;4(3):401–406. doi: 10.1111/j.1432-1033.1968.tb00226.x. [DOI] [PubMed] [Google Scholar]
- Honig G. R., Rabinovitz M. Actinomycin D: inhibition of protein synthesis unrelated to effect on template RNA synthesis. Science. 1965 Sep 24;149(3691):1504–1506. doi: 10.1126/science.149.3691.1504. [DOI] [PubMed] [Google Scholar]
- Horisberger M., Amos H. Ribonucleic acid synthesis and transport in animal cells at 27 degrees C. Biochem J. 1970 Apr;117(2):347–353. doi: 10.1042/bj1170347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KROEGER H. CHEMICAL NATURE OF THE SYSTEM CONTROLLING GENE ACTIVITIES IN INSECT CELLS. Nature. 1963 Dec 21;200:1234–1235. doi: 10.1038/2001234a0. [DOI] [PubMed] [Google Scholar]
- Kochakian C. D., Hill J. Effect of androgen on the incorporation of orotic acid-6-14C into the ribonucleic acids and free nucleotides of mouse kidney. Biochemistry. 1966 May;5(5):1696–1701. doi: 10.1021/bi00869a035. [DOI] [PubMed] [Google Scholar]
- LUBIN M., ENNIS H. L. ON THE ROLE OF INTRACELLULAR POTASSIUM IN PROTEIN SYNTHESIS. Biochim Biophys Acta. 1964 Apr 27;80:614–631. doi: 10.1016/0926-6550(64)90306-8. [DOI] [PubMed] [Google Scholar]
- Landesman R., Gross P. R. Patterns of macromolecule synthesis during development of Xenopus laevis. I. Incorporation of radioactive precursors into dissociated embryos. Dev Biol. 1968 Dec;18(6):571–589. doi: 10.1016/0012-1606(68)90027-4. [DOI] [PubMed] [Google Scholar]
- Landesman R., Gross P. R. Patterns of macromolecule synthesis during development of Xenopus laevis. II. Identification of the 40 S precursor to ribosomal RNA. Dev Biol. 1969 Mar;19(3):244–260. doi: 10.1016/0012-1606(69)90063-3. [DOI] [PubMed] [Google Scholar]
- Lovett J. S., Leaver C. J. High-molecular-weight artifacts in RNA extracted from Blastocladiella at elevated temperatures. Biochim Biophys Acta. 1969 Dec 16;195(2):319–327. doi: 10.1016/0005-2787(69)90639-x. [DOI] [PubMed] [Google Scholar]
- Lucas Z. J. Pyrimidine nucleotide synthesis: regulatory control during transformation of lymphocytes in vitro. Science. 1967 Jun 2;156(3779):1237–1240. doi: 10.1126/science.156.3779.1237. [DOI] [PubMed] [Google Scholar]
- Malamud D., Baserga R. Pool size and specific activity of UTP in isoproterenol-stimulated salivary glands. Biochim Biophys Acta. 1969 Nov 19;195(1):258–261. doi: 10.1016/0005-2787(69)90626-1. [DOI] [PubMed] [Google Scholar]
- Malt R. A. Rapidly labeled nuclear and cytoplasmic renal RNA. J Exp Med. 1966 Oct 1;124(4):679–688. doi: 10.1084/jem.124.4.679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munro H. N. The determination of nucleic acids. Methods Biochem Anal. 1966;14:113–176. doi: 10.1002/9780470110324.ch5. [DOI] [PubMed] [Google Scholar]
- Muramatsu M., Fujisawa T. Methylation of ribosomal RNA precursor and tRNA in rat liver. Biochim Biophys Acta. 1968 May 21;157(3):476–492. doi: 10.1016/0005-2787(68)90147-0. [DOI] [PubMed] [Google Scholar]
- Nicholls D. M., Ryan M. P., Miall S. H., Cappon I. D. Factors affecting protein synthesis in rat kidney ribosome preparations. Can J Biochem. 1970 Jan;48(1):105–112. doi: 10.1139/o70-017. [DOI] [PubMed] [Google Scholar]
- PORTER G. A., BOGOROCH R., EDELMAN I. S. ON THE MECHANISM OF ACTION OF ALDOSTERONE ON SODIUM TRANSPORT: THE ROLE OF RNA SYNTHESIS. Proc Natl Acad Sci U S A. 1964 Dec;52:1326–1333. doi: 10.1073/pnas.52.6.1326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peacock A. C., Dingman C. W. Molecular weight estimation and separation of ribonucleic acid by electrophoresis in agarose-acrylamide composite gels. Biochemistry. 1968 Feb;7(2):668–674. doi: 10.1021/bi00842a023. [DOI] [PubMed] [Google Scholar]
- Penman S. RNA metabolism in the HeLa cell nucleus. J Mol Biol. 1966 May;17(1):117–130. doi: 10.1016/s0022-2836(66)80098-0. [DOI] [PubMed] [Google Scholar]
- Perry R. P., Cheng T. Y., Freed J. J., Greenberg J. R., Kelley D. E., Tartof K. D. Evolution of the transcription unit of ribosomal RNA. Proc Natl Acad Sci U S A. 1970 Mar;65(3):609–616. doi: 10.1073/pnas.65.3.609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porter G. A. In vitro inhibition of aldosterone-stimulated sodium transport by steroidal spirolactones. Mol Pharmacol. 1968 May;4(3):224–237. [PubMed] [Google Scholar]
- Prestayko A. W., Tonato M., Busch H. Low molecular weight RNA associated with 28 s nucleolar RNA. J Mol Biol. 1970 Feb 14;47(3):505–515. doi: 10.1016/0022-2836(70)90318-9. [DOI] [PubMed] [Google Scholar]
- REVEL M., HIATT H. H., REVEL J. P. ACTINOMYCIN D: AN EFFECT ON RAT LIVER HOMOGENATES UNRELATED TO ITS ACTION ON RNA SYNTHESIS. Science. 1964 Dec 4;146(3649):1311–1313. doi: 10.1126/science.146.3649.1311. [DOI] [PubMed] [Google Scholar]
- SCOTT J. F., FRACCASTORO A. P., TAFT E. B. Studies in histochemistry. I. Determination of nucleic acids in microgram amounts of tissue. J Histochem Cytochem. 1956 Jan;4(1):1–10. doi: 10.1177/4.1.1. [DOI] [PubMed] [Google Scholar]
- Sharp G. W., Leaf A. Mechanism of action of aldosterone. Physiol Rev. 1966 Oct;46(4):593–633. doi: 10.1152/physrev.1966.46.4.593. [DOI] [PubMed] [Google Scholar]
- WILLIAMSON H. E. MECHANISM OF THE ANTINATRIURETIC ACTION OF ALDOSTERONE. Biochem Pharmacol. 1963 Dec;12:1449–1450. doi: 10.1016/0006-2952(63)90220-x. [DOI] [PubMed] [Google Scholar]
- Weinberg R. A., Loening U., Willems M., Penman S. Acrylamide gel electrophoresis of HeLa cell nucleolar RNA. Proc Natl Acad Sci U S A. 1967 Sep;58(3):1088–1095. doi: 10.1073/pnas.58.3.1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinberg R. A., Penman S. Processing of 45 s nucleolar RNA. J Mol Biol. 1970 Jan 28;47(2):169–178. doi: 10.1016/0022-2836(70)90337-2. [DOI] [PubMed] [Google Scholar]
- Weinberg R. A., Penman S. Small molecular weight monodisperse nuclear RNA. J Mol Biol. 1968 Dec;38(3):289–304. doi: 10.1016/0022-2836(68)90387-2. [DOI] [PubMed] [Google Scholar]
- Wiesner R., Acs G., Reich E., Shafiq A. Degradation of ribonucleic acid in mouse fibroblasts treated with actinomycin. J Cell Biol. 1965 Oct;27(1):47–52. doi: 10.1083/jcb.27.1.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willems M., Musilova H. A., Malt R. A. Giant nucleoplasmic RNA in the switch-on of compensatory renal growth. Proc Natl Acad Sci U S A. 1969 Apr;62(4):1189–1194. doi: 10.1073/pnas.62.4.1189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmerman E. F., Holler B. W. Methylation of 45 s ribosomal RNA precursor in HeLa cells. J Mol Biol. 1967 Jan 28;23(2):149–161. doi: 10.1016/s0022-2836(67)80023-8. [DOI] [PubMed] [Google Scholar]
- Zomzely C. E., Roberts S., Gruber C. P., Brown D. M. Cerebral protein synthesis. II. Instability of cerebral messenger ribonucleic acid-ribosome complexes. J Biol Chem. 1968 Oct 25;243(20):5396–5409. [PubMed] [Google Scholar]


