Abstract
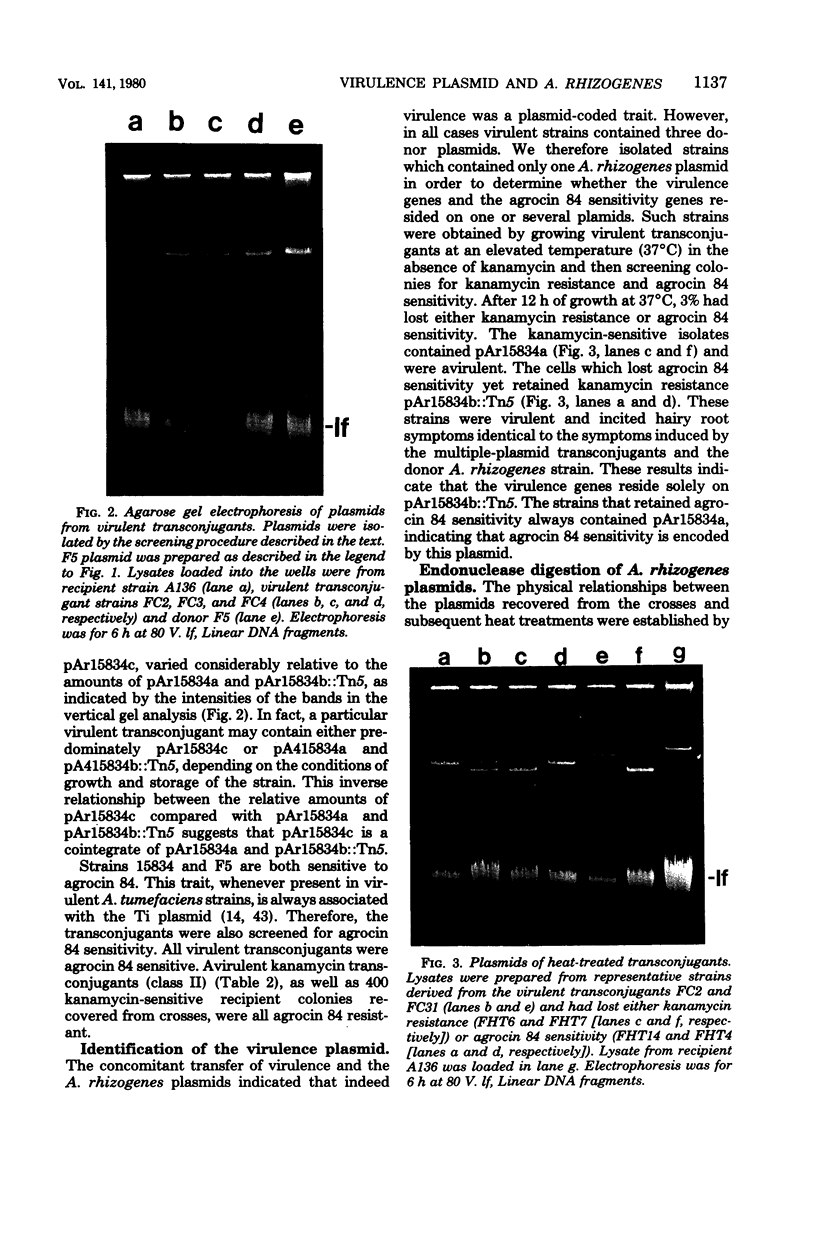
Agrobacterium rhizogenes strain 15834, which incites hairy root disease in plants, harbors three large plasmids: pAr15834a (107 x 10(6) daltons), pAr15834b (154 x 10(6) daltons), and pAr15834c (258 x 10(6) daltons). Kanamycin-resistant transconjugants were selected in a cross of kanamycin-resistant derivate of strain 15834 and an avirulent recipient. The transconjugants belonging to one class were virulent and contained all three donor plasmids. These transconjugants also acquired sensitivity to the bacteriocin agrocin 84. The loss of plasmids from virulent transconjugants during growth at 37 degrees C indicated that virulence genes reside on pAr15834b, whereas agrocin 84 sensitivity genes reside on pAr15834a. The pathology induced by the virulent transconjugants containing only pAr15834b was identical to that produced by the wild-type strain of A. rhizogenes. Restriction endonuclease fragment analysis of plasmids from the transconjugants and the donor revealed that pAr15834c is a cointegrate of pAr15834a and pAr15834b. Kanamycin-resistant transconjugants belonging to a second class were avirulent and contained an altered form of pAr15834b. Strain 15834 can utilize octopine. However, this trait was not detected in any of the transconjugants. Octopine is not synthesized by infected plant tissue.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berg D. E., Davies J., Allet B., Rochaix J. D. Transposition of R factor genes to bacteriophage lambda. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3628–3632. doi: 10.1073/pnas.72.9.3628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bomhoff G., Klapwijk P. M., Kester H. C., Schilperoort R. A., Hernalsteens J. P., Schell J. Octopine and nopaline synthesis and breakdown genetically controlled by a plasmid of Agrobacterium tumefaciens. Mol Gen Genet. 1976 May 7;145(2):177–181. doi: 10.1007/BF00269591. [DOI] [PubMed] [Google Scholar]
- Chilton M. D., Currier T. C., Farrand S. K., Bendich A. J., Gordon M. P., Nester E. W. Agrobacterium tumefaciens DNA and PS8 bacteriophage DNA not detected in crown gall tumors. Proc Natl Acad Sci U S A. 1974 Sep;71(9):3672–3676. doi: 10.1073/pnas.71.9.3672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chilton M. D., Drummond M. H., Merio D. J., Sciaky D., Montoya A. L., Gordon M. P., Nester E. W. Stable incorporation of plasmid DNA into higher plant cells: the molecular basis of crown gall tumorigenesis. Cell. 1977 Jun;11(2):263–271. doi: 10.1016/0092-8674(77)90043-5. [DOI] [PubMed] [Google Scholar]
- Chilton M. D., Montoya A. L., Merlo D. J., Drummond M. H., Nutter R., Gordon M. P., Nester E. W. Restriction endonuclease mapping of a plasmid that confers oncogenicity upon Agrobacterium tumefaciens strain B6-806. Plasmid. 1978 Feb;1(2):254–269. doi: 10.1016/0147-619x(78)90043-4. [DOI] [PubMed] [Google Scholar]
- Currier T. C., Nester E. W. Evidence for diverse types of large plasmids in tumor-inducing strains of Agrobacterium. J Bacteriol. 1976 Apr;126(1):157–165. doi: 10.1128/jb.126.1.157-165.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Currier T. C., Nester E. W. Isolation of covalently closed circular DNA of high molecular weight from bacteria. Anal Biochem. 1976 Dec;76(2):431–441. doi: 10.1016/0003-2697(76)90338-9. [DOI] [PubMed] [Google Scholar]
- Engler G., Holsters M., Van Montagu M., Schell J., Hernalsteens J. P., Schilperoort Agrocin 84 sensitivity: a plasmid determined property in Agrobacterium tumefaciens. Mol Gen Genet. 1975 Jul 10;138(4):345–349. doi: 10.1007/BF00264804. [DOI] [PubMed] [Google Scholar]
- Gresshoff P. M., Skotnicki M. L., Rolfe B. G. Crown gall teratoma formation is plasmid and plant controlled. J Bacteriol. 1979 Feb;137(2):1020–1021. doi: 10.1128/jb.137.2.1020-1021.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen J. B., Olsen R. H. Isolation of large bacterial plasmids and characterization of the P2 incompatibility group plasmids pMG1 and pMG5. J Bacteriol. 1978 Jul;135(1):227–238. doi: 10.1128/jb.135.1.227-238.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holsters M., Silva B., Genetello C., Engler G., van Vliet F., de Block M., Villarroel R., van Montagu M., Schell J. Spontaneous formation of cointegrates of the oncogenic Ti-plasmid and the wide-host-range P-plasmid RP4. Plasmid. 1978 Sep;1(4):456–467. doi: 10.1016/0147-619x(78)90004-5. [DOI] [PubMed] [Google Scholar]
- KLEINSCHMIDT A. K., LANG D., JACHERTS D., ZAHN R. K. [Preparation and length measurements of the total desoxyribonucleic acid content of T2 bacteriophages]. Biochim Biophys Acta. 1962 Dec 31;61:857–864. [PubMed] [Google Scholar]
- Kleckner N., Roth J., Botstein D. Genetic engineering in vivo using translocatable drug-resistance elements. New methods in bacterial genetics. J Mol Biol. 1977 Oct 15;116(1):125–159. doi: 10.1016/0022-2836(77)90123-1. [DOI] [PubMed] [Google Scholar]
- Lang D. Molecular weights of coliphages and coliphage DNA. 3. Contour length and molecular weight of DNA from bacteriophages T4, T5 and T7, and from bovine papilloma virus. J Mol Biol. 1970 Dec 28;54(3):557–565. doi: 10.1016/0022-2836(70)90126-9. [DOI] [PubMed] [Google Scholar]
- Lippincott J. A., Beiderbeck R., Lippincott B. B. Utilization of octopine and nopaline by Agrobacterium. J Bacteriol. 1973 Oct;116(1):378–383. doi: 10.1128/jb.116.1.378-383.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merlo D. J., Nester E. W. Plasmids in avirulent strains of Agrobacterium. J Bacteriol. 1977 Jan;129(1):76–80. doi: 10.1128/jb.129.1.76-80.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyers J. A., Sanchez D., Elwell L. P., Falkow S. Simple agarose gel electrophoretic method for the identification and characterization of plasmid deoxyribonucleic acid. J Bacteriol. 1976 Sep;127(3):1529–1537. doi: 10.1128/jb.127.3.1529-1537.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montoya A. L., Chilton M. D., Gordon M. P., Sciaky D., Nester E. W. Octopine and nopaline metabolism in Agrobacterium tumefaciens and crown gall tumor cells: role of plasmid genes. J Bacteriol. 1977 Jan;129(1):101–107. doi: 10.1128/jb.129.1.101-107.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montoya A. L., Moore L. W., Gordon M. P., Nester E. W. Multiple genes coding for octopine-degrading enzymes in Agrobacterium. J Bacteriol. 1978 Dec;136(3):909–915. doi: 10.1128/jb.136.3.909-915.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore L., Warren G., Strobel G. Involvement of a plasmid in the hairy root disease of plants caused by Agrobacterium rhizogenes. Plasmid. 1979 Oct;2(4):617–626. doi: 10.1016/0147-619x(79)90059-3. [DOI] [PubMed] [Google Scholar]
- New P. B., Kerr A. A selective medium for Agrobacterium radiobacter biotype 2. J Appl Bacteriol. 1971 Mar;34(1):233–236. doi: 10.1111/j.1365-2672.1971.tb02281.x. [DOI] [PubMed] [Google Scholar]
- Roberts W. P., Tate M. E., Kerr A. Agrocin 84 is a 6-N-phosphoramidate of an adenine nucleotide analogue. Nature. 1977 Jan 27;265(5592):379–381. doi: 10.1038/265379a0. [DOI] [PubMed] [Google Scholar]
- Sciaky D., Montoya A. L., Chilton M. D. Fingerprints of Agrobacterium Ti plasmids. Plasmid. 1978 Feb;1(2):238–253. doi: 10.1016/0147-619x(78)90042-2. [DOI] [PubMed] [Google Scholar]
- Van Larebeke N., Engler G., Holsters M., Van den Elsacker S., Zaenen I., Schilperoort R. A., Schell J. Large plasmid in Agrobacterium tumefaciens essential for crown gall-inducing ability. Nature. 1974 Nov 8;252(5479):169–170. doi: 10.1038/252169a0. [DOI] [PubMed] [Google Scholar]
- Watson B., Currier T. C., Gordon M. P., Chilton M. D., Nester E. W. Plasmid required for virulence of Agrobacterium tumefaciens. J Bacteriol. 1975 Jul;123(1):255–264. doi: 10.1128/jb.123.1.255-264.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Vliet F., Silva B., van Montagu M., Schell J. Transfer of RP4::mu plasmids to Agrobacterium tumefaciens. Plasmid. 1978 Sep;1(4):446–455. doi: 10.1016/0147-619x(78)90003-3. [DOI] [PubMed] [Google Scholar]