INTRODUCTION
More than 2 million children live with HIV worldwide and more than 90% of them live in sub-Saharan Africa.[1] In the absence of antiretroviral therapy (ART), a third of children infected perinatally will not survive to their first birthday, and more than half will not survive their second birthday.[2] Despite recent international efforts to increase access to ART in low and middle income countries, the scaling up of ART in sub-Saharan Africa faces at least two major challenges.
First, in developed nations, the monitoring of ART consists of evaluating CD4 count and viral load every 3–4 months.[3] However, the measurement of viral load and CD4 cells count requires expensive and sophisticated technologies that cannot always be easily transferred or sustained in some of the poorest settings. In settings where viral load or CD4 cells count are not available, the World Health Organization (WHO) recommends that clinical parameters be used for monitoring ART, particularly the gains in weight or height in children.[4]
Second, in addition to the lack of the infrastructures, sub-Saharan Africa also faces an enormous shortage of trained health care personnel.[5] In the context of efforts to rapidly increase access to HIV services, the WHO recommends that specific tasks be moved, where appropriate, from highly qualified health workers to health workers with shorter training and fewer qualifications.[6] For this task shifting to be efficient, simplified protocols with clear decisional algorithms must be available. Contrary to viral load, which has a clear and simple target cut-point (below detection limit), cut-points for weight, height, and CD4 cell gain that correlate with subsequent treatment outcomes have not been clearly established.
In this study, we aimed to construct percentile curves for gains in weight, height, absolute CD4, and CD4% in the first 6 months of ART, and to test the value of the lower centile curves (3rd, 10th, 25th, 33rd, and 50th) as predictors of subsequent death, virological suppression, or treatment failure.
METHODS
During the first 4 years (April, 2004 to March, 2008) of government sponsored HIV treatment and care program for HIV-infected children at the Harriet Shezi Children’s Clinic, a pediatric outpatient clinic at Chris Hani Baragwanath Hospital, Soweto, Johannesburg, 2193 children 15 years or younger were initiated on ART. Of these, 1549 (70.6%) had at least 6 months of follow-up on ART and 1394 (90.0%) with baseline and 6 months follow up weight, height, absolute CD4, and CD4% data were included in this analysis. ART eligibility was in accordance with national guidelines [7]. The first line regimen included stavudine, lamivudine, and ritonavir-boosted lopinavir (LPV/r) for children 3 years or younger; or stavudine, lamivudine and efavirenz for those over 3 years and over 10kg of weight. Second line regimen included zidovudine, didanosine, and nevirapine for children 3 years or younger; zidovudine, didanosine, and LPV/r or efavirenz for children older than 3 years. Children who were started on ART were clinically reevaluated at 1 month, at 3 months, and every 3 months thereafter or at any other time if clinically needed.
At each scheduled visit, children were assessed clinically and weight and height (children 2 years or older) or length (children < 2years) were recorded. Laboratory investigations (hematology, chemistry, viral load, and CD4 cell count) were done at baseline and every 6 months unless otherwise indicated.
Statistical Analysis
Construction of percentile curves of the 6-month gains in weight, height, CD4, and CD4%
For each individual child, with n sequence data of weight, height or length, CD4 count, and CD4% measurements, the response curves were obtained by smoothing the n measurements over the chronological age at the time of the measurement using locally weighted quadratic regression. The 6-month estimates of weight, height, CD4, and CD4% gain were obtained by subtracting the response curve estimates at 6 months from the estimates at ART initiation. [8]
To obtain the reference percentile curves, we used methods similar to those used by the WHO to construct recent international growth curves. [9] The 6-month estimates of weight, height, CD4 count, and CD4% gain were regressed on chronological age using the generalized additive model for location, scale and shape, a method that requires a parametric distribution assumption for the response variable while allowing the modeling of the distribution parameter as nonparmetric (smooth) functions of the explanatory variables [10]. For the response variable, we assumed a Box-Cox power exponential distribution with four parameters relating to location (μ, median), scale (σ, coefficient of variation), skewness (υ, transformation for symmetry), and kurtosis (τ, power exponential parameter), respectively[11]. To specify the model, the user must choose the number of degrees of freedom (df) to be used for each parameter. Starting with the simplest model that includes age and the fitting of μ and σ curves while keeping the degree of freedom for υ and τ fixed at zero, we searched for df(μ) and then df(σ) that minimized the global deviance as indicated by the generalized Akaike Information Criterion (with penalty 3 for each degree of freedom used). In the next step, using the df(μ) and df(σ) selected in the previous, we sequentially searched for the df(υ) and df(τ) that minimized the global deviance. In the last step, Q statistic [12] and worm plots [13] were used to fine tune the selected df(μ), df(σ), df(υ), and df(τ) [11]. To facilitate the convergence of the models and obtain smoother curves [9, 10, 16], extreme values of six-month gains in weight, height, absolute CD4, and CD4%, i.e. values that looked far apart on visual inspection, were set to missing during the construction of the centiles curves: 22 (1.8%) observations for weight, 17 (1.4%) for height, 16 (1.5%) for CD4 count, and 6 (0.6%) for CD4%. In subsequent analyses, children with extreme values were classified as falling below the lowest or above the highest centiles.
The selected optimum set of degrees of freedom was used to fit a final model and the age-specific mean and standard deviation curves, μ(t) and σ(t), were estimated. Centile curves for selected centile τ in the interval [0, 1] were then constructed as Q̂(τ| t)=μ̂(t)+σ̂(t)Φ−1(t) [14]. Where Φ−1(t) denotes the inverse of the standard normal distribution function. These analyses were done using the gamlss package in R [15, 16]. Growth velocity is usually assessed in pediatric HIV using reference values from the Fels Institute cohort.[17–20] Estimates of the 3rd, 50th, and 90th percentile of the six-month post-ART gains in weight and height were compared to that of a cohort of 818 non-HIV infected and otherwise healthy white American children from the Fels Institute. [21, 22]
Association of lower percentiles with subsequent responses to ART
Three outcomes were considered: time to death (survival), time to first virological suppression and time to treatment failure. For two children whose exact date of death was missing, the last visit in the clinic was used as date of death. Virological suppression was defined as the first viral load measurement below 400 HIV RNA copies/ml. Treatment failure was defined as failure to achieve virological suppression after at least 1 year of ART, failure to achieve virological suppression prior to switch to second line regimen, or 2 viral load measurements above 1000 RNA copies/ml after initial viral suppression.
Kaplan Meier survival curves stratifying by the selected lower centiles (3rd, 10th, 25th, 33rd, 50th) of the 6 month weight, height, CD4, and CD4% gains and log-rank test were used to assess the association with each of the outcomes.[23] For each of the 3 outcomes, five Cox proportional hazard models were fitted with each of the 5 selected centiles as predictors. Baseline WHO clinical stage (stage I/II and III/IV); level of immunosuppression (mild or not significant, advanced, and severe according to age specific CD4% and/or CD4 count values);[4] viral load (≥ or <5 log copies); tuberculosis treatment at ART initiation, age at ART initiation (< 1.5 years, 1.5–3 years, 3–5 years, 5–8 years, and 8–15 years) and weight-for-age z score (WAZ) (≥ minus 2SD, −2 to – 3SD, and <−3SD) [24, 25] were included in the initial model. Using a stepwise backward selection procedure and Wald test, all covariates that did not contribute significantly to the fit of each model were dropped. The Hazard ratio and 95% confidence interval (CI) from each of the final models were reported. All variables included in the model met the proportional hazard assumption formally evaluated using the Kolmogorov-type supremum test [26]. Because attained weight- and height-for-age percentile curves are commonly available and familiar to health care workers (Road-to-Health), to evaluate whether attained growth can be used directly to monitor ART response, we also assessed the association between the lower percentile of the attained weight- and height-for-age at six months post-ART in our cohort and subsequent treatment outcomes. Analyses were done using SAS 9.1 (SAS Institute, Cary, NC). All tests were conducted using a 2-sided 0.05 significance level, without correction for multiple comparisons (or uncertainty due to model selection).
RESULTS
Description of the cohort
As of March 31, 2008, 54 (3.7%) of the 1394 children included in the analysis were loss to follow-up, 18 (1.3%) were dead, and 66 (4.7%) have been transferred out. Of the 1394 children, 699 (50.2%) were male, 872 (67.4%) in WHO clinical stage III&IV, 1120 (81.9%) were severely immuno-suppressed, 772 (58.3%) had viral load > 5 log RNA copies/ml, 378 (27.9%) had WAZ <−3SD, 512 (38.1%) had height-for-age Z score (HAZ) <−3SD, 353 (25.3%) were on TB treatment, and 262 (18.8%) were 17 months or younger at the time of ART initiation (table 1). Of the 1394 children, 1249 (89.6%) had baseline and 6 months data on weight, 1228 (88.1%) on height, 1071 (76.8%) on CD4 count, and 1066 (76.5%) on CD4%. The distribution of baseline characteristics did not vary by availability of follow–up data.
Table 1.
Characteristics at ART initiation of 1394 children included in the analysis of six-month weight, height, CD4 cell gain*
| Characteristic at ART initiation | Frequency** | Percentage |
|---|---|---|
| Sex | ||
| Male | 699 | 50.18 |
| Female | 694 | 49.82 |
| WHO Clinical stage | ||
| 1 or 2 | 422 | 32.61 |
| 3 or 4 | 872 | 67.39 |
| Severity of immunosuppression (WHO) | ||
| Mild or not significant | 158 | 11.55 |
| Advanced | 90 | 6.58 |
| Severe | 1120 | 81.87 |
| Viral load | ||
| >=100 000 copies/ml | 772 | 58.31 |
| <100000 copies/ml | 552 | 41.69 |
| Weight-for-age z score† | ||
| >=−2SD | 668 | 49.34 |
| −2 to −3SD | 308 | 22.75 |
| < −3SD | 378 | 27.92 |
| Height-for-age z score† | ||
| >=−2SD | 430 | 32.02 |
| −2 to −3SD | 401 | 29.86 |
| < −3SD | 512 | 38.12 |
| TB treatment at ART initiation | ||
| No | 1041 | 74.68 |
| Yes | 353 | 25.32 |
| Age at ART initiation | ||
| < 1.5 years | 262 | 18.79 |
| 1.5 – 2.9 years | 239 | 17.14 |
| 3.0 – 4.9 years | 254 | 18.22 |
| 5.0 – 7.9 years | 349 | 25.04 |
| 8.0 – 15.0 years | 290 | 20.8 |
| Time on ART in months median (IQR) | 25.2 | (17.9 to 33.5)‡ |
Children were included if they had at least 6 months of follow-up on ART and at least a pre-HAART and a 6-month measurement needed to estimate the six-month gain. Those children were part of a cohort of HIV-infected children who were initiated on ART at HSCC between April, 2004 and March, 2008. IQR = interquartile range.
Gender specific weight- and height- for-age z score were obtained by plotting the weight measurements at baseline against the World Health Organization (WHO) weight- and height-for-age charts for children <= 5years and against the Center for Disease and Control prevention (CDC) for children older than 5 years.
Minimum = 6 months, Maximum = 47 months.
Totals varies by baseline characteristics due to missing data
Six-month weight and height gain and their distribution compared to that of normal white American children of the Fels Institute cohort
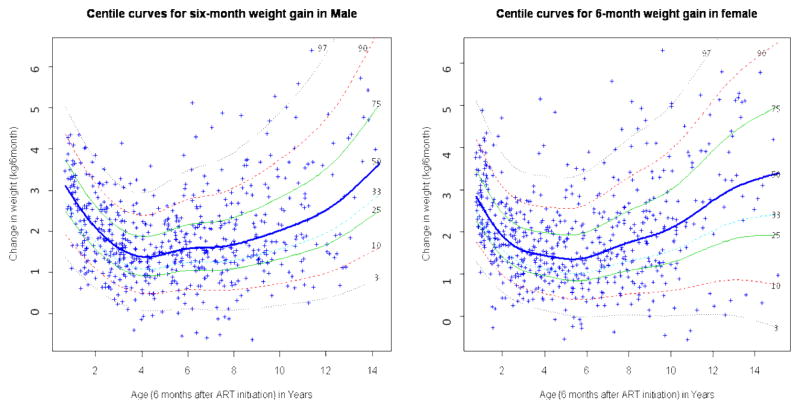
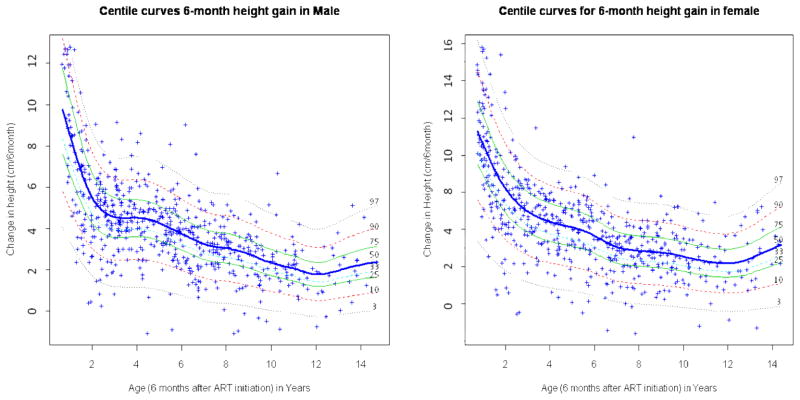
In the first 6 months post-ART, the median weight gain among boys was 2.73 kg among 1 year-olds, decreased to 1.41 kg among 4 year-olds, after which it increased to 3.05 kg among 14 year-olds (figure 1). Similarly, median weight gain in girls was 2.63 kg among 1 year-olds, decreased to 1.37 kg among 5 year-olds, and rose to 3.16 kg among 15 year-olds. The median height gain decreased from 8.54 cm among 1 year-old to 1.79 cm among 12 year-old males, and from 8.58 cm among 1 year-old to a 2.18 cm among 12 year-old females (figure 2).
Figure 1.
Six-month gender- and age-specific weight gain reference curves in children who were initiated on HAART at HSCC (Soweto) between April, 2004 and March, 2008
Curves were obtained using Box Cox power exponential distribution (BCPE) LMS and GAMLSS. HSCC: Harriet Shezi Children’s Clinic Model for male: BCPE (age, df(μ) = 4, df(σ) = 0.5, df(υ) =0, df(τ)=1). Model for female: BCPE (age, df(μ) = 5.3, df(σ) = 0, df(υ) =0, df(τ)=0)
Figure 2.
Six-month gender- and age-specific height gain reference curves among children who were initiated on ART at HSCC (Soweto) between April, 2004 and March, 2008
The curves were obtained using Box Cox power exponential distribution (BCPE) LMS and GAMLSS. HSCC: Harriet Shezi Children’s Clinic Model for male: BCPE (age, df(μ) =9.1, df(σ) =0, df(υ) =0, df(τ)=2). Model for female: BCPE (age, df(μ)=7, df(σ) =0, df(υ) =0, df(τ)=0).
Compared to the gain observed among healthy US children (Fels Institute Cohort), the six-month weight gain whether at the lower or upper tails of the distribution was consistently higher in our cohort of HIV infected South African children initiating ART, and the height gain particularly in the lower tails of the distribution was substantially and consistently lower, with differences amounting to up to 2 cm for the 3rd percentile.
Six-month CD4 count and CD4% gain
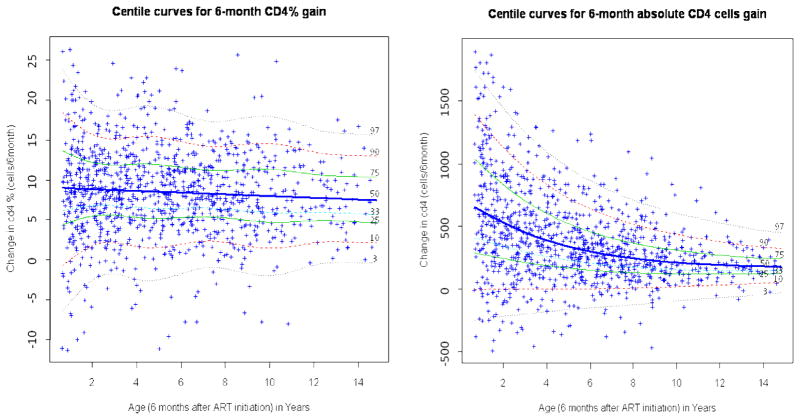
The CD4 count and CD4% improved substantially after ART initiation. While the gain in absolute CD4 count decreased with increasing age (from 672 cells among 1 year-olds to 310 cells among 6 year-olds and 154 cells among 15 year-olds), the gain in CD4% remained relatively stable across ages, ranging from 9.1% among 1 year-olds to 7.5% among 15 year-olds. (figure 3). Children below the 3rd percentile all had negative gains, and those below the 10th percentile barely maintained their baseline level of absolute CD4 or CD4% count (figure 3).
Figure 3.
Six-month age-specific CD4 count and CD4% gain reference curves among children who were initiated on HAART at HSCC (Soweto) between April, 2004 and March, 2008
The curves were obtained using Box Cox power exponential distribution (BCPE) LMS and GAMLSS. HSCC: Harriet Shezi Children’s Clinic Model for CD4 count: BCPE (age, df(μ) = 3, df(σ) = 0, df(υ) =0, df(τ)=1). Model for CD4%: BCPE (age, df(μ) = 0, df(σ) = 5, df(υ) =0, df(τ)=0).
Association between the 3rd, 10th, 25th, 33rd, and 50th percentile of six-month post-ART weight, height, and CD4 gains and survival
Of the 1394 children included in the analysis, 18 (1.3%) deaths occurred over the 2792.2 years of follow-up, corresponding to a mortality rate of 6.4 (95%CI: 4.1, 10.2) deaths per 1000 child-years between 6 months and 47 months. Children in the lower percentile of weight, CD4 count, and CD4% gain at 6 months of ART had statistically higher crude hazard of death than those with greater gains (table 2). After adjustment for WHO clinical stage, WAZ, and TB treatment at ART initiation, the hazard ratios comparing children below the 33rd percentile of weight, CD4, and CD4% gains to those above were 4.52 (95%CI: 1.47, 13.90) for weight, 3.03 (95%CI: 1.01, 9.11) for CD4 count, and 2.60 (95%CI: 0.87, 7.74) for CD4%. Hazard ratios increased with lower percentiles. There was no association between six-month height gain and survival.
Table 2.
Association between lower percentile of weight, height, absolute CD4 count, and CD4% gains at 6 months of ART and time to death, virological suppression, and treatment failure in a cohort of 1394 HIV-infected children from Soweto, South Africa
| Time to Death after 6 months | Time to Viral suppression** | Time to Treatment Failure£ | ||||
|---|---|---|---|---|---|---|
| HR (95%Confidence Interval) |
HR (95%Confidence Interval) |
HR (95%Confidence Interval) |
||||
| crude | Adjusted* | crude | adjusted† | crude | adjusted‡ | |
|
Weight | ||||||
| 3rd | 8.32 (2.32, 29.84) | 6.45 (1.69, 24.61) | 0.71 (0.52, 0.98) | 0.66 (0.48, 0.97) | 1.83 (0.75, 4.51) | 2.56 (1.04, 6.32) |
| 10th | 6.04 (2.02, 18.04) | 6.70 (2.15, 20.08) | 0.83 (0.68, 1.02) | 0.78 (0.63, 0.98) | 1.57 (0.86, 2.85) | 1.84 (1.00, 3.38) |
| 25th | 3.60 (1.26, 10.27) | 5.07 (1.68, 15.23) | 0.86 (0.75, 0.99) | 0.83 (0.71, 0.96) | 1.21 (0.80, 1.83) | 1.31 (0.86, 2.01) |
| 33rd | 2.85 (0.99, 8.21) | 4.09 (1.32, 12.74) | 0.83 (0.73, 0.94) | 0.80 (0.70, 0.91) | 1.44 (1.00, 2.07) | 1.56 (1.07, 2.28) |
| 50th | 2.38 (0.75, 7.59) | 3.07 (0.93, 10.12) | 0.87 (0.78, 0.98) | 0.84 (0.74, 0.95) | 1.30 (0.89, 1.84) | 1.47 (1.01, 2.16) |
|
Height | ||||||
| 3rd | 1.62 (0.21, 12.38) | 1.88 (0.24, 14.57) | 1.00 (0.77, 1.30) | 0.97 (0.74, 1.29) | 0.15 (0.02, 1.06) | 0.22 (0.03, 1.55) |
| 10th | 2.76 (0.87, 8.81) | 2.34 (0.72, 7.67) | 1.08 (0.91, 1.28) | 1.01 (0.84, 1.21) | 0.81 (0.49, 1.36) | 0.84 (0.49, 1.44) |
| 25th | 1.36 (0.46, 4.07) | 1.39 (0.45, 4.24) | 1.08 (0.95, 1.23) | 1.03 (0.89, 1.17) | 0.74 (0.49, 1.11) | 0.73 (0.48, 1.13) |
| 33rd | 1.25 (0.43, 3.61) | 1.30 (0.44, 3.88) | 1.08 (0.96, 1.22) | 1.03 (0.91, 1.17) | 0.77 (0.53, 1.12) | 0.76 (0.51, 1.13) |
| 50th | 1.72 (0.58, 5.12) | 2.22 (0.68, 7.26) | 1.05 (0.93, 1.18) | 0.99 (0.87, 1.12) | 0.75 (0.52, 1.07) | 0.75 (0.51, 1.10) |
|
CD4 count | ||||||
| 3rd | 0.61 (0.22, 1.68) | 0.42 (0.15, 1.20) | 0.84 (0.59, 1.20) | 0.86 (0.58, 1.28) | 0.48 (0.07, 3.44) | - |
| 10th | 3.74 (1.04, 13.42) | 5.40 (1.46, 20.00) | 0.57 (0.45, 0.72) | 0.54 (0.42, 0.69) | 3.37 (2.01, 5.64) | 3.42 (1.96, 5.99) |
| 25th | 3.57 (1.25, 10.19) | 3.89 (1.29, 11.71) | 0.75 (0.65, 0.87) | 0.75 (0.64, 0.88) | 1.57 (1.07, 2.31) | 1.78 (1.18, 2.69) |
| 33rd | 2.95 (1.02, 8.49) | 3.03 (1.01, 9.11) | 0.80 (0.70, 0.91) | 0.79 (0.69, 0.92) | 1.43 (0.99, 2.05) | 1.56 (1.06, 2.30) |
| 50th | 1.59 (0.53, 4.73) | 1.51 (0.49, 4.66) | 0.92 (0.81, 1.04) | 0.90 (0.79, 1.03) | 1.21 (0.84, 1.75) | 1.23 (0.82, 1.83) |
|
CD4 count Percentage | ||||||
| 3rd | 11.01 (3.07, 39.50) | 19.43 (4.88, 77.38) | 0.52 (0.35, 0.77) | 0.50 (0.32, 0.78) | 7.00 (3.51, 13.96) | 8.75 (3.93, 19.48) |
| 10th | 8.31 (2.88, 23.97) | 9.11 (3.03, 27.34) | 0.65 (0.52, 0.81) | 0.59 (0.47, 0.76) | 4.28 (2.70, 6.80) | 4.40 (2.68, 7.23) |
| 25th | 3.48 (1.22, 9.91) | 4.31 (1.44, 12.88) | 0.75 (0.65, 0.87) | 0.73 (0.62, 0.85) | 2.75 (1.92, 3.95) | 2.73 (1.85, 4.04) |
| 33rd | 2.28 (0.80, 6.50) | 2.60 (0.87, 7.74) | 0.75 (0.66, 0.86) | 0.73 (0.63, 0.84) | 2.39 (1.67, 3.41) | 2.43 (1.66, 3.57) |
| 50th | 1.20 (0.42, 3.45) | 1.11 (0.37, 3.31) | 0.82 (0.73, 0.93) | 0.81 (0.71, 0.92) | 1.57 (1.07, 2.28) | 1.57 (1.05, 2.34) |
Adjusted for Baseline WHO clinical stage, Baseline weight-for-age z score (WAZ), and TB treatment at ART initiation.
Adjusted for age for weight and height, baseline WHO clinical stage and age for CD4 count and CD4 percentage.
Adjusted for baseline WHO clinical stage and severity of immunosuppression for weight and height, baseline WHO clinical stage and WAZ for CD4 cell count and CD4%.
Viral load < 400 HIV RNA copies/ml,
2 viral load > 1000 after initial suppression or never suppress or > 1 year follow without viral suppression and regimen change
Association between the 3rd, 10th, 25th, 33rd, and 50th percentile of six-month post-ART weight, height, and CD4 gain and subsequent viral suppression
Almost all children achieved virological suppression, with Kaplan Meier estimates of 84.4% by 12 months and 96.4% by 24 months. Children in the lower percentile of weight, CD4 count, and CD4% gain at 6 months of ART were at lower hazard of virological suppression than those at higher percentiles (table 2). After adjustment for age (weight), or age and baseline WHO clinical stage (CD4 count and CD4%), the hazard ratios for viral suppression comparing children below the 33rd percentile to those above for weight, CD4, and CD4% gain at 6 months of ART were 0.80 (95%CI: 0.70, 0.91), 0.79 (95%CI: 0.69, 0.92), and 0.73 (95%CI: 0.63, 0.84), respectively (table 2). The hazard ratios for viral suppression were smaller with lower percentiles. Height gain in the first 6 months of ART was not associated with virological suppression.
Association between the 3rd, 10th, 25th, 33rd, and 50th percentile of six-month post-ART weight, height, and CD4 gain and subsequent treatment failure
The Kaplan Meier estimates of the proportion of children who failed treatment rose from 0.1% by 12 months to 5.5% by 24 months and 20.1% at 36 months. Children in the lower percentiles of the distribution of weight, CD4 count, and CD4% gain at 6 months of ART were at increased hazard of treatment failure. After adjustment for WHO clinical stage and severity of immunosuppression at ART initiation, the hazard ratios for treatment failure comparing children with weight gain below the 33rd percentile to those with gain above was 1.56 (95%CI: 1.07, 2.28) (table 2). For children falling below the 33rd percentile of CD4 and CD4% gain, the hazard ratios were 1.46 (95%CI: 1.06, 2.30) and 2.43 (95%CI: 1.66, 3.57), respectively, after adjustment for WHO clinical stage and WAZ (table 2). As for survival, the strength of the association increased with lower percentiles.
Association between the 3rd, 10th, 25th, 33rd, and 50th, percentile of attained weight and height for age (Road-to-Health chart) at six months post-ART and subsequent treatment outcomes
Six-month after ART, the median weight-for-age and height-for-age attained in our cohort was below the 8th (WAZ = −1.41) percentile for weight and below the 1st (HAZ = −2.37) percentile for height on the Road-to-Health chart. Children below the third percentile for weight tended to be at higher risk for death (aHR 4.63, 95% CI, 0.78, 27.42), but there was no correlation between weight or height for age at 6 months of ART and virological suppression or treatment failure aHRs: 0.98 (95%CI, 0.86, 1.11) and 0.74 (0.50, 1.10), respectively.
DISCUSSION
It is well established that poor nutritional status and stunting at time of ART initiation are correlated with poor prognosis of HIV infection in children.[27, 28] Monitoring height and weight is routinely performed in the follow up of children, but there are no reference values for weight or height gain among children receiving ART.[4] We constructed reference curves for monitoring 6 month gains in weight, height, CD4 count, and CD4% in children initiating ART, and demonstrated that, among children who survived the first six months of ART, lower percentiles of CD4% gains, weight and CD4 count, but not height, were associated with treatment outcomes, with increasing strength of association with decreasing percentiles.
In our cohort of South African children, six-month weight gain was consistently and substantially higher than that observed in the Fels cohort of white American children enrolled prenatally and followed-up between 1929 to 1978, while six-month height gain was consistently lower across all ages and gender.[21, 22] Similar differences have been observed with South African children born between April and June, 1990, (when the HIV prevalence among pregnant women was 0.7%),[29] suggesting that the differences between our cohort of HIV infected children and healthy US children may only in part be due to the presence of HIV infection.
The Fels reference values have been used in several pediatric antiretroviral drug trials in the US. [17–20] Our results demonstrate that using the Fels cohort as a reference for HIV infected children in South Africa, may result in an important underestimation of the number of children at risk of poor ART outcomes. For example, a boy who initiates ART at age 18 months and gains 1.5 kg in the first 6 months has a weight gain of the 90th percentile (+1SD) on the Fels cohort growth velocity curves, but scores below the 10th percentile (−1SD) on our newly developed reference curves of weight gain for HIV infected children on ART. A close follow up of this child would be warranted as his weight gain predicts a 6.7-fold hazard of death, 1.8 fold hazard of virologic failure and reduced likelihood (adjusted HR 0.8) of virological suppression, compared to children with gender- and age-specific weight gain above the 10th percentile.
In contrast to six month weight gain plotted on our newly developed reference charts for HIV-infected children, weight at six months of ART plotted on standard weight-for-age charts, did not appear appropriate for the purpose of monitoring ART response in our cohort. In our cohort, though as expected children with weight-for-age or height-for-age below the 3rd percentile were at increased hazard for subsequent mortality, there was no correlation with subsequent virological suppression or treatment failure.
CD4% gained in the first 6 months of ART showed the strongest association with poor ART outcomes. Monitoring gains in CD4% are particularly attractive as the CD4% gain was relatively stable across ages. In our cohort, children whose CD4% gain was less than 6% after 6 months of ART fell below the 33rd percentile of CD4% gain and had a 2.6-folds increased hazard of death, 2.4-fold greater hazard of treatment failure and were less likely (adjusted HR 0.7) to achieve viral suppression compared to children who gained 6% or more in CD4%. However, as with viral load, access to CD4% measurement is not widespread in resource poor settings, limiting its usefulness in monitoring ART response.
Our analysis has many strengths including a large sample size and a long follow-up (median follow-up time on ART 25 months), and the use of three different outcomes limiting the impact of potential errors that might result from measurement of each. Our analysis also has a number of limitations. First, we only focus on one interval: the first 6-month of ART, chosen to match the recommended time for viral load or CD4 measurement [7] and in accordance with previous studies [27, 28]. Second, most children were referred after hospitalization, with few children being referred from primary health care clinics, thus selecting for children that may have been sicker at ART initiation and have greater gains in weight, height, and CD4 cell count if they survive the early weeks of treatment.[30] Although children in our cohort do not appear to differ substantially at ART initiation from children in other cohorts in the region [31, 32], reference curves constructed from a more representative population are needed. Third, despite ART treatment some children had negative weight, height, CD4, and CD4% gains at 6 months. Although negative gains in CD4 cell count and weight can occur, negative height or length gains are likely due to measurement error occurring under routine conditions in a busy clinic. It has been shown that because of measurement errors, a single height velocity measurement even at 12 months lacks the precision to provide a reliable index of current growth, particularly in short children.[33] Fourth, some important confounders like adherence were not measured and could not be adjusted for.
Conclusion
We demonstrated that though weight gain is an excellent tool for monitoring the early response to ART, growth references from non HIV-infected children do not properly discriminate HIV-infected children with inappropriate growth response to ART. Our results also suggest that even with the reference distributions we constructed, the usual -2SD (3rd percentile) cut-off for growth failure definition misses a lot of children that are at high risk of failing ART and that substantially higher cut-offs should be considered. Gain in CD4% in the first six month of ART was the best predictor of poor subsequent ART outcomes. But in areas with limited access to viral load or CD4 measurement, weight gain post-ART using our newly developed reference distributions for HIV-infected children on ART may be a good alternative. Developing an international collaboration aimed at gathering data from ART pediatric programs in other resource-constrained settings would be of crucial interest such that international reference curves can be constructed.
Acknowledgments
Marcel Yotebieng was an NIH Fogarty Fellow sponsored by grant No. DHHS/NIH/FIC 5 D43 TW01039-08 AIDS International Training and Research Program at the University of North Carolina at Chapel Hill. Wits Echo and Harriet Shezi children’s clinic in addition to the South African government funding received support from USAID/PEPFAR. We are extremely grateful to the dedicated staff of Harriet Shezi Children’s clinic for their amazing work. We thank Drs Frieda Behets, Stephen Cole, and Adaora Adimora for their helpful comments.
References
- 1.UNAIDS. 2007 AIDS epidemic update. Geneva, Switzerland: 2007. [Google Scholar]
- 2.Newell ML, Coovadia H, Cortina-Borja M, Rollins N, Gaillard P, Dabis F. Mortality of infected and uninfected infants born to HIV-infected mothers in Africa: a pooled analysis. Lancet. 2004;364(9441):1236–43. doi: 10.1016/S0140-6736(04)17140-7. [DOI] [PubMed] [Google Scholar]
- 3.Guidelines for the Use of Antiretroviral Agents in Pediatric HIV Infection. Feb 28, 2008. [cited 2008 March 21] [Google Scholar]
- 4.World Health Organization; W.H. Organization. Antiretroviral Therapy of HIV Infection in Infants and Children in Resource-Limited Settings: Towards Universal Access. World Health Organization; Geneva, Switzerland: 2006. [Google Scholar]
- 5.Kober K, Van Damme W. Scaling up access to antiretroviral treatment in southern Africa: who will do the job? Lancet. 2004;364(9428):103–7. doi: 10.1016/S0140-6736(04)16597-5. [DOI] [PubMed] [Google Scholar]
- 6.World Health Organization . Task shifting: rational redistribution of tasks among health workforce teams, global recommendations and guidelines. 2008. [cited 2009 January 4] [Google Scholar]
- 7.Meyers T, Eley B, Leoning W. Guidelines for the management of HIV-infected children -2005, Health. Jacana Media; 2005. [Google Scholar]
- 8.Carey VJ, Yong FH, Frenkel LM, McKinney RM. Growth velocity assessment in paediatric AIDS: smoothing, penalized quantile regression and the definition of growth failure. Stat Med. 2004;23(3):509–26. doi: 10.1002/sim.1578. [DOI] [PubMed] [Google Scholar]
- 9.WHO Multicentre Growth Reference Study Group. WHO Child Growth Standards: Growth velocity based on weight, length and head circumference: Methods and development. World Health Organization; Geneva: 2009. p. 242. [Google Scholar]
- 10.Rigby RA, Stasinopoulos DM. Generalized additive models for location, scale and shape. Journal of the Royal Statistical Society: Series C (Applied Statistics) 2005;54(3):507–554. [Google Scholar]
- 11.Rigby RA, Stasinopoulos DM. Smooth centile curves for skew and kurtotic data modelled using the Box-Cox power exponential distribution. Stat Med. 2004;23(19):3053–76. doi: 10.1002/sim.1861. [DOI] [PubMed] [Google Scholar]
- 12.Royston P, Wright EM. Goodness-of-fit statistics for age-specific reference intervals. Stat Med. 2000;19(21):2943–62. doi: 10.1002/1097-0258(20001115)19:21<2943::aid-sim559>3.0.co;2-5. [DOI] [PubMed] [Google Scholar]
- 13.van Buuren S, Fredriks M. Worm plot: a simple diagnostic device for modelling growth reference curves. Stat Med. 2001;20(8):1259–77. doi: 10.1002/sim.746. [DOI] [PubMed] [Google Scholar]
- 14.Wei Y, Pere A, Koenker R, He X. Quantile regression methods for reference growth charts. Stat Med. 2006;25(8):1369–82. doi: 10.1002/sim.2271. [DOI] [PubMed] [Google Scholar]
- 15.Stasinopoulos D, Rigby R. Generalized additive models for location scale and shape (GAMLSS) in R. Journal of Statistical Software. 2007;23(7) [Google Scholar]
- 16.Stasinopoulos D, Rigby R, Akantziliotou C. Instructions on how to use the gamlss package in R. 2. 2008. [Google Scholar]
- 17.Bakshi SS, Britto P, Capparelli E, Mofenson L, Fowler MG, Rasheedetal S. Evaluation of pharmacokinetics, safety, tolerance, and activity of combination of zalcitabine and zidovudine in stable, zidovudine-treated pediatric patients with human immunodeficiency virus infection. AIDS Clinical Trials Group Protocol 190 Team. J Infect Dis. 1997;175(5):1039–50. doi: 10.1086/520351. [DOI] [PubMed] [Google Scholar]
- 18.Englund JA, Baker CJ, Raskino C, McKinney RE, Petrie B, Fowler MG, et al. Zidovudine, didanosine, or both as the initial treatment for symptomatic HIV-infected children. AIDS Clinical Trials Group (ACTG) Study 152 Team. N Engl J Med. 1997;336(24):1704–12. doi: 10.1056/NEJM199706123362403. [DOI] [PubMed] [Google Scholar]
- 19.Kline MW, Van Dyke RB, Lindsey JC, Gwynne M, Culnane M, McKinney RE, Jr, et al. A randomized comparative trial of stavudine (d4T) versus zidovudine (ZDV, AZT) in children with human immunodeficiency virus infection. AIDS Clinical Trials Group 240 Team. Pediatrics. 1998;101(2):214–20. doi: 10.1542/peds.101.2.214. [DOI] [PubMed] [Google Scholar]
- 20.McKinney RE, Jr, Johnson GM, Stanley K, Yong FH, Keller A, O’Donnell KJ, et al. A randomized study of combined zidovudine-lamivudine versus didanosine monotherapy in children with symptomatic therapy-naive HIV-1 infection. The Pediatric AIDS Clinical Trials Group Protocol 300 Study Team. J Pediatr. 1998;133(4):500–8. doi: 10.1016/s0022-3476(98)70057-5. [DOI] [PubMed] [Google Scholar]
- 21.Baumgartner RN, Roche AF, Himes JH. Incremental growth tables: supplementary to previously published charts. Am J Clin Nutr. 1986;43(5):711–22. doi: 10.1093/ajcn/43.5.711. [DOI] [PubMed] [Google Scholar]
- 22.Roche AF, Himes JH. Incremental growth charts. Am J Clin Nutr. 1980;33(9):2041–52. doi: 10.1093/ajcn/33.9.2041. [DOI] [PubMed] [Google Scholar]
- 23.Collett D. Modelling Survival data in Medical Research. 2. Florida: Chapman&Hall/CDC; 2003. [Google Scholar]
- 24.CDC. A SAS program for CDC growth charts 2000. 2007 May 22; [cited 2008 December 31]; Available from: http://www.cdc.gov/NCCDPHP/dnpa/growthcharts/resources/sas.htm.
- 25.WHO Multicentre Growth Reference Study Group ; W. Press. WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height, and Body Mass Index-for-Age: Methods and Development. WHO; Geneva, Switzerland: 2006. [Google Scholar]
- 26.Lin DY, Wei LJ, Ying Z. Checking the Cox Model with Cumulative Sums of Martingale-Based Residuals. Biometrika. 1993;80(3):557–572. [Google Scholar]
- 27.Benjamin DK, Jr, Miller WC, Ryder RW, Weber DJ, Walter E, McKinney RE., Jr Growth patterns reflect response to antiretroviral therapy in HIV-positive infants: potential utility in resource-poor settings. AIDS Patient Care STDS. 2004;18(1):35–43. doi: 10.1089/108729104322740901. [DOI] [PubMed] [Google Scholar]
- 28.Carey VJ, Yong FH, Frenkel LM, McKinney RE., Jr Pediatric AIDS prognosis using somatic growth velocity. Aids. 1998;12(11):1361–9. doi: 10.1097/00002030-199811000-00019. [DOI] [PubMed] [Google Scholar]
- 29.Cameron N, De Wet T, Ellison G, Bogin B. Growth in height and weight of South African urban infants from birth to five years: The Birth to Ten Study. Am J Hum Biol. 1998;10(4):495–504. doi: 10.1002/(SICI)1520-6300(1998)10:4<495::AID-AJHB9>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- 30.Moultrie H, Yotebieng M, Kuhn L, Meyers T. Mortality and Virological Outcomes of 2105 HIV-infected Children Receiving ART in Soweto, South Africa. 16th Conference on Retroviruses and Oportunistic Infections; Montreal, Canada. 2009. [Google Scholar]
- 31.Bolton-Moore C, Mubiana-Mbewe M, Cantrell RA, Chintu N, Stringer EM, Chi BH, et al. Clinical outcomes and CD4 cell response in children receiving antiretroviral therapy at primary health care facilities in Zambia. Jama. 2007;298(16):1888–99. doi: 10.1001/jama.298.16.1888. [DOI] [PubMed] [Google Scholar]
- 32.Sutcliffe CG, van Dijk JH, Bolton C, Persaud D, Moss WJ. Effectiveness of antiretroviral therapy among HIV-infected children in sub-Saharan Africa. Lancet Infect Dis. 2008;8(8):477–89. doi: 10.1016/S1473-3099(08)70180-4. [DOI] [PubMed] [Google Scholar]
- 33.Voss LD, Wilkin TJ, Bailey BJ, Betts PR. The reliability of height and height velocity in the assessment of growth (the Wessex Growth Study) Arch Dis Child. 1991;66(7):833–7. doi: 10.1136/adc.66.7.833. [DOI] [PMC free article] [PubMed] [Google Scholar]