Abstract
Paracrine cross-talk between tumor cells and immune cells within the tumor microenvironment underlies local mechanisms of immune evasion. Signal Transducer and Activator of Transcription 3 (STAT3), which is constitutively activated in diverse cancer types, has been shown to be a key regulator of cytokine and chemokine expression in murine tumors, resulting in suppression of both innate and adaptive anti-tumor immunity. However, the immunologic effects of STAT3 activation in human cancers have not been studied in detail. To investigate how STAT3 activity in human head and neck squamous cell carcinoma (HNSCC) might alter the tumor microenvironment to enable immune escape, we used siRNA and small molecule inhibitors to suppress STAT3 activity. STAT3 inhibition in multiple primary and established human squamous carcinoma lines resulted in enhanced expression and secretion of both proinflammatory cytokines and chemokines. While conditioned medium containing supernatants from human HNSCC inhibited LPS-induced dendritic cell activation in vitro, supernatants from STAT3 silenced tumor cells reversed this immune evasion mechanism. Moreover, supernatants from STAT3 silenced tumor cells were able to stimulate the migratory behavior of lymphocytes from human peripheral blood in vitro. These results demonstrate the importance of STAT3 activation in regulating the immunomodulatory mediators by human tumors and further validate STAT3 as a promising target for therapeutic intervention.
Introduction
Human solid malignancies, notably, head and neck squamous cell carcinoma, as well as glioblastoma multiforme, melanoma, prostate, and breast cancer display constitutive activation of STAT3 that regulates multiple genes associated with angiogenesis, apoptosis, cell cycle progression, and inflammation (1),(2),(3). Subsequently, the characterization of STAT3’s pleiotrophic role in tumorigenesis has prompted development of drugs to disrupt STAT3 signaling (4),(5),(6),(7). Interestingly, in preclinical studies, STAT3 targeting in tumor cells elicited a “bystander” anti-tumor effect that was attributed to infiltration of immune cells in the tumor microenvironment (8),(9). STAT3 can serve as a negative regulator of chronic inflammatory responses in vivo but is also critical for the generation of Th17 cell response, characterized by production of IL-17A (10),(11),(12). STAT3 null mice in the myeloid compartment induced inflammatory bowel disease and its macrophages were abnormally activated, corroborating its in vivo role in mediating an immunological “brake” against certain destructive inflammatory responses (13),(14). In this vein, IL-6 dependent suppression of DC maturation was found to be STAT3 dependent (15). On the other hand, STAT3-driven Th17 responses can induce inflammation, which in one case has recently been shown to be procarcinogenic (16).
In the context of immunological responses to established tumors in mice, STAT3 has been noted to orchestrate the immune components of the tumor microenvironment (17),(18),(22). In the B16 model, STAT3 activity inhibited the expression of multiple Th1 cytokines that can potentially induce DC maturation, resulting in immune evasion response in vivo (18),(19). In a follow-up study, Kortylewski et al. showed that genetic ablation of STAT3 in the myeloid hematopoietic compartment also elicited an anti-tumor immune response characterized by activation of NK cells, neutrophils and T cells within the tumor microenvironment (20). These studies, together with the finding that STAT3 suppression in DCs can break tumor-antigen specific T cell anergy (21), demonstrated that tumor cells can utilize STAT3 signaling as an important mechanism to suppress the anti-tumor potential of the immune cells that infiltrate their microenvironment. From these murine studies, there are now over 30 downstream STAT3 dependent factors that can potentially immunomodulate the tumor microenvironment (22). Given the complexity of the tumor microenvironment, one valid hypothesis is that these STAT3 dependent factors may act combinatorially to induce the anti-tumor phenotypes.
However, despite these findings in murine models, there have been no studies to determine whether STAT3 plays a parallel role in human cancer. As an initial approach to address this question, we explored the immunologic consequences of STAT3 blockade in HNSCC, a human cancer with consistently high levels of constitutive STAT3 activation. Specifically, we investigated the role of tumor cell STAT3 signaling in the regulation of proinflammatory cytokine expression, human dendritic cell activation and human leukocytic migration.
Materials & Methods
Cell lines
Human Cal27, HN11, and Fadu cells lines were purchased from American Type Culture Collection. HN6, HN22, HN28, and HN29 were derived from tumor specimens and kindly provided by Dr. David Sidransky (Johns Hopkins School of Medicine).
siRNA transfection and lentiviral transduction
Transient transfections of tumor cells with STAT3 small interfering RNA (siRNA) oligonucleotide (Santa Cruz) were conducted using LipofectAMINE 2000 (Invitrogen Life Technologies). For control samples, cells were transfected with scrambled small interfering RNA oligonucleotide or LipofectAMINE alone. The cells were transfected with 15nM of STAT3 or scrambled siRNA and cultured for two days.
Mission TRC-Hs (Sigma) clone sets of sequence-verified shRNA lentiviral plasmids were obtained from the JHU High Throughput Biology Center. The sequences of B7 and B8 shRNAs are reported in Supplementary Table 1. VSV-G pseudotyped virus was produced by the Johns Hopkins Neurosurgery Vector Core by co-transfecting 293T cells with an shRNA transducing vector and two packaging vectors: psPAX2 and PMD2.G.
Annexin V staining
Cells were stained with 5μl of PE Annexin V according to the manufacturer’s protocol (BD Pharmingen) and analyzed by flow cytometry within 1 hr.
Quantitative Real-Time PCR (qRT-PCR)
Total RNA was extracted and DNAse I-digested using the RNeasy kit according to the manufacturer’s protocol (Qiagen). 1μg of RNA was used as template for the reverse transcription reaction (SuperScript III; Invitrogen). qRT-PCR was performed using a ICycler MyiQ detection system (Biorad). The primer sequences are listed in the Supplementary Table 2. To analyze the relative changes in gene expression of the treatment group versus the untreated control, the 2-ΔΔCT method was used (23).
ELISA
Human IP-10, IL-6, IL-8, and VEGF-A were measured by ELISA (Quantikine HS®; R&D Systems) in the culture supernatants of siRNA treated and untreated cell lines according to the manufacturer’s instructions. The lower detection limit was 1.0 pg/ml.
Electrophoresis Mobility Shift Assay (EMSA)
Nuclear extracts were prepared and 5-10 μg of crude extracts were incubated with the 32P-labeled high-affinity SIE probe derived from the c-fos gene promoter that binds STAT1 and STAT3 as previously described (24). Protein-DNA complexes were resolved on 5% nondenaturating polyacrylammide gels and analyzed by autoradiography using Kodak film. Supershift binding reaction was performed using polyclonal rabbit antibody specific for human STAT3 (Santa Cruz).
DC maturation assay
Human dendritic cells (DC) were prepared from Buffy coat layers purchased from Baxter Healthcare Corporation. CD14+ monocytes were isolated from peripheral blood mononuclear cells (PBMC) by positive selection using a MACS system (Miltenyi Biotech), according to the manufacturer’s protocol, and were cultured for 6 days in 10% FCS RPMI-1640 supplemented with 1000U/ml GM-CSF (R&D Systems) and 500U/ml IL-4 (Peprotech). Subsequently, the immature DCs were incubated with 100ng/ml of LPS from Escherichia coli 026:B6 (Sigma) for 48 hours. Immature DCs and mature DCs were labeled with fluorescein isothiocyanate (FITC)- conjugated IgG specific for HLA-DR (BD Bioscience), phycoerythrin (PE)- conjugated IgG specific for CD86 (eBioscience), and Allophycocyanin (APC)-conjugated IgG mAb specific for CD11c (BD Bioscience) for 20 min at 4°C.
DC maturation inhibition experiments were performed with CD14+ monocytes in standard dendritic cell medium supplemented with tumor cell supernatants (50%). Tumor cell supernatant was added to the culture on day 0, 2, 4, and 6, at which point LPS was added. On day 8, cultures were stained and analyzed by flow cytometry.
Migration assay
Functional ability to induce lymphocytic chemotaxis was assessed with the ChemoTx system (3μm pore, 5.7mm site, 300μL 96-well, Neuro Probe) according to the manufacturer’s protocol. Tumor cell supernatant, serum-free media (negative control), or 100% FCS (positive control) were placed in the lower wells. In 11 wells, the supernatant was replaced by a serial dilution of PBMC to serve as a standard curve for the CyQuant cell proliferation assay. PBMCs from normal donors were placed on top of each filter site. Cell numbers were quantified with the CyQuant NF Cell Proliferation Assay (Invitrogen). The fluorescence was measured with the SpectraMax Gemini XS Fluorometer (Molecular Devices) with a 485/538 nm filter set. The fluorescence readout was correlated to cell numbers by the PBMC standard curve.
Small molecule inhibitor
Stattic (5) was purchased from Calbiochem and diluted in DMSO following the manufacturer’s protocol (final DMSO concentration of 1%). Cell lines were treated with Stattic at 1, 10, and 20μM concentration or DMSO. Cells were harvested after 24, 48 and 72 hours. Annexin V staining was performed to test for apoptosis.
Statistical analysis
We used paired t-test to calculate two-tailed p value to estimate statistical significance of differences between two treatment groups. Statistically significant p values were labeled as follow: **p < 0.01 and *p < 0.05. Data were analyzed using Excel software.
Results
Suppression of STAT3 alters the proinflammatory cytokine and chemokine profile of human HNSCC tumor cell lines
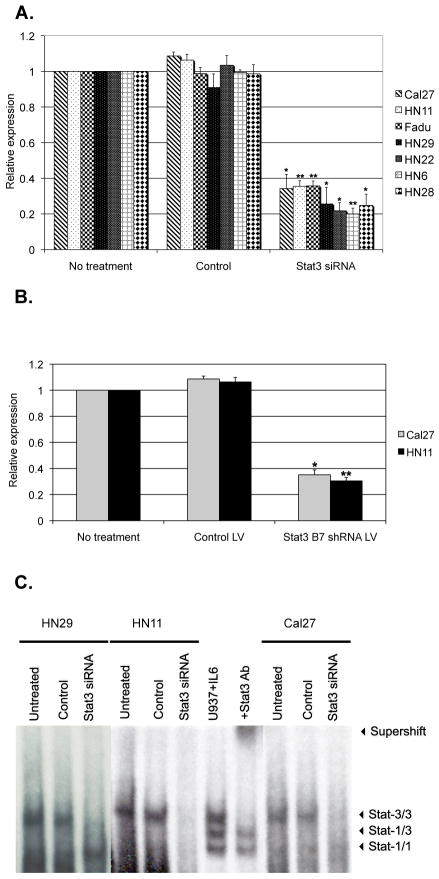
In order to study the immunologic consequences of STAT3 activation of human tumor cells, we focused on commercially available human squamous cell carcinoma (HNSCC) cell lines Cal27, HN11, and Fadu, as well as four other human HNSCC primary culture cell lines (HN6, HN22, HN28, and HN29) which are known to express high levels of constitutively active phospho-STAT3 (p-STAT3)(25). Previously, the immunomodulatory role of STAT3 was evaluated by transient transfection of STAT3 antisense oligonucleotides that had modest transfection efficiency (19). Therefore, we chose to optimize the blockade of STAT3 signaling by using siRNA knockdown to specifically suppress its expression. We optimized the transfection conditions to assure greater than 60% STAT3 mRNA suppression in all the cell lines by using a Lipofectamine system. We also transduced two of the HNSCC cell lines, HN11 and Cal27, with STAT3 siRNA lentivirus to obtain stably transduced cell lines. We observed minimal change in apoptosis (<10%) as confirmed by Annexin V staining that was performed at the end of each siRNA transfection and infection (data not shown). As shown in Figure 1A, quantitative qRT-PCR on STAT3 siRNA transfected cell lines detected a statistically significant suppression of STAT3 mRNA compared to untreated cells or cells transfected with scrambled siRNA used as controls. Comparable results were noted for HN11 and Cal27 cell lines transduced with STAT3 siRNA lentivirus (Figure 1B). In order to demonstrate that STAT3 siRNA suppression resulted in decreased p-STAT3 activation in the nuclei, Electrophoresis Mobility Shift Assay (EMSA) was performed. As shown in Figure 1C, there was a significant reduction of DNA binding p-STAT3 in the siRNA treated cell lines in comparison to the controls.
Figure 1.
STAT3 suppression in HNSCC cell lines. qRT-PCR demonstrated down-regulation of STAT3 mRNA by (a) siRNA treatment, as compared to no treatment or controls (scramble siRNA), or by (b) B7 shRNA lentivirus (LV) treatment, as compared to no treatment or control lentivirus. Histograms represent the means of at least three experiments normalized to mRNA expression of GAPDH. Data are presented as fold difference relative to control-untreated cell lines. * p <0.05; ** p <0.01. (c) STAT3 activation in tumor cells, as detected by EMSA. Stat-3/3, Stat3 homodimer; Stat-3/1, Stat3-Stat1 heterodimer, Stat-1/1, Stat1 homodimer. The STAT3 homodimer band in HN29, HN11and Cal27 cell lines decreased after treatment with STAT3 siRNA, compared to control. U937 stimulated with IL-6 represents the positive control (lane 7). Pre-incubation with an anti-STAT3 antibody shifts the STAT3 homodimer band (supershift band; lane 8).
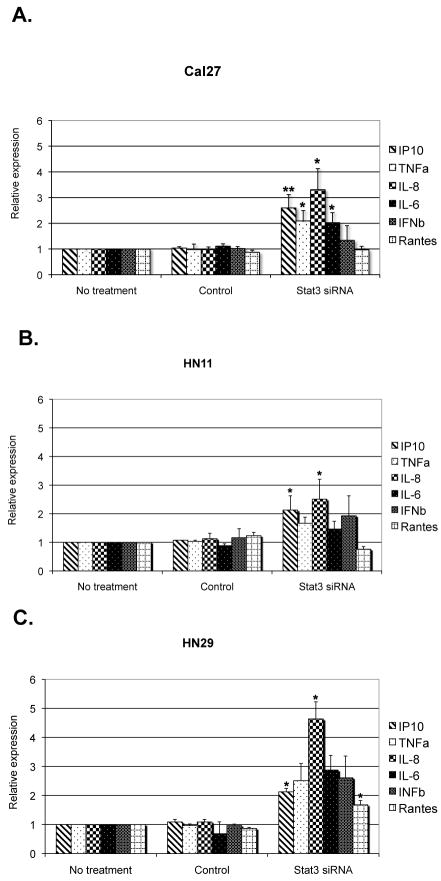
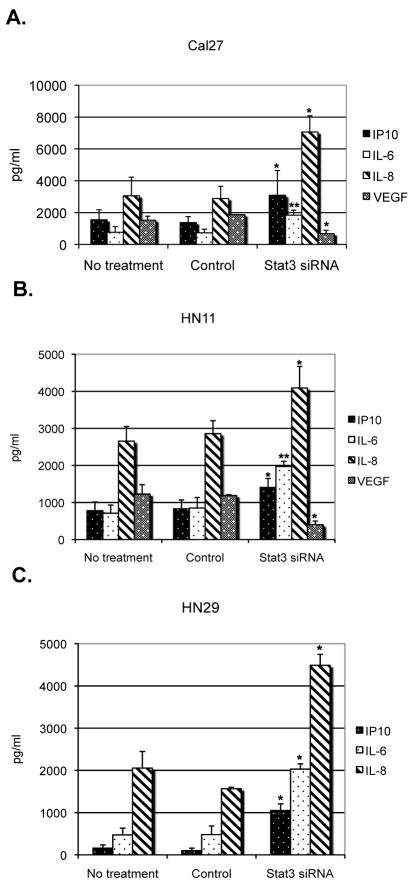
In order to evaluate the immunologic consequences of STAT3 blockade in these human HNSCC lines we initially tested the mRNA expression of several cytokines and chemokines by qRT-PCR. We noticed a pattern of expression similar to the murine models. In particular, we found that IFNγ-inducible protein 10 (IP10), IL-8, TNFα and IL-6 mRNAs were elevated in all seven HNSCC cell lines transfected with STAT3 siRNA (Figure 2 and Supplementary Figure 1). Interestingly, in contrast to observations in murine tumor systems, STAT3 suppression did not consistently result in upregulation of RANTES or IFNβ mRNAs in all of the human HNSCC tumor cell lines tested. We next investigated whether these STAT3 mediated changes in cytokine/chemokine transcription correlated with protein expression. Using ELISA assays, we demonstrated that STAT3 suppression in tumor cell lines resulted in significantly higher secretion of IP-10, IL-8, and IL-6, in the culture supernatant of each of the STAT3 siRNA treated cell lines, compared to controls (Figure 3). Since STAT3 is known to directly control VEGF transcription in mice (39), we also tested for VEGF secretion and we noticed a statistically significant reduction in the samples treated with STAT3 siRNA (Figure 3). qRT-PCR using VEGF primers in the STAT3 suppressed tumor cells also showed reduced expression of VEGF mRNA (Supplementary Figure 2), correlating with the reduction of VEGF protein. Therefore, we hypothesized that constitutive expression of STAT3 in human tumor cells that is responsible for inhibiting the production of inflammatory mediators in the tumor microenvironment may induce the tumor infiltrated immune cells to differentiate along an immunosuppressive phenotype. Moreover, given the differential expression patterns of paracrine factors among the HNSCC cell lines tested, we initially focused on the combinatorial effects of STAT3-dependent cytokines and chemokines in the tumor microenvironment.
Figure 2.
STAT-3 suppression in tumor cells increases expression of proinflammatory mediators. qRT-PCR analysis showed up-regulation of proinflammatory cytokine and chemokines mRNA by siRNA treatment only, as compared to no treatment and to controls in (a) Cal27, (b) HN11, and (c) HN29 cell lines. Histograms represent the means of at least three experiments normalized to GAPDH. Data are presented as fold difference relative to control-untreated cell lines. * p <0.05; ** p <0.01.
Figure 3.
Proinflammatory cytokines and chemokines can be detected in STAT3 siRNA treated HNSCC cell lines. ELISAs were performed from treated or control (a) Cal27, (b) HN11, and (c) HN29 cell line culture supernatants. Results are presented as the mean ± s.e.m., n=3, for each ELISA. * p <0.05; ** p <0.01.
STAT3 suppression in the tumor cell can affect dendritic cell maturation
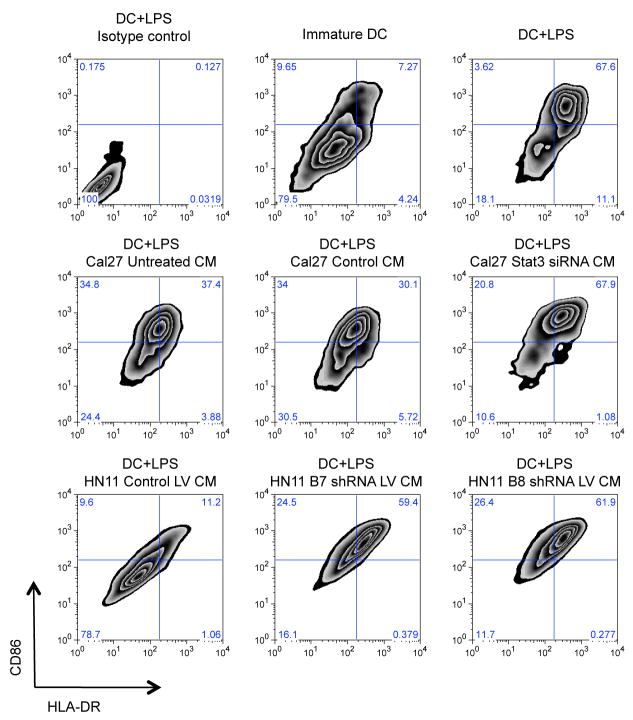
One mechanism by which cancer cells can potentially modulate the immune response is to regulate the expression of dendritic cell (DC) maturation inhibitory factors, such as VEGF and IL10, into the tumor microenvironment. In the B16 murine melanoma model, STAT3 overexpression prevented efficient maturation of murine bone marrow-derived dendritic cells (19). In order to test whether an analogous phenomenon is operative in the crosstalk between human tumor cells and human DCs, we exposed human immature monocyte-derived DCs to conditioned medium (CM) containing supernatants from STAT3 siRNA treated or untreated HNSCC cell lines. In the presence of LPS, human DCs are activated, as defined by an augmented co-expression of MHC class II and CD86 (Figure 4, upper row, right panel). The majority of DCs cultured in the presence of conditioned media containing supernatant of HNSCC cell lines were shown to remain immature after LPS stimulation (Figure 4, middle and lower rows, left panels). However, LPS induced maturation of DCs in the presence of CM containing STAT3 siRNA treated cell line supernatants revealed that the percentage of MHC class IIhigh and CD86high DCs was restored to that level observed by standard LPS stimulation (Figure 4, middle rows, right panels). Comparable results were noted using HN11 cell lines transduced with lentivirus containing B7 and B8 STAT3 shRNA (Figure 4, lower row panels). This indicated that blocking STAT3 signaling in human tumor cell lines may result in an enhanced LPS induced activation of DC in vitro. DC maturation assays were also performed without LPS as control experiments. The percentage of MHC class IIhigh and CD86high were slightly elevated without LPS induction when incubated with conditioned medium from STAT3 suppressed cell lines as shown in Supplementary Figure 3. However, the differential effect on DC maturation without LPS was minimal in comparison to the significant effect on the DC maturation assay in the presence of LPS. Given that the DC maturation is sensitive to variable such as temperature, time, and even mechanical manipulation, the quantitative effect of STAT3 signaling in the HNSCC cells to suppress LPS induced DC maturation underscores how STAT3 can reverse a potent immunostimulant to render the DC immature to potentially produce an immunosuppressive phenotype in the tumor microenvironment.
Figure 4.
Inhibition of DC maturation by tumor derived factors is abrogated upon STAT3 knockdown. Flow cytometry plots demonstrate surface expression of HLA-DR vs. CD86 of CD11c+ gated DCs. Upper row: immature monocyte-derived DCs (middle panel) and LPS-matured DCs (right panel). Left panel showing isotype controls on LPS-matured DCs. Middle row: LPS-matured, DCs cultured in presence of condition medium from untreated (left panel), control (middle panel), and STAT3 siRNA treated Cal27 supernatant (right panel). Lower row: LPS-matured, DCs cultured in presence of condition medium from control lentivirus (LV) vector transduced HN11 supernatant (left panel), STAT3 B7 and B8 shRNA LV vector transduced HN11 supernatant (middle and left panels, respectively).
VEGF, whose expression is controlled by STAT3, has been demonstrated as a potential mediator that can suppress DC maturation (1). By ELISA (Figure 3) and qPCR (Supplementary Figure 2) we found that the level of VEGF mRNA and secreted protein is statistically diminished in the supernatant from STAT3 siRNA treated HNSCC cell lines. Preliminary experiments were performed to directly test whether VEGF by itself can suppress LPS-induced DC maturation. Several concentrations of human recombinant VEGF were titrated back into cultured medium obtained from HNSCC cell lines with suppressed STAT3 siRNA, but even concentrations as high as 20ng/ml did not inhibit DC maturation to the level noted with cultured medium from untreated HNSCC cell lines (Supplementary Figure 4).
STAT3 suppression enhances trafficking of leukocytes in vitro
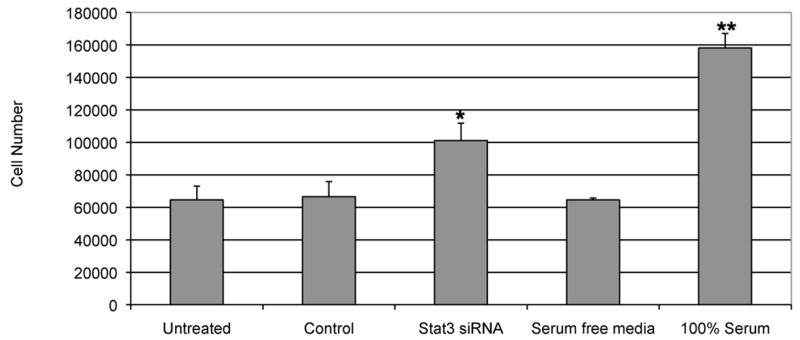
Given that suppression of STAT3 resulted in upregulation of potent chemoattractant chemokines such as IP10 (26) or IL-8 (27), we also hypothesized that suppression of STAT3 signaling in the tumor cells may improve immune cell trafficking into the tumor microenvironment, comparable to the B16 model (19). In order to test this in a human system, we used a standard in vitro chemotaxis assay. We incubated human PBMC in presence of CM containing supernatant from STAT3 siRNA treated Cal27 tumor cell line. CM containing supernatant from cells transfected with scrambled siRNAs was used as controls. We used a 3 mm pore size filter in the chemotaxis plate to allow predominantly lymphocytic migration. We used a fluorescence-based assay to quantify the number of migrated cells. A standard curve with known numbers of PBMC demonstrated the linear range of the assay. We observed a distinct, statistically significant migration of leukocytes only towards the supernatant from Cal27 transfected with STAT3 siRNA (Figure 5). Further work is in progress to establish if a differential combination of STAT3 dependent chemotactic mediators can stimulate lymphocyte migration.
Figure 5.
Supernatant from STAT3 siRNA suppressed tumor cells enhances leukocytes migration. Chemotaxis assay demonstrated enhanced PBMC migration in the presence of CM containing siRNA treated and control Cal27 culture supernatants. PBMCs were incubated in presence of serum free medium and 100% serum as negative and positive controls, respectively. Results are presented as the mean ± s.e.m., n=3, for each ELISA. * p <0.05; ** p <0.01.
Effects of pharmacological inhibition of STAT3
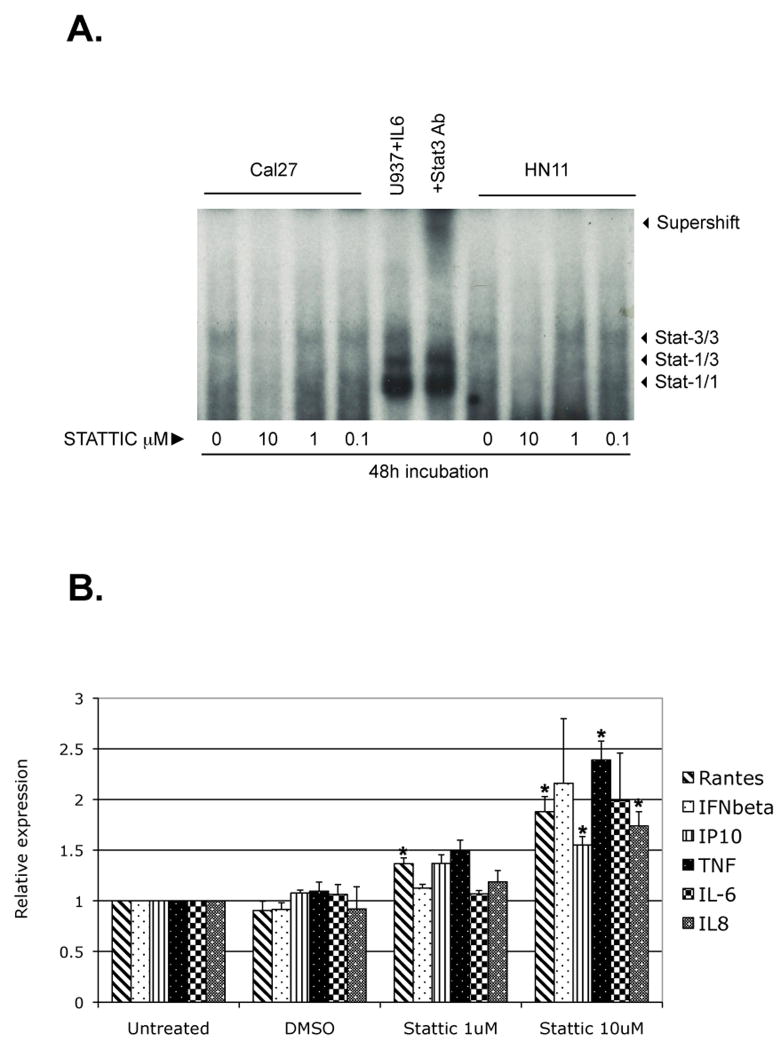
Despite advances in siRNA technology, the feasibility of siRNA in cancer therapy is still unknown. Fortunately, since STAT3 has been extensively studied as an oncogenic factor, there are several STAT3 specific small molecule inhibitors with translational potentials. One such STAT3 inhibitor that is commercially available is Stattic, a cell-permeable vinyl-sulfone compound that acts as an inhibitor of STAT3 cellular function by targeting the STAT3-SH2 domain and preventing its association with upstream kinases (5). First, we titrated the concentration of Stattic needed to specifically reduce the activation, dimerization, and nuclear translocation of STAT3 in human HN11 and Cal27 cell lines. At concentrations between 1-10 μM, which is below the reported concentrations at which apoptosis is induced, Stattic resulted in substantial reduction in p-STAT3 and the DNA binding of STAT3 homodimers, while binding of STAT1 homodimers to the same DNA probe was not significantly inhibited at those concentrations in all human cell lines tested according to the EMSA assay (Figure 6A). We next evaluated the battery of pro-inflammatory chemokine and cytokine after 48 hour incubation in the tumor cell lines. We were able to observe a dose-dependent increase in mRNA for RANTES, TNFα, IP-10, IL-8, IFNβ and IL-6 in both HNSSC cell lines (Figure 6B). It is informative that the small molecule STAT3 inhibitor did result in increased mRNA for RANTES and IFNβ, suggesting that the lack of effect on these genes with STAT3 siRNA transfection may have been due to incomplete STAT3 knockdown rather than a fundamental difference between mouse and human tumor lines. These results suggest that targeting STAT3 with a small molecule inhibitor at doses that may not induce apoptosis may still induce a therapeutic anti-tumor effect by activating loco-regional dendritic cells and by attracting leukocytes into the tumor microenvironment.
Figure 6.
Stattic treatment decreases STAT3 activation and induces proinflammatory mediator expression. (a) STAT3 activation in tumor cells, as detected by EMSA. Stat-3/3, Stat3 homodimer; Stat-3/1, Stat3/Stat1 heterodimer, Stat-1/1, Stat1 homodimer. The STAT3 homodimer band in both Cal27 and HN11 cell lines was decreased after treatment with 10μM Stattic for 48h, compared to untreated and to 1-0.1 μM Stattic treatments. U937 stimulated with IL-6 is the positive control (lane 5). Pre-incubation with an anti-STAT3 antibody shifted the STAT3 homodimer band (supershift band; lane 6). (b) qRT-PCR analysis showed up-regulation of proinflammatory cytokine and chemokines mRNA by 10mM Stattic for 48h, compared to untreated, DMSO only (drug diluent), and to 1 μM Stattic treatment. Histograms represent the means of at least three experiments normalized to GAPDH mRNA expression (internal control). Data are presented as fold difference relative to untreated cell lines. * p <0.05.
Discussion
Physiologically, STAT3 transcription factor is a critical negative regulator of tissue wound healing that induces pleiotrophic brake on inflammatory cytokines, chemokines, and angiogenic factors to prevent tissue injury during the chronic phase of wound healing (28), (18), (29). It is not surprising that neoplastic epithelial cells have been selected for constitutively active STAT3 as a potent immune evasion mechanism. Previous work from our lab in the murine system showed that tumor cells could co-opt STAT3’s physiological function to prevent an anti-tumor host immune response (19),(22).
In this report, we demonstrated that STAT3 inhibition in human HNSCC cells could also induce upregulation and secretion of multiple cytokines and chemokines for potential antitumor response in the microenvironment. Both siRNA silencing and pharmacologic small molecule inhibition of STAT3 were used to substantially reduce STAT3 signaling in the human tumor cells. As demonstrated by this report, HNSCC differentially expressed multiple paracrine factors that can potentially inhibit LPS-induced DC maturation. STAT3 suppression in HNSCC cell lines dramatically reversed this immunosuppressive phenotype in vitro. We also showed that these STAT3 dependent tumor derived factors could stimulate migration of lymphocytes in vitro. The control tumor cells were treated with scrambled siRNA to ensure that variables that can be attributed to transfection or transduction of small RNA sequences did not confound the paracrine effects. Moreover, the two independent methods of STAT3 inhibition inducing comparable effects corroborate our hypothesis that these paracrine cross talk are STAT3-dependent.
We reasoned that focusing on a single cytokine or chemokine may not reveal the combinatorial effects of multiple factors that can direct cellular behaviors within the tumor microenvironment. To directly examine the various combination of STAT3 induced cytokines and chemokines, we tested the unbiased conditioned media from the STAT3 siRNA treated tumor cell line culture supernatants to examine the paracrine crosstalk between the human tumor cells and the isolated human immune cells. Although we tested for IP-10, IL-6, IL-8, and VEGF protein levels in the culture supernatants, this report does not address the possibility of the differential expression pattern of other proinflammatory factors that may shape the tumor microenvironment. Our preliminary experiments with VEGF to examine if a single STAT3 dependent factor can mediate some of the anti-tumor phenotypes of the tumor microenvironment suggest that STAT3 signaling induces transcriptional variations of multiple paracrine factors that work combinatorially to produce an anti-tumor response. We are currently investigating the presence of other important cytokines, such as IL12 and IL23, as well as combinatorial effects of multiple paracrine factors mediating DC activation and lymphocytic chemotaxis in vitro. It would be also very important to study the relationship between corresponding receptors of these proinflammatory mediators and STAT3 signaling suppression in the same tumor cells as well as in the immune cells.
Demonstrating IP10 upregulation consistently and significantly in all the cell lines treated with STAT3 siRNA was an interesting phenomena because it can potentially mediate antitumor activity through two independent mechanisms: by stimulation of leukocytic migration into tumor microenvironment and by inhibition of tumor angiogenesis. It is also well documented in mouse tumor models the synergistic antitumor effects of IP10 in conjunction with IL12 or with TNFα (30),(31). This “attraction-expansion” hypothesis is based on the idea that colocalization of proinflammatory and chemotactic mediators could enhance synergistically the anti-tumoral immune response (32). Our results showed concomitant upregulation of various STAT3 dependent immunomodulatory factors that can act as the “recruiters” as well as “activators”.
The finding that blockade of STAT3 in HNSCC cancer cell lines could reverse the inhibitory effects of tumor cell supernatant on DC maturation suggested that VEGF may be a critical STAT3-dependent immunomodulator than can interfere with DC differentiation and function in cancer patients. Gabrilovich et al. documented that there was a direct association between the elevated numbers of immature DCs in cancer patients and high levels of circulating VEGF (33). In our in vitro studies, STAT3 dependent VEGF alone did not appear to directly mediate DC maturation. Interestingly, although we focused our initial studies on DC maturation, STAT3 dependent VEGF may induce other hematopoietic cells such as myeloid derived suppressor cells to shape the tumor microenvironment towards an immunosuppressive phenotype.
The role of IL-6 in tumorigenesis has been well documented, and a recent cohort study showed that serum IL-6 may serve as a biomarker for prognosis among head and neck cancer patients (34). In our study, as well as in the murine models, we consistently noted increase level of IL-6 with STAT3 suppression. IL-6 is currently hypothesized as one cytokine that can mediate the STAT3 feed forward loop, which may be dependent on NF-kB signaling (22), (40). However, given the complex relationship between inflammation and tumorigenesis within the tumor microenvironment, the consequences of STAT3 dependent IL-6 variations may be difficult to predict. Since we did not observe any increased growth rate of the tumor cells tested in vitro, this elevation of IL-6 is not correlated with STAT3-independent tumorigenesis. Despite IL-6’s possible use as a biomarker, however, it may not account for the full combinatorial effects of multiple cytokines and chemokines to direct the development of the tumor. Rather than focusing on a single cytokine, therefore, the focus of our study was to evaluate the role of STAT3 signaling in human tumor cells.
In terms of STAT3 signaling, our lab and others have shown that STAT3 signaling in the hematopoietic compartment also plays a significant role in the development of the murine tumor microenvironment (20). Although our study was directed at the STAT3 activity from human carcinoma cells, murine studies have demonstrated that STAT3 signaling in the tumor microenvironment can also induce the expression of IL-23 on tumor infiltrating macrophages, while inhibiting NF-kB dependent IL-12/p39 gene expression (35). STAT3 also appears to prolong NF-kB nuclear retention in both tumor cells as well as hematopoietic cells (36). Moreover, STAT3 signaling regulates the level of Th17 cells in vivo, and this proinflammatory environment has been shown to be pro-carcinogenic in the colon carcinoma models (12),(16). Cumulatively, STAT3 signaling affects multiple downstream pathways to promote a pro-carcinogenic “soil” that suppresses an anti-tumor response.
From a clinical standpoint, therefore, targeting STAT3 signaling in the tumor microenvironment appears to be attractive. The data presented in this report show that the paracrine effects of STAT3 activation in human carcinoma can regulate the human tumor microenvironment analogously to the murine system. Given that STAT3 signaling in tumor cells promotes proliferation and prevents apoptosis, our report provides more evidence that STAT3 targeting may be an avenue of translational research as immunomodulator for clinical trials. As a transcription factor, STAT3 may be difficult to target, but we were able to demonstrate that Stattic, a small molecule inhibitor available commercially, can inhibit STAT3 signaling in the human tumor cells. Interestingly, its initial report used several log-fold higher concentrations for induction of apoptosis (5). Using doses from 1-10μM range, which did not induce apoptosis of the tumor cells in our studies, we showed that Stattic can induce an upregulation of RANTES, TNFα, IP-10, IL-8, IFNβ and IL-6 to a comparable level as seen with the STAT3 siRNA suppression. These results illustrate the feasibility of pharmacologic STAT3 blockade to induce an immunomodulatory effect.
Currently several clinical trials are underway that can block STAT3 signaling in various cancer patients (37),(38). Some are upstream regulators of STAT3 such as EGFR inhibitors and neutralizing antibodies, as well as Src inhibitors. Given the findings in our report, immunomodulatory effects of STAT3 signaling should be investigated as potential mechanisms of clinical efficacy among patients who are treated with biological agents that work upstream to STAT3 signaling.
Supplementary Material
Acknowledgments
Grant Support: NIH K23-DE018464-02 (YJK) and Flight Attendant Medical Research Institute Grant (YJK).
We thank Dr. David Sidransky (Johns Hopkins Oncology) for providing primary HNSCC cell lines, and Dr. Tullia Bruno (Johns Hopkins Oncology) for help in setting the DC maturation assay.
References
- 1.Yu H, Jove R. The STATs of cancer--new molecular targets come of age. Nat Rev Cancer. 2004;4(2):97–105. doi: 10.1038/nrc1275. [DOI] [PubMed] [Google Scholar]
- 2.Darnell JE. Validating Stat3 in cancer therapy. Nat Med. 2005;11(6):595–6. doi: 10.1038/nm0605-595. [DOI] [PubMed] [Google Scholar]
- 3.Nagpal JK, Mishra R, Das BR. Activation of stat-3 as one of the early events in tobacco chewing-mediated oral carcinogenesis. Cancer. 2002;94(9):2393–400. doi: 10.1002/cncr.10499. [DOI] [PubMed] [Google Scholar]
- 4.Sen M, Tosca PJ, Zwayer C, et al. Lack of toxicity of a STAT3 decoy oligonucleotide. Cancer Chemother Pharmacol. 2008 doi: 10.1007/s00280-008-0823-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schust J, Sperl B, Hollis A, Mayer TU, Berg T. Stattic: A small-molecule inhibitor of STAT3 activation and dimerization. Chem Biol. 2006;13(11):1235–42. doi: 10.1016/j.chembiol.2006.09.018. [DOI] [PubMed] [Google Scholar]
- 6.Kong LY, Wei J, Sharma AK, et al. A novel phosphorylated STAT3 inhibitor enhances T cell cytotoxicity against melanoma through inhibition of regulatory T cells. Cancer Immunol Immunother. 2008 doi: 10.1007/s00262-008-0618-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Homsi J, Cubitt C, Daud A. The src signaling pathway: A potential target in melanoma and other malignancies. Expert Opin Ther Targets. 2007;11(1):91–100. doi: 10.1517/14728222.11.1.91. [DOI] [PubMed] [Google Scholar]
- 8.Gabrilovich DI, Chen HL, Girgis KR, et al. Production of vascular endothelial growth factor by human tumors inhibits the functional maturation of dendritic cells. Nat Med. 1996;2(10):1096–103. doi: 10.1038/nm1096-1096. [DOI] [PubMed] [Google Scholar]
- 9.Heinrich PC, Behrmann I, Muller-Newen G, Schaper F, Graeve L. Interleukin-6-type cytokine signaling through the gp130/Jak/STAT pathway. Biochem J. 1998;334(Pt 2):297–314. doi: 10.1042/bj3340297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Darnell JE., Jr STATs and gene regulation. Science. 1997;277(5332):1630–5. doi: 10.1126/science.277.5332.1630. [DOI] [PubMed] [Google Scholar]
- 11.Hackenmiller R, Kim J, Feldman RA, Simon MC. Abnormal stat activation, hematopoietic homeostasis, and innate immunity in c-fes-/- mice. Immunity. 2000;13(3):397–407. doi: 10.1016/s1074-7613(00)00039-x. [DOI] [PubMed] [Google Scholar]
- 12.Harris TJ, Grosso JF, Yen HR, et al. Cutting edge: An in vivo requirement for STAT3 signaling in TH17 development and TH17-dependent autoimmunity. J Immunol. 2007;179(7):4313–7. doi: 10.4049/jimmunol.179.7.4313. [DOI] [PubMed] [Google Scholar]
- 13.Takeda K, Clausen BE, Kaisho T, et al. Enhanced Th1 activity and development of chronic enterocolitis in mice devoid of Stat3 in macrophages and neutrophils. Immunity. 1999;10(1):39–49. doi: 10.1016/s1074-7613(00)80005-9. [DOI] [PubMed] [Google Scholar]
- 14.Welte T, Zhang SS, Wang T, et al. STAT3 deletion during hematopoiesis causes Crohn’s disease-like pathogenesis and lethality: A critical role of STAT3 in innate immunity. Proc Natl Acad Sci U S A. 2003;100(4):1879–84. doi: 10.1073/pnas.0237137100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Park SJ, Nakagawa T, Kitamura H, et al. IL-6 regulates in vivo dendritic cell differentiation through STAT3 activation. J Immunol. 2004;173(6):3844–54. doi: 10.4049/jimmunol.173.6.3844. [DOI] [PubMed] [Google Scholar]
- 16.Wu S, Rhee KJ, Albesiano E, et al. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nat Med. 2009;15(9):1016–22. doi: 10.1038/nm.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yu H, Jove R. The STATs of cancer--new molecular targets come of age. Nat Rev Cancer. 2004;4(2):97–105. doi: 10.1038/nrc1275. [DOI] [PubMed] [Google Scholar]
- 18.Yu H, Kortylewski M, Pardoll D. Crosstalk between cancer and immune cells: Role of STAT3 in the tumour microenvironment. Nat Rev Immunol. 2007;7(1):41–51. doi: 10.1038/nri1995. [DOI] [PubMed] [Google Scholar]
- 19.Wang T, Niu G, Kortylewski M, et al. Regulation of the innate and adaptive immune responses by stat-3 signaling in tumor cells. Nat Med. 2004;10(1):48–54. doi: 10.1038/nm976. [DOI] [PubMed] [Google Scholar]
- 20.Kortylewski M, Kujawski M, Wang T, et al. Inhibiting Stat3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nat Med. 2005;11(12):1314–21. doi: 10.1038/nm1325. [DOI] [PubMed] [Google Scholar]
- 21.Cheng F, Wang HW, Cuenca A, et al. A critical role for Stat3 signaling in immune tolerance. Immunity. 2003;19(3):425–36. doi: 10.1016/s1074-7613(03)00232-2. [DOI] [PubMed] [Google Scholar]
- 22.Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nature Reviews – Cancer. 2009;9:798–809. doi: 10.1038/nrc2734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods. 2001;25(4):402–8. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 24.Yu CL, Meyer DJ, Campbell GS, et al. Enhanced DNA-binding activity of a Stat3-related protein in cells transformed by the src oncoprotein. Science. 1995;269(5220):81–3. doi: 10.1126/science.7541555. [DOI] [PubMed] [Google Scholar]
- 25.Xi S, Gooding WE, Grandis JR. In vivo antitumor efficacy of STAT3 blockade using a transcription factor decoy approach: Implications for cancer therapy. Oncogene. 2005;24(6):970–9. doi: 10.1038/sj.onc.1208316. [DOI] [PubMed] [Google Scholar]
- 26.Taub DD, Lloyd AR, Conlon K, et al. Recombinant human interferon-inducible protein 10 is a chemoattractant for human monocytes and T lymphocytes and promotes T cell adhesion to endothelial cells. J Exp Med. 1993;177(6):1809–14. doi: 10.1084/jem.177.6.1809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Van Damme J, Decock B, Lenaerts JP, et al. Identification by sequence analysis of chemotactic factors for monocytes produced by normal and transformed cells stimulated with virus, double-stranded RNA or cytokine. Eur J Immunol. 1989;19(12):2367–73. doi: 10.1002/eji.1830191228. [DOI] [PubMed] [Google Scholar]
- 28.Dvorak HF. Tumors: Wounds that do not heal. similarities between tumor stroma generation and wound healing. N Engl J Med. 1986;315(26):1650–9. doi: 10.1056/NEJM198612253152606. [DOI] [PubMed] [Google Scholar]
- 29.Sano S, Itami S, Takeda K, et al. Keratinocyte-specific ablation of Stat3 exhibits impaired skin remodeling, but does not affect skin morphogenesis. EMBO J. 1999;18(17):4657–68. doi: 10.1093/emboj/18.17.4657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Narvaiza I, Mazzolini G, Barajas M, et al. Intratumoral coinjection of two adenoviruses, one encoding the chemokine IFN-gamma-inducible protein-10 and another encoding IL-12, results in marked antitumoral synergy. J Immunol. 2000;164(6):3112–22. doi: 10.4049/jimmunol.164.6.3112. [DOI] [PubMed] [Google Scholar]
- 31.Enderlin M, Kleinmann EV, Struyf S, et al. TNF-alpha and the IFN-gamma-inducible protein 10 (IP-10/CXCL-10) delivered by parvoviral vectors act in synergy to induce antitumor effects in mouse glioblastoma. Cancer Gene Ther. 2009;16(2):149–60. doi: 10.1038/cgt.2008.62. [DOI] [PubMed] [Google Scholar]
- 32.Paillard F. Cytokine and chemokine: A stimulating couple. Hum Gene Ther. 1999;10(5):695–6. doi: 10.1089/10430349950018454. [DOI] [PubMed] [Google Scholar]
- 33.Gabrilovich D, Ishida T, Oyama T, et al. Vascular endothelial growth factor inhibits the development of dendritic cells and dramatically affects the differentiation of multiple hematopoietic lineages in vivo. Blood. 1998;92(11):4150–66. [PubMed] [Google Scholar]
- 34.Duffy SA, Taylor JM, Terrell JE, et al. Interleukin-6 predicts recurrence and survival among head and neck cancer patients. Cancer. 2008;113(4):750–7. doi: 10.1002/cncr.23615. [DOI] [PubMed] [Google Scholar]
- 35.Kortylewski M, Xin H, Kujawski M, et al. Regulation of the IL-23 and IL-12 balance by Stat3 signaling in the tumor microenvironment. Cancer Cell. 2009;15(2):114–23. doi: 10.1016/j.ccr.2008.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lee H, Herrmann A, Deng JH, et al. Persistently activated Stat3 maintains constitutive NF-kappaB activity in tumors. Cancer Cell. 2009;15(4):283–93. doi: 10.1016/j.ccr.2009.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pan Q, Gorin MA, Teknos TN. Pharmacotherapy of head and neck squamous cell carcinoma. Expert Opin Pharmacother. 2009;10(14):2291–302. doi: 10.1517/14656560903136754. [DOI] [PubMed] [Google Scholar]
- 38.Gupta S, El-Rayes BF. Small molecule tyrosine kinase inhibitors in pancreatic cancer. Biologics. 2008;2(4):707–15. doi: 10.2147/btt.s3003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xu Q, Briggs J, Park S, et al. Targeting Stat3 blocks both HIF-1 and VEGF expression induced by multiple oncogenic growth signaling pathways. Oncogene. 2005;24:5552–5560. doi: 10.1038/sj.onc.1208719. [DOI] [PubMed] [Google Scholar]
- 40.Squarize C, Castilho R, Sriuranpong V, et al. Molecular cross-Talk between NF-kB and STAT3 signaling pathways in head and neck squamous carcinoma. Neoplasia. 2009;8(9):733–746. doi: 10.1593/neo.06274. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.