Abstract
Rationale
The Purkinje fiber network has been proposed as the source of arrhythmogenic Ca2+ release events in catecholaminergic polymorphic ventricular tachycardia (CPVT) yet evidence supporting this mechanism at the cellular level is lacking.
Objective
We sought to determine the frequency and severity of spontaneous Ca2+ release events and the response to the anti-arrhythmic agent flecainide in Purkinje cells and ventricular myocytes from RyR2R4496C/+ CPVT mutant mice and littermate controls.
Methods and Results
We crossed RyR2R4496C/+ knock-in mice with the newly described Cntn2-EGFP BAC transgenic mice, which express a fluorescent reporter gene in cells of the cardiac conduction system, including the distal Purkinje fiber network. Isolated VMs (EGFP-) and Purkinje cells (EGFP+) from wild type hearts and mutant hearts were distinguished by epifluorescence and intracellular Ca2+ dynamics recorded by microfluorimetry. Both wild type and RyR2R4496C/+ mutant Purkinje cells displayed significantly slower kinetics of activation and relaxation compared to ventricular myocytes of the same genotype, and τdecay in the mutant Purkinje cells was significantly slower than that observed in wild type Purkinje cells. Of the four groups studied, RyR2R4496C/+ mutant Purkinje cells were also most likely to develop spontaneous Ca2+ release events and the number of events per cell was also significantly greater. Furthermore, with isoproterenol treatment, while all four groups showed increases in the frequency of arrhythmogenic Ca2+i events, the RyR2R4496C/+ Purkinje cells responded with the most profound abnormalities in intracellular Ca2+ handling, including a significant increase in the frequency of unstimulated Ca2+i events, the development of alternans, as well as isolated and sustained runs of triggered beats. Both Purkinje cells and ventricular myocytes from wild type mice showed suppression of spontaneous Ca2+ release events with flecainide, whereas in RyR2R4496C/+ mice, the Purkinje cells were preferentially responsive to drug. In contrast, the RyR2 blocker tetracaine was equally efficacious in mutant Purkinje cells and ventricular myocytes.
Conclusions
Purkinje cells display a greater propensity to develop abnormalities in intracellular Ca2+ handling than ventricular myocytes. This pro-arrhythmic behavior is enhanced by disease-causing mutations in the RyR2 Ca2+ release channel and greatly exacerbated by catecholaminergic stimulation, with the development of arrhythmogenic triggered beats. These data support the concept that Purkinje cells are critical contributors to arrhythmic triggers in animal models and humans with CPVT and suggest a broader role for the Purkinje fiber network in the genesis of ventricular arrhythmias.
Keywords: Purkinje fiber, arrhythmia, triggered beat, arrhythmia, mouse
Introduction
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is an inherited syndrome characterized by adrenergically mediated ventricular tachycardias, often resulting in syncope and sudden cardiac death1. First described more than 20 years ago by Coumel, recent genetic studies have demonstrated that mutations in the ryanodine receptor (RyR2) and calsequestrin (CASQ2) genes underlie some cases of this syndrome2, 3, implicating aberrant control of Ca2+ homeostasis in the molecular pathogenesis.
The specialized cardiac conduction system (CCS) comprises a heterogeneous network of cells that orchestrate the initiation and propagation of a wave of electrical excitation throughout the myocardium4, 5. Purkinje cells (PCs) are the most distal component of the CCS and they deliver the depolarizing impulse to the working myocytes of the ventricular myocardium. More than forty years ago, Hoffman and colleagues proposed that PCs might “be a significant factor in human arrhythmias6.” Since that time, substantial, but somewhat indirect experimental data has accumulated supporting the concept that PCs play a key mechanistic role triggering a broad range of life-threatening ventricular arrhythmias. Indeed, recent optical mapping data of RyR2R4496C/+ mutant mice, which to a remarkable extent phenocopy human CPVT, support the hypothesis that Ca2+-dependent arrhythmic triggers may be arising from the Purkinje fiber network7.
A mechanistic basis for the pro-arrhythmic behavior of PCs is slowly emerging. Underlying ultrastructural features such as a decreased T-tubular density8, the presence of IP3-sensitive Ca2+ channels9, and susceptibility to Ca2+ overload due to greater Na+ load10 may in part account for the propensity of these specialized cells to develop abnormal Ca2+ release events. Moreover, recent data suggest that higher diastolic Cai-voltage coupling gain in Purkinje fibers may underlie the generation delayed after-depolarizations (DADs) and triggered beats11.
In addition to these cell-autonomous pro-arrhythmic properties, the macroscopic structural features of the specialized ventricular conduction system network may also contribute to the enhanced capacity of PCs to trigger arrhythmias. While electrical loading will normally prevent spontaneous Ca2+ release events (SCaEs) in ventricular myocytes (VMs) from evoking propagated action potentials, the same is not true of events arising within PCs. Electronic interactions between PCs and the underlying myocardium at Purkinje-ventricular junctions (PVJ) are limited, such that stochastic Ca2+ release events in PCs may trigger APs that successfully propagate into working myocytes12-14. Moreover, propagation across the PV junction is highly sensitive to the extent of coupling, as evidenced by a variety of experimental approaches including computational studies and genetically engineered mouse model systems12, 15.
As a consequence of these multi-scale mechanisms, when challenged with stressors that promote electrical remodeling or changes in intracellular Ca2+ handling, such as mutations in Ca2+-handling proteins as seen in CPVT, PCs may be preferentially “primed” to develop arrhythmic triggers16-22.
Through a transcriptional screen our laboratory recently identified a novel cell adhesion molecule named Cntn2, which is specifically expressed in cells of the specialized cardiac conduction system, including the distal Purkinje fiber network23. Moreover, we demonstrated that genetically engineered mice harboring a Cntn2-EGFP BAC transgene faithfully recapitulated expression of the endogenous protein, thereby providing a tool to specifically identify and distinguish PCs from working SCaEs. Interestingly, in our initial study we found that even wild type PCs were more arrhythmogenic than VMs, with more frequent unstimulated Ca2+i release events, DADs and triggered beats. Using this same approach, we now have examined intracellular Ca2+ dynamics in dissociated adult PCs and VMs from both wild type and RyR2R4496C/+ mutant mice, at baseline and following catecholaminergic stimulation as might be seen during arrhythmic episodes in CPVT. We also determined the anti-arrhythmic effects of RyR2 blockers in PCs and VMs from wild type and mutant mice.
Methods
An expanded Methods section is available in the Online Data Supplement.
Mutant Mice
All experiments were performed according to protocols approved by the NYU School of Medicine Institutional Animal Care and Use Committee and conformed to the National Institutes of Health (NIH) guidelines for the care and use of Laboratory Animals. Cntn2-EGFP BAC transgenic mice and RyR2R4496C/+ mutant mice have both been previously described23, 24. All mice studied were F1 crosses between the two strains and used at 8-12 weeks of age. To minimize experimental variability, data from VMs and PCs from the same preparation were always collected on the same day.
Immunohistochemistry
Dissociated myocytes from adult hearts were stained as previously described23. Primary antibodies were directed again ankyrin-G, Scn5a and α-actinin and visualized by confocal microscopy.
Ca2+ imaging and voltage measurements
Adult myocytes were isolated using an established enzymatic digestion protocol25 and intracellular Ca2+ imaging was performed using a microfluorimetry system (IonOptix, Corp), as previously described23. Spontaneous Ca2+ release events (SCaEs) were defined as unstimulated increases in intracellular Ca2+ and were quantified during the 6 second unpaced interval following the 10 beat pacing train. Transmembrane potential were recorded in whole cell current-clamp mode (Axon Instruments), also as previously described23,25.
Statistics
Kinetic parameters were compared using Student's t-test. The proportion of cells displaying SCaEs was compared using Fisher's exact test. The number of SCaEs per cell was compared by the Mann-Whitney test. P values < 0.05 were considered significant.
Results
Identification of Adult Murine Purkinje Cells
We previously demonstrated that the Cntn2-EGFP BAC transgenic mouse strain could be used to specifically identify the relatively rare PCs from a mixed population of dissociated ventricular cells23. The PCs could be visualized both in situ, in the subendocardial region of the ventricles (Figure 1A) and in dissociated cells, where both VMs and PCs express the cardiac specific proteins Scn5a and sarcomeric α-actinin (Figure 1B-E), but only the PCs express EGFP.
Figure 1. Visualization of Murine Purkinje Cells.
Immunofluorescent image (A) of adult Cntn2-EGFP transgenic mouse ventricle showing membrane-enriched staining of ankyrin-G (red) and intrinsic EGFP fluorescence (green). Typical subendocardial location of Purkinje cells is seen. Paired phase (B) and epifluorescent (C) image showing an EGFP-negative ventricular myocyte (VM) and EGFP-positive Purkinje cell (PC) in the same field. Ventricular myocyte (D) and Purkinje cell (E) co-stained for Scn5a (red) and α-actinin (blue). Individual channels are shown below. Both cell types express the two cardiac markers, but EGFP fluorescence is only observed in the Purkinje cell. Magnification is indicated.
Kinetic Properties in Purkinje Cells and Ventricular Myocytes
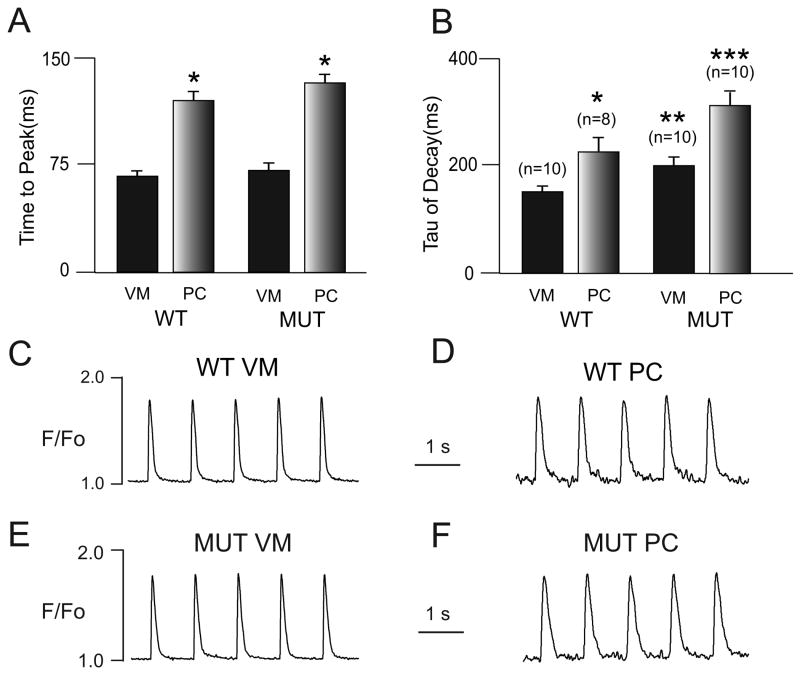
Fluorescence photometry with the calcium-sensitive dye x-Rhod-1 was used to study intracellular Ca2+ dynamics (Figure 2). Confirming our previous observation23, wild type PCs displayed significantly slower kinetics of activation and relaxation compared to VMs, with rise times of 117 ± 6.6 ms (n=8) vs. 68 ± 2.3 (n=10); p<0.00001; and τdecay of 225 ± 24 ms (n=8) vs. 155 ± 10 (n=10); p<0.01. A similar large difference in kinetics was observed comparing the RyRR4496C/+ mutant PCs and mutant myocytes, with rise times of 129 ± 6.5 ms (n=10) vs. 71 ± 5.4 ms (n=10); p<0.000002; and τdecay of 312 ± 23 ms (n=10) vs. 201 ± 15 ms (n=10); p<0.001. Interestingly, the τdecay in the mutant PCs was significantly longer than in wild type PCs (p<.01).
Figure 2. Global Intracellular Ca2+ Transient Kinetics.
Time to peak of the Ca2+ transients (A) and τdecay (B) in wild type (WT) and RyRR4496C/+ mutant (MUT) ventricular myocytes (VM) and Purkinje cells (PC). Examples of Ca2+ transients in each of the four cell types are also shown (C-F). The slower kinetics in WT and MUT PCs are evident. * p<0.05 compared to VMs of same genotype; **p<0.05 compared to WT VMs; ***p<0.05 vs. all other groups.
Spontaneous Ca2+ release events in RyRR4496C/+ Purkinje Cells
We next determined the propensity of VMs and PCs to develop SCaEs following field stimulation at a range of pacing frequencies. We first examined wild type VMs and PCs. As expected, wild type VMs were quite stable; none of 15 cells studies developed SCaEs at 1 Hz and only one cell displayed unpaced SCaEs after pacing at 3Hz or 5 Hz. Wild type PCs had a greater tendency to develop unpaced SCaEs compared to the VMs, especially following higher pacing frequencies (3 of 10 cells at 3 Hz and 5 Hz), as shown in Figure 3. We next examined the behavior of cells from RyRR4496C/+ mutant mice. Mutant VMs and mutant PCs were more arrhythmogenic than their wild type counterparts, both with respect to the proportion of cells displaying SCaEs, as well as the number of SCaEs per cell. This was especially the case for the RyRR4496C/+ mutant PCs, with as many as 62% of cells studied showing arrhythmic activity, as shown in Figure 3 and summarized in Table 1.
Figure 3. Rate Dependence of Arrhythmogenic Spontaneous Calcium Release Events.
Proportion of cells displaying SCaEs (A, B) and number of SCaEs per cell (C, D) in dissociated VMs and PCs from WT and RyR2R4496C/+ MUT mice. *p<0.05 for PCs compared to VMs of same genotype and pacing frequency.
Table 1. Spontaneous Calcium Release Events in Ventricular Myocytes and Purkinje Cells.
| Cells with SCaEs | SCaEs Per Cell | |||||||
|---|---|---|---|---|---|---|---|---|
| - ISO | +ISO | -ISO | + ISO | |||||
| 1 Hz | 3 Hz | 5 Hz | 1 Hz | 1 HZ | 3 Hz | 5 Hz | 1 Hz | |
| WT VM | 0% (15) | 7% (15) | 7% (15) | 14% (7) | 0 ± 0 (15) | 0.07 ± .07 (15) | 0.07 ± .07 (15) | 0.14 ± .14 (7) |
| WT PC | 10% (10) | 30% (10) | 30% (10) | 57% (7) | 0.10 ± .10 (10) | 0.40 ± .22 (10) | 0.40 ± .22 (10) | 3.00 ± 1.38 (7) |
| MUT VM | 22% (41) | 24% (41) | 29% (41) | 40% (10) | 0.29 ± .09 (41) | 0.37 ± .11 (41) | .34 ± .09 (41) | 1.10 ± .48 (10) |
| MUT PC | 52% (29) | 62% (29) | 62% (29) | 90% (10) | 1.07 ± .32 (29) | 1.72 ± .36 (29) | 1.65 ± .36 (29) | 12.2 ± 1.48 (10) |
Data are shown as percentage of of cells with spontaneous Ca2+ release events (SCaEs) and the mean ± SEM SCaEs/cell at each pacing frequency. The number of cells studied for each condition are shown in parentheses. WT, wild type; MUT, mutant; VM, ventricular myocyte; PC, Purkinje cell; ISO, isoproterenol.
Increased Sensitivity to Catecholaminergic Stimulation
Inasmuch as arrhythmic behavior in patients and animal models of CPVT is provoked during catecholaminergic stimulation, the effects of isoproterenol treatment (30nM) on intracellular Ca2+ dynamics in wild type and mutant VMs and PCs was assessed. When paced at 1 Hz, wild type VMs, wild type PCs and mutant VMs all showed modest increases in the proportion of cells that developed ScaEs compared to untreated controls, as shown in Figure 4 and Table 1. However, the effect was most profound in mutant PCs; almost all (90%; 9 of 10) developed arrhythmogenic Ca2+ activity. Moreover, the quantity of SCaEs per cell was significantly greater for the RyR2R4496C PCs (12.2 ± 1.5/cell) compared to RyR2R4496C VMs (1.1 ± .48/cell). Furthermore, even at the slow pacing frequency of 1 Hz, the PCs developed marked abnormalities in intracellular Ca2+ handling, including oscillations, alternans and sustained salvos of presumed triggered beats following the end of the pacing train (Figure 4 D-G). To confirm that these abnormalities in calcium regulation were indeed sufficient to trigger action potentials, we performed simultaneous Ca2+ imaging and recordings of transmembrane potentials in whole cell current-clamp mode. These studies indeed demonstrated that SCaEs could successfully trigger action potentials in the mutant PCs (Figure 4H). Thus, the RyRR4496C/+ mutant PCs appeared to be especially sensitive to the pro-arrhythmic effects of catecholaminergic stimulation.
Figure 4. Effects of Isoproterenol.
Proportion of cells displaying SCaEs in VMs and PCs from WT and MUT mice after exposure to isoproterenol (A). Number of SCaEs per cell (B). Intracellular Ca2+ recordings showing SCaEs in a RyR2R4496C/+ mutant VM (C) and in several RyR2R4496C/+ mutant PCs (D-G). Simultaneous recording of [Ca2+]i and transmembrane voltage demonstrate that SCaEs can lead to triggered action potentials, highlighted in boxed area (H). Arrows indicate field stimulation, which is at 1 Hz in these examples. *p< 0.05 compared to VM of same genotype.
Preferential Suppression of Spontaneous Ca2+ Release Events in Mutant Purkinje Cells
The anti-arrhythmic agent flecainide has shown efficacy in preventing ventricular arrhythmias in mice and humans with Casq2-associated CPVT26. We therefore tested the effects of flecainide on the magnitude of spontaneous Ca2+ release events in VMs and PCs. In agreement with previous studies27, the modest arrhythmic activity observed in wild type VMs was diminished with flecainide, both with respect to the number of cells developing SCaEs, as well as the number of SCaEs per cell. We also observed efficacy in wild type PCs from these same hearts, as shown in Figure 5 and Table 2. As expected, cells from RyR2R4496C/+ mutant mice were substantially more arrhythmic at baseline than their wild type counterparts. Surprisingly, however, the mutant VMs appeared resistant to the anti-arrhythmic effects of flecainide, whereas the mutant PCs showed a significant reduction in the number of cells with SCaEs as well as a reduction in SCaEs per cell, as shown in Figure 5B, D. We also tested the effects of tetracaine, which preferentially reduces RyR2 open probability28. In contrast to flecainide, this agent suppressed SCaEs in mutant PCs and VMs with equal efficacy, as shown in Figure 5E, F. These data are summarized in Table 2.
Figure 5. Effects of Flecainide and Tetracaine on SCaEs.
Proportion of cells displaying SCaEs (A, B, E) and number of SCaEs per cell (C, D, F) in dissociated VMs and PCs from WT and RyR2R4496C/+ MUT mice in the absence (CON) or presence of flecainide (FLEC) or tetracaine (TETR). *p< 0.05 compared to control cells.
Table 2.
Anti-arrhythmic Effects of Flecainide and Tetracaine in Ventricular Myocytes and Purkinje Cells
| Cells with SCaEs | SCaEs Per Cell | |||
|---|---|---|---|---|
| CON | FLEC | CON | FLEC | |
| WT VM | 14% (28) | 3% (36) | .18 ± .09 (28) | .03 ± .03 (36) |
| WT PC | 45% (20) | 11% (18) | .80 ± .24 (20) | .22 ± .17 (18) |
| MUT VM | 43% (56) | 34% (71) | .68 ± .13 (56) | .61 ± .12 (71) |
| MUT PC | 76% (25) | 27% (37) | 2.64 ± .47 (25) | .57 ± .19 (37) |
| CON | TETR | CON | TETR | |
| MUT VM | 54% (24) | 16% (25) | .79 (24) | .16 (25) |
| MUT PC | 89% (19) | 27% (11) | 3.58 (19) | .73 (11) |
Data are shown as percentage of of cells with spontaneous Ca2+ release events (SCaEs) and the mean ± SEM SCaEs/cell. The number of cells studied for each condition are shown in parentheses.WT, wild type; MUT, mutant; VM, ventricular myocyte; PC, Purkinje cell.
Discussion
Recent data suggest that SR Ca2+-release channels comprised of the CPVT-causing RyR2R4496C mutant protein display increased sensitivity to both cytosolic and luminal Ca2+, predisposing them to the development of arrhythmogenic DADs and triggered beats29-32. In this study we show that PCs from mice harboring this same mutation are preferentially and exquisitely sensitive to this underlying molecular defect. Compared to genetically identical VMs, the mutant PCs show more profound abnormalities in intracellular Ca2+ handling, including the development of spontaneous Ca2+ release events at baseline and, as demonstrated by simultaneous calcium and voltage measurements, the progression to triggered beats in response to relatively modest catecholaminergic stimulation.
This study extends our prior observation indicating that even wild type PCs are more likely to develop DADs and triggered beats compared to VMs23. The basis for this heightened proclivity of PCs to develop arrhythmic behavior is not fully known, although the diminished T-tubular density8, altered action potential morphology33, altered kinetics of Ca2+ handling (Fig 2) and increased intracellular Na+ load10 may all be contributory factors. In addition, the participation of IP3-sensitive Ca2+ channels as part of the excitation-contraction coupling machinery may also influence this arrhythmogenic behavior9. As such, when confronted with another perturbation, in this case a defective intracellular Ca2+ release channel, spontaneous Ca2+ release events may arise. If of sufficient number, magnitude or location, these events may result in DADs. Moreover, PCs appear especially vulnerable to the development of DADs by virtue of their increased diastolic Cai-voltage coupling gain, behavior which may be due in part to diminished inwardly rectifying potassium currents (IK1)11. Furthermore, in contrast to well-coupled and electrically loaded working myocytes, in PCs, oscillations in membrane potential are uniquely capable of evoking propagated action potentials that can traverse the Purkinje-ventricular junction into the myocardium proper12.
Our analysis of myocytes from RyR2R4496C mutant mice strongly support the recent suggestion that the Purkinje network may serve as the arrhythmogenic trigger in CPVT associated with RyR2 mutations. This initial hypothesis was based in part upon the characteristic bidirectional ventricular tachycardia seen in patients and mice with this syndrome24, 34 and data demonstrating that ablation of the right ventricular subendocardium (where Purkinje fibers are found) with Lugol's solution converted the biventricular tachycardia to a monomorphic arrhythmia7. Whether or not PCs serve as arrhythmic triggers in CPVT associated with mutations in the calsequestrin gene remains to be seen35, 36, but comparable studies in Casq2 mutant mice should be informative. More broadly, the Purkinje fiber network has been implicated in the initiation of ventricular tachycardia not only in CPVT, but following myocardial infarction, in dilated cardiomyopathies, in idiopathic ventricular fibrillation, in post-shock arrhythmias, and other inherited channelopathies including Brugada and long QT syndromes7, 37-42. A similar analysis of PC behavior at the cellular level of resolution in these diseases should be equally revealing.
Several experimental and clinically approved agents have recently been shown to inhibit arrhythmogenic Ca2+ waves26, 27, 43. In particular, the class 1C anti-arrhythmic agent flecainide was shown to inhibit RyR2 channel activity by reducing the duration of channel openings, especially when the RyR2 channel was activated by high luminal Ca2+ concentration. Moreover, flecainide showed efficacy in preventing ventricular arrhythmias in mice and in humans with Casq2-associated CPVT26. Here we show that flecainide is indeed effective in suppressing spontaneous Ca2+ release events not only in VMs but in PCs as well. Unexpectedly, however, whereas flecainide appears equally efficacious in VMs and PCs from wild type mice, in RyR2R4496C/+ mutant mice the beneficial effect appears restricted to only the PCs. The suppressive effect we observed in PCs occurs at substantially lower concentrations than the reported IC50 for inhibition of RyR2 open probability27. Conceivably, flecainide may be acting through additional targets such as voltage-gated sodium channels, secondarily influencing intracellular Ca2+ load and the propensity for arrhythmogenic SCaEs. Interestingly, we have recently found that Nav1.8/Scn10a is preferentially expressed in PCs, which could account for the differential sensitivity of these cells to flecainide compared to VMs. Indeed, with the more pure RyR2 inhibitor tetracaine, we observed equal efficacy suppressing SCaEs in both VMs and PCs. Accordingly, it will be particularly informative to see if flecainide has clinical utility not only in Casq2-associated CPVT, but also in RyR2-dependent forms of this syndrome.
This study was facilitated by our recent discovery of Cntn2 in the specialized conduction system and the recognition that Cntn2-EGFP mice could facilitate the identification and isolation of conduction system cells, including PCs23. This approach is similar in principal to using Cx40-EGFP knock-in mice44, although that strategy may be less specific, as Cx40 is expressed not only in the conduction system, but also in atrial myocytes, and vascular endothelium and smooth muscle cells45.
In summary, our study provides compelling evidence at the cellular level that the Purkinje fiber network are critical contributors to arrhythmia initiation in CPVT and that defects in intracellular Ca2+ regulation underlie the disease phenotype. These data add to an accumulating body of literature suggesting that the unique structural and functional properties of PCs and the network they comprise play a mechanistic role in a broad range of arrhythmic syndromes. Moreover, our data provide impetus for focusing on PCs as specific anti-arrhythmic targets.
Novelty and Significance
What is known?
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is an inherited arrhythmia that is characterized by aberrant regulation of intracellular calcium handling.
Studies at the organ or organismal level of resolution suggest that the Purkinje fiber network may serve as the arrhythmic trigger in CPVT.
What new information does this article contribute?
We determined the kinetic properties of intracellular calcium transients in adult ventricular myocytes and Purkinje cells from both wild type and RyR2R4496C/+ mutant mice.
We found that both wild type and RyR2R4496C/+ mutant Purkinjes cells have a great propensity to develop unstimulated spontaneous calcium release events and triggered action potentials compared to working ventricular myocytes of the same genotype.
We determined that flecainide and tetracaine both potently suppress spontaneous calcium release events in Purkinje cells.
Multiple lines of evidence suggest that the cardiac Purkinje fiber network serves as the arrhythmic trigger in a range of genetic and acquired forms of heart disease. However, to date, the evidence is largely indirect, including electrical recordings from the subendocardium in the intact or isolated heart, or suppression of arrhythmias following ablation of presumptive cells from the Purkinje fiber network. Here, using a novel genetic reporter gene to specifically identify dissociated adult murine Purkinje cells, we show for the first time that Purkinje cells are indeed more likely than working ventricular myocytes to develop unstimulated spontaneous calcium release events that are capable of triggering action potentials. This preferential behavior is exacerbated in in mice harboring a CPVT disease-causing mutation in the RyR2 channel, especially so in the presence of isoproterenol. Moreover, we show that Purkinje cells can respond to anti-arrhythmic agents, but their sensitivities may differ from working ventricular myocytes. Our study provides new insight into disease pathogenesis at the cellular level of resolution, and also provides impetus for the development of Purkinje cell-specific anti-arrhythmic strategies. Such strategies may find utility both in inherited arrhythmias such as CPVT, but potentially also in acquired syndromes including post-myocardial infarction ventricular arrhythmias.
Supplementary Material
Acknowledgments
Funding Sources
This work was supported by NIH R01HL64757, R01HL081336, R01HL82727 and a New York State Stem Cell Science Award to G.I.F and a Glorney-Raisbeck Fellowship in Cardiovascular Diseases to S.F.G.
Non-standard Abbreviations and Acronyms
- CPVT
Catecholaminergic polymorphic ventricular tachycardia
- RYR2
ryanodine receptor type 2
- CASQ2
calsequestrin
- CCS
cardiac conduction system
- PC
Purkinje cell
- VM
ventricular myocyte
- SCaE
spontaneous calcium release event
Footnotes
Disclosures
None
References
- 1.Cerrone M, Napolitano C, Priori SG. Catecholaminergic polymorphic ventricular tachycardia: A paradigm to understand mechanisms of arrhythmias associated to impaired Ca(2+) regulation. Heart Rhythm. 2009;6:1652–1659. doi: 10.1016/j.hrthm.2009.06.033. [DOI] [PubMed] [Google Scholar]
- 2.Priori SG, Napolitano C, Tiso N, Memmi M, Vignati G, Bloise R, Sorrentino V, Danieli GA. Mutations in the cardiac ryanodine receptor gene (hRyR2) underlie catecholaminergic polymorphic ventricular tachycardia. Circulation. 2001;103:196–200. doi: 10.1161/01.cir.103.2.196. [DOI] [PubMed] [Google Scholar]
- 3.Lahat H, Pras E, Olender T, Avidan N, Ben-Asher E, Man O, Levy-Nissenbaum E, Khoury A, Lorber A, Goldman B, Lancet D, Eldar M. A missense mutation in a highly conserved region of CASQ2 is associated with autosomal recessive catecholamine-induced polymorphic ventricular tachycardia in Bedouin families from Israel. Am J Hum Genet. 2001;69:1378–1384. doi: 10.1086/324565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Myers DC, Fishman GI. Toward an understanding of the genetics of murine cardiac pacemaking and conduction system development. Anat Rec A Discov Mol Cell Evol Biol. 2004;280:1018–1021. doi: 10.1002/ar.a.20077. [DOI] [PubMed] [Google Scholar]
- 5.Pennisi DJ, Rentschler S, Gourdie RG, Fishman GI, Mikawa T. Induction and patterning of the cardiac conduction system. Int J Dev Biol. 2002;46:765–775. [PubMed] [Google Scholar]
- 6.Singer DH, Lazzara R, Hoffman BF. Interrelationship between automaticity and conduction in Purkinje fibers. Circ Res. 1967;21:537–558. doi: 10.1161/01.res.21.4.537. [DOI] [PubMed] [Google Scholar]
- 7.Cerrone M, Noujaim SF, Tolkacheva EG, Talkachou A, O'Connell R, Berenfeld O, Anumonwo J, Pandit SV, Vikstrom K, Napolitano C, Priori SG, Jalife J. Arrhythmogenic mechanisms in a mouse model of catecholaminergic polymorphic ventricular tachycardia. Circ Res. 2007;101:1039–1048. doi: 10.1161/CIRCRESAHA.107.148064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Di Maio A, Ter Keurs HE, Franzini-Armstrong C. T-tubule profiles in Purkinje fibres of mammalian myocardium. J Muscle Res Cell Motil. 2007;28:115–121. doi: 10.1007/s10974-007-9109-6. [DOI] [PubMed] [Google Scholar]
- 9.Hirose M, Stuyvers B, Dun W, Ter Keurs H, Boyden PA. Wide long lasting perinuclear Ca2+ release events generated by an interaction between ryanodine and IP3 receptors in canine Purkinje cells. J Mol Cell Cardiol. 2008;45:176–184. doi: 10.1016/j.yjmcc.2008.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vassalle M, Lin CI. Calcium overload and cardiac function. J Biomed Sci. 2004;11:542–565. doi: 10.1007/BF02256119. [DOI] [PubMed] [Google Scholar]
- 11.Maruyama M, Joung B, Tang L, Shinohara T, On YK, Han S, Choi EK, Kim DH, Shen MJ, Weiss JN, Lin SF, Chen PS. Diastolic intracellular calcium-membrane voltage coupling gain and postshock arrhythmias: role of purkinje fibers and triggered activity. Circ Res. 106:399–408. doi: 10.1161/CIRCRESAHA.109.211292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rawling DA, Joyner RW, Overholt ED. Variations in the functional electrical coupling between the subendocardial Purkinje and ventricular layers of the canine left ventricle. Circ Res. 1985;57:252–261. doi: 10.1161/01.res.57.2.252. [DOI] [PubMed] [Google Scholar]
- 13.Huelsing DJ, Spitzer KW, Pollard AE. Electrotonic suppression of early afterdepolarizations in isolated rabbit Purkinje myocytes. Am J Physiol Heart Circ Physiol. 2000;279:H250–259. doi: 10.1152/ajpheart.2000.279.1.H250. [DOI] [PubMed] [Google Scholar]
- 14.Ben Caref E, Boutjdir M, Himel HD, El-Sherif N. Role of subendocardial Purkinje network in triggering torsade de pointes arrhythmia in experimental long QT syndrome. Europace. 2008;10:1218–1223. doi: 10.1093/europace/eun248. [DOI] [PubMed] [Google Scholar]
- 15.Morley GE, Danik SB, Bernstein S, Sun Y, Rosner G, Gutstein DE, Fishman GI. Reduced intercellular coupling leads to paradoxical propagation across the Purkinje-ventricular junction and aberrant myocardial activation. Proc Natl Acad Sci U S A. 2005;102:4126–4129. doi: 10.1073/pnas.0500881102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Han W, Chartier D, Li D, Nattel S. Ionic remodeling of cardiac Purkinje cells by congestive heart failure. Circulation. 2001;104:2095–2100. doi: 10.1161/hc4201.097134. [DOI] [PubMed] [Google Scholar]
- 17.Han W, Bao W, Wang Z, Nattel S. Comparison of ion-channel subunit expression in canine cardiac Purkinje fibers and ventricular muscle. Circ Res. 2002;91(9):790–797. doi: 10.1161/01.res.0000039534.18114.d9. [DOI] [PubMed] [Google Scholar]
- 18.Boyden PA, Pinto JM. Reduced calcium currents in subendocardial Purkinje myocytes that survive in the 24- and 48-hour infarcted heart. Circulation. 1994;89:2747–2759. doi: 10.1161/01.cir.89.6.2747. [DOI] [PubMed] [Google Scholar]
- 19.Jeck C, Pinto J, Boyden P. Transient outward currents in subendocardial Purkinje myocytes surviving in the infarcted heart. Circulation. 1995;92:465–473. doi: 10.1161/01.cir.92.3.465. [DOI] [PubMed] [Google Scholar]
- 20.Pinto JM, Boyden PA. Reduced inward rectifying and increased E-4031-sensitive K+ current density in arrhythmogenic subendocardial purkinje myocytes from the infarcted heart. J Cardiovasc Electrophysiol. 1998;9:299–311. doi: 10.1111/j.1540-8167.1998.tb00915.x. [DOI] [PubMed] [Google Scholar]
- 21.Pinto JM, Sosunov EA, Gainullin RZ, Rosen MR, Boyden PA. Effects of mibefradil, a T-type calcium current antagonist, on electrophysiology of Purkinje fibers that survived in the infarcted canine heart. J Cardiovasc Electrophysiol. 1999;10:1224–1235. doi: 10.1111/j.1540-8167.1999.tb00300.x. [DOI] [PubMed] [Google Scholar]
- 22.Boyden PA, Pu J, Pinto J, Keurs HE. Ca(2+) transients and Ca(2+) waves in purkinje cells : role in action potential initiation. Circ Res. 2000;86:448–455. doi: 10.1161/01.res.86.4.448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pallante BA, Giovannone S, Liu FY, Zhang J, Liu N, Kang G, Dun W, Boyden PA, Fishman GI. Contactin-2 Expression in the Cardiac Purkinje Fiber Network. Circ Arrhythm Electrophysiol. 2010;3:186–194. doi: 10.1161/CIRCEP.109.928820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cerrone M, Colombi B, Santoro M, di Barletta MR, Scelsi M, Villani L, Napolitano C, Priori SG. Bidirectional ventricular tachycardia and fibrillation elicited in a knock-in mouse model carrier of a mutation in the cardiac ryanodine receptor. Circ Res. 2005;96:e77–82. doi: 10.1161/01.RES.0000169067.51055.72. [DOI] [PubMed] [Google Scholar]
- 25.Liu N, Colombi B, Memmi M, Zissimopoulos S, Rizzi N, Negri S, Imbriani M, Napolitano C, Lai FA, Priori SG. Arrhythmogenesis in catecholaminergic polymorphic ventricular tachycardia: insights from a RyR2 R4496C knock-in mouse model. Circ Res. 2006;99:292–298. doi: 10.1161/01.RES.0000235869.50747.e1. [DOI] [PubMed] [Google Scholar]
- 26.Watanabe H, Chopra N, Laver D, Hwang HS, Davies SS, Roach DE, Duff HJ, Roden DM, Wilde AA, Knollmann BC. Flecainide prevents catecholaminergic polymorphic ventricular tachycardia in mice and humans. Nat Med. 2009;15:380–383. doi: 10.1038/nm.1942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hilliard FA, Steele DS, Laver D, Yang Z, Le Marchand SJ, Chopra N, Piston DW, Huke S, Knollmann BC. Flecainide inhibits arrhythmogenic Ca2+ waves by open state block of ryanodine receptor Ca2+ release channels and reduction of Ca2+ spark mass. J Mol Cell Cardiol. 48:293–301. doi: 10.1016/j.yjmcc.2009.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Venetucci LA, Trafford AW, Diaz ME, O'Neill SC, Eisner DA. Reducing ryanodine receptor open probability as a means to abolish spontaneous Ca2+ release and increase Ca2+ transient amplitude in adult ventricular myocytes. Circ Res. 2006;98:1299–1305. doi: 10.1161/01.RES.0000222000.35500.65. [DOI] [PubMed] [Google Scholar]
- 29.Fernandez-Velasco M, Rueda A, Rizzi N, Benitah JP, Colombi B, Napolitano C, Priori SG, Richard S, Gomez AM. Increased Ca2+ sensitivity of the ryanodine receptor mutant RyR2R4496C underlies catecholaminergic polymorphic ventricular tachycardia. Circ Res. 2009;104:201–209. doi: 10.1161/CIRCRESAHA.108.177493. 212p following 209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jiang D, Xiao B, Zhang L, Chen SR. Enhanced basal activity of a cardiac Ca2+ release channel (ryanodine receptor) mutant associated with ventricular tachycardia and sudden death. Circ Res. 2002;91:218–225. doi: 10.1161/01.res.0000028455.36940.5e. [DOI] [PubMed] [Google Scholar]
- 31.Jiang D, Xiao B, Yang D, Wang R, Choi P, Zhang L, Cheng H, Chen SR. RyR2 mutations linked to ventricular tachycardia and sudden death reduce the threshold for store-overload-induced Ca2+ release (SOICR) Proc Natl Acad Sci U S A. 2004;101:13062–13067. doi: 10.1073/pnas.0402388101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sedej S, Heinzel FR, Walther S, Dybkova N, Wakula P, Groborz J, Gronau P, Maier LS, Vos MA, Lai FA, Napolitano C, Priori SG, Kockskamper J, Pieske B. Na+-dependent SR Ca2+ overload induces arrhythmogenic events in mouse cardiomyocytes with a human CPVT mutation. Cardiovasc Res. 2010;87:50–59. doi: 10.1093/cvr/cvq007. [DOI] [PubMed] [Google Scholar]
- 33.Boyden PA, Albala A, Dresdner KP., Jr Electrophysiology and ultrastructure of canine subendocardial Purkinje cells isolated from control and 24-hour infarcted hearts. Circ Res. 1989;65:955–970. doi: 10.1161/01.res.65.4.955. [DOI] [PubMed] [Google Scholar]
- 34.Liu N, Ruan Y, Priori SG. Catecholaminergic polymorphic ventricular tachycardia. Prog Cardiovasc Dis. 2008;51:23–30. doi: 10.1016/j.pcad.2007.10.005. [DOI] [PubMed] [Google Scholar]
- 35.Liu N, Priori SG. Disruption of calcium homeostasis and arrhythmogenesis induced by mutations in the cardiac ryanodine receptor and calsequestrin. Cardiovasc Res. 2008;77:293–301. doi: 10.1093/cvr/cvm004. [DOI] [PubMed] [Google Scholar]
- 36.Rizzi N, Liu N, Napolitano C, Nori A, Turcato F, Colombi B, Bicciato S, Arcelli D, Spedito A, Scelsi M, Villani L, Esposito G, Boncompagni S, Protasi F, Volpe P, Priori SG. Unexpected structural and functional consequences of the R33Q homozygous mutation in cardiac calsequestrin: a complex arrhythmogenic cascade in a knock in mouse model. Circ Res. 2008;103:298–306. doi: 10.1161/CIRCRESAHA.108.171660. [DOI] [PubMed] [Google Scholar]
- 37.Bogun F, Good E, Reich S, Elmouchi D, Igic P, Tschopp D, Dey S, Wimmer A, Jongnarangsin K, Oral H, Chugh A, Pelosi F, Morady F. Role of Purkinje fibers in post-infarction ventricular tachycardia. J Am Coll Cardiol. 2006;48:2500–2507. doi: 10.1016/j.jacc.2006.07.062. [DOI] [PubMed] [Google Scholar]
- 38.Hayashi M, Kobayashi Y, Iwasaki YK, Morita N, Miyauchi Y, Kato T, Takano T. Novel mechanism of postinfarction ventricular tachycardia originating in surviving left posterior Purkinje fibers. Heart Rhythm. 2006;3:908–918. doi: 10.1016/j.hrthm.2006.04.019. [DOI] [PubMed] [Google Scholar]
- 39.Morishima I, Nogami A, Tsuboi H, Sone T. Verapamil-sensitive left anterior fascicular ventricular tachycardia associated with a healed myocardial infarction: changes in the delayed Purkinje potential during sinus rhythm. J Interv Card Electrophysiol. 2008;22:233–237. doi: 10.1007/s10840-008-9268-4. [DOI] [PubMed] [Google Scholar]
- 40.Sinha AM, Schmidt M, Marschang H, Gutleben K, Ritscher G, Brachmann J, Marrouche NF. Role of left ventricular scar and Purkinje-like potentials during mapping and ablation of ventricular fibrillation in dilated cardiomyopathy. Pacing Clin Electrophysiol. 2009;32:286–290. doi: 10.1111/j.1540-8159.2008.02233.x. [DOI] [PubMed] [Google Scholar]
- 41.Haissaguerre M, Extramiana F, Hocini M, Cauchemez B, Jais P, Cabrera JA, Farre J, Leenhardt A, Sanders P, Scavee C, Hsu LF, Weerasooriya R, Shah DC, Frank R, Maury P, Delay M, Garrigue S, Clementy J. Mapping and ablation of ventricular fibrillation associated with long-QT and Brugada syndromes. Circulation. 2003;108:925–928. doi: 10.1161/01.CIR.0000088781.99943.95. [DOI] [PubMed] [Google Scholar]
- 42.Haissaguerre M, Shah DC, Jais P, Shoda M, Kautzner J, Arentz T, Kalushe D, Kadish A, Griffith M, Gaita F, Yamane T, Garrigue S, Hocini M, Clementy J. Role of Purkinje conducting system in triggering of idiopathic ventricular fibrillation. Lancet. 2002;359:677–678. doi: 10.1016/S0140-6736(02)07807-8. [DOI] [PubMed] [Google Scholar]
- 43.Kobayashi S, Yano M, Suetomi T, Ono M, Tateishi H, Mochizuki M, Xu X, Uchinoumi H, Okuda S, Yamamoto T, Koseki N, Kyushiki H, Ikemoto N, Matsuzaki M. Dantrolene, a therapeutic agent for malignant hyperthermia, markedly improves the function of failing cardiomyocytes by stabilizing interdomain interactions within the ryanodine receptor. J Am Coll Cardiol. 2009;53:1993–2005. doi: 10.1016/j.jacc.2009.01.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Miquerol L, Meysen S, Mangoni M, Bois P, van Rijen HV, Abran P, Jongsma H, Nargeot J, Gros D. Architectural and functional asymmetry of the His-Purkinje system of the murine heart. Cardiovasc Res. 2004;63:77–86. doi: 10.1016/j.cardiores.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 45.Severs NJ, Rothery S, Dupont E, Coppen SR, Yeh HI, Ko YS, Matsushita T, Kaba R, Halliday D. Immunocytochemical analysis of connexin expression in the healthy and diseased cardiovascular system. Microsc Res Tech. 2001;52:301–322. doi: 10.1002/1097-0029(20010201)52:3<301::AID-JEMT1015>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.