Abstract
Individuals with chronic craniofacial pain experience symptoms that are consistent with central sensitization. In fact, central sensitization may constitute the major disease process in these conditions, particularly if the original injury has healed or the condition is idiopathic. To understand central sensitization we have developed a conjugate of substance P and cholera toxin (SP-CTA). SP-CTA is selectively taken up by cells that express neurokinin receptors. Twenty four hours following intracisternal administration of SP-CTA wild type rats and mice demonstrated signs of persistent background nociception, but when tested for facial cold sensitivity they did not differ from controls. However, treating the SP-CTA injected animals with naloxone exposed cold hypersensitivity in the face. Mu opioid receptor knockout mice treated with SP-CTA demonstrated hypersensitivity without naloxone treatment. These findings suggest that central sensitization leads to activation of an endogenous opioid system. The data also demonstrate that the intracisternal administration of SP-CTA in rodents is a useful model for studying central sensitization as a disease process without having to induce a peripheral injury.
Keywords: neurokinin receptors, substance P, central sensitization, cholera toxin, endogenous opioids
Introduction
Craniofacial pain comprises a host of conditions ranging from migraine headache to neuralgias and toothaches. It is estimated that most individuals will experience persistent craniofacial pain at some point in their lives. If the pain does not resolve within a few months the condition is considered chronic. Approximately 10 percent of adults and as many as 50 percent of the elderly have chronic craniofacial pain 27,26,33. Many of these chronic pain conditions are not well understood and therapies are based mainly on trial and error rather than empirical evidence. Thus novel models of craniofacial pain are needed for the development of rational therapies.
As a normal part of the healing process following a disease or injury the nociceptive pathways are sensitized. Sensitization alters the input/output relationship of nociceptive neurons resulting in nociceptive and non-nociceptive signals being amplified. Behaviorally this amplification is expressed as hyperalgesia, allodynia and referred pain. Thus minimally painful stimuli or normally innocuous stimuli, such as a cool breeze, can become excruciatingly painful. This peripheral and central sensitization aids the healing process by encouraging the person or animal to protect the damaged area. The sensitization usually reverses when the tissue has been repaired 22. Occasionally, however, central sensitization does not reverse when the tissue is healed or central sensitization occurs without an apparent injury as in idiopathic pain. When this happens central sensitization becomes the primary disease. In these instances an understanding of the molecular processes leading to central sensitization and the failure of these processes to reverse would greatly enhance our ability to treat the disease.
Several groups previously demonstrated that activation of serine/threonine kinases in dorsal horn neurons is a critical step in the evolution of central sensitization 2,18,58,56,57,24,45,55,60,61,6,7,40,53. The activated kinases phosphorylate several proteins leading to enhanced synaptic transmission and altered gene expression. Toward the goal of understanding central sensitization as a disease process we have developed a conjugate of substance P and the catalytic subunit of cholera toxin (SP-CTA) 5. SP-CTA is taken up by cells that express neurokinin-1 or neurokinin-2 receptors and produces an increase in intracellular levels of cAMP by ADP-ribosylating the stimulatory g-protein Gs, which then activates adenylate cyclase 16. By elevating cAMP levels the conjugate stimulates the serine/threonine kinase Protein Kinase A (PKA). Activation of PKA was found to induce central sensitization 20,44. Previous work demonstrated that when SP-CTA was injected into the intrathecal space of the lumbar spinal cord of rats it induces the phosphorylation of cAMP Response Element Binding protein (CREB) and the expression of c-Fos in dorsal horn neurons. These findings are consistent with previous studies on injury induced central sensitization 13,28. Behaviorally, intrathecal SP-CTA produces hypersensitivity to thermal stimuli in the hind paws 5. These findings indicated that intrathecal SP-CTA treatment produces central sensitization. Interestingly, with higher intrathecal doses of SP-CTA hypersensitivity to peripheral input is suppressed even though the animals still appear agitated by the treatment. Since spinothalamic tract neurons express neurokinin-1 receptors 12 our findings suggested that the SP-CTA activated neurons are transmitting nociceptive signals rostrally, which in turn stimulates descending inhibition. The net result is that there is a suppression of peripheral nociceptive input while a tonic background nociception is maintained by the SP-CTA sensitized spinothalamic tract neurons. These findings indicate that SP-CTA is a useful tool for examining central sensitization and the resulting compensatory mechanisms. In an effort to understand central sensitization as a disease process in the head and face the current study used SP-CTA to sensitize the Trigeminal Nucleus Caudalis (TNC) in rats and mice.
Materials and Methods
Animals
Male hairless Sprague–Dawley rats (200–350g) (Charles River, Raleigh, NC), C57BL/6J mice, Balb/c mice, Neurokinin-1 receptor knockout mice (NK1-KO) from a line originally derived by Bozic et al. 1, MOR knockout mice on a C57BL/6J background and hairless SKH1-Hrhr mice (25–40g, both sexes) (The Jackson Laboratories, Bar Harbor, ME) were used for these studies. Animals were maintained in AAALAC approved facilities with 12 hour light/dark cycles. The animals were given free access to standard rodent chow and water. All experiments were reviewed and approved by the University of Florida Institutional Animal Care and Use Committee and conformed to the standards of the International Association for the Study of Pain’s guidelines for animal research.
Preparation of SP-CTA
The conjugate of substance P and the A subunit of cholera toxin (SP-CTA) was synthesized as previously described 5(Advanced Targeting Systems, San Diego, CA). Briefly, substance P (Sigma inc., St. Louis, MO) was linked to the A subunit of cholera toxin (List Biological Laboratories inc., Campbell, CA) using Pierce’s (Pierce Chemical Co., Rockford, IL) Sulfosuccinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (Sulfo-SMCC) cross linking agent. The manufacturer’s directions were followed for the cross linking procedure. The resulting product had an amine from either the N-terminal arginine or lysine of substance P covalently bound to one of two cysteine residues in the C-terminal region of the cholera toxin A subunit. The final product was washed with phosphate buffered saline (PBS) and concentrated using Centricon filters with a 10kd molecular weight cutoff (Millipore, Billerica, MA). Protein concentration was determined using the BCA Assay 46 (Pierce Chemical Co., Rockford, IL). This conjugate was previously demonstrated to be taken up selectively by cells that express neurokinin-1 (NK-1) or 2 (NK-2) receptors 5. Cells that did not express either of these two receptors were not influenced by SP-CTA.
Intracisternal Injections
Rats and mice were anesthetized with isoflurane (1 to 3%) and injections were made into the cisterna magna via percutaneous puncture through the atlanto-occipital membrane using a Hamilton syringe and 30 gauge needles. SP-CTA was diluted in PBS and injected in a volume of 10μl (rats) or 1μl (mice) over the course of three minutes. Vehicle treated animals received the indicated volume of PBS without SP-CTA. The animals were then allowed to recover from the anesthesia.
Immunohistochemistry
Rats (N = 2) used for immunohistochemistry were allowed to recover from anesthesia for one hour following the intracisternal injections of SP-CTA (10 μg) and then euthanized with pentobarbital (100mg/kg, ip.). The one hour time point was chosen because the intracellular half life for the A subunit of cholera toxin is approximately 8 hours 15. The animals were then perfused transcardially with ice cold PBS followed by PBS with paraformaldehyde (4%). The brainstem was removed and post fixed overnight in 4% paraformaldehyde in PBS. The tissue was then cryoprotected in 30% sucrose, sectioned in a cryostat (20μ) and mounted on slides. The sections were blocked with 3% normal goat serum in PBS with 0.3% tween-20 for 60 minutes. The neurokinin-1 receptor (Zymed Laboratories inc., San Francisco, CA) and the cholera toxin A subunit (Sigma inc., St Louis, MO) primary antibodies were added to the blocking solution (1:500) and the sections were incubated for 24 hours at 4°C. The sections were then washed (3 × 10 mins) in PBS. Following the wash the sections were incubated for 1 hour in 3% normal goat serum/0.3% tween-20 in PBS with secondary antibodies that were coupled to a fluorescent label (1:1000) (Alexa Fluor 488 for NK-1 receptors and Alexa Fluor 594 for the cholera toxin A subunit, Invitrogen Corporation, Carlsbad, CA). Sections were washed (3 × 10 mins) in PBS and cover slipped with Prolong Gold anti-fade reagent (Invitrogen Corporation, Carlsbad, CA).
Western Blots
Two hours following the intracisternal injection of either PBS or SP-CTA brainstems were harvested from NK1-KO mice (N = 4) and wild type mice (N = 4). The brainstems were homogenized in 10 mls/g tissue of 50 mM tris buffer containing a protease inhibitor cocktail (Sigma, St Louis, MO) (4-(2-aminoethyl) benzenesulfonyl fluoride (AEBSF), E-64, bestatin, leupeptin, aprotinin, and sodium EDTA). After homogenization 30 μl of the suspension was diluted with 30 μl 2X electrophoresis buffer containing 250 mM Tris pH 6.8, 4% SDS, 10% glycerol, 0.006% bromophenol blue and 2 % mercaptoethanol. The solution was boiled for 5 minutes and centrifuged (16,000 g, 5 mins, 4°C). The supernatant was loaded onto SDS-Polyacrylamide gels (4 – 20% gradient) and electrophoresed. The running buffer for the electrophoresis contained 24 mM Tris base, 180 mM glycine and 0.1 % SDS. When the bromophenol blue had migrated to the bottom, the gel was removed and the proteins were transferred to Polyvinylidene fluoride (PVDF) membranes using a semi-dry transfer device. The transfer buffer was composed of 20% methanol, 48 mM Tris pH 9.2, and 39 mM glycine. The membrane was then placed in TTBS (20 mM Tris pH 7.6, 0.9% NaCl, and 0.05% Tween-80, pH 7.4) containing 5% non-fat dry milk for 1 hr. The primary antibody anti-cholera A (Sigma, St Louis, MO) (1:500) was added to the solution and the membrane was incubated overnight at 4°C. The membrane was washed with TTBS (3X) and secondary anti-rabbit antibodies (Sigma, ST Louis, MO) linked to horse radish peroxidase were added (1:1000). The solution was incubated at room temperature for 1 hour and the membrane washed with TTBS (3X). The bands were viewed using enhanced chemiluminescence and film.
Operant Facial Nociception Assay
Rats and mice were tested for facial nociception in an operant assay as previously described 30,32,29,31,39,38. Briefly, the animals were placed into a plastic box with an opening in one wall. On the edges of the opening were two aluminum tubes through which water was circulated at the desired testing temperature. Just outside the box a rodent water bottle containing a diluted solution of sweetened condensed milk (1:3 with water) was positioned so that the animals had to contact the temperature controlled tubes with their faces in order to obtain the milk solution. In animals with fur the animals’ cheeks were shaved under isoflurane anesthesia (1 to 3%) prior to testing. The animals were trained for 6 thirty minute sessions over the course of 2 weeks and then used for the experiments. For testing sessions the animals were food restricted for 6 to 12 hours and then placed in the apparatus. Rats were tested for 30 minutes and mice were tested for 15 minutes. Animal contacts with the thermal stimuli and with the watering bottle were monitored electronically, digitized and recorded on a computer. The data were then analyzed using a custom Lab View program, Excel and SPSS or Graphpad Prism statistical software.
Thermal Place Preference Assay
Thermal place preference was performed as previously described by Vierck and colleagues 54. Briefly, rats were randomly placed into one chamber of a box with two chambers. The chambers were separated by a wall with an opening in the center that was just large enough for the animals to comfortably walk through (8 cm wide × 10 cm high). In one chamber the floor was heated to 45°C and in the other chamber the floor was cooled to 15°C. The animals were then monitored for 900 seconds to determine which floor temperature they preferred. The location of the rats was monitored electronically and recorded on a computer.
Statistics
Data are expressed as the mean ± SEM. Behavioral data were analyzed using Analysis of Variance (ANOVA) followed by Dunnett’s post hoc test and two way ANOVAs followed by Bonferroni’s post hoc tests. Student’s paired t-tests were used when appropriate. An alpha ≤ 0.05 was considered significant.
Results
Localization of Intracisternally Injected SP-CTA
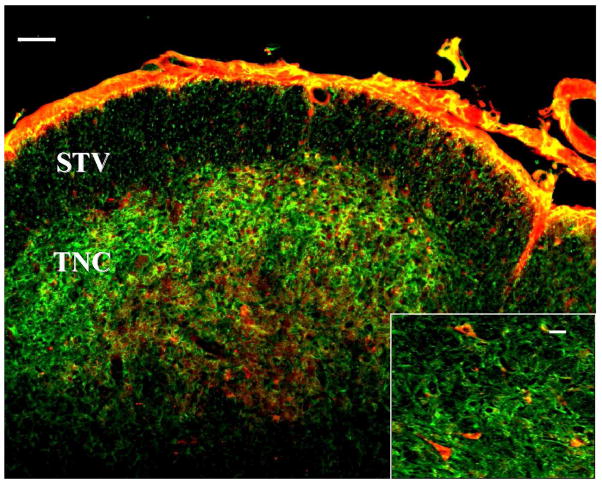
To determine if the Trigeminal Nucleus Caudalis (TNC) neurons that express neurokinin receptors take up SP-CTA immunohistochemistry was performed on tissue from rats (N = 2) that were treated with intracisternal injections of 10μg SP-CTA in 10 μl PBS one hour prior to tissue harvest. The brainstems of the rats were sectioned transversely (20μ). Brainstem sections were labeled with antibodies to both the NK-1 receptor and the A subunit of cholera toxin. Figure 1 demonstrates that neurons in the TNC that express NK-1 receptors (green) also take up SP-CTA (red). Dual labeling is viewed as either orange or yellow in the images. The area immediately medial to the surface of the brainstem is the spinal tract of V, which was not significantly labeled by antibodies to NK-1 receptors or cholera toxin. These findings indicate that intracisternally injected SP-CTA is taken up by TNC neurons that express NK-1 receptors.
Figure 1.
Localization of SP-CTA in rat brainstem following intracisternal injection. SP-CTA (10μg) was injected into the cisterna magna via percutaneous puncture (N = 2). One hour following the injections the brainstems were harvested and sectioned (20μ) on a cryostat and labeled for the NK-1 receptor and the A subunit of cholera toxin. The large image demonstrates immunohistochemical co-localization of NK-1 receptors (green) and SP-CTA (red)(bar = 50μ). Note that there is a significant amount of SP-CTA that still remained on the surface of the tissue at this time point. Labels: STV - Spinal Tract of V; TNC – Trigeminal Nucleus Caudalis. The insert illustrates individual TNC neurons labeling for both NK-1 receptors and SP-CTA (bar = 10μ).
To verify that NK-1 receptors were required for uptake of SP-CTA in the TNC, NK-1 knockout mice (NK1-KO) and wild type Balb/c mice were injected intracisternally with 10 μg of SP-CTA (1μl) or vehicle. Two NK1-KO and two wild type animals received SP-CTA and another set of four animals received vehicle. The animals were euthanized with CO2 two hours following the injections and the brainstems were removed. The tissue was rinsed with PBS and prepared for western blots. The blots were probed with antibodies to the A subunit of cholera toxin (CTA). As figure 2 illustrates CTA was only observed in the brainstems of wild type mice that had received SP-CTA injections. The CTA immunoreactive band from the wild type mice migrated at the same molecular weight as the SP-CTA control sample (approximately 30kd). This is consistent with the predicted molecular weight of SP-CTA 5. NK1-KO animals and vehicle injected animals did not demonstrate the presence of CTA. These findings confirm that intracisternally injected SP-CTA requires NK-1 receptors for uptake in the TNC.
Figure 2.
Uptake of SP-CTA in NK1-KO mice. NK1-KO mice and Balb/c wild type mice were injected intracisternally with either SP-CTA (10μg, 1μl PBS) or with 1μl PBS (2 animals per treatment group). Two hours following the injections the brainstems were harvested, rinsed with PBS and run on western blots. The blots were probed with an anti-body to the A subunit of cholera toxin. The far left lane on the western blot was a 5 μg sample of the original SP-CTA stock solution.
Behavioral Effects of Intracisternally Injected SP-CTA
To determine the behavioral consequences of SP-CTA rats were injected intracisternally with either 10μg SP-CTA in 10μl PBS or PBS alone. Twenty four hours following the injections the SP-CTA animals were agitated and displayed aggressive behavior towards their cage mates. To determine if the animals exhibited enhanced sensitivity to nociceptive stimuli on the face we utilized an operant facial nociception assay as previously described 32,29,31,39,38. In this assay the rats are required to place their face against thermally controlled tubes in order to obtain a sweetened milk reward. Twenty four hours following the intracisternal injections of SP-CTA we found that rats responded to a −4°C stimulus in a manner that was not statistically different from vehicle treated animals in the number of licking events (Figure 3A) or the number of contacts with the cold stimulus (Figure 3B). However, animals receiving 10mg/kg naloxone (sc.) 24 hours after receiving SP-CTA and 30 minutes prior to testing did not access the reward solution to any significant extent (N = 5 rats/treatment group, p < 0.05, ANOVA followed by Dunnett’s test when compared to baseline measurements. Two way ANOVA p < 0.05 Vehicle versus SP-CTA) indicating that the animals were highly sensitized to the cold temperature. Naloxone did not influence the performance of the vehicle injected control rats.
Figure 3.
Effect of SP-CTA and naloxone on nociception in rats at −4°C in the operant facial nociception assay. The rats were tested prior to injections (Baseline: vehicle treated, N = 10; SP-CTA, N = 10) and 24 hours following the intracisternal injection of either SP-CTA (10μg, 10μl) or Vehicle (10μl PBS). Thirty minutes prior to testing half of the animals received injections of naloxone (10mg/kg, sc.). N = 5 rats per treatment group. A. Number of licking events in the 30 min. trial. B. Number of contacts with the stimulus. Asterisks indicate p<0.05 ANOVA followed by Dunnett’s test when compared to baseline. Two way ANOVA p < 0.05 Vehicle versus SP-CTA, # indicates p < 0.05 Bonferroni’s post hoc test when comparing the naloxone treated animals that received SP-CTA versus those that did not receive SP-CTA.
Thermal Place preference
One concern with the operant data was that the combination of SP-CTA and naloxone may have suppressed motor function resulting in reduced access to the reward solution. To evaluate the possible motor effects of this treatment rats were tested in Vierck et al’s thermal place preference assay 54. Rats (N = 8) were tested in the place preference assay without treatment and then injected intracisternally with SP-CTA (10μg in 10μl PBS). Twenty four hours following the SP-CTA treatment the animals received a subcutaneous injection of naloxone (10mg/kg) and were tested one half hour later. The animals spent an average of 393 ± 81 seconds in the warm chamber and 507 ± 81 seconds in the cool chamber prior to treatment. Following the SP-CTA/naloxone treatment the animals spent 565 ± 107 seconds in the warm chamber and 335 ± 107 seconds in the cool chamber (p = 0.24 students’ paired t-test). The number of crossings between chambers were 20 ± 3 and 15 ± 4 for the pre and post treatment conditions respectively (p = 0.21 students’ paired t-test). These data indicate that the SP-CTA/naloxone treatment did not significantly influence crossings between chambers and suggested that motor function was not compromised by the treatment to the extent that the rats could not perform the operant facial nociception assay.
SP-CTA Dose Response Relationship in Mice
To evaluate the dose response relationship of intracisternal SP-CTA hairless SKH1-Hrhr mice were used. These animals do not require shaving in order to perform on the operant facial nociception assay. We previously demonstrated that the performance of hairless mice is similar to shaved C57BL/6J mice 29. The animals (N = 36) were trained as previously described and then tested for a baseline response. The mice then received intracisternal injections of 0.1, 1.0 or 10 μg of SP-CTA in 1 μl of PBS. Twenty four hours following the injections half of the animals received injections of naloxone (10 mg/kg) and the other half received vehicle injections (0.25 mls, sc.)(N = 6 mice per treatment group). Half an hour later the mice were tested in the operant facial nociception assay with the thermal stimulus set to 10°C. The temperature of 10°C was chosen instead of −4°C because preliminary studies with mice indicated that, unlike rats, mice avoided the reward solution when the stimulus was set lower than 10°C. Figure 4 demonstrates that the responses of mice that had received SP-CTA alone did not differ from their baseline responses in the assay (ANOVA, p > 0.05). However, the animals that received the naloxone injections in addition to the SP-CTA demonstrated a clear dose dependent decrease in licking on the reward bottle (p < 0.05, ANOVA followed by Dunnett’s test, Two way ANOVA p < 0.05 naloxone versus vehicle, p < 0.05 for dose of SP-CTA). These findings are consistent with the previously described results in rats.
Figure 4.
SP-CTA dose response relationship in mice. Hairless SKH1-Hrhr mice were tested in the operant facial nociception assay prior to treatment (Baseline, N = 36) and then 24 hours following intracisternal injections of SP-CTA (0.1, 1.0, or 10μg in 1μl PBS). Thirty minutes prior to testing half of the animal in each group received injections of naloxone (10mg/kg, sc.) and the other half received vehicle injections (PBS, 0.25mls, sc.) (N = 6 animals per treatment group). Asterisks indicate p < 0.05, ANOVA followed by Dunnett’s test when compared to the Baseline responses. Two way ANOVA p < 0.05 Vehicle versus Naloxone treatment and p < 0.05 for dose of SP-CTA.
SP-CTA in MOR Knockout Mice
The naloxone data in both rats and mice indicated that an endogenous opioid mediated antinociceptive system was activated in response to the sensitization produced by intracisternal SP-CTA. This further suggested that individuals with a defective endogenous opioid system may be more adversely affected by central sensitization than normal individuals. To test this idea MOR knockout mice were injected intracisternally with SP-CTA and tested in the operant facial nociception assay at 10°C. Their data were compared to data collected in C57BL/6J wild type mice. Figure 5A illustrates that 24 hours following an intracisternal injection of SP-CTA (10μg, 1μl PBS) MOR knockout mice were hypersensitive to 10°C without having to inject them with naloxone (Baseline N = 23 mice; Vehicle N = 8 mice; SP-CTA N = 13 mice) (p < 0.05 ANOVA followed by Dunnett’s test). Wild type mice, on the other hand, did not demonstrate hypersensitivity to 10°C following intracisternal SP-CTA unless they also received an injection of naloxone (Figure 5B)(Baseline N = 27 mice; Vehicle/Nal N = 7 mice; SP-CTA N = 7 mice; SP-CTA/Nal N = 7 mice) (p < 0.05 ANOVA followed by Dunnett’s test). These findings confirmed the hypothesis that a defective endogenous opioid system may make an animal more susceptible to the consequences of central sensitization.
Figure 5.
Effect of intracisternal SP-CTA in mu opioid receptor knockout (MOR) mice. Baseline testing was completed at 10°C prior to intracisternal injections. Animals were treated with either 10μg SP-CTA or PBS via percutaneous intracisternal injection (1μl). The animals were tested 24 hrs later at 10°C using the operant facial nociception assay. A. MOR knockout mice (Baseline N = 23 mice; Vehicle N = 8 mice; SP-CTA N = 13 mice). Asterisks indicate p < 0.05, ANOVA followed by Dunnett’s test when compared to the Baseline responses. # indicates p < 0.05 Bonferroni’s test on SP-CTA versus vehicle treated animals.
B. In wild type C57BL/6J mice some of the animals received subcutaneous injections of naloxone (10mg/kg) 30 minutes prior to testing. (Baseline N = 27 mice; Vehicle/Nal N = 7 mice; SP-CTA N = 7 mice; SP-CTA/Nal N = 7 mice). Asterisks indicate p < 0.05, ANOVA followed by Dunnett’s test when compared to the Baseline responses. # indicates p < 0.05 Bonferroni’s test on SP-CTA/Nal versus SP-CTA treated animals that did not receive naloxone.
Discussion
Over the last decade several authors have suggested the hypothesis that central sensitization may be the primary disease in many chronic pain conditions 11,22,14,37,3,25,35,41,43,48. This may be particularly true for idiopathic conditions where there is no clear injury that might serve as a site for neuromas or other peripheral pain generators. The hypothesis is supported by the fact that many of the pain syndromes are associated with allodynia to both mechanical and thermal peripheral stimuli, referred pain, expanded receptive fields and a persistent or recurring background pain sensation. For example, individuals diagnosed with fibromyalgia have neurophysiological signs of central sensitization which include diffuse pain, hypersensitivity to peripheral stimuli, areas of secondary hypersensitivity and a constant or recurring background pain. Additionally, these individuals frequently present with comorbid conditions such as irritable bowel syndrome, migraine headache and temporomandibular joint disorders 10,49,50,52,51,59. The comorbid nature of these syndromes suggests that a peripheral generator is not responsible for maintaining the disease. The most parsimonious explanation for the etiology of these comorbid conditions is that there is one central mechanism. The presence of signs of central sensitization indicates that central sensitization is the major concern in these patients. Thus central sensitization models may be useful for determining the molecular mechanisms associated with chronic pain and for evaluating novel analgesics.
In order to understand central sensitization in the Trigeminal Nucleus Caudalis (TNC) as a disease process for craniofacial pain we have begun to use SP-CTA as a tool to induce central sensitization. SP-CTA produces central sensitization by activation of adenylate cyclase in neurons expressing neurokinin receptors 5. SP-CTA produces central sensitization without a peripheral injury and, thus, it eliminates the possibility of a peripheral generator that maintains the sensitized state. In this study we found that intracisternally injected SP-CTA co-localizes with NK-1 receptors in the TNC (Figures 1 and 2). We examined the uptake of SP-CTA at one to two hours after the intracisternal injections because intracellular cholera toxin is degraded with a half life of approximately eight hours 15. We confirmed this published work by examining rats twenty four hours after intracisternal injections of SP-CTA and found that we could not observe the presence of CTA in the tissue (data not shown). Thus the biochemical and behavioral effects of SP-CTA are maintained for at least 24 hours by the reaction products of cholera toxin rather than by its continued presence in the cells. We also found that SP-CTA did not migrate rostral to the TNC following intracisternal injection (data not shown). This finding is consistent with the caudal flow of CSF from the fourth ventricle, which may have prevented the diffusion of SP-CTA to the trigeminal nucleus interpolaris or other brainstem nuclei.
In our previous study we found that intrathecally administered SP-CTA produced hypersensitivity to thermal stimuli in the hind limbs of rats 5. The present study confirmed those findings in the TNC and face of both rats and mice using cold temperatures. Similar, but less robust, results were observed with 48°C in rats (data not shown). However, Latremoliere and Woolf suggest that heat hypersensitivity is most likely due to peripheral sensitization rather than central sensitization whereas mechanical and cold hypersensitivity may be the result of central sensitization 22. Thus additional work is needed to determine the mechanism of the thermal hypersensitivity observed following intrathecal 5 and intracisternal SP-CTA.
We also found that SP-CTA activated an endogenous opioid dependent inhibitory system that appeared to suppress peripheral hypersensitivity. When wild type rats and mice were treated intracisternally with SP-CTA an injection of naloxone was required to reveal enhanced sensitivity in the face. MOR knockout mice, on the other hand, demonstrated hypersensitivity without the naloxone treatment. These findings indicate that sensitization of the TNC results in the activation of an endogenous opioid system. It is currently not known if this activated opioid pathway involves descending or local TNC mechanisms. We speculate, however, that the sensitized TNC neurons are stimulating a descending control system. Previous work by Lee and colleagues demonstrated that stimulation of the ventral PAG inhibits reflex hypersensitivity associated with peripheral nerve injury in rats 23. Furthermore, Sohn et al., and Jones and Gebhart demonstrated that injecting an opioid into the PAG could also suppress sensitivity following peripheral nerve injury 47,19 and, in a spinal cord injury model, Hao and colleagues demonstrated that endogenous opioid systems were suppressing peripheral hypersensitivity to mechanical and cold stimuli 17. Our data are consistent with these previous studies and suggest that activation of spinothalamic track neurons in the TNC stimulates the opioid dependent descending inhibitory system in the PAG to suppress incoming nociceptive signals from the face. Supporting this idea we observed that all animals that received intracisternal SP-CTA, except NK1-KO mice, demonstrated agitated behaviors 24 hours following the injections. These behaviors consisted of unprovoked aggression toward cage mates, rearing and defensive posturing. These observations suggested that the animals were uncomfortable and may have had spontaneous pain evoked by the SP-CTA sensitized TNC. However, SP-CTA treated wild type animals still performed in the operant facial nociception assay as well as control animals indicating that they were not experiencing hypersensitivity to peripheral stimuli. Only when they received an injection of naloxone did the SP-CTA treated wild type animals demonstrate hypersensitivity in the assay. Thus, we suspect that the peripheral stimuli were blunted by descending inhibition. Further data are needed to confirm this hypothesis.
In previous studies we and others found that wild type cholera toxin inhibited the hyperalgesia and allodynia produced by a variety of peripheral injuries 4,8. The inhibitory effect of the toxin was blocked by the opioid antagonist naloxone. The mechanism that produced this endogenous opioid mediated inhibition is not known, but Shen and Crain found that cholera toxin can enhance the inhibitory effects of opioids on primary afferent neurons 42. Based on these data we speculate that wild type cholera toxin, which binds primarily to GM1 ganglioside, acts on primary afferent neurons to inhibit peripherally mediated nociception. SP-CTA on the other hand, affects NK-1 receptor expressing central neurons to generate central sensitization. These data indicate that targeting the A subunit of cholera toxin to specific cell types in vivo with various targeting ligands can produce profound differences in behavior.
One aspect of our findings that may have clinical relevance is that MOR knockout mice demonstrated hypersensitivity to cold temperatures after receiving intracisternal SP-CTA without needing an injection of naloxone. This suggests that individuals with a defective endogenous opioid system could be more vulnerable to central sensitization than normal subjects, just as the MOR knockout mice were more susceptible to the SP-CTA induced central sensitization than the wild type mice. If an injury elicits central sensitization or central sensitization arises spontaneously in such a compromised individual the pain may go unchecked by the endogenous descending control system. The net result might be that the pain is more disabling than it would be in a normal individual. It is also possible that individuals with defective descending control systems may be more vulnerable to the development of chronic pain. Recent work has demonstrated functionally significant mutations in the mu opioid receptor in many individuals. For example, the A118G single nucleotide polymorphism in the mu opioid receptor was found to reduce the efficacy of opioids in both homo- and heterozygous individuals 34,21. Presumably the efficacy of endogenous opioid peptides would also be diminished as a result of this mutation suggesting that the endogenous opioid mediated pain control system may be compromised. Since it was found that approximately 4 percent of the population was homozygous for this mutation and another 17 percent were heterozygous it is possible that a significant portion of the population is vulnerable to chronic pain conditions as a result of this single mutation 34,21.
In addition to genetic and developmental flaws in descending inhibition, previous work with a rat bladder inflammation model has demonstrated that injury as a neonate reduces the effectiveness of the endogenous opioid system in the animal as an adult. The defective descending inhibition results in a greater nociceptive response to bladder inflammation in the adult animal 36,9. These findings indicate that early life experiences may also make an individual susceptible to central sensitization and chronic pain.
The mu opioid receptor is not the only vulnerability in the endogenous pain control network. The inhibitory descending control system is a complicated arrangement of neurons with many possible molecular and anatomical sites that could serve as loci for disrupting descending inhibition. For example, kappa and delta opioid receptors are obvious sites of vulnerability that need to be investigated. Thus many people may be susceptible to chronic pain due to inefficient descending inhibitory control as a result of mutations in various genes, through developmental abnormalities or through early life experiences. The use of SP-CTA in mice provides a rapid assay to evaluate the potential of various genetic mutations or neonatal manipulations to disrupt descending control mechanisms that may lead to enhanced vulnerability to chronic pain syndromes. Given the ease of injecting SP-CTA into the cisterna magna and the simplicity of testing mice in the operant facial nociception assay a rapid survey of the contribution of many genes to facial nociception could be undertaken. Each strain of mice or genetically altered mice could, in theory, be fully characterized within a 24 hour period using this model of central sensitization. Thus it is possible to quickly identify specific genes, such as the mu opioid receptor, or deficits that could be involved in central sensitization mediated pain syndromes. Further work is needed to determine if this central sensitization model can provide clues as to the mechanisms that transition acute central sensitization into chronic pain.
In summary, we have developed an easy method to evaluate central sensitization in the TNC of rodents. The injection of SP-CTA induces sensitization without a peripheral injury and our operant facial nociception assay can be used to rapidly evaluate this central sensitization. The data from this study also demonstrated that an endogenous opioid mediated inhibitory system is activated by TNC sensitization. Since mice can be used in the operant facial nociception assay this model system should be very useful for identifying specific genetic vulnerabilities for the development of central sensitization mediated pain syndromes and for evaluating novel therapeutics to treat chronic pain.
Perspective
Central sensitization is a concern in many craniofacial pain conditions. In this project we utilize a conjugate of substance P and the catalytic subunit of cholera toxin to induce central sensitization in the nucleus caudalis of rodents. The data indicate that the injected animals become hypersensitive in the face.
Acknowledgments
This work was supported by National Institutes of Health grants NS045614 and DA016562. The University of Florida has licensed SP-CTA to Advanced Targeting Systems, San Diego, CA.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Contributor Information
Robert M. Caudle, Email: caudle@ufl.edu.
Christopher King, Email: cking@dental.ufl.edu.
Todd A. Nolan, Email: tnolan@dental.ufl.edu.
Shelby K. Suckow, Email: shelbyks@gmail.com.
Charles J. Vierck, Jr., Email: vierck@mbi.ufl.edu.
John K. Neubert, Email: jneubert@dental.ufl.edu.
References
- 1.Bozic CR, Lu B, Hopken UE, Gerard C, Gerard NP. Neurogenic amplification of immune complex inflammation. Science. 1996;273:1722–1725. doi: 10.1126/science.273.5282.1722. [DOI] [PubMed] [Google Scholar]
- 2.Brenner GJ, Ji RR, Shaffer S, Woolf CJ. Peripheral noxious stimulation induces phosphorylation of the NMDA receptor NR1 subunit at the PKC-dependent site, serine-896, in spinal cord dorsal horn neurons. Eur J Neurosci. 2004;20:375–384. doi: 10.1111/j.1460-9568.2004.03506.x. [DOI] [PubMed] [Google Scholar]
- 3.Burstein R, Cutrer MF, Yarnitsky D. The development of cutaneous allodynia during a migraine attack clinical evidence for the sequential recruitment of spinal and supraspinal nociceptive neurons in migraine. Brain. 2000;123 (Pt 8):1703–1709. doi: 10.1093/brain/123.8.1703. [DOI] [PubMed] [Google Scholar]
- 4.Caudle RM, Mannes AJ, Benoliel R, Eliav E, Iadarola MJ. Intrathecally administered cholera toxin blocks allodynia and hyperalgesia in persistent pain models. Journal of Pain. 2001;2:118–127. doi: 10.1054/jpai.2000.19948. [DOI] [PubMed] [Google Scholar]
- 5.Caudle RM, Mannes AJ, Keller J, Perez FM, Suckow SK, Neubert JK. Sensitization of Spinal Cord Nociceptive Neurons with a Conjugate of Substance P and Cholera Toxin. BMC Neurosci. 2007;8:30. doi: 10.1186/1471-2202-8-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chaban VV, Li J, Ennes HS, Nie J, Mayer EA, McRoberts JA. N-methyl-D-aspartate receptors enhance mechanical responses and voltage-dependent Ca2+ channels in rat dorsal root ganglia neurons through protein kinase C. Neuroscience. 2004;128:347–357. doi: 10.1016/j.neuroscience.2004.06.051. [DOI] [PubMed] [Google Scholar]
- 7.Chen L, Huang LY. Protein kinase C reduces Mg2+ block of NMDA-receptor channels as a mechanism of modulation. Nature. 1992;356:521–523. doi: 10.1038/356521a0. [DOI] [PubMed] [Google Scholar]
- 8.Chung KM, Suh HW. Pretreatment with cholera or pertussis toxin differentially modulates morphine- and beta-endorphin-induced antinociception in the mouse formalin test. Neuropeptides. 2001;35:197–203. doi: 10.1054/npep.2001.0862. [DOI] [PubMed] [Google Scholar]
- 9.Deberry J, Ness TJ, Robbins MT, Randich A. Inflammation-induced enhancement of the visceromotor reflex to urinary bladder distention: modulation by endogenous opioids and the effects of early-in-life experience with bladder inflammation. J Pain. 2007;8:914–923. doi: 10.1016/j.jpain.2007.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Desmeules JA, Cedraschi C, Rapiti E, Baumgartner E, Finckh A, Cohen P, Dayer P, Vischer TL. Neurophysiologic evidence for a central sensitization in patients with fibromyalgia. Arthritis Rheum. 2003;48:1420–1429. doi: 10.1002/art.10893. [DOI] [PubMed] [Google Scholar]
- 11.Dodick D, Silberstein S. Central sensitization theory of migraine: clinical implications. Headache. 2006;46 (Suppl 4):S182–S191. doi: 10.1111/j.1526-4610.2006.00602.x. [DOI] [PubMed] [Google Scholar]
- 12.Dougherty PM, Palecek J, Paleckova V, Willis WD. Neurokinin 1 and 2 antagonists attenuate the responses and NK1 antagonists prevent the sensitization of primate spinothalamic tract neurons after intradermal capsaicin. J Neurophysiol. 1994;72:1464–1475. doi: 10.1152/jn.1994.72.4.1464. [DOI] [PubMed] [Google Scholar]
- 13.Draisci G, Iadarola MJ. Temporal analysis of increases in c-fos, preprodynorphin and preproenkephalin mRNAs in rat spinal cord. Brain Res Mol Brain Res. 1989;6:31–37. doi: 10.1016/0169-328x(89)90025-9. [DOI] [PubMed] [Google Scholar]
- 14.Dubner R, Ren K. Brainstem mechanisms of persistent pain following injury. J Orofac Pain. 2004;18:299–305. [PubMed] [Google Scholar]
- 15.Fishman PH. Internalization and degradation of cholera toxin by cultured cells: relationship to toxin action. J Cell Biol. 1982;93:860–865. doi: 10.1083/jcb.93.3.860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ganguly NK, Kaur T. Mechanism of action of cholera toxin & other toxins. Indian J Med Res. 1996;104:28–37. [PubMed] [Google Scholar]
- 17.Hao JX, Yu W, Xu XJ. Evidence that spinal endogenous opioidergic systems control the expression of chronic pain-related behaviors in spinally injured rats. Exp Brain Res. 1998;118:259–268. doi: 10.1007/s002210050280. [DOI] [PubMed] [Google Scholar]
- 18.Ji RR, Woolf CJ. Neuronal plasticity and signal transduction in nociceptive neurons: implications for the initiation and maintenance of pathological pain. Neurobiol Dis. 2001;8:1–10. doi: 10.1006/nbdi.2000.0360. [DOI] [PubMed] [Google Scholar]
- 19.Jones SL, Gebhart GF. Inhibition of spinal nociceptive transmission from the midbrain, pons and medulla in the rat: activation of descending inhibition by morphine, glutamate and electrical stimulation. Brain Res. 1988;460:281–296. doi: 10.1016/0006-8993(88)90373-3. [DOI] [PubMed] [Google Scholar]
- 20.Kawasaki Y, Kohno T, Zhuang ZY, Brenner GJ, Wang HB, Van der Meer C, Befort K, Woolf CJ, Ji RR. Ionotropic and metabotropic receptors, protein kinase A, protein kinase C, and Src contribute to C-fiber-induced ERK activation and cAMP response element-binding protein phosphorylation in dorsal horn neurons, leading to central sensitization. Journal of Neuroscience. 2004;24:8310–8321. doi: 10.1523/JNEUROSCI.2396-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Klepstad P, Rakvag TT, Kaasa S, Holthe M, Dale O, Borchgrevink PC, Baar C, Vikan T, Krokan HE, Skorpen F. The 118 A > G polymorphism in the human mu-opioid receptor gene may increase morphine requirements in patients with pain caused by malignant disease. Acta Anaesthesiol Scand. 2004;48:1232–1239. doi: 10.1111/j.1399-6576.2004.00517.x. [DOI] [PubMed] [Google Scholar]
- 22.Latremoliere A, Woolf CJ. Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J Pain. 2009;10:895–926. doi: 10.1016/j.jpain.2009.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee BH, Park SH, Won R, Park YG, Sohn JH. Antiallodynic effects produced by stimulation of the periaqueductal gray matter in a rat model of neuropathic pain. Neurosci Lett. 2000;291:29–32. doi: 10.1016/s0304-3940(00)01375-6. [DOI] [PubMed] [Google Scholar]
- 24.Lin Q, Peng YB, Willis WD. Possible role of protein kinase C in the sensitization of primate spinothalamic tract neurons. J Neurosci. 1996;16:3026–3034. doi: 10.1523/JNEUROSCI.16-09-03026.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lovati C, D’Amico D, Bertora P. Allodynia in migraine: frequent random association or unavoidable consequence? Expert Rev Neurother. 2009;9:395–408. doi: 10.1586/14737175.9.3.395. [DOI] [PubMed] [Google Scholar]
- 26.Madland G, Feinmann C. Chronic facial pain: a multidisciplinary problem. J Neurol Neurosurg Psychiatry. 2001;71:716–719. doi: 10.1136/jnnp.71.6.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Madland G, Newton-John T, Feinmann C. Chronic idiopathic orofacial pain: I: What is the evidence base? Br Dent J. 2001;191:22–24. doi: 10.1038/sj.bdj.4801081. [DOI] [PubMed] [Google Scholar]
- 28.Messersmith DJ, Kim DJ, Iadarola MJ. Transcription factor regulation of prodynorphin gene expression following rat hindpaw inflammation. Brain Res Mol Brain Res. 1998;53:260–269. doi: 10.1016/s0169-328x(97)00308-2. [DOI] [PubMed] [Google Scholar]
- 29.Neubert JK, King C, Malphurs W, Wong F, Weaver JP, Jenkins AC, Rossi HL, Caudle RM. Characterization of mouse orofacial pain and the effects of lesioning TRPV1-expressing neurons on operant behavior. Mol Pain. 2008;4:43. doi: 10.1186/1744-8069-4-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Neubert JK, Rossi HL, Malphurs W, Vierck CJ, Jr, Caudle RM. Differentiation between capsaicin-induced allodynia and hyperalgesia using a thermal operant assay. Behav Brain Res. 2006;170:308–315. doi: 10.1016/j.bbr.2006.03.008. [DOI] [PubMed] [Google Scholar]
- 31.Neubert JK, Rossi HL, Pogar J, Jenkins AC, Caudle RM. Effects of mu- and kappa-2 opioid receptor agonists on pain and rearing behaviors. Behav Brain Funct. 2007;3:49. doi: 10.1186/1744-9081-3-49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Neubert JK, Widmer CG, Malphurs W, Rossi HL, Vierck CJ, Jr, Caudle RM. Use of a novel thermal operant behavioral assay for characterization of orofacial pain sensitivity. Pain. 2005;116:386–395. doi: 10.1016/j.pain.2005.05.011. [DOI] [PubMed] [Google Scholar]
- 33.Newton-John T, Madland G, Feinmann C. Chronic idiopathic orofacial pain: II. What can the general dental practitioner do? Br Dent J. 2001;191:72–73. doi: 10.1038/sj.bdj.4801098. [DOI] [PubMed] [Google Scholar]
- 34.Oertel BG, Kettner M, Scholich K, Renne C, Roskam B, Geisslinger G, Schmidt PH, Lotsch J. A Common Human {micro}-Opioid Receptor Genetic Variant Diminishes the Receptor Signaling Efficacy in Brain Regions Processing the Sensory Information of Pain. J Biol Chem. 2009;284:6530–6535. doi: 10.1074/jbc.M807030200. [DOI] [PubMed] [Google Scholar]
- 35.Pietrobon D. Migraine: new molecular mechanisms. Neuroscientist. 2005;11:373–386. doi: 10.1177/1073858405275554. [DOI] [PubMed] [Google Scholar]
- 36.Randich A, Mebane H, DeBerry JJ, Ness TJ. Rostral ventral medulla modulation of the visceromotor reflex evoked by urinary bladder distension in female rats. J Pain. 2008;9:920–926. doi: 10.1016/j.jpain.2008.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ren K, Dubner R. Central nervous system plasticity and persistent pain. Journal of Orofacial Pain. 1999;13:155–163. [PubMed] [Google Scholar]
- 38.Rossi HL, Neubert JK. Effects of environmental enrichment on thermal sensitivity in an operant orofacial pain assay. Behav Brain Res. 2008;187:478–482. doi: 10.1016/j.bbr.2007.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rossi HL, Vierck CJ, Jr, Caudle RM, Neubert JK. Characterization of cold sensitivity and thermal preference using an operant orofacial assay. Mol Pain. 2006;2:37. doi: 10.1186/1744-8069-2-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Scott DB, Blanpied TA, Ehlers MD. Coordinated PKA and PKC phosphorylation suppresses RXR-mediated ER retention and regulates the surface delivery of NMDA receptors. Neuropharmacology. 2003;45:755–767. doi: 10.1016/s0028-3908(03)00250-8. [DOI] [PubMed] [Google Scholar]
- 41.Sessle BJ. Acute and chronic craniofacial pain: brainstem mechanisms of nociceptive transmission and neuroplasticity, and their clinical correlates. Crit Rev Oral Biol Med. 2000;11:57–91. doi: 10.1177/10454411000110010401. [DOI] [PubMed] [Google Scholar]
- 42.Shen KF, Crain SM. Cholera toxin-A subunit blocks opioid excitatory effects on sensory neuron action potentials indicating mediation by Gs-linked opioid receptors. Brain Res. 1990;525:225–231. doi: 10.1016/0006-8993(90)90868-c. [DOI] [PubMed] [Google Scholar]
- 43.Silberstein SD. Migraine pathophysiology and its clinical implications. Cephalalgia. 2004;24 (Suppl 2):2–7. doi: 10.1111/j.1468-2982.2004.00892.x. [DOI] [PubMed] [Google Scholar]
- 44.Sluka KA. Activation of the cAMP transduction cascade contributes to the mechanical hyperalgesia and allodynia induced by intradermal injection of capsaicin. Br J Pharmacol. 1997;122:1165–1173. doi: 10.1038/sj.bjp.0701486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sluka KA, Rees H, Chen PS, Tsuruoka M, Willis WD. Capsaicin-induced sensitization of primate spinothalamic tract cells is prevented by a protein kinase C inhibitor. Brain Res. 1997;772:82–86. doi: 10.1016/s0006-8993(97)00876-7. [DOI] [PubMed] [Google Scholar]
- 46.Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC. Measurement of Protein Using Bicinchoninic Acid. Analytical Biochemistry. 1985;150:76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- 47.Sohn JH, Lee BH, Park SH, Ryu JW, Kim BO, Park YG. Microinjection of opiates into the periaqueductal gray matter attenuates neuropathic pain symptoms in rats. Neuroreport. 2000;11:1413–1416. doi: 10.1097/00001756-200005150-00012. [DOI] [PubMed] [Google Scholar]
- 48.Srikiatkhachorn A. Chronic daily headache: a scientist’s perspective. Headache. 2002;42:532–537. doi: 10.1046/j.1526-4610.2002.02132.x. [DOI] [PubMed] [Google Scholar]
- 49.Staud R, Bovee CE, Robinson ME, Price DD. Cutaneous C-fiber pain abnormalities of fibromyalgia patients are specifically related to temporal summation. Pain. 2008;139:315–323. doi: 10.1016/j.pain.2008.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Staud R, Craggs JG, Perlstein WM, Robinson ME, Price DD. Brain activity associated with slow temporal summation of C-fiber evoked pain in fibromyalgia patients and healthy controls. Eur J Pain. 2008;12:1078–1089. doi: 10.1016/j.ejpain.2008.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Staud R, Koo E, Robinson ME, Price DD. Spatial summation of mechanically evoked muscle pain and painful aftersensations in normal subjects and fibromyalgia patients. Pain. 2007;130:177–187. doi: 10.1016/j.pain.2007.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Staud R, Robinson ME, Price DD. Temporal summation of second pain and its maintenance are useful for characterizing widespread central sensitization of fibromyalgia patients. J Pain. 2007;8:893–901. doi: 10.1016/j.jpain.2007.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tingley WG, Roche KW, Thompson AK, Huganir RL. Regulation of NMDA receptor phosphorylation by alternative splicing of the C-terminal domain. Nature. 1993;364:70–73. doi: 10.1038/364070a0. [DOI] [PubMed] [Google Scholar]
- 54.Vierck CJ, Acosta-Rua AJ, Johnson RD. Bilateral chronic constriction of the sciatic nerve: a model of long-term cold hyperalgesia. J Pain. 2005;6:507–517. doi: 10.1016/j.jpain.2005.03.003. [DOI] [PubMed] [Google Scholar]
- 55.Willis WD. Role of neurotransmitters in sensitization of pain responses. Role of Neural Plasticity in Chemical Intolerance. 2001;933:142–156. doi: 10.1111/j.1749-6632.2001.tb05821.x. [DOI] [PubMed] [Google Scholar]
- 56.Woolf CJ. The pathophysiology of peripheral neuropathic pain--abnormal peripheral input and abnormal central processing. Acta Neurochir Suppl (Wien) 1993;58:125–130. doi: 10.1007/978-3-7091-9297-9_29. [DOI] [PubMed] [Google Scholar]
- 57.Woolf CJ, Thompson SW, King AE. Prolonged primary afferent induced alterations in dorsal horn neurones, an intracellular analysis in vivo and in vitro. J Physiol (Paris) 1988;83:255–266. [PubMed] [Google Scholar]
- 58.Woolf CJ, Thompson SWN. The Induction and Maintenance of Central Sensitization Is Dependent on N-Methyl-D-Aspartic Acid Receptor Activation - Implications for the Treatment of Postinjury Pain Hypersensitivity States. Pain. 1991;44:293–299. doi: 10.1016/0304-3959(91)90100-C. [DOI] [PubMed] [Google Scholar]
- 59.Zoppi M, Maresca M. Symptoms accompanying fibromyalgia. Reumatismo. 2008;60:217–220. doi: 10.4081/reumatismo.2008.217. [DOI] [PubMed] [Google Scholar]
- 60.Zou X, Lin Q, Willis WD. Role of protein kinase a in phosphorylation of NMDA receptor 1 subunits in dorsal horn and spinothalamic tract neurons after intradermal injection of capsaicin in rats. Neuroscience. 2002;115:775–786. doi: 10.1016/s0306-4522(02)00490-6. [DOI] [PubMed] [Google Scholar]
- 61.Zou XJ, Lin Q, Willis WD. Enhanced phosphorylation of NMDA receptor 1 subunits in spinal cord dorsal horn and spinothalamic tract neurons after intradermal injection of capsaicin in rats. Journal of Neuroscience. 2000;20:6989–6997. doi: 10.1523/JNEUROSCI.20-18-06989.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]