Five years ago, a retrovirus resembling a murine leukemia virus (MLV) was found in patients with prostate cancer (1), and last year, a similar gammaretrovirus was identified in patients with chronic fatigue syndrome (CFS) (2). The agent was named xenotropic MLV-related virus (XMRV), because its env gene was nearly identical to that of xenotropic MLV, an infectious endogenous MLV that preferentially infects cells from foreign species, including humans (Fig. 1A) (3). The two reports struck a common chord, because the viral sequences found in prostate cancer and CFS were nearly identical. A second common theme emerged in reports from Europe that XMRV was rarely found, if at all, in prostate-cancer samples or patients with CFS; however, other investigators confirmed the presence of XMRV in prostate-cancer samples from North America (4). Although a recent report from the Centers for Disease Control and Prevention (CDC) did not find a link between XMRV and CFS (5), distinct MLV-related sequences are found in serial samples collected from the mid-1990s to 2010 from patients with CFS reported in the study by Lo et al. (6) in PNAS. However, the reasons for the current geographical restriction and the source of the infection are baffling.
Fig. 1.
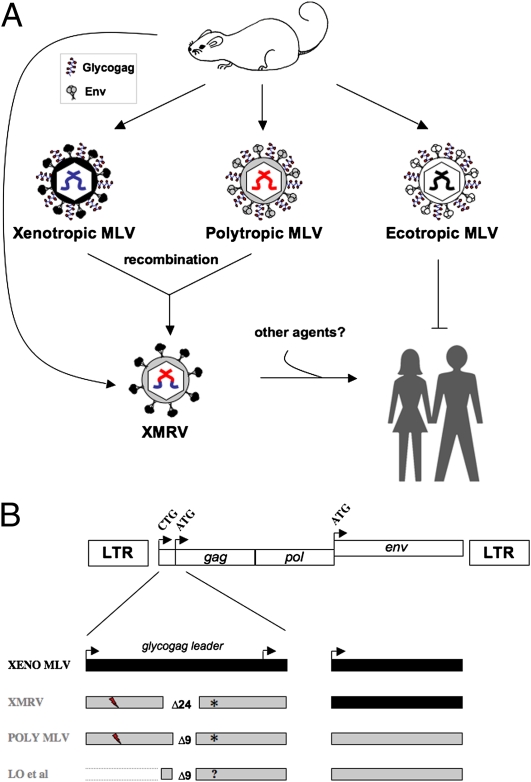
MLV-related sequences in CFS patients. (A) Virions are represented with color-coded genomic RNA caged in hexagonal gag-encoded capsids, with glycogag and the env-encoded envelope (Env) glycoproteins represented as zigzag lines, circles, and sticks, respectively. Env determines tropism for mouse and not human cells (ecotropic), nonmouse cells (xenotropic), or both cell types (polytropic). XMRV seems to be a xenotropic/polytropic recombinant that may both lack glycogag (see below) and require complementation with another infectious agent to spread in humans. (B) Schematic MLV proviral DNA genome with the glycogag, gag, and env CTG and ATG translation initiation codons. The xenotropic (black) or polytropic (gray) origin for env, when known, and the glycogag leader sequence (upstream of the gag ATG) are shown with the corresponding in-frame 24- (xenotropic) or 9-bp (polytropic) deletion and a frameshift (lightning) that leads to a stop codon (star). Sequences reported in PNAS (6) do not allow predictions on glycogag production.
Originally, a viral etiology of familial prostate cancer was pursued in patients with RNase L deficiency, an antiviral endoribonuclease that relays the antiviral IFN response (7). Because modulated RNase L activity has been linked with CFS, similar case-control studies were conducted, and XMRV was found in 67% of CFS patients (2). Since then, XMRV has been found in prostate-cancer patients with no link to RNase L deficiency (4) and in 3.7% of healthy donors in the original CFS study (2). Lo et al. detect MLV-related sequences in 88% of their CFS samples and 6.8% of control blood donors (6). If verified, this perplexing frequency of detecting MLV-like sequences in blood donors suggests a more widespread source of infection.
The gag gene sequences identified by Lo et al. (6) share 96.6% homology with XMRV, but they seem divergent in a region that has long puzzled virologists. Over 30 y ago, extracellular glycosylated forms of the MLV Gag proteins were identified (8) and found to be translated from an alternative CTG initiation codon in-frame with the conventional gag ATG to append a type II leader sequence to the Gag precursor (9) (Fig. 1B). Glycosylated forms of Gag, or glycogag, can be incorporated in virions (10) and are required for efficient viral release (11), spread, and pathogenesis (12, 13) (Fig. 1A). Intriguingly, the hepatitis B virus harbors a similar precore region that encodes the e antigen (HBeAg) in-frame with the core protein, and natural mutants with stop codons preventing expression of HBeAg have been reported to arise during the natural course of infection (14). Similarly, the 24-bp in-frame deletion common to all XMRV isolates located in the glycogag leader sequence maintains the ORF, whereas the ORF is eventually occluded by an upstream point deletion (Fig.1B). The sequences reported by Lo et al. (6) with a 9-bp deletion are distinct from that of XMRV. Although the available sequence also maintains the glycogag ORF, sequence information on the CTG region and availability of the ORF is still outstanding.
Lo et al. (6) report that the MLV-related sequences that they identify correspond to polytropic and not xenotropic MLV sequences that differ in their Env host range. Both xenotropic and polytropic MLV Env bind the polymorphic Xpr1 receptor to mediate cell entry, although with different efficiencies in laboratory and wild-mouse strains (15–18). Therefore, identifying the precise origins and relatedness of both these MLV-related agents found in humans becomes of paramount importance to clarify the source and epidemiology of infection and ultimately, the potential role in disease. Indeed, XMRV genomes (1, 2) are actually hybrids between polytropic endogenous MLV sequences for their 5′ half up to approximately the middle of pol and xenotropic MLV for their 3′ half that harbors env. The glycogag leader of XMRV matches 100% of a polytropic endogenous sequence of the 129 × 1/SvJ laboratory mouse strain (accession no. AAHY01591888.1), a commonly used strain for gene knock-outs in embryonic stem cells. However, the glycogag/gag sequences reported by Lo et al. (6) best match endogenous polytropic sequences of the C57BL/6J laboratory strain of Mus musculus, with 99% nucleotide homology, and the env sequences share 97% homology with similar endogenous polytropic MLV and mink cell focus-forming viruses.
Env cross-dressing between different MLV, known as pseudotyping, is a common feature in dually infected hosts (19), allowing MLV to extend their original tropism. Indeed, the multiple origins of these xenotropic sequences, the hybrid nature of the XMRV genome, and the occlusion of the otherwise necessary glycogag ORF underscore the potential complementation and recombinational events that may lead to their transmission into humans. Interestingly, MLV glycogag can both increase the production of HIV-1 (11) and efficiently substitute for Nef to reestablish HIV-1 spread (20). These observations suggest a scenario in which retroviruses, MLV-related agents, and potentially, other viral agents may cross-complement to promote coinfection and enable pathogenicity.
The current data suggest that a variety of xenotropic and polytropic MLV can be found in North Americans with and without disease. To add to this bewilderment, it is likely that more than one environmental agent impacts on the development of both CFS and prostate cancer. At this juncture, it would seem reasonable to conduct extensive case-control studies in North America, as suggested by Lo et al. (6), using coded control samples from subjects with inflammatory disease to determine the frequency of MLV infection in patients with CFS. The potential transmission of MLV-related sequences from human to human should also be epidemiologically evaluated.
As we currently lack postulates to prove a causal association with a prevalent agent and a chronic disease with genetic predisposition, it would also be appropriate to conduct interventional studies. Indeed, the Helicobacter pylori hypothesis of peptic ulcer disease was only accepted after Barry Marshall showed that bacterial eradication with antibiotics cured peptic ulcer disease (21). Studies to gain proof of principle have been performed with antivirals in other chronic, idiopathic diseases linked to retroviral infection, such as primary biliary cirrhosis associated with mouse mammary tumor virus, another possible murine zoonosis (22). Trials using a combination of reverse transcriptase inhibitors led to significant improvements in clinical, histological, and biochemical outcomes in these patients, albeit with some evidence of viral resistance to therapy (23). Such studies are now feasible for CFS, because reverse-transcriptase inhibitors, such as tenofovir and emtracitabine, and the integrase inhibitor raltegravir can inhibit XMRV (24).
The caveats for conducting clinical trials in patients with CFS and MLV infection are that the potential benefits of treatment should outweigh the risks; also, studies should be conducted as randomized controlled trials with meaningful and feasible endpoints using robust therapies. At this juncture, studies to establish proof of principle are justified to determine whether safe antiviral regimens can impact on CFS and to determine whether xenotropic or polytropic MLV is causally associated with this debilitating disease.
Acknowledgments
We thank Gina Mason (Sunrise Productions, Edmonton, AB, Canada) for artwork. J.-L.B. and M.S. are supported by the Institut National de la Santé et de la Recherche Médicale, and work on retrovirus transmission in M.S.’s laboratory is supported in part by grants from the Fondation de la Recherche Medicale, Fondation de France, and Sidaction. A.L.M. is supported by the Alberta Heritage Foundation for Medical Research, Broad Foundation, Canadian Liver Foundation, and Canadian Institute of Health Research.
Footnotes
The authors declare no conflict of interest.
See companion article on page 15874.
References
- 1.Urisman A, et al. Identification of a novel Gammaretrovirus in prostate tumors of patients homozygous for R462Q RNASEL variant. PLoS Pathog. 2006;2:e25. doi: 10.1371/journal.ppat.0020025. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 2.Lombardi VC, et al. Detection of an infectious retrovirus, XMRV, in blood cells of patients with chronic fatigue syndrome. Science. 2009;326:585–589. doi: 10.1126/science.1179052. [DOI] [PubMed] [Google Scholar]
- 3.Levy JA. Xenotropic viruses: Murine leukemia viruses associated with NIH Swiss, NZB, and other mouse strains. Science. 1973;182:1151–1153. doi: 10.1126/science.182.4117.1151. [DOI] [PubMed] [Google Scholar]
- 4.Schlaberg R, Choe DJ, Brown KR, Thaker HM, Singh IR. XMRV is present in malignant prostatic epithelium and is associated with prostate cancer, especially high-grade tumors. Proc Natl Acad Sci USA. 2009;106:16351–16356. doi: 10.1073/pnas.0906922106. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 5.Switzer WM, et al. Absence of evidence of xenotropic murine leukemia virus-related virus infection in persons with chronic fatigue syndrome and healthy controls in the United States. Retrovirology. 2010;7:57. doi: 10.1186/1742-4690-7-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lo S-C, et al. Detection of MLV-related virus gene sequences in blood of patients with chronic fatigue syndrome and healthy blood donors. Proc Natl Acad Sci USA. 2010;107:15874–15879. doi: 10.1073/pnas.1006901107. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 7.Silverman RH. Viral encounters with 2′,5′-oligoadenylate synthetase and RNase L during the interferon antiviral response. J Virol. 2007;81:12720–12729. doi: 10.1128/JVI.01471-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Evans LH, Dresler S, Kabat D. Synthesis and glycosylation of polyprotein precursors to the internal core proteins of Friend murine leukemia virus. J Virol. 1977;24:865–874. doi: 10.1128/jvi.24.3.865-874.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Prats AC, De Billy G, Wang P, Darlix JL. CUG initiation codon used for the synthesis of a cell surface antigen coded by the murine leukemia virus. J Mol Biol. 1989;205:363–372. doi: 10.1016/0022-2836(89)90347-1. [DOI] [PubMed] [Google Scholar]
- 10.Fujisawa R, McAtee FJ, Favara C, Hayes SF, Portis JL. N-terminal cleavage fragment of glycosylated Gag is incorporated into murine oncornavirus particles. J Virol. 2001;75:11239–11243. doi: 10.1128/JVI.75.22.11239-11243.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nitta T, Kuznetsov Y, McPherson A, Fan H. Murine leukemia virus glycosylated Gag (gPr80gag) facilitates interferon-sensitive virus release through lipid rafts. Proc Natl Acad Sci USA. 2010;107:1190–1195. doi: 10.1073/pnas.0908660107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chun R, Fan H. Recovery of glycosylated gag virus from mice infected with a glycosylated gag-negative mutant of moloney murine leukemia virus. J Biomed Sci. 1994;1:218–223. doi: 10.1007/BF02253305. [DOI] [PubMed] [Google Scholar]
- 13.Corbin A, Prats AC, Darlix JL, Sitbon M. A nonstructural gag-encoded glycoprotein precursor is necessary for efficient spreading and pathogenesis of murine leukemia viruses. J Virol. 1994;68:3857–3867. doi: 10.1128/jvi.68.6.3857-3867.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Santantonio T, et al. Familial clustering of HBV pre-C and pre-S mutants. J Hepatol. 1997;26:221–227. doi: 10.1016/s0168-8278(97)80034-7. [DOI] [PubMed] [Google Scholar]
- 15.Battini JL, Rasko JE, Miller AD. A human cell-surface receptor for xenotropic and polytropic murine leukemia viruses: Possible role in G protein-coupled signal transduction. Proc Natl Acad Sci USA. 1999;96:1385–1390. doi: 10.1073/pnas.96.4.1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tailor CS, Nouri A, Lee CG, Kozak C, Kabat D. Cloning and characterization of a cell surface receptor for xenotropic and polytropic murine leukemia viruses. Proc Natl Acad Sci USA. 1999;96:927–932. doi: 10.1073/pnas.96.3.927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yan Y, Liu Q, Kozak CA. Six host range variants of the xenotropic/polytropic gammaretroviruses define determinants for entry in the XPR1 cell surface receptor. Retrovirology. 2009;6:87. doi: 10.1186/1742-4690-6-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yang YL, et al. Receptors for polytropic and xenotropic mouse leukaemia viruses encoded by a single gene at Rmc1. Nat Genet. 1999;21:216–219. doi: 10.1038/6005. [DOI] [PubMed] [Google Scholar]
- 19.Sitbon M, Nishio J, Wehrly K, Chesebro B. Pseudotyping of dual-tropic recombinant viruses generated by infection of mice with different ecotropic murine leukemia viruses. Virology. 1985;140:144–151. doi: 10.1016/0042-6822(85)90453-2. [DOI] [PubMed] [Google Scholar]
- 20.Pizzato M. MLV glycosylated-Gag is an infectivity factor that rescues Nef-deficient HIV-1. Proc Natl Acad Sci USA. 2010;107:9364–9369. doi: 10.1073/pnas.1001554107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Marshall BJ, et al. Prospective double-blind trial of duodenal ulcer relapse after eradication of Campylobacter pylori. Lancet. 1988;2:1437–1442. doi: 10.1016/s0140-6736(88)90929-4. [DOI] [PubMed] [Google Scholar]
- 22.Xu L, et al. Does a betaretrovirus infection trigger primary biliary cirrhosis? Proc Natl Acad Sci USA. 2003;100:8454–8459. doi: 10.1073/pnas.1433063100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mason AL, Wasilenko ST. Other potential medical therapies: The use of antiviral agents to investigate and treat primary ciliary cirrhosis. Clin Liver Dis. 2008;12:445–460. doi: 10.1016/j.cld.2008.02.006. [DOI] [PubMed] [Google Scholar]
- 24.Singh IR, Gorzynski JE, Drobysheva D, Bassit L, Schinazi RF. Raltegravir is a potent inhibitor of XMRV, a virus implicated in prostate cancer and chronic fatigue syndrome. PLoS ONE. 2010;5:e9948. doi: 10.1371/journal.pone.0009948. [DOI] [PMC free article] [PubMed] [Google Scholar]