SUMMARY
Protective immunity to avian influenza (AI) virus can be elicited in chickens by in ovo or intramuscular vaccination with replication-competent adenovirus (RCA)-free human recombinant adenovirus serotype 5 (Ad5) encoding AI virus H5 (AdTW68.H5) or H7 (AdCN94.H7) hemagglutinins. We evaluated bivalent in ovo vaccination with AdTW68.H5 and AdCN94.H7 and determined that vaccinated chickens developed robust hemagglutination inhibition (HI) antibody levels to both H5 and H7 AI strains. Additionally, we evaluated immune responses of 1-day-old chickens vaccinated via spray with AdCN94.H7. These birds showed increased immunoglobulin A responses in lachrymal fluids and increased interleukin-6 expression in Harderian gland–derived lymphocytes. However, specific HI antibodies were not detected in the sera of these birds. Because pigs might play a role as a “mixing vessel” for the generation of pandemic influenza viruses we explored the use of RCA-free adenovirus technology to immunize pigs against AI virus. Weanling piglets vaccinated intramuscularly with a single dose of RCA-free AdTW68.H5 developed strong systemic antibody responses 3 wk postvaccination. Intranasal application of AdTW68.H5 in piglets resulted in reduced vaccine coverage, i.e., 33% of pigs (2/6) developed an antibody response, but serum antibody levels in those successfully immunized animals were similar to intramuscularly vaccinated animals.
Keywords: avian influenza, vaccination, chickens, pigs, recombinant, adenovirus
Highly pathogenic (HP) avian influenza (AI) viruses belonging to the H5 or H7 subtypes continue to threaten the world poultry industry and are zoonotic agents with pandemic potential for humans (29). In 2005 the World Organization for Animal Health and the Food and Agriculture Organization of the United Nations recommended that vaccination of poultry be considered for the control of AI viruses (36). Mass vaccination increases the resistance of chicken populations and reduces viral shedding. Ideal AI vaccines should not only meet the normal requirements for all poultry vaccine products, i.e., induction of protective immunity, cost effectiveness, safety in the food chain, and feasibility for mass delivery, but they should also comply with a differentiation between infected and vaccinated animals (DIVA) strategy and increased biosafety regulations both at vaccine production centers and in the field. Numerous AI vaccines, both commercially available and experimentally produced, are capable of inducing appropriate levels of protection in chickens. Live AI vaccines based on naturally avirulent or attenuated strains are associated with an inherent risk for generating new reassortant influenza viruses (10) with unpredictable characteristics. Conventional whole AI virus inactivated vaccines induce appropriate immunity but have serious disadvantages such as incompatibility with DIVA, withdrawal time of as long as 42 days, and individual delivery, and are a biohazard during manufacturing. In addition, the vaccine production process is difficult because of the high lethality of most influenza virus strains for chicken embryos. Inactivated “marker” vaccines (4) overcome only some of these disadvantages. Multiple experimental recombinant vaccines have been developed in recent years, some of which have been reported to efficiently protect chickens against HPAI challenge (13,18,22). However, live recombinant vectors have the risk of generating revertants and allowing spread of genetically modified organisms in both target and nontarget species in the environment. In addition, it is not clear if preexisting immunity against some of these virus vectors, which are endemic in the poultry industry, may preclude the development of an efficacious immune response against the antigen, as has been shown for the fowlpox vector (23).
We previously reported that protective immunity against AI can be elicited in chickens in a single-dose regimen by in ovo vaccination with a replication-competent adenovirus (RCA)-free human adenovirus serotype 5 (Ad5) vector encoding either the AI virus H5 (AdTW68.H5) or H7 (AdCN94.H7) hemagglutinins (HA). Vaccinated chickens were protected against HPAI virus challenges (26,27). We have shown that chickens vaccinated in ovo with AdTW68.H5 and subsequently vaccinated intramuscularly with AdCN94.H7 after hatch develop antibodies against both the H5 and H7 HA proteins. This result suggests that preexisting Ad5 immunity in chickens does not significantly interfere with the potency of Ad5-vectored vaccines. We demonstrated that the use of a synthetic AI H5 gene with codons optimized to match the chicken transfer-RNA pool is more immunogenic than its cognate counterpart without codon optimization (27). Thus, propagation of influenza virus during recombinant Ad5 vaccine production is not required. In addition to in ovo injection, ocular administration of Ad5-vectored AI vaccines shows promise (8) and may allow mass immunization of poultry by spray. Vaccination by coarse spray is likely the most efficient route for mass vaccine delivery to existing chicken populations. Efforts on spray vaccination with live replicating respiratory viruses were started as early as 1946 by Jungherr and Luginbuhl (6,11). Live vaccine viruses applied by spray enter the host via different routes including direct contact with ocular and nasal mucosal membranes, inhalation (a mechanism that deposits the virus at different levels of the respiratory tract depending on the size of the aerosolized droplets), and via the intestinal tract, which occurs when spray-vaccinated birds beak-clean droplets of vaccine product left on their plumage. Vaccination via these mucosal routes not only triggers systemic immune responses but also mucosal immunity which is effected at mucosal-associated lymphoid tissues such as the Harderian gland (glandula lacrimalis accesoria; HG). Vaccination via this route with live attenuated poultry vaccines results in both systemic and mucosal immune responses. Mucosal antibodies can be detected and quantified in secretions such as the lachrymal fluid (1,2,3,7,25).
Domestic pigs (Sus domestica) have low transmissibility of H5N1 AI viruses (5) and low susceptibility to HPAI strains (15). However, some swine influenza A viruses have been shown to contain genes originated from avian, human, and pig influenza viruses (31). This emphasizes that pigs might play a role as a “mixing vessel” for the possible generation of pandemic influenza viruses (17). Human Ad5 recombinant viruses expressing the HA of an H3N2 swine influenza virus have been shown to elicit a strong antibody response and protection against swine influenza virus challenge in vaccinated pigs (34,35). Wesley et al. (35) used the 293 packaging cell line to propagate recombinant adenoviruses. However, contamination by RCA, generated by a double crossover event between the homologous overlapping sequences present in the Ad5 vector and the genome of the 293 packaging cell, appears to be an intrinsic problem for the 293 cell line (16,37).
The use of RCA-free recombinant Ad5 technology solves this contamination problem. RCA-free Ad5-vectored AI vaccines can be produced at a large scale in the well-characterized PER.C6 cell line in serum-free suspension bioreactors (14) in conjunction with chromatography-mediated purification (12). The PER.C6 cell line contains only Ad5 nucleotides 459–3510, which preclude double crossover-type homologous recombination between the PER.C6 genome and a matched Ad5 vector with this E1 gene deleted. Safe and cost-effective vaccine delivery to large chicken populations is feasible via routinely used automated in ovo injectors (19,32) in response to an emerging AI pandemic. In addition, this Ad5-vectored AI vaccine is DIVA compliant because the vector only elicits an immune response against the viral HA. In contrast to replicating live virus vaccines that may generate undesirable new reassortants with concurrently circulating wild influenza viruses (10), it is not possible for the DNA genome of Ad5 to undergo reassortment with the segmented RNA genome of an influenza virus. Finally, the RCA-free Ad5 vector will not propagate even in human cells in the absence of Ad E1 gene expression.
Because of the continuous threat posed by HPAI viruses belonging to the H5 or H7 types and thus the importance of adopting an immunization strategy with a broad antigen repertoire, in this study we evaluated bivalent in ovo vaccination with AdTW68.H5 and AdCN94.H7. In addition, because of the potential need of vaccinating existing chicken populations, we evaluated immune responses of 1-day-old chickens vaccinated via spray with AdTW68.H5. Finally, because of the relevance of pigs in influenza epidemiology, we report the use of RCA-free Ad5-vectored AI vaccines for the immunization of this species.
MATERIALS AND METHODS
Construction of RCA–free recombinant adenovirus vectors encoding the HA gene of the AI virus
Adenovirus (Ad)-vectored AI vaccines encoding either the avian influenza virus H5 or H7 HAs were developed as previously described (26,27). In brief, RCA-free Ad encoding the H5 AI HA (AdTW68.H5) was developed using the A/tk/WI/68 H5 HA gene which was PCR-amplified from a plasmid template (21) using a previously described primer pair (26). The fragment containing the full-length HA gene was inserted into the HindIII-BamHI site of the shuttle plasmid pAdApt (provided by Crucell Holland BV; Leiden, The Netherlands) to generate the plasmid pAdApt-TW68.H5 with the HA gene under transcriptional control of the human cytomegalovirus early promoter. An RCA-free, E1/E3-defective Ad5 vector encoding this H5 HA gene (AdTW68.H5) was subsequently constructed in human PER.C6 cells (provided by Crucell) by recombination following cotransfection of pAdApt-TW68.H5 with the Ad5 backbone plasmid pAdEasy1 (9) as described (20). The Ad5 recombinant vaccine with the H7 transgene was developed similarly. In brief, a fragment containing the full-length H7 gene of the A/Ch/NY/13142-5/94 AI virus was inserted into the HindIII-BamHI site of the shuttle plasmid pAdApt to generate the plasmid pAdApt-NY94.H7. An RCA-free E1/E3-defective Ad5 vector encoding the H7 gene was subsequently constructed in human PER.C6 cells by cotransfection of pAdApt-NY94.H7 with the Ad5 backbone plasmid pJM17, followed by multiple cycles of plaque purification. Both the AdTW68.H5 and AdCN94.H7 recombinant viruses were validated by DNA sequencing. Titers (infectious units [ifu]/ml) were determined by the Adeno-X rapid titer kit (BD Clontech; Mountain View, CA).
Animals
White leghorn specific-pathogen-free (SPF) chickens (Sunrise Farms, Inc., Catskill, NY) were used in all chicken vaccine experiments. Hatched chickens were maintained in Horsfall-type isolators in biosafety level 2 facilities. Feed and water were provided ad libitum. Pure-breed Yorkshire piglets were born and reared at the Auburn University Swine Research and Education Center (AU-SREC) in floor pens in indoor facilities. Water was provided ad libitum. Feeding followed the normal program for pig production used at AU-SREC using nutritionally balanced formulas for the weaning and growing period. Experimental procedures and animal care were performed in compliance with all applicable federal and institutional animal use guidelines. Auburn University College of Veterinary Medicine is an Association for Assessment and Accreditation of Laboratory Animal Care-accredited institution.
Experiment 1. Determine if in ovo vaccination with a bivalent RCA-free recombinant Ad vaccine encoding the H5 and H7 AI hemagglutinins elicits antibody responses against both expressed HAs
We evaluated bivalent in ovo vaccination with AdTW68.H5 and AdCN94.H7. A total of 60 SPF fertile eggs were divided into four groups and vaccinated in ovo on day 18 of incubation as follows: group A of 12 fertile eggs was vaccinated with 200 µl per egg of AdTW68.H5 containing 5.0 × 109 ifu, group B of 12 eggs was vaccinated with 200 µl per egg of AdCN94.H7 containing 5.0 × 107 ifu, group C of 24 eggs was vaccinated with a combination of both vaccine constructs (200 µl of each construct) containing the same doses applied to groups A and B, and group D of 12 eggs served as the unvaccinated control. Blood samples were obtained from all chicken groups by wing vein puncture on day 20 posthatch and at 10-day intervals through day 40 of age. Individual serum samples were inactivated in a water bath at 56 C for 30 min and tested by hemagglutination inhibition (HI) as described (24) for antibodies against the A/tk/OR/71 (H7N3) and the A/tk/WI/68 (H5N9) AI strains.
Experiment 2. Determine if RCA-free recombinant Ad vector applied by spray elicits immune responses to the expressed AI HA
We evaluated mucosal and systemic antibody levels in chickens vaccinated via coarse spray with AdCN94.H7. We also performed initial cytokine analyses (interferon gamma [IFN-γ] and interleukin 6 [IL-6]) on lymphocytes derived from the HG after spray immunization with this recombinant virus. Chickens hatched from SPF embryonated eggs were spray-vaccinated at 1 day of age using a commercial spray cabinet (Merial Select Inc., Gainesville, GA) which provides a droplet size of 140–150 microns. Each vaccine shot expels 8 ml of vaccine suspension in spray form into the cabinet space. The AdCN94.H7 vaccine suspension used for spray vaccination was prepared to contain 1.1 × 1010 ifu/ml. Group A (n = 15) was sprayed once (8 ml) and group B (n = 15) received three times the dose of group A (24 ml) and a boost (8 ml) on day 16 of age. Blood samples were obtained from the wing vein and tear samples were collected as described (25) on days 10, 21, 30, and 42 postvaccination. Serum antibodies were evaluated by HI test as described above against the A/tk/OR/71 (H7N3) AI strain. Local specific immunoglobulin A (IgA) in tear fluids was assessed by enzyme-linked immunosorbent assay (ELISA) mainly as previously described for antibodies to other viruses (28). In brief, ELISA tests were performed in flat-bottomed 96-well immunoplates (NUNC, Rothskilde, Denmark) coated with a 1:10 dilution of A/tk/OR/71 (H7N3) virus stock (hemagglutination titer of 10 log2) overnight at 4 C. Wells were blocked with phosphate-buffered saline (PBS) containing 1% bovine serum albumin and 0.05% Tween 20. A 1:10 dilution of individual tear samples was added and incubated for 60 min at room temperature (25 C). AI-specific antibody detection was performed using horseradish peroxidase (HRP)-conjugated goat anti-chicken IgA (Gallus Immunotech Inc., Fergus, Canada). Between each step, plates were washed five times with PBS-Tween 20 (0.05%). Tetramethylbenzidine (TMB) substrate was used to detect enzymatic activity. Optical density (OD) values were determined at 650 nm.
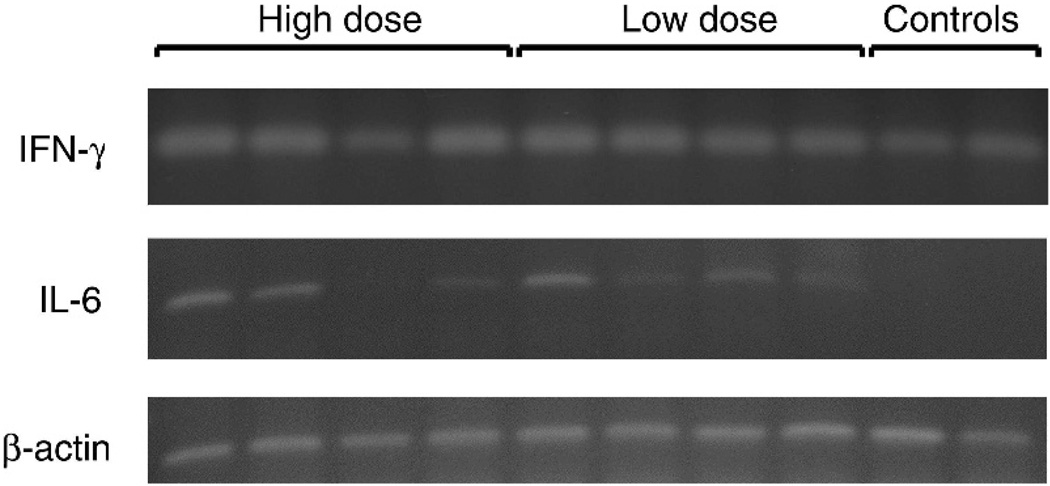
We hypothesized that aerosolized Ad5 particles would likely target the HG and induce a local immune response due to ocular exposure. Four birds of each vaccinated group were separated and spray-boosted with AdCN94.H7 on day 38 of age (4 days prior to termination of the experiment) for cytokine analyses. IFN-γ and IL-6 RNA was detected by reverse transcriptase–PCR (RT-PCR) on HG-derived lymphocytes in these chickens (4 days after spray immunization with AdCN94.H7). Total RNA was isolated from the HG of chickens using Tri-reagent (Molecular Research Inc., Cincinnati, OH) according to the manufacturer’s protocols. One microgram of total RNA was reverse-transcribed using random hexamers and amplified by 32 PCR cycles of 94 C, 1 min.; 58 C, 1 min to detect expression of the chicken cytokines IFN-γ and IL-6 and the housekeeping gene β-actin. The forward primer and the reverse primer were designed in Vector-NTI and are listed in Table 1. The resulting amplicon was separated on a 2.0% agarose gel and stained with ethidium bromide. The bands were visualized following ultraviolet exposure (Fotodyne Incorporated, Hartland, WI) and analyzed by ImageQuant software.
Table 1.
Forward and reverse primers (designed in Vector-NTI) and amplicon sizes for IFN-γ,A IL-6,B and the housekeeping gene β-actin are shown. RNA was detected by RT-PCRC on lymphocytes derived from the HGs of chickens after AdCN94.H7 spray immunization.
| Cytokine | Sense primer | Anti-sense primer | Amplicon |
|---|---|---|---|
| IFN-γ | GTCATTCAGATGTAGCTGACGGTGGA | CGCCATCAGGAAGGTTGTTTTTCA | 201 bp |
| IL-6 | GACGAGGAGAAATGCCTGACGAAG | CCACTTCATCGGGATTTATCACCATC | 175 bp |
| β-actin | GCCATCTTTCTTGGGTATGGAGTCCT | GGTGGGGCAATGATCTTGATTTTCA | 210 bp |
IFN-γ = interferon γ.
IL-6 = interleukin 6.
RT-PCR = reverse transcriptase polymerase chain reaction.
Experiment 3. Determine if RCA-free recombinant Ad vectors encoding the H5 HA elicits an antibody response against the expressed AI HA in pigs
Topical and intramuscular vaccine delivery routes were evaluated. Sixteen piglet weaners were identified by ear marks and divided into three groups in fence-separated pens within the same room. Two groups of piglets (each n = 6) were vaccinated with AdTW68.H5 at 5 wk of age. A third group of four pigs served as unvaccinated control. In group 1 each of six piglet weaners was vaccinated intramuscularly in the neck muscle (up to 70 mm behind the base of the ear using an 18-gauge 1-inch needle) with 1 ml of vaccine construct containing 109 ifu of AdTW68.H5. In group 2, six weaners were vaccinated intranasally at 5 and 7 wk of age. Nasal vaccination was performed with a 1-ml syringe. The syringe (without needle) was used to squirt 1 ml AdTW68.H5 containing 1010 ifu/ml into each nostril. Blood samples (from the jugular vein) as well as samples of nasal and eye secretions (nasal and eye swabs) were obtained from all animals for AI-specific antibody detection by ELISA at 7 days postvaccination and at 7-day intervals through day 42 postvaccination. Serum antibodies were evaluated by HI test as described above using 4 HA units of the A/tk/WI/68 (H5N9) AI strain. Sera of vaccinated pigs were also tested by ELISA against the same AI strain. In brief, flat-bottomed 96-well immunoplates (NUNC) were coated with a 1:10 dilution of A/tk/WI/68 (H5N9) virus stock (hemagglutination titer of 8 log2) overnight at 4 C. Wells were blocked as described above. A 1:100 dilution of individual serum samples was added and incubated for 60 min at room temperature (25 C). Detection of pig immunoglobulin G (IgG) was performed with a goat anti-pig IgG (Innovative Research Inc., Novi, MI) followed by rabbit anti-goat IgG HRP conjugate (Innovative Research Inc., Novi, MI) both antibodies diluted 2000-fold. TMB substrate was used to detect enzymatic activity. ODs were determined at 650 nm. Serum samples of pigs preimmunization as well as unvaccinated controls were also tested by ELISA for antibodies possibly binding to Ad5. ELISAs for Ad-specific antibodies were performed as described above with modifications previously reported (30). In brief, 96-well immunoplates were coated with 2 × 108 ifu of heat-inactivated Ad5 and blocked. Serial twofold dilutions were made of each serum sample starting at a 1:16 dilution. Detection of pig IgG was performed as described above. The endpoint titers were defined by the highest serum dilution with an OD405 above 0.100.
Nasal and eye swabs were obtained from each piglet for AI-specific antibody detection by ELISA. Swabs were placed in tubes containing 1 ml of PBS and vortexed. Samples were tested for specific IgA and IgG by ELISA. ELISA tests were performed mainly as described above but samples were diluted 1:10. IgG detection was performed as described above and IgA detection was performed using an anti-pig IgA goat antibody (Innovative Research Inc.).
RESULTS
Experiment 1
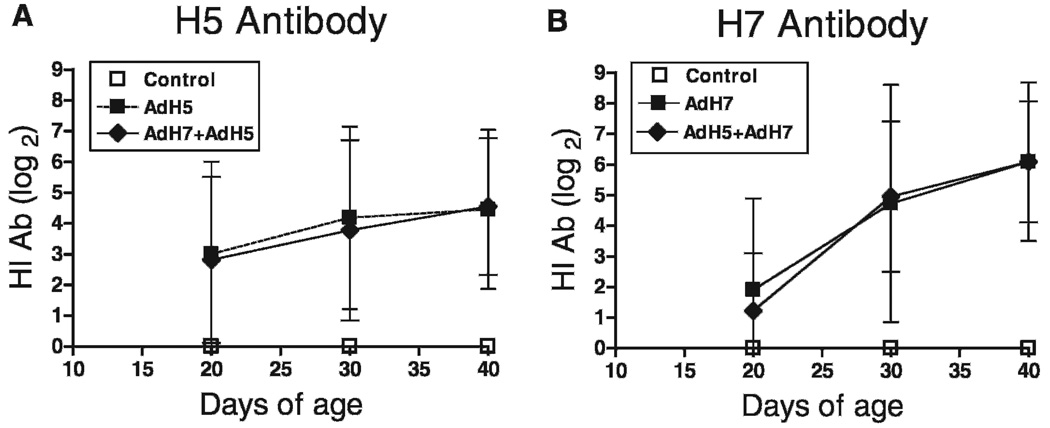
As seen in Fig. 1 bivalent in ovo vaccination with AdTW68.H5 and AdCN94.H7 elicited antibody responses against both expressed AI HAs. On day 20 posthatch similar (P > 0.05) mean H5 antibody titers of ~3 log2 were detected in chickens inoculated with either the monovalent or bivalent vaccines. H5 antibody levels continued to rise in both chicken groups achieving mean values of ~4.5 log2 on day 40 of age (Fig. 1A). On the other hand, H7 antibody levels achieved HI titers of ~2 log2 on day 20 posthatch (Fig. 1B). Similarly, no significant differences (P > 0.05) were detected between chickens vaccinated with AdCN94.H7 only and chickens vaccinated with both constructs simultaneously. H7 antibody levels achieved a maximum (~6 log2) on day 40 of age. On day 20 of age not only were H5 antibody levels higher than H7 antibody levels in all groups but also the number of birds showing detectable antibodies in chickens vaccinated with the monovalent and the bivalent vaccines differed: for AdTW68.H5 only, 7/11 (63.6%) were positive for H5 antibodies; for AdCN94.H7 only, 4/11 (36.3%) were positive for H7 antibodies; for the bivalent group H5 antibodies, 15/22 (68.1%) were positive, and bivalent group H7 antibodies, 7/22 (31.8%) were positive. There was a significant difference (Fisher’s exact test) for H5 and H7 detectable antibodies in chickens inoculated with the bivalent vaccine (P < 0.03) on day 20 of age. This difference can be explained by the lower dose of AdCN94.H7 used at the time of vaccination (5.0 × 107ifu AdCN94.H7 vs. 5.0 × 109 ifu AdTW68.H5). However, when comparing all antibody levels for H5 and H7 throughout the experimental period no significant differences (P > 0.05) were detected by ANOVA. At the termination of the experiment on day 40 of age, 9/11 (81%) of the chickens vaccinated with AdTW68.H5 only were positive for H5 antibodies, 10/11 (90%) vaccinated with AdCN94.H7 only were positive for H7 antibodies, 21/22 (95%) chickens vaccinated dually (AdCN94.H7 + AdTW68.H5) were positive for H5 antibodies and 22/22 (100%) were positive for H7 antibodies. Unvaccinated controls maintained a negative antibody status throughout the experimental period and were significantly different from vaccinated chickens (P < 0.01) throughout the experimental period.
Fig. 1.
In ovo vaccination (day 18 of embryonation) with AdTW68.H5 and AdChNY.H7 elicited serum HI antibody responses against both expressed AI HAs. (A) HI (H5) antibodies (mean ± SD) to A/tk/WI/68 (H5N9) in chickens (n = 12) vaccinated with AdTW68.H5 alone (200 µl/egg containing 5.0 × 109 ifu) or in chickens (n = 24) vaccinated with AdTW68.H5 (same dose as above) in combination with AdCN94.H7 (200 µl/egg containing 5.0 × 107 ifu). (B) HI (H7) antibodies (mean ± SD) to A/Tk/OR/71 (H7N3) in chickens (n = 12) vaccinated with AdCN94.H7 alone or in chickens (n = 24) vaccinated with AdTW68.H5 in combination with AdCN94.H7 (same doses as above). All control birds produced no measurable AI antibody titers.
Experiment 2
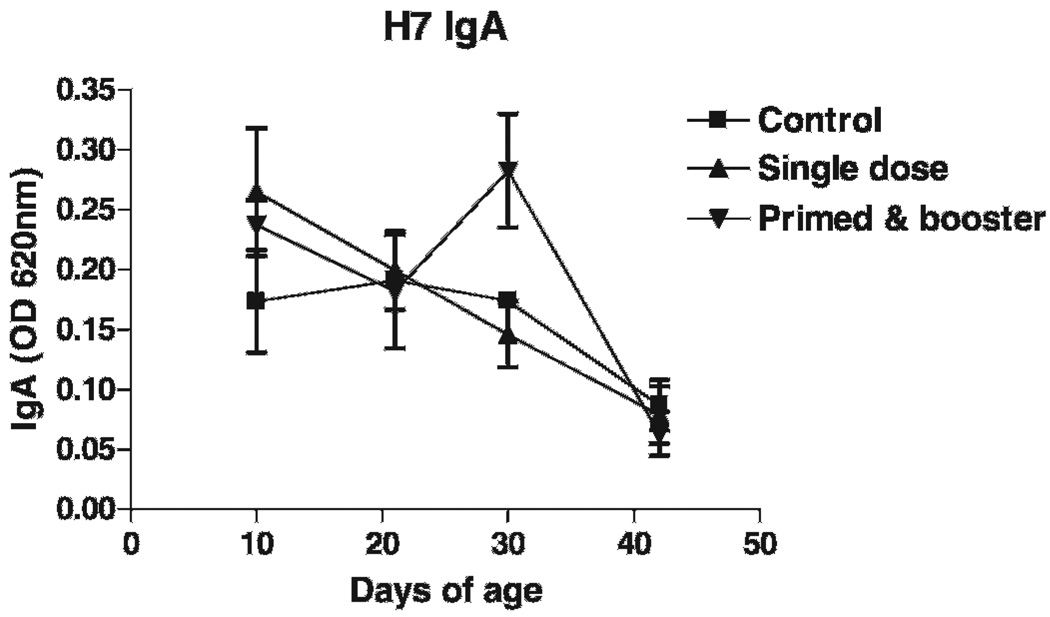
Spray-vaccinated chickens did not show any signs of distress in the 24-hr period following vaccine administration. No antibody to H7 were detected by HI testing against A/tk/OR/71 (H7N3) throughout the experimental period either in chickens vaccinated with a low dose or in chickens vaccinated three times with the dose. As seen in Fig. 2 both spray-vaccinated chicken groups showed higher specific IgA levels in tear fluids on day 10 of age (9 days postvaccination). IgA levels declined with time in singly vaccinated chickens. Chickens receiving a booster vaccination on day 16 of age showed an increase of specific IgA on day 30 of age.
Fig. 2.
ELISA IgA levels in chickens vaccinated with AdCN94.H7 by spray at 1 day of age using a commercial spray cabinet. Group “Single dose” (n = 15) was sprayed once with 8 ml of virus suspension containing 1.1 × 1010 ifu/ml. Group “Primed & booster” (n = 15) received three times the dose of group A (24 ml) and a boost (8 ml) on day 16 of age. Spray-vaccinated chickens showed higher specific IgA levels in tear fluids on day 10 of age (9 days postvaccination). IgA levels declined with time in singly vaccinated chickens. Chickens receiving a booster vaccination on day 16 of age showed an increase of specific IgA on day 30 of age. Unvaccinated controls (n = 15) were negative for specific IgA throughout the experimental period.
Cytokine analyses on HG-derived lymphocytes after spray immunization with AdCN94.H7 are shown in Fig. 3. As seen in this figure IFN-γ was detected in both controls and vaccinated birds and did not differ significantly between the control and vaccinated groups. The increase of IFN-γ in the HG in the control group has been previously observed (van Ginkel, pers. obs.) and seemed to occur around 10–14 days posthatch, when maternal antibodies in the chicks decline. IL-6 was not detected in control chickens but both chickens initially receiving a low dose or a high dose displayed similar levels of IL-6 in the HG. No significant differences were detected between the bands by ImageQuant software.
Fig. 3.
IFN-γ and IL-6 RNA by RT-PCR on lymphocytes derived from the HGs 4 days after spray booster vaccination with AdCN94.H7. IFN-γ was detected in both controls (n = 2) and vaccinated birds without significant differences. Chickens initially receiving a low (n = 4) or high (n = 4) dose of vaccine displayed similar levels of IL-6 in the HG as determined by ImageQuant analysis. IL-6 was not detected in unvaccinated control chickens. β-actin gene was used as the housekeeping gene.
Experiment 3
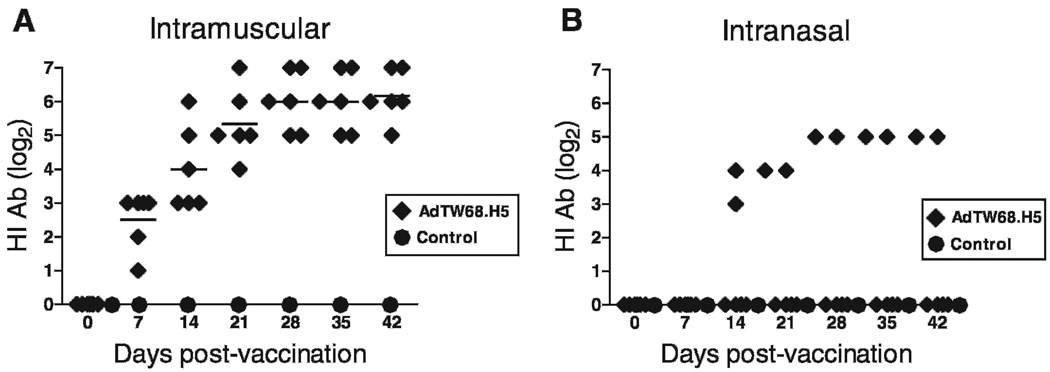
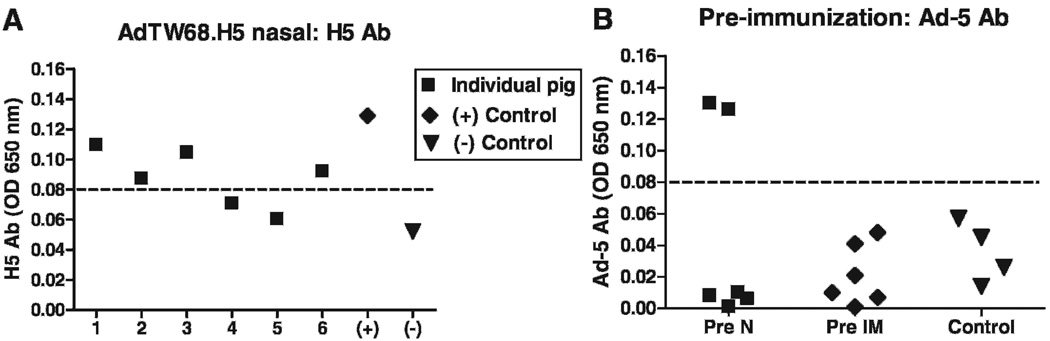
All pigs were bright, alert, and responsive throughout the experimental period. No changes were detected at the injection site. All pigs (6/6) vaccinated intramuscularly with AdTW68.H5 were positive for HI antibodies against the A/tk/WI/68 (H5N9) AI strain as soon as 1 wk after vaccination. As seen in Fig. 4A antibody levels continued to rise achieving a plateau on 28 days postvaccination (mean = 6.2 [SE = 0.3] log2). This antibody level was maintained through day 42 postvaccination. On the other hand, 2/6 pigs vaccinated intranasally developed antibody levels (5 log2) to the H5 HA (Fig. 4B). The rest of the pigs in this group (four pigs) maintained a negative HI antibody status throughout the experimental period. ELISA results were expressed as OD values because of unavailability of a positive control, i.e., a standard serum and/or mucosal secretion from pigs immunized with RCA-free adenovirus vector. Animals were considered positive for antibodies when their OD values were higher than the mean of the unvaccinated controls plus three times the SD. All pigs vaccinated intramuscularlly were positive for antibodies to A/tk/WI/68 by ELISA (not shown). On the other hand, 4/6 pigs vaccinated intranasally were positive for specific IgG determined by ELISA (Fig. 5A). Interestingly, ELISA testing for antibodies reacting with Ad5 demonstrated two animals with such antibodies higher than the mean + 3 SD of all other piglets (Fig. 5B). All pigs were negative for IgG both in tear fluid and in nasal secretions (eye or nasal swabs; not shown). OD values obtained for specific IgA in tear and nasal secretions were low compared to OD values for serum IgG (Fig. 6A). At week 11 of age, three-fourths of the pigs showed IgA ELISA values higher than the mean of controls + 3 SD both in tear and nasal secretion. One pig showed IgA OD values that were markedly higher than controls both in tears and nasal secretion (Fig. 6B). All unvaccinated control pigs, which were separated only by a fence from vaccinated animals, maintained a negative antibody status to A/tk/WI/68 both by HI and ELISA throughout the experimental period.
Fig. 4.
Serum H5 antibodies induced by RCA-free recombinant Ad-vector encoding AI H5 (AdTW68.H5) vaccination in pure-breed Yorkshire piglet weaners. (A) Intramuscular vaccination (n = 6) performed at 5 wk of age with 1 ml of virus suspension containing 109 ifu of AdTW68.H5 in the neck muscle (~70 mm behind the base of the ear). (B) Intranasal vaccination (n = 6) performed at 5 and 7 wk of age with 1 ml of virus suspension containing 1010 ifu of AdTW68.H5 in each nostril. HI antibody titers were measured in chickens on day 7 and at 7 day intervals through 42 days of age. All naïve control piglets (n = 4) produced no measurable AI antibody titers.
Fig. 5.
Serum ELISA antibodies in Yorkshire piglet weaners. (A) H5 antibodies in pigs (n = 6) subjected to intranasal vaccination at 5 and 7 wk of age with 1 ml of virus suspension containing 1010 ifu of AdTW68.H5 in each nostril. Positive control serum (+) is a pool of sera from two pigs vaccinated intramuscularly in experiment 3. Negative control (−) is the mean OD value of unvaccinated controls. The baseline was established from the mean of unvaccinated controls plus three times their SD. (B) Antibodies reacting with Ad5 in each group of pigs prenasal (Pre-N) and pre-intramuscular (Pre-IM) immunizations: unvaccinated controls (n = 4), Pre-N (n = 6), and Pre-IM (n = 6). Two piglets within the Pre-N group showed ELISA antibody levels higher than the mean + 3 SD of all other animals.
Fig. 6.
Specific antibody levels determined by ELISA in Yorkshire piglet weaners vaccinated intranasally with AdTW68.H5 at 5 and 7 wk of age. (A) IgA in individual unvaccinated controls (n = 4) in tear fluid (Ctr-Tear) or nasal secretion (Ctr-N) and in individual intranasally vaccinated piglets (N-Tear; N-N) on week 10. (B) IgA in tear or nasal secretions on week 11. Baselines obtained from the mean OD values of unvaccinated controls plus three times the SD. IgA OD values in tear and nasal secretions were low compared to values for serum IgG (Fig. 5). Even though a few animals show OD values above the baseline, only one pig on week 11 seem to have developed a significant IgA response.
DISCUSSION
Because HPAI viruses belonging to the H5 or H7 types continue to threaten the world poultry industry and are zoonotic agents with pandemic potential for humans (29), it is crucial to adopt an immunization strategy with a broader antigen repertoire. Ad-vectored AI vaccines encoding either the AI virus H5 (AdTW68.H5) or H7 (AdCN94.H7) were successfully constructed (26,27) and the immune response was then evaluated in chickens vaccinated in ovo with either construct alone or in a combined vaccine regimen. Our results (Fig. 1) show that bivalent in ovo vaccination with AdTW68.H5 and AdChNY.H7 elicits antibody responses against both encoded AI HAs in chickens. From an applied perspective, this result is relevant because it demonstrates the feasibility of adopting an immunization strategy with a broad antigenic repertoire.
Spray vaccination with AdChNY.H7 in chickens induced weak specific IgA levels in the tear fluids and increased IL-6 expression in the HG, a cytokine secreted mainly by the TH2 type of T helper cells but also by monocytes, macrophages, and bone marrow cells. However, none of the birds developed specific HI antibodies. We have shown previously that chickens vaccinated by eye drop with an RCA-free recombinant Ad-vector expressing the H5 HA gene of the A/tk/WI/68 AI virus (AdTW68.H5) develop IgA and IgG antibody-secreting cells in the HGs specific for both H5 and the Ad as measured by enzyme-linked immunosorbent spot assay (ELISPOT). In addition ocular immunization also induced Ad5- and H5-specific antibodies in the systemic compartment (8). Thus, it seems that spray vaccination, as performed in this study, does not result in enough recombinant viruses reaching the ocular and nasal mucosa to allow elicitation of a strong immune response. In the current experiment we used a small droplet size (~130 microns) which allows even low concentrations of the vaccine virus to reach deeper in the respiratory tract when inhaled by the chickens. This is not a problem when the vaccine virus efficiently replicates in the respiratory tract as most attenuated respiratory viruses do. We showed previously that Ad-vectored vaccination follows a dose response kinetic, i.e., higher virus dosage induces not only higher antibody responses but also increased percentage of antibody-positive chickens. Higher doses likely result in greater numbers of host cells being transduced by the Ad vector and increased transgene expression (27). Thus, it is conceivable that devices and/or application methods able to deposit a high viral concentration on large mucosal surfaces will allow transduction of a greater number of epithelial cells and effective transgene expression. Spray application using for example an increased droplet size and/or spray application onto the chicken’s faces (e.g., at the time of feeding in cage layer chickens) might improve the immune responses.
Vaccination of pigs against AI is relevant because pigs could potentially facilitate genetic reassortment with human influenza A viruses to acquire efficient interhuman transmissibility (17). Ad5-vectored vaccines propagated in the 293 cell line have been shown to elicit a protective immune response against H3N2 swine influenza virus challenge (35). Ad5 vectors produced in human 293-derived cells are contaminated by RCA autonomously generated through homologous recombination between the Ad5 vector and the E1 region in the 293 genome (37). Intramuscular injection of Ad5 stocks containing substantial amounts of RCA induced severe inflammation and muscular atrophy in newborn rats whereas E1-defective recombinant Ad5 was not associated with pathogenicity (16). Because the ratio between RCA and recombinant Ad5 kept increasing in culture and a ratio of 1:40 had been detected after multiple passages owing to a replicative advantage of RCA (16), it is conceivable that large-scale production of Ad5 vectors in 293 cells may result in stocks containing RCA at a level beyond safety margin. Our results show that intramuscular vaccination with RCA-free Ad recombinant virus encoding the H5 of avian influenza elicits a strong antibody response in pigs. RCA-free Ad5-vectored AI vaccines produced in large scale in the PER.C6 cell line bypass the homologous recombination event for RCA generation. Intranasal application of the RCA-free Ad5-vectored vaccine resulted in only 2/6 pigs developing an antibody response. Interestingly, the antibody response in those two animals was strong (5 log2), i.e., similar as to the response seen after intramuscular injection (6 log2). We speculate that epithelial cells of the lamina propria of the turbinates were effectively transduced by the Ad5 vector resulting in a successful immune response. We see two possible explanations for the lack of vaccine coverage in the intranasally vaccinated piglets. One possible explanation for reduced vaccine coverage might be the mode of intranasal vaccine application used. Other intranasally delivered vaccines use a nasal applicator tip applied to the syringe. By closing the nostril with the applicator tip it is conceivable that the increased intranasal pressure facilitated vaccine aerosolization resulting in exposure of a wider surface of the pseudostratified columnar ciliated respiratory epithelium of the turbinates to vaccine antigen. Because we used a syringe without an applicator tip, we speculate that the lower intranasal pressure resulted in reduced vaccine aerosolization causing most of the vaccine to remain in the liquid state. Thus the vaccine was likely deposited onto the reduced surface of the inferior meatus. Thus, the use of nasal applicator tips might result in better vaccine coverage in this species. Previous reports indicate successful use of a needle-free device (Biojector 2000, Bioject, Tualatin, OR) for Ad-vectored vaccines in pigs (33). However, those results can not be compared with intranasal inoculation results because the needle-free devices force liquids through the skin depositing the medication subcutaneously. A second plausible explanation is that preexisting antibodies to Ad5 in the piglets that were vaccinated by the nasal route might have interfered with efficient Ad5 vaccine virus infection of mucosal cells. The ELISA results support this assumption because 4/6 of intranasally vaccinated pigs were positive for antibodies to AI virus and at the same time the inverse number (2/6) of pigs were positive for antibodies binding to Ad5 (Fig. 5A and B) at preimmunization.
ACKNOWLEDGMENTS
We wish to thank Cassandra Breedlove and Stephen Gulley for outstanding technical assistance and Ray Feng for mass production of the constructs. We also wish to thank Dr. Rodrigo Gallardo and Mr. Brian Anderson for their great assistance on pig sampling and husbandry. This work was supported by USDA AICAP 2007-35203-18070 and by NIH-SBIR 2-R44-AI-068285-02.
Abbreviations
- Ad
adenovirus
- Ad5
adenovirus serotype 5
- AI
avian influenza
- ANOVA
analysis of variance
- AU-SREC
Auburn University Swine Research and Education Center
- DIVA
differentiation between infected and vaccinated animals
- ELISA
enzyme-linked immunosorbent assay
- HA
hemagglutinin
- HG
Harderian gland
- HI
hemagglutination inhibition
- HP
highly pathogenic
- HRP
horseradish peroxidase
- IgA
immunoglobulin A
- IgG
immunoglobulin G
- IM
intramuscular
- IFN-γ
interferon gamma
- IL-6
interleukin 6
- ifu
infectious units
- OD
optical density
- PBS
phosphate-buffered saline
- RCA
replication-competent adenovirus
- RT-PCR
reverse transcriptase–PCR
- SPF
specific-pathogen-free
- TMB
tetramethylbenzidine
REFERENCES
- 1.Albini B, Wick G, Rose E, Orlans E. Immunoglobulin production in chicken Harderian glands. Int. Arch. Allergy Appl. Immunol. 1974;47:23–34. doi: 10.1159/000231198. [DOI] [PubMed] [Google Scholar]
- 2.Bienenstock J, Gauldie J, Perey DY. Synthesis of IgG, IgA, IgM by chicken tissues: immunofluorescent and 14C amino acid incorporation studies. J. Immunol. 1973;111:1112–1118. [PubMed] [Google Scholar]
- 3.Burns RB. Specific antibody production against a soluble antigen in the Harderian gland of the domestic fowl. Clin. Exp. Immunol. 1976;26:371–374. [PMC free article] [PubMed] [Google Scholar]
- 4.Capua I, Marangon S, Dalla Pozza M, Santucci U. Vaccination for avian influenza in Italy. Vet. Rec. 2000;147:751. [PubMed] [Google Scholar]
- 5.Choi YK, Nguyen TD, Ozaki H, Webby RJ, Puthavathana P, Buranathal C, Chaisingh A, Auewarakul P, Hanh NT, Ma SK, Hui PY, Guan Y, Peiris JS, Webster RG. Studies of H5N1 influenza virus infection of pigs by using viruses isolated in Vietnam and Thailand in 2004. J. Virol. 2005;79:10821–10825. doi: 10.1128/JVI.79.16.10821-10825.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Crawley JF. Present status of infectious bronchitis immunization. Proc. Am. Vet. Med. Assoc. 1955:343–350. [Google Scholar]
- 7.Davelaar FG, Noordizij A, van der Donk JA. A study on the synthesis and secretion of immunoglobulins by the Harderian gland of the fowl after eyedrop vaccination against infectious bronchitis at 1-day-old. Avian Pathol. 1982;11:63–79. doi: 10.1080/03079458208436082. [DOI] [PubMed] [Google Scholar]
- 8.Ginkel FWv, Tang DC, Gulley SL, Toro H. Induction of mucosal immunity in the avian Harderian gland with a replication deficient Ad-5 vector expressing avian influenza H5 hemagglutinin. Develop. Comp. Immunol. 2009;33:28–34. doi: 10.1016/j.dci.2008.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B. A simplified system for generating recombinant adenoviruses. Proc. Natl. Acad. Sci. U. S. A. 1998;95:2509–2514. doi: 10.1073/pnas.95.5.2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hilleman MR. Realities and enigmas of human viral influenza: pathogenesis, epidemiology and control. Vaccine. 2002;20:3068–3087. doi: 10.1016/s0264-410x(02)00254-2. [DOI] [PubMed] [Google Scholar]
- 11.Jungherr EL, Luginbuhl RE. Report on poultry respiratory disease control program for 1949 and suggestions for 1950. Proc. 22nd Annual Conference of Laboratory Workers Pullorum Disease Control; Burlington, VT. 1949. [Google Scholar]
- 12.Konz JO, Livingood RC, Bett AJ, Goerke AR, Laska ME, Sagar SL. Serotype specificity of adenovirus purification using anion-exchange chromatography. Hum. Gene Ther. 2005;16:1346–1353. doi: 10.1089/hum.2005.16.1346. [DOI] [PubMed] [Google Scholar]
- 13.Lee CW, Senne DA, Suarez DL. Generation of reassortant influenza vaccines by reverse genetics that allows utilization of a DIVA (Differentiating Infected from Vaccinated Animals) strategy for the control of avian influenza. Vaccine. 2004;22:3175–3181. doi: 10.1016/j.vaccine.2004.01.055. [DOI] [PubMed] [Google Scholar]
- 14.Lewis JA, Brown EL, Duncan PA. Approaches to the release of a master cell bank of PER.C6 cells; a novel cell substrate for the manufacture of human vaccines. Dev. Biol. (Basel) 2006;123:165–176. [PubMed] [Google Scholar]
- 15.Lipatov AS, Kwon YK, Sarmento LV, Lager KM, Spackman E, Suarez DL, Swayne DE. Domestic pigs have low susceptibility to H5N1 highly pathogenic avian influenza viruses. PLoS Pathog. 2008;4:e1000102. doi: 10.1371/journal.ppat.1000102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lochmuller H, Jani A, Huard J, Prescott S, Simoneau M, Massie B, Karpati G, Acsadi G. Emergence of early region 1–containing replication-competent adenovirus in stocks of replication-defective adenovirus recombinants (delta E1 + delta E3) during multiple passages in 293 cells. Hum. Gene Ther. 1994;5:1485–1491. doi: 10.1089/hum.1994.5.12-1485. [DOI] [PubMed] [Google Scholar]
- 17.Ludwig S, Stitz L, Planz O, Van H, Fitch W, Scholtissek C. European swine virus as a possible source for the next influenza pandemic? Virology. 1995;212:555–561. doi: 10.1006/viro.1995.1513. [DOI] [PubMed] [Google Scholar]
- 18.Park MS, Steel J, Garcia-Sastre A, Swayne D, Palese P. Engineered viral vaccine constructs with dual specificity: avian influenza and Newcastle disease. Proc. Natl. Acad. Sci. U. S. A. 2006;103:8203–8208. doi: 10.1073/pnas.0602566103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sharma JM, Burmester BR. Resistance to Marek’s disease at hatching in chickens vaccinated as embryos with the turkey herpesvirus. Avian Dis. 1982;26:134–149. [PubMed] [Google Scholar]
- 20.Shi Z, Zeng M, Yang G, Siegel F, Cain LJ, Van Kampen KR, Elmets CA, Tang DC. Protection against tetanus by needle-free inoculation of adenovirus-vectored nasal and epicutaneous vaccines. J. Virol. 2001;75:11474–11482. doi: 10.1128/JVI.75.23.11474-11482.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Suarez DL, Schultz-Cherry S. The effect of eukaryotic expression vectors and adjuvants on DNA vaccines in chickens using an avian influenza model. Avian Dis. 2000;44:861–868. [PubMed] [Google Scholar]
- 22.Swayne DE. Vaccines for List A poultry diseases: emphasis on avian influenza. Dev. Biol. (Basel) 2003;114:201–212. [PubMed] [Google Scholar]
- 23.Swayne DE, Beck JR, Kinney N. Failure of a recombinant fowl poxvirus vaccine containing an avian influenza hemagglutinin gene to provide consistent protection against influenza in chickens pre-immunized with a fowl pox vaccine. Avian Dis. 2000;44:132–137. [PubMed] [Google Scholar]
- 24.Swayne DE, Senne DA, Beard CW. Influenza. In: Swayne DE, Glisson JR, Jackwood MW, Pearson JE, Reed WM, editors. Isolation and identification of avian pathogens. 4th ed. Kennett Square, PA: American Association of Avian Pathologists; 1998. pp. 150–155. [Google Scholar]
- 25.Toro H, Lavaud P, Vallejos P, Ferreira A. Transfer of IgG from serum to lacrimal fluid in chickens. Avian Dis. 1993;37:60–66. [PubMed] [Google Scholar]
- 26.Toro H, Tang DC, Suarez DL, Sylte MJ, Pfeiffer J, van Kampen KR. Protective avian influenza in ovo vaccination with non-replicating human adenovirus vector. Vaccine. 2007;25:2886–2891. doi: 10.1016/j.vaccine.2006.09.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Toro H, Tang DC, Suarez DL, Zhang J, Shi Z. Protection of chickens against avian influenza with non-replicating adenovirus-vectored vaccine. Vaccine. 2008;26:2640–2646. doi: 10.1016/j.vaccine.2008.02.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Toro H, van Santen VL, Li L, Lockaby SB, van Santen E, Hoerr FJ. Epidemiological and experimental evidence for immunodeficiency affecting avian infectious bronchitis. Avian Pathol. 2006;35:1–10. doi: 10.1080/03079450601028811. [DOI] [PubMed] [Google Scholar]
- 29.Ungchusak K, Auewarakul P, Dowell SF, Kitphati R, Auwanit W, Puthavathana P, Uiprasertkul M, Boonnak K, Pittayawonganon C, Cox NJ, Zaki SR, Thawatsupha P, Chittaganpitch M, Khontong R, Simmerman JM, Chunsutthiwat S. Probable person-to-person transmission of avian influenza A (H5N1) N. Engl. J. Med. 2005;352:333–340. doi: 10.1056/NEJMoa044021. [DOI] [PubMed] [Google Scholar]
- 30.van Ginkel FW, Liu C-J, Simecka JW, Dong J-Y, Greenway T, Frizzell RA, Kiyono H, McGhee JR, Pascual DW. Intratracheal gene delivery with adenoviral vector induces elevated systemic IgG and mucosal IgA antibodies to adenovirus and b-galactosidase. Hum. Gene Ther. 1995;6:895–903. doi: 10.1089/hum.1995.6.7-895. [DOI] [PubMed] [Google Scholar]
- 31.Van Reeth K. Avian and swine influenza viruses: our current understanding of the zoonotic risk. Vet. Res. 2007;38:243–260. doi: 10.1051/vetres:2006062. [DOI] [PubMed] [Google Scholar]
- 32.Wakenell PS, Bryan T, Schaeffer J, Avakian A, Williams C, Whitfill C. Effect of in ovo vaccine delivery route on herpesvirus of turkeys/SB-1 efficacy and viremia. Avian Dis. 2002;46:274–280. doi: 10.1637/0005-2086(2002)046[0274:EOIOVD]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 33.Wesley RD, Lager KM. Evaluation of a recombinant human adenovirus-5 vaccine administered via needle-free device and intramuscular injection for vaccination of pigs against swine influenza virus. Am. J. Vet. Res. 2005;66:1943–1947. doi: 10.2460/ajvr.2005.66.1943. [DOI] [PubMed] [Google Scholar]
- 34.Wesley RD, Lager KM. Overcoming maternal antibody interference by vaccination with human adenovirus 5 recombinant viruses expressing the hemagglutinin and the nucleoprotein of swine influenza virus. Vet. Microbiol. 2006;118:67–75. doi: 10.1016/j.vetmic.2006.07.014. [DOI] [PubMed] [Google Scholar]
- 35.Wesley RD, Tang M, Lager KM. Protection of weaned pigs by vaccination with human adenovirus 5 recombinant viruses expressing the hemagglutinin and the nucleoprotein of H3N2 swine influenza virus. Vaccine. 2004;22:3427–3434. doi: 10.1016/j.vaccine.2004.02.040. [DOI] [PubMed] [Google Scholar]
- 36.World Organization for Animal Health (OIE), Food and Agriculture Organization of the United Nations (FAO) Recommendations of the OIE/FAO International Scientific Conference on Avian Influenza [Internet]; Paris, France: [modified 2005 Apr 7–8 April]. Available from: www.oie.int/eng/avian_influenza/OIE_FAO_Recom_05.pdf. [Google Scholar]
- 37.Zhu J, Grace M, Casale J, Chang AT, Musco ML, Bordens R, Greenberg R, Schaefer E, Indelicato SR. Characterization of replication-competent adenovirus isolates from large-scale production of a recombinant adenoviral vector. Hum. Gene Ther. 1999;10:113–121. doi: 10.1089/10430349950019246. [DOI] [PubMed] [Google Scholar]