Abstract
Leukoencephalopathies in dogs encompass presumably inherited conditions such as leukodystrophies, hypomyelination or spongiform degeneration, but other causes, such as virus infections and toxic or nutritional factors, might also play a contributory role. In this report, we provide evidence of parvovirus infection and replication in the brains of five 6-week-old Cretan hound puppies suffering from a puppy shaker syndrome and leukoencephalopathy. Although these puppies belonged to two different litters, they were closely related, tracing back two generations to the same sire. Histologically, a mild to moderate lymphohistiocytic meningitis, with focal lymphohistiocytic leukoencephalitis in two animals, and a mild to moderate vacuolation with myelin loss, mainly in the white matter of the cerebellum was detected. Vacuolation was also found in the corpus callosum, fimbria hippocampi, mesencephalon, capsula interna, basal ganglia, and hypothalamus. By immunohistology and in situ hybridization, either parvoviral antigen, DNA, mRNA, or replicative intermediate DNA were detected in the cerebellum, hippocampus, periventricular areas, corpus callosum, cerebral cortex, medulla oblongata, and spinal cord. Parvovirus antigen, DNA, and mRNA were present in cells of the outer granular layer of the cerebellum and in periventricular cells, most likely representing spongioblasts, glial cells, neurons, endothelial cells, occasional macrophages, and ependymal cells. Sequencing revealed canine parvovirus type 2 stretches. Thus, an association of parvovirus infection with the leukoencephalopathy seems likely, possibly facilitated by a genetic predisposition due to the mode of inbreeding in this particular dog breed.
Leukoencephalopathies include a variety of different conditions of the central nervous system (CNS) that have been variously designated as leukodystrophies, hypomyelination, or spongiform degeneration (38, 52). Leukodystrophies refers to a series of primary, progressive neurological diseases of undetermined but presumably inherited etiology and are characterized by disturbances of myelin synthesis and maintenance, lack of inflammation, and symmetrical lesions in the brain. In dogs, they have been described in various breeds such as dalmatians, afghan hounds, miniature poodles, and rottweilers (6, 8, 13, 20, 61). Hypomyelination represents one end of the spectrum of leukodystrophies characterized by almost complete myelin loss, accumulation of abnormal myelin, or the presence of inadequate amounts of physiological myelin. Canine hypomyelination has been described for chows, lurcher hounds, springer spaniels, samoyeds, weimaraner dogs, Bernese mountain dogs, and dalmatians (8, 14, 22, 34, 37, 39, 52, 58). The most representative form of white matter spongy degeneration in humans is Canavan's disease, while in veterinary medicine comparable conditions have been described in Labrador retrievers, samoyeds, silkie terriers, and Shetland sheepdogs (36, 45, 60, 62). The hallmark of these conditions is vacuolization in the CNS, which is most likely due to myelin alterations and/or astrocytic swelling and gliosis often associated with myelin loss. A few congenital leukodystrophies are caused by viruses that act as fetal teratogen in these cases, e.g., border disease in sheep and parvovirus B19 infections in humans (3, 4, 27, 52). Parvovirus infection as a cause of leukoencephalopathy in the dog has not yet been described, whereas cerebellar hypoplasia represents a common sequelae of congenital parvovirus infection in the cat (32, 44, 50, 52).
Canine parvovirus is normally associated with enteritis primarily in two to 6-month-old puppies. Canine parvovirus type 2 (CPV-2) was first identified in the late 1970s and was replaced few years after its emerging by two antigenetic variants, CPV-2a and CPV-2b (9, 55). More recently, a new antigenetic variant, CPV-2c, was reported in Italy (10). In general, parvoviruses replicate in the nucleus. However, this depends on certain helper functions of the host cells, such as DNA polymerase, which is expressed in mammalian cells during the S phase (5). Subsequently, parvovirus replication is mainly seen in highly mitotically active cells that progress through the S phase of the cell cycle, such as lymphoid tissue, bone marrow, and the intestine. Surprisingly, parvovirus replication has also been found in the feline brain (56). Similarly, mRNA of parvovirus in brains of dogs could be amplified by using real-time reverse transcription-PCR (RT-PCR), indicating the presence of parvovirus also in the canine brain (16, 18). In contrast, in another study no evidence of parvovirus antigen was found in the central nervous systems of dogs using polyclonal antiserum against canine parvoviruses (57). Furthermore, in a canine case report of necrotizing myocarditis and diffuse leukoencephalomalacia, parvovirus antigen was only detected in the heart and not in the brain (1). In the latter case, leukoencephalomalacia was interpreted as a secondary change as a sequel of cardiovascular disturbances caused by the massive alterations in the heart. Similarly, malacia due to vascular necrosis following canine parvovirus infection has been described previously (35). In summary, the detection of parvovirus in the CNS of dogs is still discussed controversially. Furthermore, although mRNA of parvovirus was amplified in canine brain tissue using real-time RT-PCR, this technique does not allow the identification of the cellular source of the viral mRNA from the brain.
To date, an association of parvovirus infection with meningitis, encephalitis, leukoencephalopathies or peripheral neuropathies has only been described in humans infected with parvovirus B19 (3, 4, 25, 30, 54). Most frequently immunocompromised patients seem to be affected. Interestingly, there is an increasing number of reports on immunocompetent humans with parvovirus B19 infection and associated neurological signs (7, 25). Brain cell susceptibility for parvovirus infection and the underlying pathogenesis still awaits further understanding. It is assumed that direct viral toxicity, autoimmune processes, and cytokine upregulation could represent contributory factors.
In this report, a leukoencephalopathy in Cretan hound puppies suffering from a puppy shaker syndrome and exhibiting parvovirus infection of the CNS is described (49). Furthermore, the cellular source of parvovirus replication in the brain was investigated in order to provide insights in the pathogenesis of leukoencephalopathies and the possible impact of associated virus infections of the CNS.
MATERIALS AND METHODS
Animals.
Two litters of Cretan hound puppies, a dog breed popular in the Greek island of Crete, presented with a history of neurological signs that appeared at the age of 2 to 3 weeks. The first litter included four puppies (two males and two females), and the second litter included two puppies (one male and one female). Efforts from the local breeders to establish the desired phenotypic characteristics of the breed resulted in an intensive inbreeding, using a limited number of dams and sires. The parents of these two litters, although different, were closely related, had repeatedly produced affected puppies in the past, and were clinically normal. Although all dogs were born uneventfully and appeared normal at birth, one puppy from the first and two puppies from the second litter failed to thrive and died of unknown causes within the perinatal period. The remaining puppies were then referred to the Clinic of Companion Animals, Faculty of Veterinary Medicine, Aristotle University of Thessaloniki, by the local attending veterinarians for further evaluation. All litter 1 puppies (four animals) were unvaccinated, whereas those of litter 2 (two animals) had been administered one vaccination (Nobivac Puppy DP; Intervet, Netherlands) with vaccine containing two live attenuated strains of canine distemper and parvovirus days prior to their transportation to the clinic.
The animals were euthanized due to the poor prognosis even after the attempt to treat resistant neurological deficits. A postmortem examination was performed in the four puppies. The entire CNSs (brains and spinal cords) were collected from litter the 1 and 2 dogs.
In addition, the brains of seven dogs between 2 and 6 months of age suffering from parvovirus enteritis were used to obtain more data on the presence of parvovirus antigen in the brain.
Diagnostic investigation.
Complete physical, neurological (according to the method of Thomas and Dewey [53]), and clinicopathological studies, including complete blood counts (CBC), serum biochemistry profiles, and urinalysis, were performed for all puppies. A fecal test for parvovirus antigen was done using the Idexx Snap Parvo test.
Histology.
All tissue samples were fixed by immersion in 10% buffered neutral formalin and processed for routine histopathological, histochemical, and immunohistochemical examination. Briefly, CNS sections from each case, using at least seven different brain areas (cerebral cortex, hippocampus, thalamus, hypothalamus, mesencephalon, cerebellum, and medulla oblongata) and spinal cords were embedded in paraffin wax, cut at ∼3 μm, and stained with hematoxylin and eosin (H&E) for evaluation of the histological alterations. Furthermore, special stains were applied for the demonstration of myelin loss (Luxol fast blue [LFB]), neuronal degeneration (cresyl violet), and storage of lysosomal substances (periodic acid-Schiff [PAS]) according to the method of Romeis and Böck (46).
Immunohistochemistry.
Immunohistochemistry to detect parvovirus antigen was performed using a monoclonal mouse anti-CPV1-2A1 antibody (Custom Monoclonal Antibodies International, Sacramento, CA) according to the method of Kipar et al. (33). Briefly, after incubation with the primary antibody, a biotin-conjugated goat anti-mouse antibody (Vector Laboratories, Inc., Burlingame, CA) was applied. The antigen-antibody complex was visualized by using the avidin-biotin-complex (ABC) method (Vector Laboratories). Tissue sections were counterstained with Mayer's hematoxylin. To exclude other infections, additional immunohistological investigations were carried out against rabies virus, canine distemper virus, canine herpesvirus, canine adenovirus 1, canine parainfluenza virus, tick-borne encephalitis virus, West Nile virus, Borna disease virus, and Prion proteinSc as described previously (49). Furthermore, polyclonal rabbit antibodies specific for S-100 (Sigma, Taufkirchen, Germany) and glial fibrillary acidic protein (GFAP; Dako, Glostrup, Denmark) were used to further characterize the reactive cells within the CNS, as previously described (47).
ISH.
Two different in situ hybridization (ISH) assays were carried out on paraffin-embedded tissue sections. First, ISH was performed with a 315-bp digoxigenin (DIG)-labeled DNA-probe directed against the VP1 gene (48), which served to confirm the presence of parvoviral DNA. The second approach was performed to visualize a potential parvoviral replicatory activity in the CNS by using 222-bp DIG-labeled sense and antisense specific RNA probes, which labeled specifically the VP2 gene. RNA probes were synthesized as previously described (23). Briefly, DNA was extracted from a commercially available parvovirus vaccine (Virbagen canis SHAPPi; Virbac, Bad Oldesloe, Germany). PCR was performed with capsid protein VP-2 specific primers (GenBank accession number NC001539; forward primer, GAAAACGGATGGGTGGAAAT; reverse primer, AGTTGCCAATCTCCTGGATT [51]). Subsequently, PCR products were cloned by using a TOPO TA cloning kit for sequencing (Invitrogen GmbH, Karlsruhe, Germany) according to the protocol provided by Invitrogen. Thereafter, templates were generated by PCR using VP2 forward and reverse specific primers in combination with primers complementary to the M13 forward and M13 reverse priming sites of the vector, respectively. These templates contained the T3 or T7 RNA polymerase binding site and the specific fragment of the parvovirus gene VP2. After purification, RNA probes were DIG labeled and transcribed in vitro using T3 and T7 RNA polymerase (Roche Diagnostics, Mannheim, Germany) in order to obtain strand-specific RNA probes. The RNA probes transcribed by the T3 RNA polymerase were directed against the parvovirus genome, whereas the RNA probe synthesized by the T7 RNA polymerase-labeled viral mRNA and antigenome.
ISH using the DNA probe was performed according to the method of Gaedke et al. (19). For the RNA probes the protocol described by Gröters et al. (23) was used. The detection systems of both ISH procedures consisted of an anti-DIG antibody conjugated with alkaline phosphatase (1:200; Roche Diagnostics) and the substrates nitroblue tetrazolium chloride (NBT) and BCIP (5- bromo-4-chloro-3-indolylphosphate; X-Phosphate; both from Sigma-Aldrich, Taufkirchen, Germany).
PCR detection of CPV-2 in brain tissues of affected puppies.
Formalin-fixed and paraffin-embedded cerebellar tissue was used from one animal to perform PCR. Briefly, DNA was extracted from 10 sections of 10-μm thickness of paraffin-embedded brain tissue. After dewaxing, tissue was digested and DNA extracted and purified by using an ENZA tissue DNA minikit (Peqlab, Erlangen, Germany) according to the manufacturer's protocol. Two different real-time PCR were performed to obtain PCR products using the Mx3005 multiplex quantitative PCR system (Stratagene, La Jolla, CA) and fluorogenic Sybr green (Stratagene, La Jolla, CA) as DNA-binding dye. Primers used for the first PCR were AAAGTGAAAATTATAGAAGAGTGGTTG (forward) and CATGAATATCATCTAAAGCCATGTT (reverse), producing an 84-bp product of the VP-2 gene; the primers used for the second PCR were TGCCTCAATCTGAAGGAGCTA (forward) and CCAACCTCAGCTGGTCTCAT (reverse), producing a 130-bp product of the VP-2 gene. The reaction buffer of both PCRs contained 2.5 mM MgCl2, 150 nM forward and reverse primer, 8.0% glycerol, and 3.0% dimethyl sulfoxide. The thermal profile was set as follows: 95°C for 10 min (1 cycle) and then 95°C for 30 s, 59°C for 30 s, and 72°C for 30 s (45 cycles). DNA extracted from a commercially available parvovirus vaccine (Virbagen canis SHAPPi) served as a positive control. As nontemplate control water was used. The specificity of each reaction was controlled by melting curve analysis, as well as gel electrophoresis. Subsequently, PCR products were cloned by using the TOPO TA cloning kit for sequencing according to the protocol provided by Invitrogen. A second standard PCR with plasmid specific primers was performed; the amplicons obtained were purified by using a NucleoSpin extract kit (Macherey-Nagel, Düren, Germany) and sequenced at SeqLab laboratories, Göttingen, Germany.
Nucleotide sequence accession numbers.
The sequences obtained in the first and second PCRs are available in GenBank under accession numbers HM582864 and HM582865, respectively.
RESULTS
Clinical signs.
In all six puppies neurological signs became evident at the age of 2 to 3 weeks, during their first attempts to ambulate, and appeared static and nonprogressive. No other abnormalities were observed. In addition, the most severely affected puppies were ca. 30% smaller compared to less severely disturbed puppies. In particular, in litter 1 two of the least affected puppies weighed 2 and 1.8 kg, whereas the two severely affected ones weighed 1 and 1.2 kg, respectively. In addition, in litter 2 the less affected dog weighed 2.2, and the severely affected dog weighed 2 kg. This was probably due to their difficulty in feeding properly. During the first week of hospitalization, all unvaccinated first litter puppies developed acute gastrointestinal signs (anorexia, vomiting, and hemorrhagic diarrhea) and were diagnosed with parvoviral enteritis (positive fecal antigen test). Symptomatic and supportive treatment was administered immediately, and the signs subsided progressively within 3 to 4 days.
Neurological signs varied in severity and were characterized by excessive pelvic limb bouncing, intention, and generalized coarse body tremors. Signs were exacerbated by excitement of the dogs during feeding or handling but subsided during rest or sleep. Two puppies from litter 1 and one from litter 2 exhibited severe neurological dysfunction, being completely unable to ambulate, eat, and drink unassisted. In another two dogs, one from each litter, the signs were less debilitating and of moderate intensity, allowing the animals to walk with a wobbling gait but still preventing normal feeding and drinking. The more mildly affected male puppy from litter 1 showed only muscle tremors in the posterior limbs and was in a much better body condition, being almost twice the size of his affected littermates.
Histology.
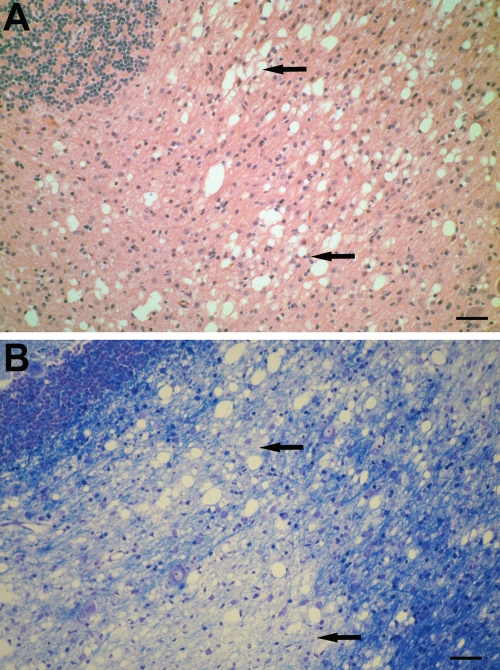
Histopathologically, all dogs showed a mild to moderate lymphohistiocytic meningitis that was accompanied by a focal lymphohistiocytic leukoencephalitis in two animals. Furthermore, a mild to moderate vacuolization of the white matter of cerebellum, corpus callosum, fimbria hippocampi, mesencephalon, capsula interna, basal ganglia, and hypothalamus was noted (Fig. 1A). White matter areas contain typical fibrous astrocytes, but in spongiform areas an increase of astrocytes was not detected by GFAP immunostaining. In two animals, there were fewer GFAP-positive astrocytes in the vacuolated areas and few cells resembling apoptotic cell death. In all animals, S-100 expression was low in white matter areas and, occasionally, small astrocytes resembling plump polygonal astrocytes might be S-100 positive (21). In the gray matter, satellitosis and only few vacuoles were present.
FIG. 1.
Histological changes in the brains of the Cretan hound puppies. (A) Vacuolization of the white matter of the cerebellum (arrows). H&E staining was performed. Bar, 50 μm. (B) Luxol fast blue (LFB) staining of vacuolated area of the white matter of the cerebellum. Note the decreased LFB staining in this area (arrows). Bar, 50 μm.
As determined by LFB staining, mild to moderate myelin loss, especially in the white matter of the cerebellum, was noted (Fig. 1B), whereas myelin staining was unaltered in other vacuolated brain areas. PAS staining did not reveal abnormal storage products in neurons or glial cells in any of the puppies. In the spinal cord and peripheral nerves no significant changes were observed.
Immunohistochemistry for parvovirus antigen.
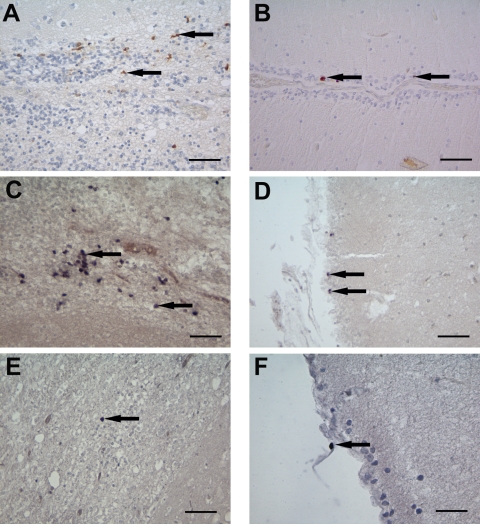
Using immunohistochemistry, parvovirus antigen was demonstrated in the brains of all five dogs, especially in the cerebellum, hippocampus, periventricular areas, corpus callosum, cerebral cortex, and medulla oblongata (Fig. 2A and B). The distribution pattern varied among the five animals; however, the cerebellum and hippocampus were the most constantly affected. In the cerebellum, parvovirus antigen was most frequently present in cells of the outer granular layer (Fig. 2B). Occasionally, the cells of the inner granular and molecular layer, as well as Bergmann glia cells and single neurons, were also infected. In the hippocampus, small positive cells could be found in the dentate gyrus. Interestingly, periventricular cells, most likely representing spongioblasts, glial cells, neurons, endothelial cells, and occasionally macrophages and ependymal cells contained parvovirus antigen in various brain areas (Fig. 2A). Parvovirus antigen was either present in the cytoplasm and/or nuclei of infected cells in all brains investigated.
FIG. 2.
Demonstration of parvovirus antigen, DNA, and mRNA or replicative intermediate in the brain of the Cretan hound puppies. (A and B) Immunohistochemical labeling of parvovirus antigen within cells (arrows) in the periventricular area (A) and cerebellum (B). (C to F) ISH using the T3 (C and D) or the T7 (E and F) RNA probe directed against the parvovirus DNA to detect positive cells (arrows) in the periventricular area (C and E) and cerebellum (D and F). Bar, 50 μm.
As determined by immunohistochemistry, none of the other above-mentioned infectious agents could be detected in the CNS of any puppy.
Of the seven control dogs suffering from parvovirus enteritis, virus antigen was detected in the brains in the cerebellum, dentate gyrus, and in periventricular areas of the lateral ventricles in four animals. In two of the seven control animals only single positive cells were found.
ISH for parvovirus DNA and mRNA.
ISH for the detection of parvoviral DNA revealed positive signals in similar brain areas, as found with immunohistochemistry. Viral DNA was found in the cerebellum, hippocampus, periventricular areas, corpus callosum, cerebral cortex, and medulla oblongata. In the spinal cord, only parvoviral DNA was detected, mostly in endothelial cells. Moreover, viral DNA was found in similar cells as a viral antigen. In detail, parvovirus DNA was found predominantly in the nuclei of cells in the outer and inner granular and molecular layer of the cerebellum, single neurons, periventricular cells, glial cells, neurons, endothelial cells, and a few macrophages and ependymal cells. In addition, faint cytoplasmic staining could be noted in neurons. Similar results were obtained by using the T3 transcribed RNA probe detecting intranuclear parvovirus genomic DNA (Fig. 2C and D).
Active parvovirus replication was confirmed by the presence of positive signals using the second ISH approach by applying the T7 transcribed probe that binds specifically to the cytoplasmic viral mRNA or monomer replicative intermediate strands during parvovirus replication. A positive signal was found within the cytoplasm of cells predominantly in the outer and inner granular and molecular layers of the cerebellum and in the periventricular area (Fig. 2E and F). In addition, single glial cells showed a positive staining within the cytoplasm. However, fewer cell were positive overall using the T7 transcribed probe compared to using the T3 transcribed probe.
PCR.
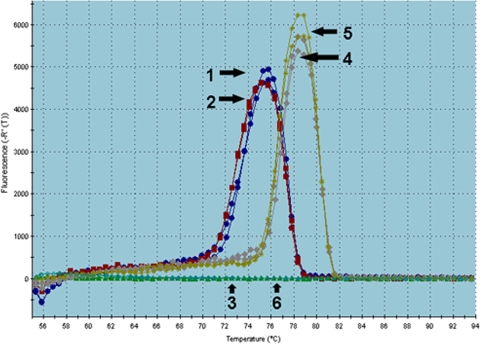
The positive control DNA from the vaccine revealed a detectable signal (critical threshold) after 30 cycles for the first PCR and after 26 cycles for the second PCR. Using the DNA from the cerebellum of the affected puppy, both PCR assays showed a detectable signal of the sample after 36 cycles. In contrast, there was no signal after 45 cycles in the negative controls (Fig. 3 and 4). After successful cloning of the amplicons, DNA sequencing, and analysis, the sequences obtained from the CNS of the affected puppy were classified as CPV genome stretches. The sequence obtained from first PCR (GenBank accession no. HM582864) showed 100% similarity to sequences from canine parvovirus type 2 (FJ197845), type 2a (GU569948), type 2b (GU569944), and type 2c (GQ865519). The sequence from the second PCR (GenBank accession no. HM582865) showed 98% similarity to canine parvovirus type 2 (GU212791), type 2a (EU441280), and type 2b (GU212792).
FIG. 3.
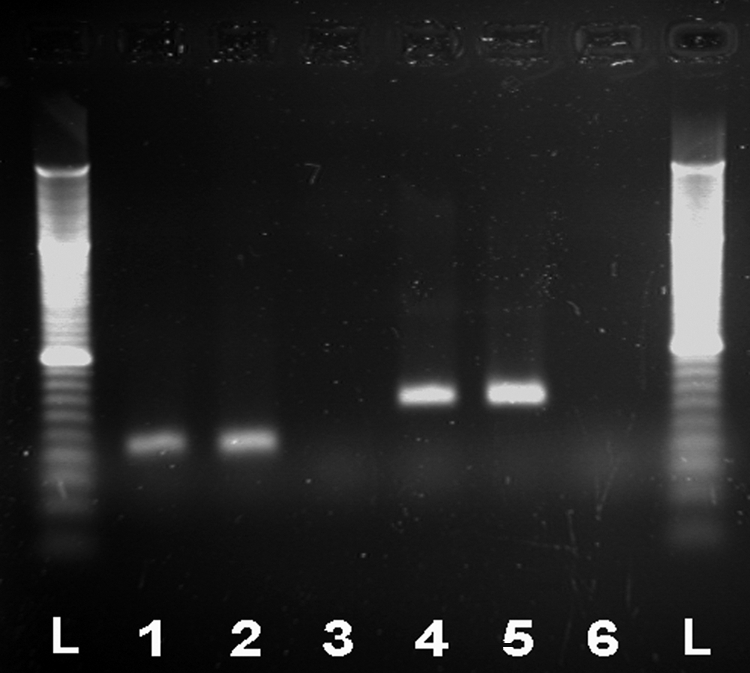
Gel electrophoresis of the real-time PCR. L, 20-bp ladder. Tested sample (lane 1) and positive control (lane 2), both at an 84-bp length, and a negative control (lane 3) of the first PCR are shown. Tested sample (lane 4) and positive control (lane 5) both at a 130-bp length, and negative control (lane 6) of the second PCR are also shown.
FIG. 4.
Melting curve of the real-time PCR. The tested sample (arrow 1) and positive control (arrow 2), both at an 84-bp length, and a negative control (arrow 3) of the first PCR are indicated. The tested sample (arrow 4) and positive control (arrow 5), both at a 130-bp length, and a negative control (arrow 6) of the second PCR are also indicated.
DISCUSSION
In this study, the brains of five Cretan Hound puppies suffering from a puppy shaker syndrome were investigated in detail to explore the cause-and-effect relationship between parvovirus infection and the neurological signs. Interestingly, parvovirus antigen, as well as parvovirus DNA and mRNA, was detected within the brains of all five dogs. Therefore, an association between parvovirus infection and leukoencephalopathy must be considered. Histologically, all five dogs exhibited a mild mononuclear meningitis and a mild-to-moderate vacuolization of white matter areas, especially in the cerebellum. The spongy degeneration is comparable with the idiopathic, most presumably inherited spongy form of leukoencephalopathies; however, the inflammatory reaction is usually lacking in the inherited condition. In the latter, lesions are also typically more severe. Spongy degeneration has thus far only been described for the Labrador retriever, samoyed, silky terrier and Shetland sheepdog (36, 45, 60, 62). In these breeds, widespread white matter vacuolization in various areas of the CNS, often accompanied by astrogliosis and myelin loss has been reported. Due to the close relationship of the parent generation and the mode of inbreeding in the Cretan hound population, a genetically based disorder cannot be excluded. Pre- or perinatal hypoxia-ischemia has been described as other cause of myelin loss in the periventricular and subcortical white matter in two dachshund puppies (43).
Surprisingly, parvovirus of CPV2 group was detected in the brains of all five puppies regardless of prior parvovirus enteritis or vaccination. Moreover, neurological signs started already at 3 weeks of age prior to parvovirus vaccination at the age of 5 to 6 weeks. This might argue for a perinatal or intrauterine infection as described for the cat (32, 44, 50, 52), or it could further indicate the presence of an inherited primary leukoencephalopathy and a secondary parvovirus infection. In humans, an association between parvovirus infection with demyelinating processes and distinct HLA-DR alleles has been described (29, 30). Thus, an inherited predisposition of the Cretan hound puppies to develop white matter vacuolization and myelin loss after parvovirus infection could be possible.
In the Cretan hound puppies parvovirus antigen was found predominantly in cells of outer granular layer of the cerebellum and in periventricular spongioblasts, other glial cells, neurons, endothelial cells, occasional macrophages, and ependymal cells (49). The possible role of parvovirus infection for the shaker syndrome was detailed by characterization of the different brain cell types that can (i) be infected by parvovirus and (ii) are capable of maintaining active virus transcription and replication. To date, canine parvovirus mRNA from the CNS of dogs has been amplified (16, 18), but whether canine brain cells can support parvovirus transcription and replication, as reported for the feline brain (56), remained elusive. As determined by ISH in the Cretan hounds, the cell types containing parvovirus antigen were also positive for viral DNA by applying the T7, as well as the T3 RNA probe, confirming at least active parvovirus transcription and translation in the canine brain. Replication of parvovirus depends on the helper functions of the host cell such as DNA polymerase. It is known that canine and feline cells from the external germinal layer continue to divide for as long as 10 weeks postnatally, hence offering favorable conditions for virus replication (17). Moreover, it is widely accepted that the brain is not a completely irreversible postmitotic tissues as previously thought and that even neurogenesis lasts until adulthood of various species in some brain regions (2, 11, 28). Replication of parvovirus in neurons has already been demonstrated in cats (56) and mice (42), and it is known that certain types of neurons, e.g., cerebellar Purkinje cells, can express the transferrin receptor that is used by canine and feline parvoviruses for viral entry (40, 59). In the present study, parvovirus antigen and DNA were detected in a few neurons, including Purkinje cells. This is in contrast to a previous report, wherein parvovirus antigen was not detectable in 40 canine brains of dogs with parvovirus enteritis (57). In the present study, in four of seven control dogs suffering from parvovirus enteritis and lacking CNS lesions, virus antigen was found in the brain, albeit in two of them only in single cells. The reason for the different results might be either due to infection with different parvovirus strains, the use of different antibodies, or the time point of infection. Recently, it has been shown that the parvovirus type 2 variants 2a, 2b, or 2c exhibit antigenic differences (12), but no differences in tissue tropism have been found thus far (16). However, tropism and host range of parvoviruses is controlled by defined viral capsid structures and canine parvovirus emerged successfully from the feline parvovirus by acquisition of the canine transferrin receptor so that further adaptation of parvoviruses seems possible (26, 40, 59). Decaro et al. (16) also found considerable amounts of parvoviral DNA in the brain by real-time PCR, further supporting the possibility of infection of the canine brain during systemic parvovirus infections.
In humans infected with parvovirus B19 the immune response, autoimmunity, or direct viral toxicity of the NS1 parvovirus protein is assumed to be involved in the pathogenesis of CNS lesions (4). It should be noted that infection of the brain with B19 of humans is considered controversial and that B19 infections have been associated with certain human autoimmune diseases (24, 30, 31, 41). In some cases with B19-associated meningoencephalitis B19 virus could not be detected in the brain (30, 54), and an inappropriate host immune response is assumed as the cause of the brain lesion. In the Cretan hound puppies, a mild-to-moderate lymphohistiocytic meningitis accompanied by a focal lymphohistiocytic leukoencephalitis was present, indicating that inflammatory processes triggered by the parvovirus infection might also play a role in the development of the lesions. For instance, neurogenesis seems to be influenced by the release of proinflammatory mediators (15), and cytokine upregulation has been proposed as a possible pathogenetic mechanism in human B19 infections of the CNS (4). As observed by light microcopy, no obvious neuronal loss was detected in the brains of the Cretan hound puppies. In other infectious conditions, including feline parvovirus infection, a direct infection of the outer granular cells in the cerebellum causes cell death (38, 42, 52). Interestingly, in the puppies, in the vacuolated areas few apoptotic cells and no increase of GFAP- or S100-positive astrocytes were detected, probably indicating a disturbance of glial cell reaction or differentiation as described for hypomyelination in dogs (8). Thus, in the Cretan hound puppies, it remains to be elucidated whether parvovirus infection and associated inflammation can affect glial cell differentiation or neurogenesis.
In summary, the puppy shaker syndrome in the Cretan hound puppies is due to a leukoencephalopathy and concurrent parvovirus infection. The determination of whether a congenital parvovirus infection was the primary cause or whether a genetic predisposition to develop a leukoencephalopathy served as a presensitizing event warrants further investigations. Nevertheless, we unequivocally show here that (i) parvovirus can replicate in distinct cell types in the young canine brain and that (ii) parvovirus infection may be associated with leukoencephalopathies and must therefore be considered as a differential diagnosis for such conditions in young dogs.
Footnotes
Published ahead of print on 30 June 2010.
REFERENCES
- 1.Agungpriyono, D. R., K. Uchida, H. Tabaru, R. Yamaguchi, and S. Tateyama. 1999. Subacute massive necrotizing myocarditis by canine parvovirus type 2 infection with diffuse leukoencephalomalacia in a puppy. Vet. Pathol. 36:77-80. [DOI] [PubMed] [Google Scholar]
- 2.Alvares-Buylla, A., B. Seri, and F. Doetsch. 2002. Identification of neural stem cells in the adult vertebrate brain. Brain Res. Bull. 57:751-758. [DOI] [PubMed] [Google Scholar]
- 3.Barah, F., P. J. Vallely, M. L. Chiswick, G. M. Cleator, and J. R. Kerr. 2001. Association of human parvovirus B19 infection with acute meningoencephalitis. Lancet 358:729-730. [DOI] [PubMed] [Google Scholar]
- 4.Barah, F., P. J. Vallely, G. M. Cleator, and J. R. Kerr. 2003. Neurological manifestations of human parvovirus B19 infection. Rev. Med. Virol. 13:185-199. [DOI] [PubMed] [Google Scholar]
- 5.Berns, K. I. 1990. Parvovirus replication. Microbiol. Rev. 54:316-329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bjerkas, I. 1977. Hereditary “cavitating” leukodystrophy in dalmatian dogs: light and electron microscopic studies. Acta Neuropathol. 40:163-169. [DOI] [PubMed] [Google Scholar]
- 7.Bonvinci, F., G. Marinacci, M. C. Pajno, G. Gallinella, M. Musiani, and M. Zerbini. 2008. Meningoencephalitis with persistent parvovirus B19 infection in an apparently healthy women. Clin. Infect. Dis. 47:385-387. [DOI] [PubMed] [Google Scholar]
- 8.Braund, K. G. 2003. Neuropathic disorders, B0241.0203. In K. G. Braund (ed.), Clinical neurology in small animals: localization, diagnosis and treatment. IVIS, Ithaca, NY.
- 9.Buonavoglia, D., A. Cavalli, A. Pratelli, V. Martella, G. Greco, M. Tempesta, and C. Buonavoglia. 2000. Antigenic analysis of canine parvovirus strains isolated in Italy. New Microbiol. 23:93-96. [PubMed] [Google Scholar]
- 10.Buonavoglia, C., V. Martella, A. Pratelli, M. Tempesta, A. Cavalli, D. Buonavoglia, G. Bozzo, G. Elia, N. Decaro, and L. Carmichael. 2001. Evidence for evolution of canine parvovirus type 2 in Italy. J. Gen. Virol. 82:3021-3025. [DOI] [PubMed] [Google Scholar]
- 11.Cai, Y., K. Xiong, Y. Chu, D. W. Luo, X. G. Luo, X. Y. Yuan, R. G. Struble, R. W. Clough, D. D. Spencer, A. Williamson, J. H. Kordower, P. R. Patrylo, and X. X. Yan. 2009. Doublecortin expression in adult cat and primate cerebral cortex relates to immature neurons that develop into GABAergic subgroups. Exp. Neurol. 216:342-356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cavalli, A., V. Martella, C. Desario, M. Camero, A. L. BellaciccoL, P. De Palo, N. Decaro, G. Elia, and C. Buonavoglia. 2008. Evaluation of the antigenic relationships among canine parvovirus type 2 variants. Clin. Vaccine Immunol. 15:534-539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cummings, J. F., and A. DeLahunta. 1978. Hereditary myelopathy of Afghan hounds, a myelinolytic disease. Acta Neuropathol. 42:173-181. [DOI] [PubMed] [Google Scholar]
- 14.Cummings, J. F., B. A. Summers, A. de Lahunta, and C. Lawson. 1986. Tremors in Samoyed pups with oligodendrocyte deficiencies and hypomyelination. Acta Neuropathol. 71:267-277. [DOI] [PubMed] [Google Scholar]
- 15.Das, S., and A. Basu. 2008. Inflammation: a new candidate in modulating adult neurogenesis. J. Neurosci. Res. 86:1199-1208. [DOI] [PubMed] [Google Scholar]
- 16.Decaro, N., V. Martella, G. Elia, C. Desario, M. Campolo, E. Lorusso, M. L. Colaianni, A. Lorusso, and C. Buonavoglia. 2007. Tissue distribution of the antigenic variants of canine parvovirus type 2 in dogs. Vet. Microbiol. 121:39-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.De Lahunta, A., and E. Glass. 2008. Development of the nervous system: malformation, p. 23-53. In A. de Lahunta and E. Grass (ed.), Veterinary neuroanatomy and clinical neurology, 3rd ed. Saunders/Elsevier, Philadelphia, PA.
- 18.Elia, G., A. Cavalli, C. Desario, E. Lorusso, M. S. Lucente, N. Decaro, V. Martella, and C. Buonavoglia. 2007. Detection of infectious canine parvovirus type 2 by mRNA real-time RT-PCR. J. Virol. Methods 146:202-208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gaedke, K., J. P. Teifke, M. Hardt, S. Alldinger, and W. Baumgärtner. 1995. Detection of distemper virus N protein RNA in the brain of dogs with spontaneous distemper encephalitis using a digoxigenin-labeled, double-stranded DNA probe for in situ hybridization. Berl. Munch. Tierarztl. Wochensch. 108:51-54. (In German.) [PubMed] [Google Scholar]
- 20.Gamble, D. A., and C. L. Chrisman. 1984. A leukoencephalomyelopathy of rottweiler dogs. Vet. Pathol. 21:274-280. [DOI] [PubMed] [Google Scholar]
- 21.Gerhauser, I., W. Baumgärtner, and C. Herden. 2007. Unusual type of reactive astrocytes in the feline central nervous system. Dtsch. Tierarztl. Wochenschr. 114:124-128. [PubMed] [Google Scholar]
- 22.Griffiths, I. R., I. D. Duncan, and M. McCulloch. 1981. Shaking pups: a disorder of central myelination in the spaniel dog. II. Ultrastructural observations on the white matter of the cervical spinal cord. J. Neurocytol. 10:847-858. [DOI] [PubMed] [Google Scholar]
- 23.Gröters, S., S. Alldinger, and W. Baumgärtner. 2005. Up-regulation of mRNA for matrix metalloproteinases-9 and-14 in advanced lesions of demyelinating canine distemper leukoencephalitis. Acta Neuropathol. 110:369-382. [DOI] [PubMed] [Google Scholar]
- 24.Hammond, C. J., and J. A. Hobbs. 2007. Parvovirus B19 infection of brain: possible role of gender in determining mental illness and autoimmune thyroid disorders. Med. Hypotheses 69:113-116. [DOI] [PubMed] [Google Scholar]
- 25.Hobbs, J. A. 2007. Parvovirus B19-brain interactions: infection, autoimmunity, or both? J. Clin. Virol. 38:364-365. [DOI] [PubMed] [Google Scholar]
- 26.Hueffer, K., and C. R. Parrish. 2003. Parvovirus host range, cell tropism and evolution. Curr. Opin. Microbiol. 6:392-398. [DOI] [PubMed] [Google Scholar]
- 27.Isumi, H., T. Nunoue, A. Nishida, and S. Takashima. 1999. Fetal brain infection with human parvovirus B19. Pediatr. Neurol. 21:661-663. [DOI] [PubMed] [Google Scholar]
- 28.Kempermann, G., L. Wiskott, and F. H. Gage. 2004. Functional significance of adult neurogenesis. Curr. Opin. Neurobiol. 14:186-191. [DOI] [PubMed] [Google Scholar]
- 29.Kerr, J. R., F. Barah, M. L. Chiswick, G. V. McDonnell, J. Smith, M. D. Chapman, J. B. Bingham, P. Kelleher, and M. N. Sheppard. 2002. Evidence for the role of demyelination, HLA-DR alleles, and cytokines in the pathogenesis of parvovirus B19 meningoencephalitis and its sequelae. J. Neurol. Neurosurg. Psychiatry 73:739-746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kerr, J. R., N. Kaushik, D. Fear, D. A. Baldwin, E. F. Nuwaysir, and I. M. Adcock. 2005. Single-nucleotide polymorphisms associated with symptomatic infection and differential human gene expression in healthy seropositive persons each implicate the cytoskeleton, integrin signaling, and oncosuppression in the pathogenesis of human parvovirus B19 infection. J. Infect. Dis. 192:276-286. [DOI] [PubMed] [Google Scholar]
- 31.Kerr, J. R., and D. L. Mattey. 2008. Preexisting psychological stress predicts acute and chronic fatigue and arthritis following symptomatic parvovirus B19 infection. Clin. Infect. Dis. 46:83-87. [DOI] [PubMed] [Google Scholar]
- 32.Kilham, L., G. Margolis, and E. D. Colby. 1971. Cerebellar ataxia and its congenital transmission in cats by feline panleukopenia virus. J. Am. Vet. Med. Assoc. 15:158. [PubMed] [Google Scholar]
- 33.Kipar, A., J. Kremendahl, C. K. Grant, I. von Bothmer, and M. Reinacher. 2000. Expression of viral proteins in feline leukemia virus-associated enteritis. Vet. Pathol. 37:129-136. [DOI] [PubMed] [Google Scholar]
- 34.Kornegay, J. N., M. A. Goodwin, and L. K. Spyridakis. 1987. Hypomyelination in Weimaraner dogs. Acta Neuropathol. 72:394-401. [DOI] [PubMed] [Google Scholar]
- 35.Lenghaus, C., and M. J. Studdert. 1982. Generalized parvovirus disease in neonatal pups. J. Am. Vet. Med. Assoc. 181:41-45. [PubMed] [Google Scholar]
- 36.Mason, R., W. Hartley, and M. Randall. 1979. Spongiform degeneration of the white matter in a Samoyed pup. Aus. Vet. Pract. 9:11-13. [Google Scholar]
- 37.Mayhew, I. G., W. F. Blakemore, A. C. Palmer, and C. J. Clarke. 1984. Tremor syndrome hypomyelination in Lurcher pups. J. Small Anim. Pract. 25:551-559. [Google Scholar]
- 38.Maxie, M. G., and S. Youssef. 2007. Nervous system, p. 378-393. In M. G. Maxie (ed.), Jubb, Kennedy, and Palmer's pathology of domestic animals, 5th ed., vol. 1. Elsevier/Saunders, Philadelphia, PA. [Google Scholar]
- 39.Palmer, A. C., W. F. Blakemore, M. E. Wallace, M. K. Wilkes, M. E. Herrtage, and S. E. Matic. 1987. Recognition of “trembler,” a hypomyelinating condition in the Bernese mountain dog. Vet. Rec. 120:609-612. [DOI] [PubMed] [Google Scholar]
- 40.Parker, J. S., W. J. Murphy, D. Wang, S. J. O'Brien, and C. R. Parrish. 2001. Canine and feline parvoviruses can use human or feline transferrin receptors to bind, enter, and infect cells. J. Virol. 75:3896-3902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pugliese, A., T. Beltramo, D. Torre, and D. Roccatello. 2007. Parvovirus B19 and immune disorders. Cell. Biochem. Funct. 25:639-641. [DOI] [PubMed] [Google Scholar]
- 42.Ramírez, J. C., A. Fairén, and J. M. Almendral. 1996. Parvovirus minute virus of mice strain multiplication and pathogenesis in the newborn mouse brain are restricted to proliferative areas and to migratory cerebellar young neurons. J. Virol. 70:8109-8116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rentmeister, K., S. Schmidbauer, M. Hewicker-Trautwein, and A. Tipold. 2004. Periventricular and subcortical leukoencephalopathy in two dachshund puppies. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2004 7-8:327-331. [DOI] [PubMed] [Google Scholar]
- 44.Résibois, A., A. Coppens, and L. Poncelet. 2007. Naturally occurring parvovirus-associated feline hypogranular cerebellar hypoplasia: a comparison to experimentally induced lesions using immunohistology. Vet. Pathol. 44:831-841. [DOI] [PubMed] [Google Scholar]
- 45.Richards, R. B., and B. A. Kakulas. 1978. Spongiform leucoencephalopathy associated with congenital myoclonia syndrome in the dog. J. Comp. Pathol. 88:317-320. [DOI] [PubMed] [Google Scholar]
- 46.Romeis, B., and P. Böck. 1989. Nervenge webe, p. 571-614. In B. Romeis and P. Böck (ed.), Mikroskopische Technik, 17th ed. Urban & Fischer, Munich, Germany.
- 47.Schaudien, D., J. M. V. Müller, and W. Baumgärtner. 2007. Omental leiomyoma in a male adult horse. Vet. Pathol. 44:722-726. [DOI] [PubMed] [Google Scholar]
- 48.Schaudien, D., H. Meyer, D. Grunwald, H. Janssen, and P. Wohlsein. 2007. Concurrent infection of a cat with cowpox virus and feline parvovirus. J. Comp. Pathol. 137:151-154. [DOI] [PubMed] [Google Scholar]
- 49.Schwab, S., C. Herden, F. Seeliger, N. Papaioannou, D. Psalla, Z. Polizopulou, and W. Baumgärtner. 2007. Non-suppurative meningoencephalitis of unknown origin in cats and dogs: an immunohistochemical study. J. Comp. Pathol. 136:96-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sharp, N. J., B. J. Davis, J. S. Guy, J. M. Cullen, S. F. Steingold, and J. N. Kornegay. 1999. Hydranencephaly and cerebellar hypoplasia in two kittens attributed to intrauterine parvovirus infection. J. Comp. Pathol. 121:39-53. [DOI] [PubMed] [Google Scholar]
- 51.Steinel, A., L. Munson, M. van Vuuren, and U. Truyen. 2000. Genetic characterization of feline parvovirus sequences from various carnivores. J. Gen. Virol. 81:345-350. [DOI] [PubMed] [Google Scholar]
- 52.Summers, B. A., J. F. Cummings, and A. de Lahunta. 1995. Inflammatory diseases of the central nervous system, p. 95-188. In B. A. Summers, J. F. Cummings, and A. de Lahunta (ed.), Veterinary neuropathology. Mosby, St. Louis, MO.
- 53.Thomas, W. B., and C. W. Dewey. 2008. Performing the neurologic examination, p. 53-74. In C. W. Dewey (ed.), A practical guide to canine and feline neurology. Wiley-Blackwell, Ames, IA.
- 54.Tonnellier, M., J. Bessereau, N. Carbonnell, B. Guidet, J. F. Méritet, J. R. Kerr, L. Monnier-Cholley, G. Offenstadt, and E. Maury. 2007. A possible parvovirus B19 encephalitis in an immunocompetent adult patient. J. Clin. Virol. 38:186-187. [DOI] [PubMed] [Google Scholar]
- 55.Truyen, U., G. Platzer, and C. R. Parrish. 1996. Antigenic type distribution among canine parvoviruses in dogs and cats in Germany. Vet. Rec. 138:365-366. [DOI] [PubMed] [Google Scholar]
- 56.Url, A., U. Truyen, B. Rebel-Bauder, H. Weissenböck, and P. Schmidt. 2003. Evidence of parvovirus replication in cerebral neurons of cats. J. Clin. Microbiol. 41:3801-3805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Url, A., and P. Schmidt. 2005. Do canine parvoviruses affect canine neurons? An immunohistochemical study. Res. Vet. Sci. 79:57-59. [DOI] [PubMed] [Google Scholar]
- 58.Vandevelde, M., K. G. Braund, T. L. Walker, and J. N. Kornegay. 1978. Dysmyelination of the central nervous system in the Chow Chow dog. Acta Neuropathol. 42:211-215. [DOI] [PubMed] [Google Scholar]
- 59.Vihinen-Ranta, M., S. Suikkanen, and C. R. Parrish. 2004. Pathways of cell infection by parvoviruses and adeno-associated viruses. J. Virol. 78:6709-6714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wood, S. L., and J. S. Patterson. 2001. Shetland sheepdog leukodystrophy. J. Vet. Intern. Med. 15:486-493. [DOI] [PubMed] [Google Scholar]
- 61.Wouda, W., and J. J. van Nes. 1986. Progressive ataxia due to central demyelination in the rottweiler. Vet. Q. 8:89-97. [DOI] [PubMed] [Google Scholar]
- 62.Zachary, J. F., and D. P. O'Brien. 1985. Spongy degeneration of the central nervous system in two canine littermates. Vet. Pathol. 22:561-571. [DOI] [PubMed] [Google Scholar]