Spatial organization of adhesion: force-dependent regulation and function in tissue morphogenesis
The Heisenberg laboratory reviews the spatial organization of signalling complexes at cell–matrix and cell–cell contact sites and its impact on cell integrity, cellular polarity and tissue morphogenesis.
Keywords: adhesion, spatial organization, tissue morphogenesis
Abstract
Integrin- and cadherin-mediated adhesion is central for cell and tissue morphogenesis, allowing cells and tissues to change shape without loosing integrity. Studies predominantly in cell culture showed that mechanosensation through adhesion structures is achieved by force-mediated modulation of their molecular composition. The specific molecular composition of adhesion sites in turn determines their signalling activity and dynamic reorganization. Here, we will review how adhesion sites respond to mecanical stimuli, and how spatially and temporally regulated signalling from different adhesion sites controls cell migration and tissue morphogenesis.
Introduction
The ability of cells to sense and respond to their mechanical environment is critical for cell and tissue rearrangement and homeostasis in development. Cells establish contact with their environment through a variety of receptors mediating adhesive interactions with the extracellular matrix (ECM) and neighbouring cells. Studies in the last decades showed that cell–cell and cell–matrix adhesions are critical for cells and tissues to respond to mechanical stimuli from their environment. Both cell–cell and cell–matrix adhesions bear intrinsic mechanosensitivity, which allows them to promptly respond to stress and effectively propagate signals controlling cell shape and motility. This mechanosensitive response has been associated with pronounced changes in the size and molecular composition of specific adhesion sites and, consequently, the signals evoked by those adhesion sites. Eventually, the combination of different signals from heterogeneous adhesion sites allows cells and tissues to undergo contiguous alterations in shape and motility.
In this review, we will present recent advances in the understanding of how signals evoked by different adhesion sites are spatially and temporally integrated during cell and tissue movements, and how mechanosensation functions therein. We will first give an overview of studies in cell culture that have addressed how stress affects local changes in cell–matrix and cell–cell adhesion site composition and how differences in the composition of those adhesion sites affect their adhesive and signalling properties. We will then describe how the spatiotemporal propagation of signals from different types of adhesion sites control cell integrity, polarity and movements.
Adhesion molecules, such as integrins and cadherins, are generally thought to have multiple functions in cell and tissue morphogenesis, including the regulation of cell adhesion, gene expression and growth factor signalling. As these different functions have already been extensively covered by excellent recent reviews (Stemmler, 2008; Winklbauer, 2009; Rozario and DeSimone, 2010), here we will primarily focus on the function of mechanosensation and spatiotemporal coordination of adhesion signalling in cell and tissue morphogenesis.
Function of mechanosensation in adhesion site modulation
Both cell–matrix and cell–cell adhesion sites are highly heterogenous structures in terms of their molecular composition, spatial organization and dynamics. In this section, we will discuss how mechanical signals can spatially and temporally control the composition of adhesion sites, and how differences in the composition of adhesion sites in turn modulate their adhesive, mechanosensitive and signalling properties.
Cell–matrix adhesion
Clustering of transmembrane integrin receptors is central to cell–matrix adhesion. Integrin receptors are functional dimers of α- and β-integrin subunits, and the combination of different α- and β-subunits determines the substrate specificity of the dimer (reviewed in Danen and Sonnenberg, 2003). Integrins bind with their extracellular domains to specific sites of ECM proteins, including vitronectin, fibronectin, laminin and collagen. The cytoplasmic tail of integrin receptors triggers the assembly of a multi-molecular network of scaffolding and signalling proteins that link the integrin receptors to the actin cytoskeleton (reviewed in Cohen et al, 2004; Geiger et al, 2009).
Studies on the spreading and migration of fibroblasts on two-dimensional fibronectin-coated substrates provided major insight into the formation and maturation of integrin-ECM adhesion sites (Figure 1A). During spreading, fibroblasts build different forms of integrin-ECM adhesion sites in a highly spatially and temporally ordered manner (Ballestrem et al, 2001; Zamir and Geiger, 2001). These adhesion sites can be morphologically subdivided in at least three types with distinct molecular composition, dynamics and adhesive properties.
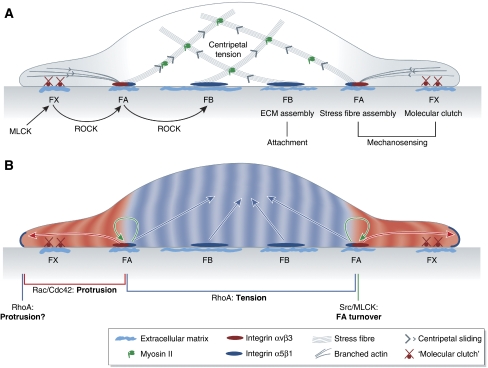
Figure 1.
Force-dependent remodelling of integrin-ECM adhesion site composition and signalling activity. (A) Tension defines the molecular composition and size of integrin-ECM adhesion sites. Assembly of nascent integrin αvβ3-containing focal complexes (FXs) at the cell edge is driven by MLCK. FXs interact with sliding actin filaments through a ‘molecular clutch', enabling tension-dependent maturation into focal adhesions (FA), which is driven by ROCK. FAs trigger stress fibre assembly. ROCK-dependent maturation of integrin α5β1-containing fibrillar adhesions is necessary for ECM assembly and stable cell-substrate attachment. The maturation of integrin-ECM adhesion structures is dependent on tension created by myosin-driven centripetal sliding of actin cytoskeleton. (B) Molecular composition of integrin-ECM adhesion sites defines their signalling activities. RhoA is activated at the edge of peripheral cell protrusion by an unknown mechanism. Integrin αvβ3-containing FAs trigger peripheral Rac/Cdc42 activation that propagates distally and is necessary for stabilizing cell protrusions and downregulating peripheral RhoA activity. FAs also trigger activation of Src and MLCK that are essential for FA turnover during cell spreading and migration. Central integrin α5β1-containing adhesions reactivate RhoA, thereby contributing to the increase of centripetal actomyosin tension.
Nascent focal complexes (FXs) are highly dynamic dot-like structures that assemble under the lamellipodium during initial cell spreading and/or migration. The formation of these complexes depends on myosin light chain kinase (MLCK)-controlled myosin contractility (Giannone et al, 2007). These nascent complexes are enriched in integrin αVβ3 and contain only a subset of focal adhesion (FA) proteins, among which vinculin and talin are most prominent (Katz et al, 2000; Zamir et al, 2000; reviewed in Zaidel-Bar et al, 2003, 2004). Within a short time span (around one minute), nascent complexes are either disassembled or undergo maturation into FAs (Zaidel-Bar et al, 2003). Assembly and maturation of integrin-based adhesion sites are strongly dependent on local tension (Galbraith et al, 2002), substrate stiffness (Saez et al, 2005) and requires Rho kinase (ROCK)-mediated cytoskeleton contractility (Ballestrem et al, 2001). Generally, force is thought to function in integrin-based adhesion assembly and maturation by facilitating integrin activation (Astrof et al, 2006; Puklin-Faucher et al, 2006; Friedland et al, 2009; reviewed in Kong et al, 2009; Puklin-Faucher and Sheetz, 2009). Integrins are activated by an allosteric conformational switch (Frelinger et al, 1990; Takagi et al, 2002; Kim et al, 2003; Xiao et al, 2004; Askari et al, 2009) that increases their ligand-binding affinity. Binding of talin (Tadokoro et al, 2003; Wegener et al, 2007; reviewed in Anthis et al, 2009) and kindlins (Ma et al, 2008; Montanez et al, 2008; Moser et al, 2008) to the cytoplasmic portion of integrin dimers also promotes integrin activation. Activated integrins form a complex with their ligand and talin, which again is needed for integrin clustering (Kim et al, 2004; Cluzel et al, 2005). Force has also been shown to promote integrin-based adhesion assembly and maturation by inducing conformational changes of talin that facilitates binding to vinculin (Lee et al, 2007; del Rio et al, 2009). The vinculin–talin complex in turn acts as a ‘molecular clutch' that enables transmission of actomyosin-mediated centripetal tension between the cytoskeleton and integrins (Hu et al, 2007). Therefore, depending on the tension exerted by the cytoskeleton and the pliability of the substrate, FXs undergo ROCK-dependent reinforcement, recognizable by an increase in integrin and vinculin density (Ballestrem et al, 2001; Humphries et al, 2007). This stress-induced increase in integrin and vinculin density then triggers the transformation of FXs into mature FAs, which are multi-molecular scaffolds consisting of >100 different components (reviewed in Geiger et al, 2009). The different components of mature FAs have both scaffolding (e.g. p130Cas and paxillin) and signalling (focal adhesion kinase (FAK) and Src) functions, some of which are in turn modulated by tension (Sawada and Sheetz, 2002). Mature FAs trigger formin-mediated polymerization of actin into stress fibres (Hotulainen and Lappalainen, 2006) and translate mechanical stimuli from the outside of the cell into intracellular signals that determine cell shape and motility (see section ‘Cell–matrix adhesion signalling' below). Some of the mature FA components are essential for haptotactic movements of cells towards stiffer substrates, suggesting that they have mechanosensory functions (Wang et al, 2001a; Frey et al, 2006), and drive FA turnover, needed for efficient cell migration (Webb et al, 2004).
Nascent FXs and FAs are predominantly required for probing the environment and for haptotactic migration. These functions are thought to be mediated by integrin β3, which is preferentially localized at FXs and FAs and has been shown to have a critical function in mechanosensing (Roca-Cusachs et al, 2009). FAs in the periphery of the cell either become disassembled in the course of cell migration or slide under the body of the migrating cell and evolve into fibrillar adhesions (FB). Fibrillar adhesions accumulate integrin α5β1 dimers, which provide stable adhesion to the ECM (Roca-Cusachs et al, 2009) and trigger ECM assembly (Huveneers et al, 2008). The transition from integrin αVβ3-enriched FAs towards integrin α5β1-enriched fibrillar adhesions is driven by ROCK-stimulated actomyosin contractility (Figure 1A) (Bershadsky et al, 2003; Wierzbicka-Patynowski and Schwarzbauer, 2003).
Disassembly of FAs also seems to be a force-dependent process. Myosin II is required for controlled FA disassembly at the trailing edge of migrating cell (Vicente-Manzanares et al, 2007). Conversely, decrease of local stress exerted at individual FAs by stress fibres leads to their rapid disassembly (Chrzanowska-Wodnicka and Burridge, 1996). Disassembly of FAs is also regulated by recruitment of specific proteins (Marshall et al, 2009) and stress-induced conformational changes of particular components (Chen et al, 2005; Papusheva et al, 2009).
Taken together, studies on integrin-mediated cell–matrix adhesion in two-dimensional cell cultures have provided evidence that mechanical stimuli regulate the molecular composition and spatial organization of cell–matrix adhesion sites, and that different adhesion sites have distinct mechanosensory properties. In three-dimensional cell culture, integrin-ECM adhesion sites are less heterogeneous (Cukierman et al, 2001), suggesting that stress-induced maturation and functional specification of adhesion sites might be less pronounced. However, the stochiometry and dynamics of FA sites, which have previously been shown to be an important factor in adhesion site maturation and segregation (Digman et al, 2009), has not yet analysed the three-dimensional cultures. It will be important to test how the complex machinery of FA site maturation and functional specification is coordinated in three-dimensional environments such as the developing embryo.
Cell–cell adhesion
Adherens junctions are localized cell–cell contacts that use classical cadherins as adhesion molecules. Adherens junction formation is initiated by the homophilic interaction between the extracellular domains of classical cadherins in neighbouring cells. The initial contact is followed by clustering of cadherins, contact zone enlargement and contact strengthening (Figure 2A).
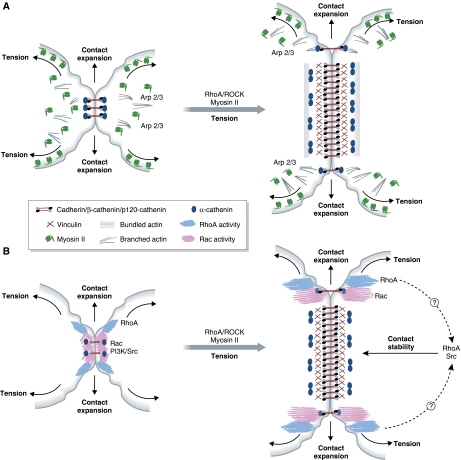
Figure 2.
Force-dependent remodelling of cell–cell adhesion site composition and signalling activity. (A) Tension drives maturation of adherens junctions. Early adherens junctions are enriched in Arp 2/3 and branched actin. Actomyosin/ROCK-mediated tension is required for the expansion of adherens junction. As the junction expands, the molecular composition of its central part changes. The α-catenin dimerizes and dissociates from the cadherin–β-catenin complex and additional actin-binding adaptors, such as vinculin, are recruited. The α-catenin dimers are thought to modulate actin reorganization by promoting the assembly of junctional actin bundles. (B) Adherens junction maturation alters its signalling activity. Upon initial cadherin engagement at cell–cell contacts, Src/PI3 kinase as well as RhoA and Rac become activated. These signals propagate as the junction extends, and are downregulated at the mature, central region of the adherens junction. At the same time, RhoA and Src/PI3 kinase activities are essential for the stability of mature adherens junctions.
The cytoplasmic domains of cadherins bind proteins that are involved in regulating the actin cytoskeleton, gene transcription and vesicular traffic. The most prominent of those proteins are β-catenin, p120-catenin and α-catenin. E-cadherin must bind to β-catenin in order to exit from the endoplasmic reticulum (Chen et al, 1999). This interaction is regulated by phosphorylation (Lickert et al, 2000; Huber and Weis, 2001). Once at the plasma membrane, E-cadherin is stabilized by the binding of p120-catenin during initial contact formation. α-catenin is thought to be involved in interactions of membrane-bound E-cadherin complexes with components of the actin cytoskeleton, although the precise mechanisms of this interaction are still debated.
Monomeric α-catenin has been proposed to bind to the E-cadherin–β-catenin complex, but not to actin (Yamada et al, 2005). It has further been suggested that a local increase of α-catenin concentration upon cadherin clustering triggers α-catenin dimerization. Dimeric α-catenin in turn binds actin filaments, but not the E-cadherin–β-catenin complex (Drees et al, 2005; Yamada et al, 2005). Binding of α-catenin dimers to actin promotes bundling of actin filaments. At the same time, dimerized α-catenin inhibits binding of Arp2/3 (actin-related proteins 2 and 3) complex to actin filaments, thereby suppressing local actin branching (Drees et al, 2005). In vivo analysis showed that α-catenin is associated with adherens junction remodelling in the early embryonic epithelium of Drosophila, and that E-cadherin clusters are stabilized at adherens junctions through connections with cortical actin in an α-catenin-independent manner (Cavey et al, 2008). An indirect link between the E-cadherin–β-catenin–α-catenin complex and actin is also provided by epithelial protein lost in neoplasm (EPLIN), an actin-binding protein also known as Lima-1 (Abe and Takeichi, 2008).
There is increasing evidence that adherens junctions are heterogenous in their molecular composition, adhesive strength and dynamics. Similar to cell–matrix adhesions sites, this heterogeneity is thought to be regulated by mechanotransduction. Recent studies showed that cadherin-mediated cell–cell junctions are modulated in a force-dependent manner (Ladoux et al, 2010). When E-cadherin-expressing cells contact each other, MLC phosphorylation increases (Shewan et al, 2005), and myosin II is recruited to the peripheral zone of the contact where it aids in expanding the contact area (Yamada and Nelson, 2007). Myosin II activity is required for cadherin-clustering and cell–cell contact zone expansion (Yamada and Nelson, 2007) and strengthening (Liu et al, 2010). Myosin II activity further enhances cadherin junctions by modulating vesicular transport of new E-cadherin molecules to the forming junction (Kametani and Takeichi, 2007). Eventually, this leads to increased cadherin density at the contact site, and consequently increased probability of cadherin binding and increased adhesion (Zhang et al, 2009).
At later stages of adherens junction maturation, myosin II is depleted from the central region of the expanding contact (Yamada and Nelson, 2007). At mature cell–cell junctions, myosin IV recruits vinculin, thereby contributing to adhesion strengthening (Maddugoda et al, 2007). Recent work showed that recruitment of vinculin to mature cell junctions is mediated by stress-dependent conformational changes in α-catenin (Yonemura et al, 2010). It appears, therefore, that cadherin-mediated cell–cell junctions undergo stress-dependent adhesion strengthening in a way reminiscent of cell–matrix adhesion sites.
Taken together, these data show that cadherin-mediated cell–cell contacts are heterogenous structures, which undergo stress-dependent changes in their molecular composition and structure. The function of this heterogeneity for adhesion persistence and strength, and the precise mechanisms controlling it, remain to be elucidated.
Spatial integration of signals from cell–cell and cell–matrix adhesion sites
Engagement of adhesion receptors triggers numerous signals that control cell shape, polarity and integrity. In this section, we will discuss how different adhesion sites translate stress into signals regulating cell morphogenesis.
Cell–matrix adhesion signalling
Integrins are thought to regulate membrane dynamics and cell shape changes by modulating the activity of Rho-family GTPases, such as RhoA, Rac, Cdc42 and Src-family kinases (Figure 1B). Integrin-dependent spreading of fibroblasts on fibronectin-coated substrates causes a rapid and transient upregulation of Rac and Cdc42, and downregulation of RhoA activity. This effect of integrins depends on formation of peripheral FAs, which are enriched in integrin αVβ3 receptors and contain phosphorylated (active) FAK/Src/p130Cas complexes and paxillin. It has further been suggested that FA-mediated Rac activation and FAK/Src complex formation synergistically downregulate RhoA activity, which in turn relieves local tension, thereby facilitating Rac- and Cdc42-driven protrusion formation (reviewed in Huveneers and Danen, 2009).
Recent advances in Förster Resonance Energy Transfer (FRET) imaging and the development of photo-activatable proteins allowed visualizing the spatiotemporal coordination of RhoA and Rac/Cdc42 signalling in individual protrusion formation (Machacek et al, 2009) (Figure 1B). At the onset of protrusion formation, a narrow band of RhoA activation becomes visible at the cell edge. This is followed by Cdc42 and Rac activation close to the cell edge, which subsequently downregulates RhoA activity. This is most clearly shown in experiments where localized activation of a photo-switchable version of Rac leads to a localized downregulation of RhoA, which then rapidly spreads across the cell (Wu et al, 2009). These data suggest that RhoA has a function in the initiation of protrusion formation, whereas Rac and Cdc42 inhibit RhoA in order to reinforce and stabilize the expanding protrusions.
It is still unclear whether adhesion signals are responsible for RhoA activation at the protruding edge. In contrast, Rac is activated at the sites of FA assembly and a wave of Rac activation propagates outwards during cell spreading (Xia et al, 2008). Moreover, integrin clustering is essential for its interaction with Rac and p21-activated kinase (PAK) (del Pozo et al, 2000), and the Rac/PAK complex is required and sufficient to initiate protrusion formation (Wu et al, 2009). This indicates that the assembly of peripheral FAs causes Rac activation and that the Rac/PAK complex causes initiation and/or stabilization of new cell protrusions.
The suppression of RhoA activity upon integrin/Rac signalling at the cell periphery of spreading cells and in advancing cell protrusions is transient and followed by an increase of RhoA activity as soon as mature integrin α5β1-containing adhesion sites and stress fibres have assembled (Dubash et al, 2007; Lim et al, 2008). However, the exact mechanisms by which integrin α5β1 engagement activates RhoA is not yet clear. RhoA activation is essential for ECM assembly (Wierzbicka-Patynowski and Schwarzbauer, 2003) and stable attachment of cells to the substrate (Danen et al, 2002). Moreover, integrin-mediated activation of RhoA and its effectors ROCK and Diaphanous is required for cell integrity and stress-induced polarization (see also section ‘Adhesion signalling and cell integrity').
Stimulation of integrins leads to assembly and activation of the FAK/Src complex, followed by downstream activation of ERK and MLCK (Schaller et al, 1994; Schlaepfer et al, 1998; Fincham et al, 2000; reviewed in Mitra et al, 2005). These signals are essential for haptotactic cell migration and modulation of FA turnover (Wang et al, 2001a; Webb et al, 2004). Local activation of Src by mechanical stimuli propagates rapidly across the cell body (Wang et al, 2005; Na et al, 2008). Integrin-mediated Src activation has been suggested to control cell shape changes in both a Rac-dependent and -independent manner (Wang et al, 2005; Ouyang et al, 2008; Poh et al, 2009).
Taken together, different cell–matrix adhesion sites propagate distinct spatial signals. Peripheral adhesion sites enriched in integrin αVβ3 activate Rac, which in turn downregulates RhoA activity and thereby facilitates protrusion formation. In contrast, more centrally located integrin α5β1-enriched fibrillar adhesion sites re-activate RhoA, which is essential for ECM assembly and stable cell attachment. Src activation is involved in mechanosensing and turnover of FAs.
Cell–cell adhesion signalling
Signals from cadherin containing cell–cell adhesion sites can substantially vary depending on the location and maturity of the junctions. Biochemical studies showed that Rac1 and Cdc42 are activated upon E-cadherin engagement, whereas RhoA activity is downregulated on a slower time scale (Noren et al, 2001; Betson et al, 2002). E-cadherin-mediated suppression of RhoA activity is thought to be mediated by p120-catenin (Anastasiadis et al, 2000). Recent works using FRET biosensors and in vivo imaging of fluorescent reporters provided detailed insight into the spatial organization of adherens junction signalling (Figure 2B) (Yamada and Nelson, 2007; Perez et al, 2008). Upon initial E-cadherin-mediated cell–cell contact, Rac is activated directly at the contact site. As contact expansion proceeds, active Rac, together with the Arp2/3 complex and associated formation of lamellipodia, is restricted to the external, newly formed contact sites. In contrast to the biochemical observations mentioned above, RhoA appears to be also activated at the newly formed contact sites, and ROCK-driven actomyosin contractility is required to expand the contact. Both RhoA and Rac activities rapidly diminish as E-cadherin density increases during contact maturation. This downregulation is accompanied by the disappearance of branched actin at the maturing contact. It is not yet clear what drives such rapid downregulation of Rho-family GTPases in the maturing adherens junction. However, as in many systems increased RhoA and Rac activities are associated with destabilization of adherens junctions (Sahai and Marshall, 2002; Yano et al, 2004; de Rooij et al, 2005), it is quite likely that this downregulation stabilizes the contact.
Cadherin-mediated modulation of Src activity was also shown to control adherens junction maturation (McLachlan et al, 2007; Ren et al, 2009). Src activation at forming cell–cell contact sites leads to an accumulation of phosphatidylinositol 3-kinase (PI3K), which propagates across the membrane while being downregulated at the initial contact site (Perez et al, 2008). PI3K in turn is thought to stabilize adherens junctions by phosphorylating the actin-regulatory protein contractin (Ren et al, 2009) at the adhesion site. Notably, constitutively active PI3K completely rescues the adherens junction defect resulting from defective Src activity, whereas the propagation of Rac activity is not affected by the inhibition of PI3K (Perez et al, 2008). It thus appears that cadherin engagement at forming cell–cell contacts triggers two signalling pathways—RhoA/Rac signalling responsible for contact expansion and Src/PI3K signalling involved in adherens junction stabilization. Questions remain as to how these two pathways interact at maturing adhesion sites in order to achieve junction stabilization and remodelling.
Taken together, adherens junctions at different stages of maturity activate different signals, which in turn control distinct aspects of peripheral cytoskeleton dynamics.
Adhesion signalling and cell integrity
Cells are exposed to mechanical stimuli and generate forces themselves through actomyosin contractions. The ability of cells to exert, transmit and resist forces without loosing their integrity requires a close coordination of local extrinsic and intrinsic stress. Force-induced signals from different adhesion sites are critical for the coordinated response of cells exposed to mechanical stimuli.
The cytoplasm of animal cells is under pressure generated by the contractile actomyosin cortex (Figure 1A) (Dai et al, 1999; Hagmann et al, 1999). In order to resist this centripetal tension and maintain their specific shape, cells have to build structures that can bear the load. The microtubule network has been suggested to represent one of those structures (Wang et al, 2001b); however, microtubules are unlikely to bear the major fraction of forces that can be measured at the periphery of adherent cells (Stamenovic et al, 2002). It has, therefore, been proposed that adhesion contacts bridged by actomyosin cables are the main structures bearing the load exerted by cortical contractions (Thery et al, 2006; reviewed in Cai and Sheetz, 2009).
The mechanosensitive nature of integrin-based cell–matrix adhesion sites allows for controlled adjustment of actomyosin assembly in response to local changes in stress. As already mentioned above, the formation of both peripheral FA sites and associated actomyosin cables is promoted by tension (Hotulainen and Lappalainen, 2006). Moreover, the rate of actomyosin assembly at the FAs is proportional to the local stress (Rossier et al, 2010). RhoA has critical functions in the stress-dependent remodelling of the actomyosin cytoskeleton at integrin-based adhesion sites (Hotulainen and Lappalainen, 2006; Rossier et al, 2010). A general function of RhoA in actomyosin cytoskeletal rearrangements was shown by the observations that the RhoA effector Diaphanous regulates actomyosin assembly at FAs (Hotulainen and Lappalainen, 2006), and that ROCK-dependent myosin II activity modulates actin cable treadmilling, stabilization and repair (Rossier et al, 2010). Furthermore, inhibition of ROCK activity leads to the loss of cytosolic coherence (Cai et al, 2010; Rossier et al, 2010) and the concomitant disappearance of mature FAs. (Totsukawa et al, 2000, 2004). RhoA is also activated by stress and RhoA activity is required for stress-induced reorientation of the FA-associated actomyosin cables, suggesting that it mediates the effect of tension at FAs on rearranging the actomyosin cytoskeleton (Goldyn et al, 2009).
Much less is known about the function of cadherin-based cell–cell adhesion in controlling cell integrity. However, recent studies in vivo have shown that cellular rearrangements in epithelial tissues are driven by apical-actomyosin networks that are contracting centripetally (Martin et al, 2009), and that components of the adherens junctions are critical for maintaining epithelial integrity during these contractions (Martin et al, 2010). It is, therefore, conceivable that cadherin-based adherens junctions function as mechanosensors during epithelial morphogenesis, ensuring that epithelial tissue remains intact during actomyosin network contractions.
Taken together, RhoA-mediated stress-induced rearrangements of the actomyosin cytoskeleton at different adhesion sites allow cells to integrate extrinsic and intrinsic mechanical stimuli while maintaining cellular integrity.
Adhesion signalling in morphogenesis
During embryonic development, cells assemble into tissues and undergo large-scale collective movements. The mechanosensory properties of cell–cell and cell–matrix adhesion have a central function in these processes. In this section, we summarize current knowledge about how spatially and temporally regulated adhesion signalling controls epithelial morphogenesis and collective cell migration, and how mechanosensation functions therein.
Adhesive crosstalk in epithelial polarization
The assembly of cells in epithelial sheets is essential for compartmentalization of the body. Specialized epithelial tissues have barrier function, enable polarized transport and secretion and control tissue architecture. The functionality of an epithelium depends on the polarization of its constituent cells along their apicobasal axis. Apicobasal cell polarization becomes apparent by the segregation of the plasma membrane into non-intermixing apical and basolateral compartments (Figure 3A). The basolateral compartment is involved in adhesive interactions with surrounding cells and the ECM. The apical compartment is a non-adhesive compartment facing an ‘outside' luminal space or equivalent structures. It can also form a primary cilium and controls secretion in secretory epithelia. The establishment of an apical–basolateral cell polarity is regulated by several molecular complexes, called polarity complexes, that become localized to the respective membrane compartments (reviewed in Bryant and Mostov, 2008; Nelson, 2009). Ectopic localization of polarity complexes disrupts apical–basolateral cell polarization and interferes with epithelia formation and function (reviewed in Bryant and Mostov, 2008; Nelson, 2009).
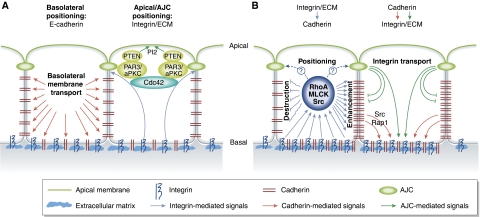
Figure 3.
Establishment of epithelial cell polarity: crosstalk between cell–cell and cell–matrix adhesion. (A) Adhesion signals control epithelial polarization. Engagement of cadherins in a cell–cell contact triggers the transport of basolateral markers to the adherent membranes. Integrin-mediated adhesion of basal surface to the ECM is essential for apical localization of PTEN, PI2 and PAR complex and activation of Cdc42. Cdc42 activation in turn is responsible for the assembly of apical junction complexes (AJC) that separate the apical and basolateral domains. (B) Crosstalk between integrins and cadherins controls epithelial integrity. Integrin-mediated regulation of RhoA, Src and MLCK activities can lead to both an enhancement and destruction of adherens junctions. The specific effect depends on the ECM composition and the cell type. Cadherins also promote integrin-ECM adhesion through Src/Rap1 signalling and targeting of integrins to the basal membrane.
Cadherin-mediated cell–cell adhesion has an important function in the assembly and polarization of epithelial tissues (Larue et al, 1994; Wang et al, 2004; Halbleib and Nelson, 2006; Capaldo and Macara, 2007). The establishment of cadherin-mediated cell–cell contacts triggers the polarized delivery of basolateral membrane proteins (Nejsum and Nelson, 2007; Shaw et al, 2007). Moreover, cell shape changes associated with the establishment of cadherin-based adherens junctions polarize the cell interior, resulting in the localization of the Golgi complex and Microtubule Organizing Center towards the non-adhesive apical surface (Dupin et al, 2009).
Although the basolateral compartment is generated as a direct consequence of cadherin-mediated cell–cell contact formation, positioning of apical surface is also controlled by external cues. In cultured epithelial cell sheets, the apical compartment is induced by both contact with the medium and the adhesion of the basal cell surface to the ECM (O'Brien et al, 2002; Zegers et al, 2003; Bryant and Mostov, 2008; Sacharidou et al, 2010; Zovein et al, 2010). How ECM adhesion controls apical compartment formation is not yet fully understood, but adhesion of the basal compartment to the ECM has been suggested to localize phosphatase and tensin homologue (PTEN) to apical junctions, which triggers subsequent apical enrichment of phosphatidylinositol (3,4)-bisphosphate (PI2) (O'Brien et al, 2001; Yu et al, 2005), Cdc42 and the partition-defective (PAR) complex (Martin-Belmonte et al, 2007). Cdc42 signalling controls apical lumen positioning by numerous downstream effectors (Koh et al, 2009). Localized Cdc42 signalling is dispensable for apicobasal polarization in individual cells, but essential for apical lumen formation in multi-cellular aggregates (Jaffe et al, 2008). Cdc42 functions in apical lumen formation by controlling the assembly of apical junctions complexes (AJC) at the boundary between basolateral and apical domains in the neighbouring cells (Wu et al, 2007).
AJC are specialized adhesion structures that separate apical and basolateral compartments in epithelial cells. AJC consist of several domains, and recruit a number of adhesion molecules, including classical cadherins, and intracellular adaptors (Gibson and Perrimon, 2003; Labouesse, 2006). AJC localize the PAR3-atypical protein kinase C (aPKC) complex at the apical membrane, which in turn recruits PTEN and triggers apical PIP2 accumulation (Martin-Belmonte et al, 2007). Recruitment of aPKC causes phosphorylation of LGL and dissociation of basal markers from the apical membrane (Betschinger et al, 2003; Plant et al, 2003; Hutterer et al, 2004). These findings suggest that AJC have central functions in apical–basolateral cell polarization, although it is not yet fully clear how the AJC interact with the polarity complexes.
The mechanosensory crosstalk between cadherin and integrin-mediated adhesion is critical for the assembly and stability of epithelial tissues (Figure 3B). Integrin-mediated cell–matrix adhesion on collagen-coated substrates promotes epithelial-to-mesenchymal transtition (EMT) and scattering of MDCK cells, whereas adhesion to other ECM proteins, such as fibronectin, vitronectin and laminin, stabilizes the epithelial state (Clark, 1994; Sander et al, 1998). Adhesion to fibronectin can also promote cell scattering (de Rooij et al, 2005). The precise effect of ECM adhesion on the integrity of epithelial cell layers appears to depend on composition of integrin dimers expressed and their specific signalling capabilities (Gimond et al, 1999; de Rooij et al, 2005; Zhou and Kramer, 2005). The cell scattering function of integrins was associated with increased activity of RhoA and MLCK, which in turn triggers the mechanical disruption of cadherin-based adhesions (Avizienyte et al, 2002; Avizienyte and Frame, 2005; de Rooij et al, 2005). Conversely, integrin signalling has been suggested to promote cell cohesion both in vitro and in vivo (Marsden and DeSimone, 2003; Yano et al, 2004) through downregulation of peripheral Rac activity and inhibiting membrane ruffling (Yano et al, 2004). Integrin-mediated adhesion was also recently shown to stimulate cadherin-mediated cell–cell cohesion by modulating RhoA/ROCK, MLCK and Src signalling (Martinez-Rico et al, 2010), consistent with recent findings that show an enhancement of cadherin-mediated adhesion under tension (see also section ‘Cell–cell adhesion'). These findings suggest that adherens junctions can differently respond to cytoskeletal contractions induced by integrin-ECM adhesion through Rho-family GTPases and Src/MLCK. Moderate contraction appears to promote adherens junction stability, probably by enhancing adhesion strength through a mechanosensory feedback loop (see also section ‘Cell–cell adhesion'). In contrast, stronger contractile stresses might disrupt adherens junctions, thereby promoting EMT. The spatiotemporal regulation of integrin-ECM adhesion signalling might also be important for its function in epithelial integrity, as adherens junctions respond differently to Rho-family GTPase and Src/MLCK activity during expansion and maturation (see also section ‘Cell–cell adhesion signalling').
Cadherins can also regulate integrin-ECM signalling. Endosomal E-cadherin signalling through Src and Rap1 promotes the adhesive function of integrins (Balzac et al, 2005). In contrast, E-cadherin at apical junctions locally represses integrin-mediated adhesion, thereby contributing to the polarized basal assembly of hemidesmosomes in the zebrafish skin (Sonawane et al, 2009). C-cadherin-dependent cell cohesion provides tissue tension that is essential for the polarized fibronectin assembly in gastrulating Xenopus embryos (Dzamba et al, 2009), suggesting a mechanosensory signalling link between cadherins and integrins.
Taken together, the crosstalk between cell–cell and cell–matrix adhesion has an important function in determining epithelial cell polarity. Cadherin-mediated cell–cell adhesion is directly involved in the generation of the basolateral compartment, whereas specialized cell–cell adhesion structures control the positioning of the apical domain. Integrin-mediated cell–matrix adhesion controls apical–basolateral cell polarity by modulating cell cohesion and positioning of AJC. The interaction between integrins and cadherins likely involves a mechanosensory signalling link through the spatial regulation of cytoskeletal contractility. This mechanosensory link between intergrins and cadherins might also be involved in the regulation of epithelial morphogenesis (see also section ‘Adhesion in epithelial remodelling').
Adhesion in epithelial remodelling
During morphogenesis, epithelial cell layers undergo dynamic shape changes. These changes involve cellular rearrangements within the plane of the epithelial sheet and cell invaginations. Planar cell rearrangements can also be a part of epithelial invaginations (Nishimura and Takeichi, 2008). In order to maintain epithelial integrity while undergoing extensive cellular rearrangements, epithelial cells must retain and dynamically remodel adherens junctions that connect them with neighbouring cells. The molecular and cellular mechanisms underlying adherens junction stability and dynamics have been extensively reviewed elsewhere (Cavey and Lecuit, 2009; Nishimura and Takeichi, 2009). Therefore, we will only briefly summarize the main principles driving planar cell rearrangements and invaginations in epithelia, and then discuss how spatial and temporal regulation of adhesion signalling contributes to these processes.
Planar cell rearrangement has been most thoroughly studied in epithelial tissues of Drosophila embryos. There, epithelial cells undergo planar rearrangements by shortening their contacts with some neighbours while extending them with others. These cell–cell contact changes are driven by polarized contraction of single or multiple cell junctions, frequently leading to the assembly of transient cell patterns, such as rosettes (Bertet et al, 2004; Blankenship et al, 2006; Zallen and Blankenship, 2008) (Figure 4A). Polarized cell junction contraction is achieved by actomyosin filaments localized at the apical junctions (Bertet et al, 2004; Fernandez-Gonzalez et al, 2009). Assembly of junctional actomyosin filaments is regulated by local tension at the respective junction, suggesting that a mechanosensory feedback is involved in junction remodelling (Fernandez-Gonzalez et al, 2009). Different components of the apical junctions, such as Shroom 3, Moesin and Bitesize, also modulate apical actin assembly, which in turn stabilizes apical E-cadherin (Pilot et al, 2006; Cavey et al, 2008; Nishimura and Takeichi, 2008). As tension can enhance cadherin-mediated cell–cell adhesion (Ladoux et al, 2010), it is conceivable that tension at apical junctions could lead to increased recruitment of junctional components, such as Moesin and Bitesize, which in turn would recruit more actin and myosin, leading to further enhancement of junctional tension and thus building up a positive mechansensory feedback loop. It has also been suggested that apical E-cadherin junctions contain stable microdomains (spot junctions), which are connected with immobilized actin patches in an α-catenin-independent manner (Cavey et al, 2008). At the same time, α-catenin links E-cadherin to a dynamic actin network, which determines the lateral mobility, or ‘sliding' of apical spot junctions.
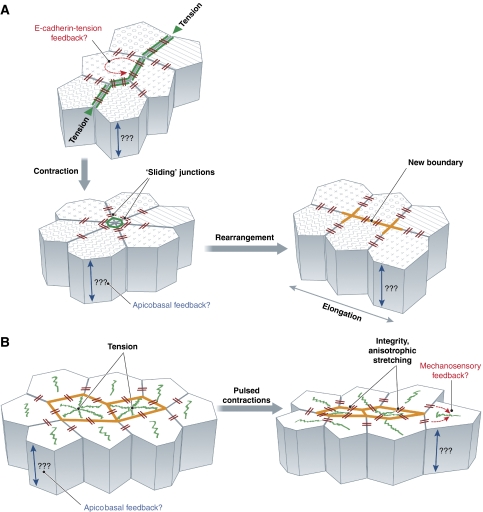
Figure 4.
Remodelling of epithelial sheets in Drosophila embryos. (A) Planar cell rearrangement is dependent on actomyosin assembly at the apical junctions. Polarized assembly of junctional actomyosin filaments is driven by tension. Actomyosin-rich junction(s) contract, which in some cases can lead to a transient ‘rosette'-like cell assembly that subsequently dissolves through the polarized expansion of new boundaries. Polarized junction contraction and expansion drives germ band elongation. During planar cell rearrangements, E-cadherin accumulated in ‘spot junctions', which are connected to stable actin patches and linked to each other through a dynamic actin network ‘slide' along the boundaries. (B) Epithelial invagination is driven by pulsed contractions of a non-junctional actomyosin network. During epithelial invagination, coalescing actomyosin networks form at the apex of the invaginating cells. These networks are connected with E-cadherin-based spot junctions. Stochastic centripetal contractions of the apical actomyosin networks, followed by the stabilization of the contracted states, drives apical constriction. E-cadherin-based spot junctions are essential for maintaining tissue integrity and stabilizing anisotropic cell shape changes during the contraction pulses.
Epithelial cell sheet bending and invagination are primarily driven by apical cell constrictions, which were originally proposed to be achieved by the continuous purse string such as contraction of apical junctional actomyosin belt (Ettensohn, 1985). However, recent studies showed that apical constrictions of epithelial cells during ventral furrow invagination and dorsal closure in Drosophila are triggered by stochastic pulsed contractions of individual cells (Martin et al, 2009; Solon et al, 2009). These contractions are driven by the coalescence of a non-junctional actomysosin web at the cell apex (Figure 4B), and each contraction cycle is followed by a partial stabilization of the contracted cell shape. Generation of contraction pulses in the ventral furrow cells seems to be not directly dependent on E-cadherin-mediated adhesion (Martin et al, 2010). However, E-cadherin-based spot junctions are needed to connect the actomyosin network between adjacent cells and thereby building up a supracellular actomysion network, which maintains epithelial integrity and transmits anisotropic tension across the tissue (Martin et al, 2010).
During epithelial remodelling, the adherens junctions are thought to primarily act as mechanical anchors that provide adhesion between cells and function as ratchets that stabilize actomyosin-driven cell shape changes. However, the engagement of cadherins is known to trigger downstream signalling (see also section ‘Cell–cell adhesion signalling'), and thus rearrangement of cadherin-based adhesion sites during epithelial movements is likely to trigger signals that also function in junctional remodelling. It is, therefore, conceivable that adherens junctions not only have mechanical and stabilizing functions in epithelia, but also actively generate and modulate forces that drive cellular rearrangements underlying epithelial morphogenesis. This assumption is supported by recent studies showing that Wiskott–Aldrich syndrome protein (WASP), a downstream effector of the Rho-family GTPases Rac and Cdc42, is involved in modulation of apical cell constriction during Drosophila germband extension (Bertet et al, 2009), and that Cdc42 functions as an important regulator of E-cadherin endocytosis during planar epithelial remodelling in the Drosophila notum (Georgiou et al, 2008). As Rac and Cdc42 signalling is also modulated by adherens junctions themselves, there are likely to be feedback mechanisms between junctional remodelling and downstream signalling influencing this process. Future studies, similar to those carried out in cell culture (Yamada and Nelson, 2007; Perez et al, 2008), will be needed to elucidate the nature, function and spatial propagation of adhesion-mediated signals during junctional remodelling in vivo.
Although junctional remodelling of the apical surface is thought to represent an important process by which epithelia change shape, activity at the basolateral side of epithelial cells is also likely to contribute. It is conceivable that the interaction between apical and basolateral cell compartments taking place in the establishment of epithelial cell polarity (see also section ‘Adhesive crosstalk in epithelial polarization'; Figure 3) is also involved in epithelial remodelling. The observation that the basal lamina is dynamically remodelled during Xenopus neurulation, and that this remodelling is critical for neural tube formation (Davidson et al, 2004), suggest that the interaction of epithelial cells at their basal side with the ECM has an important function in epithelial morphogenesis. Analysis of epithelial remodelling in three dimensions will be needed to reveal how apical and basolateral compartments of epithelial cells interact in controlling this process.
Taken together, remodelling of epithelial sheets is driven by actomyosin contractility and seems to involve two major mechanisms—actomyosin assembly at the cell apex driving pulsed apical cell constrictions and apical adherens junctions functioning as ratchets that stabilize remodelled junctions. Moreover, the assembly of supracellular junctional actomyosin cables is needed to coordinate junctional remodelling and planar cell rearrangements within the tissue. Stress-mediated regulation of junctional remodelling appears to be critical for polarized rearrangements of epithelial tissues; however, the molecular and cellular mechanisms underlying this mechanosensory function still remain to be elucidated.
Adhesion in collective cell migration
During cell migration, external stimuli need to be translated into changes in cell shape and motility. Migration is a cyclic process during which a cell extends its front and retracts its trailing end, requiring extensive reorganization of the cytoskeleton (reviewed in Vicente-Manzanares et al, 2005; Webb et al, 2005). During collective cell migration, this cyclic migration process needs to be coupled between neighbouring cells. The spatiotemporal regulation of cell adhesion and the mechanosensitivity of individual cells were proposed to have an important function for the efficient communication between cells during collective migration.
For directed migration, cells need to sense and interpret chemical and/or mechanical cues in their environment. The way cells that undergo collective migration sense these cues depends on the coupling between the cells in the group. In tightly associated cells, such as epithelial cell layers, only the leading edge cells sense the directional cues. In the zebrafish lateral line primordium, for example, a small subset of cells at the leading edge of the primordium expressing the chemokine receptor CXCR4 is able to direct the migration of large number of CXCR4-negative cells, which adhere to the leading edge cells, towards a source of the chemokine SDF1 (Haas and Gilmour, 2006). Similarly, during Drosophila border cell migration, activation of Rac in single cells is sufficient to direct migration of the whole group in the absence of other guidance clues (Wang et al, 2010). In cultured epithelial cells sheets, the specific potential of leading cells to direct the migration of cells behind is determined by the recruitment of the tight junction protein occludin to the front of these cells (Du et al, 2010). Occludin is thought to function in leading edge cells by recruiting the aPKC/PAR3 complex and thereby stimulating directed cell protrusion formation. In contrast, occludin-containing junctions are disassembled at the leading edge of the lateral line primordium (Lecaudey et al, 2008; Hava et al, 2009), indicating that other adhesive structures might support directed protrusion formation in these cells. It is not yet clear, whether collectively migrating cells that are more loosely associated, such as vertebrate mesoderm cells undergoing convergence and extension movements (Figure 5), undergo directed cell migration autonomously, or whether a small subset of cells directs their migration.
Figure 5.
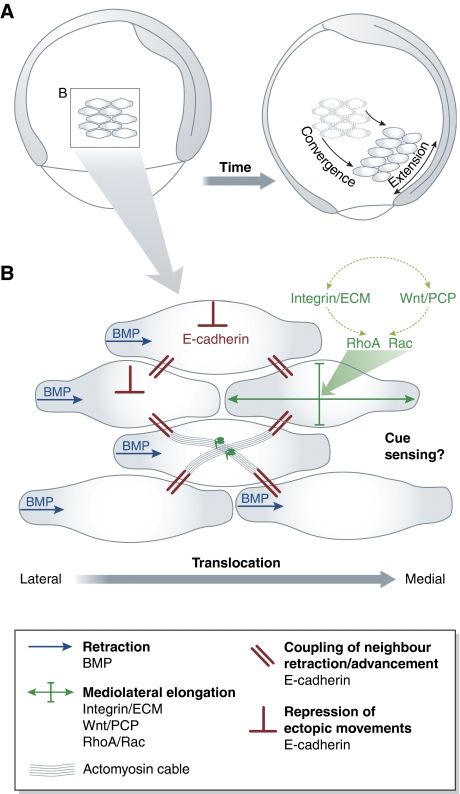
Collective migration of mesoderm progenitor cells in zebrafish gastrulation. (A) Convergence and extension movements of lateral mesoderm cells during zebrafish gastrulation. Lateral view of zebrafish embryo at onset and end of gastrulation. A group of mesoderm cells is highlighted. Cells migrate towards the dorsal side and undergo mediolateral cell intercalations. Both cell migration and intercalations contribute to convergence and extension of the forming body axis. (B) Collective migration of lateral mesoderm cells. Cell elongation is driven by integrin-ECM adhesion and Wnt/PCP signals, which together modulate Rho-family GTPase activity. BMP signalling polarizes protrusion formation in individual cells. E-cadherin-mediated cell cohesion mechanically couples and thereby aligns the movements of neighbouring cells. In addition, E-cadherin restricts movement of cells over each other.
Cells use the information provided by external cues to modulate their adhesion required for directed migration. In many cases, such modulated adhesion is needed to initiate and/or stabilize front-to-rear cell polarization. During the migration of zebrafish prechordal plate progenitor cells, for example, the extension of frontal filopodia and lamellipodia-like protrusions depends on E-cadherin expression in these cells (Montero et al, 2005). During Xenopus and zebrafish gastrulation, lateral mesoderm progenitor cells acquire a bipolar shape, which is required for these cells to undergo mediolateral cell intercalations and, as a result of this, convergent extension movements (reviewed in Green and Davidson, 2007). Both integrin-fibronectin adhesion and Wnt/planar cell polarity (Wnt/PCP) signalling are essential for mesoderm progenitors to undergo mediolateral cell intercalations (Topczewski et al, 2001; Jessen et al, 2002; Marlow et al, 2002; Kilian et al, 2003) and components of both pathways were shown to functionally interact (Marsden and DeSimone, 2001; Goto et al, 2005; Dzamba et al, 2009). Interfering with the function of Rho-family GTPases RhoA and Rac produces phenotypes in mesoderm morphogenesis reminiscent of those resulting from Wnt/PCP and integrin-fibronectin inactivation, and some aspects of Wnt/PCP and integrin-fibronectin loss-of-function phenotypes can be rescued by overexpression of these GTPases (Tahinci and Symes, 2003; Zhu et al, 2006). This suggests that Wnt/PCP and integrin-fibronectin function in mesoderm polarization and intercalation by modulating Rho-family GTPase signalling, although very little is known about the spatiotemporal activity of these GTPases in mesoderm progenitors. It also remains to be elucidated whether cell polarization in vivo involves force-mediated segregation of adhesion structures and associated signals as it occurs during in vitro cell migration (see also sections ‘Cell–matrix adhesion' and ‘Cell–matrix adhesion signalling').
Retraction of the posterior edge is essential for single cell migration and front-to-rear cell polarization (Kaverina et al, 2000). Similarly, during zebrafish gastrulation, destabilization and/or retraction of cellular protrusions contributes to cell polarization and directed migration. A ventral-to-dorsal gradient of bone morphogenetic protein (BMP) signalling within the zebrafish gastrula is thought to negatively regulate E-cadherin-mediated adhesion and consequently to destabilize ventrally oriented mesoderm cell protrusions. Consequently, mesoderm progenitors form more stable protrusions pointing dorsally, which is a prerequisite for their dorsal-directed convergence movements (von der Hardt et al, 2007).
Efficient collective cell migration requires not only polarization and directed migration of single cells, but also efficient coordination of the migratory activity between the participating cells. This coordination is achieved by mechanically coupling individual cells into a supracellular entity, and cadherin adhesion molecules are prime candidates to control this process. Studies in zebrafish suggested that cadherin-mediated cell adhesion is essential for collective mesendoderm movements, but dispensable for individual mesendoderm migration (Arboleda-Estudillo et al, 2010). Similarly, E-cadherin is required for the collective migration of Drosophila border cells by promoting their epithelial organization and frontal protrusion formation (Niewiadomska et al, 1999; Melani et al, 2008). E-cadherin is also involved in collective cell movements during mammary duct formation (Ewald et al, 2008), trachea development (Shaye et al, 2008) and cancer invasion (reviewed in Friedl and Gilmour, 2009). During collective movements, adhesion between individual cells needs to be dynamically regulated and precisely coordinated in order to allow cellular rearrangements within the group of co-migrating cells while maintaining its integrity. Wnt/PCP signalling has been suggested to regulate cell–cell adhesion dynamics in collective mesendoderm cell migration during zebrafish gastrulation by controlling E-cadherin intracellular trafficking (Ulrich et al, 2005). The formation of supracellular actomyosin cables, potentially associated with adherens junctions, was also implicated in the coordination of mediolateral cell polarization and intercalation underlying convergence and extension movements during Xenopus gastrulation (Skoglund et al, 2008).
Specific signalling proteins associated with cell–cell junctions are involved in regulating the function of E-cadherin during collective cell migration. For example, the LIM domain protein zyxin promotes collective cell migration by connecting the actin cytoskeleton between neighbouring cells at adherens junctions, thereby increasing junction stability (Sperry et al, 2009). Similarly, collective migration of Drosopila border cells depends on Jun N-terminal kinase signalling, which promotes border cell cohesion (Wang et al, 2010). These findings suggest that mechanical coupling of collectively migrating cells depends on downstream signals from cadherin-mediated cell–cell adhesions. It remains to be elucidated how such signals are spatiotemporally propagated between cells during collective migration.
In addition to providing a mechanical linkage between individual cells during collective migration, cell–cell and cell–matrix adhesion were also suggested to restrict the movement of co-migrating cells over each other (out-of-plane movements). In E-cadherin-depleted zebrafish embryos, mesendoderm progenitors crawl over each other instead of maintaining their directed and highly aligned movements during gastrulation (Arboleda-Estudillo et al, 2010). Similarly, integrin-mediated cell–matrix adhesion and Wnt/PCP signalling have been hypothesized to restrict out-of-plane cell movements during convergent extension in Xenopus gastrulation (Green and Davidson, 2007). One possible mechanism by which cadherins and Wnt/PCP signalling restrict out-of-plane cell movements is by polarizing cell protrusion formation through their effect on integrin-ECM adhesion (Goto et al, 2005; Davidson et al, 2006; Dzamba et al, 2009). Alternatively, out-of-plane cell movements might be restricted by contact inhibiton among co-migrating cells. Consistent with this assumption, Wnt/PCP has been shown to control contact inhibition between co-migrating neural crest cells in Xenopus embryos by regulating RhoA activity at the site of cell–cell contacts (Carmona-Fontaine et al, 2008). It appears, therefore, that Wnt/PCP interacts with cell–cell and cell–matrix adhesion in restricting out-of-plane movements of collectively migrating cells, although the precise mechanisms mediating this activity remain to be elucidated.
Taken together, collective cell migration requires both cell–cell and cell–matrix adhesion. Integrins and cadherins function in collective migration by both regulating individual cell protrusive activity and motility, and establishing cell–cell contact between neighbouring cells that both mechanically link these cells and restrict their movements relative to each other. It remains to be investigated, how adhesion-mediated signals are transmitted within and between cells to promote directed and aligned cell movements during collective migration.
Outlook
The mechanosensory nature of adhesion receptors has an important function in the regulation of cell adhesion by balancing adhesion strength with extracellular signals and intracellular tension. Studies on cultured cells showed that mechanosensory remodelling of cell–cell and cell–matrix adhesion sites is achieved by recruiting heterogenous components in response to mechanical stimuli. The emergent picture from these studies is that forces at adhesion sites determine their molecular composition, which again modulates their signalling activity, and that the specific signalling activity feeds back on the mechanosensory properties of these adhesion sites. Recent advances in quantitative imaging techniques, such as development of new FRET biosensors and photactivatable proteins, will facilitate the analysis of how signals originating from specific adhesion sites are propagated within cells in order to integrate the cellular response to mechanical stimuli.
The mechanosensory function of cell–cell and cell–matrix adhesion sites is also likely to have an important function in tissue morphogenesis, although only very little is known about the actual function of adhesion-mediated mechanosensing in vivo. The majority of studies published on in vivo systems consider integrin- and cadherin-mediated adhesion sites as homogenous structures with uniform function. However, recent work suggests that similar to the situation in vitro, adhesion structures in vivo are highly heterogenous, and signals emanating from these adhesion sites are modulated by changes in their molecular composition in response to mechanical stimuli. In order to understand the function of adhesion in tissue morphogenesis, it will thus be essential to consider cadherin- and integrin-based adhesions as highly heterogenous structures that dynamically change their adhesive and signalling activity in a context-dependent manner. Future studies will have to unravel how the molecular composition of adhesion sites determines their signalling activity and mechansensory properties in vivo, and how local adhesion signalling is propagated between cells in order to control tissue morphogenesis.
Footnotes
The authors declare that they have no conflict of interest.
References
- Abe K, Takeichi M (2008) EPLIN mediates linkage of the cadherin catenin complex to F-actin and stabilizes the circumferential actin belt. Proc Natl Acad Sci USA 105: 13–19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anastasiadis PZ, Moon SY, Thoreson MA, Mariner DJ, Crawford HC, Zheng Y, Reynolds AB (2000) Inhibition of RhoA by p120 catenin. Nat Cell Biol 2: 637–644 [DOI] [PubMed] [Google Scholar]
- Anthis NJ, Wegener KL, Ye F, Kim C, Goult BT, Lowe ED, Vakonakis I, Bate N, Critchley DR, Ginsberg MH, Campbell ID (2009) The structure of an integrin/talin complex reveals the basis of inside-out signal transduction. EMBO J 28: 3623–3632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arboleda-Estudillo Y, Krieg M, Stuhmer J, Licata NA, Muller DJ, Heisenberg CP (2010) Movement directionality in collective migration of germ layer progenitors. Curr Biol 20: 161–169 [DOI] [PubMed] [Google Scholar]
- Askari JA, Buckley PA, Mould AP, Humphries MJ (2009) Linking integrin conformation to function. J Cell Sci 122: 165–170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Astrof NS, Salas A, Shimaoka M, Chen J, Springer TA (2006) Importance of force linkage in mechanochemistry of adhesion receptors. Biochemistry 45: 15020–15028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avizienyte E, Frame MC (2005) Src and FAK signalling controls adhesion fate and the epithelial-to-mesenchymal transition. Curr Opin Cell Biol 17: 542–547 [DOI] [PubMed] [Google Scholar]
- Avizienyte E, Wyke AW, Jones RJ, McLean GW, Westhoff MA, Brunton VG, Frame MC (2002) Src-induced de-regulation of E-cadherin in colon cancer cells requires integrin signalling. Nat Cell Biol 4: 632–638 [DOI] [PubMed] [Google Scholar]
- Ballestrem C, Hinz B, Imhof BA, Wehrle-Haller B (2001) Marching at the front and dragging behind: differential alphaVbeta3-integrin turnover regulates focal adhesion behavior. J Cell Biol 155: 1319–1332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balzac F, Avolio M, Degani S, Kaverina I, Torti M, Silengo L, Small JV, Retta SF (2005) E-cadherin endocytosis regulates the activity of Rap1: a traffic light GTPase at the crossroads between cadherin and integrin function. J Cell Sci 118: 4765–4783 [DOI] [PubMed] [Google Scholar]
- Bershadsky AD, Balaban NQ, Geiger B (2003) Adhesion-dependent cell mechanosensitivity. Annu Rev Cell Dev Biol 19: 677–695 [DOI] [PubMed] [Google Scholar]
- Bertet C, Rauzi M, Lecuit T (2009) Repression of Wasp by JAK/STAT signalling inhibits medial actomyosin network assembly and apical cell constriction in intercalating epithelial cells. Development 136: 4199–4212 [DOI] [PubMed] [Google Scholar]
- Bertet C, Sulak L, Lecuit T (2004) Myosin-dependent junction remodelling controls planar cell intercalation and axis elongation. Nature 429: 667–671 [DOI] [PubMed] [Google Scholar]
- Betschinger J, Mechtler K, Knoblich JA (2003) The Par complex directs asymmetric cell division by phosphorylating the cytoskeletal protein Lgl. Nature 422: 326–330 [DOI] [PubMed] [Google Scholar]
- Betson M, Lozano E, Zhang J, Braga VM (2002) Rac activation upon cell-cell contact formation is dependent on signaling from the epidermal growth factor receptor. J Biol Chem 277: 36962–36969 [DOI] [PubMed] [Google Scholar]
- Blankenship JT, Backovic ST, Sanny JS, Weitz O, Zallen JA (2006) Multicellular rosette formation links planar cell polarity to tissue morphogenesis. Dev Cell 11: 459–470 [DOI] [PubMed] [Google Scholar]
- Bryant DM, Mostov KE (2008) From cells to organs: building polarized tissue. Nat Rev Mol Cell Biol 9: 887–901 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Y, Rossier O, Gauthier NC, Biais N, Fardin MA, Zhang X, Miller LW, Ladoux B, Cornish VW, Sheetz MP (2010) Cytoskeletal coherence requires myosin-IIA contractility. J Cell Sci 123: 413–423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Y, Sheetz MP (2009) Force propagation across cells: mechanical coherence of dynamic cytoskeletons. Curr Opin Cell Biol 21: 47–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capaldo CT, Macara IG (2007) Depletion of E-cadherin disrupts establishment but not maintenance of cell junctions in Madin-Darby canine kidney epithelial cells. Mol Biol Cell 18: 189–200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carmona-Fontaine C, Matthews HK, Kuriyama S, Moreno M, Dunn GA, Parsons M, Stern CD, Mayor R (2008) Contact inhibition of locomotion in vivo controls neural crest directional migration. Nature 456: 957–961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavey M, Lecuit T (2009) Molecular bases of cell-cell junctions stability and dynamics. Cold Spring Harb Perspect Biol 1: a002998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavey M, Rauzi M, Lenne PF, Lecuit T (2008) A two-tiered mechanism for stabilization and immobilization of E-cadherin. Nature 453: 751–756 [DOI] [PubMed] [Google Scholar]
- Chen H, Cohen DM, Choudhury DM, Kioka N, Craig SW (2005) Spatial distribution and functional significance of activated vinculin in living cells. J Cell Biol 169: 459–470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen YT, Stewart DB, Nelson WJ (1999) Coupling assembly of the E-cadherin/beta-catenin complex to efficient endoplasmic reticulum exit and basal-lateral membrane targeting of E-cadherin in polarized MDCK cells. J Cell Biol 144: 687–699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chrzanowska-Wodnicka M, Burridge K (1996) Rho-stimulated contractility drives the formation of stress fibers and focal adhesions. J Cell Biol 133: 1403–1415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark P (1994) Modulation of scatter factor/hepatocyte growth factor activity by cell-substratum adhesion. J Cell Sci 107 (Pt 5): 1265–1275 [DOI] [PubMed] [Google Scholar]
- Cluzel C, Saltel F, Lussi J, Paulhe F, Imhof BA, Wehrle-Haller B (2005) The mechanisms and dynamics of (alpha)v(beta)3 integrin clustering in living cells. J Cell Biol 171: 383–392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen M, Joester D, Geiger B, Addadi L (2004) Spatial and temporal sequence of events in cell adhesion: from molecular recognition to focal adhesion assembly. Chembiochem 5: 1393–1399 [DOI] [PubMed] [Google Scholar]
- Cukierman E, Pankov R, Stevens DR, Yamada KM (2001) Taking cell-matrix adhesions to the third dimension. Science 294: 1708–1712 [DOI] [PubMed] [Google Scholar]
- Dai J, Ting-Beall HP, Hochmuth RM, Sheetz MP, Titus MA (1999) Myosin I contributes to the generation of resting cortical tension. Biophys J 77: 1168–1176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danen EH, Sonnenberg A (2003) Integrins in regulation of tissue development and function. J Pathol 200: 471–480 [DOI] [PubMed] [Google Scholar]
- Danen EH, Sonneveld P, Brakebusch C, Fassler R, Sonnenberg A (2002) The fibronectin-binding integrins alpha5beta1 and alphavbeta3 differentially modulate RhoA-GTP loading, organization of cell matrix adhesions, and fibronectin fibrillogenesis. J Cell Biol 159: 1071–1086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson LA, Keller R, DeSimone DW (2004) Assembly and remodeling of the fibrillar fibronectin extracellular matrix during gastrulation and neurulation in Xenopus laevis. Dev Dyn 231: 888–895 [DOI] [PubMed] [Google Scholar]
- Davidson LA, Marsden M, Keller R, Desimone DW (2006) Integrin alpha5beta1 and fibronectin regulate polarized cell protrusions required for Xenopus convergence and extension. Curr Biol 16: 833–844 [DOI] [PubMed] [Google Scholar]
- de Rooij J, Kerstens A, Danuser G, Schwartz MA, Waterman-Storer CM (2005) Integrin-dependent actomyosin contraction regulates epithelial cell scattering. J Cell Biol 171: 153–164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- del Pozo MA, Price LS, Alderson NB, Ren XD, Schwartz MA (2000) Adhesion to the extracellular matrix regulates the coupling of the small GTPase Rac to its effector PAK. EMBO J 19: 2008–2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- del Rio A, Perez-Jimenez R, Liu R, Roca-Cusachs P, Fernandez JM, Sheetz MP (2009) Stretching single talin rod molecules activates vinculin binding. Science 323: 638–641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Digman MA, Wiseman PW, Choi C, Horwitz AR, Gratton E (2009) Stoichiometry of molecular complexes at adhesions in living cells. Proc Natl Acad Sci USA 106: 2170–2175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drees F, Pokutta S, Yamada S, Nelson WJ, Weis WI (2005) Alpha-catenin is a molecular switch that binds E-cadherin-beta-catenin and regulates actin-filament assembly. Cell 123: 903–915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du D, Xu F, Yu L, Zhang C, Lu X, Yuan H, Huang Q, Zhang F, Bao H, Jia L, Wu X, Zhu X, Zhang X, Zhang Z, Chen Z (2010) The tight junction protein, occludin, regulates the directional migration of epithelial cells. Dev Cell 18: 52–63 [DOI] [PubMed] [Google Scholar]
- Dubash AD, Wennerberg K, Garcia-Mata R, Menold MM, Arthur WT, Burridge K (2007) A novel role for Lsc/p115 RhoGEF and LARG in regulating RhoA activity downstream of adhesion to fibronectin. J Cell Sci 120: 3989–3998 [DOI] [PubMed] [Google Scholar]
- Dupin I, Camand E, Etienne-Manneville S (2009) Classical cadherins control nucleus and centrosome position and cell polarity. J Cell Biol 185: 779–786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dzamba BJ, Jakab KR, Marsden M, Schwartz MA, DeSimone DW (2009) Cadherin adhesion, tissue tension, and noncanonical Wnt signaling regulate fibronectin matrix organization. Dev Cell 16: 421–432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ettensohn CA (1985) Mechanisms of epithelial invagination. Q Rev Biol 60: 289–307 [DOI] [PubMed] [Google Scholar]
- Ewald AJ, Brenot A, Duong M, Chan BS, Werb Z (2008) Collective epithelial migration and cell rearrangements drive mammary branching morphogenesis. Dev Cell 14: 570–581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez-Gonzalez R, Simoes Sde M, Roper JC, Eaton S, Zallen JA (2009) Myosin II dynamics are regulated by tension in intercalating cells. Dev Cell 17: 736–743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fincham VJ, James M, Frame MC, Winder SJ (2000) Active ERK/MAP kinase is targeted to newly forming cell-matrix adhesions by integrin engagement and v-Src. EMBO J 19: 2911–2923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frelinger AL III, Cohen I, Plow EF, Smith MA, Roberts J, Lam SC, Ginsberg MH (1990) Selective inhibition of integrin function by antibodies specific for ligand-occupied receptor conformers. J Biol Chem 265: 6346–6352 [PubMed] [Google Scholar]
- Frey MT, Tsai IY, Russell TP, Hanks SK, Wang YL (2006) Cellular responses to substrate topography: role of myosin II and focal adhesion kinase. Biophys J 90: 3774–3782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedl P, Gilmour D (2009) Collective cell migration in morphogenesis, regeneration and cancer. Nat Rev Mol Cell Biol 10: 445–457 [DOI] [PubMed] [Google Scholar]
- Friedland JC, Lee MH, Boettiger D (2009) Mechanically activated integrin switch controls alpha5beta1 function. Science 323: 642–644 [DOI] [PubMed] [Google Scholar]
- Galbraith CG, Yamada KM, Sheetz MP (2002) The relationship between force and focal complex development. J Cell Biol 159: 695–705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geiger B, Spatz JP, Bershadsky AD (2009) Environmental sensing through focal adhesions. Nat Rev Mol Cell Biol 10: 21–33 [DOI] [PubMed] [Google Scholar]
- Georgiou M, Marinari E, Burden J, Baum B (2008) Cdc42, Par6, and aPKC regulate Arp2/3-mediated endocytosis to control local adherens junction stability. Curr Biol 18: 1631–1638 [DOI] [PubMed] [Google Scholar]
- Giannone G, Dubin-Thaler BJ, Rossier O, Cai Y, Chaga O, Jiang G, Beaver W, Dobereiner HG, Freund Y, Borisy G, Sheetz MP (2007) Lamellipodial actin mechanically links myosin activity with adhesion-site formation. Cell 128: 561–575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson MC, Perrimon N (2003) Apicobasal polarization: epithelial form and function. Curr Opin Cell Biol 15: 747–752 [DOI] [PubMed] [Google Scholar]
- Gimond C, van Der Flier A, van Delft S, Brakebusch C, Kuikman I, Collard JG, Fassler R, Sonnenberg A (1999) Induction of cell scattering by expression of beta1 integrins in beta1-deficient epithelial cells requires activation of members of the rho family of GTPases and downregulation of cadherin and catenin function. J Cell Biol 147: 1325–1340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldyn AM, Rioja BA, Spatz JP, Ballestrem C, Kemkemer R (2009) Force-induced cell polarisation is linked to RhoA-driven microtubule-independent focal-adhesion sliding. J Cell Sci 122: 3644–3651 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goto T, Davidson L, Asashima M, Keller R (2005) Planar cell polarity genes regulate polarized extracellular matrix deposition during frog gastrulation. Curr Biol 15: 787–793 [DOI] [PubMed] [Google Scholar]
- Green JB, Davidson LA (2007) Convergent extension and the hexahedral cell. Nat Cell Biol 9: 1010–1015 [DOI] [PubMed] [Google Scholar]
- Haas P, Gilmour D (2006) Chemokine signaling mediates self-organizing tissue migration in the zebrafish lateral line. Dev Cell 10: 673–680 [DOI] [PubMed] [Google Scholar]
- Hagmann J, Burger MM, Dagan D (1999) Regulation of plasma membrane blebbing by the cytoskeleton. J Cell Biochem 73: 488–499 [PubMed] [Google Scholar]
- Halbleib JM, Nelson WJ (2006) Cadherins in development: cell adhesion, sorting, and tissue morphogenesis. Genes Dev 20: 3199–3214 [DOI] [PubMed] [Google Scholar]
- Hava D, Forster U, Matsuda M, Cui S, Link BA, Eichhorst J, Wiesner B, Chitnis A, Abdelilah-Seyfried S (2009) Apical membrane maturation and cellular rosette formation during morphogenesis of the zebrafish lateral line. J Cell Sci 122: 687–695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hotulainen P, Lappalainen P (2006) Stress fibers are generated by two distinct actin assembly mechanisms in motile cells. J Cell Biol 173: 383–394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu K, Ji L, Applegate KT, Danuser G, Waterman-Storer CM (2007) Differential transmission of actin motion within focal adhesions. Science 315: 111–115 [DOI] [PubMed] [Google Scholar]
- Huber AH, Weis WI (2001) The structure of the beta-catenin/E-cadherin complex and the molecular basis of diverse ligand recognition by beta-catenin. Cell 105: 391–402 [DOI] [PubMed] [Google Scholar]
- Humphries JD, Wang P, Streuli C, Geiger B, Humphries MJ, Ballestrem C (2007) Vinculin controls focal adhesion formation by direct interactions with talin and actin. J Cell Biol 179: 1043–1057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutterer A, Betschinger J, Petronczki M, Knoblich JA (2004) Sequential roles of Cdc42, Par-6, aPKC, and Lgl in the establishment of epithelial polarity during Drosophila embryogenesis. Dev Cell 6: 845–854 [DOI] [PubMed] [Google Scholar]
- Huveneers S, Danen EH (2009) Adhesion signaling—crosstalk between integrins, Src and Rho. J Cell Sci 122: 1059–1069 [DOI] [PubMed] [Google Scholar]
- Huveneers S, Truong H, Fassler R, Sonnenberg A, Danen EH (2008) Binding of soluble fibronectin to integrin alpha5 beta1—link to focal adhesion redistribution and contractile shape. J Cell Sci 121: 2452–2462 [DOI] [PubMed] [Google Scholar]
- Jaffe AB, Kaji N, Durgan J, Hall A (2008) Cdc42 controls spindle orientation to position the apical surface during epithelial morphogenesis. J Cell Biol 183: 625–633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jessen JR, Topczewski J, Bingham S, Sepich DS, Marlow F, Chandrasekhar A, Solnica-Krezel L (2002) Zebrafish trilobite identifies new roles for Strabismus in gastrulation and neuronal movements. Nat Cell Biol 4: 610–615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kametani Y, Takeichi M (2007) Basal-to-apical cadherin flow at cell junctions. Nat Cell Biol 9: 92–98 [DOI] [PubMed] [Google Scholar]
- Katz BZ, Zamir E, Bershadsky A, Kam Z, Yamada KM, Geiger B (2000) Physical state of the extracellular matrix regulates the structure and molecular composition of cell-matrix adhesions. Mol Biol Cell 11: 1047–1060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaverina I, Krylyshkina O, Gimona M, Beningo K, Wang YL, Small JV (2000) Enforced polarisation and locomotion of fibroblasts lacking microtubules. Curr Biol 10: 739–742 [DOI] [PubMed] [Google Scholar]
- Kilian B, Mansukoski H, Barbosa FC, Ulrich F, Tada M, Heisenberg CP (2003) The role of Ppt/Wnt5 in regulating cell shape and movement during zebrafish gastrulation. Mech Dev 120: 467–476 [DOI] [PubMed] [Google Scholar]
- Kim M, Carman CV, Springer TA (2003) Bidirectional transmembrane signaling by cytoplasmic domain separation in integrins. Science 301: 1720–1725 [DOI] [PubMed] [Google Scholar]
- Kim M, Carman CV, Yang W, Salas A, Springer TA (2004) The primacy of affinity over clustering in regulation of adhesiveness of the integrin {alpha}L{beta}2. J Cell Biol 167: 1241–1253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koh W, Sachidanandam K, Stratman AN, Sacharidou A, Mayo AM, Murphy EA, Cheresh DA, Davis GE (2009) Formation of endothelial lumens requires a coordinated PKCepsilon-, Src-, Pak- and Raf-kinase-dependent signaling cascade downstream of Cdc42 activation. J Cell Sci 122: 1812–1822 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong F, Garcia AJ, Mould AP, Humphries MJ, Zhu C (2009) Demonstration of catch bonds between an integrin and its ligand. J Cell Biol 185: 1275–1284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labouesse M (2006) Epithelial junctions and attachments. WormBook 13: 1–21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ladoux B, Anon E, Lambert M, Rabodzey A, Hersen P, Buguin A, Silberzan P, Mege RM (2010) Strength dependence of cadherin-mediated adhesions. Biophys J 98: 534–542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larue L, Ohsugi M, Hirchenhain J, Kemler R (1994) E-cadherin null mutant embryos fail to form a trophectoderm epithelium. Proc Natl Acad Sci USA 91: 8263–8267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lecaudey V, Cakan-Akdogan G, Norton WH, Gilmour D (2008) Dynamic Fgf signaling couples morphogenesis and migration in the zebrafish lateral line primordium. Development 135: 2695–2705 [DOI] [PubMed] [Google Scholar]
- Lee SE, Kamm RD, Mofrad MR (2007) Force-induced activation of talin and its possible role in focal adhesion mechanotransduction. J Biomech 40: 2096–2106 [DOI] [PubMed] [Google Scholar]
- Lickert H, Bauer A, Kemler R, Stappert J (2000) Casein kinase II phosphorylation of E-cadherin increases E-cadherin/beta-catenin interaction and strengthens cell-cell adhesion. J Biol Chem 275: 5090–5095 [DOI] [PubMed] [Google Scholar]
- Lim Y, Lim ST, Tomar A, Gardel M, Bernard-Trifilo JA, Chen XL, Uryu SA, Canete-Soler R, Zhai J, Lin H, Schlaepfer WW, Nalbant P, Bokoch G, Ilic D, Waterman-Storer C, Schlaepfer DD (2008) PyK2 and FAK connections to p190Rho guanine nucleotide exchange factor regulate RhoA activity, focal adhesion formation, and cell motility. J Cell Biol 180: 187–203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Z, Tan JL, Cohen DM, Yang MT, Sniadecki NJ, Ruiz SA, Nelson CM, Chen CS (2010) Mechanical tugging force regulates the size of cell-cell junctions. Proc Natl Acad Sci USA 107: 9944–9949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma YQ, Qin J, Wu C, Plow EF (2008) Kindlin-2 (Mig-2): a co-activator of beta3 integrins. J Cell Biol 181: 439–446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machacek M, Hodgson L, Welch C, Elliott H, Pertz O, Nalbant P, Abell A, Johnson GL, Hahn KM, Danuser G (2009) Coordination of Rho GTPase activities during cell protrusion. Nature 461: 99–103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maddugoda MP, Crampton MS, Shewan AM, Yap AS (2007) Myosin VI and vinculin cooperate during the morphogenesis of cadherin cell cell contacts in mammalian epithelial cells. J Cell Biol 178: 529–540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marlow F, Topczewski J, Sepich D, Solnica-Krezel L (2002) Zebrafish Rho kinase 2 acts downstream of Wnt11 to mediate cell polarity and effective convergence and extension movements. Curr Biol 12: 876–884 [DOI] [PubMed] [Google Scholar]
- Marsden M, DeSimone DW (2001) Regulation of cell polarity, radial intercalation and epiboly in Xenopus: novel roles for integrin and fibronectin. Development 128: 3635–3647 [DOI] [PubMed] [Google Scholar]
- Marsden M, DeSimone DW (2003) Integrin-ECM interactions regulate cadherin-dependent cell adhesion and are required for convergent extension in Xenopus. Curr Biol 13: 1182–1191 [DOI] [PubMed] [Google Scholar]
- Marshall TW, Aloor HL, Bear JE (2009) Coronin 2A regulates a subset of focal-adhesion-turnover events through the cofilin pathway. J Cell Sci 122: 3061–3069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin AC, Gelbart M, Fernandez-Gonzalez R, Kaschube M, Wieschaus EF (2010) Integration of contractile forces during tissue invagination. J Cell Biol 188: 735–749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin AC, Kaschube M, Wieschaus EF (2009) Pulsed contractions of an actin-myosin network drive apical constriction. Nature 457: 495–499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin-Belmonte F, Gassama A, Datta A, Yu W, Rescher U, Gerke V, Mostov K (2007) PTEN-mediated apical segregation of phosphoinositides controls epithelial morphogenesis through Cdc42. Cell 128: 383–397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Rico C, Pincet F, Thiery JP, Dufour S (2010) Integrins stimulate E-cadherin-mediated intercellular adhesion by regulating Src-kinase activation and actomyosin contractility. J Cell Sci 123: 712–722 [DOI] [PubMed] [Google Scholar]
- McLachlan RW, Kraemer A, Helwani FM, Kovacs EM, Yap AS (2007) E-cadherin adhesion activates c-Src signaling at cell-cell contacts. Mol Biol Cell 18: 3214–3223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melani M, Simpson KJ, Brugge JS, Montell D (2008) Regulation of cell adhesion and collective cell migration by hindsight and its human homolog RREB1. Curr Biol 18: 532–537 [DOI] [PubMed] [Google Scholar]
- Mitra SK, Hanson DA, Schlaepfer DD (2005) Focal adhesion kinase: in command and control of cell motility. Nat Rev Mol Cell Biol 6: 56–68 [DOI] [PubMed] [Google Scholar]
- Montanez E, Ussar S, Schifferer M, Bosl M, Zent R, Moser M, Fassler R (2008) Kindlin-2 controls bidirectional signaling of integrins. Genes Dev 22: 1325–1330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montero JA, Carvalho L, Wilsch-Brauninger M, Kilian B, Mustafa C, Heisenberg CP (2005) Shield formation at the onset of zebrafish gastrulation. Development 132: 1187–1198 [DOI] [PubMed] [Google Scholar]
- Moser M, Nieswandt B, Ussar S, Pozgajova M, Fassler R (2008) Kindlin-3 is essential for integrin activation and platelet aggregation. Nat Med 14: 325–330 [DOI] [PubMed] [Google Scholar]
- Na S, Collin O, Chowdhury F, Tay B, Ouyang M, Wang Y, Wang N (2008) Rapid signal transduction in living cells is a unique feature of mechanotransduction. Proc Natl Acad Sci USA 105: 6626–6631 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nejsum LN, Nelson WJ (2007) A molecular mechanism directly linking E-cadherin adhesion to initiation of epithelial cell surface polarity. J Cell Biol 178: 323–335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson WJ (2009) Remodeling epithelial cell organization: transitions between front-rear and apical-basal polarity. Cold Spring Harb Perspect Biol 1: a000513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niewiadomska P, Godt D, Tepass U (1999) DE-Cadherin is required for intercellular motility during Drosophila oogenesis. J Cell Biol 144: 533–547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishimura T, Takeichi M (2008) Shroom3-mediated recruitment of Rho kinases to the apical cell junctions regulates epithelial and neuroepithelial planar remodeling. Development 135: 1493–1502 [DOI] [PubMed] [Google Scholar]
- Nishimura T, Takeichi M (2009) Remodeling of the adherens junctions during morphogenesis. Curr Top Dev Biol 89: 33–54 [DOI] [PubMed] [Google Scholar]
- Noren NK, Niessen CM, Gumbiner BM, Burridge K (2001) Cadherin engagement regulates Rho family GTPases. J Biol Chem 276: 33305–33308 [DOI] [PubMed] [Google Scholar]
- O'Brien LE, Jou TS, Pollack AL, Zhang Q, Hansen SH, Yurchenco P, Mostov KE (2001) Rac1 orientates epithelial apical polarity through effects on basolateral laminin assembly. Nat Cell Biol 3: 831–838 [DOI] [PubMed] [Google Scholar]
- O'Brien LE, Zegers MM, Mostov KE (2002) Opinion: building epithelial architecture: insights from three-dimensional culture models. Nat Rev Mol Cell Biol 3: 531–537 [DOI] [PubMed] [Google Scholar]
- Ouyang M, Sun J, Chien S, Wang Y (2008) Determination of hierarchical relationship of Src and Rac at subcellular locations with FRET biosensors. Proc Natl Acad Sci USA 105: 14353–14358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papusheva E, Mello de Queiroz F, Dalous J, Han Y, Esposito A, Jares-Erijmanxa EA, Jovin TM, Bunt G (2009) Dynamic conformational changes in the FERM domain of FAK are involved in focal-adhesion behavior during cell spreading and motility. J Cell Sci 122: 656–666 [DOI] [PubMed] [Google Scholar]
- Perez TD, Tamada M, Sheetz MP, Nelson WJ (2008) Immediate-early signaling induced by E-cadherin engagement and adhesion. J Biol Chem 283: 5014–5022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pilot F, Philippe JM, Lemmers C, Lecuit T (2006) Spatial control of actin organization at adherens junctions by a synaptotagmin-like protein Btsz. Nature 442: 580–584 [DOI] [PubMed] [Google Scholar]
- Plant PJ, Fawcett JP, Lin DC, Holdorf AD, Binns K, Kulkarni S, Pawson T (2003) A polarity complex of mPar-6 and atypical PKC binds, phosphorylates and regulates mammalian Lgl. Nat Cell Biol 5: 301–308 [DOI] [PubMed] [Google Scholar]
- Poh YC, Na S, Chowdhury F, Ouyang M, Wang Y, Wang N (2009) Rapid activation of Rac GTPase in living cells by force is independent of Src. PLoS One 4: e7886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puklin-Faucher E, Gao M, Schulten K, Vogel V (2006) How the headpiece hinge angle is opened: new insights into the dynamics of integrin activation. J Cell Biol 175: 349–360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puklin-Faucher E, Sheetz MP (2009) The mechanical integrin cycle. J Cell Sci 122: 179–186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ren G, Helwani FM, Verma S, McLachlan RW, Weed SA, Yap AS (2009) Cortactin is a functional target of E-cadherin-activated Src family kinases in MCF7 epithelial monolayers. J Biol Chem 284: 18913–18922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roca-Cusachs P, Gauthier NC, Del Rio A, Sheetz MP (2009) Clustering of alpha(5)beta(1) integrins determines adhesion strength whereas alpha(v)beta(3) and talin enable mechanotransduction. Proc Natl Acad Sci USA 106: 16245–16250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossier OM, Gauthier N, Biais N, Vonnegut W, Fardin MA, Avigan P, Heller ER, Mathur A, Ghassemi S, Koeckert MS, Hone JC, Sheetz MP (2010) Force generated by actomyosin contraction builds bridges between adhesive contacts. EMBO J 29: 1055–1068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rozario T, DeSimone DW (2010) The extracellular matrix in development and morphogenesis: a dynamic view. Dev Biol 341: 126–140 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sacharidou A, Koh W, Stratman AN, Mayo AM, Fisher KE, Davis GE (2010) Endothelial lumen signaling complexes control 3D matrix-specific tubulogenesis through interdependent Cdc42- and MT1-MMP-mediated events. Blood 115: 5259–5269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saez A, Buguin A, Silberzan P, Ladoux B (2005) Is the mechanical activity of epithelial cells controlled by deformations or forces? Biophys J 89: L52–L54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahai E, Marshall CJ (2002) ROCK and Dia have opposing effects on adherens junctions downstream of Rho. Nat Cell Biol 4: 408–415 [DOI] [PubMed] [Google Scholar]
- Sander EE, van Delft S, ten Klooster JP, Reid T, van der Kammen RA, Michiels F, Collard JG (1998) Matrix-dependent Tiam1/Rac signaling in epithelial cells promotes either cell-cell adhesion or cell migration and is regulated by phosphatidylinositol 3-kinase. J Cell Biol 143: 1385–1398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawada Y, Sheetz MP (2002) Force transduction by Triton cytoskeletons. J Cell Biol 156: 609–615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaller MD, Hildebrand JD, Shannon JD, Fox JW, Vines RR, Parsons JT (1994) Autophosphorylation of the focal adhesion kinase, pp125FAK, directs SH2-dependent binding of pp60src. Mol Cell Biol 14: 1680–1688 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlaepfer DD, Jones KC, Hunter T (1998) Multiple Grb2-mediated integrin-stimulated signaling pathways to ERK2/mitogen-activated protein kinase: summation of both c-Src- and focal adhesion kinase-initiated tyrosine phosphorylation events. Mol Cell Biol 18: 2571–2585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw RM, Fay AJ, Puthenveedu MA, von Zastrow M, Jan YN, Jan LY (2007) Microtubule plus-end-tracking proteins target gap junctions directly from the cell interior to adherens junctions. Cell 128: 547–560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaye DD, Casanova J, Llimargas M (2008) Modulation of intracellular trafficking regulates cell intercalation in the Drosophila trachea. Nat Cell Biol 10: 964–970 [DOI] [PubMed] [Google Scholar]
- Shewan AM, Maddugoda M, Kraemer A, Stehbens SJ, Verma S, Kovacs EM, Yap AS (2005) Myosin 2 is a key Rho kinase target necessary for the local concentration of E-cadherin at cell-cell contacts. Mol Biol Cell 16: 4531–4542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skoglund P, Rolo A, Chen X, Gumbiner BM, Keller R (2008) Convergence and extension at gastrulation require a myosin IIB-dependent cortical actin network. Development 135: 2435–2444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solon J, Kaya-Copur A, Colombelli J, Brunner D (2009) Pulsed forces timed by a ratchet-like mechanism drive directed tissue movement during dorsal closure. Cell 137: 1331–1342 [DOI] [PubMed] [Google Scholar]
- Sonawane M, Martin-Maischein H, Schwarz H, Nusslein-Volhard C (2009) Lgl2 and E-cadherin act antagonistically to regulate hemidesmosome formation during epidermal development in zebrafish. Development 136: 1231–1240 [DOI] [PubMed] [Google Scholar]
- Sperry RB, Bishop NH, Bramwell JJ, Brodeur MN, Carter MJ, Fowler BT, Lewis ZB, Maxfield SD, Staley DM, Vellinga RM, Hansen MD (2009) Zyxin controls migration in epithelial-mesenchymal transition by mediating actin-membrane linkages at cell-cell junctions. J Cell Physiol 222: 612–624 [DOI] [PubMed] [Google Scholar]
- Stamenovic D, Mijailovich SM, Tolic-Norrelykke IM, Chen J, Wang N (2002) Cell prestress. II. Contribution of microtubules. Am J Physiol Cell Physiol 282: C617–C624 [DOI] [PubMed] [Google Scholar]
- Stemmler MP (2008) Cadherins in development and cancer. Mol Biosyst 4: 835–850 [DOI] [PubMed] [Google Scholar]
- Tadokoro S, Shattil SJ, Eto K, Tai V, Liddington RC, de Pereda JM, Ginsberg MH, Calderwood DA (2003) Talin binding to integrin beta tails: a final common step in integrin activation. Science 302: 103–106 [DOI] [PubMed] [Google Scholar]
- Tahinci E, Symes K (2003) Distinct functions of Rho and Rac are required for convergent extension during Xenopus gastrulation. Dev Biol 259: 318–335 [DOI] [PubMed] [Google Scholar]
- Takagi J, Petre BM, Walz T, Springer TA (2002) Global conformational rearrangements in integrin extracellular domains in outside-in and inside-out signaling. Cell 110: 599–611 [DOI] [PubMed] [Google Scholar]
- Thery M, Racine V, Piel M, Pepin A, Dimitrov A, Chen Y, Sibarita JB, Bornens M (2006) Anisotropy of cell adhesive microenvironment governs cell internal organization and orientation of polarity. Proc Natl Acad Sci USA 103: 19771–19776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topczewski J, Sepich DS, Myers DC, Walker C, Amores A, Lele Z, Hammerschmidt M, Postlethwait J, Solnica-Krezel L (2001) The zebrafish glypican knypek controls cell polarity during gastrulation movements of convergent extension. Dev Cell 1: 251–264 [DOI] [PubMed] [Google Scholar]
- Totsukawa G, Wu Y, Sasaki Y, Hartshorne DJ, Yamakita Y, Yamashiro S, Matsumura F (2004) Distinct roles of MLCK and ROCK in the regulation of membrane protrusions and focal adhesion dynamics during cell migration of fibroblasts. J Cell Biol 164: 427–439 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Totsukawa G, Yamakita Y, Yamashiro S, Hartshorne DJ, Sasaki Y, Matsumura F (2000) Distinct roles of ROCK (Rho-kinase) and MLCK in spatial regulation of MLC phosphorylation for assembly of stress fibers and focal adhesions in 3T3 fibroblasts. J Cell Biol 150: 797–806 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulrich F, Krieg M, Schotz EM, Link V, Castanon I, Schnabel V, Taubenberger A, Mueller D, Puech PH, Heisenberg CP (2005) Wnt11 functions in gastrulation by controlling cell cohesion through Rab5c and E-cadherin. Dev Cell 9: 555–564 [DOI] [PubMed] [Google Scholar]
- Vicente-Manzanares M, Webb DJ, Horwitz AR (2005) Cell migration at a glance. J Cell Sci 118: 4917–4919 [DOI] [PubMed] [Google Scholar]
- Vicente-Manzanares M, Zareno J, Whitmore L, Choi CK, Horwitz AF (2007) Regulation of protrusion, adhesion dynamics, and polarity by myosins IIA and IIB in migrating cells. J Cell Biol 176: 573–580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- von der Hardt S, Bakkers J, Inbal A, Carvalho L, Solnica-Krezel L, Heisenberg CP, Hammerschmidt M (2007) The Bmp gradient of the zebrafish gastrula guides migrating lateral cells by regulating cell-cell adhesion. Curr Biol 17: 475–487 [DOI] [PubMed] [Google Scholar]
- Wang F, Dumstrei K, Haag T, Hartenstein V (2004) The role of DE-cadherin during cellularization, germ layer formation and early neurogenesis in the Drosophila embryo. Dev Biol 270: 350–363 [DOI] [PubMed] [Google Scholar]
- Wang HB, Dembo M, Hanks SK, Wang Y (2001a) Focal adhesion kinase is involved in mechanosensing during fibroblast migration. Proc Natl Acad Sci USA 98: 11295–11300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang N, Naruse K, Stamenovic D, Fredberg JJ, Mijailovich SM, Tolic-Norrelykke IM, Polte T, Mannix R, Ingber DE (2001b) Mechanical behavior in living cells consistent with the tensegrity model. Proc Natl Acad Sci USA 98: 7765–7770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, He L, Wu YI, Hahn KM, Montell DJ (2010) Light-mediated activation reveals a key role for Rac in collective guidance of cell movement in vivo. Nat Cell Biol 12: 591–597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Botvinick EL, Zhao Y, Berns MW, Usami S, Tsien RY, Chien S (2005) Visualizing the mechanical activation of Src. Nature 434: 1040–1045 [DOI] [PubMed] [Google Scholar]
- Webb DJ, Donais K, Whitmore LA, Thomas SM, Turner CE, Parsons JT, Horwitz AF (2004) FAK-Src signalling through paxillin, ERK and MLCK regulates adhesion disassembly. Nat Cell Biol 6: 154–161 [DOI] [PubMed] [Google Scholar]
- Webb DJ, Zhang H, Horwitz AF (2005) Cell migration: an overview. Methods Mol Biol 294: 3–11 [PubMed] [Google Scholar]
- Wegener KL, Partridge AW, Han J, Pickford AR, Liddington RC, Ginsberg MH, Campbell ID (2007) Structural basis of integrin activation by talin. Cell 128: 171–182 [DOI] [PubMed] [Google Scholar]
- Wierzbicka-Patynowski I, Schwarzbauer JE (2003) The ins and outs of fibronectin matrix assembly. J Cell Sci 116: 3269–3276 [DOI] [PubMed] [Google Scholar]
- Winklbauer R (2009) Cell adhesion in amphibian gastrulation. Int Rev Cell Mol Biol 278: 215–275 [DOI] [PubMed] [Google Scholar]
- Wu X, Li S, Chrostek-Grashoff A, Czuchra A, Meyer H, Yurchenco PD, Brakebusch C (2007) Cdc42 is crucial for the establishment of epithelial polarity during early mammalian development. Dev Dyn 236: 2767–2778 [DOI] [PubMed] [Google Scholar]
- Wu YI, Frey D, Lungu OI, Jaehrig A, Schlichting I, Kuhlman B, Hahn KM (2009) A genetically encoded photoactivatable Rac controls the motility of living cells. Nature 461: 104–108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia N, Thodeti CK, Hunt TP, Xu Q, Ho M, Whitesides GM, Westervelt R, Ingber DE (2008) Directional control of cell motility through focal adhesion positioning and spatial control of Rac activation. FASEB J 22: 1649–1659 [DOI] [PubMed] [Google Scholar]
- Xiao T, Takagi J, Coller BS, Wang JH, Springer TA (2004) Structural basis for allostery in integrins and binding to fibrinogen-mimetic therapeutics. Nature 432: 59–67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada S, Nelson WJ (2007) Localized zones of Rho and Rac activities drive initiation and expansion of epithelial cell-cell adhesion. J Cell Biol 178: 517–527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada S, Pokutta S, Drees F, Weis WI, Nelson WJ (2005) Deconstructing the cadherin-catenin-actin complex. Cell 123: 889–901 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yano H, Mazaki Y, Kurokawa K, Hanks SK, Matsuda M, Sabe H (2004) Roles played by a subset of integrin signaling molecules in cadherin-based cell-cell adhesion. J Cell Biol 166: 283–295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yonemura S, Wada Y, Watanabe T, Nagafuchi A, Shibata M (2010) alpha-Catenin as a tension transducer that induces adherens junction development. Nat Cell Biol 12: 533–542 [DOI] [PubMed] [Google Scholar]
- Yu W, Datta A, Leroy P, O'Brien LE, Mak G, Jou TS, Matlin KS, Mostov KE, Zegers MM (2005) Beta1-integrin orients epithelial polarity via Rac1 and laminin. Mol Biol Cell 16: 433–445 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaidel-Bar R, Ballestrem C, Kam Z, Geiger B (2003) Early molecular events in the assembly of matrix adhesions at the leading edge of migrating cells. J Cell Sci 116: 4605–4613 [DOI] [PubMed] [Google Scholar]
- Zaidel-Bar R, Cohen M, Addadi L, Geiger B (2004) Hierarchical assembly of cell-matrix adhesion complexes. Biochem Soc Trans 32: 416–420 [DOI] [PubMed] [Google Scholar]
- Zallen JA, Blankenship JT (2008) Multicellular dynamics during epithelial elongation. Semin Cell Dev Biol 19: 263–270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamir E, Geiger B (2001) Molecular complexity and dynamics of cell-matrix adhesions. J Cell Sci 114: 3583–3590 [DOI] [PubMed] [Google Scholar]
- Zamir E, Katz M, Posen Y, Erez N, Yamada KM, Katz BZ, Lin S, Lin DC, Bershadsky A, Kam Z, Geiger B (2000) Dynamics and segregation of cell-matrix adhesions in cultured fibroblasts. Nat Cell Biol 2: 191–196 [DOI] [PubMed] [Google Scholar]
- Zegers MM, O'Brien LE, Yu W, Datta A, Mostov KE (2003) Epithelial polarity and tubulogenesis in vitro. Trends Cell Biol 13: 169–176 [DOI] [PubMed] [Google Scholar]
- Zhang Y, Sivasankar S, Nelson WJ, Chu S (2009) Resolving cadherin interactions and binding cooperativity at the single-molecule level. Proc Natl Acad Sci USA 106: 109–114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou H, Kramer RH (2005) Integrin engagement differentially modulates epithelial cell motility by RhoA/ROCK and PAK1. J Biol Chem 280: 10624–10635 [DOI] [PubMed] [Google Scholar]
- Zhu S, Liu L, Korzh V, Gong Z, Low BC (2006) RhoA acts downstream of Wnt5 and Wnt11 to regulate convergence and extension movements by involving effectors Rho kinase and Diaphanous: use of zebrafish as an in vivo model for GTPase signaling. Cell Signal 18: 359–372 [DOI] [PubMed] [Google Scholar]
- Zovein AC, Luque A, Turlo KA, Hofmann JJ, Yee KM, Becker MS, Fassler R, Mellman I, Lane TF, Iruela-Arispe ML (2010) Beta1 integrin establishes endothelial cell polarity and arteriolar lumen formation via a Par3-dependent mechanism. Dev Cell 18: 39–51 [DOI] [PMC free article] [PubMed] [Google Scholar]