Abstract
The prognostic impact of minimal residual disease (MRD) was analyzed in 259 patients with mantle cell lymphoma (MCL) treated within two randomized trials of the European MCL Network (MCL Younger and MCL Elderly trial). After rituximab-based induction treatment 106/190 evaluable patients (56%) achieved a molecular remission (MR) based upon blood and/or bone marrow (BM) analysis. MR resulted in a significantly improved response duration (RD) (87% vs. 61% patients in remission at 2 years, p=0.0043) and emerged to be an independent prognostic factor for RD (HR 0.4, 95% CI 0.1–0.9, p=0.027). MR was highly predictive for prolonged RD independent of clinical response (CR, CRu, PR) (RD at 2 years: 100% in BM MRD-negative CR and 88% in BM MRD-negative CRu/PR, compared to 78% in BM MRD-positive CR and 53% in BM MRD-positive CRu/PR, p=0.0015). Sustained MR during the post-induction period was predictive for outcome in MCL Younger after ASCT (RD at 2 years 100% vs. 65%, p=0.0007) and during maintenance in MCL Elderly (RD at 2 years: 76% vs. 36%, p=0.015). ASCT in MCL Younger patients increased the proportion of patients in MR from 55% prior to high dose therapy to 72% thereafter. Sequential MRD monitoring is a powerful predictor for treatment outcome in MCL.
Keywords: Adult; Aged; Aged, 80 and over; Antineoplastic Combined Chemotherapy Protocols; therapeutic use; Cell Separation; Combined Modality Therapy; Female; Flow Cytometry; Humans; Immunohistochemistry; Immunotherapy; methods; Lymphoma, Mantle-Cell; pathology; therapy; Male; Middle Aged; Neoplasm Staging; Neoplasm, Residual; Polymerase Chain Reaction; Prognosis; Radiotherapy; Treatment Outcome
Keywords: mantle cell lymphoma, autologous stem cell transplantation, RQ-PCR, minimal residual disease, MRD, immunochemotherapy
Introduction
Mantle cell lymphoma (MCL) is characterized by a mostly advanced stage of disease at diagnosis and an aggressive clinical course with a short median overall survival (OS) of 3–4 years after standard treatment. However, recent studies have reported an improved outcome with an almost doubled median survival of 5–6 years.1;2 The biological hallmark of MCL is the chromosomal translocation t(11;14)(q13;q32) leading to cyclin D1 protein overexpression. The translocation is detectable by molecular cytogenetics in more than 95% of MCL. 3;4
Current treatment strategies include combinations of the monoclonal anti-CD20 antibody rituximab with different chemotherapy regimens as well as more intensive treatment protocols including high-dose ara-C.5–7 Furthermore, there is increasing evidence that autologous stem cell transplantation (ASCT) as part of the first-line treatment leads to a substantial prolongation of disease free survival (DFS) and OS in younger patients with MCL.8–14 Although in particular combinations of rituximab, ara-C-based consolidation and ASCT can achieve long-lasting remissions in significant proportions of patients, 7;10;15–18 individual patients may still suffer from early relapse. Recent evidence suggests that clinical relapses might be prevented by experimental consolidation treatments, such as interferon-alpha or antibody maintenance or even allogeneic SCT. Therefore prediction of quality and duration of response becomes increasingly important for early individual risk estimation.
The MCL international prognostic index (MIPI) based on the 4 independent factors age, ECOG performance status, LDH and white blood cell (WBC) count is of proven value for pre-treatment risk assessment in patients with advanced stage MCL.19 However, parameters for early response assessment and individual risk assignment during treatment are currently lacking.
Molecular monitoring of minimal residual disease (MRD) by quantitative PCR is a broadly applicable tool for the assessment of circulating residual lymphoma cells with a great impact on prognosis in different B-cell lymphoma entities.20–27 We have shown that quantitative MRD assessment during treatment allows to compare the relative impact of different treatment modalities (i.e. conventional chemotherapy, ASCT, with and without monoclonal antibodies) on the tumor load, and to study the kinetics of tumor depletion and regrowth after cytotoxic treatment in MCL.26 Clonal IGH VH-JH rearrangements as well as the t(11;14) translocation are suitable targets for molecular MRD assessment in MCL. Achievement of a molecular remission (MR) defined as achievement of MRD negativity following ASCT demonstrated a high prognostic significance for progression free survival (PFS) and OS in MCL.26 However, to date only sparse data on the prognostic impact of MRD in the setting of modern combined immuno-chemotherapy approaches are available10;11;26;28.
In the present study we therefore addressed the prognostic potential of quantitative MRD monitoring after combined immunochemotherapy followed by ASCT or maintenance treatment in MCL patients. Taking advantage of two large and homogeneously treated patient cohorts from the current Intergroup European MCL Network trials (MCL net), we evaluated the prognostic impact of MRD kinetics on disease control and compared the specific effect of different treatment modalities (combined immunochemotherapy, myeloblative radio-chemotherapy and maintenance treatment with interferon-α or rituximab) on quantitative MRD load.
Patients and methods
Patients and sample collection for MRD
Patients with histologically confirmed MCL were randomized within the clinical trials of the MCL net according to age and eligibility to receive a high dose therapy. The trials were investigating the role of different induction protocols followed by either two different high-dose regimens with ASCT (MCL Younger) or two different maintenance therapies (MCL Elderly). Inclusion criteria comprised patients up to 65 years of age in the MCL Younger trial and above 60 years in the MCL Elderly trial with previously untreated, advanced Ann-Arbor stage II to IV MCL. The histologic diagnosis was confirmed by a central pathology review at one of the designated pathology reference centers (European MCL Pathology Panel). Both protocols including the incorporated MRD analyzes had been approved by the local institutional review boards and were conducted according to the updated declaration of Helsinki, and are listed under http://www.clinicaltrials.gov (MCL Younger NCT00209222, MCL Elderly NCT00209209).
Prospective quantitative MRD monitoring was a pre-defined secondary objective of the current trials of the EU-MCL Network. However, participation in the MRD program was not a prerequisite for randomization in the clinical trials. MRD assessment was performed in national reference labs. Due to logistical reasons MRD assessment was mainly performed in Germany and France. Because this analysis was performed within ongoing trials, clinical and molecular data could not be collected concordantly in all cases.
Histological, immunhistochemical and cytogenetic analyzes
The diagnosis of MCL was established according to WHO criteria.29 Conventionally and immunohistochemically stained paraffin sections of lymph node biopsies were reviewed. Minimal requirements for immunohistochemistry included: positivity for Cyclin D1, CD20 and CD5, negativity for CD23 and CD10. The histological slides and immunohistochemical stainings (CD20, CD5, CD23, CyclinD1) were evaluated according to the Annecy criteria21 and MCL were sub-classified according to the different cytological subtypes. Histological slides were reviewed by members of the European Mantle Cell Lymphoma Study Group.
The presence of a t(11;14)(q13;q32) translocation was investigated by either PCR, FISH or conventional cytogenetics in diagnostic PB and/or BM samples.
Treatment of patients below 65 years and eligible for ASCT
After initial randomization patients received either 6 cycles of 3-weekly R-CHOP followed by stem cell mobilization with DexaBEAM and myeloablative radio-chemotherapy with autologous blood stem cell support according to a previously published protocol of the EU-MCL Study Group 14 or a total of 6 cycles of alternating R-CHOP/R-DHAP regimens with a high dose Ara-C containing myeloablative radio-chemotherapy and ASCT. 15;16 Following myeloablative therapy no further antilymphoma treatment was applied (figure 1).
Figure 1. Diagram of the two randomized EU-MCL network trials.
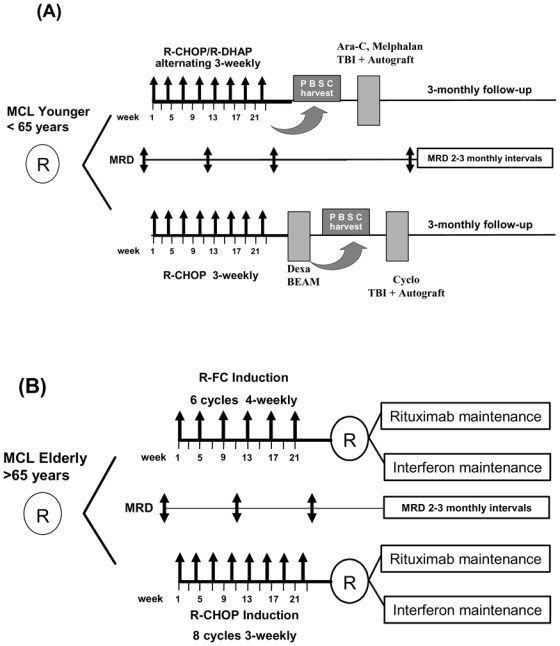
(A) MCL Younger and (B) MCL Elderly with the respective MRD sampling time points. MRD is assessed until clinical relapse or death. Maintenance treatment in both arms of the elderly protocol is given until progression or death.
Treatment of patients above 65 years or above 60 and ineligible for ASCT
Patients were randomized to induction treatment of either 8 cycles of 3-weekly R-CHOP or 6 cycles of 4-weekly R-FC chemotherapy. After a second randomization all patients in clinical remission received maintenance treatment with either interferon-α (IFN-α 3×3 M IU or PegIntron 1 μg/kg weekly) or rituximab maintenance (rituximab 375 mg/m2) at 2-monthly intervals. Maintenance treatment was given until clinical relapse (figure 1).30
Flow Cytometry
Four-color flow cytometry (4C-FC) was performed to assess the proportion of MCL cells in diagnostic blood and bone marrow samples.
This four-color flow cytometry assay had been previously standardized and tested in 281 peripheral blood and bone marrow samples from 98 MCL patients of the current EU-MCL trials and demonstrated a high specifity and sensitivity for MCL cell quantification. The principles of staining protocols for flow cytometry, gating strategies as well as specifity and sensitivity have been recently published by our group in detail.31
The degree of lymphoma involvement of the diagnostic sample was subsequently used to establish standard dilution series of the diagnostic specimen for real-time quantitative RQ-PCR for each individual patient.
Clonality assessment and PCR based MRD analysis
DNA from PB or peripheral blood mononuclear cells (PBMC) and BM was extracted with a standard proteinase K digestion and a phenol-chloroform extraction or the Qiagen Blood Mini Kit (Qiagen, Hilden; Germany). Samples were analyzed by t(11;14) PCR and IGH multiplex PCR as published to assess the clonal rearrangement.32;33 Genescanning and sequence analyzes were performed on an ABI PRISM 377 automated sequencer (Applied Biosystems, Foster City, CA, USA). Sequencing of clonal rearrangements for allel-specific PCR was performed by using the BigDye Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems).
Quantitative PCR with allel-specific oligonucleotides was performed as described previously.26 The assays were established to reach a sensitivity of 10−5, tested by analyzing 10-fold serial dilutions from diagnostic samples in polyclonal DNA derived from pooled mononuclear cells of healthy donors. For determining the quantitative MRD levels target copy numbers were related to the number of target copies at diagnosis.
MRD levels were given as fraction of numbers of MCL cells per total number of mononuclear BM or PB cells analyzed per PCR assay.
Only follow-up samples with a minimum sensitivity of at least 1×10−4 according to albumin copies as control gene 34 were included in survival analysis. Results of RQ-PCR were evaluated according to the criteria of the European Study Group on MRD detection in ALL (ESG-MRD-ALL).35
MRD status at a certain time point was assigned by investigating either PB or BM and if available both. In case of parallel investigations of PB and BM, MRD was judged positive if at least one of both samples was positive by RQ-PCR. A molecular remission was assigned as complete if both parallel samples were MRD negative. Within this study we used the pooled MRD information resulting from MRD analysis in PB and/or BM. In case of MRD positive parallel samples the higher MRD value was used for calculation.
Response criteria and evaluation
Clinical response was assessed after midterm induction therapy (3–4 cycles of induction therapy according to the protocol), at restaging after completion of induction therapy (about 4 weeks after the last cycle of induction), 3 months after ASCT (within MCL Younger) and at 2 to 3 monthly intervals during follow-up. As this analysis has been performed within ongoing clinical trials, randomized arms were pooled and analyzed together without any unblinding of the treatment arms.
Response was defined according to the International Working Group criteria.36 Response duration (RD) was defined only for patients who achieved at least a PR after induction treatment and was calculated as period from the completion of induction to documented progression or death from any cause, which were both considered as an event. Overall survival (OS) was defined as the interval between trial registration and death from any cause.
Definition of molecular remission and sampling time points for MRD analysis
PB and/or BM samples were collected at diagnosis and at follow-up according to clinical staging time points. Sampling time points corresponded to clinical response assessment and included: midterm staging (after 3–4 cycles of induction therapy according to the protocol), restaging after completion of induction therapy (about 4 weeks after the last cycle of induction with R-CHOP or R-CHOP/R-DHAP or R-FC prior to DexaBEAM and ASCT or maintenance treatment) and post-induction monitoring at 3 monthly intervals after ASCT for MCL Younger patients and at 2 to 3 monthly intervals during maintenance follow-up for MCL Elderly patients (figure 1). MRD monitoring was intended to be performed until clinical relapse in both trials.
MRD status at a certain time point was assigned by using the pooled MRD information from MRD analysis in PB or BM and if available both. In case of parallel investigation of PB and BM, MRD was judged positive if at least one of both samples was positive by RQ-PCR. In case of MRD positivity in parallel samples the higher MRD value was used for calculation. Molecular remission (MR) was defined as MRD negativity investigated by clone-specific RQ-PCR with an assay sensitivity of at least 10−4. MR was assigned in parallel analyzed PB and BM samples if both were MRD negative.
MRD status within the post-induction period (implying for MCL Younger patients the first 12 months after ASCT and for MCL Elderly patients the first 12 months after end of induction/start of maintenance) a patient was judged to be MRD positive if at least one sample demonstrated MRD positivity by RQ-PCR.
Samples collected at or subsequent to documented clinical relapse were not included in statistical analysis.
Statistical analysis
To describe quantitative MRD-values median and ranges of MRD levels were assessed. Quantitative MRD-values were compared between groups according to baseline characteristics using the Mann-Whitney-U-Test. Quantitative MRD-values in PB and BM were compared with Pearson’s r and the concordance correlation coefficient. 37 Quantitative MRD-values at different time points during induction were compared by Wilcoxon rank-sum-test. Cross-tables together with exact Fisher tests were calculated to assess the association of the achievement of a molecular remission with categorical clinical parameters and with clinical response. MR after induction and during the first year of follow-up was compared in paired samples by McNemar’s test. Response duration according to clinical or molecular remission was analyzed by Kaplan-Meier estimates and compared using the log rank test. Follow-up time was estimated using the reversed Kaplan-Meier method. Multiple Cox regression was performed to analyze the adjusted prognostic value of molecular remission in a model together with clinical remission status and MIPI prognostic score at diagnosis. The significance level was 5%. Statistical analyzes were performed using SAS 9.1, SAS Institute Inc., Cary, NC, USA.
Results
Patients and samples
From 01/01/2004 until 16/10/2008 760 patients with central review-confirmed MCL were randomized within the intergroup trials of the MCL net, 600 of these in Germany (n=356) and France (n=244) (figure S1).
Sample recruitment for MRD (at least one sample) comprised 90% of all study patients recruited in Germany and France. In the missing 10% samples were not send according to lacking center or patient compliance because MRD assessment was not a prerequisite for study enrolment. As of October 2008, 259 patients with a molecular marker and at least 2 samples from different time points had been analyzed for MRD, comprising 160 patients treated within the European MCL Younger trial and 99 patients within the MCL Elderly trial (figure S2).
The detection of a suitable molecular marker for RQ-PCR was possible in 90% of all patients with material available for MRD. In case of patients with diagnostic samples not-informative for molecular follow-up, this was principally due to failure to identify a clonal IGH population. MRD was assessed with quantitative allele specific IGH-RQ-PCR (n = 245) or allele specific IGH-BCL- RQ-PCR (n = 14).
Patients analyzed for MRD did not differ from those of the complete study cohort with respect to clinical characteristics (table 1). Overall 315 samples prior to treatment (210 PB, 105 BM) and 1324 follow-up samples (907 PB, 417 BM) were investigated. All 315 baseline samples demonstrated lymphoma infiltration by RQ-PCR.
Table 1.
Baseline clinical characteristics
| all | MCL Younger | MCL Elderly | ||
|---|---|---|---|---|
| cohort analyzed for MRD | ||||
| Variable | Value | n=259 | n=160 | n=99 |
| Age (years) | median (range) | 61 (33–81) | 55 (33–65) | 70 (60–81) |
| Sex stage | Male | 77% | 81% | 71% |
| 2 | 2% | 1% | 4% | |
| 3 | 14% | 15% | 12% | |
| 4 | 84% | 84% | 84% | |
| B-symptoms | Yes | 39% | 39% | 39% |
| LDH | Elevated | 39% | 38% | 41% |
| Extra nodal | Yes | 34% | 37% | 29% |
| Bone marrow | yes | 81% | 83% | 79% |
| MIPI risk | Low Risk | 43% | 65% | 8% |
| Intermediate Risk | 34% | 23% | 52% | |
| High Risk | 23% | 12% | 40% | |
Comparability of PB and BM for MRD detection
With regard to the ease of access to MRD samples in clinical routine we addressed the question of comparability of PB and BM for MRD assessment. At diagnosis, 95 paired BM and PB samples demonstrated similar levels of lymphoma cells with a median level of 7.1×10−2 in BM and 5.6×10−2 in PB (Pearson r=0.82, concordance correlation coefficient c=0.81).
After induction, 31 of 108 paired samples were concordantly positive in PB and BM and 50 were negative in both. In 21 paired samples discordant results were obtained with MRD negative PB but low-level MRD detectable in the corresponding BM. In contrast, only 6 BM samples failed to demonstrate persistent disease when the corresponding PB sample was MRD positive. Thus, PB analysis after induction underestimated MRD in about 19% of patients.
Kinetics of MRD
Circulating lymphoma cells (CLC) were assessed by using 4C-FC in 157 of 210 patients with available peripheral blood at diagnosis. Due to different sample processing peripheral blood samples of 53 patients were not directly accessible for CLC by 4C-FC.
Although the vast majority had no leukemic MCL by clinical parameters, all 157 patients showed CLC at a median level of 6.3×10−2 (range 2.0×10−4 to 8.3×10−1). Levels of CLC correlated significantly with the following parameters: stage (median 5.4×10−3 stage 2, 2.4×10−2 stage 3 and 7.6×10−2 stage 4, p=0.0023), elevated LDH (1.2×10−1 vs. 4.7×10−2, p= 0.0021), histological BM infiltration (7.8×10−2 vs. 1.9×10−2 p=0.0002) and MIPI prognostic index (2.7×10−2 vs. 7.9×10−2 vs. 3.3×10−1 for low, intermediate and high risk respectively, p<0.0001) (figure S3).
Monitoring of MRD kinetics during induction was possible in 190 patients and showed that induction treatment with combined immunochemotherapy protocols rapidly reduced the tumor cell load. At midterm staging, 59/190 (31%) patients achieved a MR corresponding to a median 3-log tumor cell reduction (figure 2). The median lymphoma cell level prior to treatment were comparable in PB and BM samples (6.2×10−2 compared to 7.1×10−2 (range 2.0×10−4 to 9.0×10−1 in BM and 8.3×10−1 in PB) and were significantly reduced to a median of 1.0×10−4 in PB and 1.2×10−4 in BM at midterm staging (p<0.0001) and to MRD negativity at end of induction in both (p<0.0001) (figure 2).
Figure 2. MRD quantification by RQ-PCR of 190 patients prior to, during and after induction.
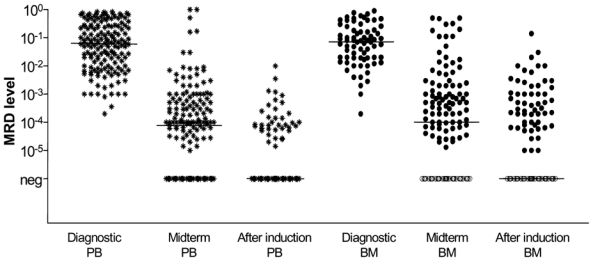
Combined immunochemotherapy resulted in a significant 3 log reduction of lymphoma cells in PB and BM from 6.2×10−2 in PB and 7.0×10−2 in BM prior treatment to 1.0×10−4 in PB and 1.2×10−4 in BM at midterm staging (p<0.0001) and to MRD negativity after end of induction (prior to DexaBEAM/ASCT or maintenance). Filled black symbols for MRD positive samples, empty symbols for MRD negative samples.
Clinical response to treatment
207/259 patients with MRD data were evaluable for clinical response after induction with 65 patients (31%) achieving a CR and 135 patients achieving a CRu or PR (ORR 97%). For patients of the MCL Younger and MCL Elderly trial the CR rate was 32% and 31%, and the ORR 99% and 95%, respectively. To date, 27 patients relapsed (12 in MCL Younger and 15 in MCL Elderly) and 5 patients died in remission (one in MCL Younger and 4 in MCL Elderly). With a median observation time of 17 months, the median response duration has not been reached with 75% patients in remission at 2 years (MCL Younger: median response duration not reached, patients in remission at 2 years 84%; MCL Elderly median response duration 37 months, patients in remission at 2 years 58%). Of 225 patients evaluable for overall survival 26 have died, with median OS not reached and 2 years OS of 86%.
Achievement of Molecular Remission
Molecular remission (MR) after induction treatment was achieved by 106 of 190 patients with MRD data (56%). Interestingly, MCL Elderly patients achieved a MR (54/81, 67%) more frequently compared to MCL Younger patients (MR 52/109, 48%) (p=0.012) despite a higher number of patients with an adverse MIPI score (MIPI high risk 49% vs. 12%).
MRD persistence after induction correlated with the following pretreatment parameters: stage (0%, 36%, 47% positive for stages II, III, and IV respectively, p=0.04), the presence of B-symptoms (53% vs. 38%, p=0.051), LDH above normal level (55% vs. 38%, p=0.024) and BM infiltration (49% vs. 25%, p=0.015). Patients achieving a clinical CR and CRu had a higher probability of obtaining a MR (both 70%) than patients achieving a PR (42%, p=0.0018).
Prognostic relevance of Molecular Remission
One hundred and fifty-six patients with MRD data and a documented clinical remission after induction were evaluable for assessment of the prognostic impact of MRD (figure S2). Patients achieving a MR after induction (n=87) demonstrated a significantly improved response duration compared to patients with residual disease (n=69) (patients in remission at 2 years 87% vs. 61%, p=0.0043) (figure 3a).
Figure 3.
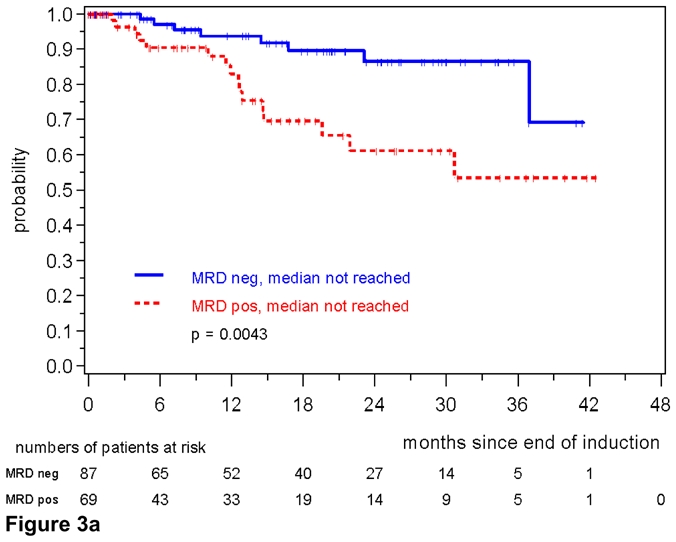
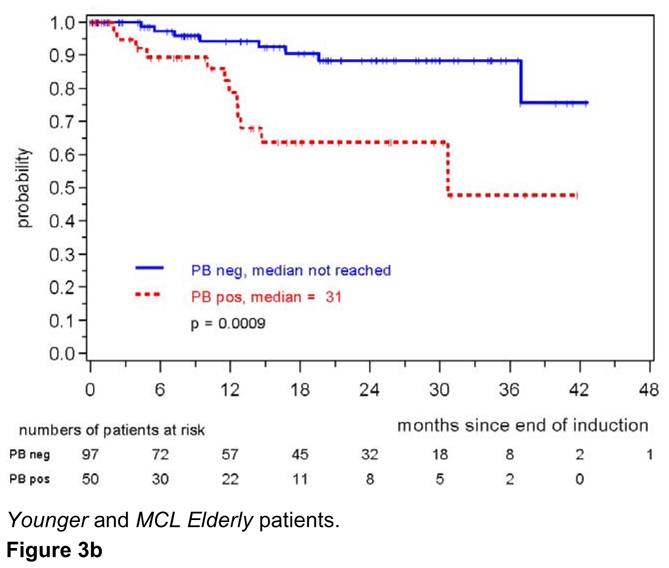
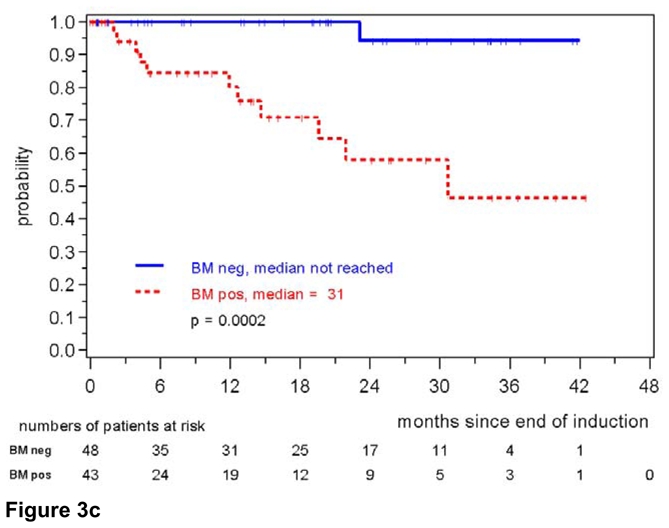
Figure 3a. Response duration according to MRD status after combined immunochemotherapy. MRD was assessed in PB and/or BM after end of induction in MCL Younger and MCL Elderly patients.
Figure 3b: Response duration according to MRD status assessed in the peripheral blood after induction with combined immunochemotherapy. MRD was assessed in MCL Younger and MCL Elderly patients.
Figure 3c: Response duration according to MRD status assessed in the bone marrow after induction with combined immunochemotherapy. MRD was assessed in MCL Younger and MCL Elderly patients.
The high impact of MR on response duration was also confirmed when only PB was analyzed (147 patients, patients in remission at 2 years: 88% for MRD negative patients compared to 64% of MRD positive patients, p=0.0009) (figure 3b). 8/97 (8%) patients in the PB-MRD negative cohort relapsed compared to 12/50 (24%) patients in the MRD positive group. The impact of MRD was even more prominent when only BM was assessed (n=91, patients in remission at 2 years 94% in MRD negative vs. 58% in MRD positive patients, p=0.0002, figure 3c). Only one patient relapsed in the BM-MRD negative group compared to 11 patients with detectable residual disease.
We also compared the significance of MRD in PB and BM within the clinical response groups after induction (CR/Cru and PR). Notably, MRD status in PB and BM was a much better predictor for response duration than the clinical response status (CR/CRu/PR). MR in the BM correlated with a significant prolongation of response duration compared to MRD positive CR or MRD positive CRu/PR patients (patients in remission at 2 years: 94% in MRD negative CR/Cru and 100% in MRD negative PR, compared to 71% in MRD positive CR/Cru and 51% in MRD positive PR, p=0.0023, (figure 4). This was also reproducible when PB alone was investigated (patients in remission at 2 years: 92% in MRD negative CR/Cru and 83% in MRD negative PR, compared to 85% in MRD positive CR/Cru and 55% in MRD positive PR, p=0.0029) (data not shown)
Figure 4. Response duration according to MRD status and clinical remission (CR/Cru/PR).
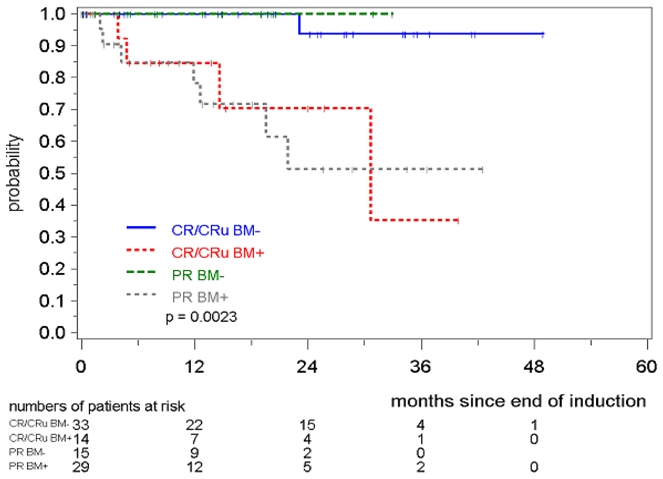
MRD was assessed in the bone marrow after induction with combined immunochemotherapy in MCL Younger and MCL Elderly patients.
MRD was prognostically significant independent of assignment to the MCL Younger or MCL Elderly trial. MRD negativity after induction was reached in 46/91 evaluable MCL Younger patients and 41/65 MCL Elderly patients and was associated with a better prognosis in both cohorts (MCL Younger: patients in remission at 2 years: 94% vs. 74%, p=0.0216, figure 5, MCL Elderly: 77% vs. 34%, p= 0.0207, figure 6). In both trials the impact of MRD on prognosis was confirmed when PB as well as BM was analyzed (figures S4 to S7).
Figure 5.
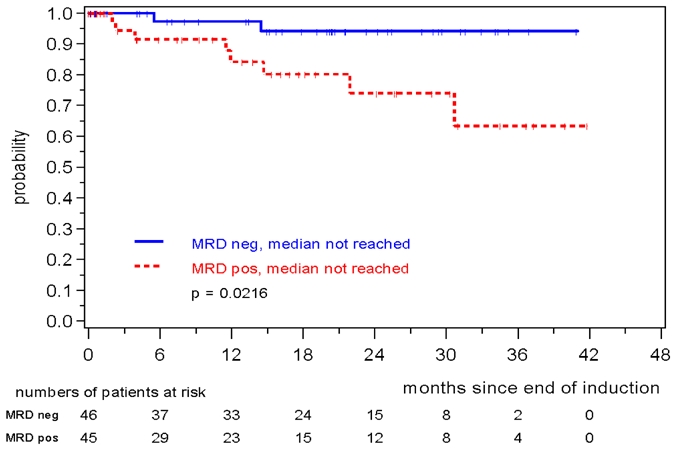
Response duration according to MRD status assessed in PB and/or BM after induction with combined immunochemotherapy in MCL Younger patients.
Figure 6.
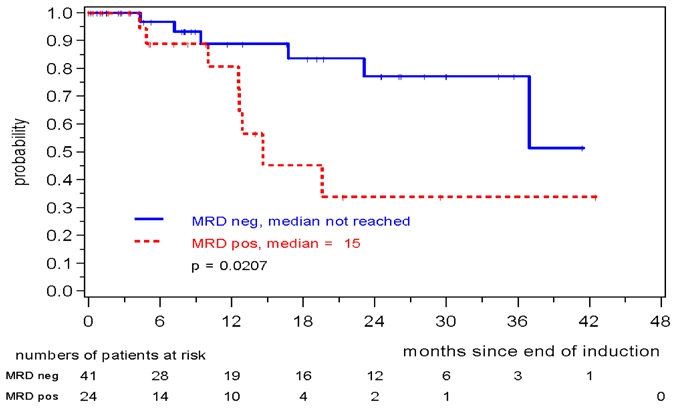
Response duration according to MRD status assessed in PB and/or BM after induction with combined immunochemotherapy in MCL Elderly patients.
A Cox regression model was used to evaluate the prognostic significance of MR together with achievement of CR/CRu vs. PR and pretreatment clinical variables summarized in the continuous MIPI score. Achievement of a MR after induction (HR 0.4, 95% CI 0.1–0.9, p=0.027) turned out to be an important prognostic factor for response duration independent from MIPI (HR 3.2, 95% CI 1.8–5.6, p<0.0001) and achievement of CR (HR 0.7, 95% CI 0.2–2.0, p=0.49).
MRD assessment in the post-induction period
Within both trials MRD assessments were performed at 2 to 3 monthly intervals during the first 12 months of the post-induction period (after ASCT in MCL Younger patients and during maintenance in MCL Elderly patients) to evaluate the prognostic impact of a sustained molecular remission. MRD data were pooled from a median of 3 (1 to 8) samples and the MRD status was judged as MRD positive if at least one sample was positive by RQ-PCR.
Amongst 60 MCL Younger patients all 41 patients with a consistently negative MRD status within the first year after ASCT remained in continuous clinical remission whereas 5/19 patients with at least one MRD positive sample within this period relapsed (patients in remission at 2 years 100% vs. 65%, medians not reached, p=0.0007) (figure 7). This was also confirmed when either only PB (n=57, p=0.0039) or only BM (n=37, p=0.0142 were assessed for the presence of MRD (data not shown).
Figure 7. Response duration according to MRD status assessed in PB and/or BM within the first 12 months after ASCT in MCL Younger patients.
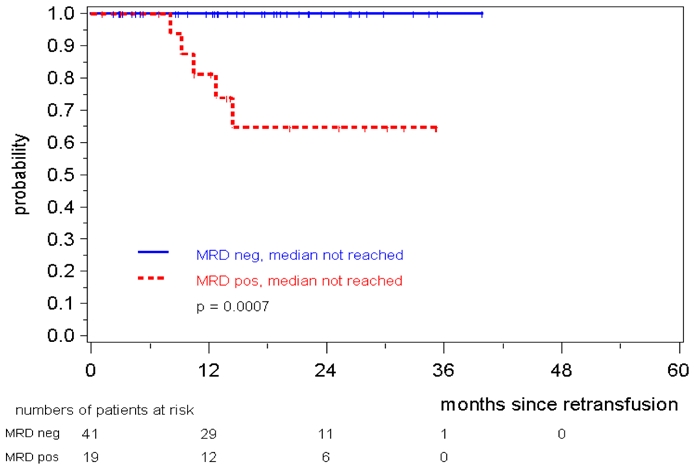
MRD status was judged as MRD positive if at least one sample of in median 3 was positive.
Similarly, in 51 MCL Elderly patients a consistently negative MRD status during the first year of maintenance was associated with a prolonged response duration, 76% of patients in MR were in clinical remission at 24 months compared to only 36% of the patients with residual disease (p=0.015) (figure 8).
Figure 8.
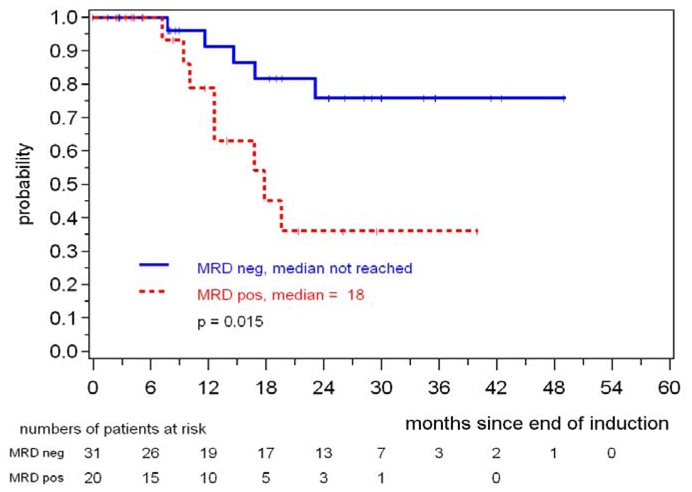
Response duration according to MRD status assessed in PB and/or BM during the first year of maintenance in MCL Elderly patients.
For younger and elderly patients the impact of sustained MRD-negativity could be confirmed in landmark analysis. Only patients in ongoing clinical remission at the respective time point 3, 6 and 12 months after induction treatment (MCL Elderly trial) or ASCT (MCL younger trial) were included in this analysis demonstrating that during all 3 time periods a sustained molecular remission retains its high clinical significance (FS8-S13)
Hence, these results demonstrate that a consistently negative MRD status in patients achieving clinical remission has a strong impact on prognosis independent from the treatment regimen that was applied to achieve it.
Impact of high-dose treatment on Molecular Remission
A direct comparison of the impact of high-dose treatment followed by ASCT on tumor cell reduction could be performed in 67 MCL Younger patients with DNA available after induction treatment and after ASCT. Amongst 67 MCL Younger patients, 37 were MRD negative after induction treatment (55%). High-dose treatment followed by ASCT increased the MR rate to 72% (48/67, p = 0.0116, Mc Nemar test) demonstrating a significant impact of high-dose treatment on tumor reduction.
Discussion
Today, a spectrum of highly effective but potentially toxic treatment modalities is available for patients with MCL. This makes sensitive and reliable assessment of treatment efficacy allowing individual estimation of response duration desirable in order to optimize patient care as well as trial design. Previous retrospective data from our group suggested that quantitative measurement of MRD during and after treatment may provide an excellent tool to achieve this objective.26
Those earlier MRD data were in keeping with the notion that persistence of residual lymphoma cells is considered to be the principal reason for relapse 21;23. However, in those earlier series patients did not receive rituximab as a part of induction treatment. We hypothesized that MRD assessment as a dynamic parameter might contribute to the prediction of prognosis, independently from pretreatment risk profile (MIPI score). This analysis was particularly important as previous data addressed this question retrospectively in small patient cohorts only. 21;23;26
The aim of our study was therefore (1) to assess the prognostic relevance of MRD in the context of two large prospective trials investigating various immunochemotherapy regimens with different consolidation treatments, and (2) to study the individual effects of different treatment components on response kinetics and tumor load in MCL patients.
Because of easier access and patient comfort MRD analysis of PB would be preferable to BM assessment. This is of particular importance as rituximab based treatment protocols induce profound peripheral B-cell depletion that might lead to a discordant PB MRD status in comparison with the clinical disease status and prognosis may be altered by prior Rituximab use due to a preferential clearance of disease from the PB compartment.
Therefore we addressed the question of the comparability of both sources for MRD assessment prior to and after combined immunochemotherapy. By analyzing 95 paired PB and BM pre-treatment samples we found comparable median CLC levels in PB and BM. However, the situation changes when paired samples after start of immunochemotherapy were compared. Analysis of PB alone failed to demonstrate persistent lymphoma cells in about 19% of patients who were simultaneously positive in BM. We thus demonstrate herein that rituximab-based immunochemotherapy more effectively clears lymphoma cells from PB than from BM. This phenomenon has been reported for alemtuzumab administered in CLL38 but is in contrast to our earlier findings in MCL demonstrating a similar predictive value of MRD assessment in PB or BM also after rituximab-free treatment. 26 Accordingly, in the present study BM MR predicted more accurately for an event-free clinical course than PB MR although even the latter was associated with a clearly superior outcome. Therefore, source of material for MRD assessment remains an important issue which must be taken into account when designing clinical trials or using MRD results as basis for consolidation or preemptive treatment decisions.
In this study MRD assessment allowed for the first time direct evaluation of the impact of individual treatment elements on tumor cell reduction within a multimodal protocol in patients with MCL. Immunochemotherapy rapidly caused a median MRD reduction of 3 log by midterm staging and induced MR in 56% of the cases after end of induction. This is in marked contrast to our previous work showing that 4–6 cycles of CHOP alone did not significantly reduce MRD levels, and none of those patients achieved MR after induction 26. This observation correlates with the superior clinical response rate of R-CHOP over CHOP in MCL 5 but can also be influenced by the potential effect of the different induction regimens that are tested within the two trials (R-DHAP and R-FC). Thus, our data suggest that rituximab improves the efficacy of CHOP chemotherapy that on its own shows only limited activity in MCL.
With regard to prediction of prognosis achievement of MR after induction was highly correlated with prolonged response duration independent of the study protocol applied. A favorable outcome could be predicted early by MRD assessment of PB or preferably BM at a single time point after induction. Thus, achieving MR after induction is clearly a desirable goal in the treatment of patients with MCL. It remains unclear, however, if the better sensitivity to cytotoxic treatment as documented by achievement of MR simply reflects a more “benign” biological profile of the tumor in individual patients, or if MR has prognostic impact per se.
Our results are in contrast to published data suggesting that achievement of MR in MCL after rituximab and CHOP or other chemotherapy schemes has no impact on prognosis 39. Although in the study by Howard et al. 9/25 patients with PCR-detectable disease at diagnosis achieved a molecular remission, no differences in outcome according to MRD status could be demonstrated. This can in part be explained by the small numbers of patients but also by technical aspects. Most previous reports on MRD detection within multimodal treatment protocols were based on qualitative PCR approaches that appear less reliable for PCR-based risk stratification in MCL due to varying sensitivity and the lack of standardized evaluation. Thus, our results underline once more the importance of highly standardized, sensitive and quantitative RQ-PCR assays for MRD assessment 26 as this has already been documented previously by our group.
Quality of clinical remission after treatment is thought to be one of the strongest parameters for prognosis in patients with MCL 5;13;40. Of note, in this series the prognostic impact of MR in responding patients exceeded that of clinical remission status. Patients achieving a MRD-negative CR or PR had a response duration which was superior to that of patients with a MRD-positive CR or PR. This implies that prediction of outcome by MRD is much more meaningful than by quality of clinical remission and strongly suggests that future treatment protocols should incorporate MRD assessment in response evaluation of MCL treatment. Similar observations on the prognostic impact of MRD have recently been reported in CLL where a MRD-negative status was the strongest predictor of clinical outcome superior to quality of 41;42 response or low level of MRD during and after induction were identified as prognostic parameter for PFS. 43 Also in MCL it will be useful to investigate the prognostic value of different MRD cut-offs in PCR positive patients to precisely define clinical risk groups.
In a multivariate analysis including the parameters MIPI and quality of clinical response in addition to MRD the achievement of MR after induction turned out to be an independent prognostic factor for response duration. These results document for the first time the independent prognostic value and high clinical relevance of MRD after combined immunochemotherapy in MCL. Comparable results in a prospective series have only been published in patients in follicular lymphoma undergoing treatment with 6 courses of CHOP followed by rituximab (CHOP-R) or -supplemented high-dose sequential chemotherapy with autografting (R-HDS). In this entity molecular remission was achieved in 44% of R-CHOP and 80% of R-HDS patients (P <0.001) 25 representing the strongest predictor of outcome.
MRD kinetics was also analyzed in the post-induction period, after ASCT consolidation in MCL Younger patients and during maintenance treatment in MCL Elderly patients.
In younger patients MRD kinetics clearly demonstrated that high-dose radio-chemotherapy followed by ASCT can further reduce the tumor load even if applied immediately after immunochemotherapy. The rate of MR increased from 55% post-induction to 72% within the first year after ASCT. These data impressively demonstrate the anti-lymphoma activity of high-dose radio chemotherapy in MCL as already inferred from clinical data 9;10;13;14 and our previous observations 26. On the other hand, patients in whom neither rituximab containing induction nor the high-dose consolidation induced MR had a significantly inferior outcome. This might suggest that for future clinical trials the post-ASCT rather than the post-induction MRD status is an excellent tool for identifying patients in need for further treatment intensification or consolidation. However, the post-induction MRD status has the advantage of providing the prognostic information more timely, thereby allowing for better planning of potential post-consolidation treatment intensification or maintenance treatment.
In summary, the addition of rituximab into the treatment of MCL has clearly improved the outcome of patients with MCL as shown by several trials10–12. The present study demonstrates for the first time, that prospective longitudinal monitoring of MRD after combined immunochemotherapy can be a powerful predictor of treatment outcome in patients with MCL allowing an early individual risk assessment already during treatment. Accordingly, MRD assessment should be integrated into future clinical trial concepts to evaluate new treatment strategies and to serve as an early surrogate marker that can be used for risk adapted treatment.
Acknowledgments
This work was supported by the European Community within the European MCL Network LSHC-CT 2004-503351 (CP, EM, MHDL, RS) and by the Lymphoma Research Foundation (CP, SB, MD) Correlative Research grant, ARC (association de recherche contre le Cancer) Subvention number 3730 (MHDL); Fondation de France (FDF) comité Leucémie; Subvention number 2004004029 (MHDL), ARTGIL (Association de recherche sur le traitement, la genétique et l’immunologie des lymphomes) (MHDL).
Appendix
The authors wish to thank the following physicians for collecting samples for MRD analysis:S. Stilgenbauer, B. Coiffier, C. Gisselbrecht, P. Lederlin, R. Forstpointner, M. Hänel, R. Bouabdallah, M. Hallek, M. Pfreundschuh, C. Sebban, H.-G. Mergenthaler, R.O. Casasnovas, C. H. Köhne, L. Kanz, E. Heidemann, C. Haioun, W. Lindemann, R. Mertelsmann, C. Hube, K. Bouabdallah, U. Vehling-Kaiser, M. Jovet, G. Schlimok, H. Tilly, H. Mallek, P. Delaby, M. Uppenkamp, P. Schmidt, H. Fuss, N. Peter, L. Trümper, C. Soussain, T. Fischer, G. Lepeu, T. Wagner, C. Peschel, S. Kremers, R. Andreesen, W.-J. Mayet, M. Macro, U. Dührsen, G. Maschmeyer, J. Wehmeyer, U. Haak, L. Balleisen, E. Jourdan, O. Fitoussi, L. Sanhes, D. Metzner, B. Kempf, W. Aulitzky, T. Reiber, W.E. Berdel, E. Kettner, B. Audhuy, N. Fegueux, M. Wilhelm, F. Griesinger, N. Schmitz, A. Neubauer, E. Lengfelder, G. Obst, A. Devidas, K. Höffken, M. Geißler, F. Boue, D. Semsek, J. Gabarre, U. R. Kleeberg, M. Boisseau, D.W. Beelen, M. Bentz, S. Klein, J. Janssen, Hentrich, C. Martin, P. Fenaux, S. Prenninger, A. Denizon, C. Fruchart, A. Grüneisen, C. Bokemeyer, S. Castaigne, W. Heit, W. Helmer, M. R. Clemens, O. Fain, W. Schmiegel, G. Dölken, S. Siehl, T. Ulshöfer, M. Sieber, R. Scholten, N. Brüllke, H. Orfeuvre, G. Salles, L. Aljassem, W.-D. Ludwig, N. Schmitz, W. Gassmann, M. Grundheber, D. Guggenberger, C. Salanoubat, U. Graeven, M. Freund, J. Grunst, E. Höring, B. Wörmann, M. Agape, M. Fabbro, A. Belhabri, S. Fetscher, H. J. Pielken, L. Heidenreich, T. Kamp, E. Thiel, I. Wehle, R. Weide, S. Hahnfeld, F. Busch, M. Stauch, M. Sandmann, O. Prümmer, F. A. Mosthaf, H. Hebart, G. Meckenstock, F. Bauduer, F. Klump, Y. Kerneis, L. Müller, C. Schadeck-Gressel, C. Priebe-Richter, H.-D. Harich, G. Michl, J. Mezger, A. Ganser, L. Theilmann, V. Lakner, M. Demandt, W. Ladda, J. Mittermüller, G. Trenn, H. Pohl, F. Dilenon, C. Aul, J. Kubin, B. Rendenbach, S. Detken, L. Fischer v. Weikersthal, M. Adler, E. Schleyer, G. Sebahoun
We thank the EU-MCL reference pathology panel for pathologic review: T. Barth; F. Berger; W. Bernd; N. Brousse; J. Cabecadas; S. Caulet; E. Campo; S. Cogliatti; A.C. Feller, M.L. Hansmann; W. Klapper; P. Kluin; R. Kodet; J. van Krieken C. Loddenkemper; P. Möller; H. K. Müller-Hermelink; G. Ott; S. Pileri; E. Ralfkiaer, A. Rosenwald; G. Rymkiewicz; H. S tein;
Footnotes
Specific contributions of all authors to published work
CP guided MRD analysis and interpretation and drafted the manuscript. CP, MHD, KB and EM, NA were responsible for sample and data collection and interpretation. JJVD assisted in data interpretation, EH conducted the statistical analysis of clinical and MRD data, MU is the responsible statistician of the EU-MCL Study Group. SB, VA and AP were responsible for flow cytometry, WK and FB performed the pathological panel review, EC-B and RS were responsible for cytogenetic and molecular cytogenetic data collection and interpretation. MD is the coordinator of the European MCL trials and together with WH, OH, HK-N, VR, AVH, MT and JW responsible for its clinical trial design and conduct. PD participated in data interpretation and manuscript preparation. MK was responsible for the overall conduct of the MRD study and participated in manuscript preparation.
Conflict of interest disclosure: The authors declare no competing financial interests.
References
- 1.Herrmann A, Hoster E, Zwingers T, et al. Improvement of overall survival in advanced stage mantle cell lymphoma. J Clin Oncol. 2009;27(4):511–518. doi: 10.1200/JCO.2008.16.8435. [DOI] [PubMed] [Google Scholar]
- 2.Meusers P, Engelhard M, Bartels H, et al. Multicentre randomized therapeutic trial for advanced centrocytic lymphoma: anthracycline does not improve the prognosis. Hematol Oncol. 1989;7(5):365–380. doi: 10.1002/hon.2900070505. [DOI] [PubMed] [Google Scholar]
- 3.Fu K, Weisenburger DD, Greiner TC, et al. Cyclin D1-negative mantle cell lymphoma: a clinicopathologic study based on gene expression profiling. Blood. 2005;106(13):4315–4321. doi: 10.1182/blood-2005-04-1753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Siebert R, Matthiesen P, Harder S, et al. Application of interphase cytogenetics for the detection of t(11;14)(q13;q32) in mantle cell lymphomas. Ann Oncol. 1998;9(5):519–526. doi: 10.1023/a:1008242729509. [DOI] [PubMed] [Google Scholar]
- 5.Lenz G, Dreyling M, Hoster E, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: results of a prospective randomized trial of the german low grade lymphoma study group (GLSG) J Clin Oncol. 2005;23(9):1984–1992. doi: 10.1200/JCO.2005.08.133. [DOI] [PubMed] [Google Scholar]
- 6.Romaguera JE, Khouri IF, Kantarjian HM, et al. Untreated aggressive mantle cell lymphoma: results with intensive chemotherapy without stem cell transplant in elderly patients. Leuk Lymphoma. 2000;39(1):77–85. doi: 10.3109/10428190009053541. [DOI] [PubMed] [Google Scholar]
- 7.Romaguera JE, Fayad L, Rodriguez MA, et al. High rate of durable remissions after treatment of newly diagnosed aggressive mantle-cell lymphoma with rituximab plus hyper-CVAD alternating with rituximab plus high-dose methotrexate and cytarabine. J Clin Oncol. 2005;23(28):7013–7023. doi: 10.1200/JCO.2005.01.1825. [DOI] [PubMed] [Google Scholar]
- 8.van ‘t Veer MB, de JD, MacKenzie M, et al. High-dose ara-c and BEAM with autograft rescue in R-CHOP responsive mantle cell lymphoma patients. Br J Haematol. 2009;144(4):524–530. doi: 10.1111/j.1365-2141.2008.07498.x. [DOI] [PubMed] [Google Scholar]
- 9.Dreger P, von NN, Kuse R, et al. Sequential high-dose therapy and autologous stem cell transplantation for treatment of mantle cell lymphoma. Ann Oncol. 1997;8(4):401–403. doi: 10.1023/a:1008251301319. [DOI] [PubMed] [Google Scholar]
- 10.Dreger P, Rieger M, Seyfarth B, et al. Rituximab-augmented myeloablation for first-line autologous stem cell transplantation for mantle cell lymphoma: effects on molecular response and clinical outcome. Haematologica. 2007;92(1):42–49. doi: 10.3324/haematol.10608. [DOI] [PubMed] [Google Scholar]
- 11.Geisler CH, Kolstad A, Laurell A, et al. Long-term progression-free survival of mantle cell lymphoma after intensive front-line immunochemotherapy with in vivo-purged stem cell rescue: a nonrandomized phase 2 multicenter study by the Nordic Lymphoma group. Blood. 2008;112(7):2687–2693. doi: 10.1182/blood-2008-03-147025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gianni AM, Magni M, Martelli M, et al. Long-term remission in mantle cell lymphoma following high-dose sequential chemotherapy and in vivo rituximab-purged stem cell autografting (R-HDS regimen) Blood. 2003;102(2):749–755. doi: 10.1182/blood-2002-08-2476. [DOI] [PubMed] [Google Scholar]
- 13.Vandenberghe E, Ruiz de Elvira C, Loberiza FR, et al. Outcome of autologous transplantation for mantle cell lymphoma: a study by the European blood and bone marrow transplant and autologous blood and marrow transplant registries. Br J Haematol. 2003;120(5):793–800. doi: 10.1046/j.1365-2141.2003.04140.x. [DOI] [PubMed] [Google Scholar]
- 14.Dreyling M, Lenz G, Hoster E, et al. early consolidation by myeloablative radiochemotherapy followed by autologous stem cell transplantation in first remission significantly prolongs progression-free survival in mantle-cell lymphoma: results of a prospective randomized trial of the European MCL Network. Blood. 2005;105(7):2677–2684. doi: 10.1182/blood-2004-10-3883. [DOI] [PubMed] [Google Scholar]
- 15.Delarue R, Haioun C, Ribrag V, et al. R-CHOP and R-DHAP followed by autologous stem cell transplantation (ASCT) in mantle cell lymphoma (MCL): Final results of a phase II study from the GELA [abstract] Blood. 2008;112(11):581. [Google Scholar]
- 16.Lefrere F, Delmer A, Levy V, et al. Sequential chemotherapy regimens followed by high-dose therapy with stem cell transplantation in mantle cell lymphoma: an update of a prospective study. Haematologica. 2004;89(10):1275–1276. [PubMed] [Google Scholar]
- 17.Ritchie DS, Seymour JF, Grigg AP, et al. The hyper-CVAD-rituximab chemotherapy programme followed by high-dose busulfan, melphalan and autologous stem cell transplantation produces excellent event-free survival in patients with previously untreated mantle cell lymphoma. Ann Hematol. 2007;86(2):101–105. doi: 10.1007/s00277-006-0193-2. [DOI] [PubMed] [Google Scholar]
- 18.Khouri IF, Romaguera J, Kantarjian H, et al. Hyper-CVAD and high-dose methotrexate/cytarabine followed by stem-cell transplantation: an active regimen for aggressive mantle-cell lymphoma. J Clin Oncol. 1998;16(12):3803–3809. doi: 10.1200/JCO.1998.16.12.3803. [DOI] [PubMed] [Google Scholar]
- 19.Hoster E, Dreyling M, Klapper W, et al. A new prognostic index (MIPI) for patients with advanced-stage mantle cell lymphoma. Blood. 2008;111(2):558–565. doi: 10.1182/blood-2007-06-095331. [DOI] [PubMed] [Google Scholar]
- 20.Ritgen M, Bottcher S, Stilgenbauer S, et al. Quantitative MRD monitoring identifies distinct GVL response patterns after allogeneic stem cell transplantation for chronic lymphocytic leukemia: results from the GCLLSG CLL3X trial. Leukemia. 2008;22(7):1377–1386. doi: 10.1038/leu.2008.96. [DOI] [PubMed] [Google Scholar]
- 21.Andersen NS, Donovan JW, Borus JS, et al. Failure of immunologic purging in mantle cell lymphoma assessed by polymerase chain reaction detection of minimal residual disease. Blood. 1997;90(10):4212–4221. [PubMed] [Google Scholar]
- 22.Corradini P, Ladetto M, Astolfi M, et al. Clinical and molecular remission after allogeneic blood cell transplantation in a patient with mantle-cell lymphoma. Br J Haematol. 1996;94(2):376–378. doi: 10.1046/j.1365-2141.1996.d01-1816.x. [DOI] [PubMed] [Google Scholar]
- 23.Corradini P, Astolfi M, Cherasco C, et al. Molecular monitoring of minimal residual disease in follicular and mantle cell Non-Hodgkin’s lymphomas treated with high-dose chemotherapy and peripheral blood progenitor cell autografting. Blood. 1997;89(2):724–731. [PubMed] [Google Scholar]
- 24.Ladetto M, Corradini P, Vallet S, et al. High rate of clinical and molecular remissions in follicular lymphoma patients receiving high-dose sequential chemotherapy and autografting at diagnosis: a multicenter, prospective study by the gruppo italiano trapianto midollo osseo (GITMO) Blood. 2002;100(5):1559–1565. doi: 10.1182/blood-2002-02-0621. [DOI] [PubMed] [Google Scholar]
- 25.Ladetto M, De Marco F, Benedetti F, et al. Prospective, multicenter randomized GITMO/IIL trial comparing intensive (R-HDS) versus conventional (CHOP-R) chemoimmunotherapy in high-risk follicular lymphoma at diagnosis: the superior disease control of R-HDS does not translate into an overall survival advantage. Blood. 2008;111(8):4004–4013. doi: 10.1182/blood-2007-10-116749. [DOI] [PubMed] [Google Scholar]
- 26.Pott C, Schrader C, Gesk S, et al. Quantitative assessment of molecular remission after high-dose therapy with autologous stem cell transplantation predicts long-term remission in mantle cell lymphoma. Blood. 2006;107(6):2271–2278. doi: 10.1182/blood-2005-07-2845. [DOI] [PubMed] [Google Scholar]
- 27.Rawstron AC, Kennedy B, Evans PAS, et al. Quantitation of minimal disease levels in chronic lymphocytic leukemia using a sensitive flow cytometric assay improves the prediction of outcome and can be used to optimize therapy. Blood. 2001;98(1):29–35. doi: 10.1182/blood.v98.1.29. [DOI] [PubMed] [Google Scholar]
- 28.Andersen NS, Donovan JW, Zuckerman A, et al. Real-time polymerase chain reaction estimation of bone marrow tumor burden using clonal immunoglobulin heavy chain gene and bcl-1/jh rearrangements in mantle cell lymphoma. Exp Hematol. 2002;30(7):703–710. doi: 10.1016/s0301-472x(02)00807-x. [DOI] [PubMed] [Google Scholar]
- 29.Swerdlow SH, Campo E, Seto M, Müller-Hermelink HK. Mature B-cell neoplasms: mantle cell lymphoma. In: Swerdlow SH, Campo E, Harris NL, et al., editors. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Lyon: IARC Press; 2008. pp. 229–232. [Google Scholar]
- 30.Dreyling M, Hoster E, Hermine O, et al. European MCL Network: An update on current first line trials. [abstract] Blood 20071101138817360939 [Google Scholar]
- 31.Bottcher S, Ritgen M, Buske S, et al. Minimal residual disease detection in mantle cell lymphoma: methods and significance of four-color flow cytometry compared to consensus igh-polymerase chain reaction at initial staging and for follow-up examinations. Haematologica. 2008;93(4):551–559. doi: 10.3324/haematol.11267. [DOI] [PubMed] [Google Scholar]
- 32.Pott C, Tiemann M, Linke B, et al. Structure of BCL-1 and IGH-CDR3 rearrangements as clonal markers in mantle cell lymphomas. Leukemia. 1998;12(10):1630–1637. doi: 10.1038/sj.leu.2401172. [DOI] [PubMed] [Google Scholar]
- 33.van Dongen JJ, Langerak AW, Bruggemann M, et al. Design and standardization of PCR primers and protocols for detection of clonal immunoglobulin and T-cell receptor gene recombinations in suspect lymphoproliferations: report of the biomed-2 concerted action BMH4-CT98-3936. Leukemia. 2003;17(12):2257–2317. doi: 10.1038/sj.leu.2403202. [DOI] [PubMed] [Google Scholar]
- 34.Pongers-Willemse MJ, Verhagen OJ, Tibbe GJ, et al. Real-time quantitative PCR for the detection of minimal residual disease in acute lymphoblastic leukemia using junctional region specific taqman probes. Leukemia. 1998;12(12):2006–2014. doi: 10.1038/sj.leu.2401246. [DOI] [PubMed] [Google Scholar]
- 35.van der Velden VHJ, Cazzaniga G, Schrauder A, et al. Analysis of minimal residual disease by IG/TCR gene rearrangements: guidelines for interpretation of real-time quantitative PCR data. Leukemia. 2007;21(4):604–611. doi: 10.1038/sj.leu.2404586. [DOI] [PubMed] [Google Scholar]
- 36.Cheson BD, Bennett JM, Kopecky KJ, et al. Revised recommendations of the international working group for diagnosis, standardization of response criteria, treatment outcomes, and reporting standards for therapeutic trials in acute myeloid leukemia. J Clin Oncol. 2003;21(24):4642–4649. doi: 10.1200/JCO.2003.04.036. [DOI] [PubMed] [Google Scholar]
- 37.Lin LI. A concordance correlation coefficient to evaluate reproducibility. Biometrics. 1989;45(1):255–268. [PubMed] [Google Scholar]
- 38.Lundin J, Kimby E, Bjorkholm M, et al. Phase II trial of subcutaneous anti-CD52 monoclonal antibody alemtuzumab (campath-1H) as first-line treatment for patients with B-cell chronic lymphocytic leukemia (B-CLL) Blood. 2002;100(3):768–773. doi: 10.1182/blood-2002-01-0159. [DOI] [PubMed] [Google Scholar]
- 39.Howard OM, Gribben JG, Neuberg DS, et al. Rituximab and CHOP induction therapy for newly diagnosed mantle-cell lymphoma: molecular complete responses are not predictive of progression-free survival. J Clin Oncol. 2002;20(5):1288–1294. doi: 10.1200/JCO.2002.20.5.1288. [DOI] [PubMed] [Google Scholar]
- 40.Andersen NS, Pedersen L, Elonen E, et al. Primary treatment with autologous stem cell transplantation in mantle cell lymphoma: outcome related to remission pretransplant. Eur J Haematol. 2003;71(2):73–80. doi: 10.1034/j.1600-0609.2003.00093.x. [DOI] [PubMed] [Google Scholar]
- 41.Bosch F, Ferrer A, Villamor N, et al. Fludarabine, cyclophosphamide, and mitoxantrone as initial therapy of chronic lymphocytic leukemia: high response rate and disease eradication. Clin Cancer Res. 2008;14(1):155–161. doi: 10.1158/1078-0432.CCR-07-1371. [DOI] [PubMed] [Google Scholar]
- 42.Moreton P, Kennedy B, Lucas G, et al. Eradication of minimal residual disease in B-cell chronic lymphocytic leukemia after alemtuzumab therapy is associated with prolonged survival. J Clin Oncol. 2005;23(13):2971–2979. doi: 10.1200/JCO.2005.04.021. [DOI] [PubMed] [Google Scholar]
- 43.Boettcher S, Fischer K, Stilgenbauer S, et al. Quantitative MRD assessment predicts progression free survival in CLL patients treated with fludarabine and cyclophosphamide with or without rituximab – a prospective analysis in 471 patients from the randomized GCLLSG CLL8 trial [abstract] Blood. 2008;112(11):326. [Google Scholar]


