Abstract
The decision of a cell to undergo programmed cell death is tightly regulated during animal development and tissue homeostasis. Here, we show that the Caenorhabditis elegans Six family homeodomain protein C. elegans homeobox (CEH-34) and the Eyes absent ortholog EYA-1 promote the programmed cell death of a specific pharyngeal neuron, the sister of the M4 motor neuron. Loss of either ceh-34 or eya-1 function causes survival of the M4 sister cell, which normally undergoes programmed cell death. CEH-34 physically interacts with the conserved EYA domain of EYA-1 in vitro. We identify an egl-1 5′ cis-regulatory element that controls the programmed cell death of the M4 sister cell and show that CEH-34 binds directly to this site. Expression of the proapoptotic gene egl-1 in the M4 sister cell requires ceh-34 and eya-1 function. We conclude that an evolutionarily conserved complex that includes CEH-34 and EYA-1 directly activates egl-1 expression through a 5′ cis-regulatory element to promote the programmed cell death of the M4 sister cell. We suggest that the regulation of apoptosis by Six and Eya family members is conserved in mammals and involved in human diseases caused by mutations in Six and Eya.
Keywords: cell death, sine oculis, eyes absent, transcription
Apoptosis, also referred to as programmed cell death, plays fundamental roles in animal development and tissue homeostasis (1). The misregulation of apoptosis is associated with many human disorders, including cancer and neurodegenerative and autoimmune diseases (2). Determining how particular cells are specified to live or die is critical to understanding both normal animal development and human diseases associated with the misregulation of apoptotic cell death.
During the development of the Caenorhabditis elegans hermaphrodite, 131 somatic cells undergo programmed cell death (3, 4). Genetic studies of programmed cell death in C. elegans have defined an evolutionarily conserved pathway that executes this process (5). This pathway consists of four genes, egl-1 [egg-laying defective (egl)], ced-9 [cell-death abnormal (ced)], ced-4, and ced-3, all of which are conserved from C. elegans to mammals. Although much is understood about the pathway responsible for the execution of programmed cell death in both C. elegans and other animals, less is known about the mechanisms that control how specific cells decide whether to survive or die by programmed cell death. In C. elegans, most of the genes identified to control cell-death specification encode transcription factors, some of which are known to directly regulate the transcription of cell-death genes (6–11). For example, the Snail family transcription factor CES-1 [cell-death specification (ces)] can directly repress expression of the BH3-only proapoptotic gene egl-1 and prevent the deaths of the NSM sister cells (6, 7). This regulatory mechanism is conserved in mammals and has been implicated in human cancer: acute lymphoblastic leukemia results from an overexpression of the CES-1 homolog Slug, which directly represses expression of the BH3-only proapoptotic gene Puma, preventing apoptosis of hematopoietic progenitor cells (12, 13).
By contrast, the C. elegans Bar homeodomain transcription factor CEH-30 [C. elegans homeobox (ceh)] protects specific neurons from undergoing programmed cell death by acting independently of egl-1 (14); ceh-30 also regulates egl-1 transcription (15). The antiapoptotic function of ceh-30 likely explains the previously observed loss of sensory hair cells of the inner ear and progressive hearing loss of mice deficient in the function of the ceh-30 homolog Barhl1 (16).
The identification and characterization of additional factors that regulate cell type-specific apoptosis in C. elegans promise to reveal both conserved molecular mechanisms that control cell-death specification and how misregulated apoptosis can cause human disease. Here, we show that the C. elegans Six family homeodomain protein CEH-34 and the Eyes absent ortholog EYA-1 directly activate expression of the proapoptotic BH3-only gene egl-1 in the M4 sister cell to promote programmed cell death. We suggest that, in mammals, Six family members and Eya control apoptosis by directly regulating the transcription of BH3-only genes and mutations in Six and Eya genes perturb normal apoptosis by misregulating the transcription of BH3-only genes.
Results
Mutants Defective in the Programmed Cell Death of the M4 Sister Cell.
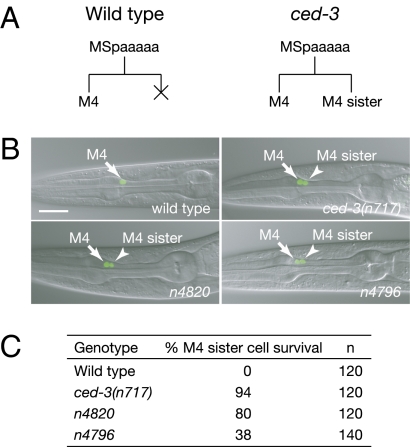
The C. elegans M4 motor neuron is located on the dorsal side of the anterior bulb of the pharynx and regulates pharyngeal muscle contraction during feeding (17, 18). The mother of the M4 neuron divides to generate an anterior daughter that survives and becomes the M4 neuron and a posterior daughter, the M4 sister cell, which dies by programmed cell death during embryogenesis (Fig. 1A) (3). To identify genes that control the specification of the M4 sister-cell death, we performed a genetic screen for mutants with a surviving M4 sister cell. We used a Pceh-28::4xNLS::gfp reporter transgene that is specifically expressed in the M4 neuron in wild-type animals and in both the M4 neuron and the surviving M4 sister cell in ced-3 mutants defective in programmed cell death (Fig. 1 A and B) (19). Thus, this reporter can be used to detect M4 sister-cell survival.
Fig. 1.
Mutants defective in M4 sister-cell death. (A) A schematic representation of the M4 lineage in the wild-type and ced-3 mutants is shown. The M4 sister cell undergoes programmed cell death in the wild-type and survives in ced-3 animals. (B) Merged epifluorescence and Nomarski images of the pharynx in the wild-type, ced-3(n717), n4820, and n4796 animals carrying the Pceh-28::4xNLS::gfp transgene are shown. Arrow, the M4 neuron; arrowhead, the surviving M4 sister cell. (Scale bar, 20 μm.) (C) The penetrances of M4 sister survival in the indicated strains are shown.
We screened the F2 progeny of hermaphrodites mutagenized with ethyl methanesulfonate (EMS) and, from a total of 72,000 haploid genomes screened, isolated 59 mutants with a surviving M4 sister cell, which was located variably as in ced-3 mutants (Fig. 1B and Fig. S1). From these 59 mutants, we identified 38 alleles of ced-3, 13 alleles of ced-4, 3 alleles of egl-1, and 5 mutants that seemed not to carry alleles of any known cell-death genes. Here, we focus on two mutants that affected the deaths of both the M4 sister cell and the pharyngeal I1 sister cells, n4820 and n4796.
In n4820 mutants, 80% of the M4 sister cells (n = 120) and 10% of the I1 sister cells (n = 240) survived (Fig. 1 B and C and Table S1). In n4796 mutants, 38% of the M4 sister cells (n = 140) and 47% of the I1 sister cells (n = 240) survived (Fig. 1 B and C and Table S1). By contrast, the n4820 and n4796 mutations did not affect the programmed cell deaths of the NSM sister cells or the VC homologs of the ventral nerve cord (Table S1). Thus, n4820 and n4796 affect M4 sister-cell and I1 sister-cell deaths but do not affect all programmed cell deaths.
n4820 Is a Noncoding Allele of the Proapoptotic BH3-Only Gene egl-1.
The mutation n4820 complemented ced-3(n717) and ced-4(n1162) but failed to complement egl-1(n1084 n3082) for M4 sister-cell survival (Table S2). We mapped n4820 to chromosome V, the same chromosome as egl-1. Whereas the egl-1(n4820) mutation caused survival of the M4 sister cell and the I1 sister cells but not of other cells fated to die (Table S1), an egl-1 null allele causes survival of essentially all somatic cells that die by programmed cell death (Table S1) (20). These data indicate that egl-1(n4820) is an atypical egl-1 allele that specifically affects the programmed cell deaths of the M4 sister cell and the I1 sister cells.
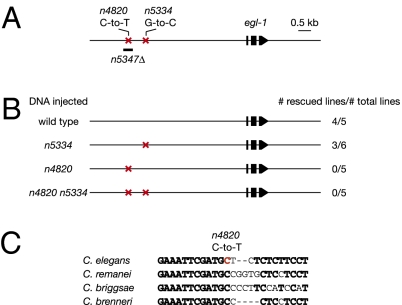
We determined the sequence of the egl-1 locus of egl-1(n4820) mutants and found two mutations. One is a C-to-T mutation located 4.9 kb upstream of the egl-1 start codon, and the other is a G-to-C mutation located 4.2 kb upstream of the egl-1 start codon (Fig. 2A). To determine which mutation caused survival of the M4 sister cell, we performed rescue experiments for the phenotype of M4 sister survival of egl-1(n4820) mutants by injecting an egl-1 genomic fragment containing 6.5 kb upstream of the egl-1 start codon, the egl-1 coding region, and 2.2 kb downstream of the egl-1 stop codon. Four of five transgenic lines that contained a wild-type egl-1 genomic fragment and three of six transgenic lines that contained an egl-1 genomic fragment carrying the G-to-C mutation were rescued for the phenotype of M4 sister survival (i.e., the M4 sister cells died in the rescued lines) (Fig. 2B and Fig. S2). Zero of five transgenic lines that contained an egl-1 genomic fragment carrying either the C-to-T mutation or both the G-to-C and C-to-T mutations were rescued for the phenotype of M4 sister survival (Fig. 2B and Fig. S2). Furthermore, we isolated a 268-bp genomic deletion, n5347Δ, lacking the region of the C-to-T mutation but not the region of the G-to-C mutation (Fig. 2A). In n5347Δ mutants, 90% of the M4 sister cells (n = 118) and 39% of the I1 sister cells (n = 240) survived; n5347Δ did not affect the deaths of the NSM sister cells (0% NSM sister survival; n = 240) or the VC homologs (0.0 extra VC homologs/animal; n = 120).
Fig. 2.
n4820 is an egl-1 noncoding allele. (A) The egl-1(n4820) isolate contained two mutations in the egl-1 5′ upstream noncoding region: C-to-T (n4820) and G-to-C (n5334). The extent of the n5347Δ deletion is shown. The exons of egl-1 are shown as black boxes. (B) The indicated egl-1 genomic fragments were injected into egl-1(n4820 n5334) animals to test for rescue of the phenotype of M4 sister-cell survival. The results of these rescue experiments are indicated. (C) DNA sequence surrounding the site altered by the n4820 mutation is shown for C. elegans and related species. Nucleotides identical to those in C. elegans are indicated by bold, and the nucleotide mutated in n4820 animals is highlighted in red.
Together, these results establish that the C-to-T mutation was responsible for the survival of the M4 sister cell and the I1 sister cells in egl-1(n4820) mutants. To distinguish the n4820 C-to-T mutation, which causes survival of the M4 sister cell and the I1 sister cells, from the G-to-C mutation, we designated the G-to-C mutation n5334 (Fig. 2A). The DNA sequence immediately surrounding the n4820 mutation is highly conserved in other nematode species (Fig. 2C). Based on these results, we suggest that the n4820 region defines a binding site for a transcriptional activator that promotes egl-1 expression in the M4 sister cell and the I1 sister cells and that this binding site is defective in n4820 and n5347Δ mutants.
n4796 Is an Allele of ceh-34, a Six Family Homeodomain Gene.
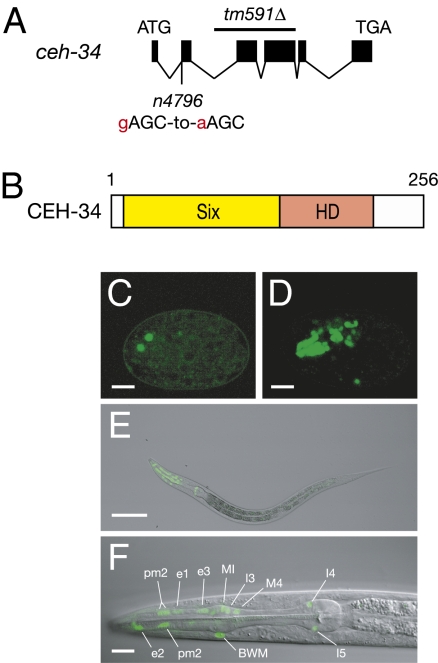
We mapped n4796 to a 150-kb interval of chromosome V almost completely covered by six cosmids (Fig. S3). We performed rescue experiments for the phenotype of M4 sister survival of n4796 animals by injecting cosmids in this interval. The cosmid C10G8 rescued the phenotype of M4 sister survival of n4796 animals (Fig. S3). C10G8 encodes eight predicted genes, including ceh-34. A genomic fragment that contains only ceh-34 rescued the phenotype of M4 sister survival of n4796 animals (Fig. S3). n4796 animals have a G-to-A mutation in the splice acceptor site of the second exon of ceh-34 (Fig. 3A). RNAi of ceh-34 caused survival of the M4 sister cell (38% of M4 sisters survived; n = 123). These results indicate that n4796 is a reduction-of-function allele of ceh-34.
Fig. 3.
n4796 is an allele of the Six family homeodomain gene ceh-34. (A) Genomic organization of the ceh-34 locus. The location and nature of the n4796 mutation are indicated. The red lowercase letter indicates the splice acceptor site of the n4796 mutation in the first intron, and the uppercase letters indicate DNA sequences in the second exon. The extent of the tm591Δ deletion is indicated. (B) Representation of the domain structure of the CEH-34 protein. The yellow box indicates the Six domain (Six), and the orange box indicates the homeodomain (HD). (C–F) Epifluorescence images showing expression of the ceh-34::gfp transgene. (C) The early embryo, (D) 1.5-fold stage embryo, (E) first-stage (L1) larva, and (F) the pharynx of an L1 larva at higher magnification. Cells expressing the ceh-34::gfp transgene are identified in F. BWM, body-wall muscle. (Scale bars: C, D, and F, 10 μm; E, 50 μm.)
ceh-34 encodes a 256 amino acid protein that is similar to Six family homeodomain proteins, which typically function as transcription factors (21). Six family homeodomain proteins contain a conserved Six domain that mediates protein–protein interactions and a homeodomain that binds DNA (21) (Fig. 3B). Both domains are evolutionarily conserved from C. elegans to humans (Fig. S4). The CEH-34 protein is 48% and 63% identical to the human protein SIX1 within the Six domain and the homeodomain, respectively.
We tested whether the functions of C. elegans CEH-34 and human SIX are evolutionarily conserved. We showed that expression of a ceh-34 cDNA under the control of the ceh-34 promoter in a ceh-34(n4796) mutant rescued the defect in M4 sister-cell death (Table S3). We then substituted a human SIX1 cDNA for the ceh-34 cDNA in this construct and found that expression of SIX1 also rescued the defect in M4 sister-cell death of ceh-34(n4796) mutants (Table S3). We conclude that the functions of C. elegans ceh-34 and human SIX are evolutionarily conserved.
CEH-34 Is Expressed Predominantly in the Nuclei of Pharyngeal Cells.
To determine the expression pattern of ceh-34, we constructed a translational ceh-34::gfp reporter transgene and showed that it can rescue the phenotype of M4 sister survival of ceh-34(n4796) mutants. Expression of this ceh-34::gfp transgene began during embryogenesis (Fig. 3 C and D). CEH-34::GFP was localized to the nuclei of expressing cells (Fig. 3 C–F), consistent with its likely function as a transcription factor. During embryonic morphogenesis and larval development and throughout adulthood, expression of the ceh-34::gfp transgene was seen predominantly in pharyngeal cells (Fig. 3 D and E). A similar expression pattern for a ceh-34::gfp transgene has been independently observed (22). We observed that our ceh-34::gfp transgene was expressed in all pharyngeal neurons (M4, I1, MI, I3, M3, NSM, MC, I2, I4, I5, I6, M1, M2, and M5), some pharyngeal muscle cells (pm1 and pm2) and pharyngeal epithelial cells (e1 and e3), and some body wall muscles around the anterior pharynx (Fig. 3F).
Eyes Absent Ortholog EYA-1 Is Required for M4 Sister- and I1 Sister-Cell Death.
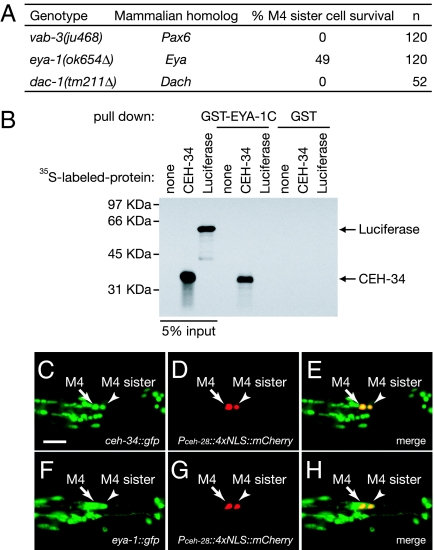
Six family homeodomain genes were originally identified in studies of Drosophila eye development (23). The Drosophila Six family homeodomain gene sine oculis is required for proper eye specification and functions in a conserved transcriptional network that includes the paired-box (Pax) homeodomain gene eyeless (ey) and the transcriptional cofactors eyes absent (eya) and dachshund (dac) (24). We tested whether the C. elegans orthologs of these three genes promote M4 sister-cell death. We found that a deletion allele of eya-1, the C. elegans eyes absent ortholog, caused survival of both the M4 sister cell (49% of M4 sisters survived; n = 120) and the I1 sister cells (19% of I1 sisters survived; n = 240) but did not affect the NSM sister cells or the VC homologs (Fig. 4A and Table S1). Null mutations in vab-3 and dac-1, the C. elegans orthologs of eyeless and dachshund, respectively, did not cause survival of the M4 sister cell (Fig. 4A). These results indicate that the C. elegans eyes absent ortholog eya-1 is required for the deaths of the M4 sister cell and the I1 sister cells and suggest that eya-1 might function with the Six family homeodomain gene ceh-34 to promote programmed cell death.
Fig. 4.
(A) The penetrances of M4 sister-cell survival in the indicated mutants are shown. Pax6, paired-like homeodomain gene; Eya, mammalian eyes absent homolog; Dach, mammalian dachshund homolog. (B) CEH-34 binds the C-terminal domain of EYA-1 (EYA-1C) in vitro; 5% of input proteins and bound proteins were subjected to electrophoresis and visualized using autoradiography. The positions of CEH-34 and luciferase are indicated by arrows. (C–H) ceh-34::gfp and eya-1::gfp transgenes are expressed in the M4 sister cells in ced-3 mutants. (C and F) Expression of the ceh-34::gfp and eya-1::gfp transgenes, respectively. (D and G) Expression of the M4 neuron and the M4 sister-cell marker Pceh-28::4xNLS::mCherry. (E and H) Merged images. Arrows indicate the M4 neurons, and arrowheads indicate the M4 sister cells. (Scale bar, 10 μm.)
EYA-1 Forms an Evolutionarily Conserved Complex with the Six Family Homeodomain Protein CEH-34.
The Six family homeodomain proteins of Drosophila and mammals physically interact with the conserved C-terminal Eya domains of Eya proteins (25, 26). Because mutations of ceh-34 and eya-1 caused the same phenotype of M4 sister-cell and I1 sister-cell survival, we examined whether CEH-34 physically interacts with EYA-1. We fused GST to the C-terminal Eya domain of EYA-1 (GST-EYA-1C) and found that this protein specifically bound CEH-34 (Fig. 4B). CEH-34 and EYA-1 also have been shown by others to both act to specify coelomocyte cell fate and interact physically (22).
If CEH-34 physically interacts and functions with EYA-1 in vivo, the two proteins must be expressed in at least some of the same cells. Indeed, the expression pattern of an eya-1::gfp transgene capable of rescuing the phenotype of eya-1 mutants is similar to that of our ceh-34::gfp transgene; eya-1 is predominantly expressed in pharyngeal cells (27). We observed expression of both the ceh-34::gfp transgene and an eya-1::gfp transgene in the M4 sister cell in ced-3(n717) mutants (Fig. 4 C–H). These results suggest that CEH-34 and EYA-1 form an evolutionarily conserved complex in the M4 sister cell to promote programmed cell death.
CEH-34 Binds to the egl-1 5′ cis-Regulatory Element Defined by egl-1(n4820).
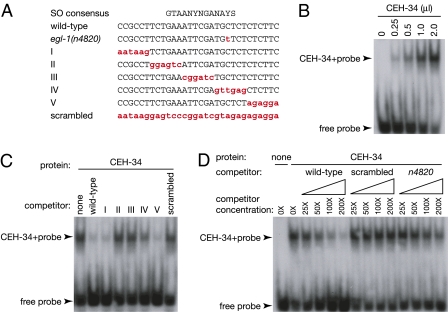
Because egl-1(n4820) and ceh-34(n4796) both specifically affected the programmed cell deaths of the M4 sister cell and the I1 sister cells, we examined whether egl-1 and ceh-34 might functionally interact to regulate these programmed cell deaths. Interestingly, the DNA sequence surrounding the egl-1(n4820) mutation is similar to the consensus-binding sequence of Sine Oculis (SO), the Drosophila homolog of CEH-34 (Fig. 5A) (28). To test if CEH-34 can bind directly to this presumptive egl-1 cis-regulatory element, we performed gel-shift assays. We observed that CEH-34 indeed binds to this sequence (Fig. 5B).
Fig. 5.
CEH-34 binds to the egl-1 5′ cis-regulatory element defined by egl-1(n4820). (A) Sine Oculis (SO) consensus-binding sequence and the DNA sequences of oligonucleotide probes used in the gel-shift assays are shown. Mutated bases are shown in red lowercase letters. Y indicates C or T, S indicates G or C, and N indicates A, C, G, or T. (B) The indicated amount of reticulocyte lysate expressing CEH-34 was mixed with the wild-type radioactive probe. The shifted band and free probe are indicated by arrowheads. (C) The indicated unlabeled competitors were preincubated in 200-fold molar excess to the radiolabeled probe. The shifted band and free probe are indicated by arrowheads. (D) The indicated unlabeled competitors were preincubated in 0- to 200-fold molar excess to the radiolabeled probe. The shifted band and free probe are indicated by arrowheads.
To test the specificity of this interaction, we performed competition experiments with unlabeled competitors. Both the wild-type competitor and competitors with mutations outside the SO consensus-binding sequence (wild type, I, and V in Fig. 5A) competed with the labeled wild-type probe for CEH-34 binding (Fig. 5C). By contrast, competitors with mutations in the SO consensus-binding sequence (II, III, IV, and scrambled in Fig. 5A) did not efficiently compete with the labeled wild-type probe for CEH-34 binding (Fig. 5C). These results indicate that CEH-34 binds to the SO consensus-binding sequence of an egl-1 cis-regulatory element surrounding the egl-1(n4820) mutation.
Because the egl-1(n4820) mutation is located within the SO consensus-binding sequence, we examined whether the egl-1(n4820) mutation affects CEH-34 binding. Whereas wild-type competitor efficiently competed for CEH-34 binding, a competitor containing the egl-1(n4820) mutation did not compete for CEH-34 binding as efficiently as the wild-type competitor (Fig. 5D). These results indicate that the egl-1(n4820) mutation reduces CEH-34 binding to this egl-1 cis-regulatory element.
egl-1 Expression in the M4 Sister Cell Requires ceh-34 and eya-1 Function.
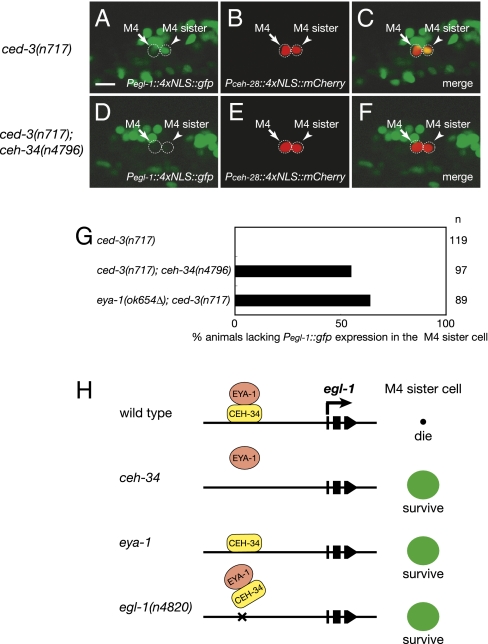
Given that CEH-34 directly binds to the egl-1 cis-regulatory element in vitro and that CEH-34 forms a complex with EYA-1, we examined if ceh-34 or eya-1 mutations alter egl-1 expression in the M4 sister cell. In ced-3 mutants, a Pegl-1::4xNLS::gfp reporter transgene that contains the egl-1 cis-regulatory element required for CEH-34 binding was expressed in the M4 sister cell (100% of animals; n = 119) but not in the M4 neuron (0% of animals; n = 119), strongly suggesting that egl-1 expression regulates the death of the M4 sister cell (Fig. 6 A, B, C, and G). We observed that in 55% of ced-3; ceh-34(n4796) animals (n = 97) and 64% of eya-1(ok654Δ); ced-3 animals (n = 89), the M4 sister cell did not express the Pegl-1::4xNLS::gfp transgene (Fig. 6 D–G). These results establish that ceh-34 and eya-1 are necessary for normal egl-1 expression in the M4 sister cell and suggest that ceh-34 and eya-1 activate egl-1 expression to promote M4 sister-cell death.
Fig. 6.
egl-1 expression in the M4 sister cell requires ceh-34 and eya-1. (A–F) The Pegl-1::4xNLS::gfp and Pceh-28::4xNLS::mCherry transgenes were coexpressed in ced-3(n717) animals (A–C) and ced-3(n717); ceh-34(n4796) animals (D–F). (A and D) Expression of the Pegl-1::4xNLS::gfp transgene. (B and E) Expression of the Pceh-28::4xNLS::mCherry transgene in the M4 neuron and M4 sister cell. (C and F) Merged images. Arrows indicate the M4 neuron; arrowheads indicate the M4 sister cell. Both cells are circled. (Scale bar, 5 μm.) (G) Percent of animals lacking expression of the Pegl-1::4xNLS::gfp transgene in the M4 sister cell is indicated for ced-3(n717), ced-3(n717); ceh-34(n4796), and eya-1(ok654Δ); ced-3(n717) animals. (H) A model for the control of M4 sister-cell death by CEH-34 and EYA-1 is shown for the wild-type, ceh-34, eya-1, or egl-1(n4820) mutants.
Discussion
ceh-34 and eya-1 Promote M4 Sister-Cell Death by Directly Activating Transcription of the Proapoptotic Gene egl-1.
From a genetic screen for mutants defective in the death of the M4 sister cell and a subsequent candidate-gene approach, we identified two genes that specify the cell-death fate of the M4 sister cell: the Six family homeodomain gene ceh-34 and the eyes absent ortholog eya-1. Based on our findings, we propose a model for the regulation of M4 sister-cell death (Fig. 6H). In wild-type animals, a complex consisting of the CEH-34 and EYA-1 proteins binds to the egl-1 5′ cis-regulatory element and activates egl-1 expression in the M4 sister cell, causing the M4 sister cell to die. In ceh-34 or eya-1 mutants, a functional CEH-34-EYA-1 complex is not formed, and egl-1 is not expressed, causing the M4 sister cell to survive. In egl-1(n4820) mutants, the CEH-34-EYA-1 complex cannot bind to the egl-1 5′ cis-regulatory element, and egl-1 is not expressed, causing the M4 sister cell to survive. The same transcriptional regulatory mechanism likely, at least in part, promotes I1 sister-cell death, because ceh-34(n4796), eya-1(ok654Δ), and egl-1(n4820) also caused survival of the I1 sister cells.
We propose that CEH-34 and EYA-1 function cell autonomously in the M4 sister cell to activate egl-1 expression and promote programmed cell death. Consistent with this hypothesis, we observed that ceh-34 and eya-1 were expressed in the M4 sister cell. Whereas ceh-34 and eya-1 were expressed in both the M4 neuron and M4 sister cell, egl-1 was expressed only in the M4 sister cell destined to die but not in the M4 neuron destined to survive. This observation suggests that ceh-34 and eya-1 are necessary but not sufficient to activate egl-1 expression in the M4 sister cell. Other factors that activate egl-1 expression in the M4 sister cell or that repress egl-1 expression in the M4 neuron likely also interact with CEH-34 and EYA-1 in causing the M4 sister cell to die or the M4 neuron to survive (Fig. 6H).
ceh-34 and eya-1 Function More Broadly in Pharyngeal Development.
Although ceh-34 and eya-1 regulate the programmed cell deaths of the M4 sister cell and the I1 sister cells, these genes also function in other aspects of development. ceh-34(tm591Δ) deletion mutants arrest as early larvae and exhibit abnormal pharyngeal morphology with decreased expression of the ceh-28 reporter transgene that we used as a reporter for the M4 neuron and M4 sister cell. eya-1 deletion mutants exhibit various defects, including abnormal pharyngeal morphology (27). Both eya-1 (27) and ceh-34 (our results) are predominantly expressed in pharyngeal cells. These observations suggest that ceh-34 and eya-1 are involved in aspects of pharyngeal development besides the regulation of the programmed cell deaths of the M4 sister cell and I1 sister cells.
Functions of ceh-34 and eya-1 in Regulating Programmed Cell Death Are Evolutionarily Conserved.
CEH-34 family Six proteins and EYA-1 family Eya proteins function in a diversity of organisms in a variety of developmental processes, including apoptosis, cell survival, and cell differentiation (24). We observed that human SIX1 can rescue the defect in M4 sister-cell death of ceh-34(n4796) mutants. This result suggests that the mechanisms of Six family protein function in regulation of apoptosis are evolutionarily conserved between C. elegans and humans.
Eya function in apoptosis is also likely conserved from C. elegans to mammals. Just as eya-1 promotes M4 sister-cell death in C. elegans, the overexpression of mammalian Eya proteins promotes apoptosis of murine myeloid cells (29). By contrast, during embryonic development, C. elegans eya-1 mutants have excessive programmed cell deaths (27), suggesting a protective function of eya-1. Such a protective function of eya-1 might also be evolutionarily conserved, because loss of Eya function causes increased apoptosis in Drosophila (30), zebrafish (31), and mice (32). Eya proteins act as protein phosphatases (33) as well as transcriptional cofactors, and the phosphatase activity of Eya proteins on histone variant H2AX is required to protect mammalian kidney embryonic cells from apoptosis after DNA damage (34). These observations indicate that Eya proteins can function in either promoting or inhibiting apoptosis, depending on cellular context, and more generally, that the function of Eya in regulating apoptosis is evolutionarily conserved from C. elegans to mammals.
Human Diseases Caused by Mutations in Six Family Homeodomain Genes or eyes absent Homologs Likely Result in Part from Misregulation of Transcription of BH3-Only Genes and Apoptosis.
Six family homeodomain genes and eyes absent genes have been implicated in mammalian apoptosis and various human diseases, including cancer (35, 36), cardiomyopathy (37), and developmental disorders (38–40). Mutations in SIX3 were identified in human patients with holoprosencephaly (HPE), a severe malformation of the forebrain characterized by an incomplete segregation of the cerebral hemispheres (38). In mice, a reduction of Six3 function can cause an HPE-like phenotype as well as decreased apoptosis along the ventral midline of the telencephalon and in optic stalks and increased apoptosis in the lateral dorsal telencephalon of embryos of a phenotypically normal heterozygous Sonic hedgehog mutant (41). Mutations in SIX1 and EYA1 were identified in human patients with branchio-oto-renal (BOR) syndrome, which is characterized by craniofacial abnormalities, hearing loss, and kidney defects (39, 40). Six1- or Eya1-deficient mice showed increased apoptosis in the inner ear and kidney, likely causing a lack of inner ear structure and failure of kidney development (32, 42, 43). In these mice, increased or decreased apoptosis is seen in tissues in which abnormalities also are observed in human HPE or BOR patients. These observations suggest that the misregulation of apoptosis caused by a mutation in Six or Eya is likely involved in these diseases. Because Six and Eya likely function in transcriptional regulation, transcriptional misregulation of target genes likely causes altered apoptosis in mice with a mutation in Six or Eya. Based on our findings, we suggest that mammalian Six and Eya proteins directly regulate transcription of BH3-only genes, that altered apoptosis in mammals carrying Six or Eya mutations is caused by misregulation of transcription of BH3-only genes, and that, in humans, such altered apoptosis is at least in part responsible for the HPE and BOR syndromes.
Materials and Methods
See SI Materials and Methods for a full description.
Strains and Genetics.
C. elegans strains were cultured and maintained on NGM plates at 20 °C as described (44). The Bristol strain N2 was the standard wild-type strain, and the Hawaiian strain CB4856 was used for SNP mapping.
In Vitro GST Fusion Protein-Binding Experiments.
In vitro GST fusion protein-binding experiments were performed essentially as described (25).
Gel Mobility Shift Assays.
Gel mobility shift experiments were performed essentially as described (28).
Supplementary Material
Acknowledgments
We thank Brian Harfe (University of Florida College of Medicine, Gainesville, FL) and Andrew Fire (Stanford University School of Medicine, Stanford, CA) for providing pBH46.53, Asako Sugimoto (RIKEN Center for Developmental Biology, Kobe, Japan) for eya-1 strains and the eya-1::gfp construct, Yuichi Iino (University of Tokyo, Tokyo) for pENTR-gcy-10, Barbara Conradt (Dartmouth Medical School, Hanover, NH) for bcIs24, Karen Oegema (University of California at San Diego, La Jolla, CA) for mCherry, Yuji Kohara (National Institute of Genetics, Mishima, Japan) for ceh-34 and eya-1 cDNAs, Andrew Fire for gfp vectors, and members of the H.R.H. laboratory for technical support and discussions. We also thank Nirav Amin (Cornell University) and Jun Liu (Cornell University, Ithaca, NY) for sharing information about ceh-34 and eya-1, the Caenorhabditis Genetic Center, which is funded by the National Institutes of Health, National Center for Research Resources, for strains, the Sanger Institute (Hinxton, UK) for cosmids, and Shohei Mitani at the National BioResource project (Tokyo Women's Medical University, Tokyo) for the ceh-34(tm591) strains. This work was supported by the Howard Hughes Medical Institute. T.H. was supported in part by the Ministry of Education, Science, Technology, Sports, and Culture of Japan. H.R.H is an Investigator of the Howard Hughes Medical Institute and the David H. Koch Professor of Biology at Massachusetts Institute of Technology.
Footnotes
The authors declare no conflict of interest.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1010023107/-/DCSupplemental.
References
- 1.Baehrecke EH. How death shapes life during development. Nat Rev Mol Cell Biol. 2002;3:779–787. doi: 10.1038/nrm931. [DOI] [PubMed] [Google Scholar]
- 2.Hipfner DR, Cohen SM. Connecting proliferation and apoptosis in development and disease. Nat Rev Mol Cell Biol. 2004;5:805–815. doi: 10.1038/nrm1491. [DOI] [PubMed] [Google Scholar]
- 3.Sulston JE, Schierenberg E, White JG, Thomson JN. The embryonic cell lineage of the nematode Caenorhabditis elegans. Dev Biol. 1983;100:64–119. doi: 10.1016/0012-1606(83)90201-4. [DOI] [PubMed] [Google Scholar]
- 4.Sulston JE, Horvitz HR. Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Dev Biol. 1977;56:110–156. doi: 10.1016/0012-1606(77)90158-0. [DOI] [PubMed] [Google Scholar]
- 5.Horvitz HR. Worms, life, and death (Nobel lecture) ChemBioChem. 2003;4:697–711. doi: 10.1002/cbic.200300614. [DOI] [PubMed] [Google Scholar]
- 6.Metzstein MM, Horvitz HR. The C. elegans cell death specification gene ces-1 encodes a snail family zinc finger protein. Mol Cell. 1999;4:309–319. doi: 10.1016/s1097-2765(00)80333-0. [DOI] [PubMed] [Google Scholar]
- 7.Thellmann M, Hatzold J, Conradt B. The Snail-like CES-1 protein of C. elegans can block the expression of the BH3-only cell-death activator gene egl-1 by antagonizing the function of bHLH proteins. Development. 2003;130:4057–4071. doi: 10.1242/dev.00597. [DOI] [PubMed] [Google Scholar]
- 8.Conradt B, Horvitz HR. The TRA-1A sex determination protein of C. elegans regulates sexually dimorphic cell deaths by repressing the egl-1 cell death activator gene. Cell. 1999;98:317–327. doi: 10.1016/s0092-8674(00)81961-3. [DOI] [PubMed] [Google Scholar]
- 9.Liu H, Strauss TJ, Potts MB, Cameron S. Direct regulation of egl-1 and of programmed cell death by the Hox protein MAB-5 and by CEH-20, a C. elegans homolog of Pbx1. Development. 2006;133:641–650. doi: 10.1242/dev.02234. [DOI] [PubMed] [Google Scholar]
- 10.Potts MB, Wang DP, Cameron S. Trithorax, Hox, and TALE-class homeodomain proteins ensure cell survival through repression of the BH3-only gene egl-1. Dev Biol. 2009;329:374–385. doi: 10.1016/j.ydbio.2009.02.022. [DOI] [PubMed] [Google Scholar]
- 11.Maurer CW, Chiorazzi M, Shaham S. Timing of the onset of a developmental cell death is controlled by transcriptional induction of the C. elegans ced-3 caspase-encoding gene. Development. 2007;134:1357–1368. doi: 10.1242/dev.02818. [DOI] [PubMed] [Google Scholar]
- 12.Wu WS, et al. Slug antagonizes p53-mediated apoptosis of hematopoietic progenitors by repressing puma. Cell. 2005;123:641–653. doi: 10.1016/j.cell.2005.09.029. [DOI] [PubMed] [Google Scholar]
- 13.Inukai T, et al. SLUG, a ces-1-related zinc finger transcription factor gene with antiapoptotic activity, is a downstream target of the E2A-HLF oncoprotein. Mol Cell. 1999;4:343–352. doi: 10.1016/s1097-2765(00)80336-6. [DOI] [PubMed] [Google Scholar]
- 14.Schwartz HT, Horvitz HR. The C. elegans protein CEH-30 protects male-specific neurons from apoptosis independently of the Bcl-2 homolog CED-9. Genes Dev. 2007;21:3181–3194. doi: 10.1101/gad.1607007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nehme R, et al. Transcriptional upregulation of both egl-1 BH3-only and ced-3 caspase is required for the death of the male-specific CEM neurons. Cell Death Differ. 2010;17:1266–1276. doi: 10.1038/cdd.2010.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li S, et al. Hearing loss caused by progressive degeneration of cochlear hair cells in mice deficient for the Barhl1 homeobox gene. Development. 2002;129:3523–3532. doi: 10.1242/dev.129.14.3523. [DOI] [PubMed] [Google Scholar]
- 17.Avery L, Horvitz HR. A cell that dies during wild-type C. elegans development can function as a neuron in a ced-3 mutant. Cell. 1987;51:1071–1078. doi: 10.1016/0092-8674(87)90593-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.White JG, Southgate E, Thomson JN, Brenner S. The structure of the nervous system of the nematode Caenorabditis elegans. Philos Trans R Soc Lond B Biol Sci. 1986;314:1–340. doi: 10.1098/rstb.1986.0056. [DOI] [PubMed] [Google Scholar]
- 19.Schwartz HT. A protocol describing pharynx counts and a review of other assays of apoptotic cell death in the nematode worm Caenorhabditis elegans. Nat Protoc. 2007;2:705–714. doi: 10.1038/nprot.2007.93. [DOI] [PubMed] [Google Scholar]
- 20.Conradt B, Horvitz HR. The C. elegans protein EGL-1 is required for programmed cell death and interacts with the Bcl-2-like protein CED-9. Cell. 1998;93:519–529. doi: 10.1016/s0092-8674(00)81182-4. [DOI] [PubMed] [Google Scholar]
- 21.Kawakami K, Sato S, Ozaki H, Ikeda K. Six family genes—structure and function as transcription factors and their roles in development. Bioessays. 2000;22:616–626. doi: 10.1002/1521-1878(200007)22:7<616::AID-BIES4>3.0.CO;2-R. [DOI] [PubMed] [Google Scholar]
- 22.Amin NM, Lim SE, Shi H, Chan TL, Liu J. A conserved Six-Eya cassette acts downstream of Wnt signaling to direct non-myogenic versus myogenic fates in the C. elegans postembryonic mesoderm. Dev Biol. 2009;331:350–360. doi: 10.1016/j.ydbio.2009.05.538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cheyette BN, et al. The Drosophila sine oculis locus encodes a homeodomain-containing protein required for the development of the entire visual system. Neuron. 1994;12:977–996. doi: 10.1016/0896-6273(94)90308-5. [DOI] [PubMed] [Google Scholar]
- 24.Jemc J, Rebay I. The eyes absent family of phosphotyrosine phosphatases: Properties and roles in developmental regulation of transcription. Annu Rev Biochem. 2007;76:513–538. doi: 10.1146/annurev.biochem.76.052705.164916. [DOI] [PubMed] [Google Scholar]
- 25.Pignoni F, et al. The eye-specification proteins So and Eya form a complex and regulate multiple steps in Drosophila eye development. Cell. 1997;91:881–891. doi: 10.1016/s0092-8674(00)80480-8. [DOI] [PubMed] [Google Scholar]
- 26.Heanue TA, et al. Synergistic regulation of vertebrate muscle development by Dach2, Eya2, and Six1, homologs of genes required for Drosophila eye formation. Genes Dev. 1999;13:3231–3243. doi: 10.1101/gad.13.24.3231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Furuya M, Qadota H, Chisholm AD, Sugimoto A. The C. elegans eyes absent ortholog EYA-1 is required for tissue differentiation and plays partially redundant roles with PAX-6. Dev Biol. 2005;286:452–463. doi: 10.1016/j.ydbio.2005.08.011. [DOI] [PubMed] [Google Scholar]
- 28.Pauli T, Seimiya M, Blanco J, Gehring WJ. Identification of functional sine oculis motifs in the autoregulatory element of its own gene, in the eyeless enhancer and in the signalling gene hedgehog. Development. 2005;132:2771–2782. doi: 10.1242/dev.01841. [DOI] [PubMed] [Google Scholar]
- 29.Clark SW, Fee BE, Cleveland JL. Misexpression of the eyes absent family triggers the apoptotic program. J Biol Chem. 2002;277:3560–3567. doi: 10.1074/jbc.M108410200. [DOI] [PubMed] [Google Scholar]
- 30.Bonini NM, Leiserson WM, Benzer S. The eyes absent gene: Genetic control of cell survival and differentiation in the developing Drosophila eye. Cell. 1993;72:379–395. doi: 10.1016/0092-8674(93)90115-7. [DOI] [PubMed] [Google Scholar]
- 31.Kozlowski DJ, Whitfield TT, Hukriede NA, Lam WK, Weinberg ES. The zebrafish dog-eared mutation disrupts eya1, a gene required for cell survival and differentiation in the inner ear and lateral line. Dev Biol. 2005;277:27–41. doi: 10.1016/j.ydbio.2004.08.033. [DOI] [PubMed] [Google Scholar]
- 32.Xu PX, et al. Eya1-deficient mice lack ears and kidneys and show abnormal apoptosis of organ primordia. Nat Genet. 1999;23:113–117. doi: 10.1038/12722. [DOI] [PubMed] [Google Scholar]
- 33.Li X, et al. Eya protein phosphatase activity regulates Six1-Dach-Eya transcriptional effects in mammalian organogenesis. Nature. 2003;426:247–254. doi: 10.1038/nature02083. [DOI] [PubMed] [Google Scholar]
- 34.Cook PJ, et al. Tyrosine dephosphorylation of H2AX modulates apoptosis and survival decisions. Nature. 2009;458:591–596. doi: 10.1038/nature07849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang L, et al. Transcriptional coactivator Drosophila eyes absent homologue 2 is up-regulated in epithelial ovarian cancer and promotes tumor growth. Cancer Res. 2005;65:925–932. [PubMed] [Google Scholar]
- 36.Ford HL, Kabingu EN, Bump EA, Mutter GL, Pardee AB. Abrogation of the G2 cell cycle checkpoint associated with overexpression of HSIX1: A possible mechanism of breast carcinogenesis. Proc Natl Acad Sci USA. 1998;95:12608–12613. doi: 10.1073/pnas.95.21.12608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schönberger J, et al. Mutation in the transcriptional coactivator EYA4 causes dilated cardiomyopathy and sensorineural hearing loss. Nat Genet. 2005;37:418–422. doi: 10.1038/ng1527. [DOI] [PubMed] [Google Scholar]
- 38.Wallis DE, et al. Mutations in the homeodomain of the human SIX3 gene cause holoprosencephaly. Nat Genet. 1999;22:196–198. doi: 10.1038/9718. [DOI] [PubMed] [Google Scholar]
- 39.Abdelhak S, et al. A human homologue of the Drosophila eyes absent gene underlies branchio-oto-renal (BOR) syndrome and identifies a novel gene family. Nat Genet. 1997;15:157–164. doi: 10.1038/ng0297-157. [DOI] [PubMed] [Google Scholar]
- 40.Ruf RG, et al. SIX1 mutations cause branchio-oto-renal syndrome by disruption of EYA1-SIX1-DNA complexes. Proc Natl Acad Sci USA. 2004;101:8090–8095. doi: 10.1073/pnas.0308475101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Geng X, et al. Haploinsufficiency of Six3 fails to activate Sonic hedgehog expression in the ventral forebrain and causes holoprosencephaly. Dev Cell. 2008;15:236–247. doi: 10.1016/j.devcel.2008.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zheng W, et al. The role of Six1 in mammalian auditory system development. Development. 2003;130:3989–4000. doi: 10.1242/dev.00628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xu PX, et al. Six1 is required for the early organogenesis of mammalian kidney. Development. 2003;130:3085–3094. doi: 10.1242/dev.00536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Brenner S. The genetics of Caenorhabditis elegans. Genetics. 1974;77:71–94. doi: 10.1093/genetics/77.1.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.