Abstract
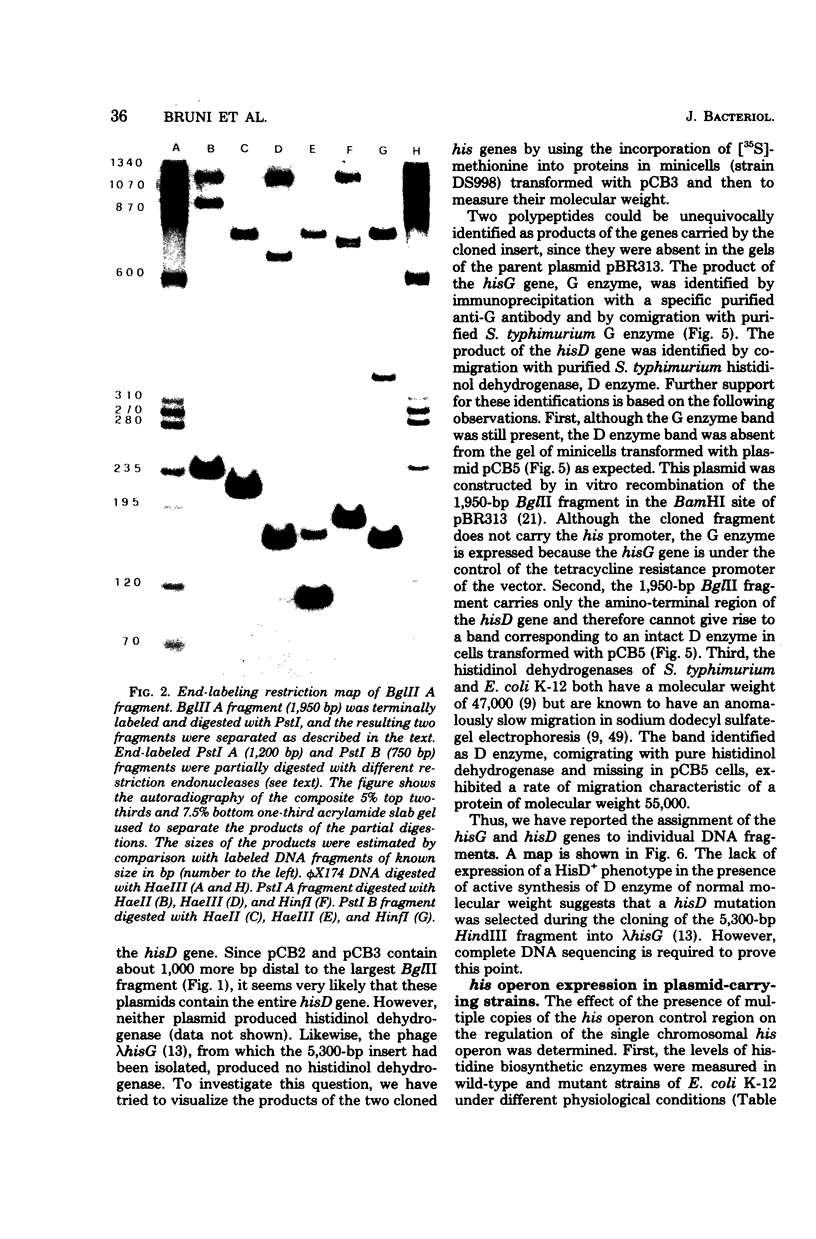
A fragment of deoxyribonucleic acid 5,300 base paris long and containing the promoter-proximal portion of the histidine operon of Escherichia coli K-12, has been cloned in plasmid pBR313 (plasmids pCB2 and pCB3). Restriction mapping, partial nucleotide sequencing, and studies on functional expression in vivo and on protein synthesis in minicells have shown that the fragment contains the regulatory region of the operon, the hisG, hisD genes, and part of the hisC gene. Another plasmid (pCB5) contained the hisG gene and part of the hisD gene. Expression of the hisG gene in the latter plasmid was under control of the tetracycline promoter of the pBR313 plasmid. The in vivo expression of the two groups of plasmids described above, as well as their effect on the expression of the histidine genes not carried by the plasmids but present on the host chromosome, has been studied. The presence of multiple copies of pCB2 or pCB3, but not of pCB5, prevented derepression of the chromosomal histidine operon. Possible interpretations of this phenomenon are discussed.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Artz S. W., Broach J. R. Histidine regulation in Salmonella typhimurium: an activator attenuator model of gene regulation. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3453–3457. doi: 10.1073/pnas.72.9.3453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atkins J. F., Loper J. C. Transcription initiation in the histidine operon of Salmonella typhimurium. Proc Natl Acad Sci U S A. 1970 Apr;65(4):925–932. doi: 10.1073/pnas.65.4.925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avitabile A., Bruni C. B., Carlomagno-Cerillo M. S., Meyers M., Vigliar G., Blasi F. Deletion mapping and orientation of the histidine operon of Escherichia coli on a transducing bacteriophage. J Bacteriol. 1973 Nov;116(2):656–662. doi: 10.1128/jb.116.2.656-662.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avitabile A., Bruni C. B., Covelli A., Di Nocera P. P., Sbordone L., Blasi F. In vitro transcription of the Escherichia coli histidine operon. Mol Gen Genet. 1974;132(1):1–12. doi: 10.1007/BF00268225. [DOI] [PubMed] [Google Scholar]
- Avitabile A., Carlomagno-Cerillo S., Favvre R., Blasi F. Isolation of transducing bacteriophages for the histidine and isoleucine-valine operons in Escherichia coli K-12. J Bacteriol. 1972 Oct;112(1):40–47. doi: 10.1128/jb.112.1.40-47.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachmann B. J., Low K. B., Taylor A. L. Recalibrated linkage map of Escherichia coli K-12. Bacteriol Rev. 1976 Mar;40(1):116–167. doi: 10.1128/br.40.1.116-167.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes W. M. DNA sequence from the histidine operon control region: seven histidine codons in a row. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4281–4285. doi: 10.1073/pnas.75.9.4281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bickle T. A., Pirrotta V., Imber R. A simple, general procedure for purifying restriction endonucleases. Nucleic Acids Res. 1977 Aug;4(8):2561–2572. doi: 10.1093/nar/4.8.2561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bitar K. G., Firca J. R., Loper J. C. Histidinol dehydrogenase from salmonella typhimurium and Escherichia coli. Purification, some characteristics and the amino acid sequence around a reactive thiol group. Biochim Biophys Acta. 1977 Aug 23;493(2):429–440. doi: 10.1016/0005-2795(77)90199-4. [DOI] [PubMed] [Google Scholar]
- Blasi F., Bruni C. B., Avitabile A., Deeley R. G., Goldberger R. F., Meyers M. M. Inhibition of transcription of the histidine operon in vitro by the first enzyme of the histidine pathway. Proc Natl Acad Sci U S A. 1973 Sep;70(9):2692–2696. doi: 10.1073/pnas.70.9.2692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolivar F., Rodriguez R. L., Betlach M. C., Boyer H. W. Construction and characterization of new cloning vehicles. I. Ampicillin-resistant derivatives of the plasmid pMB9. Gene. 1977;2(2):75–93. doi: 10.1016/0378-1119(77)90074-9. [DOI] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Borck K., Beggs J. D., Brammar W. J., Hopkins A. S., Murray N. E. The construction in vitro of transducing derivatives of phage lambda. Mol Gen Genet. 1976 Jul 23;146(2):199–207. doi: 10.1007/BF00268089. [DOI] [PubMed] [Google Scholar]
- Brenner M., De Lorenzo F., Ames B. N. Energy charge and protein synthesis. Control of aminoacyl transfer ribonucleic acid synthetases. J Biol Chem. 1970 Jan 25;245(2):450–452. [PubMed] [Google Scholar]
- Bruni C. B., Colantuoni V., Sbordone L., Cortese R., Blasi F. Biochemical and regulatory properties of Escherichia coli K-12 hisT mutants. J Bacteriol. 1977 Apr;130(1):4–10. doi: 10.1128/jb.130.1.4-10.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chessin H., Summers W. C. Initiation by RNA polymerase on UV or x-ray damaged T7 DNA. Biochem Biophys Res Commun. 1970 Jan 6;38(1):40–45. doi: 10.1016/0006-291x(70)91080-6. [DOI] [PubMed] [Google Scholar]
- Clewell D. B. Nature of Col E 1 plasmid replication in Escherichia coli in the presence of the chloramphenicol. J Bacteriol. 1972 May;110(2):667–676. doi: 10.1128/jb.110.2.667-676.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Nocera P. P., Avitabile A., Blasi F. In vitro transcription of the Escherichia coli histidine operon primed by dinucleotides. Effect of the first histidine biosynthetic enzyme. J Biol Chem. 1975 Nov 10;250(21):8376–8381. [PubMed] [Google Scholar]
- Di Nocera P. P., Blasi F., Di Lauro R., Frunzio R., Bruni C. B. Nucleotide sequence of the attenuator region of the histidine operon of Escherichia coli K-12. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4276–4280. doi: 10.1073/pnas.75.9.4276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ely B. Physiological studies of salmonella histidine operator-promoter mutants. Genetics. 1974 Oct;78(2):593–606. doi: 10.1093/genetics/78.2.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernández V. M., Martíndelrío R., Tébar A. R., Guisán J. M., Ballesteros A. O. Derepression and repression of the histidine operon: role of the feedback site of the first enzyme. J Bacteriol. 1975 Dec;124(3):1366–1373. doi: 10.1128/jb.124.3.1366-1373.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiil N., Friesen J. D. Isolation of "relaxed" mutants of Escherichia coli. J Bacteriol. 1968 Feb;95(2):729–731. doi: 10.1128/jb.95.2.729-731.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner J. F. Regulation of the threonine operon: tandem threonine and isoleucine codons in the control region and translational control of transcription termination. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1706–1710. doi: 10.1073/pnas.76.4.1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrick-Silversmith L., Hartman P. E. Histidine-requiring mutants of Escherichia coli K12. Genetics. 1970 Oct;66(2):231–244. doi: 10.1093/genetics/66.2.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gemmill R. M., Wessler S. R., Keller E. B., Calvo J. M. leu operon of Salmonella typhimurium is controlled by an attenuation mechanism. Proc Natl Acad Sci U S A. 1979 Oct;76(10):4941–4945. doi: 10.1073/pnas.76.10.4941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberger R. F., Kovach J. S. Regulation of histidine biosynthesis in Salmonella typhimurium. Curr Top Cell Regul. 1972;5:285–308. doi: 10.1016/b978-0-12-152805-8.50014-9. [DOI] [PubMed] [Google Scholar]
- Goldschmidt E. P., Cater M. S., Matney T. S., Butler M. A., Greene A. Genetic analysis of the histidine operon in Escherichia coli K12. Genetics. 1970 Oct;66(2):219–229. doi: 10.1093/genetics/66.2.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallewell R. A., Sherratt D. J. Isolation and characterization of Co1E2 plasmid mutants unable to kill colicin-sensitive cells. Mol Gen Genet. 1976 Aug 2;146(3):239–245. doi: 10.1007/BF00701246. [DOI] [PubMed] [Google Scholar]
- Hardies S. C., Wells R. D. Preparative fractionation of DNA restriction fragments by reversed phase column chromatography. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3117–3121. doi: 10.1073/pnas.73.9.3117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hershfield V., Boyer H. W., Yanofsky C., Lovett M. A., Helinski D. R. Plasmid ColEl as a molecular vehicle for cloning and amplification of DNA. Proc Natl Acad Sci U S A. 1974 Sep;71(9):3455–3459. doi: 10.1073/pnas.71.9.3455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston H. M., Barnes W. M., Chumley F. G., Bossi L., Roth J. R. Model for regulation of the histidine operon of Salmonella. Proc Natl Acad Sci U S A. 1980 Jan;77(1):508–512. doi: 10.1073/pnas.77.1.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasai T. Regulation of the expression of the histidine operon in Salmonella typhimurium. Nature. 1974 Jun 7;249(457):523–527. doi: 10.1038/249523a0. [DOI] [PubMed] [Google Scholar]
- Keller E. B., Calvo J. M. Alternative secondary structures of leader RNAs and the regulation of the trp, phe, his, thr, and leu operons. Proc Natl Acad Sci U S A. 1979 Dec;76(12):6186–6190. doi: 10.1073/pnas.76.12.6186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klungsoyr L., Atkinson D. E. Regulatory properties of phosphoribosyladenosine triphosphate synthetase. Synergism between adenosine monophosphate, phosphoribosyladenosine triphosphate, and histidine. Biochemistry. 1970 Apr 28;9(9):2021–2027. doi: 10.1021/bi00811a025. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lawther R. P., Hatfield G. W. Effects of altered rho gene product on the expression of the Escherichia coli histidine operon. J Bacteriol. 1978 Dec;136(3):1201–1204. doi: 10.1128/jb.136.3.1201-1204.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee F., Bertrand K., Bennett G., Yanofsky C. Comparison of the nucleotide sequences of the initial transcribed regions of the tryptophan operons of Escherichia coli and Salmonella typhimurium. J Mol Biol. 1978 May 15;121(2):193–217. doi: 10.1016/s0022-2836(78)80005-9. [DOI] [PubMed] [Google Scholar]
- Lewis J. A., Ames B. N. Histidine regulation in Salmonella typhimurium. XI. The percentage of transfer RNA His charged in vivo and its relation to the repression of the histidine operon. J Mol Biol. 1972 Apr 28;66(1):131–142. doi: 10.1016/s0022-2836(72)80011-1. [DOI] [PubMed] [Google Scholar]
- Maniatis T., Jeffrey A., Kleid D. G. Nucleotide sequence of the rightward operator of phage lambda. Proc Natl Acad Sci U S A. 1975 Mar;72(3):1184–1188. doi: 10.1073/pnas.72.3.1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. A new method for sequencing DNA. Proc Natl Acad Sci U S A. 1977 Feb;74(2):560–564. doi: 10.1073/pnas.74.2.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oxender D. L., Zurawski G., Yanofsky C. Attenuation in the Escherichia coli tryptophan operon: role of RNA secondary structure involving the tryptophan codon region. Proc Natl Acad Sci U S A. 1979 Nov;76(11):5524–5528. doi: 10.1073/pnas.76.11.5524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons S. M., Koshland D. E., Jr A rapid isolation of phosphoribosyladenosine triphosphate synthetase and comparison to native enzyme. J Biol Chem. 1974 Jul 10;249(13):4104–4109. [PubMed] [Google Scholar]
- Piszkiewicz D., Tilley B. E., Rand-Meir T., Parsons S. M. Amino acid sequence of ATP phosphoribosyltransferase of Salmonella typhimurium. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1589–1592. doi: 10.1073/pnas.76.4.1589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rechler M. M., Bruni C. B. Properties of a fused protein formed by genetic manipulation. Histidinol dehydrogenase-imidazolylacetol phosphate: L-glutamate aminotransferase. J Biol Chem. 1971 Mar 25;246(6):1806–1813. [PubMed] [Google Scholar]
- Roth J. R., Antón D. N., Hartman P. E. Histidine regulatory mutants in Salmonella typhimurium. I. Isolation and general properties. J Mol Biol. 1966 Dec 28;22(2):305–323. doi: 10.1016/0022-2836(66)90134-3. [DOI] [PubMed] [Google Scholar]
- Singer C. E., Smith G. R., Cortese R., Ames B. N. [Mutant tRNA His ineffective in repression and lacking two pseudouridine modifications]. Nat New Biol. 1972 Jul 19;238(81):72–74. doi: 10.1038/newbio238072a0. [DOI] [PubMed] [Google Scholar]
- Smith H. O., Birnstiel M. L. A simple method for DNA restriction site mapping. Nucleic Acids Res. 1976 Sep;3(9):2387–2398. doi: 10.1093/nar/3.9.2387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stephens J. C., Artz S. W., Ames B. N. Guanosine 5'-diphosphate 3'-diphosphate (ppGpp): positive effector for histidine operon transcription and general signal for amino-acid deficiency. Proc Natl Acad Sci U S A. 1975 Nov;72(11):4389–4393. doi: 10.1073/pnas.72.11.4389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tait R. C., Rodriguez R. L., Boyer H. W. Altered tetracycline resistance in pSC101 recombinant plasmids. Mol Gen Genet. 1977 Mar 16;151(3):327–331. doi: 10.1007/BF00268797. [DOI] [PubMed] [Google Scholar]
- Tébar A. R., Ballesteros A. O. Kinetic properties of ATP phosphoribosyltransferase of Escherichia coli. Mol Cell Biochem. 1976 Jun 15;11(3):131–136. doi: 10.1007/BF01744993. [DOI] [PubMed] [Google Scholar]
- Tébar A. R., Fernández V. M., Martíndelrío R., Ballesteros A. O. Fluorescence studies of phosphoribosyladenosine triphosphate synthetase of Escherichia coli. FEBS Lett. 1975 Feb 1;50(2):239–242. doi: 10.1016/0014-5793(75)80497-2. [DOI] [PubMed] [Google Scholar]
- Umbarger H. E. Amino acid biosynthesis and its regulation. Annu Rev Biochem. 1978;47:532–606. doi: 10.1146/annurev.bi.47.070178.002533. [DOI] [PubMed] [Google Scholar]
- VOGEL H. J., BONNER D. M. Acetylornithinase of Escherichia coli: partial purification and some properties. J Biol Chem. 1956 Jan;218(1):97–106. [PubMed] [Google Scholar]
- Winkler M. E. Expression of the histidine operon in rho mutants of Escherichia coli. J Bacteriol. 1978 Aug;135(2):721–725. doi: 10.1128/jb.135.2.721-725.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler M. E., Roth D. J., Hartman P. E. Promoter- and attenuator-related metabolic regulation of the Salmonella typhimurium histidine operon. J Bacteriol. 1978 Feb;133(2):830–843. doi: 10.1128/jb.133.2.830-843.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zurawski G., Brown K., Killingly D., Yanofsky C. Nucleotide sequence of the leader region of the phenylalanine operon of Escherichia coli. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4271–4275. doi: 10.1073/pnas.75.9.4271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zurawski G., Elseviers D., Stauffer G. V., Yanofsky C. Translational control of transcription termination at the attenuator of the Escherichia coli tryptophan operon. Proc Natl Acad Sci U S A. 1978 Dec;75(12):5988–5992. doi: 10.1073/pnas.75.12.5988. [DOI] [PMC free article] [PubMed] [Google Scholar]