Abstract
Background
Endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) of mediastinal lymphadenopathy has been shown to be a valuable diagnostic tool in high-volume EUS centers (≥50 mediastinal EUS-FNA/endoscopist/year). Our goal was to assess the diagnostic accuracy of EUS-FNA and its impact on clinical management and costs in low-volume EUS centers (<50 mediastinal EUS-FNA/endoscopist/year).
Methods
Consecutive patients referred to two Dutch endoscopy centers in the period 2002–2008 for EUS-FNA of mediastinal lymphadenopathy were reviewed. The gold standard for a cytological diagnosis was histological confirmation or clinical follow-up of more than 6 months with repeat imaging. The impact of EUS-FNA on clinical management was subdivided into a positive impact by providing (1) adequate cytology that influenced the decision to perform surgery or (2) a diagnosis of a benign inflammatory disorder, and a negative impact which was subdivided into (1) false-negative or inconclusive cytology or (2) an adequate cytological diagnosis that did not influence patient management. Costs of an alternative diagnostic work-up without EUS-FNA, as established by an expert panel, were compared to costs of the actual work-up.
Results
In total, 213 patients (71% male, median age = 61 years, range = 23–88 years) underwent EUS-FNA. Sensitivity, specificity, and negative and positive predictive values were 89%, 100%, 80%, and 100%, respectively. EUS-FNA had a positive impact on clinical management in 84% of cases by either influencing the decision to perform surgery (49%) or excluding malignant lymphadenopathy (35%), and a negative impact in 7% of cases because of inadequate (3%) or false-negative (4%) cytology. In 9% of cases, EUS-FNA was performed without an established indication. Two nonfatal perforations occurred (0.9%). Total cost reduction was €100,593, with a mean cost reduction of €472 (SD = €607) per patient.
Conclusions
Mediastinal EUS-FNA can be performed in low-volume EUS centers without compromising diagnostic accuracy. Moreover, EUS-FNA plays an important role in the management of patients with mediastinal lymphadenopathy and reduces total diagnostic costs.
Keywords: EUS-FNA, Costs, Accuracy, Mediastinal lymphadenopathy
Mediastinal lymphadenopathy may be secondary to malignancy or inflammation. Mediastinoscopy and computed tomography (CT)-guided biopsy can be used for obtaining tissue, although both techniques have several limitations and may cause morbidity [1].
Endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) has been proven to be an accurate diagnostic tool for determining the nature of mediastinal lesions [2–4]. Its impact on patient management has been studied mostly in clinical trials with highly defined groups, i.e., in patients with lymphadenopathy of unknown origin [5–7], non-small-cell lung cancer (NSCLC) [8–16], and esophageal cancer [17]. The majority of these studies were performed in high-volume centers with experienced endoscopists performing EUS-FNA [2, 3, 8, 10–12, 14, 16, 17].
It is known that there is a learning curve for EUS(-FNA) in staging malignancies [18–21]. Moreover, it has been shown that high-volume EUS centers (≥50 EUS/endoscopist/year) perform better in staging esophageal cancer than low-volume centers (<50 EUS/endoscopist/year) [22]. However, whether this issue also applies to EUS-FNA of mediastinal lymphadenopathy is unknown. Another important yet underreported topic is the potential of EUS-FNA to reduce costs by establishing a diagnosis. Only a few studies have looked into this subject, including only patients with mediastinal nodes in NSCLC and esophageal cancer [16, 23–25].
The aim of this study was to assess the diagnostic performance of EUS-FNA in mediastinal lymphadenopathy in low-volume EUS centers and its impact on clinical management and costs.
Materials and methods
All consecutive patients who underwent EUS-FNA of enlarged mediastinal lymph nodes, detected on CT, positron emission tomography (PET) scan, or previous EUS, in the period 2002–2008 at the University Medical Center Utrecht (a tertiary referral center) and the Meander Medical Center, Amersfoort (a teaching hospital), were reviewed. Patients were subdivided into two categories according to the use of EUS-FNA: for staging a malignancy (n = 86) or diagnosis (n = 145). In the latter, patients presented with mediastinal nodes of unknown origin or with disseminated disease for which other staging procedures had already been performed but cytological confirmation of malignancy was required. In the staging group, patients already had confirmed malignant disease or were strongly suspected of having a malignancy.
Procedure
EUS-FNA was performed by four endoscopists (MS, FV, MB, and BO). Each had performed more than 25 EUS-FNA procedures before the beginning of the study. Administration of anticoagulants was discontinued 3 days before the procedure. Patients were placed in the left lateral decubitus position. Pulse rate and oxygen saturation were monitored. Patients received conscious sedation with intravenous midazolam and pethidine.
EUS was performed with linear array echoendoscopes (GF-UC140P or GF-UCT140, Olympus, Hamburg, Germany). The endoscopist selected the largest and/or the most malignant-appearing node for FNA. In some cases more than one enlarged lymph node was aspirated. FNA was performed with a 22-gauge needle (Echotip Endoscopic Ultrasound needle, Cook, Limerick, Ireland). The aspirated material was smeared directly on glass slides, air-dried, and stained with Diff-Quick stain. Slides were immediately reviewed by a pathology resident or cytotechnician for adequacy of the specimen. The procedure was repeated until adequacy was confirmed.
Accuracy
The gold standard for final diagnosis included histology or a clinical follow-up of at least 6 months, with repeat imaging to assess disease progression [2–4]. A positive cytological result was considered true positive if it was highly suggestive of malignancy in patients with compatible clinical progress of the disorder or a response to chemotherapy or radiation. In patients with a high probability of an inflammatory disorder such as sarcoidosis, detection of noncaseating granulomas was considered true positive for sarcoidosis, but in patients suspected for disseminated malignancy, detection of granulomas was considered true negative for malignancy. Inconclusive or benign cytology was considered false negative if further diagnostic workup or clinical follow-up showed signs positive for malignancy or a benign inflammatory disorder. Negative cytology was considered to be true negative when histology did not show any abnormality or when imaging studies during follow-up showed spontaneous resolution or lack of progression of mediastinal lymph nodes. Patients in whom no final diagnosis could be made were excluded from analysis.
Impact on clinical management
A prepuncture diagnosis (diagnosis most likely before performing EUS-FNA) was obtained from clinical data, including radiology reports and endoscopy reports, and compared to the final cytodiagnosis. The impact of EUS-FNA on clinical management was divided into two categories based on a classification by Chen et al. [26]: The impact was considered to be positive if it provided an adequate cytological result that (1) influenced the decision to perform surgery or start chemotherapy or radiotherapy or (2) diagnosed a benign inflammatory disorder or a reactive lymph node. The impact was considered to be negative if the cytology (1) provided a false-negative or inconclusive result and therefore no indication for further diagnostic investigation or (2) did not influence patient management despite adequate cytology.
The impact of the cytological result on patient management was independently determined for each patient by a panel that included a gastroenterologist (KT), a pulmonologist (NS), and a researcher (MH), all of whom were not involved in the EUS-FNA procedures. In inconclusive cases, consensus was achieved by a panel discussion in which a second gastroenterologist (FV) was involved.
Cost reduction analysis
To assess potential cost reduction by performing EUS-FNA in the diagnostic evaluation of mediastinal lymphadenopathy, the above-mentioned panel determined for each individual patient which diagnostic tool would have been used if EUS-FNA had not been available (theoretical diagnostic work-up). Costs of EUS-FNA, alternative investigations, hospital stay, anesthesia, and pathology processing and interpretation were obtained from a standard price list. The prices were calculated by averaging the prices of these procedures in ten Dutch medical centers. If the actual work-up required further diagnostic investigations, these extra costs were added. Total cost of a complication in the actual work-up comprised the costs of prolonged hospital admission (€239.15 per day) and additional investigations and therapy, if necessary. As it is not possible to predict the course and thereby the costs of a theoretical complication; we assumed that both actual and theoretical complications had a similar course and equal costs [16, 23]. The number of theoretical complications was calculated from published complication rates (2% for mediastinoscopy) [1]. The total cost of the actual diagnostic work-up, including complications and additional investigations, was compared with the total cost of the alternative diagnostic work-up, including theoretical complications. Cost of therapy was not included since both diagnostic strategies were assumed to have equivalent outcomes.
Statistical analysis
The cytodiagnosis was compared with the gold standards for assessment of accuracy. SPSS v15.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. Comparison between categorical parameters was performed with a χ2 test or Fischer’s exact test. For continuous parameters, the two-sided Student t test was used. A value of p < 0.05 was considered to be significant.
Results
Of the 231 patients who underwent EUS-FNA, 18 (8%) were excluded because no final diagnosis was made (Fig. 1). In total, 213 patients (152 men) with a median age of 61 years (range = 23-88) were included (Table 1). Of these, 92 (43%) patients had a history of malignancy, 51 (24%) had a strong clinical suspicion of malignancy, and 70 (33%) had no suspicion for or history of malignancy.
Fig. 1.
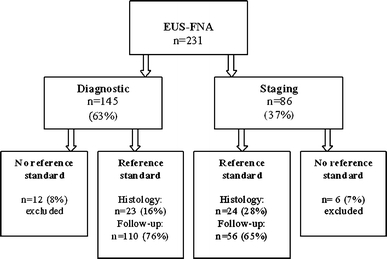
Flowchart of all EUS-FNA procedures for mediastinal lymphadenopathy and their reference standards
Table 1.
Baseline characteristics of all patients who underwent mediastinal EUS-FNA with a final diagnosis
| Overall n (%) | Diagnostic n (%) | Staging n (%) | |
|---|---|---|---|
| Patients | 213 | 133 (62) | 80 (38) |
| Median age (years) (range) | 61 (23–88) | 56 (23–86) | 66 (40–88) |
| Male (% of patients) | 152 (71) | 92 (69) | 60 (75) |
| History of malignancy (% of patients) | |||
| No | 70 (33) | 70 (53) | 0 (0) |
| Clinical suspicion but no histological proof | 51 (24) | 23 (17) | 28 (35) |
| Yes, with previous histological proof | 92 (43) | 40 (30) | 52 (65) |
| Lung cancer | 30 (33) | 3 (8) | 27 (52) |
| Esophageal cancer | 14 (15) | 0 (0) | 14 (27) |
| Non-Hodgkin’s lymphoma | 8 (9) | 8 (20) | 0 (0) |
| Breast cancer | 7 (7) | 6 (15) | 1 (2) |
| Other | 33 (36) | 23 (57) | 10 (19) |
| Previous investigation (% of patients) | |||
| CT | 189(89) | 127 (95) | 62 (78) |
| EUS | 16 (8) | 1 (0.7) | 15 (19) |
| PET | 69 (36) | 33 (25) | 36 (45) |
Accuracy
Cytology was diagnostic in 205 (96%) patients and nondiagnostic in 8 patients (4%) (Table 2). Lymph node localization and size and median number of passes to obtain cytology are listed in Table 2. Diagnostic characteristics are given in Table 3. No significant differences were found between the results for diagnostic or staging EUS-FNA with an accuracy of 92 and 94%, respectively. Table 4 presents the final diagnoses of the false-negative and inconclusive specimens.
Table 2.
Results of all EUS-FNA procedures for mediastinal lymphadenopathy with a final diagnosis
| Overall n (%) | Diagnostic n (%) | Staging n (%) | |
|---|---|---|---|
| Patients with a final diagnosis | 213 | 133 | 80 |
| Prepuncture diagnosis | 193 (91) | 115 (86) | 78 (98) |
| Confirmed | 119 (62) | 74 (64) | 45 (58) |
| Corrected | 74 (38) | 41 (36) | 33 (42) |
| Location of lymph node | |||
| Aorta-pulmonary window | 44 (21) | 23 (17) | 21 (26) |
| Subcarinal | 113 (53) | 76 (57) | 37 (47) |
| Paratracheal | 9 (4) | 5 (4) | 4(5) |
| Other | 17 (8) | 8 (6) | 9 (11) |
| >1 lymph node station | 30(14) | 21(16) | 9 (11) |
| Cytology | |||
| Malignant | 87 (41) | 44 (33) | 43 (54) |
| Benign | 118 (55) | 82 (62) | 36 (45) |
| Nondiagnostic | 8 (4) | 7 (5) | 1 (1) |
| Mean size of lymph node in mm (± SD) | 24 (±13) | 26 (±14) | 21 (±10) |
| Median number of passes (range) | 3 (1–9) | 3 (1–9) | 3 (1–6) |
| Additional investigations performed | 43 (20) | 26 (20) | 17(21) |
Table 3.
Diagnostic characteristics of EUS-FNA for mediastinal lymphadenopathy
| n | True negative | True positive | False negative | Sensitivity (%) | Specificity (%) | NPV (%) | PPV (%) | p value | |
|---|---|---|---|---|---|---|---|---|---|
| Overall | 213 | 64 | 131 | 16a | 89 | 100 | 80 | 100 | |
| Diagnostic procedure | 133 | 38 | 84 | 11 | 88 | 100 | 77 | 100 | 0.59 |
| Staging procedure | 80 | 28 | 47 | 5 | 90 | 100 | 85 | 100 | |
| Eloubeidi et al. [27] | 246 | 123 | 108 | 11 | 92 | 99 | 93 | 99 |
NPV negative predictive value, PPV positive predictive value
Comparison for diagnostic accuracy in staging and diagnostic group (χ2 test)
aFalse-negative cytology included benign (8) and nondiagnostic (8) specimens
Table 4.
Final diagnosis in patients with false-negative (benign or nondiagnostic) specimens after EUS-FNA for mediastinal lymphadenopathy
| False negative (8) | Nondiagnostic (8) |
|---|---|
| Sarcoidosis (4) | Sarcoidosis (6) |
| Tuberculosis (1) | NSCLC (1) |
| Adenocarcinoma esophagus (1) | Small cell lung cancer (1) |
| Gastric cancer (1) | |
| Chronic lymhocytic leukemia (1) |
NSCLC non-small-cell lung cancer
Impact on management
A prepuncture diagnosis, available in 193 of 213 (91%) procedures, was confirmed by cytology in 119 (62%) procedures and adjusted in 74 (38%) procedures (Table 2). EUS-FNA had a positive impact on clinical management in 178 of 213 (84%) procedures (Table 5), particularly by excluding malignant lymphadenopathy (35%) or influencing the decision to perform surgery (49%). Only a minority of patients underwent surgery as a consequence of the cytological result (n = 19), whereas in most cases surgery was avoided (n = 85).
Table 5.
Impact on clinical management of EUS-FNA for mediastinal lymphadenopathy
| Overall (n = 213) | Diagnostic EUS-FNA (n = 133) | Staging EUS-FNA (n = 80) | |
|---|---|---|---|
| Positive impact (%) by providing tissue diagnosis | 178 (84) | 113 (85) | 65 (81) |
| and influencing decision on surgery | 104 (49) | 42 (32) | 62 (77) |
| and diagnosing an inflammatory disorder or reactive node | 74 (35) | 71 (53) | 3 (4) |
| Negative impact (%) by providing | 35 (16) | 20 (15) | 15 (19) |
| false-negative or inconclusive specimen | 16 (7) | 11 (8) | 5 (7) |
| no effect on management despite adequate cytology | 19 (9) | 9 (7) | 10 (12) |
In 16 of 213 (8%) procedures, clinical management was negatively affected by the cytological results by providing false-negative or inconclusive cytology. In 19 of 213 (9%) procedures, EUS-FNA did not affect patient management despite adequate cytological results. In four patients, the enlarged lymph node could not be detected by EUS and a nonsuspicious lymph node was punctured instead or there was confusion about which lymph node was punctured after the procedure. Four patients were referred for staging but subsequently refused further treatment. Eleven patients underwent EUS-FNA although, in retrospect, all these patients were already inoperable at the time of EUS-FNA due to invasive tumor growth (n = 4), distant metastases (n = 2), or known comorbidity (n = 5). Most inoperable patients had esophageal cancer (n = 8).
No significant difference with respect to the impact on management was found between diagnostic and staging EUS-FNA (85 and 81%, respectively; p = 0.14).
Complications
Two perforations in 213 (0.9%) procedures occurred due to a technical problem with the needle. One patient, known to have brain metastases, developed mediastinitis. He was treated with antibiotics and was discharged 25 days after the procedure. He died 6 days later due to progression of his malignancy. The second patient was treated with antibiotics and was discharged 6 days after EUS-FNA in good clinical condition.
Cost reduction analysis
Table 6 gives the total cost of the actual diagnostic workup versus the total cost of the alternative diagnostic workup, including the costs of all the components of the workups. In the alternative diagnostic workup, mediastinoscopy instead of EUS-FNA would have been performed in 143 patients. Since the complication rate of a cervical or parasternal mediastinoscopy is estimated to be 2%, two complications would have occurred in this cohort. The costs of the theoretical complications were assumed to be equal to the costs of the complications that occurred in the actual follow-up (€3,755; see Table 6). The total amount saved by performing 213 EUS-FNA procedures was €100,593, which results in average cost savings of €472 per patient.
Table 6.
Comparison of the cost of an alternative diagnostic workup (without EUS-FNA) and the actual diagnostic workup (by using EUS-FNA) for mediastinal lymphadenopathy
| Diagnostic tools | Costa (€) | Alternative workup | Actual workup | ||
|---|---|---|---|---|---|
| Number of investigations | Theoretical cost (€) | Number of investigations | Actual cost (€) | ||
| EUS-FNA | 175 | 0 | 0 | 213 | 37,275 |
| Mediastinoscopy/parasternal mediastinotomy | 961 | 143 | 137,423 | 15 | 14,415 |
| Video-assisted thoracoscopy (VATS) | 1,096 | 2 | 2192 | 4 | 4384 |
| PET scan | 998 | 4 | 3992 | 1 | 998 |
| Upper endoscopy | 102 | 4 | 408 | 5 | 510 |
| FNA/biopsy with other method | 76 | 14 | 1064 | 10 | 760 |
| Bronchoscopy | 294 | 29 | 8526 | 5 | 1470 |
| Expectative | 0 | 17 | 0 | 0 | 0 |
| Complications of investigation | 3755b | 2 | 7511 | 2 | 7511 |
| Death through investigation | 100,000b | 0 | 0 | 0 | 0 |
| Total cost of alternative workup (€) | 161,116 | Total cost of actual workup (€) | 60,523 | ||
aIf necessary, pathology processing and interpretation, anesthesia, and hospital stay are included in costs
bFixed costs for complications and death through complications were used for both theoretical and actual complications
Discussion
This study shows that the performance characteristics of EUS-FNA in patients with mediastinal lymphadenopathy performed in low-volume EUS centers are comparable to those in studies from high-volume EUS centers. Despite the relatively low case volume (213 cases in 6 years), our testing characteristics were the same as those obtained in a high-volume center by one very experienced endoscopist (Table 3) [27]. The diagnostic accuracy of EUS-FNA for mediastinal nodes in high-volume centers has been reported to be high (89–98%) [2–4, 8, 16, 27–29]. In most of those studies, EUS-FNA was performed by only one or two experienced endoscopists, which may well explain these excellent results [4, 8, 16, 27–29]. In our study, EUS-FNA procedures were performed by four endoscopists at two centers, each of whom had performed fewer than 25 EUS-FNA procedures before the start of the study. Since it has been suggested that the results of EUS-FNA are directly proportional to the experience of the endoscopist [30], our study may be a more realistic reflection of EUS-FNA results in normal daily clinical practice. A learning curve with regard to diagnostic accuracy [18, 19, 21] could not be detected in our study. This might be the result of the relatively low complexity of EUS-FNA of mediastinal lymphadenopathy.
Interestingly, in 10 of 16 patients (63%) with false-negative or nondiagnostic specimens, the final diagnosis turned out to be sarcoidosis (Table 4). This result raises the question of whether cytology is sufficient to diagnose sarcoidosis, which is traditionally diagnosed by histology. Although several studies have shown an accurate cytological diagnosis in 86–94% of sarcoidosis cases, these results were obtained mainly in small, selected patient populations with a high pretest probability for sarcoidosis [31–34]. Interestingly, a recent study comparing cytological and histological specimens showed significantly better results for histology [34/36 (94%) correct diagnoses] than for cytology [28/32 (82%) correct diagnoses, p = 0.04] [35].
In most patients in our study (84%), EUS-FNA had a positive impact on clinical management [26] since false-negative or inconclusive cytological results occurred infrequently (7%). However, the impact could have been even higher. This is explained by the observation that in another 9% of cases the impact was considered negative despite adequate cytological specimens. In retrospect, the indication for performing EUS-FNA in these patients was incorrect since the clinical management of these patients did not depend on this result. These were mostly patients with distal esophageal cancer in whom mediastinal FNA may be valuable for directing the appropriate surgical approach (transthoracic versus transhiatal esophageal resection) [17]. Nonetheless, EUS-FNA should be performed only in patients who are already considered to be clinically fit to undergo a resection.
Previous studies on EUS-FNA of mediastinal lymphadenopathy focused mainly on lung cancer staging. It has been shown that in 49–68% of patients with lung cancer, surgery or additional surgical staging was avoided as a consequence of the cytological result [8, 11]. A recent randomized trial showed that adding EUS-FNA to the conventional workup of lung cancer patients reduced the number of futile thoracotomies from 25% in the conventional workup to 9% in the workup with EUS-FNA [10]. Annema et al. [36] also reported that 16% of thoracotomies were avoided by using EUS-FNA in addition to mediastinoscopy in preoperative staging. A few studies have assessed the clinical impact of EUS-FNA in patients with mediastinal lymphadenopathy of unknown origin [5–7]. Catalano et al. [5] found that EUS-FNA affected clinical decision-making in 87% of cases in a series of 62 patients with mediastinal lymphadenopathy of unknown cause. These results are in line with our findings.
Until now, the evaluation of the cost-effectiveness of EUS-FNA compared to other lymph node sampling methods has been relatively uncommon [16, 23–25]. Eloubeidi et al. [16] showed an average cost reduction of $11,033 per patient by using EUS-FNA instead of mediastinoscopy as the primary staging tool in patients with NSCLC. In our heterogeneous patient population, various diagnostic tools such as mediastinoscopy or CT-guided FNA would have been performed if EUS-FNA had not been available. The use of EUS-FNA resulted in a cost reduction of €472 per patient by avoiding other more invasive and definitely more expensive procedures. The absolute cost reduction for our patient population was substantially less pronounced compared to other cost studies. This is probably because in our study the costs saved by abstaining from surgery were not included in the analysis. Furthermore, we did not include the costs of additional investigations in the alternative workup, which would probably have been performed in some patients because of an inadequate diagnostic result. This would have further increased the total cost difference between the two diagnostic strategies in favor of EUS-FNA.
Two esophageal perforations occurred in our study population, resulting in a complication rate of 0.9%. Other studies that prospectively assessed the safety of EUS-FNA in various organs have reported comparable complication rates (0.3–2.2%) [37–39], with esophageal perforation accounting for 50% of complications in one study [38]. Since our patients were not followed up after EUS-FNA according to a predefined protocol, our complication rate might be an underestimate. Furthermore, most studies on mediastinal EUS-FNA do not report any complications at all, which suggests that our complication rate may be higher than that of most high-volume centers. This may well reflect the fact that the endoscopists in our study were less experienced.
Our study has some limitations. The lack of histological confirmation in a large number of patients may reduce the validity of these results. Although most studies in this field use long-term follow-up as a reference standard for cytology results [2, 3, 27], this may give rise to a biased accuracy in favor of EUS-FNA. In addition, a retrospective judgment of the impact on clinical management may introduce classification bias. Therefore, classification was performed independently by a panel consisting of a researcher, a pulmonologist, and a gastroenterologist. To compensate for classification bias, special emphasis was placed on a critical judgment of the indication for EUS-FNA. Furthermore, the retrospective assessment of costs saved by performing EUS-FNA was cumbersome as well due to the heterogeneity of our population. Therefore, the alternative diagnostic or staging work-up was determined for each individual patient (taking into account indication for EUS-FNA, medical history, and lymph node station) and not for a diagnostic category.
Finally, in the theoretical workup we did not include the use of endobronchial ultrasound-guided fine-needle aspiration (EBUS-FNA) because it was not available in our institutions. Currently, EBUS-FNA might be a more elegant way to analyze mediastinal lymphadenopathy compared to mediastinoscopy [1, 40]. Performing EBUS-FNA instead of mediastinoscopy in the theoretical workup might change the outcome of the cost analysis by decreasing the number of mediastinoscopies performed. Furthermore, some high-volume EUS centers have shown that adding EBUS-FNA to EUS-FNA can improve the diagnostic yield since both methods are complementary, reaching different lymph node stations [41, 42]. However, just like mediastinoscopy, EBUS alone is hampered by the inability to sample the lymph nodes in the aorta pulmonary window [1, 41].
In conclusion, our study demonstrated that EUS-FNA is a valuable diagnostic tool to guide patient management and reduces the costs of clinical evaluation in a large heterogeneous group of patients being analyzed for mediastinal lymphadenopathy. Moreover, mediastinal EUS-FNA can be performed in low-volume EUS-FNA centers without compromising diagnostic accuracy.
Disclosures
Prof. Dr. P. D. Siersema receives research support from Cook Ireland Ltd., Ireland; Avantis Medical Systems, Inc., USA; Merit Medical, USA; Philips BV, Eindhoven, The Netherlands; and Boston Scientific Corp., USA; and has an advisory role at Niti Medical Technologies Ltd., Israel; Boston Scientific Corp., USA; and Cook Ireland Ltd., Ireland. Drs. M. M. C. Hirdes, M. P. Schwartz, K. M. Tytgat, N. Schlösser, D. M. Sie-Go, M. Brink, B. Oldenburg, and F. P Vleggaar have no conflicts of interest or financial ties to disclose.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Detterbeck FC, Jantz MA, Wallace M, Vansteenkiste J, Silvestri GA. Invasive mediastinal staging of lung cancer: ACCP evidence-based clinical practice guidelines (2nd edition) Chest. 2007;132:202S–220S. doi: 10.1378/chest.07-1362. [DOI] [PubMed] [Google Scholar]
- 2.Williams DB, Sahai AV, Aabakken L, Penman ID, van Velse VA, Webb J, Wilson M, Hoffman BJ, Hawes RH. Endoscopic ultrasound guided fine needle aspiration biopsy: a large single centre experience. Gut. 1999;44:720–726. doi: 10.1136/gut.44.5.720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wiersema MJ, Vilmann P, Giovannini M, Chang KJ, Wiersema LM. Endosonography-guided fine-needle aspiration biopsy: diagnostic accuracy and complication assessment. Gastroenterology. 1997;112:1087–1095. doi: 10.1016/S0016-5085(97)70164-1. [DOI] [PubMed] [Google Scholar]
- 4.Fritscher-Ravens A, Sriram PV, Bobrowski C, Pforte A, Topalidis T, Krause C, Jaeckle S, Thonke F, Soehendra N. Mediastinal lymphadenopathy in patients with or without previous malignancy: EUS-FNA-based differential cytodiagnosis in 153 patients. Am J Gastroenterol. 2000;95:2278–2284. doi: 10.1111/j.1572-0241.2000.02243.x. [DOI] [PubMed] [Google Scholar]
- 5.Catalano MF, Nayar R, Gress F, Scheiman J, Wassef W, Rosenblatt ML, Kochman M. EUS-guided fine needle aspiration in mediastinal lymphadenopathy of unknown etiology. Gastrointest Endosc. 2002;55:863–869. doi: 10.1067/mge.2002.124637. [DOI] [PubMed] [Google Scholar]
- 6.Devereaux BM, Leblanc JK, Yousif E, Kesler K, Brooks J, Mathur P, Sandler A, Chappo J, Lehman GA, Sherman S, Gress F, Ciaccia D. Clinical utility of EUS-guided fine-needle aspiration of mediastinal masses in the absence of known pulmonary malignancy. Gastrointest Endosc. 2002;56:397–401. doi: 10.1016/S0016-5107(02)70045-X. [DOI] [PubMed] [Google Scholar]
- 7.Yasuda I, Tsurumi H, Omar S, Iwashita T, Kojima Y, Yamada T, Sawada M, Takami T, Moriwaki H, Soehendra N. Endoscopic ultrasound-guided fine-needle aspiration biopsy for lymphadenopathy of unknown origin. Endoscopy. 2006;38:919–924. doi: 10.1055/s-2006-944665. [DOI] [PubMed] [Google Scholar]
- 8.Larsen SS, Krasnik M, Vilmann P, Jacobsen GK, Pedersen JH, Faurschou P, Folke K. Endoscopic ultrasound guided biopsy of mediastinal lesions has a major impact on patient management. Thorax. 2002;57:98–103. doi: 10.1136/thorax.57.2.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Savides TJ, Perricone A. Impact of EUS-guided FNA of enlarged mediastinal lymph nodes on subsequent thoracic surgery rates. Gastrointest Endosc. 2004;60:340–346. doi: 10.1016/S0016-5107(04)01709-2. [DOI] [PubMed] [Google Scholar]
- 10.Larsen SS, Vilmann P, Krasnik M, Dirksen A, Clementsen P, Maltbaek N, Lassen U, Skov BG, Jacobsen GK. Endoscopic ultrasound guided biopsy performed routinely in lung cancer staging spares futile thoracotomies: preliminary results from a randomised clinical trial. Lung Cancer. 2005;49:377–385. doi: 10.1016/j.lungcan.2005.04.005. [DOI] [PubMed] [Google Scholar]
- 11.Tournoy KG, De Ryck F, Vanwalleghem LR, Vermassen F, Praet M, Aerts JG, Van Maele G, van Meerbeeck JP. Endoscopic ultrasound reduces surgical mediastinal staging in lung cancer: a randomized trial. Am J Respir Crit Care Med. 2008;177:531–535. doi: 10.1164/rccm.200708-1241OC. [DOI] [PubMed] [Google Scholar]
- 12.Eloubeidi MA, Desmond R, Desai S, Mehra M, Bryant A, Cerfolio RJ. Impact of staging transesophageal EUS on treatment and survival in patients with non-small-cell lung cancer. Gastrointest Endosc. 2008;67:193–198. doi: 10.1016/j.gie.2007.06.052. [DOI] [PubMed] [Google Scholar]
- 13.Fritscher-Ravens A, Soehendra N, Schirrow L, Sriram PV, Meyer A, Hauber HP, Pforte A. Role of transesophageal endosonography-guided fine-needle aspiration in the diagnosis of lung cancer. Chest. 2000;117:339–345. doi: 10.1378/chest.117.2.339. [DOI] [PubMed] [Google Scholar]
- 14.Tournoy KG, De Ryck F, Vanwalleghem L, Praet M, Vermassen F, Van Maele G, van Meerbeeck JP. The yield of endoscopic ultrasound in lung cancer staging: does lymph node size matter? J Thorac Oncol. 2008;3:245–249. doi: 10.1097/JTO.0b013e3181653cbb. [DOI] [PubMed] [Google Scholar]
- 15.Annema JT, Versteegh MI, Veselic M, Voigt P, Rabe KF. Endoscopic ultrasound-guided fine-needle aspiration in the diagnosis and staging of lung cancer and its impact on surgical staging. J Clin Oncol. 2005;23:8357–8361. doi: 10.1200/JCO.2005.01.1965. [DOI] [PubMed] [Google Scholar]
- 16.Eloubeidi MA, Tamhane A, Chen VK, Cerfolio RJ. Endoscopic ultrasound-guided fine-needle aspiration in patients with non-small cell lung cancer and prior negative mediastinoscopy. Ann Thorac Surg. 2005;80:1231–1239. doi: 10.1016/j.athoracsur.2005.04.001. [DOI] [PubMed] [Google Scholar]
- 17.Marsman WA, Brink MA, Bergman JJ, Tytgat GN, ten Kate FJ, van Lanschot JJ, Fockens P. Potential impact of EUS-FNA staging of proximal lymph nodes in patients with distal esophageal carcinoma. Endoscopy. 2006;38:825–829. doi: 10.1055/s-2006-944611. [DOI] [PubMed] [Google Scholar]
- 18.Fockens P, Van den Brande JH, van Dullemen HM, van Lanschot JJ, Tytgat GN. Endosonographic T-staging of esophageal carcinoma: a learning curve. Gastrointest Endosc. 1996;44:58–62. doi: 10.1016/S0016-5107(96)70230-4. [DOI] [PubMed] [Google Scholar]
- 19.Eloubeidi MA, Tamhane A. EUS-guided FNA of solid pancreatic masses: a learning curve with 300 consecutive procedures. Gastrointest Endosc. 2005;61:700–708. doi: 10.1016/S0016-5107(05)00363-9. [DOI] [PubMed] [Google Scholar]
- 20.Mertz H, Gautam S. The learning curve for EUS-guided FNA of pancreatic cancer. Gastrointest Endosc. 2004;59:33–37. doi: 10.1016/S0016-5107(03)02028-5. [DOI] [PubMed] [Google Scholar]
- 21.Schlick T, Heintz A, Junginger T. The examiner’s learning effect and its influence on the quality of endoscopic ultrasonography in carcinoma of the esophagus and gastric cardia. Surg Endosc. 1999;13:894–898. doi: 10.1007/s004649901128. [DOI] [PubMed] [Google Scholar]
- 22.van Vliet EP, Eijkemans MJ, Poley JW, Steyerberg EW, Kuipers EJ, Siersema PD. Staging of esophageal carcinoma in a low-volume EUS center compared with reported results from high-volume centers. Gastrointest Endosc. 2006;63:938–947. doi: 10.1016/j.gie.2006.01.053. [DOI] [PubMed] [Google Scholar]
- 23.Harewood GC, Wiersema MJ, Edell ES, Liebow M. Cost-minimization analysis of alternative diagnostic approaches in a modeled patient with non-small cell lung cancer and subcarinal lymphadenopathy. Mayo Clin Proc. 2002;77:155–164. doi: 10.4065/77.2.155. [DOI] [PubMed] [Google Scholar]
- 24.Harewood GC, Wiersema MJ. A cost analysis of endoscopic ultrasound in the evaluation of esophageal cancer. Am J Gastroenterol. 2002;97:452–458. doi: 10.1111/j.1572-0241.2002.05499.x. [DOI] [PubMed] [Google Scholar]
- 25.Aabakken L, Silvestri GA, Hawes R, Reed CE, Marsi V, Hoffman B. Cost-efficacy of endoscopic ultrasonography with fine-needle aspiration vs. mediastinotomy in patients with lung cancer and suspected mediastinal adenopathy. Endoscopy. 1999;31:707–711. doi: 10.1055/s-1999-74. [DOI] [PubMed] [Google Scholar]
- 26.Chen VK, Eloubeidi MA. Endoscopic ultrasound-guided fine-needle aspiration of intramural and extraintestinal mass lesions: diagnostic accuracy, complication assessment, and impact on management. Endoscopy. 2005;37:984–989. doi: 10.1055/s-2005-870272. [DOI] [PubMed] [Google Scholar]
- 27.Eloubeidi MA, Tamhane A. Prospective assessment of diagnostic utility and complications of endoscopic ultrasound-guided fine needle aspiration. Results from a newly developed academic endoscopic ultrasound program. Dig Dis. 2008;26:356–363. doi: 10.1159/000177022. [DOI] [PubMed] [Google Scholar]
- 28.Eloubeidi MA, Cerfolio RJ, Chen VK, Desmond R, Syed S, Ojha B. Endoscopic ultrasound-guided fine needle aspiration of mediastinal lymph node in patients with suspected lung cancer after positron emission tomography and computed tomography scans. Ann Thorac Surg. 2005;79:263–268. doi: 10.1016/j.athoracsur.2004.06.089. [DOI] [PubMed] [Google Scholar]
- 29.Larsen SS, Vilmann P, Krasnik M, Dirksen A, Clementsen P, Skov BG, Jacobsen GK. Endoscopic ultrasound-guided biopsy versus mediastinoscopy for analysis of paratracheal and subcarinal lymph nodes in lung cancer staging. Lung Cancer. 2005;48:85–92. doi: 10.1016/j.lungcan.2004.10.002. [DOI] [PubMed] [Google Scholar]
- 30.Barthet M. Endoscopic ultrasound teaching and learning. Minerva Med. 2007;98:247–251. [PubMed] [Google Scholar]
- 31.Annema JT, Veselic M, Rabe KF. Endoscopic ultrasound-guided fine-needle aspiration for the diagnosis of sarcoidosis. Eur Respir J. 2005;25:405–409. doi: 10.1183/09031936.05.00098404. [DOI] [PubMed] [Google Scholar]
- 32.Fritscher-Ravens A, Sriram PV, Topalidis T, Hauber HP, Meyer A, Soehendra N, Pforte A. Diagnosing sarcoidosis using endosonography-guided fine-needle aspiration. Chest. 2000;118:928–935. doi: 10.1378/chest.118.4.928. [DOI] [PubMed] [Google Scholar]
- 33.Michael H, Ho S, Pollack B, Gupta M, Gress F. Diagnosis of intra-abdominal and mediastinal sarcoidosis with EUS-guided FNA. Gastrointest Endosc. 2008;67:28–34. doi: 10.1016/j.gie.2007.07.049. [DOI] [PubMed] [Google Scholar]
- 34.Mishra G, Sahai AV, Penman ID, Williams DB, Judson MA, Lewin DN, Hawes RH, Hoffman BJ. Endoscopic ultrasonography with fine-needle aspiration: an accurate and simple diagnostic modality for sarcoidosis. Endoscopy. 1999;31:377–382. doi: 10.1055/s-1999-32. [DOI] [PubMed] [Google Scholar]
- 35.Iwashita T, Yasuda I, Doi S, Kato T, Sano K, Yasuda S, Nakashima M, Hirose Y, Takaimi T, Moriwaki H. The yield of endoscopic ultrasound-guided fine needle aspiration for histological diagnosis in patients suspected of stage I sarcoidosis. Endoscopy. 2008;40:400–405. doi: 10.1055/s-2007-995593. [DOI] [PubMed] [Google Scholar]
- 36.Annema JT, Versteegh MI, Veselic M, Welker L, Mauad T, Sont JK, Willems LN, Rabe KF. Endoscopic ultrasound added to mediastinoscopy for preoperative staging of patients with lung cancer. JAMA. 2005;294:931–936. doi: 10.1001/jama.294.8.931. [DOI] [PubMed] [Google Scholar]
- 37.Al-Haddad M, Wallace MB, Woodward TA, Gross SA, Hodgens CM, Toton RD, Raimondo M. The safety of fine-needle aspiration guided by endoscopic ultrasound: a prospective study. Endoscopy. 2008;40:204–208. doi: 10.1055/s-2007-995336. [DOI] [PubMed] [Google Scholar]
- 38.Mortensen MB, Pless T, Durup J, Ainsworth AP, Plagborg GJ, Hovendal C. Clinical impact of endoscopic ultrasound-guided fine needle aspiration biopsy in patients with upper gastrointestinal tract malignancies. A prospective study. Endoscopy. 2001;33:478–483. doi: 10.1055/s-2001-14966. [DOI] [PubMed] [Google Scholar]
- 39.Bournet B, Migueres I, Delacroix M, Vigouroux D, Bornet JL, Escourrou J, Buscail L. Early morbidity of endoscopic ultrasound: 13 years’ experience at a referral center. Endoscopy. 2006;38:349–354. doi: 10.1055/s-2005-921173. [DOI] [PubMed] [Google Scholar]
- 40.Vilmann P, Puri R. The complete “medical” mediastinoscopy (EUS-FNA + EBUS-TBNA) Minerva Med. 2007;98:331–338. [PubMed] [Google Scholar]
- 41.Wallace MB, Pascual JM, Raimondo M, Woodward TA, McComb BL, Crook JE, Johnson MM, Al-Haddad MA, Gross SA, Pungpapong S, Hardee JN, Odell JA. Minimally invasive endoscopic staging of suspected lung cancer. JAMA. 2008;299:540–546. doi: 10.1001/jama.299.5.540. [DOI] [PubMed] [Google Scholar]
- 42.Vilmann P, Krasnik M, Larsen SS, Jacobsen GK, Clementsen P. Transesophageal endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) and endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) biopsy: a combined approach in the evaluation of mediastinal lesions. Endoscopy. 2005;37:833–839. doi: 10.1055/s-2005-870276. [DOI] [PubMed] [Google Scholar]


