Abstract
The differences and efficacy of standard preservation solutions in kidney transplantation, University of Wisconsin (UW) and histidine-tryptophan-ketoglutarate (HTK), remain a topic of debate in recent clinical studies. P-selectins represent glycoproteins expressed on endothelial cells and platelets responsible for the earliest events in ischemia/reperfusion injury in kidney transplantation. This study aimed to compare the levels of P-selectin expression between cold preserved kidney tissues in UW and HTK solutions. Thirty kidneys were procured from male Lewis rats and stored in cold (4°C) solutions for periods of 4, 12, 16, 20, and 24h. Group 1 (n = 15) kidneys were stored in UW solutions, and group 2 (n = 15) kidneys were submerged in HTK solutions. At the end of each time point, the kidneys underwent preparation and levels of P-selectin expression in the tissues were measured using Immunoblot analyses and adjusted volumetric quantification of Western blot signals. For all periods of cold preservation, P-selectin expression was significantly down-regulated in kidney tissues stored in UW compared with HTK solutions (P < 0.001). In summary, UW demonstrated a significant benefit over HTK solution in down-regulating P-selectin expression in cold preserved kidney grafts.
Keywords: University of Wisconsin, histidine-tryptophan-ketoglutarate, P-selectin, kidney transplantation
INTRODUCTION
Differences and efficacy of standard preservation solutions in kidney transplantation, University of Wisconsin (UW) and histidine-tryptophan-ketoglutarate (HTK), are a matter of discourse in recent clinical studies [1–11]. One of the earliest pathophysiologic events in reperfusion injury is endothelial cell dysfunction characterized, among other signs, by a dramatic adherence of polymorphonuclear leukocytes (PMNs) to the reperfused endothelium [12]. Adhesive interactions with the vascular endothelium initiate the migration of leukocytes to sites of injury and inflammation. In this cascade of events (rolling, attachment, spreading, and transendothelial migration), the selectin family (P-, E-, and L-selectins) is largely involved in the earlier events, rolling and attachment [13].
The selectins are a family of glycoproteins that are important in the regulation of cell adhesion as well as in cell signaling. P-selectin is found preformed in Weibel-Palade bodies of endothelial cells and α-granules of platelets, and is exteriorized to the cell surfaces within minutes after reperfusion. Such expression is activated by humoral stimuli, including thrombin, histamine, hydrogen peroxide, inhibitors of nitric oxide synthase [14, 15]. P-selectin expression and adherence of PMNs to the endothelium peaks in 10 to 20 min post-reperfusion [16, 17], and with minimal contribution from L-selectin [18], strongly implicating P-selectin as a major initial mediator of leukocyte rolling. After approximately 20 min, the role of P-selectin diminishes secondary to internal degradation. L-selectin, surface glycoprotein expressed constitutively on a wide variety of leukocytes, represents the principal mediator of leukocyte rolling beyond 20 min post-reperfusion [19]. In contrast, E-selectin is expressed on endothelial cells only and requires de novo synthesis before expression. Thus, synthesis of E-selectin follows activation of endothelial cells by proinflammatory cytokines; tumor necrosis factor alpha (TNF-α), interleukin-1 (IL-1), and endotoxin, a process requiring up to 4–6 h [20]. P-selectin is also transcriptionally regulated and shows transiently up-regulated expression in cytokine-activated endothelial cells after 2–4 h [21]. Moreover, P-selectin has been demonstrated to have an important role not only in earliest cellular responses but in chronic inflammations as well [22–24].
University of Wisconsin solution (UW) is the standard and most commonly used solution in human abdominal organ transplantation, although histidine-tryptophan ketoglutarate (HTK) solution, developed in the 1970s by Bretschneider as a cardioplegia solution [25], is being increasingly used for kidney [4–7, 26, 27], pancreas [28–32], and liver transplantation [33, 34]. The composition of these two solutions is summarized in Table 1. UW contains metabolically inert substrates like lactobionate and raffinose, colloid carrier hydroxyethyl-starch, oxygen radical scavengers, glutathione, allopurinol, and adenosine as a nucleotide precursor. HTK is a low viscosity, low potassium solution containing large concentration of histidine as a strong hydrogen ion buffer, tryptophan as a membrane stabilizer, and ketoglutarate as energy substrate. HTK is also reported to be less expensive to use than UW, despite the need for higher volumes of fluid per donor recovery [35].
TABLE 1.
Compositions of UW and HTK Solutions
| Component | UW (mmol/L) | HTK (mmol/L) |
|---|---|---|
| Na | 30 | 15 |
| K | 120 | 9 |
| pH | 7.4 | 7.1 |
| Lactobionate | 100 | |
| Glutathione | 3 | |
| Raffinose | 30 | |
| Hydroxyethyl starch | 5 gm% | |
| Adenosine | 5 | |
| Histidine | 180 | |
| Tryptophan | 2 | |
| Ketoglutarate | 1 | |
| Mannitol | 30 |
Ischemic periods associated with hypothermic organ preservation prime the vasculature for its characteristic response during reperfusion by displaying P-selectin prominently on endothelial cell surfaces before reperfusion, serving as a spark that ignites and amplifies the subsequent inflammatory response. Furthermore, preservation solutions play a pivotal role in organ protection, although it seems paradoxical that potential of common preservation solutions to modify important constituents of I-R injury such as P-selectins expression have not been thoroughly evaluated. Numerous studies have shown equal effectiveness of HTK and UW solutions in clinical kidney transplantation [2, 3, 5–7]. In contrast, other studies have demonstrated superiority of UW preservation over HTK [1, 4, 8, 9]. However, interpreting data from clinical studies is often associated with difficulties owing to a nonrandomized study design, not exactly matched groups, or insufficient end points in these trials. Our study aimed to investigate the effect of common storage solutions, UW and HTK, on the expression of renal P-selectin in the late ischemic period.
MATERIALS AND METHODS
Animals
Inbred male Lewis rats weighing 250 to 350 g were obtained from Charles Rivers Laboratories, Inc. (Wilmington, MA), and maintained in laminar flow cages in a specific pathogen-free animal facility at Tulane University School of Medicine. Animals were fed with a standard diet and water ad libitum. All procedures in this experiment were preformed according to the guidelines of the Institutional Animal Care and Use Committee at Tulane University and the National Research Council's Guide for the Humane Care and Use of Laboratory Animals.
Reagents
UW (Duramed Pharmaceutical, Pomona, NY) and HTK (Essential Pharmaceutical, Newton, PA) preservation solutions were used for cold storage of organs in the experiment. UW and HTK were used in the unaltered, commercially available form.
Experimental Groups
Thirty kidneys were procured from male Lewis rats and preserved in cold (4 °C) solutions for periods of 4, 12, 16, 20, and 24 h. Group 1 (n = 15) kidneys were stored in UW solutions, and group 2 (n = 15) kidneys were submerged in HTK solutions. At the end of each time periods the kidneys were stored at –80 °C for fewer than 2 mo before analysis.
Immunoblotting
Western blotting was performed on protein lysates obtained from whole kidney tissue. After separation by SDS-polyacrylamide gel electrophoresis (4%–15% gradient PAGE; Biorad, Hercules, CA) proteins were transferred electrophoretically (110 V, 90 min) to polyvinylidene diflouride membranes (Biorad) that were then blocked with a solution containing 5% blotting grade nonfat dry milk, 20 mM Tris, pH 7.4, NaCl 500 mM, Tween 20 0.05% (TBS-Tween) for 2 h at room temperature. Membranes subsequently were incubated with antibodies against P-selectin (1:1000) (Santa Cruz Biotechnology, Santa Cruz, CA). The anti-P-selectin antibody was diluted in the solution containing 5% blotting grade nonfat dry milk, 20 mM Tris, pH 7.4, NaCl 500 mM, Tween 20 0.05% (TBS-Tween). Binding of primary antibodies was followed by incubation for 3 h at room temperature with a secondary HRP-conjugated goat anti-mouse IgG (Santa Cruz Biotechnology) in 5% blotting grade nonfat milk. Signals were visualized by enhanced chemiluminescence signals captured on X-ray films. Data were normalized to β-actin internal control. Adjusted volume of immunoblots was measured using Molecular Imager Gel Doc XR + System (Biorad). Expression of P-selectin was calculated by dividing the adjusted volume of P-selectin measured for each sample by each respective adjusted volume for β-actin.
Renal Histology
At the end of each storage time point in UW and HTK solutions, kidneys were preserved in 10% buffered formalin. Such preserved kidneys were then dehydrated and embedded in paraffin. Sections (3 μm) were stained with hematoxylin and eosin and periodic acid-Schiff (PAS) using standard procedures. Examination of the sections of each kidney for hallmarks of acute tubular necrosis (ATN) at different time points were conducted by a renal pathologist.
Statistical Analysis
Statistical analysis was carried out in SPSS 16.0 for Windows (SPSS Inc., Chicago, IL). All values are reported as means ± SEM. The data within groups were compared using nonparametric Kruskal-Wallis and Mann-Whitney U tests. P value ≤ 0.05 was considered statistically significant.
RESULTS
P-Selectin Expression Showed Increase With Prolonged Duration of Cold Preservation
There is no information on expression pattern of P-selectin over variable period of cold renal preservation in UW and HTK solutions. To address this question, we performed immunoblotting studies after preservation of whole kidneys for 4, 12, 16, 20, and 24 h durations. P-selectin showed increase in expression with incrementally longer durations of cold preservation in both UW solution (P = 0.046) and HTK solution (Table 2).
TABLE 2.
Mean P-Selectin Expression Normalized to β-Actin Internal Control, Expressed as Adjusted Volumetric Units
| UW |
HTK |
||||||
|---|---|---|---|---|---|---|---|
| Duration of preservation | Mean P-selectin | Mean β-actin | Mean P-selectin/β-actin | Mean P-selectin | Mean β-actin | Mean P-selectin/β-actin | P values |
| 4 h | 408.33 | 17195.8 | 0.027 | 29597.67 | 20931.67 | 1.667 | 0.05 |
| 12 h | 3116.7 | 11981.23 | 0.259 | 24245 | 22892.67 | 1.034 | 0.05 |
| 16 h | 3000.67 | 16870.33 | 0.154 | 28939 | 17568 | 1.884 | 0.05 |
| 20 h | 2979.33 | 17133.9 | 0.195 | 27538.33 | 14981 | 3.598 | 0.05 |
| 24 h | 4115.93 | 14677 | 0.279 | 23964 | 17706.33 | 1.747 | 0.05 |
P-selectin Expression was Significantly Down-Regulated in UW Cold Preserved Kidneys
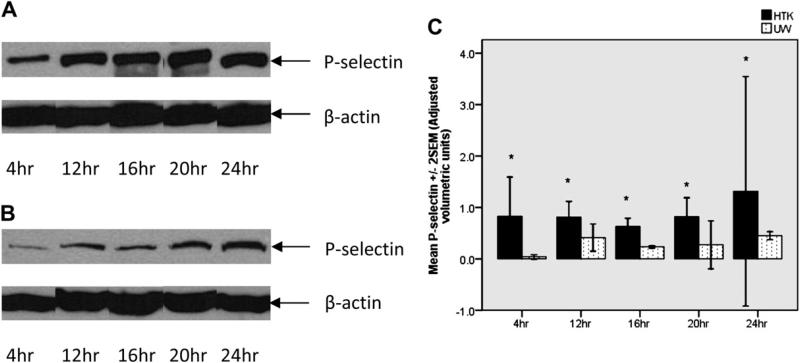
We also compared the P-selectin expression level in kidney tissues cold preserved in UW and HTK solutions, respectively. At the end of all preservation periods examined, P-Selectin expression was consistently less in kidney tissues cold-stored in UW solution compared with HTK solution (P < 0.001) (Fig. 1A, B, and C).
FIG. 1.
UW solution significantly down-regulated P-selectin expression in cold preserved rat kidneys. (A) Western blot analyses of P-selectin in HTK cold preserved kidneys for 4, 12, 16, 20, and 24 h. (B) Western blot analyses of P-selectin in UW cold preserved kidneys for 4, 12, 16, 20, and 24 h. (C) The adjusted volume of the Western blot signal at each time point was measured using a Moleculare Imager Gel Doc XR system and the data normalized to β-actin internal control. As depicted in the graph, P-selectin was significantly down-regulated in UW as compared with HTK preserved kidneys, for all time periods considered.
HTK Preserved Kidneys Manifested Features of Acute Tubular Necrosis (ATN) Earlier
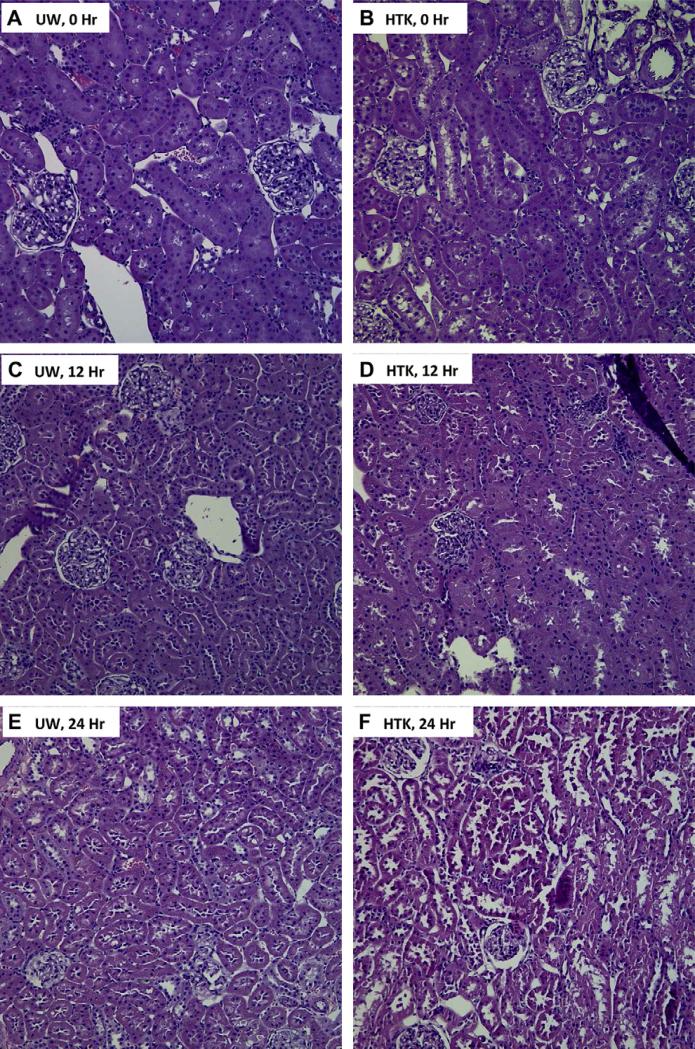
A histologic study was carried out comparing sections of kidneys preserved in UW and HTK solutions for variable length of time. Global and more severe disruption of linings of tubular cells was noted in HTK preserved as early as 24 h, while sections of 24 h UW preserved kidneys maintained normal looking tubules except for some focal and mild necrotic changes. The glomeruli remained largely intact, following the 24 h of cold storage in both UW and HTK solutions (Fig. 2).
FIG. 2.
A. Section of normal kidney tissue which has been flushed with cold (4 °C) UW solution during procurement but which has not been preserved for any duration (0 h). The normal tubules and glomeruli serve for comparison with the preserved specimens. (B) Section of normal kidney tissue which has been flushed with cold (4 °C) HTK solution during procurement but which has not been preserved for any duration (0 h). The normal tubules and glomeruli serve for comparison with the preserved specimens. (C) Section of kidney preserved for 12 h in a cold (4 °C) UW solution. Note normal tubules and glomeruli. (D) Section of kidney preserved for 12 h in a cold (4 °C) HTK solution. Intact glomeruli but edematous looking tubules noted. (E). Section of kidney preserved for 24 h in a cold (4 °C) UW solution. Normal glomeruli are noted. Tubules show focal areas of limited disruption of epithelial lining. (F) Section of kidney preserved for 24 h in a cold (4 °C) HTK solution. Global and much more severe disruption of tubular lining cells is noted. The glomeruli, however, remained intact.
DISCUSSION
The results from this study demonstrate a striking difference in P-selectin expression between UW and HTK preserved kidneys. UW solution has been shown to offer a statistically significant benefit in down-regulating P-selectin expression in ischemic renal grafts, for all periods of cold storage considered in the study. These findings were supported by histologic evidence illustrating earlier appearance of diffuse acute tubular necrotic changes in HTK preserved kidneys than those preserved in UW solution. Furthermore, increased expression of P-selectin was associated with longer preservation periods in both UW and HTK solutions, demonstrating the relevance of P-selectin as a marker of severity of cold ischemia.
Clinical and experimental studies illustrate that antigen-independent events like donor brain death and ischemia/reperfusion injury influence the quality of transplanted solid organs, and are important risk factors for later dysfunction [36–38]. In particular, with increasing use of non-heart-beating donors and expanded criteria donors in clinical renal transplantation, the short-term and late effects of initial ischemia/reperfusion injury are increasingly important. Ischemic insult to organs prior to transplantation up-regulates P-selectin molecules, which are rapidly translocated to endothelial cell surfaces within 5 min of revascularization of the organ, initiating steps leading to transient sticking (tethering) and slowing (rolling) of PMNs to the vascular endothelium [39, 40]. With stronger attachment to endothelium and diapedesis into the tissues via the sequential activity of other adhesion molecules, PMN become activated by locally produced chemokines and cytokines. Activated PMN trigger a further cascade of inflammatory events, stimulating activities of other leukocyte populations, and promoting expression of cytotoxic products.
Such inflammatory response, in turn, is believed to trigger and amplify acute host immunologic activity against the graft [41]. In addition, at a later stage following resolution of the acute insults, the progression of chronic changes developing within a compromised allograft may become autonomous from any host-mediated processes, leading to its increasing deterioration and failure over time [42]. Ultimately, the common pathway for chronic renal dysfunction and failure, the effect over time of decreased numbers of functioning nephrons, as noted clinically [43, 44], unfolds. In contrast, selectin blockade with P-selectin glycoprotein ligand (rPSGL-Ig) was found to decrease inflammation in early post-transplant rat model such that lower doses of maintenance immunosuppression were sufficient to maintain long-term graft function [45–47]. Similar inhibition of P-selectin with a soluble P-selectin glycoprotein ligand, which blocks P- and E-selectins, has also been shown to decrease renal injury secondary to ischemia-reperfusion in rat [48–53] and mice models [54].
In the clinical setting, superior efficacy of UW preservation has been shown for long-term cold storage [1, 4, 8, 9]. Several other studies showed similar outcomes for less-extended cold ischemic times [2, 3, 5–7]. The Euro-transplant multicenter study [7] was the only large randomized multicenter study specifically comparing the two preservation solutions in renal transplantation, and reported no differences in percentage of initial non-function and 3-y survival as end points. Our study is the first to compare the effects of UW and HTK preservation solutions on P-selectin expressions in cold ischemic kidney grafts. UW solution was found to be superior in inhibiting P-selectins expression in cold preserved rat kidneys for up to 24 h. However, a direct comparison of our results with the findings of clinical studies is very difficult, as multiple and hard to control variables are involved in the setting of a clinical trial, which may influence observable differences.
Although it was beyond the scope of this study to define the mechanisms of P-selectin expression differences noted between UW and HTK solutions, contrasts in formulation of the two solutions may contribute. One main difference between these solutions is the addition of antioxidative substances to UW, namely allupurionol and glutathione, whereas HTK does not contain equivalent antioxidative agents. Endothelium, an important source of P-selectin, is also the source and primary target of oxidative stress in ischemia/reperfusion leading to microcirculatory impairment [55]. UW solution was reported to provide superior protection of endothelial cells compared with HTK [56]. Loss of mitochondrial integrity is very likely to contribute to release of reactive oxygen species. UW preservation has been proven to be superior to HTK in preventing mitochondrial respiratory defects occurring only after 8 h of anoxic preservation and reoxygenation [57].
In conclusion, it was demonstrated that UW is superior to HTK solution, in down-regulating P-Selectin expression in cold ischemic rat kidneys, hence potential accumulation of neutrophils and propagation of I-R injury upon reperfusion. The differences in the effects on renal P-selection expression have also emphasized the need for precise evaluation of preservation solutions in organ transplantation. Future studies exploring potential targets of optimizing organ preservation, in addition to correction of deleterious processes by immunosuppressive agents, are warranted.
REFERENCES
- 1.Opelz G, Dohler B. Comparison of histidine-tryptophan-ketoglutarate and University of Wisconsin preservation in renal transplantation. Am J Transplant. 2008;8:1957. doi: 10.1111/j.1600-6143.2008.02271.x. [DOI] [PubMed] [Google Scholar]
- 2.Lynch RJ, Kubus RH, Chenault RH, et al. Comparison of histidine-tryptophan-ketoglutarate and University of Wisconsin preservation in renal transplantation. Am J Transplant. 2008;8:567. doi: 10.1111/j.1600-6143.2007.02065.x. [DOI] [PubMed] [Google Scholar]
- 3.Klaus F, Castro DB, Bittar CM, et al. Kidney transplantation with Belzer or Custodiol solution: A randomized prospective study. Transplant Proc. 2007;39:353. doi: 10.1016/j.transproceed.2007.01.009. [DOI] [PubMed] [Google Scholar]
- 4.Opelz G, Dohler B. Multicenter analysis of kidney preservation. Transplantation. 2007;83:247. doi: 10.1097/01.tp.0000251781.36117.27. [DOI] [PubMed] [Google Scholar]
- 5.Agarwal A, Murdock P, Fridell JA. Comparison of histidine-tryptophan ketoglutarate solution and University of Wisconsin solution in prolonged cold preservation of kidney allografts. Transplantation. 2006;81:480. doi: 10.1097/01.tp.0000196724.89757.79. [DOI] [PubMed] [Google Scholar]
- 6.Agarwal A, Goggins WC, Pescovitz ML, et al. Comparison of histidine-tryptophan Ketoglutarate and University of Wisconsin solutions as primary preservation in renal allografts undergoing pulsatile perfusion. Transplant Proc. 2005;37:2016. doi: 10.1016/j.transproceed.2005.03.038. [DOI] [PubMed] [Google Scholar]
- 7.de Boer J, Smits JMA, De Meester J, et al. A randomized multicenter study on kidney preservation comparing HTK with UW. Transplant Proc. 1999;31:2065. doi: 10.1016/s0041-1345(99)00263-8. [DOI] [PubMed] [Google Scholar]
- 8.Roels L, Coosemans W, Donck J, et al. Inferior outcome of cadaveric kidneys preserved for more than 24 hours in histidine-tryptophan ketoglutarate solution. Transplantation. 1998;66:1660. doi: 10.1097/00007890-199812270-00015. [DOI] [PubMed] [Google Scholar]
- 9.Opelz G, Wujciak T. Comparative analysis of kidney preservation methods. Transplant Proc. 1996;28:87. [PubMed] [Google Scholar]
- 10.Groenewoud AF, de Boer J. A report of the eurotransplant randomized multicenter study comparing kidney graft preservation with HTK, UW, EC solutions. Transpl Int. 1997;7:S479. doi: 10.1111/j.1432-2277.1994.tb01423.x. [DOI] [PubMed] [Google Scholar]
- 11.Groenewoud AF, Thorogood J. Current status of the eurotransplant randomized multicenter study comparing kidney graft preservation with histidine-tryptophan-ketoglutarate, University of Wisconsin, and Euro-Collins solutions. Transplant Proc. 1993;25:1582. [PubMed] [Google Scholar]
- 12.Lefer AM, Lefer DJ. The role of nitric oxide and cell adhesion molecules on the microcirculation in ischemia-reperfusion. Cardiovasc Res. 1996;32:743. [PubMed] [Google Scholar]
- 13.Butcher EC. Leucocyte-endothelial cell recognition: Three (or more) steps to specificity and diversity. Cell. 1991;67:1033. doi: 10.1016/0092-8674(91)90279-8. [DOI] [PubMed] [Google Scholar]
- 14.Lorant DE, Patel KD, McIntyre TM, et al. Co-expression of GMP-140 and PAF by endothelium stimulated by histamine or thrombin: A juxtacrine system for adhesion and activation of neutrophils. J Cell Biol. 1991;115:223. doi: 10.1083/jcb.115.1.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kansas GS. Selectins and their ligands: Current concepts and controversies. Blood. 1996;88:3259. [PubMed] [Google Scholar]
- 16.McEver RP. GMP-140: A receptor for neutrophils and monocytes on activated platelet and endothelium. J Cell Biochem. 1991;45:156. doi: 10.1002/jcb.240450206. [DOI] [PubMed] [Google Scholar]
- 17.Weyrich AS, Buerke M, Albertine KH, et al. Time course of coronary vascular endothelial adhesion molecule expression during reperfusion of the ischemic feline myocardium. J Leukocyte Biol. 1995;57:45. doi: 10.1002/jlb.57.1.45. [DOI] [PubMed] [Google Scholar]
- 18.Griffin JD, Spertini O, Ernst TJ, et al. Granulocyte-macrophage colony-stimulation factor and other cytokines regulate surface expression of the leukocyte adhesion molecule-1 on human neutrophils, monocytes, and their precursors. J Immunol. 1990;145:576. [PubMed] [Google Scholar]
- 19.Chmoun F, Burne M, O'Donnell M, et al. Pathophysiologic role of selectins and their ligands in ischemia reperfusion injury. Front Biosci. 2000;5:E103. doi: 10.2741/chamoun. [DOI] [PubMed] [Google Scholar]
- 20.Bevilacqua MB, Stengelin S, Gimbrone MA, Jr., et al. Endothelial leukocyte adhesion molecule 1: An inducible receptor for neutrophils related to complement regulatory proteins and lectins. Science. 1989;243:1160. doi: 10.1126/science.2466335. [DOI] [PubMed] [Google Scholar]
- 21.Kansas GS. Selectins and their ligands: Current concepts and controversies. Blood. 1996;88:3259. [PubMed] [Google Scholar]
- 22.Johnson RC, Mayadas TN, Frenette PS, et al. Blood cell dynamics in P-selectin-deficient mice. Blood. 1995;86:1106. [PubMed] [Google Scholar]
- 23.Subramaniam M, Saffaropour S, Watson SR, et al. Reduced recruitment of inflammatory cells in a contact hypersensitivity response in P-selectin –deficient mice. J Exp Med. 1995;181:2277. doi: 10.1084/jem.181.6.2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Grober JS, Bowen BL, Ebling H, et al. Monocyte-endothelial adhesion in chronic rheumatoid arthritis. J Clin Invest. 1993;91:2609. doi: 10.1172/JCI116500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bretschneider HJ. Myocardial protection. Thorac Cardiovasc Surg. 1980;28:295. doi: 10.1055/s-2007-1022099. [DOI] [PubMed] [Google Scholar]
- 26.Lynch RJ, Kubus RH, Chenault RH, et al. Comparison of histidine-tryptophan-ketoglutarate and University of Wisconsin preservation in renal transplantation. Am J Transplant. 2008;8:567. doi: 10.1111/j.1600-6143.2007.02065.x. [DOI] [PubMed] [Google Scholar]
- 27.Klaus F, Castro DB, Bittar CM, et al. Kidney transplantation with Belzer or Custodiol solution: A randomized prospective study. Transplant Proc. 2007;39:353. doi: 10.1016/j.transproceed.2007.01.009. [DOI] [PubMed] [Google Scholar]
- 28.Stewart ZA, Lonze BE, Warren DS, et al. Histidine-Tryptophan-Ketoglutarate (HTK) is associated with reduced graft survival of deceased donor kidney transplants. Am J Transplant. 2009;9:1048. doi: 10.1111/j.1600-6143.2008.02545.x. [DOI] [PubMed] [Google Scholar]
- 29.Schneeberger S, Biebl M, Steurer W, et al. A prospective randomized multicenter trial comparing histidine-tryptophan-ketoglutarate versus University of Wisconsin perfusion solution in clinical pancreas transplantation. Transpl Int. 2009;22:217. doi: 10.1111/j.1432-2277.2008.00773.x. [DOI] [PubMed] [Google Scholar]
- 30.Becker T, Ringe B, Nyibata M, et al. Pancreas transplantation with histidine-tryptophan-ketoglutarate (HTK) solution and University of Wisconsin (UW) solution: Is there a difference? JOP. 2007;8:304. [PubMed] [Google Scholar]
- 31.Salehi P, Hansen MA, Avila JG, et al. Human islet isolation outcomes from pancreata preserved with histidine-tryptophan-ketoglutarate versus University of Wisconsin solution. Transplantation. 2006;82:982. doi: 10.1097/01.tp.0000232310.49237.06. [DOI] [PubMed] [Google Scholar]
- 32.Englesbe MJ, Moyer A, Kim DY, et al. Early pancreas transplant outcomes with histidine-tryptophan-ketoglutarate preservation: A multicenter study. Transplantation. 2006;82:136. doi: 10.1097/01.tp.0000225764.21343.e3. [DOI] [PubMed] [Google Scholar]
- 33.Mangus RS, Fridell JA, Vianna RM, et al. Comparison of histidine-tryptophan-ketoglutarate solution and University of Wisconsin solution in extended criteria liver donors. Liver Transpl. 2008;14:365. doi: 10.1002/lt.21372. [DOI] [PubMed] [Google Scholar]
- 34.Mangus RS, Tector AJ, Agarwal A, et al. Comparison of histidine-tryptophan-ketoglutarate solution (HTK) and University of Wisconsin solution (UW) in adult liver transplantation. Liver Transpl. 2006;12:226. doi: 10.1002/lt.20552. [DOI] [PubMed] [Google Scholar]
- 35.Englesbe MJ, Heidt D, Sung R, et al. Does using HTK solution for cold perfusion of cadaveric kidneys save money? Transplantation. 2006;82:580. doi: 10.1097/01.tp.0000231845.38735.a5. [DOI] [PubMed] [Google Scholar]
- 36.Takada M, Nadeau KC, Hancock WW, et al. Effects of explosive brain death on cytokine activation of peripheral organs in the rat. Transplantation. 1998;65:1533. doi: 10.1097/00007890-199806270-00001. [DOI] [PubMed] [Google Scholar]
- 37.Terasaki PI, Cecka JM, Gjertson DW, et al. High survival rates of kidney transplants from spousal and living unrelated donors. N Engl J Med. 1995;333:333. doi: 10.1056/NEJM199508103330601. [DOI] [PubMed] [Google Scholar]
- 38.Takada M, Nadeau KC, Shaw GD, et al. The cytokine-adhesion molecule cascade in ischemia/reperfusion injury of rat kidney. Inhibition by a soluble P-selectin ligand. J Clin Invest. 1997;99:2682. doi: 10.1172/JCI119457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ley K, Tedder TF. Leukocyte interactions with vascular endothelium. J Immunol. 1995;155:525. [PubMed] [Google Scholar]
- 40.McEver RP, Moor KL, Cummings RD. Leukocyte trafficking mediated by selectin carbohydrate interactions. J Biol Chem. 1995;270:11025. doi: 10.1074/jbc.270.19.11025. [DOI] [PubMed] [Google Scholar]
- 41.Pratschke J, Wilhelm MJ, Kusaka M, et al. Accelerated rejection of renal allografts from brain-dead donors. Ann Surg. 2000;232:263. doi: 10.1097/00000658-200008000-00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tullius SG, Hancock WW, Heemann U, et al. Reversibility of chronic renal allograft rejection. Critical effect of time after transplantation suggests both host immune dependent and independent phases of progressive injury. Transplantation. 1994;58:93. [PubMed] [Google Scholar]
- 43.Brenner BM, Milford EL. Nephron under dosing: A programmed cause of chronic renal allograft failure. Am J Kidney Dis. 1993;21:66. doi: 10.1016/0272-6386(93)70097-i. [DOI] [PubMed] [Google Scholar]
- 44.Chertow GM, Milford EL, Mackenzie HS, et al. Antigen-independent determinants of cadaver kidney transplant failure. JAMA. 1996;276:1732. [PubMed] [Google Scholar]
- 45.Gasser M, Waaga-Gasser AM, Grimm MW, et al. Selectin blockade plus therapy with low-dose sirolimus and cyclosporine A prevent brain death induced renal allograft dysfunction. Am J Transplant. 2005;5:662. doi: 10.1111/j.1600-6143.2005.00763.x. [DOI] [PubMed] [Google Scholar]
- 46.Kusaka M, Zandi-Nejad K, Kato S, et al. Exploitation of the continuum between early ischemia/reperfusion injury and host alloresponsiveness: Indefinite kidney allograft survival by treatment with a soluble P-selectin ligand and low-dose cyclosporine in combination. Transplantation. 1999;67:1255. doi: 10.1097/00007890-199905150-00010. [DOI] [PubMed] [Google Scholar]
- 47.Kusaka M, Nadeau KC, Takada M, et al. Influence of initial antigen-independent events on acute allograft rejection: Inhibition by a soluble P-selectin ligand and low-dose cyclosporine in combination. Transplant Proc. 1998;30:1027. doi: 10.1016/s0041-1345(98)00136-5. [DOI] [PubMed] [Google Scholar]
- 48.Gasser M, Waaga AM, Kist-Van Holthe JE, et al. Normalization of brain death-induced injury to rat renal allograft by recombinant soluble P-selectin glycoprotein ligand. J Am Soc Nephrol. 2002;13:1937. doi: 10.1097/01.asn.0000019401.12257.c4. [DOI] [PubMed] [Google Scholar]
- 49.Pratschke J, Kofla G, Wilhelm M, et al. Improvements in early behavior of kidney allografts after donor treatment. Transplant Proc. 2002;34:2213. doi: 10.1016/s0041-1345(02)03206-2. [DOI] [PubMed] [Google Scholar]
- 50.Takada M, Nadeau KC, Hancock WW, et al. Effects of explosive brain death on cytokine activation of peripheral organs in the rat. Transplantation. 1998;65:1533. doi: 10.1097/00007890-199806270-00001. [DOI] [PubMed] [Google Scholar]
- 51.Takada M, Nadeau KC, Shaw GD, et al. The cytokine-adhesion molecule cascade in ischemia/reperfusion injury of the rat kidney. Inhibition by a soluble P-selectin ligand. J Clin Invest. 1997;99:2682. doi: 10.1172/JCI119457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Takada M, Nadeau KC, Shaw GD, et al. Early cellular and molecular changes in ischemia/reperfusion injury: Inhibition by a selectin antagonist, P-selectin glycoprotein ligand-1. Transplant Proc. 1997;29:1324. doi: 10.1016/s0041-1345(96)00577-5. [DOI] [PubMed] [Google Scholar]
- 53.Takada M, Nadeau KC, Shaw GD, et al. Prevention of late renal changes after initial ischemia/reperfusion injury by blocking early selectin binding. Transplantation. 1997;64:1520. doi: 10.1097/00007890-199712150-00003. [DOI] [PubMed] [Google Scholar]
- 54.Singbartl K, Green SA, Ley K. Blocking P-selectin protects from ischemia/reperfusion-induced acute renal failure. FASEB J. 2000;14:48. doi: 10.1096/fasebj.14.1.48. [DOI] [PubMed] [Google Scholar]
- 55.Koo DD, Welsh KI, West NE, et al. Endothelial cell protection against ischemia/reperfusion injury by lecithinized superoxide dismutase. Kidney Int. 2001;60:786. doi: 10.1046/j.1523-1755.2001.060002786.x. [DOI] [PubMed] [Google Scholar]
- 56.Janssen H, Janssen PH, Broelsch CE. UW is superior to Celsior and HTK in the protection of human liver endothelial cells against preservation injury. Liver Transpl. 2004;10:1514. doi: 10.1002/lt.20309. [DOI] [PubMed] [Google Scholar]
- 57.Steinlechner-Maran R, Eberl T, Kunc M, et al. Respiratory defect as an early event in preservation-reoxygenation injury of endothelial cells. Transplantation. 1997;63:136. doi: 10.1097/00007890-199701150-00025. [DOI] [PubMed] [Google Scholar]