Abstract
Rationale
Increased aortic stiffness, an important feature of many vascular diseases, e.g., aging, hypertension, atherosclerosis and aortic aneurysms, is assumed due to changes in extracellular matrix (ECM).
Objective
We tested the hypothesis that the mechanisms also involve intrinsic stiffening of vascular smooth muscle cells (VSMCs).
Methods and Results
Stiffness was measured in vitro both by atomic force microscopy (AFM) and in a reconstituted tissue model, using VSMCs from aorta of young versus old male monkeys (Macaca fascicularis, n=7/group), where aortic stiffness increases by 200 % in vivo. The apparent elastic modulus was increased (P<0.05) in old VSMCs (41.7±0.5 kPa) versus young (12.8±0.3 kPa), but not after disassembly of the actin cytoskeleton with cytochalasin D. Stiffness of the VSMCs in the reconstituted tissue model was also higher (P<0.05) in old (23.3±3.0 kPa) than in young (13.7±2.4 kPa).
Conclusions
These data support the novel concept, not appreciated previously, that increased vascular stiffness with aging is due not only to changes in ECM, but also to intrinsic changes in VSMCs.
Keywords: vascular smooth muscle cell, stiffness, AFM, aging
INTRODUCTION
Increased aortic stiffness is an important feature of aging and of cardiovascular disease, and is assumed to be due to changes in the extracellular matrix (ECM)1, 2. However, the treatment of large artery stiffening in patients involves the utilization of calcium channel blockers and of angiotensin II receptor antagonists, which seems internally inconsistent, since these drugs primarily affect the tone of vascular smooth muscle cells (VSMCs) but not the ECM. Accordingly, our hypothesis, not examined previously, is that the intrinsic mechanical properties of VSMCs contribute to increased vascular stiffness in aging. An important feature of this investigation was the use of a non-human primate model (M. fascicularis) because of its phylogenetic proximity to humans and because the aging process occurs gradually over 20-30 years. In a prior study, we demonstrated that aortic stiffness, assessed in vivo by measurements of aortic pressure and diameter, increased in old compared to young monkeys2. In that study, as in another3, it was assumed that increased stiffness was due to an increase in collagen and a decrease in elastin in the vascular media. In this investigation, we isolated VSMCs from thoracic aorta from old and young male monkeys and assessed their mechanical properties in vitro using atomic force microscopy (AFM) and a reconstituted tissue model.
METHODS
Further details of the Methods used are provided in a supplementary file.
Animal model
Young (6.4±0.1 yr) and old (25±0.4 yr) male monkeys (Macaca fascicularis, n=7/group) were used and maintained in accordance with the Guide for the Care and Use of Laboratory Animals (NIH 83-23, revised 1996).
VSMC isolation and culture
VSMCs from thoracic aorta of monkeys were used at passages 2-4. The phenotype was confirmed by immunofluorescence for α-smooth muscle actin.
Reconstituted tissue model measurements
Cultured VSMCs were mixed with collagen type I and incubated for 2 days in a cylindrical well with a central mandrel to create a ring mold that was connected to an isometric force transducer4. The elasticity modulus (E) was calculated as: E= F*L/(A*ΔL), where F is force generated by the construct in response to stretch, L is original length of tissue, A is cross-sectional area, and ΔL the variation of length after stretch.
AFM imaging and force measurement
To obtain topographical images of VSMC, the AFM was operated in contact mode. Transverse VSMC stiffness was measured in force mode using silicon nitride cantilevers. For combined AFM and fluorescent imaging, VSMCs were fluorescently labeled with Alexa 568-phalloidin and recorded with an integrated AFM-confocal microscope system (Veeco Bioscope 2-Olympus FV1000). VSMCs were treated with cytochalasin D (CD, 10 μmol/L) to disrupt the actin cytoskeleton, with the myosin light chain kinase inhibitor ML7 (10 μmol/L) to inhibit myosin light phosphorylation, or with colchicine (10 μmol/L) to depolymerize microtubules. AFM probes with fibronectin (FN, 1mg/ml) operated in force mode were used to assess β1-integrin binding to the ECM.
Statistical analysis
Data reported as mean ± SE are compared using Student’s t-test. A P value<0.05 was considered significant.
RESULTS
Increased stiffness of aging VSMCs in reconstituted tissue
Typical force recordings of VSMCs are shown for the reconstituted tissues (Fig. 1A, young monkey and 1B, old monkey). The total modulus of elasticity (E) of the VSMC construct in old males was significantly higher (23.3±3.0 kPa, P<0.05) than that in young (13.7±2.4 kPa) (Fig. 1C), indicating that the VSMC tissue construct in aging monkeys was stiffer.
Figure 1. Mechanical properties of VSMC from the reconstituted tissue model.
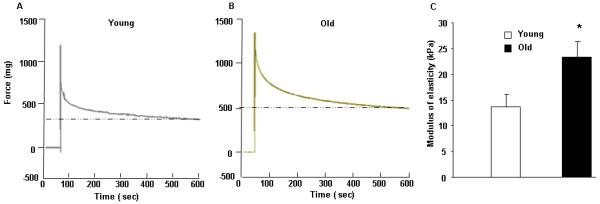
Recording of the force generated by the aortic VSMCs from young (A) and old (B) monkeys in the reconstituted tissue. (C) Modulus of elasticity in reconstituted tissue. *, P<0.05 versus young monkeys (n=7/group).
Mechanical properties of single VSMC measured by AFM
Single VSMC stiffness was measured using cell nano-indentation with AFM. As shown in Figure 2 A and D, the apparent elastic modulus was significantly higher in VSMCs from old (41.7±0.5 kPa, 76 cells from 3 animals) compared with young (12.8±0.3 kPa, 40 cells from 3 animals) male monkeys, demonstrating that VSMCs from old male monkeys are inherently stiffer than those from young males. Treatment of VSMCs with CD (Figure 2 B, C and D) or ML7 (Figure 2 E) resulted in a dramatic reduction of E in VSMCs from young and old monkeys. Treatment of VSMCs with colchine reduced cell stiffness, however to a lower extent than that observed with CD or with ML7, although old monkey VSMCs remained significantly stiffer than young monkey VSMCs(Figure 2 F). Collectively, these observations indicate that VSMC stiffness depends on the actin component of the cytoskeleton.
Figure 2. Mechanical properties of single VSMC measured by AFM.

(A) Distribution of force as a function of indentation in young (pink, n=40 cells) and old (blue, n=76 cells) monkeys. (B-C) Time course of elasticity for one single VSMC before and after CD treatment in young (B) versus old (C) monkeys. (D-E) Response of VSMC stiffness to CD (D) or ML7 (E) treatment in young and old monkeys. (F) Response of VSMC stiffness to colchicine treatment in young and old monkeys. *, P<0.05 versus young monkeys.
Expression of cytoskeletal proteins and adhesion molecules
The density of cortical cytoskeletal filaments, quantified by analyzing the AFM topography images of single VSMCs, was increased in VSMCs from old monkeys (Figure 3A-D). The expression of α-smooth muscle actin was significantly higher in old monkey VSMCs than in young monkeys. There was no difference in the expression of vimentin and tubulin between both groups, again suggesting that α-smooth muscle actin plays a predominant role in VSMC stiffening with aging (Figure 3D, 4A and B). The abundance of β1-integrin, a major adhesion molecule between VSMC and ECM, was significantly higher in old versus young males (Figure 4A and B). In addition, AFM probes coupled to an antibody against β1-integrin confirmed the increased adhesiveness of old versus young VSMCs (Figure 4C and D), as measured by increased binding probability (48±1% vs 31±2%) and by a higher peak binding force (50±1 pN vs 29±1 pN). Adhesive interactions with FN were also enhanced in old VSMCs, as evidenced by their increased adhesion to FN-coupled AFM probes and higher binding force (Figure 4 E and F).
Figure 3. Cortical cytoskeletal density of VSMC by topographical AFM images.

(A) Deflection Images of VSMC (left panel), gradient image (middle panel) and intermediate process image mask (right panel). (B) Example of deflection images in VSMCs from young versus old monkeys. (C) AFM image analysis. Old VSMC showed significantly greater surface gradients consistent with denser cortical cytoskeleton. (D) Fluorescent 3D image set (top panel) of young (left) and old (right) VSMC labeled for f-actin (red). AFM topographical image of same cell (middle panel) and overlay of AFM with fluorescent images (lower panel). *, P<0.05 versus young monkeys.
Figure 4. Alteration of cytoskeletal proteins in aging VSMCs.

(A) Immunostaining for α-smooth muscle actin, α-tubulin, vimentin and β1-integrin in cultured VSMCs. (B) Western blotting of the corresponding proteins. *, P< 0.05 versus young monkeys. Identification of β1-integrin (C for young and D for old) and active integrin adhesion to FN measured by AFM on the surface of VSMCs from young (E) and old (F) monkeys. The binding probability and peak binding force (see supplementary file of the Methods) in VSMC was higher in old vs. young monkeys.
DISCUSSION
Our data show that aortic VSMCs from old male monkeys have a higher intrinsic stiffness compared to young monkeys. In contrast to current concepts indicating that increased vascular stiffness of aging is simply due to ECM changes, primarily collagen and elastin, our study demonstrates that intrinsic mechanisms in VSMCs contribute to stiffness as well.
Few data are available about the contribution of VSMCs per se to increased stiffness with aging, in part because this is difficult to assess in intact tissue in vivo. Therefore, a model of reconstituted tissue provides an appealing alternative to quantitatively characterize the stiffness of VSMCs. We have shown in our previous study6 that the expression of multiple genes encoding proteins of the cytoskeleton and membrane receptors is regulated by aging in the vasculature. These observations led to the hypothesis, tested and confirmed in the present study, that increased stiffness of the aging aorta correlates with changes in the mechanical properties of the VSMCs. To further explore whether VSMC stiffness during the aging process was independent of the ECM, we used AFM for direct measurements of VSMC stiffness, confirming that VSMCs become stiffer with aging. To compare the increase in stiffness of VSMCs with that of the entire aorta, we found that the stiffness index β increased by 214±38% in aortas of old versus young monkeys used in the current investigation, similar to the increase observed in our prior study2. Interestingly, the stiffness of the aortic VSMCs measured by AFM in the current study increased by an almost identical amount, 223±5% (Figure 2). It would also be important to understand the quantitative contribution of the VSMC component to global stiffness, but that is not possible, since the two models, in vivo and in vitro, are so different.
We further show that expression of α-smooth muscle actin was increased in VSMCs from aging monkeys, while expression of vimentin and tubulin did not differ. Increased expression of actin was confirmed by immunolabeling and AFM topography analysis. These observations suggest that α-smooth muscle actin plays a key role in the enhancement of VSMC stiffness during aging. Changes in the regulation of actin polymerization and depolymerization could also contribute to increased actin expression and to increased VSMC stiffness. α-smooth muscle actin is a stress fiber-specific isoform for VSMCs and is the most abundant component (70% of total actin) in adult VSMCs7. It is also central to mechano-sensation, -transduction and -transmission through the integrin by transferring force to the ECM, thereby contributing to matrix remodeling8.
The integrins couple the ECM to cytoskeleton9, thereby providing a mechanical connection between VSMCs and the extracellular environment10. Integrin-ECM adhesion results in “outside-in” signaling that promotes cytoskeletal remodeling and expression of genes encoding cytoskeletal proteins that modulate the mechanical resistance of the VSMCs11. We also show that the increased expression of β1-integrin correlates with increased adhesiveness to FN, suggesting an increased “inside-out” interaction of VSMCs with the ECM. Therefore, both β1-integrin and α-smooth muscle actin are likely major players in the increased stiffness of VSMCs during aging.
In conclusion, this investigation demonstrates for the first time the relative importance of changes in intrinsic VSMC stiffness to the global increase in aortic stiffening with aging, thereby providing a mechanistic rationale for the use of pharmacological agents that affect VSMC tone in the treatment of increased vascular stiffness. It is conceivable that this novel mechanism may also mediate increased vascular stiffness in other disease states, e.g., hypertension, atherosclerosis and aortic aneurysm, thereby permitting a paradigm shift in the pharmacological targeting of vascular stiffening.
Novelty and Significance
What is known?
One of the most significant cardiovascular changes with aging is increased arterial pulse pressure, which is due to increased stiffness of large arteries, e.g., the aorta.
Considered an important risk factor, it is widely held that the increased aortic stiffness observed with aging is due to changes in the extracellular matrix (ECM), i.e., increase in collagen and decrease in elastin.
There is virtually no information on whether changes in the mechanical properties of vascular smooth muscle cells (VSMCs) can contribute to the increased stiffness of the aorta with aging.
What new information does this article contribute?
The increased aortic stiffness of aging is due not only to changes in the ECM, but also to increases in stiffness of the VSMCs.
This novel mechanism for vascular stiffness may also be involved in the increased aortic stiffness observed in hypertension, atherosclerosis, and aortic aneurysm.
Targeting the mechanisms that regulate VSMC stiffness could be a novel therapeutic approach for these diseases.
Summary
Increased aortic stiffness contributing to many vascular diseases is assumed to be due to changes in ECM. Whether its mechanism also involves VSMCs is unknown. We describe that VSMC stiffness, measured both by atomic force microscopy and in a reconstituted tissue model, significantly increases in aorta from old versus young monkeys, which is suppressed after disassembly of the actin cytoskeleton. These data demonstrate the novel concept that increased vascular stiffness with aging is at least partly due to intrinsic changes in VSMCs, which may open new therapeutic avenues for the treatment of vascular stiffening by targeting directly VSMC stiffness.
Supplementary Material
Acknowledgments
Source of funding:
This work supported in part by NIH grants HL102472; AG027211; HL033107; HL069752; HL095888; HL069020; HL-58960; HL-062863; AG023567.
Non-standard Abbreviations and Acronyms
- AFM
atomic force microscopy
- CD
Cytochalasin D
- E
elasticity modulus
- ECM
extracellular matrix
- FN
Fibronectin
- VSMCs
vascular smooth muscle cells
Footnotes
Disclosures:
None
This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Mitchell GF, Guo CY, Benjamin EJ, Larson MG, Keyes MJ, Vita JA, Vasan RS, Levy D. Cross-sectional correlates of increased aortic stiffness in the community: the Framingham Heart Study. Circulation. 2007;20:2628–2636. doi: 10.1161/CIRCULATIONAHA.106.667733. [DOI] [PubMed] [Google Scholar]
- 2.Qiu H, Depre C, Ghosh K, Resuello RG, Natividad FF, Rossi F, Peppas A, Shen YT, Vatner DE, Vatner SF. Mechanism of gender-specific differences in aortic stiffness with aging in nonhuman primates. Circulation. 2007;6:669–676. doi: 10.1161/CIRCULATIONAHA.107.689208. [DOI] [PubMed] [Google Scholar]
- 3.Gaballa MA, Jacob CT, Raya TE, Liu J, Simon B, Goldman S. Large artery remodeling during aging: biaxial passive and active stiffness. Hypertension. 1998;3:437–443. doi: 10.1161/01.hyp.32.3.437. [DOI] [PubMed] [Google Scholar]
- 4.Wakatsuki T, Kolodney MS, Zahalak GI, Elson EL. Cell mechanics studied by a reconstituted model tissue. Biophys J. 2000;5:2353–2368. doi: 10.1016/S0006-3495(00)76481-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Karlon WJ, Hsu PP, Li S, Chien S, McCulloch AD, Omens JH. Measurement of orientation and distribution of cellular alignment and cytoskeletal organization. Ann Biomed Eng. 1999;6:712–720. doi: 10.1114/1.226. [DOI] [PubMed] [Google Scholar]
- 6.Qiu H, Tian B, Resuello RG, Natividad FF, Peppas A, Shen YT, Vatner DE, Vatner SF, Depre C. Sex-specific regulation of gene expression in the aging monkey aorta. Physiol Genomics. 2007;2:169–180. doi: 10.1152/physiolgenomics.00229.2006. [DOI] [PubMed] [Google Scholar]
- 7.Gabbiani G, Schmid E, Winter S, Chaponnier C, de Ckhastonay C, Vandekerckhove J, Weber K, Franke WW. Vascular smooth muscle cells differ from other smooth muscle cells: predominance of vimentin filaments and a specific alpha-type actin. Proc Natl Acad Sci U S A. 1981;1:298–302. doi: 10.1073/pnas.78.1.298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang J, Zohar R, McCulloch CA. Multiple roles of alpha-smooth muscle actin in mechanotransduction. Exp Cell Res. 2006;3:205–214. doi: 10.1016/j.yexcr.2005.11.004. [DOI] [PubMed] [Google Scholar]
- 9.Schwartz MA, Ginsberg MH. Networks and crosstalk: integrin signalling spreads. Nat Cell Biol. 2002;4:E65–68. doi: 10.1038/ncb0402-e65. [DOI] [PubMed] [Google Scholar]
- 10.Delon I, Brown NH. Integrins and the actin cytoskeleton. Curr Opin Cell Biol. 2007;1:43–50. doi: 10.1016/j.ceb.2006.12.013. [DOI] [PubMed] [Google Scholar]
- 11.Chiquet M, Gelman L, Lutz R, Maier S. From mechanotransduction to extracellular matrix gene expression in fibroblasts. Biochim Biophys Acta. 2009;5:911–920. doi: 10.1016/j.bbamcr.2009.01.012. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


