Abstract
Background
Varicella zoster virus (VZV) vasculopathy produces stroke secondary to viral infection of cerebral arteries. Not all patients have rash before cerebral ischemia or stroke. Furthermore, other vasculitides produce similar clinical features and comparable imaging, angiographic, and CSF abnormalities.
Methods
We review our 23 published cases and 7 unpublished cases of VZV vasculopathy. All CSFs were tested for VZV DNA by PCR and anti-VZV IgG antibody and were positive for either or both.
Results
Among 30 patients, rash occurred in 19 (63%), CSF pleocytosis in 20 (67%), and imaging abnormalities in 29 (97%). Angiography in 23 patients revealed abnormalities in 16 (70%). Large and small arteries were involved in 15 (50%), small arteries in 11 (37%), and large arteries in only 4 (13%) of 30 patients. Average time from rash to neurologic symptoms and signs was 4.1 months, and from neurologic symptoms and signs to CSF virologic analysis was 4.2 months. CSF of 9 (30%) patients contained VZV DNA while 28 (93%) had anti-VZV IgG antibody in CSF; in each of these patients, reduced serum/CSF ratio of VZV IgG confirmed intrathecal synthesis.
Conclusions
Rash or CSF pleocytosis is not required to diagnose varicella zoster virus (VZV) vasculopathy, whereas MRI/CT abnormalities are seen in almost all patients. Most patients had mixed large and small artery involvement. Detection of anti-VZV IgG antibody in CSF was a more sensitive indicator of VZV vasculopathy than detection of VZV DNA (p < 0.001). Determination of optimal antiviral treatment and benefit of concurrent steroid therapy awaits studies with larger case numbers.
The clinical diagnosis of varicella zoster virus (VZV) vasculopathy is usually based on a history of recent zoster followed by neurologic symptoms and signs; imaging abnormalities indicating cerebral ischemia, infarction, or hemorrhage; angiographic evidence of narrowing or beading in cerebral arteries; and a CSF pleocytosis. Vasculopathy typically involves one or more cerebral arteries. Unifocal vasculopathy follows ophthalmic-distribution zoster in elderly adults or childhood chickenpox and commonly affects large arteries of the anterior (figure 1A) or posterior circulation. Multifocal vasculopathy usually affects branches of large cerebral arteries (figure 1B) or small cerebral arteries (figure 1C), mostly in immunocompromised individuals. The diagnosis of VZV vasculopathy is not always straightforward since 1) neurologic disease often develops weeks and sometimes months after zoster, so that TIAs or stroke is often attributed to arteriosclerotic disease rather than virus infection in cerebral arteries; 2) not all patients with pathologically and virologically verified disease have a history of zoster rash or chickenpox; 3) vasculopathies of other etiologies produce the same neurologic symptoms, signs, and CSF and imaging abnormalities; and 4) virologic analysis is often limited to a search only for VZV DNA in CSF, which is negative in most cases of VZV vasculopathy, in contrast to the detection of anti-VZV IgG antibody in CSF, which is the virologic test of choice to diagnose disease.1
Figure 1. Characteristic angiographic, imaging, and pathologic abnormalities in varicella zoster virus (VZV) vasculopathy.
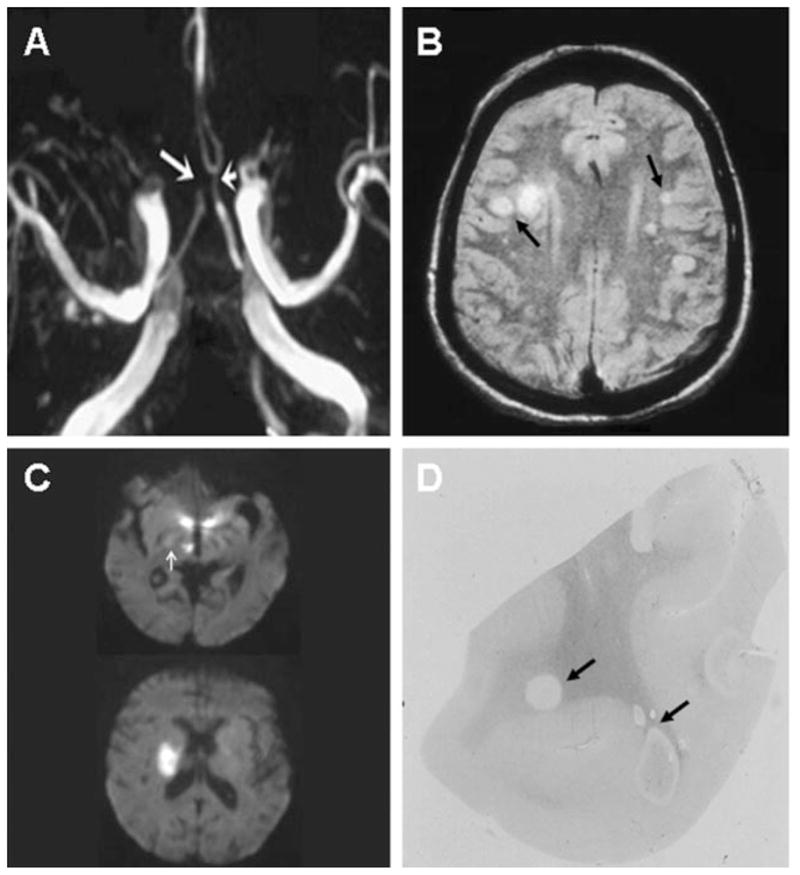
(A) Three-dimensional time-of-flight magnetic resonance angiography of the Circle of Willis shows marked narrowing of the left anterior cerebral artery (short arrow) and occlusion of the right anterior cerebral artery (long arrow). (B) Brain MRI scan shows multiple areas of infarction in both hemispheres, primarily involving white matter and gray-white matter junctions (arrows). (C) Diffusion-weighted MRI in a patient with small vessel VZV vasculopathy. Top figure shows two ischemic lesions in the posterior thalamus and one in the hypothalamus; in addition, a small ischemic lesion is seen in the posterior limb of the internal capsule (white arrow); 1 week later, the patient became hemiplegic. Although the ischemic thalamic and hypothalamic lesions had resolved, a new MRI (bottom figure) showed a discrete infarct in the area of the posterior limb of the internal capsule. (D) Macroscopic changes in brain from a patient who died of chronic VZV vasculopathy; arrows indicate ovoid areas of ischemia/demyelination of varying size, primarily at gray-white matter junctions. (A) Reprinted from Gilden DH, Lipton HL, Wolf JS, et al. Two patients with unusual forms of varicella zoster virus vasculopathy. N Engl J Med 2002;347:1500–1503. Reprinted with permission from Massachusetts Medical Society. Copyright 2002. All rights reserved. (B) Reprinted from Gilden DH, Mahalingam R, Cohrs RJ, Kleinschmidt-DeMasters BK, Forghani B. The protean manifestations of varicella-zoster virus vasculopathy. J Neurovirol 2002;8:75–79. Reprinted with permission from Journal of Neurovirology. (D) Reprinted from Nagel MA, Gilden DH. Protean neurologic manifestations of varicella zoster virus infection. Cleve Clin J Med 2007;74: 489–504. Reprinted with permission from the Cleveland Clinic Journal of Medicine.
Most of the literature on VZV vasculopathy has been individual case reports. The largest review, nearly 25 years ago, summarized clinical, imaging, angiographic, and CSF abnormalities in 29 patients with herpes zoster ophthalmicus followed by contralateral hemiparesis.2 Arterial disease was restricted to large cerebral arteries, and most cases were not verified virologically by isolation of VZV from CSF or detection of anti-VZV IgG antibody in CSF. At the time of that review, VZV vasculopathy in the absence of rash and small vessel disease produced by VZV was not recognized, MRI and MRA scanning had not been developed, and PCR had not been applied to the detection of VZV DNA in CSF.
Herein, we provide an updated review of 30 virologically verified cases of VZV vasculopathy (23 published and 7 unpublished). We determined the frequency of rash, CSF pleocytosis, imaging abnormalities on CT or MRI, angiographic abnormalities by conventional angiography or MRA, and the value of detecting VZV DNA and anti-VZV IgG antibody in the CSF to confirm the diagnosis. We also examined the time from rash to the onset of neurologic symptoms and signs as well as the time from the onset of neurologic symptoms and signs to virologic analysis. We further determined the frequency of large artery involvement, small artery involvement, or both, and studied the effect of treatment on outcome.
METHODS
We reviewed all 30 patients with neurologic symptoms or signs, and imaging, angiographic, or CSF abnormalities consistent with CNS vasculopathy, and whose CSF tested positive for VZV DNA by PCR or for anti-VZV IgG antibody by enzyme immunoabsorbent assay (EIA), or both. Of the 30 cases, CSF samples from 17 were analyzed for VZV DNA by nested PCR which detects one copy of VZV DNA per microgram of DNA.3 For 2 other patients, PCR data with a sensitivity of 50 to 250 copies of VZV DNA/mL of CSF were available; in 11 patients, the sensitivity of PCR was unknown. Anti-VZV IgG antibody was assessed in 18 patients as previously described.1 Among the remaining 12 patients, EIA on 8 was performed using kits from commercial laboratories (Behring Diagnostics, Marburg; SRL, Inc., Tokyo; Denka-Seiken Co., Ltd., Tokyo; and Bio-Merieux, Marcy l’Etoile); the laboratory that performed EIA on the remaining 4 patients is unknown. In every patient in whom anti-VZV IgG antibody was detected in the CSF, intrathecal synthesis of antibody was confirmed by comparing the serum/CSF ratio of anti-VZV IgG antibody to the serum/CSF ratio of albumin and total IgG as described.4
Based on brain imaging as well as vascular studies in many cases, arterial disease was classified as exclusively large artery, exclusively small artery, or mixed (both large and small artery involvement). Large arteries included the internal or external carotid arteries, the anterior cerebral arteries or their major branches (i.e., the pericallosal or callosal marginal arteries), the middle cerebral arteries or their major branches (M2, M3), the posterior cerebral arteries, the basilar artery, vertebral arteries, and the anterior or posterior inferior cerebellar arteries, or ischemic lesions in their corresponding distribution on brain imaging. In contrast, involvement of any areas of the brain that would be supplied by penetrating arteries emanating from the base of the brain that would supply deep-seated structures such as the thalamus or brainstem in which the basilar and posterior cerebral artery were normal was considered small artery disease.
RESULTS
The combined clinical, imaging, angiographic, or CSF abnormalities identified 30 patients with a clinical diagnosis consistent with VZV vasculopathy. Every patient had their CSF tested for both VZV DNA and anti-VZV IgG antibody; positivity for either or both confirmed the clinical diagnosis of VZV vasculopathy. In patients with anti-VZV IgG antibody in their CSF, the diagnosis was further corroborated by intrathecal synthesis of the antibody.
Clinical features, CSF pleocytosis, brain imaging, and vascular studies
Table 1 lists the clinical, CSF, and imaging features and virologic findings. The age range was from 1 to 88 years. Fifteen patients (50%) were male and 15 were female (50%). Two patients had AIDS and 3 were HIV+; relevant underlying disorders in 6 other patients included leukemia (2), the CREST syndrome (1), lymphoma (1), low CD4 count (1), and immunosuppressive treatment for rheumatoid arthritis and systemic lupus erythematosus (1).
Table 1.
Clinical features, CSF pleocytosis, imaging abnormalities, artery involvement, and virologic findings in patients with varicella zoster virus (VZV) vasculopathy
| Case | Age, y | Sex | Relevant underlying disorders | Rash | CSF pleocytosis | MRI/CT focal lesions | Focal vascular abnormalities* | Artery involvement | CSF VZV | Reduced serum/CSF ratio of VZV IgG | Reference | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DNA | IgG | |||||||||||
| 1 | 61 | M | None | + | + | + | + | Mixed | − | + | + | 5 |
| 2 | 18 | F | None | − | + | + | ND | Small | − | + | + | 6 |
| 3 | 54 | M | AIDS | − | + | + | ND | Small | + | + | + | 6 |
| 4 | 34 | M | AIDS | + | + | + | ND | Small | − | + | + | 6 |
| 5 | 42 | M | None | − | + | + | + | Mixed | − | + | + | 7 |
| 6 | 2 | M | None | + | + | + | + | Mixed | − | + | + | 8 |
| 7 | 7 | F | None | + | + | + | + | Mixed | + | − | ND | 9 |
| 8 | 2 | M | None | + | − | + | ND | Mixed | + | − | ND | 10 |
| 9 | 4 | F | None | + | + | + | ND | Small | − | + | + | 11 |
| 10 | 1 | F | None | − | + | + | − | Mixed | − | + | + | 11 |
| 11 | 28 | M | HIV+ | − | + | + | − | Mixed | + | + | + | 12 |
| 12 | 4 | M | None | + | + | + | + | Mixed | − | + | + | 13 |
| 13 | 71 | M | Leukemia | + | − | + | + | Large | − | + | + | 14 |
| 14 | 76 | F | None | + | − | − | − | Small | − | + | + | 14 |
| 15 | 51 | F | CREST syndrome | − | − | + | + | Mixed | − | + | + | 15 |
| 16 | 36 | M | HIV+ | + | + | + | + | Mixed | + | + | + | 16 |
| 17 | 56 | F | None | − | − | + | + | Large | − | + | + | 1 |
| 18 | 77 | F | None | + | − | + | + | Mixed | − | + | + | 1 |
| 19 | 52 | F | None | + | + | + | + | Small | − | + | + | 1 |
| 20 | 52 | M | HIV+ | + | + | + | − | Small | + | + | + | 1 |
| 21 | 76 | F | Lymphoma | − | + | + | − | Small | − | + | + | 1 |
| 22 | 58 | M | None | + | − | + | + | Large | − | + | + | 1 |
| 23 | 88 | M | None | + | + | + | ND | Large | − | + | + | 1 |
| 24 | 54 | F | None | + | − | + | − | Small | − | + | + | Unpublished |
| 25 | 20 | F | None | + | − | + | + | Mixed | + | + | + | Unpublished |
| 26 | 63 | F | None | − | + | + | ND | Small | − | + | + | Unpublished |
| 27 | 25 | F | None | − | − | + | + | Mixed | − | + | + | Unpublished |
| 28 | 73 | M | CLL | + | + | + | + | Mixed | + | + | + | Unpublished |
| 29 | 78 | M | Low C4 | − | + | + | + | Mixed | + | + | + | Unpublished |
| 30 | 63 | F | RA, SLE† | + | + | + | − | Small | − | + | + | Unpublished |
Determined by four-vessel angiography or MRA.
Patient on chronic prednisone and hydroxyurea.
ND = not done; CREST = calcinosis, Raynaud phenomenon, esophageal dysmotility, sclerodactyly, and telangiectasia; CLL = chronic lymphocytic leukemia; RA = rheumatoid arthritis; SLE = systemic lupus erythematosus.
Of the 30 patients, 19 (63%) had rash, 20 (67%) had a CSF pleocytosis (>5 WBCs), and 29 (97%) had abnormal imaging studies on brain MRI or CT scanning. Lesions in white matter were more frequent than in gray matter, and gray-white matter junction lesions were prominent on imaging (figure 1B) and as seen macroscopically (figure 1D). Of 23 patients who underwent vascular studies (conventional angiography or MRA), 16 (70%) showed vascular abnormalities. Brain imaging and vascular studies (table 1) revealed mixed large and small artery involvement in 15 (50%), pure small artery involvement in 11 (37%), and pure large artery disease in 4 (13%) patients (table 1).
Immunocompetent vs immunocompromised patients
Of the 11 immunocompromised patients, 6 (54%) had rash compared to 13 of 19 (68%) immunocompetent patients. In the 11 immunocompromised patients, 1 had large artery disease, 5 had small artery disease, and 5 had mixed artery disease; in the 19 immunocompetent patients, 3 had larger artery disease, 6 had small artery disease, and 10 had mixed artery disease. In the 11 immunocompromised patients, 9 (82%) had a CSF pleocytosis compared to 11 of 19 (58%) immunocompetent patients. In the 11 immunocompromised patients, 6 (54%) had VZV DNA in their CSF compared to 3 of 19 (16%) immunocompetent patients. In the 11 immunocompromised patients, all 11 (100%) had anti-VZV IgG antibody in their CSF compared to 17 of 19 (89%) of the immunocompetent patients.
Statistical analysis did not reveal a significant difference in the presence of rash, CSF pleocytosis, and anti-VZV IgG antibody in immunocompromised vs immunocompetent patients. The presence of VZV DNA in CSF of immunocompromised patients was significantly greater than in immunocompetent patients (p = 0.0419, Fisher exact test).
Virologic studies
Of the 30 patients, 9 (30%) had VZV DNA in CSF, while 28 (93%) had CSF anti-VZV IgG antibody; all of the latter patients had a reduced serum/CSF ratio of VZV IgG. The two patients who had no detectable anti-VZV IgG antibody in their CSF were children who developed vasculopathy after chickenpox. Based on the sample size (n = 30), the sensitivity of PCR as a test for VZV vasculopathy was 30% (9/30) with 95% confidence limits (14.73 to 49.40) for binomial proportions, and the sensitivity of anti-VZV IgG antibody in the CSF as a test for VZV vasculopathy was 93.33% (28/30) with 95% confidence limits of 77.93 to 99.18 for binomial proportions. As a test for VZV vasculopathy, the sensitivity of detection of VZV IgG was greater than that of VZV DNA detection by PCR (p < 0.001 using the Mc-Nemar test for correlated proportions). In the 19 patients with rash, the interval from rash to the onset of neurologic symptoms and signs ranged from the same day to 2.5 years (average 4.1 months) (table 2). In all 30 patients with virologically verified VZV vasculopathy, the interval from neurologic symptoms and signs to virologic analysis for the presence of VZV DNA or anti-VZV IgG antibody in CSF ranged from 1 day to 2 years (average 4.2 months) (table 2).
Table 2.
Time from rash to neurologic symptoms/signs and from neurologic symptoms/signs to virologic analysis in varicella zoster virus (VZV) vasculopathy patients
| Case* | Rash to neurologic symptoms and signs† | Neurologic symptoms and signs to virologic analysis‡ |
|---|---|---|
| 1 | 3 wk | 1–2 d |
| 2 | NA | 1–2 d |
| 3 | NA | 3 d |
| 4 | 6 mo | 3 wk |
| 5 | NA | 10–14 d |
| 6 | 3 mo | 8 mo |
| 7 | 6 d | 4 d |
| 8 | 20 mo | 6–8 d |
| 9 | 1 wk | 1–2 d |
| 10 | NA | 3 mo |
| 11 | NA | 3 mo |
| 12 | 3 mo | 3 mo |
| 13 | 1 mo | 6 mo |
| 14 | 6 mo | 2 d |
| 15 | NA | 2 mo |
| 16 | 7 d | 14 d |
| 17 | NA | 2 y |
| 18 | 10 d | 3 d |
| 19 | Concurrent | 16 mo |
| 20 | 1 mo | 6 d |
| 21 | NA | 1 mo |
| 22 | 2.5 y | 3 wk |
| 23 | 4 wk | 1 d |
| 24 | 2 wk | 6 mo |
| 25 | 3 mo | 3 wk |
| 26 | NA | 7 mo |
| 27 | NA | 5 wk |
| 28 | 7 wk | 1 wk |
| 29 | NA | 17 d |
| 30 | 2 wk | 1 d |
Cases correspond to those in table 1 with respect to age, sex, relevant underlying disorders, rash status, CSF VZV DNA and VZV IgG findings and references.
Average 4.1 months.
Average 4.2 months.
NA = not applicable.
Treatment and outcome
Drug treatment, dose, duration of treatment, outcomes, and other medical conditions that might have affected outcome varied among patients (table e-1 on the Neurology® Web site at www.neurology.org). In many instances, the specific antiviral agent or steroid, and the dose and length of treatment were unknown. Improvement was defined as partial to complete resolution of the neurologic deficits. Fifteen patients (13 adults and 2 children) were treated with IV acyclovir for 10 to 28 days without steroids (Cases 3, 4, 8, 9, 11, 13, 14, 16, 18, 19, 20, 23, 24, 26, and 30). Of these 15, 9 (60%) improved immediately (Cases 8, 11, 13, 14, 18, 19, 20, 23, and 24), and 1 (7%) stabilized (Case 4), 2 (13%) worsened and died (Cases 3 and 26); 3 others (20%) worsened until steroids were added to the acyclovir, which then was followed by improvement (Cases 9, 16, and 30). In Patient 14, symptoms recurred after acyclovir was discontinued, but resolved after a second course of IV acyclovir.
Twelve patients were treated with both IV acyclovir and steroids (prednisone or prednisolone) (Cases 1, 2, 5, 7, 10, 15, 17, 21, 22, 27, 28, and 29). Of these 12, 8 (67%) improved (Cases 2, 5, 7, 10, 17, 22, 27, and 28), 1 (8%) remained stable (Case 15), and 3 (25%) worsened (Cases 1, 21, and 29); 1 (8%) improved initially (Case 10), but symptoms recurred 2 days after treatment was discontinued, although subsequent treatment with prednisolone alone resulted in slow improvement. Patient 28 improved clinically even though new deep-seated infarcts developed during treatment with IV acyclovir and steroids.
Two children (Cases 6 and 12) did not receive IV acyclovir or steroids; one stabilized and the other improved slowly. One adult patient (Case 25) was treated for 7 days with oral valacyclovir and acetylsalicylic acid, and experienced recurrent TIAs for the next 3 months.
Irrespective of type of treatment, all 4 patients (100%) with exclusive large artery disease improved. Of 11 patients with exclusive small artery disease, 7 (64%) improved, 1 (9%) stabilized, 1 (9%) progressed, and 2 (18%) died. Of 15 patients with both large and small artery disease, 10 (67%) improved, 2 (13%) stabilized, and 3 (20%) progressed.
DISCUSSION
A number of important findings emerged from our study of 30 patients with virologically verified VZV vasculopathy. First, nearly 40% of the patients had no history of zoster or varicella rash. Thus, VZV must be considered as a possible cause of TIA or stroke in adults or children even without a history of rash. Second, when rash occurred, there was often a long delay from the onset of rash to the occurrence of neurologic symptoms and signs (average interval 4.1 months). Given this temporal dissociation, even a history of zoster may not be recognized as significant because TIAs and stroke in elderly individuals are more commonly caused by arteriosclerotic disease. Third, there was no CSF pleocytosis in 33% of the VZV vasculopathy patients. Normally, the absence of a CSF pleocytosis would argue against an infectious cause of a vasculopathy of unknown etiology, and further virologic studies would not be undertaken. Clearly, the absence of a CSF pleocytosis does not rule out the diagnosis of VZV vasculopathy. Fourth, only 16 (70%) of 23 patients who had vascular studies (MRA or conventional four-vessel angiography) showed evidence of focal narrowing or stenosis in cerebral arteries, most likely because disease in small arteries is not as readily detected as in large arteries. Thus, although the presence of stenosis or occlusion is helpful in diagnosing VZV vasculopathy, a negative angiogram does not exclude this diagnosis. In contrast to the variable presence of rash, CSF pleocytosis, and vascular abnormalities, the most consistent conventional criterion supporting the clinical diagnosis of VZV vasculopathy is evidence of ischemic lesions on MRI or CT in the setting of a CSF positive VZV PCR or VZV IgG. Of our 30 patients, 29 (97%) had abnormal MRI or CT findings; the only patient with a normal MRI had small vessel disease restricted to the posterior ciliary artery.14 Thus, normal brain imaging studies argue strongly against a diagnosis of VZV vasculopathy.
Furthermore, in contrast to the longstanding notion that VZV vasculopathy predominantly or exclusively affects large cerebral arteries, combined brain imaging and vascular studies showed that involvement of both large and small arteries in the same patient was most common (50%), followed by exclusive small artery involvement (37%). Large artery involvement occurred in only 13% of our patients. In addition, among immunocompromised patients, the entire spectrum of large, small, and mixed artery involvement was seen compared to a predominance of exclusively small artery involvement as previously described.17 Although all four patients with exclusive large artery disease improved, no definitive correlation can be made between arterial involvement and the outcome after various treatments.
A comparison of clinical and virologic features in immunocompromised and immunocompetent patients did not reveal any statistically significant difference between the presence of rash, CSF pleocytosis, or presence of anti-VZV IgG antibody in CSF in the two groups. However, the presence of VZV DNA in 54% of immunocompromised patients compared to 16% of immunocompetent patients was statistically significant.
When a clinical diagnosis of VZV vasculopathy is suspected and supported by single or multiple characteristic lesions on MRI or CT, virologic confirmation is required. We recently found that in 14 patients, the diagnostic value of detecting anti-VZV IgG antibody in CSF was greater than that of detecting VZV DNA.1 In the present study with 16 additional patients from all our institutions, 93% of the 30 patients with VZV vasculopathy had anti-VZV IgG in the CSF compared to only 30% with VZV DNA in CSF. This finding is consistent with our earlier work showing that detection of VZV antibody in CSF is a more sensitive test (p < 0.001) than detection of VZV DNA in CSF.1 Although a positive PCR for VZV DNA in CSF is helpful, a negative PCR does not exclude the diagnosis; only negative results in both VZV PCR and anti-VZV IgG antibody tests in CSF can reliably exclude the diagnosis of VZV vasculopathy.
The superiority of VZV antibody detection compared to VZV DNA might rest in the often protracted clinical course of VZV vasculopathy, usually lasting for weeks to months. This longer course contrasts with the acute encephalitis caused by HSV-1, in which the CSF is positive for HSV-1 DNA by PCR and negative for antibody to HSV-1 during the first week of disease, while viral DNA begins to disappear from the CSF as anti-HSV-1 antibody becomes detectable during the second week.18 In one of our patients, serial CSF analysis for VZV DNA revealed its presence on the day of admission and 7 days later; VZV DNA became undetectable on days 14 to 50 when anti-VZV IgG antibody was found (table 1, Case 16). In our 30 patients, the average time from onset of neurologic symptoms to virologic analysis was 4.2 months; thus, it is not surprising that most patients did not have VZV DNA in their CSF but did have anti-VZV IgG antibody. Nevertheless, compared to the common clinical practice of testing for VZV DNA in CSF only, testing for anti-VZV IgG antibody identifies more cases of VZV vasculopathy. Finally, it is important to recognize that whereas in HSV encephalitis, the diagnostic value of PCR was shown by its comparison to HSV-positive brain biopsies, which has served as the gold standard for diagnosis, a gold standard which demonstrates VZV in the arterial wall of patients with VZV vasculopathy is not readily established.
VZV vasculopathy is caused by a productive viral infection in arteries, as evidenced by the presence of multinucleated giant cells (figure 2A), Cowdry A inclusion bodies (figure 2B), and herpes virus particles (figure 2C). Postmortem virologic analysis reveals VZV antigen (figure 2D) in cerebral vessels.19 Thus, all patients are typically treated with IV acyclovir based on Category 3 evidence (opinions of respected authorities based on clinical experience, descriptive studies, or reports of expert committees). Herein, of all patients treated with acyclovir alone, a total of 66% improved or stabilized compared to 75% who improved or stabilized when treated with both acyclovir and steroids. Because our patients received different treatment regimens at different institutions in an uncontrolled setting, the determination of optimal dose, duration of antiviral treatment, and benefit of concurrent steroid therapy awaits studies with larger case numbers.
Figure 2. Artery of a patient who died of varicella zoster virus (VZV) vasculopathy and immunohistochemical analysis of cerebral artery from a patient with VZV vasculopathy.
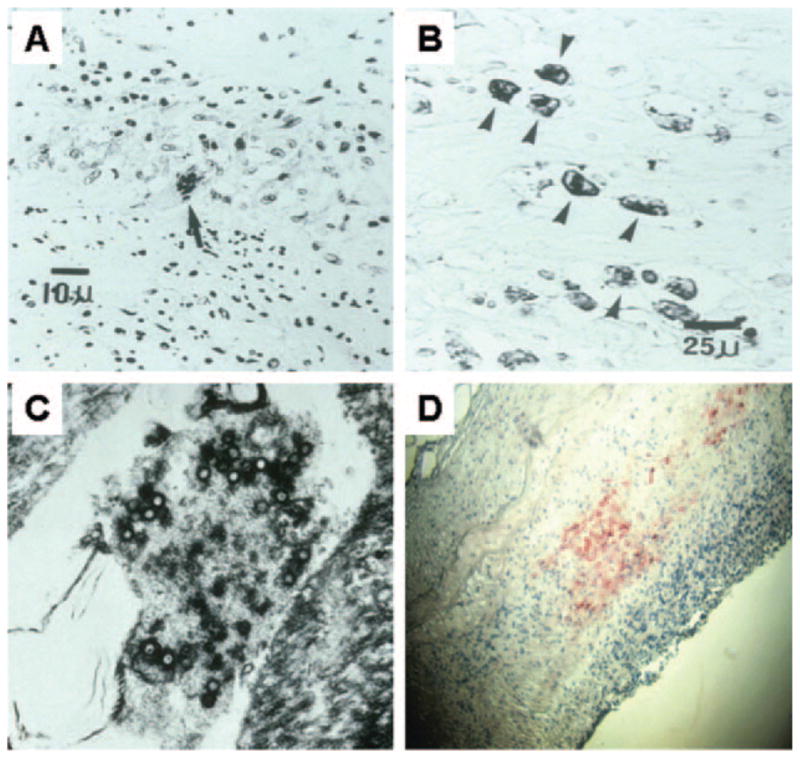
(A) Artery of a patient who died of VZV vasculopathy reveals a multinucleated giant cell (arrow) with abundant histiocytes and lymphocytes. (B) Same artery as in A; avidin-biotin-peroxidase staining shows VZV-specific antigen within Cowdry A inclusion bodies in the cytoplasm of histiocytes (arrows). (C) Same artery as in A; electron microscopy shows herpes virus particles. (D) Immunohistochemical analysis of cerebral artery from a patient with VZV vasculopathy; VZV antigen (red staining) was detected using rabbit antiserum directed against the VZV open reading frame 63 protein (magnification ×86). (A, B) Reprinted from Fukumoto S, Kinjo M, Hokamura K, Tanaka K. Subarachnoid hemorrhage and granulomatous angiitis of the basilar artery: demonstration of the varicella-zoster virus in the basilar artery lesions. Stroke 1986;17:1024 –1028. Reprinted with permission from Lippincott Williams & Wilkins. (C) Reprinted from Gilden DH. Shingles and postherpetic neuralgia: an unmet need? Consultant 2005;45:S13–19. Reprinted with permission from Cliggott Publishing, CMP Healthcare Media LLC. (D) Reprinted from Gilden DH, Mahalingam R, Cohrs RJ, Kleinschmidt-DeMasters BK, Forghani B. The protean manifestations of varicella-zoster virus vasculopathy. J Neurovirol 2002;8:75–79. Reprinted with permission from Journal of Neurovirology.
Acknowledgments
Supported by grants AG06127, NS32623, and NS07321 to Dr. Gilden from the NIH.
The authors thank Dr. Gary Zerbe for statistical analysis, Marina Hoffman for editorial review, and Cathy Allen for assistance in manuscript preparation.
GLOSSARY
- EIA
enzyme immunoabsorbent assay
- VZV
varicella zoster virus
Footnotes
Disclosure: The authors report no conflicts of interest.
References
- 1.Nagel MA, Forghani B, Mahalingam R, et al. The value of detecting anti-VZV IgG antibody in CSF to diagnose VZV vasculopathy. Neurology. 2007;68:1069–1073. doi: 10.1212/01.wnl.0000258549.13334.16. [DOI] [PubMed] [Google Scholar]
- 2.Hilt DC, Buchholz D, Krumholz A, Weiss H, Wolinsky J. Herpes zoster ophthalmicus and delayed contralateral hemiparesis caused by cerebral angiitis: diagnosis and management approaches. Ann Neurol. 1983;14:543–553. doi: 10.1002/ana.410140509. [DOI] [PubMed] [Google Scholar]
- 3.Gilden DH, Gesser R, Smith J, et al. Presence of VZV and HSV-1 DNA in human nodose and celiac ganglia. Virus Genes. 2001;23:145–147. doi: 10.1023/a:1011883919058. [DOI] [PubMed] [Google Scholar]
- 4.Reiber H, Lange P. Quantification of virus-specific antibodies in cerebrospinal fluid and serum: sensitive and specific detection of antibody synthesis in brain. Clin Chem. 1991;7:1153–1160. [PubMed] [Google Scholar]
- 5.Terborg C, Busse O. Granulomatous vasculitis of the CNS as a complication of herpes zoster ophthalmicus. Fortschr Neurol Psychiatr. 1995;63:383–387. doi: 10.1055/s-2007-1002284. [DOI] [PubMed] [Google Scholar]
- 6.Gilden DH, Bennett JL, Kleinschmidt-DeMasters BK, Song DD, Yee AS, Steiner I. The value of cerebrospinal fluid antiviral antibody in the diagnosis of neurologic disease produced by varicella zoster virus. J Neurol Sci. 1998;159:140–144. doi: 10.1016/s0022-510x(98)00153-1. [DOI] [PubMed] [Google Scholar]
- 7.Nau R, Lantsch M, Stiefel M, Polak T, Reiber H. Varicella zoster virus-associated focal vasculitis without herpes zoster: Recovery after treatment with acyclovir. Neurology. 1998;51:914–915. doi: 10.1212/wnl.51.3.914. [DOI] [PubMed] [Google Scholar]
- 8.Hattori H, Higuchi Y, Tsuji M. Recurrent strokes after varicella. Ann Neurol. 2000;47:136. [PubMed] [Google Scholar]
- 9.Caruso JM, Tung GA, Brown WD. Central nervous system and renal vasculitis associated with primary varicella infection in a child. Pediatrics. 2001;107:9–12. doi: 10.1542/peds.107.1.e9. [DOI] [PubMed] [Google Scholar]
- 10.Chiappini E, Calabri G, Galli L, Salvi G, de Martino M. Varicella-zoster virus acquired at 4 months of age reactivates at 24 months and causes encephalitis. J Pediatr. 2002;140:250–251. doi: 10.1067/mpd.2002.121934. [DOI] [PubMed] [Google Scholar]
- 11.Hausler M, Schaade L, Kemmeny S, Schweizer K, Schoenmackers C, Ramaekers VT. Encephalitis related to primary varicella-zoster virus infection in immunocompetent children. J Neurol Sci. 2002;195:111–116. doi: 10.1016/s0022-510x(02)00017-5. [DOI] [PubMed] [Google Scholar]
- 12.Kronenberg A, Schupbach R, Schuknecht B, et al. Multifocal vasculopathy due to varicella-zoster virus (VZV): serial analysis of VZV DNA and intrathecal synthesis of VZV antibody in cerebrospinal fluid. Clin Infect Dis. 2002;35:330–333. doi: 10.1086/341492. [DOI] [PubMed] [Google Scholar]
- 13.Ueno M, Oka A, Koeda T, Okamoto R, Takeshita K. Unilateral occlusion of the middle cerebral artery after varicella-zoster virus infection. Brain Dev. 2002;24:106–108. doi: 10.1016/s0387-7604(02)00005-0. [DOI] [PubMed] [Google Scholar]
- 14.Gilden DH, Lipton HL, Wolf JS, et al. Two patients with unusual forms of varicella-zoster virus vasculopathy. N Engl J Med. 2002;347:1500–1503. doi: 10.1056/NEJMoa020841. [DOI] [PubMed] [Google Scholar]
- 15.Russman AN, Lederman RJ, Calabrese LH, Embi PJ, Forghani B, Gilden DH. Multifocal varicella-zoster virus vasculopathy without rash. Arch Neurol. 2003;60:1607–1609. doi: 10.1001/archneur.60.11.1607. [DOI] [PubMed] [Google Scholar]
- 16.Saraya T, Shimura C, Wada H, Aoshima M, Goto H. Evidence for vascular spread of varicella zoster-associated vasculopathy. Ann Intern Med. 2006;144:535–537. doi: 10.7326/0003-4819-144-7-200604040-00022. [DOI] [PubMed] [Google Scholar]
- 17.Kleinschmidt-DeMasters BK, Gilden DH. Varicella-zoster virus infections of the nervous system: clinical and pathologic correlates. Arch Pathol Lab Med. 2001;125:770–780. doi: 10.5858/2001-125-0770-VZVIOT. [DOI] [PubMed] [Google Scholar]
- 18.Aurelius E, Johansson B, Skoldenberg B, Staland A, Forsgren M. Rapid diagnosis of herpes simplex encephalitis by nested polymerase chain reaction assay of cerebrospinal fluid. Lancet. 1991;337:189–192. doi: 10.1016/0140-6736(91)92155-u. [DOI] [PubMed] [Google Scholar]
- 19.Gilden DH, Kleinschmidt-DeMasters BK, Wellish M, Hedley-Whyte ET, Rentier B, Mahalingam R. Varicella zoster virus, a cause of waxing and waning vasculitis: the N Engl J Med case 5-1995 revisited. Neurology. 1996;47:1441–1446. doi: 10.1212/wnl.47.6.1441. [DOI] [PubMed] [Google Scholar]


