Abstract
Heterochrony, or the evolution of ontogeny, has been well studied in embryology and skeletal development, providing insight into morphological and genetic mechanisms of evolution.1–5 However, heterochronic studies of behavior and cognition lag behind in comparison. In a recent study we investigated the ontogeny of social behavior and cognition in humans’ closest living relatives, chimpanzees (Pan troglodytes) and bonobos (Pan paniscus). These two species are estimated to have had a chimpanzeelike common ancestor between 0.86 and 1.8 mya.6,7 Bonobos have been argued to exhibit morphological indications of paedomorphism relative to chimpanzees, especially in the cranium, and to exhibit paedomorphic behavior as adults.6–11 We found that bonobos exhibit developmental delays relative to chimpanzees in several aspects of their social behavior and cognition. Here, we describe how placing these results in the framework of heterochrony contributes to understanding behavioral and cognitive differences between adults of these two species and to our knowledge of hominid evolution in general.
Key words: heterochrony, chimpanzee, bonobo, behavior, cognition, ontogeny, evolution
Heterochrony research quantifies how developmental processes evolve, through characterization of the relationship between a trait’s growth in size and shape over time. Heterochronic processes are divided into two major categories—paedomorphosis, where a trait in the descendant species resembles a juvenile form of the ancestral species, and peramorphosis, where a trait in the descendant species develops beyond the adult form of the ancestral species. Within each of these categories, heterochrony researchers have created precise definitions of how selection can act on development through changing the start and end points of development, the rate of development, and/or the means by which development occurs.12,13
In general, it is difficult to apply morphological concepts of heterochronic change to behavioral and cognitive traits because such traits do not often grow at measurable rates and in predictable stages of form.13,14 However, a few behaviors can be studied in this manner because they can be characterized as having distinct “sizes” and “shapes,” attributes used by the heterchrony model in the study of morphology. For example, in some species agonistic signals increase in both number (“size”) and complexity (“shape”) through infancy and juvenility.15,16 If an adult of a descendant species shows a reduced number and complexity of agonistic signals, this meets the morphological criteria for paedomorphosis.17
The heterochrony model may prove to be applied more easily to cognitive abilities than to behavioral traits, because there are more obvious matches of developmental traits to “size” and “shape.” The order in which cognitive abilities emerge over the course of development can vary across species,18 thus the relative proficiencies at differing tasks can provide the equivalent of a shape component. In turn, proficiency across cognitive tasks, or more general intelligence, can be indexed by the size component. Research into human cognitive heterochrony has suggested that children acquire cognitive abilities both at a different rate and in a different sequence relative to other primates, suggesting a fruitful area for further study.19,20
Investigating heterochrony in several dimensions (behavioral and/or cognitive in addition to morphological) enables the possibility of finding correlations across seemingly unrelated processes and assessing whether such correlations are due to common developmental mechanisms. Current research suggests that although in some poikilotherms morphological and behavioral paedomorphic processes are decoupled, in others they may be selected as a unit.21,22 Little work has examined whether correlations exist between morphological and behavioral heterochrony in non-human primates.
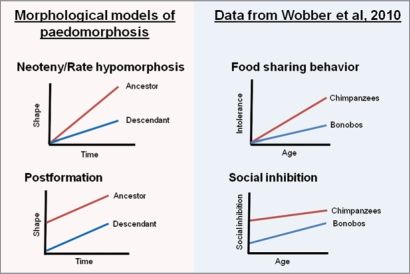
In our recent study, we demonstrated that bonobos exhibit delayed development in several aspects of social behavior and cognition relative to chimpanzees.23 This finding is in line with prior evidence that bonobos exhibit cranial paedomorphosis, suggesting potential broad-scale developmental evolution in bonobos.7 Here, we present our data in relation to the models of paedomorphosis defined in previous work.8 We utilize age in years as our time variable and performance on each behavioral or cognitive task as our shape component, with no relevant size component for these analyses. In our first experiment, we found that chimpanzees increased in their intolerance during food sharing with age, while bonobos maintained juvenile, low, levels of intolerance into adulthood. This resulted in adult bonobos sharing food more readily than adult chimpanzees.23,24 The observed pattern best resembles neoteny or rate hypomorphosis in bonobos, since juveniles of the two species showed comparable levels of intolerance and intolerance increased at differing rates with age in the two species (Fig. 1).
Figure 1.
Depictions of the patterns found in our recent study23 in comparison to models of paedomorphosis.8 Bonobos appear to exhibit neoteny or rate hypomorphosis in their ontogeny of intolerant behavior, while in their patterns of social inhibition across two tasks they appear to exhibit postformation, the process found to create paedomorphosis in aspects of the bonobo cranium.8
In our second experiment, we found that chimpanzees showed consistent performance across ages on a social response inhibition task, while bonobos improved at a slower rate across infancy.23 This conforms to the morphological concept of postformation (paedomorphosis due to initial shape under-development) in bonobos, with the youngest bonobos having more difficulty than the youngest chimpanzees (Fig. 1). Finally, we found that on a social reversal learning task,25 bonobos started at a lower level of performance than chimpanzees in juvenility, and while both species improved with age, adult bonobos never reached a level of performance comparable to adult chimpanzees.23 Again, this appears to represent postformation in the bonobos, with there being a difference in early levels of ability that persists into adulthood (Fig. 1).
Together, our results suggest that behavioral and cognitive traits can be subject to ontogenetic evolution, and that models of heterochrony can be usefully extended to traits not typically assessed in the evolutionary-developmental framework. One result is to encourage further cross-disciplinary comparisons of traits subject to developmental shifts in evolution. In particular, our finding that behavioral and cognitive heterochrony appears correlated with cranial heterochrony in bonobos raises provocative questions about whether the developmental mechanisms responsible for these changes reflect an underlying uniformity.
Acknowledgements
This work was supported in part by a European Research Commission Advanced Grant Agreement 233297 and a National Science Foundation grant NSFBCS-08-27552-02 to B.H., along with NSF DDIG 0851291 and a Wenner-Gren Foundation Grant to V.W.
Footnotes
Previously published online: www.landesbioscience.com/journals/cib/article/11762
References
- 1.Zelditch M. Beyond heterochrony: the evolution of development. New York: Wiley-Liss, Inc; 2001. [Google Scholar]
- 2.McKinney M, McNamara K. Heterochrony: the evolution of ontogeny. New York: Plenum Press; 1991. [Google Scholar]
- 3.Abzhanov A, Kuo W, Hartmann C, Grant B, Grant P, Tabin C. The calmodulin pathway and evolution of elongated beak morphology in Darwin’s finches. Nature. 2006;442:563–567. doi: 10.1038/nature04843. [DOI] [PubMed] [Google Scholar]
- 4.Leigh S, Shea B. Ontogeny of body size variation in African apes. Am J Phys Anthropol. 1996;99:43–65. doi: 10.1002/(SICI)1096-8644(199601)99:1<43::AID-AJPA3>3.0.CO;2-0. [DOI] [PubMed] [Google Scholar]
- 5.Meaney M. Maternal care, gene expression, and the transmission of individual differences in stress reactivity across generations. Ann Rev Neurosci. 2001;24:1161–1192. doi: 10.1146/annurev.neuro.24.1.1161. [DOI] [PubMed] [Google Scholar]
- 6.Won Y, Hey J. Divergence population genetics of chimpanzees. Mol Biol Evol. 2005;22:297–307. doi: 10.1093/molbev/msi017. [DOI] [PubMed] [Google Scholar]
- 7.Wrangham R, Pilbeam D. African apes as time machines. In: Galdikas B, Briggs N, Sheeran L, Shapiro G, Goodall J, editors. All apes great and small. New York: Kluwer Academic/Plenum Publishers; 2001. pp. 5–18. [Google Scholar]
- 8.Lieberman DE, Carlo J, Ponce de Leon M, Zollikofer C. A geometric morphometric analysis of heterochrony in the cranium of chimpanzees and bonobos. J Human Evol. 2007;52:647–662. doi: 10.1016/j.jhevol.2006.12.005. [DOI] [PubMed] [Google Scholar]
- 9.Kuroda S. Developmental retardation and behavioral characteristics of pygmy chimpanzees. In: Heltne P, Marquardt L, editors. Understanding chimpanzees. Cambridge, Massachusetts: Harvard University Press; 1989. pp. 184–193. [Google Scholar]
- 10.Zihlman A, Cramer D. Skeletal differences between pygmy (Pan paniscus) and common chimpanzees (Pan troglodytes) Folia Primatol. 1978;29:86–94. doi: 10.1159/000155830. [DOI] [PubMed] [Google Scholar]
- 11.Fruth B, Hohmann G. How bonobos handle hunts and harvests: why share food? In: Boesch C, Hohmann G, Marchant L, editors. Behavioural diversity in chimpanzees and bonobos. 231-43. Cambridge: Cambridge University Press; 2002. [Google Scholar]
- 12.Godfrey L, Sutherland M. Paradox of peramophic paedomorphosis: heterochrony and human evolution. Am J Phys Anthropol. 1996;99:17–42. doi: 10.1002/ajpa.1330990102. [DOI] [PubMed] [Google Scholar]
- 13.Klingenberg C. Heterochrony and allometry: the analysis of evolutionary change in ontogeny. Biol Rev. 1998;73:79–123. doi: 10.1017/s000632319800512x. [DOI] [PubMed] [Google Scholar]
- 14.Alberch P, Gould S, Oster G, Wake D. Size and shape in ontogeny and phylogeny. Paleobiology. 1979;5:296–317. [Google Scholar]
- 15.Fox M. The anatomy of aggression and its ritualization in Canidae: a developmental and comparative study. Behaviour. 1969;35:242–258. [Google Scholar]
- 16.Cairns R, MacComble D, Hood K. A developmentalgenetic analysis of aggressive behavior in mice: I. Behavioral outcomes. J Comp Psychol. 1983;97:69–89. [PubMed] [Google Scholar]
- 17.Gariepy J, Bauer D, Cairns R. Selective breeding for differential aggression in mice provides evidence for heterochrony in social behaviours. Animal Behav. 2001;61:933–947. [Google Scholar]
- 18.Tomasello M, Carpenter M. The emergence of social cognition in three young chimpanzees. Monogr Soc Res Child Dev. 2005;70:1–155. doi: 10.1111/j.1540-5834.2005.00324.x. [DOI] [PubMed] [Google Scholar]
- 19.Langer J. The descent of cognitive development. Dev Sci. 2000;3:361–388. [Google Scholar]
- 20.Gomez JC. Species comparative studies and cognitive development. Trends Cogn Sci. 2005;9:118–125. doi: 10.1016/j.tics.2005.01.004. [DOI] [PubMed] [Google Scholar]
- 21.Denoel M. Paedomorphosis in the Alpine newt (Triturus alpestris): decoupling behavioural and morphological change. Behav EcolSociobiol. 2002;52:394–399. [Google Scholar]
- 22.Hastings P. Evolution of morphological and behavioral ontogenies in females of a highly dimorphic clade of blennioid fishes. Evolution. 2002;56:1644–1654. doi: 10.1111/j.0014-3820.2002.tb01476.x. [DOI] [PubMed] [Google Scholar]
- 23.Wobber V, Wrangham R, Hare B. Bonobos exhibit delayed development of social behavior and cognition relative to chimpanzees. Curr Biol. 2010;20:226–230. doi: 10.1016/j.cub.2009.11.070. [DOI] [PubMed] [Google Scholar]
- 24.Hare B, Melis AP, Woods V, Hastings S, Wrangham R. Tolerance allows bonobos to outperform chimpanzees on a cooperative task. Curr Biol. 2007;17:619–623. doi: 10.1016/j.cub.2007.02.040. [DOI] [PubMed] [Google Scholar]
- 25.Wobber V, Hare B. Testing the social dog hypothesis: are dogs also more skilled than chimpanzees in non-communicative social tasks? Behav Procs. 2009;81:423–428. doi: 10.1016/j.beproc.2009.04.003. [DOI] [PubMed] [Google Scholar]